Waist Circumference as a Preventive Tool of Atherogenic Dyslipidemia and Obesity-Associated Cardiovascular Risk in Young Adults Males: A Cross-Sectional Pilot Study
Abstract
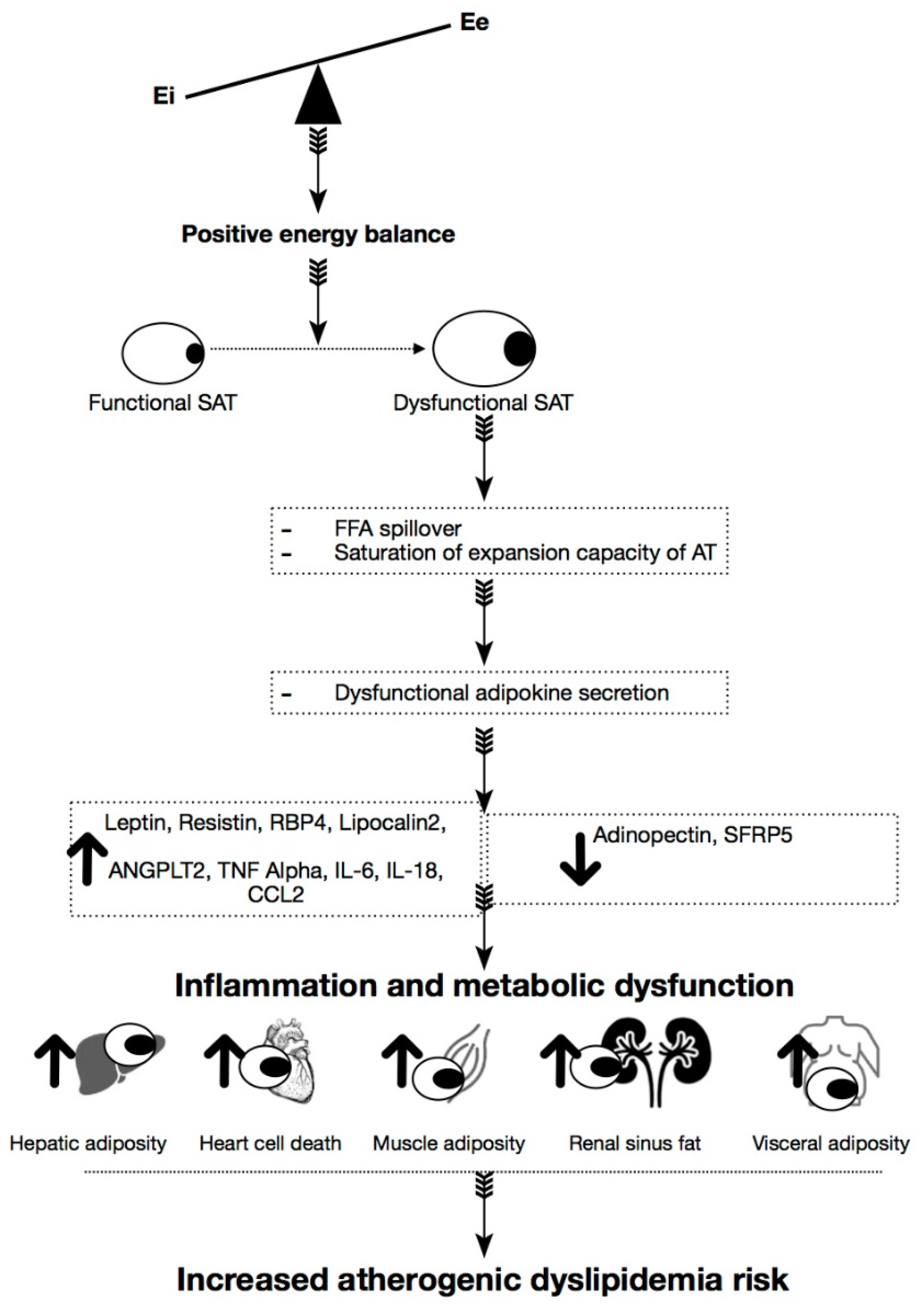
1. Introduction
2. Materials and Methods
2.1. Study Design
2.2. Inclusion/Exclusion Criteria
2.3. Data Collection and Sampling Procedure
2.4. Cardiovascular Risk Assessment
2.5. Statistical Analyses
3. Results
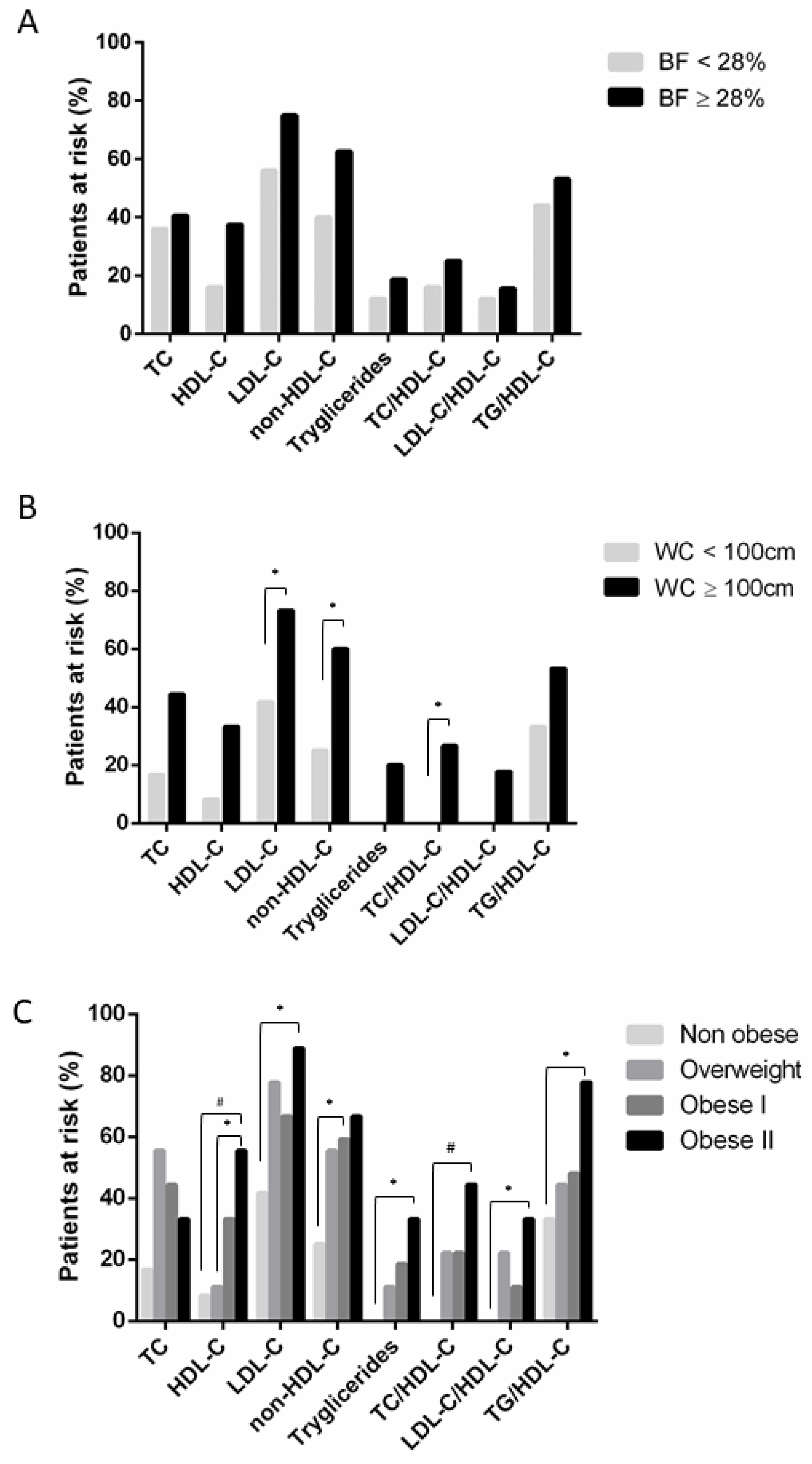
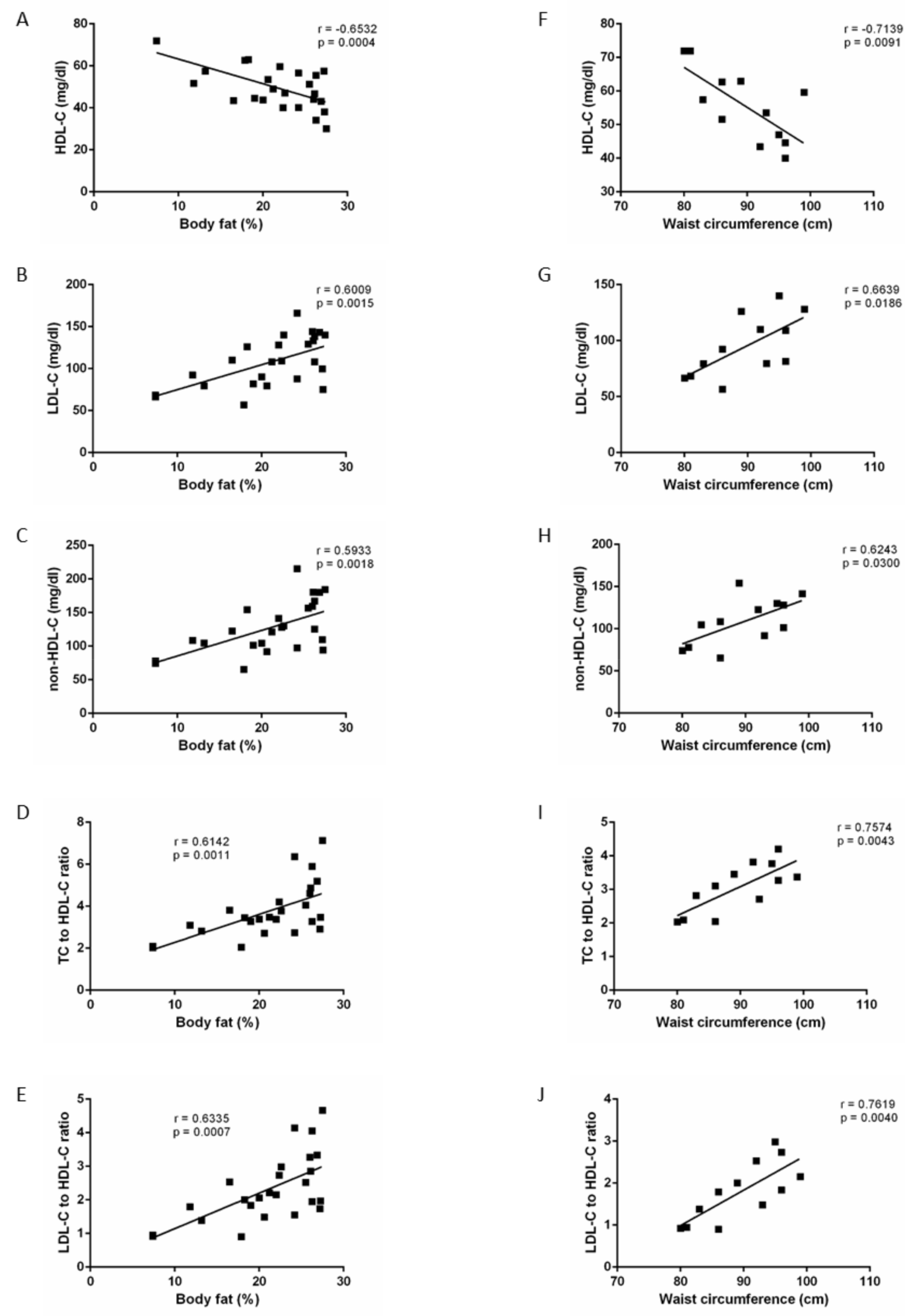
3.1. Body Fat Percentage and Lipid Profile Evaluation
3.2. Waist Circumference and Lipid Profile Evaluation
3.3. Study of Obesity Evolution on Lipid Profile
3.4. Cardiovascular Risk Assessment Associated with Atherogenic Dyslipidaemia
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- WHO. Obesity: Preventing and Managing the Global Epidemic: Report of a WHO Consultation; WHO Technical Report Series No. 894; WHO: Geneva, Switzerland, 2000; Available online: https://www.who.int/nutrition/publications/obesity/WHO_TRS_894/en/ (accessed on 1 November 2020).
- OECD. The Heavy Burden of Obesity: The Economics of Prevention; OECD Health Policy Studies; OECD Publishing: Paris, France, 2019. [Google Scholar] [CrossRef]
- WHO. The Top 10 Causes of Death 2018. 2018. Available online: https://www.who.int/news-room/fact-sheets/detail/the-top-10-causes-of-death (accessed on 1 November 2020).
- Tune, J.D.; Goodwill, A.G.; Sassoon, D.J.; Mather, K.J. Cardiovascular consequences of metabolic syndrome. Transl. Res. 2017, 183, 57–70. [Google Scholar] [CrossRef]
- Youmans, Q.R.; Hastings-Spaine, L.; Princewill, O.; Shobayo, T.; Okwuosa, I.S. Disparities in cardiovascular care: Past, present, and solutions. Clevel. Clin. J. Med. 2019, 86, 621–632. [Google Scholar] [CrossRef]
- Daviglus, M.L.; Liu, K.; Yan, L.L.; Pirzada, A.; Garside, D.B.; Schiffer, L.; Dyer, A.R.; Greenland, P.; Stamler, J. Body mass index in middle age and health-related quality of life in older age: The Chicago heart association detection project in industry study. Arch. Intern. Med. 2003, 163, 2448–2455. [Google Scholar] [CrossRef][Green Version]
- Hernández-Reyes, A.; Cámara-Martos, F.; Molina-Luque, R.; Romero-Saldaña, M.; Molina-Recio, G.; Moreno-Rojas, R. Changes in body composition with a hypocaloric diet combined with sedentary, moderate and high-intense physical activity: A randomized controlled trial. BMC Womens Health 2019, 19, 167. [Google Scholar] [CrossRef]
- Ghachem, A.; Paquin, J.; Brochu, M.; Dionne, I.J. Should Waist Circumference Cutoffs in the Context of Cardiometabolic Risk Factor Assessment be Specific to Sex, Age, and BMI? Metab. Syndr. Relat. Disord. 2018, 16, 366–374. [Google Scholar] [CrossRef]
- Ross, R.; Neeland, I.J.; Yamashita, S.; Shai, I.; Seidell, J.; Magni, P.; Santos, R.D.; Arsenault, B.; Cuevas, A.; Hu, F.B.; et al. Waist circumference as a vital sign in clinical practice: A Consensus Statement from the IAS and ICCR Working Group on Visceral Obesity. Nat. Rev. Endocrinol. 2020, 16, 177–189. [Google Scholar] [CrossRef]
- Song, X.; Jousilahti, P.; Stehouwer, C.D.; Söderberg, S.; Onat, A.; Laatikainen, T.; Yudkin, J.S.; Dankner, R.; Morris, R.; Tuomilehto, J.; et al. Comparison of various surrogate obesity indicators as predictors of cardiovascular mortality in four European populations. Eur. J. Clin. Nutr. 2013, 67, 1298–1302. [Google Scholar] [CrossRef]
- Nam, G.E.; Kim, Y.H.; Han, K.; Jung, J.H.; Park, Y.G.; Lee, K.W.; Rhee, E.J.; Son, J.W.; Lee, S.S.; Kwon, H.S.; et al. Obesity Fact Sheet in Korea, 2018: Data Focusing on Waist Circumference and Obesity-Related Comorbidities. J. Obes. Metab. Syndr. 2019, 28, 236–245. [Google Scholar] [CrossRef]
- Shen, W.; Punyanitya, M.; Chen, J.; Gallagher, D.; Albu, J.; Pi-Sunyer, X.; Lewis, C.E.; Grunfeld, C.; Heshka, S.; Heymsfield, S.B. Waist circumference correlates with metabolic syndrome indicators better than percentage fat. Obesity (Silver Spring) 2006, 14, 727–736. [Google Scholar] [CrossRef]
- Zhang, A.; Yao, Y.; Xue, Z.; Guo, X.; Dou, J.; Lv, Y.; Shen, L.; Yu, Y.; Jin, L. A Study on the Factors Influencing Triglyceride Levels among Adults in Northeast China. Sci. Rep. 2018, 8, 6388. [Google Scholar] [CrossRef]
- Rydén, M.; Arner, P. Subcutaneous Adipocyte Lipolysis Contributes to Circulating Lipid Levels. Arter. Thromb. Vasc. Biol. 2017, 37, 1782–1787. [Google Scholar] [CrossRef]
- Pouliot, M.C.; Després, J.P.; Lemieux, S.; Moorjani, S.; Bouchard, C.; Tremblay, A.; Nadeau, A.; Lupien, P.J. Waist circumference and abdominal sagittal diameter: Best simple anthropometric indexes of abdominal visceral adipose tissue accumulation and related cardiovascular risk in men and women. Am. J. Cardiol. 1994, 73, 460–468. [Google Scholar] [CrossRef]
- Carr, D.B.; Utzschneider, K.M.; Hull, R.L.; Kodama, K.; Retzlaff, B.M.; Brunzell, J.D.; Shofer, J.B.; Fish, B.E.; Knopp, R.H.; Kahn, S.E. Intra-abdominal fat is a major determinant of the National Cholesterol Education Program Adult Treatment Panel III criteria for the metabolic syndrome. Diabetes 2004, 53, 2087–2094. [Google Scholar] [CrossRef]
- Meigs, J.B.; Wilson, P.W.; Fox, C.S.; Vasan, R.S.; Nathan, D.M.; Sullivan, L.M.; D’Agostino, R.B. Body mass index, metabolic syndrome, and risk of type 2 diabetes or cardiovascular disease. J. Clin. Endocrinol. Metab. 2006, 91, 2906–2912. [Google Scholar] [CrossRef]
- Ardern, C.I.; Janssen, I.; Ross, R.; Katzmarzyk, P.T. Development of health-related waist circumference thresholds within BMI categories. Obes. Res. 2004, 12, 1094–1103. [Google Scholar] [CrossRef]
- Ballantyne, C.M.; Olsson, A.G.; Cook, T.J.; Mercuri, M.F.; Pedersen, T.R.; Kjekshus, J. Influence of low high-density lipoprotein cholesterol and elevated triglyceride on coronary heart disease events and response to simvastatin therapy in 4S. Circulation 2001, 104, 3046–3051. [Google Scholar] [CrossRef]
- Vega, G.L.; Barlow, C.E.; Grundy, S.M.; Leonard, D.; DeFina, L.F. Triglyceride-to-high-density-lipoprotein-cholesterol ratio is an index of heart disease mortality and of incidence of type 2 diabetes mellitus in men. J. Investig. Med. 2014, 62, 345–349. [Google Scholar] [CrossRef]
- Ascaso, J.; Gonzalez Santos, P.; Hernandez Mijares, A.; Mangas Rojas, A.; Masana, L.; Millan, J.; Pallardo, L.F.; Pedro-Botet, J.; Perez Jimenez, F.; Pintó, X.; et al. Management of dyslipidemia in the metabolic syndrome: Recommendations of the Spanish HDL-Forum. Am. J. Cardiovasc. Drugs 2007, 7, 39–58. [Google Scholar] [CrossRef]
- Encuesta Nacional de Salud 2011/2012. Ministerio de Sanidad, Servicios Sociales e Igualdad. Available online: http://www.msssi.gob.es/estadEstudios/estadisticas/encuestaNacional/encuesta2011.htm (accessed on 20 December 2016).
- Gidding, S.S.; Allen, N.B. Cholesterol and Atherosclerotic Cardiovascular Disease: A Lifelong Problem. J. Am. Heart Assoc. 2019, 8, e012924. [Google Scholar] [CrossRef]
- Robinson, J.G.; Williams, K.J.; Gidding, S.; Borén, J.; Tabas, I.; Fisher, E.A.; Packard, C.; Pencina, M.; Fayad, Z.A.; Mani, V.; et al. Eradicating the Burden of Atherosclerotic Cardiovascular Disease by Lowering Apolipoprotein B Lipoproteins Earlier in Life. J. Am. Heart Assoc. 2018, 7, e009778. [Google Scholar] [CrossRef]
- Robinson, J.G.; Huijgen, R.; Ray, K.; Persons, J.; Kastelein, J.J.; Pencina, M.J. Determining When to Add Nonstatin Therapy: A Quantitative Approach. J. Am. Coll. Cardiol. 2016, 68, 2412–2421. [Google Scholar] [CrossRef]
- American Heart Association. Cardiovascular Disease: A Costly Burden for America. Cost Projections through 2035. 2017. Available online: http://www.heart.org/idc/groups/heart-public/@wcm/@adv/documents/downloadable/ucm_491543.pdf (accessed on 10 January 2020).
- Ascaso, J.F.; Millán, J.; Hernández-Mijares, A.; Blasco, M.; Brea, Á.; Díaz, Á.; Mantilla, T.; Pedro-Botet, J.; Pintó, X.; Grupo de trabajo sobre Dislipemia Aterogénica de la SEA. Consensus document on the management of the atherogenic dyslipidaemia of the Spanish Society of Arteriosclerosis. Clin. Investig. Arterioscler. 2017, 29, 86–91. [Google Scholar] [CrossRef]
- Tomlinson, D.J.; Erskine, R.M.; Morse, C.I.; Onambélé, G.L. Body Fat Percentage, Body Mass Index, Fat Mass Index and the Ageing Bone: Their Singular and Combined Roles Linked to Physical Activity and Diet. Nutrients 2019, 11, 195. [Google Scholar] [CrossRef]
- Schubert, M.M.; Seay, R.F.; Spain, K.K.; Clarke, H.E.; Taylor, J.K. Reliability and validity of various laboratory methods of body composition assessment in young adults. Clin. Physiol. Funct. Imaging 2019, 39, 150–159. [Google Scholar] [CrossRef]
- Lean, M.E.; Han, T.S.; Morrison, C.E. Waist circumference as a measure for indicating need for weight management. BMJ 1995, 311, 158–161. [Google Scholar] [CrossRef]
- Friedewald, W.T.; Levy, R.I.; Fredrickson, D.S. Estimation of the concentration of low-density lipoprotein cholesterol in plasma, without use of the preparative ultracentrifuge. Clin. Chem. 1972, 18, 499–502. [Google Scholar] [CrossRef]
- von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P.; STROBE Initiative. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. Int. J. Surg. 2014, 12, 1495–1499. [Google Scholar] [CrossRef]
- Vesa, C.M.; Popa, L.; Popa, A.R.; Rus, M.; Zaha, A.A.; Bungau, S.; Tit, D.M.; Corb Aron, R.A.; Zaha, D.C. Current Data Regarding the Relationship between Type 2 Diabetes Mellitus and Cardiovascular Risk Factors. Diagnostics 2020, 10, 314. [Google Scholar] [CrossRef]
- Gheorghe, G.; Toth, P.P.; Bungau, S.; Behl, T.; Ilie, M.; Pantea Stoian, A.; Bratu, O.G.; Bacalbasa, N.; Rus, M.; Diaconu, C.C. Cardiovascular Risk and Statin Therapy Considerations in Women. Diagnostics 2020, 10, 483. [Google Scholar] [CrossRef]
- Song, X.; Tabák, A.G.; Zethelius, B.; Yudkin, J.S.; Söderberg, S.; Laatikainen, T.; Stehouwer, C.D.; Dankner, R.; Jousilahti, P.; Onat, A.; et al. Obesity attenuates gender differences in cardiovascular mortality. Cardiovasc. Diabetol. 2014, 13, 144. [Google Scholar] [CrossRef]
- Wilkins, E.; Wilson, L.; Wickramasinghe, K.; Bhatnagar, P.; Leal, J.; Luengo-Fernandez, R.; Burns, R.; Rayner, M.; Townsend, N. European Cardiovascular Disease Statistics; Eur Hear Network: Brussels, Belgium, 2017; pp. 1–192. Available online: http://www.ehnheart.org/images/CVD-statistics-report-August-2017.pdf (accessed on 11 November 2020).
- Khan, S.S.; Ning, H.; Wilkins, J.T.; Allen, N.; Carnethon, M.; Berry, J.D.; Sweis, R.N.; Lloyd-Jones, D.M. Association of Body Mass Index With Lifetime Risk of Cardiovascular Disease and Compression of Morbidity. JAMA Cardiol. 2018, 3, 280–287. [Google Scholar] [CrossRef]
- Clark, L.T. Cholesterol and heart disease: Current concepts in pathogenesis and treatment. J. Natl. Med. Assoc. 1986, 78, 743–751. [Google Scholar]
- Furtado, J.M.; Almeida, S.M.; Mascarenhas, P.; Ferraz, M.E.; Ferreira, J.C.; Vilanova, M.; Monteiro, M.P.; Ferraz, F.P. Anthropometric features as predictors of atherogenic dyslipidemia and cardiovascular risk in a large population of school-aged children. PLoS ONE 2018, 13, e0197922. [Google Scholar] [CrossRef]
- Holvoet, P.; Jenny, N.S.; Schreiner, P.J.; Tracy, R.P.; Jacobs, D.R.; Multi-Ethnic Study of Atherosclerosis. The relationship between oxidized LDL and other cardiovascular risk factors and subclinical CVD in different ethnic groups: The Multi-Ethnic Study of Atherosclerosis (MESA). Atherosclerosis 2007, 194, 245–252. [Google Scholar] [CrossRef]
- Kwon, D.; Yi, J.J.; Ohrr, H.; Yi, S.W. Total cholesterol and mortality from ischemic heart disease and overall cardiovascular disease in Korean adults. Medicine (Baltimore) 2019, 98, e17013. [Google Scholar] [CrossRef]
- Stamler, J.; Daviglus, M.L.; Garside, D.B.; Dyer, A.R.; Greenland, P.; Neaton, J.D. Relationship of baseline serum cholesterol levels in 3 large cohorts of younger men to long-term coronary, cardiovascular, and all-cause mortality and to longevity. JAMA 2000, 284, 311–318. [Google Scholar] [CrossRef]
- Klag, M.J.; Ford, D.E.; Mead, L.A.; He, J.; Whelton, P.K.; Liang, K.Y.; Levine, D.M. Serum cholesterol in young men and subsequent cardiovascular disease. N. Engl. J. Med. 1993, 328, 313–318. [Google Scholar] [CrossRef]
- Jeong, S.M.; Choi, S.; Kim, K.; Kim, S.M.; Lee, G.; Park, S.Y.; Kim, Y.Y.; Son, J.S.; Yun, J.M.; Park, S.M. Effect of Change in Total Cholesterol Levels on Cardiovascular Disease Among Young Adults. J. Am. Heart Assoc. 2018, 7, e008819. [Google Scholar] [CrossRef]
- Vinik, A.I. The metabolic basis of atherogenic dyslipidemia. Clin. Cornerstone 2005, 7, 27–35. [Google Scholar] [CrossRef]
- Osadnik, K.; Osadnik, T.; Lonnie, M.; Lejawa, M.; Regula, R.; Fronczek, M.; Gawlita, M.; Wądolowska, L.; Gąsior, M.; Pawlas, N. Metabolically healthy obese and metabolic syndrome of the lean: The importance of diet quality. Analysis of MAGNETIC cohort. Nutr. J. 2020, 19, 19. [Google Scholar] [CrossRef]
- Siles Gutiérrez, M.; Goldaracena Tanco, M.; Avila Muñoz, L.; Sánchez-Eznarriaga, B.C. Consumo de hipolipemiantes en España, 1987-2000. Rev. Esp. Salud. Publica 2001, 75, 129–141. [Google Scholar] [CrossRef]
- Rodríguez Artalejo, F.; del Rey Calero, J. La prevención secundaria de la enfermedad cardiovascular es prioritaria pero resulta insuficiente. Rev. Esp. Salud. Publica 1999, 73, 441–443. [Google Scholar] [CrossRef]
- Perk, J.; De Backer, G.; Gohlke, H.; Graham, I.; Reiner, Ž.; Verschuren, M.; Albus, C.; Benlian, P.; Boysen, G.; Cifkova, R.; et al. European Guidelines on cardiovascular disease prevention in clinical practice (version 2012). The Fifth Joint Task Force of the European Society of Cardiology and Other Societies on Cardiovascular Disease Prevention in Clinical Practice (constituted by representatives of nine societies and by invited experts). Eur. Hearth J. 2012, 33, 1635–1701. [Google Scholar] [CrossRef]
- Kazi, D.S.; Moran, A.E.; Coxson, P.G.; Penko, J.; Ollendorf, D.A.; Pearson, S.D.; Tice, J.A.; Guzman, D.; Bibbins-Domingo, K. Cost-effectiveness of PCSK9 Inhibitor Therapy in Patients With Heterozygous Familial Hypercholesterolemia or Atherosclerotic Cardiovascular Disease. JAMA 2016, 316, 743–753. [Google Scholar] [CrossRef]
- Olry de Labry Lima, A.; Gimeno Ballester, V.; Sierra Sánchez, J.F.; Matas Hoces, A.; González-Outón, J.; Alegre Del Rey, E.J. Cost-effectiveness and Budget Impact of Treatment with Evolocumab Versus Statins and Ezetimibe for Hypercholesterolemia in Spain. Rev. Esp. Cardiol. (Engl. Ed.) 2018, 71, 1027–1035. [Google Scholar] [CrossRef]
| Biomarkers and Ratios | Values |
|---|---|
| TG | >150 mg/dl |
| HDL-C | <40 mg/dl |
| LDL-C | >100 mg/dl |
| non-HDL-C | >130 mg/dl |
| TC/HDL-C | >5 |
| TG/HDL-C | >2 |
| LDL-C/HDL-C | >2.5 |
| Variable | Total (n = 57) |
|---|---|
| Age (years) | 35.9 ± 10.85 |
| Height (m) | 1.77 ± 0.07 |
| Weight (Kg) | 101.2 ± 19.02 |
| BMI (Kg/m2) | 32.4 ± 6.08 |
| Body fat (%) | 27.4 ± 7.13 |
| Muscle mass (Kg) | 68.8 ± 8.38 |
| WC (cm) | 112.2 ± 15.90 |
| Variable | Total (n = 57) |
|---|---|
| Triglycerides (mg/dl) | 105.4 ± 56.58 |
| Total Cholesterol (mg/dl). | 185.6 ± 42.15 |
| HDL-C (mg/dl) | 48.1 ± 11.87 |
| LDL-C (mg/dl) | 118.9 ± 33.09 |
| non-HDL-C (mg/dl) | 137.5 ± 40.41 |
| TC/HDL-C ratio | 4.02 ± 1.11 |
| TG/HDL-C ratio | 2.39 ± 1.54 |
| LDL-C/HDL-C ratio | 2.60 ± 0.89 |
| Variable | Non-Obese (n = 25) | Obese (n = 32) | p-Value |
|---|---|---|---|
| Age (years) | 33.1 ± 9.63 | 38.1 ± 11.4 | 0.0854 |
| Height (m) | 1.77 ± 0.07 | 1.76 ± 0.06 | 0.5188 |
| Weight (Kg) | 88.9 ± 15.27 | 110.8 ± 16.03 | <0.0001 |
| BMI (Kg/m2) | 28.3 ± 4.76 | 35.7 ± 4.98 | <0.0001 |
| Body fat (%) | 21.1 ± 6.01 | 32.2 ± 2.80 | <0.0001 |
| Muscle mass (Kg) | 66.0 ± 8.61 | 71.1 ± 7.59 | 0.0212 |
| WC (cm) | 100.7 ± 12.68 | 121.3 ± 11.84 | <0.0001 |
| Variable | Non-Obese (n = 25) | Obese (n = 32) | p-Value |
|---|---|---|---|
| Triglycerides (mg/dl) | 100.1 ± 58.97 | 126.2 ± 55.23 | 0.5336 |
| Total Cholesterol (mg/dl) | 178.0 ± 33.41 | 191.5 ± 47.56 | 0.2320 |
| HDL-C (mg/dl) | 50.2 ± 10.78 | 46.4 ± 12.57 | 0.2356 |
| LDL-C (mg/dl) | 107.9 ± 29.64 | 127.5 ± 33.54 | 0.0254 |
| non-HDL-C (mg/dl) | 127.7 ± 38.55 | 145.1 ± 40.78 | 0.1089 |
| TC/HDL-C ratio | 3.76 ± 1.31 | 4.22 ± 0.90 | 0.1243 |
| TG/HDL-C ratio | 2.23 ± 1.71 | 2.51 ± 1.40 | 0.4979 |
| LDL-C/HDL-C ratio | 2.32 ± 1.00 | 2.82 ± 0.72 | 0.0331 |
| Variable | Non-Obese (n = 12) | Obese (n = 45) | p-Value |
|---|---|---|---|
| Age (years) | 30.7 ± 8.73 | 37.3 ± 11.0 | 0.0605 |
| Height (m) | 1.78 ± 0.08 | 1.76 ± 0.06 | 0.3523 |
| Weight (Kg) | 78.1 ± 8.92 | 107.4 ± 16.01 | <0.0001 |
| BMI (Kg/m2) | 24.5 ± 2.06 | 34.5 ± 4.95 | <0.0001 |
| Body fat (%) | 16.6 ± 5.47 | 30.2 ± 4.14 | <0.0001 |
| Muscle mass (Kg) | 61.5 ± 6.86 | 70.8 ± 7.68 | 0.0004 |
| WC (cm) | 89.7 ± 6.42 | 118.3 ± 11.64 | <0.0001 |
| Variables | Non-Obese (n = 12) | Obese (n = 45) | p-Value |
|---|---|---|---|
| Triglycerides (mg/dl) | 75.3 ± 33.84 | 113.5 ± 58.94 | 0.0366 |
| TC (mg/dl) | 163.8 ± 25.02 | 192.4 ± 44.06 | 0.0434 |
| HDL-C (mg/dl) | 55.5 ± 10.71 | 46.1 ± 11.46 | 0.0131 |
| LDL-C (mg/dl) | 94.7 ± 27.28 | 125.3 ± 31.72 | 0.0035 |
| non-HDL-C (mg/dl) | 108.3 ± 27.85 | 145.2 ± 39.88 | 0.0039 |
| TC/HDL-C ratio | 3.06 ± 0.73 | 4.27 ± 1.06 | 0.0004 |
| TG/HDL-C ratio | 1.42 ± 0.69 | 2.64 ± 1.60 | 0.0131 |
| LDL-C/HDL-C ratio | 1.80 ± 0.71 | 2.81 ± 0.80 | 0.0002 |
| Variable | Non-Obese (n = 12) | Overweight (n = 9) | Obese I (n= 27) | Obese II (n = 9) |
|---|---|---|---|---|
| TG (mg/dl) | 75.3 ± 33.84 | 107.1 ± 64.99 | 106.8 ± 51.92 | 139.9 ± 71.71 a |
| TC (mg/dl) | 163.8 ± 25.02 | 199.0 ± 43.61 | 190.0 ± 46.67 | 187.8 ± 40.09 |
| HDL-C (mg/dl) | 55.5 ± 10.71 | 49.0 ± 7.31 | 47.6 ± 13.01 a | 38.7 ± 6.45 a,b,c |
| LDL-C (mg/dl) | 94.7 ± 27.28 | 126.2 ± 31.57 a | 124.1 ± 33.90 a | 128.2 ± 28.03 a |
| non-HDL-C (mg/dl) | 108.3 ± 27.85 | 150.0 ± 44.87 a | 142.4 ± 39.96 a | 149.0 ± 38.41 a |
| TC/HDL-C ratio | 3.06 ± 0.73 | 4.15 ± 1.19 a | 4.10 ± 0.93 a | 4.92 ± 1.16 a,c |
| TG/HDL-C ratio | 1.42 ± 0.69 | 2.28 ± 1.46 | 2.41 ± 1.35 a | 3.72 ± 2.11 a,b,c |
| LDL-C/HDL-C ratio | 1.80 ± 0.71 | 2.63 ± 0.84 a | 2.68 ± 0.68 a | 3.39 ± 0.94 a,b,c |
| BFP Correlation | WC Correlation | |
|---|---|---|
| TG (mg/dl) | NS | NS |
| TC (mg/dl) | r = 0.2790; p = 0.0356 | NS |
| HDL-C (mg/dl) | r = −0.2863; p = 0.0309 | r = −0.4295; p = 0.0009 |
| LDL-C (mg/dl) | r = 0.4354; p = 0.0007 | r = 0.2976; p = 0.0246 |
| non-HDL-C (mg/dl) | r = 0.3751; p = 0.0040 | r = 0.2615; p = 0.0494 |
| TC/HDL-C ratio | r = 0.3859; p = 0.0030 | r = 0.4272; p = 0.0009 |
| TG/HDL-C ratio | NS | r = 0.3163; p = 0.0165 |
| LDL-C/HDL-C ratio | r = 0.4475; p = 0.0005 | r = 0.4831; p = 0.0001 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hernández-Reyes, A.; Vidal, Á.; Moreno-Ortega, A.; Cámara-Martos, F.; Moreno-Rojas, R. Waist Circumference as a Preventive Tool of Atherogenic Dyslipidemia and Obesity-Associated Cardiovascular Risk in Young Adults Males: A Cross-Sectional Pilot Study. Diagnostics 2020, 10, 1033. https://doi.org/10.3390/diagnostics10121033
Hernández-Reyes A, Vidal Á, Moreno-Ortega A, Cámara-Martos F, Moreno-Rojas R. Waist Circumference as a Preventive Tool of Atherogenic Dyslipidemia and Obesity-Associated Cardiovascular Risk in Young Adults Males: A Cross-Sectional Pilot Study. Diagnostics. 2020; 10(12):1033. https://doi.org/10.3390/diagnostics10121033
Chicago/Turabian StyleHernández-Reyes, Alberto, Ángela Vidal, Alicia Moreno-Ortega, Fernando Cámara-Martos, and Rafael Moreno-Rojas. 2020. "Waist Circumference as a Preventive Tool of Atherogenic Dyslipidemia and Obesity-Associated Cardiovascular Risk in Young Adults Males: A Cross-Sectional Pilot Study" Diagnostics 10, no. 12: 1033. https://doi.org/10.3390/diagnostics10121033
APA StyleHernández-Reyes, A., Vidal, Á., Moreno-Ortega, A., Cámara-Martos, F., & Moreno-Rojas, R. (2020). Waist Circumference as a Preventive Tool of Atherogenic Dyslipidemia and Obesity-Associated Cardiovascular Risk in Young Adults Males: A Cross-Sectional Pilot Study. Diagnostics, 10(12), 1033. https://doi.org/10.3390/diagnostics10121033






