Who Bites Me? A Tentative Discriminative Key to Diagnose Hematophagous Ectoparasites Biting Using Clinical Manifestations
Abstract
1. Introduction
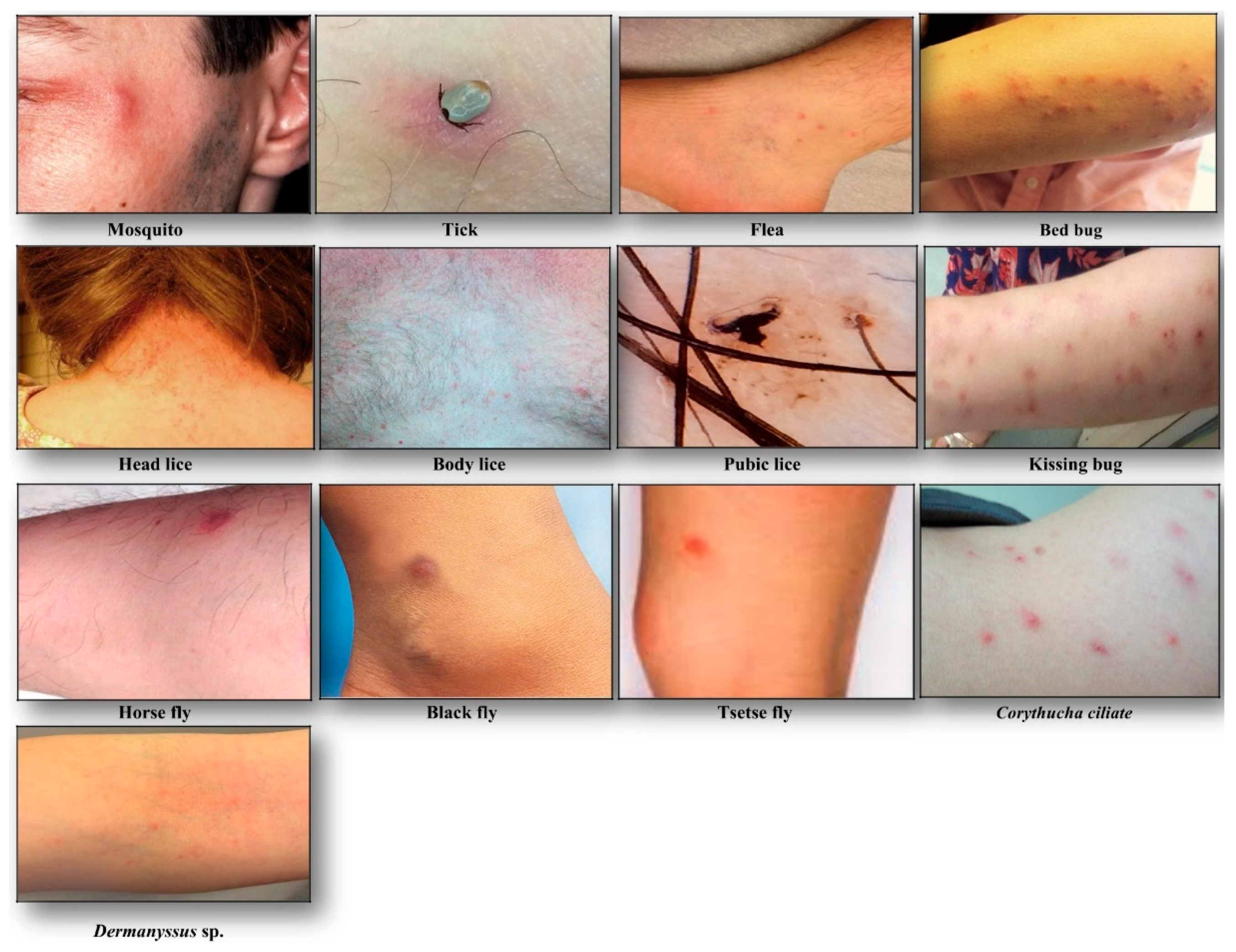
2. Clinical Manifestations of Arthropod Bites
2.1. Culicidae (Mosquitoes)
2.2. Ceratopogonidae (Biting Midges) and Phlebotominae (Sandflies)
2.3. Pediculidae, Pthiridae (Lice)
2.4. Ixodidae, Argasidae (Ticks)
2.5. Glossinidae (Tsetse Flies)
2.6. Pulicidae (Fleas)
2.7. Cimicidae (Bed Bugs)
2.8. Reduviidae (Kissing Bugs)
2.9. Simuliidae (Blackflies)
2.10. Tabanidae (Horse Flies)
3. Other Non-Common Hematophagous Arthropods
4. Treatment
5. Discussion and Conclusions
Funding
Conflicts of Interest
References
- Jensen, S. The Proterozoic and Earliest Cambrian Trace Fossil Record; Patterns, Problems and Perspectives. Integr. Comp. Biol. 2003, 43, 219–228. [Google Scholar] [CrossRef]
- Ribeiro, J.M.; Francischetti, I.M. Role of arthropod saliva in blood feeding: Sialome and post-sialome perspectives. Annu. Rev. Entomol. 2003, 48, 73–88. [Google Scholar] [CrossRef] [PubMed]
- Choe, J.C. Encyclopedia of Animal Behavior; Elsevier: San Diego, CA, USA, 2019; Volume 2. [Google Scholar]
- Bouchet, F.; Lavaud, F. Solenophagy and telmophagy: Biting mechanisms among various hematophagous insects. Allerg. Immunol. 1999, 31, 346–350. [Google Scholar]
- Andrade, B.B.; Teixeira, C.R.; Barra, A.; Barral-Netto, M. Haematophagous arthropod saliva and host defense system: A tale of tear and blood. An. Acad. Bras. Ciênc. 2005, 77, 15. [Google Scholar] [CrossRef]
- Rizzo, M.C.; Arruda, L.K.; Chapman, M.D.; Fernandez-Caldas, E.; Baggio, D.; Platts-Mills, T.A.; Naspitz, C.K. IgG and IgE antibody responses to dust mite allergens among children with asthma in Brazil. Ann. Allergy 1993, 71, 152–158. [Google Scholar] [PubMed]
- Harbach, R.E. Mosquito Taxonomic Inventory. Available online: http://mosquito-taxonomic-inventory.info (accessed on 6 December 2017).
- Rozendaal, J.A. Chapter 1-Mosquitos and other biting Diptera. In Vector Control-Methods for Use by Individuals and Communities; World Health Organization: Geneva, Switzerland, 1997. [Google Scholar]
- Engler, R.J. Mosquito bite pathogenesis in necrotic skin reactors. Curr. Opin. Allergy. Clin. Immunol. 2001, 1, 349–354. [Google Scholar] [CrossRef]
- Chiu, T.M.; Lin, Y.M.; Wang, S.C.; Tsai, Y.G. Hypersensitivity to mosquito bites as the primary clinical manifestation of an Epstein-Barr virus infection. J. Microbiol. Immunol. Infect. 2016, 49, 613–616. [Google Scholar] [CrossRef]
- Manuyakorn, W.; Itsaradisaikul, S.; Benjaponpitak, S.; Kamchaisatian, W.; Sasisakulporn, C.; Jotikasthira, W.; Matangkasombut, P. Mosquito allergy in children: Clinical features and limitation of commercially-available diagnostic tests. Asian Pac. J. Allergy Immunol. 2017, 35, 186–190. [Google Scholar]
- Boorman, J. Biting midges (Ceratopogonidae). In Medical Insects and Arachnids; Lane, R.P., Crosskey, R.W., Eds.; Springer: Dordrecht, The Netherlands, 1993; Volume 1, pp. 288–309. [Google Scholar]
- Linley, J.R. Biting Midges (Diptera: Ceratopogonidae) as Vectors of Nonviral Animal Pathogens. J. Med. Entomol. 1985, 22, 589–599. [Google Scholar] [CrossRef]
- Akhoundi, M.; Kuhls, K.; Cannet, A.; Votýpka, J.; Marty, P.; Delaunay, P.; Sereno, D. A Historical Overview of the Classification, Evolution, and Dispersion of Leishmania Parasites and Sandflies. PLoS Negl. Trop. Dis. 2016, 10, e0004349. [Google Scholar] [CrossRef]
- Pali-Schöll, I.; Blank, S.; Verhoeckx, K.; Mueller, R.; Janda, J.; Marti, E.; Seida, A.A.; Rhyner, C.; DeBoer, D.J.; Jensen-Jarolim, E. EAACI position paper: Comparing insect hypersensitivity induced by bite, sting, inhalation or ingestion in human beings and animals. Allergy 2019, 74, 874–887. [Google Scholar] [CrossRef] [PubMed]
- Borkent, A. The biting midges, the Ceratopogonidae (Diptera). In Biology of Disease Vectors, 2nd ed.; Marquardt, W.C., Ed.; Elsevier: Burlington, MA, USA, 2004; Volume 2, pp. 113–126. [Google Scholar]
- Linley, J.R.; Hoch, A.L.; Pinheiro, F.P. Biting midges (Diptera: Ceratopogonidae) and human health. J. Med. Entomol. 1983, 20, 347–364. [Google Scholar] [CrossRef] [PubMed]
- Price, R.D.; Hellenthal, R.A.; Palma, R.L.; Johnson, K.P.; Clayton, D.H. The Chewing Lice: World Checklist and Biological Overview; Illinois Natural History Survey: Urbana, IL, USA, 2003; Volume 24, 501p. [Google Scholar]
- Durden, L.A.; Musser, G.G. The sucking lice (Insecta: Anoplura) of the world: A taxonomic checklist with records of mammalian hosts and geographical distributions. Bull. Am. Mus. Nat. Hist. 1994, 218, 1–90. [Google Scholar]
- Mazurek, C.M.; Lee, C.P. How to manage head lice. West J. Med. 2000, 172, 342–345. [Google Scholar] [CrossRef]
- Burkhart, C.N.; Burkhart, C.G. Head lice: Scientific assessment of the nit sheath with clinical ramifications and therapeutic options. J. Am. Acad. Dermatol. 2005, 53, 129–133. [Google Scholar] [CrossRef]
- Badiaga, S.; Brouqui, P. Human louse-transmitted infectious diseases. Clin. Microbiol. Infect. 2012, 18, 332–337. [Google Scholar] [CrossRef]
- Bonilla, D.L.; Durden, L.A.; Eremeeva, M.E.; Dasch, G.A. The Biology and Taxonomy of Head and Body Lice—Implications for Louse-Borne Disease Prevention. PLoS Pathog. 2013, 9, e1003724. [Google Scholar] [CrossRef]
- Raoult, D.; Roux, V. The body louse as a vector of reemerging human diseases. Clin. Infect. Dis. 1999, 29, 888–911. [Google Scholar] [CrossRef]
- Brouqui, P. Arthropod-Borne Diseases Associated with Political and Social Disorder. Annu. Rev. Entomol. 2011, 56, 357–374. [Google Scholar] [CrossRef]
- Anderson, A.L.; Chaney, E. Pubic Lice (Pthirus pubis): History, Biology and Treatment vs. Knowledge and Beliefs of US College Students. Int. J. Environ. Res. Public Health 2009, 6, 592–600. [Google Scholar] [CrossRef]
- Akhoundi, M.; Cannet, A.; Arab, M.K.; Marty, P.; Delaunay, P. An old lady with Pediculosis pubis on the head hair. J. Eur. Acad. Dermatol. Venereol. 2016, 30, 885–887. [Google Scholar] [CrossRef] [PubMed]
- Veraldi, S.; Schianchi, R.; Ramoni, S.; Nazzaro, G. Pubic hair removal and Phthirus pubis infestation. Int. J. STD AIDS 2018, 29, 103–104. [Google Scholar] [CrossRef] [PubMed]
- Furman, D.P.; Loomis, E.C. The ticks of California (Acari: Ixodida). Bull. California Insect Survey 1984, 25, 1–239. [Google Scholar]
- Guglielmone, A.A.; Robbing, R.G.; Dmitry, A.; Trevor, N.P.; Estrada-Peña, A.; Horak, I.G.; Shao, R.; Barker, S.C. The Argasidae, Ixodidae and Nuttalliellidae (Acari: Ixodida) of the world: A list of valid species names. Zootaxa 2010, 2528, 1–28. [Google Scholar] [CrossRef]
- Brites-Neto, J.; Roncato Duarte, K.M.; Fernandes Martins, T. Tick-borne infections in human and animal population worldwide. Vet. World 2015, 8, 301–315. [Google Scholar] [CrossRef]
- Parola, P.; Paddock, C.D.; Raoult, D. Tick-Borne Rickettsioses around the World: Emerging Diseases Challenging Old Concepts. Clin. Microbiol. Rev. 2005, 18, 719–756. [Google Scholar] [CrossRef]
- Van Wye, J.E.; Hsu, Y.P.; Terr, A.I.; Moss, R.B.; Lane, R.S. Anaphylaxis from a tickbite. NEJM 1991, 324, 777–778. [Google Scholar]
- Rahlenbeck, S.; Fingerle, V.; Doggett, S. Prevention of tick-borne diseases: An overview. Br. J. Gen. Pract. 2016, 66, 492–494. [Google Scholar] [CrossRef]
- Haddad, V., Jr.; Raineri Haddad, M.; Santos, M.; Luiz Costa Cardoso, J. Skin manifestations of tick bites in humans. An. Bras. Dermatol. 2018, 93, 251–255. [Google Scholar] [CrossRef]
- Krafsur, E.S. Tsetse flies: Genetics, evolution, and role as vectors. Infect. Genet. Evol. 2009, 9, 124–141. [Google Scholar] [CrossRef]
- Gooding, R.H.; Krafsur, E.S. Tsetse Genetics: Contributions to Biology, Systematics, and Control of Tsetse Flies. Annu. Rev. Entomol. 2005, 50, 101–123. [Google Scholar] [CrossRef] [PubMed]
- Service, M.W. A Guide to Medical Entomology; Palgrave Macmillan: London, UK, 1980. [Google Scholar]
- Muhanguzi, D.; Mugenyi, A.; Bigirwa, G.; Kamusiime, M.; Kitibwa, A.; Gloria Akurut, G.; Ochwo, S.; Amanyire, W.; George Okech, S.; Hattendorf, J.; et al. African animal trypanosomiasis as a constraint to livestock health and production in Karamoja region: A detailed qualitative and quantitative assessment. BMC Vet. Res. 2017, 13, 355. [Google Scholar]
- Lewis, R.E. Resume of the Siphonaptera (Insecta) of the World. J. Med. Entomol. 1999, 35, 377–389. [Google Scholar]
- Lewis, R.E. Notes on the geographical distribution and host preferences in the order Siphonaptera. Part 8. New taxa described between 1984 and 1990, with a current classification of the order. J. Med. Entomol. 1993, 30, 239–256. [Google Scholar] [CrossRef]
- Dobler, G.; Pfeffer, M. Fleas as parasites of the family Canidae. Parasit. Vectors 2011, 4, 139. [Google Scholar] [CrossRef]
- Leulmi, H.; Socolovschi, C.; Laudisoit, A.; Houemenou, G.; Davoust, B.; Bitam, I.; Raoult, D.; Parola, P. Detection of Rickettsia felis, Rickettsia typhi, Bartonella species and Yersinia pestis in fleas (Siphonaptera) from Africa. PLoS Negl. Trop. Dis. 2014, 8, e3152. [Google Scholar] [CrossRef]
- Mathison, B.A.; Prittb, B.S. Laboratory Identification of Arthropod Ectoparasites. Clin. Microbiol. Rev. 2014, 27, 48–67. [Google Scholar] [CrossRef]
- Galy, A.; Loubet, P.; Peiffer-Smadja, N.; Yazdanpanah, Y. The plague: An overview and hot topics. Rev. Med. Interne. 2018, 39, 863–868. [Google Scholar] [CrossRef]
- Mullen, G.R.; Durden, L.A. Medical and Veterinary Entomology, 2nd ed.; Academic Press: Boston, MA, USA, 2009; 637p. [Google Scholar]
- Goddard, J.; deShazo, R. Bed bugs (Cimex lectularius) and clinical consequences of their bites. JAMA 2009, 301, 1358–1366. [Google Scholar] [CrossRef]
- Salazar, R.; Castillo-Neyra, R.; Tustin, A.W.; Borrini-Mayorí, K.; Náquira, C.; Levy, M.Z. Bed bugs (Cimex lectularius) as vectors of Trypanosoma cruzi. Am. J. Trop. Med. Hyg. 2015, 92, 331–335. [Google Scholar] [CrossRef]
- Leulmi, H.; Bitam, I.; Berenger, J.M.; Lepidi, H.; Rolain, J.M.; Almeras, L.; Raoult, D.; Parola, P. Competence of Cimex lectularius Bed Bugs for the Transmission of Bartonella quintana, the Agent of Trench Fever. PLoS Negl. Trop. Dis. 2015, 9, e0003789. [Google Scholar]
- Bircher, A.J. Systemic immediate allergic reactions to arthropod stings and bites. Dermatology 2005, 210, 119–127. [Google Scholar] [CrossRef] [PubMed]
- Galvão, C.; Carcavallo, R.; Rocha, D.S.; Jurberg, J. A checklist of the current valid species of the subfamily Triatominae Jeannel, 1919 (Hemiptera, Reduviidae) and their geographical distribution, with nomenclatural and taxonomic notes. Zootaxa 2003, 202, 1–36. [Google Scholar] [CrossRef]
- Schofield, C.J.; Galvão, C. Classification, evolution, and species groups within the Triatominae. Acta Trop. 2009, 110, 88–100. [Google Scholar] [PubMed]
- Clayton, J. Chagas disease 101. Nature 2010, 465, S4–S5. [Google Scholar] [CrossRef]
- Klotz, J.H.; Dorn, P.L.; Logan, J.L.; Stevens, L.; Pinnas, J.L.; Schmidt, J.O.; Klotz, S.A. “Kissing Bugs”: Potential Disease Vectors and Cause of Anaphylaxis. Clin. Infect. Dis. 2010, 50, 1629–1634. [Google Scholar] [CrossRef]
- Anderson, C.; Belnap, C. The Kiss of Death: A Rare Case of Anaphylaxis to the Bite of the “Red Margined Kissing Bug”. Hawaii J. Med. Public Health 2015, 74, 33–35. [Google Scholar]
- Steen, C.J.; Carbonaro, P.A.; Schwartz, R.A. Arthropods in dermatology. J. Am. Acad. Dermatol. 2004, 50, 819–842. [Google Scholar] [CrossRef]
- Adler, P.H. World Blackflies (Diptera: Simuliidae): A Comprehensive Revision of the Taxonomic and Geographical Inventory. Ph.D. Thesis, Department of Plant and Environmental Sciences, Clemson University, Clemson, SC, USA, 2017. Available online: https://biomia.sites.clemson.edu/pdfs/blackflyinventory.pdf (accessed on 29 May 2018).
- Lock, K.; Adriaen, T.; Goethal, P. Effect of water quality on blackflies (Diptera: Simuliidae) in Flanders (Belgium). Limnologica 2014, 44, 58–65. [Google Scholar] [CrossRef]
- Adler, P.H.; Cheke, R.A.; Post, R.J. Evolution, Epidemiology, and population genetics of black flies (Diptera: Simuliidae). Infect. Gen. Evol. 2010, 10, 846–865. [Google Scholar] [CrossRef]
- Procunier, W.S. Cytological approaches to simuliid biosystematics in relation to the epidemiology and control of human onchocerciasis. Genome 1989, 32, 559–569. [Google Scholar] [CrossRef] [PubMed]
- Chattopadhyay, P.; Goyary, D.; Dhiman, S.; Rabha, B.; Hazarika, S.; Veer, V. Immunomodulating effects and hypersensitivity reactions caused by Northeast Indian black fly salivary gland extract. J. Immunotoxicol. 2014, 11, 126–132. [Google Scholar] [CrossRef]
- Farkas, J. Simuliosis. Analysis of dermatological manifestations following blackfly (Simuliidae) bites as observed in the years 1981–1983 in Bratislava (Czechoslovakia). Derm. Beruf. Umwelt. 1984, 32, 171–173. [Google Scholar] [PubMed]
- Morita, S.I.; Bayless, K.M.; Yeates, D.K.; Wiegmann, B.M. Molecular phylogeny of the horse flies: A framework for renewing tabanid taxonomy. Systematic Entomol. 2015, 41, 56–72. [Google Scholar] [CrossRef]
- Croof, H.; Nour, M.; Ali, N. Morphological Identification of Horse Flies (Diptera: Tabanidae) and Estimation of their Seasonal Abundance in Al-Showak District, Gedaref State, Eastern Sudan. IRA-Int. J. Appl. Sci. 2017, 6, 41–54. [Google Scholar] [CrossRef]
- Cheng, T.C. General Parasitology, 2nd ed.; Elsevier Science: Amsterdam, The Netherlands, 2012; 660p. [Google Scholar]
- Hemmer, W.; Focke, M.; Vieluf, D.; Berg-Drewniok, B.; Götz, M.; Jarisch, R. Anaphylaxis induced by horsefly bites: Identification of a 69 kd IgE-binding salivary gland protein from Chrysops spp. (Diptera Tabanidae) by western blot analysis. J. Allergy Clin. Immunol. 1998, 101, 134–136. [Google Scholar] [CrossRef]
- Veraldi, S.; Esposito, L. Skin lesions caused by Tabanus bovinus bites. J. Travel Med. 2017, 24, 1–5. [Google Scholar] [CrossRef][Green Version]
- Sparagano, O.A.E.; George, D.R.; Harrington, D.W.J.; Giangaspero, A. Significance and Control of the Poultry Red Mite, Dermanyssus gallinae. Ann. Rev. Entomol. 2014, 59, 447–466. [Google Scholar] [CrossRef]
- Izri, A.; Andriantsoanirina, V.; Chosidow, O.; Durand, R. Dermatosis Caused by Blood-Sucking Corythucha Ciliata. JAMA Dermatol. 2015, 151, 909–910. [Google Scholar] [CrossRef]
- Foex, B.A. Oral antihistamines for insect bites. Emerg. Med. J. 2006, 23, 721–727. [Google Scholar] [CrossRef]
- Kemp, S.F.; Lockey, R.F.; Simons, F.E.R.; World Allergy Organization ad hoc Committee on Epinephrine in Anaphylaxis. Epinephrine: The Drug of Choice for Anaphylaxis-A Statement of the World Allergy Organization. World Allergy Organ J. 2008, 1, S18–S26. [Google Scholar] [CrossRef] [PubMed]
- Aberer, E. What should one do in case of a tick bite? Curr. Probl. Dermatol. 2009, 37, 155–166. [Google Scholar] [PubMed]
- Pariser, D.M.; Meinking, T.L.; Bell, M.; Ryan, W.G. Topical 0.5% Ivermectin Lotion for Treatment of Head Lice. NEJM 2012, 367, 1687–1693. [Google Scholar] [CrossRef] [PubMed]

| Hematophagous Arthropod | Vectorial Role | Sex/Bloodfeeding | Stage/ Bloodfeeding | Telmophagy/ Solenophagy | Time/ Bloodfeeding | Seasonal Activity | Geographical Dispersion | |
|---|---|---|---|---|---|---|---|---|
| Disease | Pathogenic Agent | |||||||
| Culicidae (Mosquito) | Malaria, yellow fever, chikungunya, dengue fever, Zika, lymphatic filariasis, and japanese encephalitis | Plasmodium sp., Flaviviridae, Togaviridae, Wuchereria sp., Brugia sp., Japanese encephalitis virus | Female | Adult | S 1 | Sunset and night * | Commonly warm seasons | Global |
| Ceratopogonidae (Biting midges) Phlebotominae (Sandflies) | Leishmaniasis, bartonellosis, bluetongue disease, African horse sickness and epizootic hemorrhagic disease, pappataci fever | Leishmania sp., Bartonella sp., Bluetongue virus, Phlebovirus, Toscana virus, African horse virus, Epizootic hemorrhagic disease virus | Female | Adult | T 2 | Sunset and night | Commonly warm seasons | Global |
| Pediculidae, Pthiridae (Lice) | Epidemic typhus, trench fever and louse-borne relapsing fever | Rickettsia prowazekii, Bartonella quintana, Borrelia recurrentis | Both sexes | Nymph and adult | S | Every time | Throughout the year | Global |
| Ixodidae, Argasidae (Tick) | Lyme disease, Rocky Mountain spotted fever, tularemia, Colorado tick fever, human tick-borne ehrlichiosis, babesiosis, tick paralysis, relapsing fever, Q fever | Borrelia burgdorferi, Rickettsia rickettsii, Francisella tularensis, Colorado tick fever (CTF) virus, Ehrlichia chaffeensis, E. ewingii, Babesia microti, Borrelia sp., Coxiella burnetii | Both sexes | Larva, Nymph and adult | T | Every time | Throughout the year | Global |
| Pulicidae (Flea) | Plague, murine typhus, Tungiansis, Tularemia | Yersinia pestis, Rickettsia typhi, Francisella tularensis | Both sexes | Adult | T | Every time | Throughout the year but mainly inwarm seasons | Global |
| Glossinidae (Tsetse fly) | Sleep sickness | Trypanosoma brucei gambiense, Trypanosoma brucei rhodesiense | Both sexes | Adult | T | Diurnal | Throughout the year | Sub-Sahara countries |
| Reduviidae (Kissing bug, Triatomine bug) | Chagas | Trypanosoma cruzi | Both sexes | Nymph and adult | S | Nocturnal | Throughout the year | Latin and South America |
| Simuliidae (Black fly) | Onchocerciasis (River blindness) | Onchocerca volvulus | Female | Adult | T | Diurnal | Throughout the year | Africa, Latin and South America |
| Tabanidae (Horse fly) | Loiasis | Loa loa | Female | Adult | T | Diurnal | Throughout the year | Global |
| Cimicidae (Bed bug) | - | - ** | Both sexes | Nymph and adult | S | Nocturnal | Throughout the year | Global |
| Criteria | Hematophagous Arthropod | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| Mosquito | Biting Midge, Sandfly | Louse | Tsetse Fly | Horse Fly | Black Fly | Triatomine Bug | Tick | Flea | Bed Bug | ||
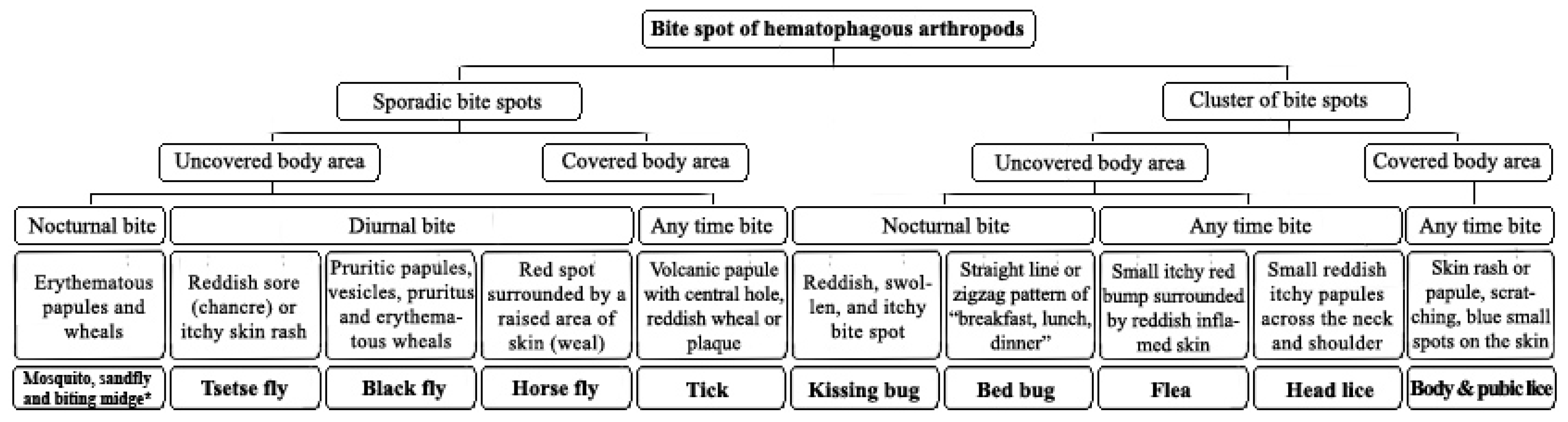
| Clinical manifestations | Red itchy papules | Small reddish swollen bump | Small painful red spot or skin rash, inflammation and irritation with blue spots or small spots of blood on the skin | Reddish sore (chancre), itchy skin rash, boil-like swelling | Reddish raised rash | Pruritic papules, vesicules, pruritus and erythematus weals | Red, swollen and itchy skin, anaphylactic shock | Volcanic papule with central hole, reddish wheal or plaque | Small itchy bumps surrounded by reddish inflamed skin, macule, papule, nodule | Macule, papule, nodule, vesicle, bullae, erythematous and pruritic symptoms, allergy, systematic reaction | |
| Bite spot diagnosis | Bite feeling | Painless | Painful | Painful | Painful | Painful | Painful | Painless | Painless | Painful | Painless |
| Bite spot pattern | Sporadic and separated | Sporadic and separated | Cluster of separated small red bumps | Sporadic and separated | Sporadic and separated | Sporadic and separated | Cluster of separated small red bumps | separated and sporadic | Cluster of 2, 3 or more bites | Zigzag or straight line | |
| Biting time | Evening and night | Evening and night | Any time | Diurnal | Diurnal | Diurnal | Night | Any time | Any time | Night | |
| Location | Exposed area | Exposed area | Throughout body particularly on the scalp, neck and shoulders | Exposed area | Exposed area | Exposed area | Exposed area | Exposed area | Lower extremities, rarely upper body | Exposed area | |
| Treatment | Washing with soap and water, ice compress, antihistamine such as Benadryl, Calamine | Washing with soap and water, ice compress, antihistamine such as Benadryl, Calamine | Combing, pediculicidal shampoo or lotion | Cold compress, calamine, hydrocortisone | Washing with soap and water, ice compress, Benadryl, Calamine | Washing with soap and water, antihistamine such as Benadryl, Calamine | Washing with soap and water, ice compress, epinephrine (in case of systemic reaction) | Removing the tick by tweezers, topical steroids and oral antihistamines | Washing with soap and water, ice compress, Antihistamine such as Benadryl, Calamine | Washing with soap and water, corticosteroids (triamcinolone), antipruritic medications (paroxime, doxepin) | |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Akhoundi, M.; Sereno, D.; Marteau, A.; Bruel, C.; Izri, A. Who Bites Me? A Tentative Discriminative Key to Diagnose Hematophagous Ectoparasites Biting Using Clinical Manifestations. Diagnostics 2020, 10, 308. https://doi.org/10.3390/diagnostics10050308
Akhoundi M, Sereno D, Marteau A, Bruel C, Izri A. Who Bites Me? A Tentative Discriminative Key to Diagnose Hematophagous Ectoparasites Biting Using Clinical Manifestations. Diagnostics. 2020; 10(5):308. https://doi.org/10.3390/diagnostics10050308
Chicago/Turabian StyleAkhoundi, Mohammad, Denis Sereno, Anthony Marteau, Christiane Bruel, and Arezki Izri. 2020. "Who Bites Me? A Tentative Discriminative Key to Diagnose Hematophagous Ectoparasites Biting Using Clinical Manifestations" Diagnostics 10, no. 5: 308. https://doi.org/10.3390/diagnostics10050308
APA StyleAkhoundi, M., Sereno, D., Marteau, A., Bruel, C., & Izri, A. (2020). Who Bites Me? A Tentative Discriminative Key to Diagnose Hematophagous Ectoparasites Biting Using Clinical Manifestations. Diagnostics, 10(5), 308. https://doi.org/10.3390/diagnostics10050308





