High-grade B-cell Lymphoma with MYC and BCL2 Rearrangement Arising from Follicular Lymphoma: Presentation as a Large Peripancreatic Mass
Abstract
1. Introduction
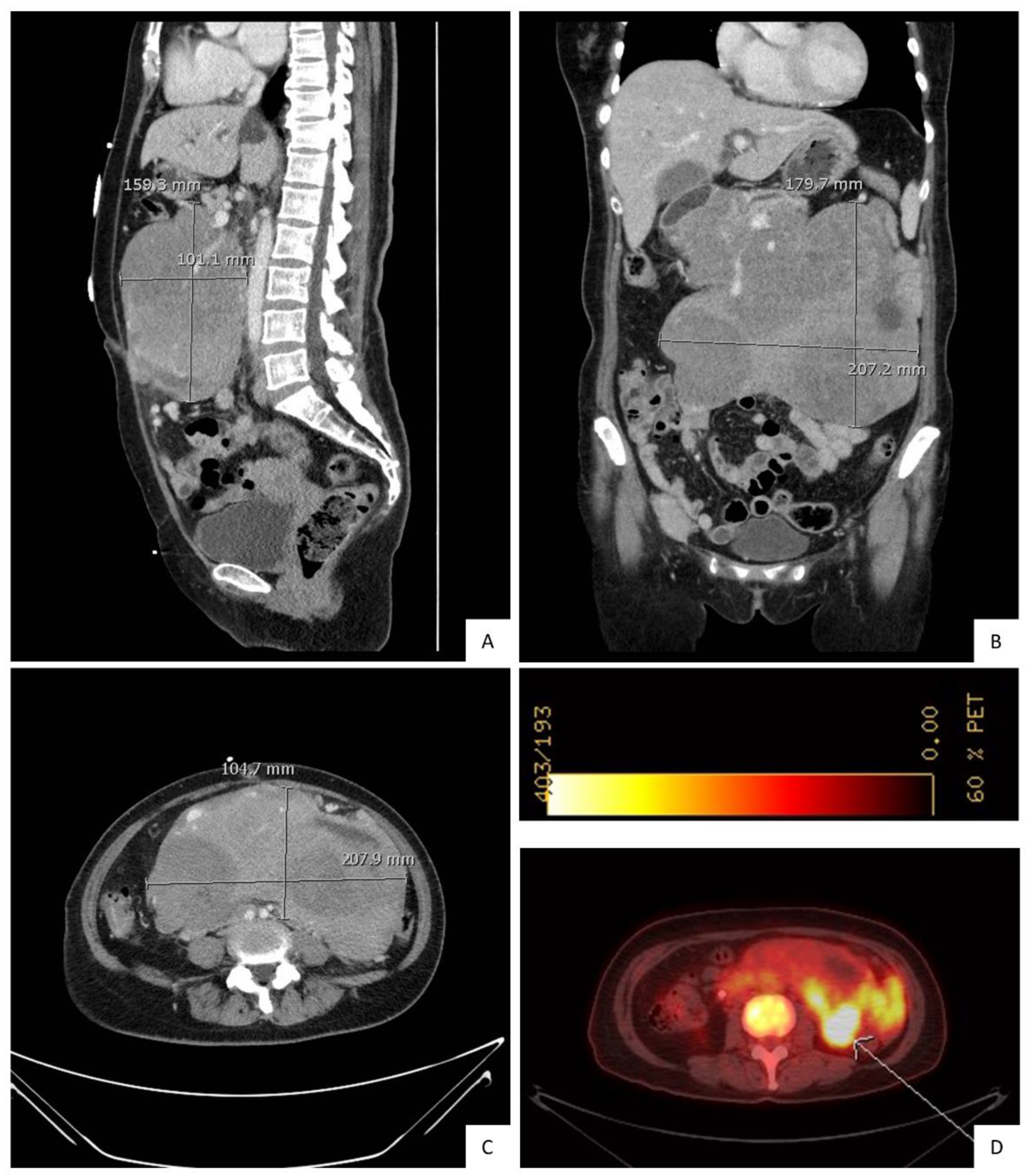
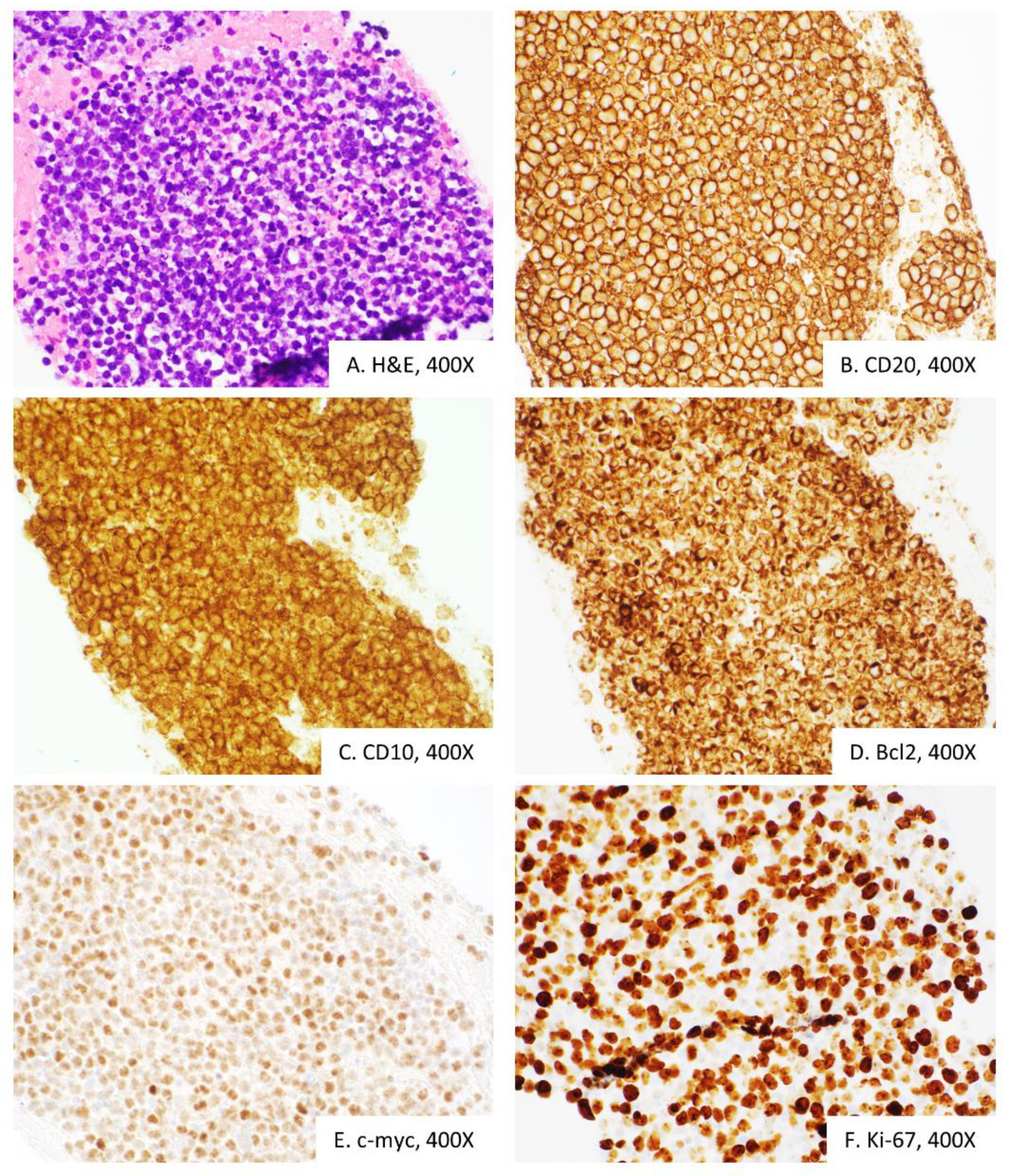
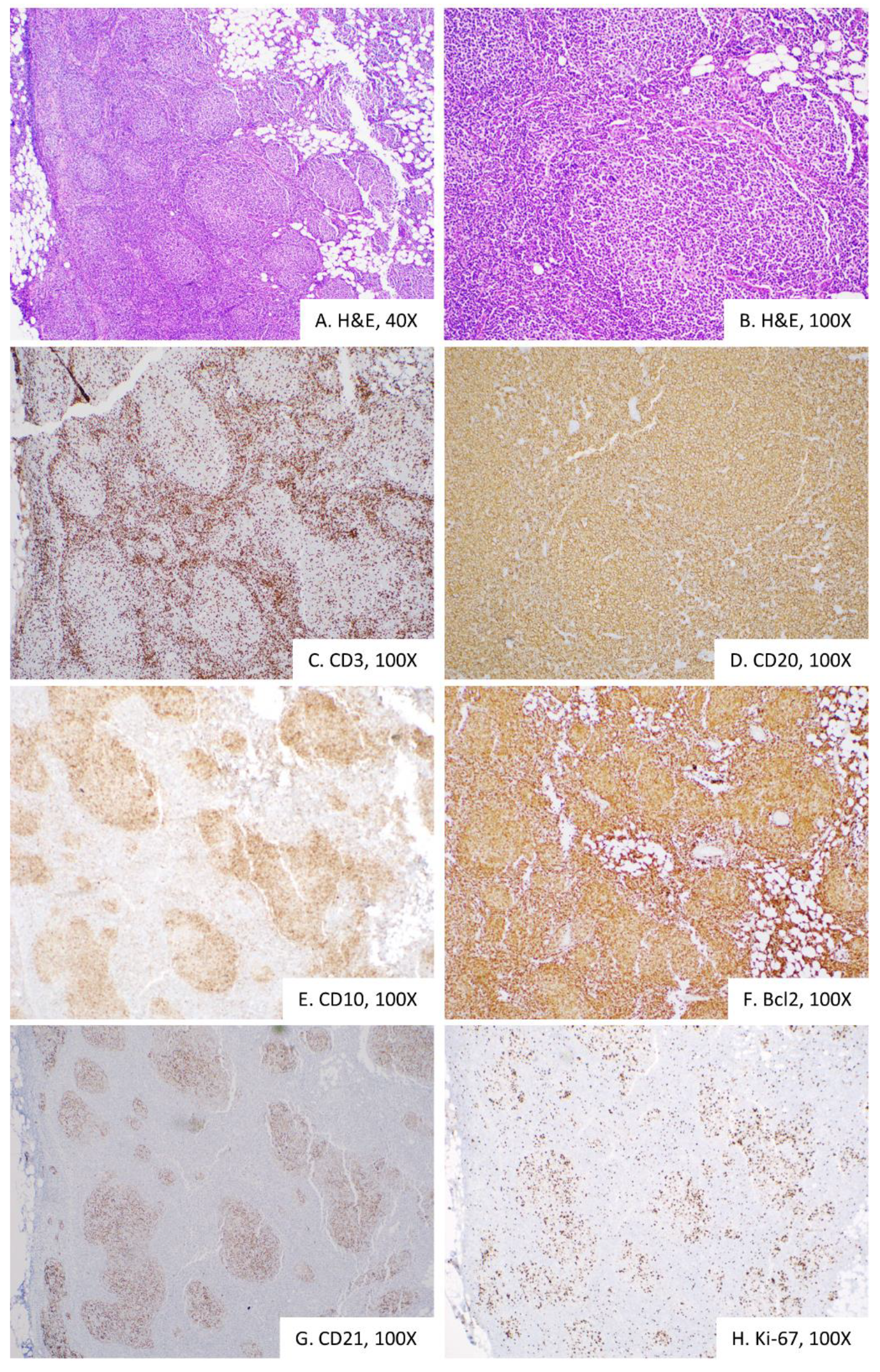
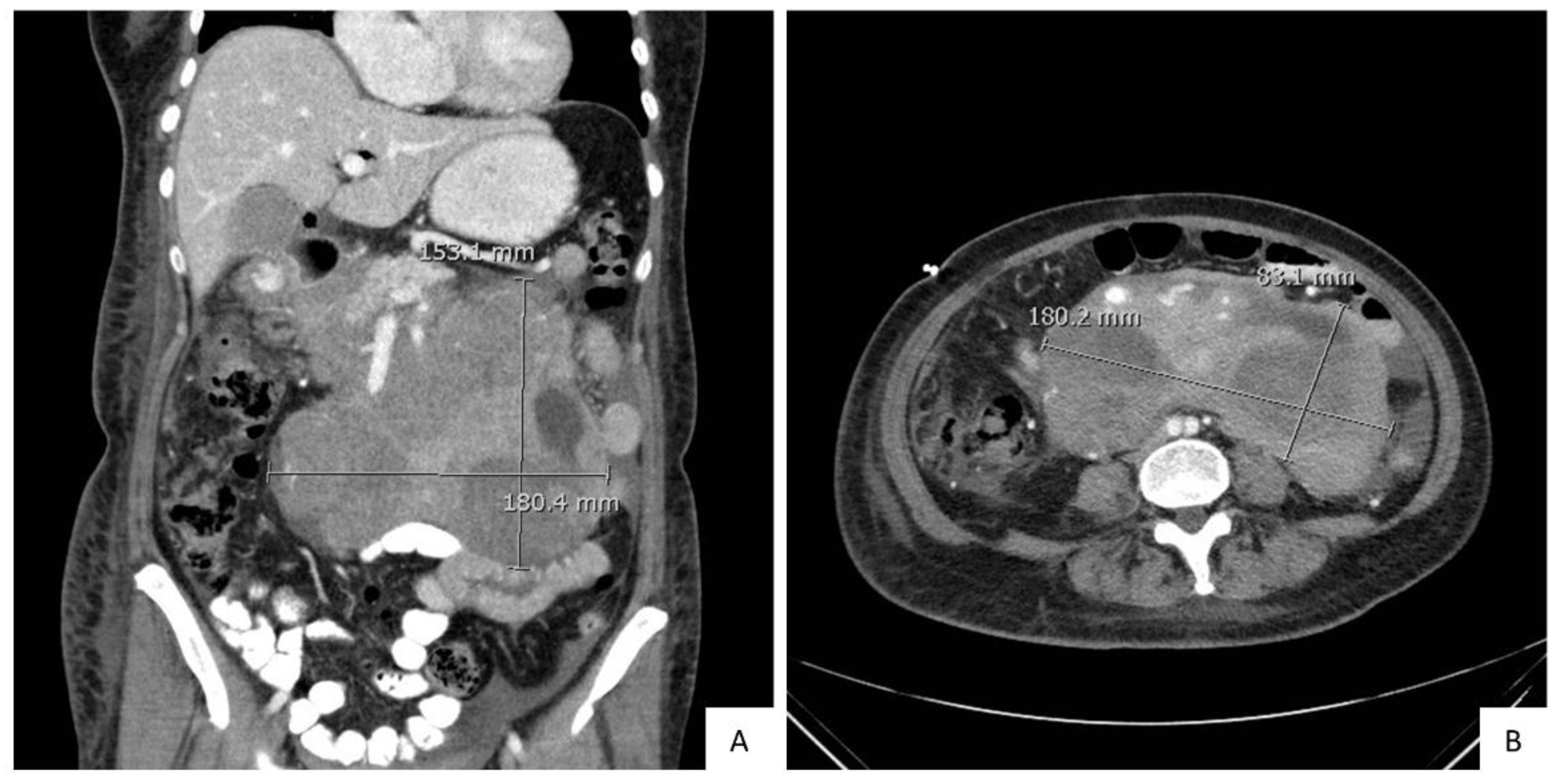
2. Case Presentation
3. Discussion
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Morton, L.M.; Wang, S.S.; Devesa, S.S.; Hartge, P.; Weisenburger, D.D.; Linet, M.S. Lymphoma incidence patterns by WHO subtype in the United States, 1992–2001. Blood 2006, 107, 265–276. [Google Scholar] [CrossRef] [PubMed]
- Conconi, A.; Ponzio, C.; Lobetti-Bodoni, C.; Motta, M.; Rancoita, P.M.; Stathis, A.; Moccia, A.A.; Mazzucchelli, L.; Bertoni, F.; Ghielmini, M.; et al. Incidence, risk factors and outcome of histological transformation in follicular lymphoma. Br. J. Haematol. 2012, 157, 188–196. [Google Scholar] [CrossRef] [PubMed]
- Sant, M.; Allemani, C.; Tereanu, C.; De Angelis, R.; Capocaccia, R.; Visser, O.; Marcos-Gragera, R.; Maynadié, M.; Simonetti, A.; Lutz, J.-M.; et al. Incidence of hematologic malignancies in Europe by morphologic subtype: Results of the HAEMACARE project. Blood 2010, 116, 3724–3734. [Google Scholar] [CrossRef] [PubMed]
- Anderson, T.; Chabner, B.A.; Young, R.C.; Berard, C.W.; Garvin, A.J.; Simon, R.M.; DeVita, V.T. Malignant lymphoma I. The histology and staging of 473 patients at the national cancer institute. Cancer 1982, 50, 2699–2707. [Google Scholar] [CrossRef]
- Gallamini, A.; Borra, A. FDG-PET Scan: A New Paradigm for Follicular Lymphoma Management. Mediterr. J. Hematol. Infect. Dis. 2017, 9, e2017029. [Google Scholar] [CrossRef]
- Swerdlow, S.; Campo, E.; Harris, N.; Jaffe, E.; Pileri, S.; Stein, H.; Thiele, J. WHO Classification of Tumours of Haematopoietic and Lymphoid Tissues; International Agency for Research on Cancer: Lyon, France, 2017; ISBN 978-92-832-4494-3. [Google Scholar]
- Guo, Y.; Karube, K.; Kawano, R.; Yamaguchi, T.; Suzumiya, J.; Huang, G.-S.; Ohshima, K. Low-grade follicular lymphoma with t(14;18) presents a homogeneous disease entity otherwise the rest comprises minor groups of heterogeneous disease entities with Bcl2 amplification, Bcl6 translocation or other gene aberrances. Leukemia 2005, 19, 1058–1063. [Google Scholar] [CrossRef]
- Al-Tourah, A.J.; Gill, K.K.; Chhanabhai, M.; Hoskins, P.J.; Klasa, R.J.; Savage, K.J.; Sehn, L.H.; Shenkier, T.N.; Gascoyne, R.D.; Connors, J.M. Population-Based Analysis of Incidence and Outcome of Transformed Non-Hodgkin’s Lymphoma. J. Clin. Oncol. 2008, 26, 5165–5169. [Google Scholar] [CrossRef]
- Wagner-Johnston, N.D.; Link, B.; Byrtek, M.; Dawson, K.L.; Hainsworth, J.; Flowers, C.R.; Friedberg, J.W.; Bartlett, N. Outcomes of transformed follicular lymphoma in the modern era: A report from the National LymphoCare Study (NLCS). Blood 2015, 126, 851–857. [Google Scholar] [CrossRef]
- Pasqualucci, L.; Khiabanian, H.; Fangazio, M.; Vasishtha, M.; Messina, M.; Holmes, A.B.; Ouillette, P.; Trifonov, V.; Rossi, D.; Tabbò, F.; et al. Genetics of follicular lymphoma transformation. Cell Rep. 2014, 6, 130–140. [Google Scholar] [CrossRef]
- Godfrey, J.; Leukam, M.; Smith, S.M. An update in treating transformed lymphoma. Best Pract. Res. Clin. Haematol. 2018, 31, 251–261. [Google Scholar] [CrossRef]
- Howlett, C.; Snedecor, S.J.; Landsburg, D.J.; Svoboda, J.; Chong, E.A.; Schuster, S.J.; Nasta, S.D.; Feldman, T.; Rago, A.; Walsh, K.M.; et al. Front-line, dose-escalated immunochemotherapy is associated with a significant progression-free survival advantage in patients with double-hit lymphomas: A systematic review and meta-analysis. Br. J. Haematol. 2015, 170, 504–514. [Google Scholar] [CrossRef] [PubMed]
- Biagi, J.J.; Seymour, J.F. Insights into the molecular pathogenesis of follicular lymphoma arising from analysis of geographic variation. Blood 2002, 99, 4265–4275. [Google Scholar] [CrossRef] [PubMed]
- Bloomfield, C.D.; Arthur, D.C.; Frizzera, G.; Levine, E.G.; Peterson, B.; Gajl-Peczalska, K.J. Nonrandom chromosome abnormalities in lymphoma. Cancer Res. 1983, 43, 2975–2984. [Google Scholar] [PubMed]
- Sarkozy, C.; Trněný, M.; Xerri, L.; Wickham, N.; Feugier, P.; Leppä, S.; Brice, P.; Soubeyran, P.; Silva, M.R.; Mounier, C.; et al. Risk Factors and Outcomes for Patients with Follicular Lymphoma Who Had Histologic Transformation after Response to First-Line Immunochemotherapy in the PRIMA Trial. J. Clin. Oncol. 2016, 34, 2575–2582. [Google Scholar] [CrossRef] [PubMed]
- Eide, M.B.; Liestøl, K.; Lingjærde, O.C.; Hystad, M.E.; Kresse, S.H.; Meza-Zepeda, L.; Myklebost, O.; Trøen, G.; Aamot, H.V.; Holte, H.; et al. Genomic alterations reveal potential for higher grade transformation in follicular lymphoma and confirm parallel evolution of tumor cell clones. Blood 2010, 116, 1489–1497. [Google Scholar] [CrossRef] [PubMed]
- Montoto, S.; Davies, A.J.; Matthews, J.; Calaminici, M.; Norton, A.; Amess, J.; Vinnicombe, S.; Waters, R.; Rohatiner, A.Z.; Lister, T.A. Risk and Clinical Implications of Transformation of Follicular Lymphoma to Diffuse Large B-Cell Lymphoma. J. Clin. Oncol. 2007, 25, 2426–2433. [Google Scholar] [CrossRef]
- Bischin, A.M.; Dorer, R.; Aboulafia, D. Transformation of Follicular Lymphoma to a High-Grade B-Cell Lymphoma with MYC and BCL2 Translocations and Overlapping Features of Burkitt Lymphoma and Acute Lymphoblastic Leukemia: A Case Report and Literature Review. Clin. Med. Insights Blood Disord. 2017, 10. [Google Scholar] [CrossRef]
- Kapur, S.; Miles, L. Transformation of Follicular Lymphoma to Double Hit B-Cell Lymphoma Causing Hypercalcemia in a 69-Year-Old Female: A Case Report and Review of the Literature. Case Rep. Hematol. 2014, 2014, 619760. Available online: https://www.hindawi.com/journals/crihem/2014/619760/ (accessed on 6 March 2020). [CrossRef]
- Xu, X.; Zhang, L.; Wang, Y.; Zhang, Q.; Zhang, L.; Sun, B.; Zhang, Y. Double-hit and triple-hit lymphomas arising from follicular lymphoma following acquisition of MYC: Report of two cases and literature review. Int. J. Clin. Exp. Pathol. 2013, 6, 788–794. [Google Scholar]
- Lossos, I.S.; Gascoyne, R.D. Transformation of follicular lymphoma. Best Pract. Res. Clin. Haematol. 2011, 24, 147–163. [Google Scholar] [CrossRef]
- Bonavida, B. Postulated mechanisms of resistance of B-cell non-Hodgkin lymphoma to rituximab treatment regimens: Strategies to overcome resistance. Semin. Oncol. 2014, 41, 667–677. [Google Scholar] [CrossRef] [PubMed]
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shestakova, A.; Rezk, S.; Ghasemizadeh, D.; Nael, A.; Zhao, X. High-grade B-cell Lymphoma with MYC and BCL2 Rearrangement Arising from Follicular Lymphoma: Presentation as a Large Peripancreatic Mass. Diagnostics 2020, 10, 157. https://doi.org/10.3390/diagnostics10030157
Shestakova A, Rezk S, Ghasemizadeh D, Nael A, Zhao X. High-grade B-cell Lymphoma with MYC and BCL2 Rearrangement Arising from Follicular Lymphoma: Presentation as a Large Peripancreatic Mass. Diagnostics. 2020; 10(3):157. https://doi.org/10.3390/diagnostics10030157
Chicago/Turabian StyleShestakova, Anna, Sherif Rezk, Dara Ghasemizadeh, Ali Nael, and Xiaohui Zhao. 2020. "High-grade B-cell Lymphoma with MYC and BCL2 Rearrangement Arising from Follicular Lymphoma: Presentation as a Large Peripancreatic Mass" Diagnostics 10, no. 3: 157. https://doi.org/10.3390/diagnostics10030157
APA StyleShestakova, A., Rezk, S., Ghasemizadeh, D., Nael, A., & Zhao, X. (2020). High-grade B-cell Lymphoma with MYC and BCL2 Rearrangement Arising from Follicular Lymphoma: Presentation as a Large Peripancreatic Mass. Diagnostics, 10(3), 157. https://doi.org/10.3390/diagnostics10030157




