Characterizing Interstellar Medium, Planetary Surface and Deep Environments by Spectroscopic Techniques Using Unique Simulation Chambers at Centro de Astrobiologia (CAB)
Abstract
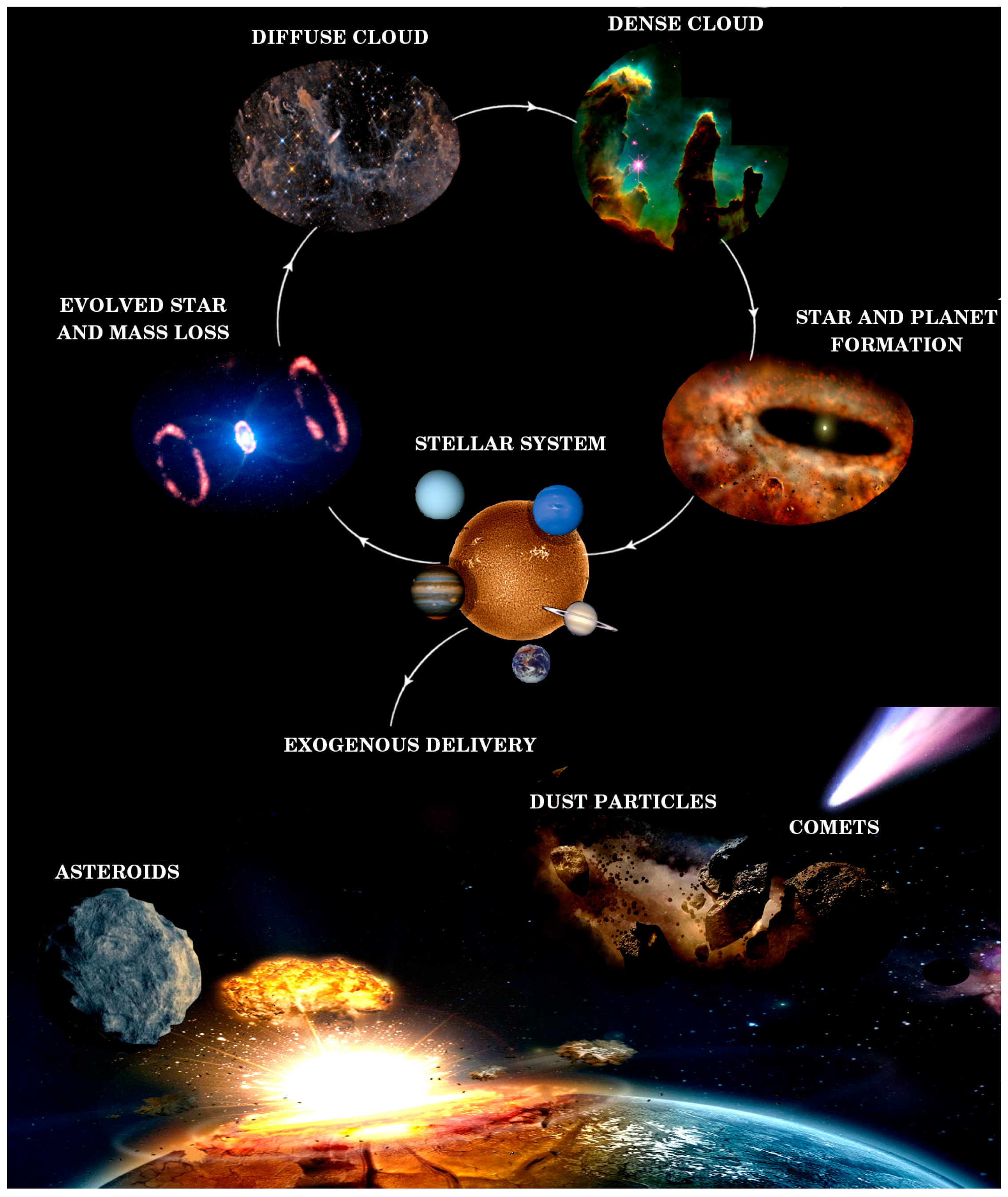
1. Introduction
2. Materials and Methods
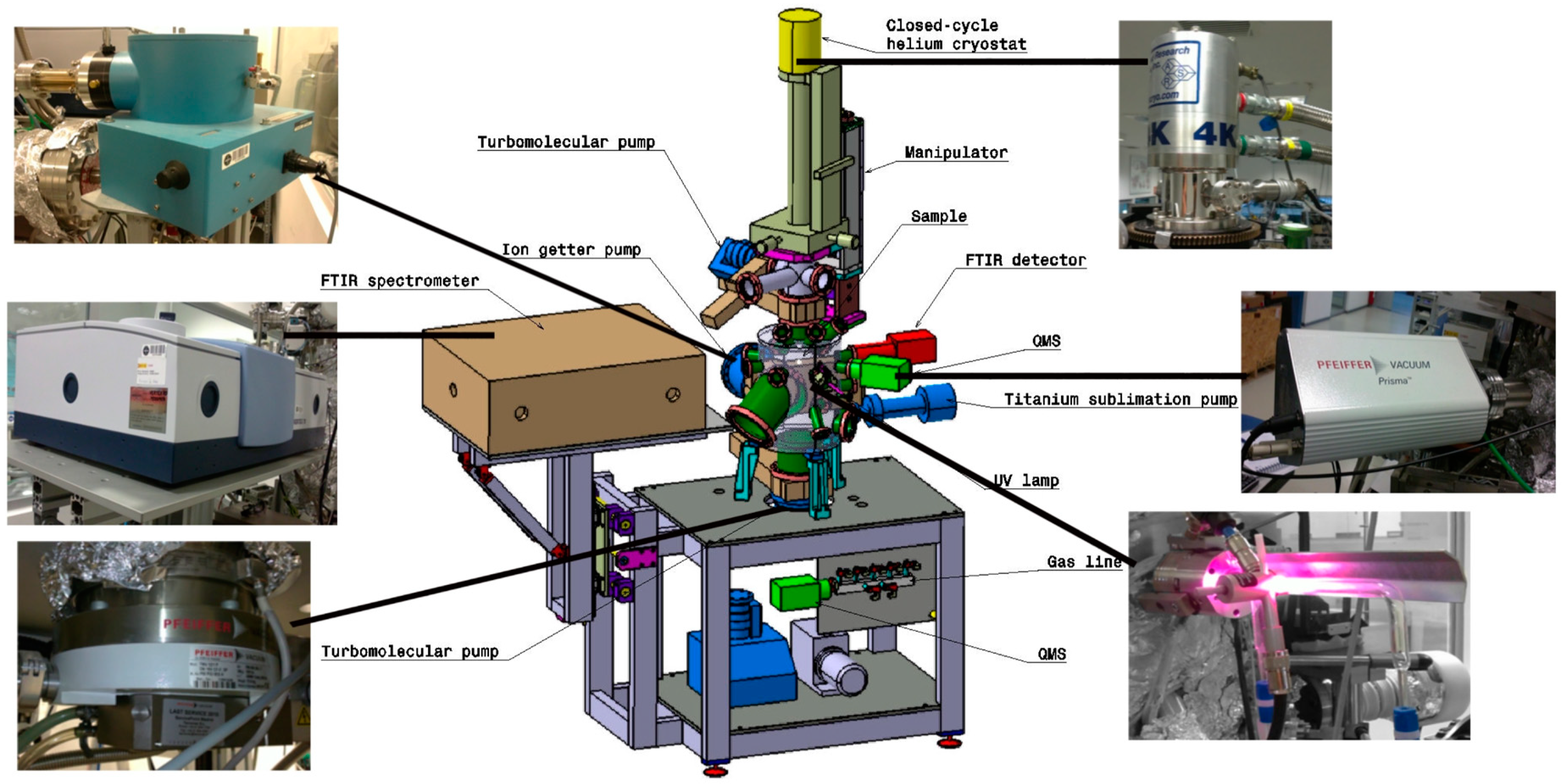
2.1. ISAC
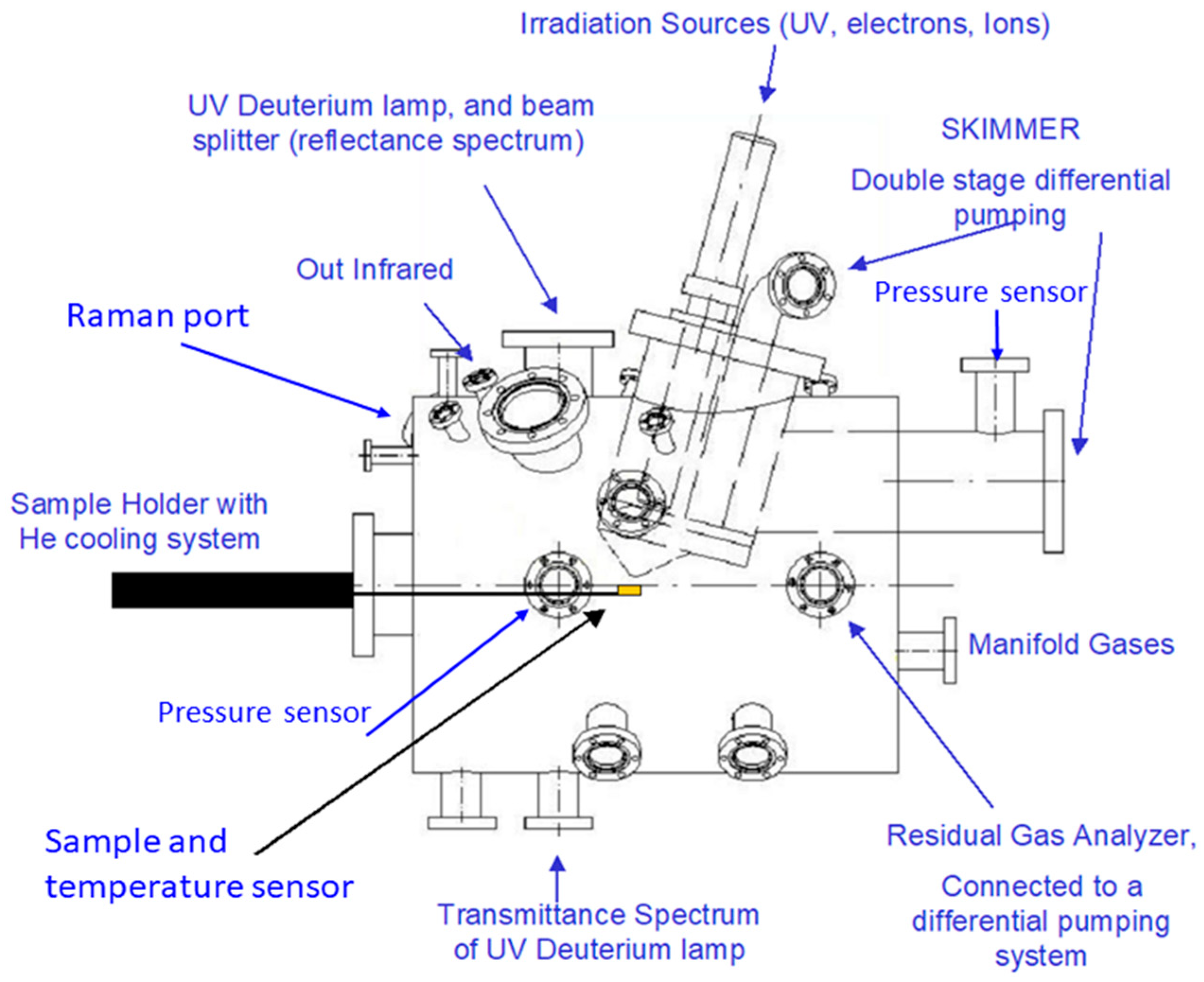
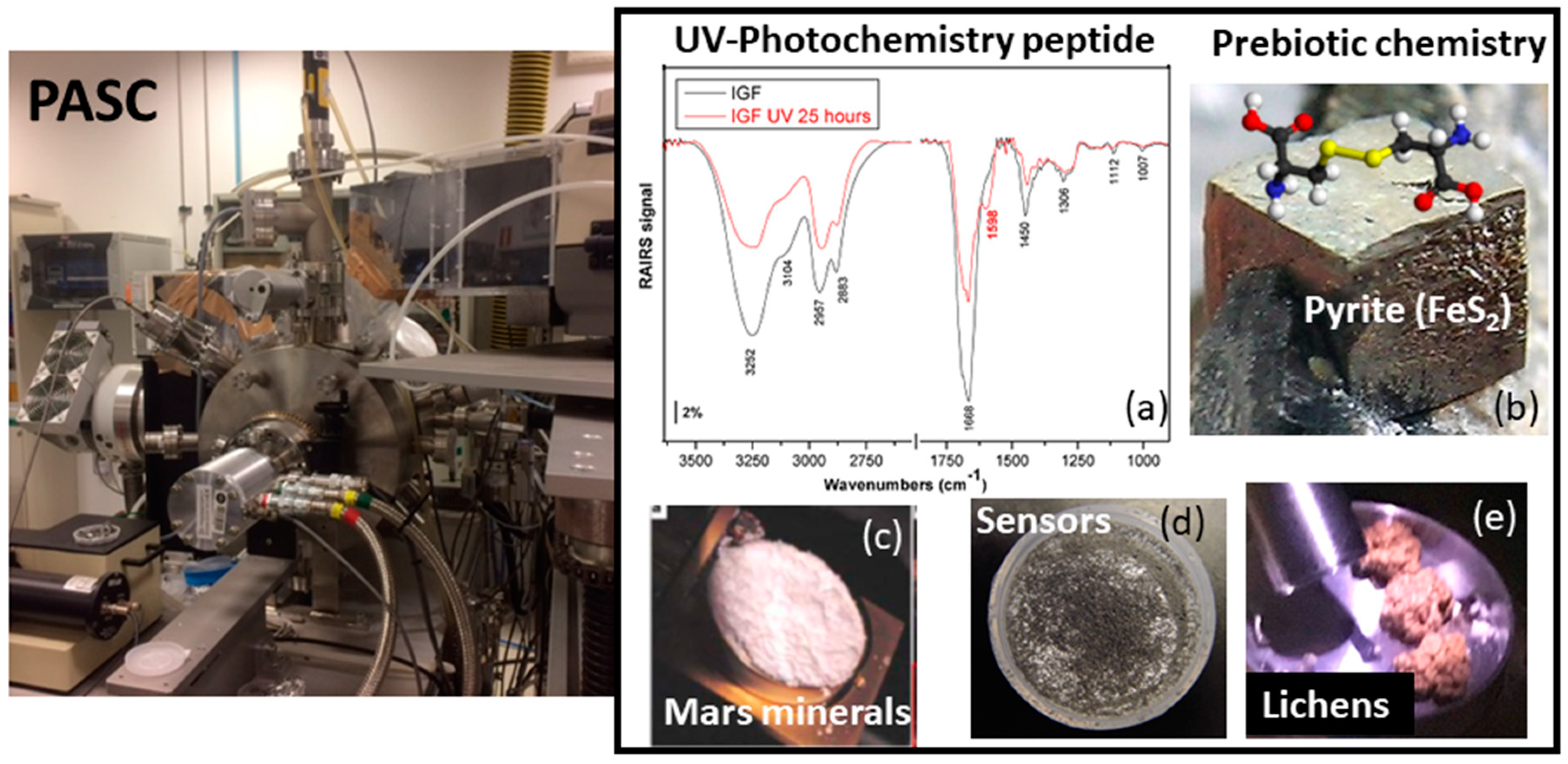
2.2. PASC
2.3. HPPECs
3. Review of Experimental Results
3.1. Interstellar Medium
3.2. Planetary Surfaces
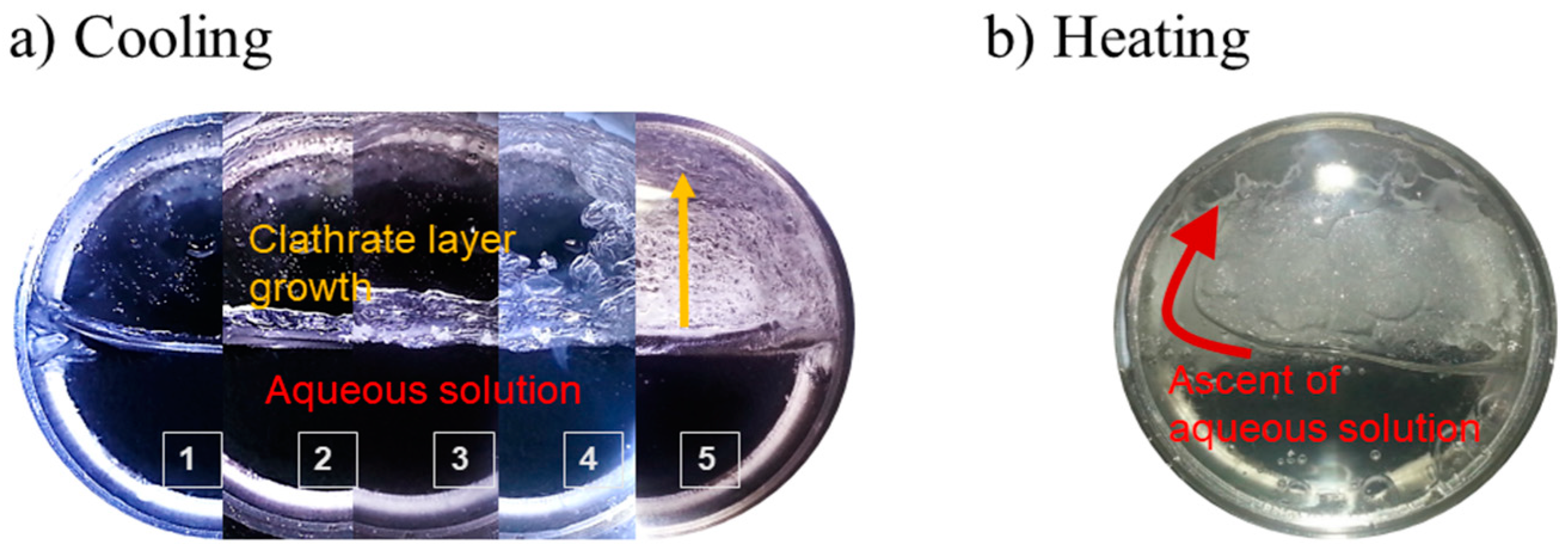
3.3. Deep Environments
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Öberg, K.I. Photochemistry and astrochemistry: Photochemical pathways to interstellar complex organic molecules. Chem. Rev. 2016, 116, 9631–9663. [Google Scholar] [CrossRef] [PubMed]
- Grotzinger, J.P.; Crisp, J.; Vasavada, A.R.; Anderson, R.C.; Baker, C.J.; Barry, R.; Blake, D.F.; Conrad, P.; Edgett, K.S.; Ferdowski, B.; et al. Mars science laboratory mission and science investigation. Space Sci. Rev. 2012, 170, 5–56. [Google Scholar] [CrossRef]
- Vago, J.L.; Westall, F.; Coates, A.J.; Jaumann, R.; Korablev, O.; Ciarletti, V.; Mitrofanov, I.; Josset, J.L.; De Sanctis, M.C.; Bibring, J.-P.; et al. Habitability on early mars and the search for biosignatures with the exomars rover. Astrobiology 2017, 17, 471–510. [Google Scholar] [CrossRef] [PubMed]
- Grasset, O.; Dougherty, M.K.; Coustenis, A.; Bunce, E.J.; Erd, C.; Titov, D.; Blanc, M.; Coates, A.; Drossart, P.; Fletcher, L.N.; et al. Jupiter icy moons explorer (JUICE): An ESA mission to orbit ganymede and to characterise the jupiter system. Planet Space Sci. 2013, 78, 1–21. [Google Scholar] [CrossRef]
- Phillips, C.B.; Pappalardo, R.T. Europa clipper mission concept: Exploring jupiter’s ocean moon. Eos Trans. Agu. 2014, 95, 165–167. [Google Scholar] [CrossRef]
- Mateo-Marti, E. Planetary atmosphere and surfaces chamber (PASC): A platform to address various challenges in astrobiology. Challenges 2014, 5, 213–223. [Google Scholar] [CrossRef]
- Jensen, L.L.; Merrison, J.; Hansen, A.A.; Mikkelsen, K.A.; Kristoffersen, T.; Nørnberg, P.; Lomstein, B.A.; Finster, K. A Facility for long-term mars simulation experiments: The mars environmental simulation chamber (MESCH). Astrobiology 2008, 8, 537–548. [Google Scholar] [CrossRef] [PubMed]
- Sears, D.W.G.; Benoit, P.H.; McKeever, S.W.S.; Banerjee, D.; Kral, T.; Stites, W.; Roe, L.; Jansma, P.; Mattioli, G. Investigation of biological chemical and physical processes on and in planetary surfaces by laboratory simulation. Planet Space Sci. 2002, 50, 821–828. [Google Scholar] [CrossRef]
- Martin, D.; Cockell, C.S. PELS (Planetary Environmental Liquid Simulator): A new type of simulation facility to study extraterrestrial aqueous environments. Astrobiology 2015, 15, 111–118. [Google Scholar] [CrossRef] [PubMed]
- Sobron, P.; Wang, A. A planetary environment and analysis chamber (PEACh) for coordinated Raman-LIBS-IR measurements under planetary surface environmental conditions. J. Raman Spectrosc. 2012, 43, 212–227. [Google Scholar] [CrossRef]
- Cruz-Diaz, G.A.; Muñoz Caro, G.M.; Chen, Y.J.; Yih, T.S. Vacuum-UV spectroscopy of interstellar ice analogs: I. Absorption cross-sections of polar-ice molecules. Astron. Astrophys. 2014, 562, A119. [Google Scholar] [CrossRef]
- Cruz-Diaz, G.A.; Muñoz Caro, G.M.; Chen, Y.J.; Yih, T.S. Vacuum-UV spectroscopy of interstellar ice analogs: II. Absorption cross-sections of nonpolar ice molecules. Astron. Astrophys. 2014, 562, A120. [Google Scholar] [CrossRef]
- Cruz-Diaz, G.A.; Caro, G.M.M.; Chen, Y.J. Vacuum-UV absorption spectroscopy of interstellar ice analogues. III. Isotopic effects. Mon. Not. the R. Astron. Soc. 2014, 439, 2370–2376. [Google Scholar] [CrossRef]
- Chen, Y.J.; Juang, K.J.; Nuevo, M.; Jiménez-Escobar, A.; Muñoz Caro, G.M.; Qiu, J.M.; Chu, C.C.; Yih, T.S.; Wu, C.Y.R.; Fung, H.S.; et al. Formation of S-bearing species by VUV/EUV irradiation of H2S-containing ice mixtures: Photon energy and carbon source effects. Astrophys. J. 2014, 798, 80. [Google Scholar] [CrossRef]
- Gónzalez Díaz, C.; Carrascosa de Lucas, H.; Aparicio, S.; Muñoz Caro, G.M.; Sie, N.E.; Hsiao, L.C.; Cazaux, S.; Chen, Y.J. Accretion and photodesorption of CO ice as a function of the incident angle of deposition. Mon. Not. R. Astron. Soc. 2019, 486, 5519–5525. [Google Scholar] [CrossRef]
- Muñoz Caro, G.M.; Jiménez-Escobar, A.; Martín-Gago, J.A.; Rogero, C.; Atienza, C.; Puertas, S.; Sobrado, J.M.; Torres-Redondo, J. New results on thermal and photodesorption of CO ice using the novel inter stellar astrochemistry chamber (ISAC). Astron. Astrophys. 2010, 522, A108–A121. [Google Scholar] [CrossRef]
- Mateo-Martí, E.; Prieto-Ballesteros, O.; Sobrado, J.M.; Gómez-Elvira, J.; Martín-Gago, J.A. A chamber for studying planetary environments and its applications to astrobiology. Meas. Sci. Technol. 2006, 17, 2274–2280. [Google Scholar] [CrossRef]
- Boogert, A.C.A.; Gerakines, P.A.; Whittet, D.C.B. Observations of the icy universe. Annu. Rev. Astron. Astrophys. 2015, 53, 541–581. [Google Scholar] [CrossRef]
- Dartois, E.; Marco, O.; Muñoz Caro, G.M.; Brooks, K.; Deboffle, D.; d’Hendecourt, L. Organic matter in seyfert 2 nuclei: Comparison with our galactic center lines of sight. Astron. Astrophys. 2004, 423, 549–558. [Google Scholar] [CrossRef]
- Dartois, E.; Muñoz Caro, G.M.; Deboffle, D.; d’Hendecourt, L. Diffuse interstellar medium organic polymers. photoproduction of the 3.4 6.85 and 7.25 μm features. Astron. Astrophys. 2004, 423, L33–L36. [Google Scholar] [CrossRef]
- Dartois, E.; Muñoz Caro, G.M.; Deboffle, D.; Montagnac, G.; d’Hendecourt, L. Ultraviolet photoproduction of ISM dust-laboratory characterisation and astrophysical relevance. Astron. Astrophys. 2005, 432, 895–908. [Google Scholar] [CrossRef]
- Matrajt, G.; Muñoz Caro, G.M.; Dartois, E.; D’Hendecourt, L.; Deboffle, D.; Borg, J. FTIR analysis of the organics in IDPs: Comparison with the IR spectra of the diffuse interstellar medium. Astron. Astrophys. 2005, 433, 979–995. [Google Scholar] [CrossRef]
- Muñoz Caro, G.M.; Matrajt, G.; Dartois, E.; Nuevo, M.; D’Hendecourt, L.; Deboffle, D.; Montagnac, G.; Chauvin, N.; Boukari, C.; Le Du, D. Nature and evolution of the dominant carbonaceous matter in interplanetary dust particles: Effects of irradiation and identification with a type of amorphous carbon. Astron. Astrophys. 2006, 459, 147–159. [Google Scholar] [CrossRef]
- Dartois, E.; Muñoz Caro, G.M. Carbonaceous dust grains in luminous infrared galaxies. Spitzer/IRS reveals a-C:H as an abundant and ubiquitous ISM component. Astron. Astrophys. 2007, 476, 1235–1242. [Google Scholar] [CrossRef]
- Muñoz Caro, G.M.; Dartois, E.; Nakamura-Messenger, K. Characterization of the carbon component in cometary stardust samples by means of infrared and Raman spectroscopy. Astron. Astrophys. 2008, 485, 743–751. [Google Scholar] [CrossRef]
- Godard, M.; Geballe, T.R.; Dartois, E.; Muñoz Caro, G.M. The deep 3.4 μm interstellar absorption feature toward the IRAS 18511+0146 cluster. Astron. Astrophys. 2012, 537, A27. [Google Scholar] [CrossRef]
- Muñoz Caro, G.M.; Rietmeijer, F.J.M.; Souza-Egipsy, V.; Valles-González, M.P. A potentially new type of nonchondritic interplanetary dust particle with hematite organic carbon amorphous Na, Ca-aluminosilicate and FeO-spheres. Meteorit. Planet. Sci. 2012, 47, 248–261. [Google Scholar] [CrossRef]
- Alata, I.; Cruz-Diaz, G.A.; Muñoz Caro, G.M.; Dartois, E. Vacuum ultraviolet photolysis of hydrogenated amorphous carbons: I. Interstellar H2 and CH4 formation rates. Astron. Astrophys. 2014, 569, A119. [Google Scholar] [CrossRef]
- Alata, I.; Jallat, A.; Gavilan, L.; Chabot, M.; Cruz-Diaz, G.A.; Muñoz Caro, G.M.; Béroff, K.; Dartois, E. Vacuum ultraviolet of hydrogenated amorphous carbons: II. Small hydrocarbons production in photon dominated regions. Astron. Astrophys. 2015, 584, A123. [Google Scholar] [CrossRef]
- Martín-Doménech, R.; Dartois, E.; Muñoz Caro, G.M. Vacuum ultraviolet photolysis of hydrogenated amorphous carbons: III. Diffusion of photo-produced H2 as a function of temperature. Astron. Astrophys. 2016, 591, A107. [Google Scholar] [CrossRef]
- Dartois, E.; Jallat, A.; Alata, I.; Gavilan, L.; Cruz-Diaz, G.A.; Chabot, M.; Beroff, K.; Muñoz Caro, G.M. UV photolysis of hydrogenated amorphous carbons of astrophysical interest. Polycycl. Aromat. Compd. 2017, 37, 94–100. [Google Scholar] [CrossRef]
- Dartois, E.; Augé, B.; Boduch, P.; Brunetto, R.; Chabot, M.; Domaracka, A.; Ding, J.J.; Kamalou, O.; Lv, X.Y.; Rothard, H.; et al. Heavy ion irradiation of crystalline water ice: Cosmic ray amorphisation cross-section and sputtering yield. Astron. Astrophys. 2015, 576, A125. [Google Scholar] [CrossRef]
- Martín-Doménech, R.; Manzano-Santamaría, J.; Muñoz Caro, G.M.; Cruz-Díaz, G.A.; Chen, Y.-J.; Herrero, V.J.; Tanarro, I. UV photoprocessing of CO2 ice: A complete quantification of photochemistry and photon-induced desorption processes. Astron. Astrophys. 2015, 584, A14. [Google Scholar] [CrossRef]
- Chen, Y.J.; Muñoz Caro, G.M.; Aparicio, S.; Jiménez-Escobar, A.; Lasne, J.; Rosu-Finsen, A.; McCoustra, M.R.S.; Cassidy, A.M.; Field, D. Wannier-Mott excitons in nanoscale molecular ices. Phys. Rev. Lett. 2017, 119, 157703. [Google Scholar] [CrossRef] [PubMed]
- Muñoz Caro, G.M.; Chen, Y.J.; Aparicio, S.; Jiménez-Escobar, A.; Rosu-Finsen, A.; Lasne, J.; McCoustra, M.R.S. Photodesorption and physical properties of CO ice as a function of temperature. Astron. Astrophys. 2016, 589, A19. [Google Scholar] [CrossRef]
- Martín-Doménech, R.; Muñoz Caro, G.M.; Cruz-Díaz, G.A. Study of the photon-induced formation and subsequent desorption of CO3OH and H2CO in interstellar ice analogs. Astron. Astrophys. 2016, 589, A107. [Google Scholar] [CrossRef]
- Cruz-Diaz, G.A.; Martín-Doménech, R.; Muñoz Caro, G.M.; Chen, Y.-J. Negligible photodesorption of methanol ice and active photon-induced desorption of its irradiation products. Astron. Astrophys. 2016, 592, A68. [Google Scholar] [CrossRef]
- Martín-Doménech, R.; Cruz-Díaz, G.A.; Muñoz Caro, G.M. UV photoprocessing of NH3 ice: Photon-induced desorption mechanisms. Mon. Not. R. Astron. Soc. 2018, 473, 2575–2582. [Google Scholar] [CrossRef]
- Cruz-Diaz, G.A.; Martín-Doménech, R.; Moreno, E.; Muñoz Caro, G.M.; Chen, Y.J. New measurements on water ice photodesorption and product formation under ultraviolet irradiation. Mon. Not. R. Astron. Soc. 2018, 474, 3080–3089. [Google Scholar] [CrossRef]
- Jiménez-Escobar, A.; Ciaravella, A.; Cecchi-Pestellini, C.; Huang, C.H.; Sie, N.E.; Chen, Y.J.; Muñoz Caro, G.M. X-Ray photo-desorption of H2O:CO:NH3 circumstellar ice analogs: Gas-phase enrichment. Astrophys. J. 2018, 868, 73. [Google Scholar] [CrossRef]
- Sie, N.E.; Caro, G.M.M.; Huang, Z.H.; Martín-Doménech, R.; Fuente, A.; Chen, Y.J. On the photodesorption of CO2 ice analogs: The formation of atomic C in the ice and the effect of the VUV emission spectrum. Astrophys. J. 2019, 874, 35. [Google Scholar] [CrossRef]
- Carrascosa, H.; Hsiao, L.C.; Sie, N.E.; Muñoz Caro, G.M.; Chen, Y.J. 13CO and 13CO2 ice mixtures with N2 in photon energy transfer studies. Mon. Not. R. Astron. Soc. 2019, 486, 1985–1994. [Google Scholar] [CrossRef]
- Jiménez-Escobar, A.; Muñoz Caro, G.M. Sulfur depletion in dense clouds and circumstellar regions: I. H2S ice abundance and UV-photochemical reactions in the H2O-matrix. Astron. Astrophys. 2011, 536, A91. [Google Scholar] [CrossRef]
- Jiménez-Escobar, A.; Muñoz Caro, G.M.; Ciaravella, A.; Cecchi-Pestellini, C.; Candia, R.; Micela, G. Soft X-Ray irradiation of H2S ice and the presence of S2 in comets. Astrophys. J. 2012, 751, L40–L44. [Google Scholar] [CrossRef]
- Jiménez-Escobar, A.; Muñoz Caro, G.M.; Chen, Y.J. Sulphur depletion in dense clouds and circumstellar regions. organic products made from UV photoprocessing of realistic ice analogs containing H2S. Mon. Not. R. Astron. Soc. 2014, 443, 343–354. [Google Scholar] [CrossRef]
- Martín-Doménech, R.; Jiménez-Serra, I.; Muñoz Caro, G.M.; Müller, H.S.P.; Occhiogrosso, A.; Testi, L.; Woods, P.M.; Viti, S. The sulfur depletion problem: Upper limits on the H2S2, HS2, and S2 gas-phase abundances toward the low-mass warm core IRAS 16293-2422. Astron. Astrophys. 2016, 585, A112. [Google Scholar] [CrossRef]
- Fuente, A.; Goicoechea, J.R.; Pety, J.; Le Gal, R.; Martín-Doménech, R.; Gratier, P.; Guzmán, V.; Roueff, E.; Loison, J.C.; Muñoz Caro, G.M.; et al. First detection of interstellar S2H. Astrophys. J. 2017, 851, L49. [Google Scholar] [CrossRef] [PubMed]
- Ciaravella, A.; Muñoz Caro, G.M.; Jimenez Escobar, A.; Cecchi-Pestellini, C.; Giarrusso, S.; Barbera, M.; Collura, A. Soft X-ray irradiation of a methanol ice. Astrophys. J. Lett. 2010, 722, L45–L48. [Google Scholar] [CrossRef]
- Ciaravella, A.; Jiménez-Escobar, A.; Muñoz Caro, G.M.; Cecchi-Pestellini, C.; Candia, R.; Giarrusso, S.; Barbera, M.; Collura, A. Soft X-ray irradiation of the pure carbon monoxide interstellar ice analogues. Astrophys. J. 2012, 746, L1–L5. [Google Scholar] [CrossRef]
- Ciaravella, A.; Cecchi-Pestellini, C.; Chen, Y.J.; Muñoz Caro, G.M.; Huang, C.H.; Jiménez-Escobar, A.; Venezia, A.M. Soft X-ray irradiation of silicates: implications on dust evolution in protoplanetary disks. Astrophys. J. 2016, 828, 29. [Google Scholar] [CrossRef]
- Ciaravella, A.; Chen, Y.J.; Cecchi-Pestellini, C.; Jiménez-Escobar, A.; Muñoz Caro, G.M.; Chuang, K.J.; Huang, C.H. Chemical evolution of a CO ice induced by soft X-rays. Astrophys. J. 2016, 819, 38. [Google Scholar] [CrossRef]
- Ciaravella, A.; Jimenez-Escobar, A.; Cecchi-Pestellini, C.; Huang, C.H.; Sie, N.E.; Muñoz-Caro, G.M.; Chen, Y.J. Synthesis of prebiotic molecules in soft X-ray irradiated ices. Astrophys. J. 2019, 879, 21. [Google Scholar] [CrossRef]
- Chen, Y.J.; Chuang, K.J.; Muñoz Caro, G.M.; Nuevo, M.; Chu, C.C.; Yih, T.S.; Ip, W.H.; Wu, C.Y.R. Vacuum ultraviolet emission spectrum measurement of a microwave-discharge hydrogen-flow lamp in several configurations: Application to photodesorption of CO ice. Astrophys. J. 2013, 781, 15. [Google Scholar] [CrossRef]
- Chen, Y.J.; Ciaravella, A.; Muñoz Caro, G.M.; Cecchi-Pestellini, C.; Jiménez-Escobar, A.; Juang, K.J.; Yih, T.S. soft X-Ray irradiation of methanol ice: formation of products as a function of photon energy. Astrophys. J. 2013, 778, 162. [Google Scholar] [CrossRef]
- Muñoz Caro, G.M.; Ciaravella, A.; Jiménez-Escobar, A.; Cecchi-Pestellini, C.; Chen, Y.J. X-ray versus UV irradiation of astrophysical ice analogs leading to COMs formation. ACS Earth Space Chem. 2019. [Google Scholar] [CrossRef]
- Muñoz Caro, G.M.; Dartois, E.; Boduch, P.; Rothard, H.; Domaracka, A.; Jiménez-Escobar, A. Comparison of UV and high-energy ion irradiation of methanol: Ammonia ice. Astron. Astrophys. 2014, 566, A93. [Google Scholar] [CrossRef]
- Jiménez-Escobar, A.; Giuliano, B.M.; Caro, G.M.M.; Cernicharo, J.; Marcelino, N. Investigation of HNCO isomers formation in ice mantles by UV and thermal processing: An experimental approach. Astrophys. J. 2014, 788, 19–26. [Google Scholar] [CrossRef]
- Escribano, R.M.; Muñoz Caro, G.M.; Cruz-Diaz, G.A.; Rodriguez-Lazcano, Y.; Mate, B. Crystallization of CO2 ice and the absence of amorphous CO2 ice in space. Proc. Natl. Acad. Sci. USA 2013, 110, 12899–12904. [Google Scholar] [CrossRef] [PubMed]
- Giuliano, B.M.; Escribano, R.M.; Martín-Doménech, R.; Dartois, E.; Muñoz Caro, G.M. Interstellar ice analogs: band strengths of H2O CO2 CH3OH and NH3 in the far-infrared region. Astron. Astrophys. 2014, 565, A108. [Google Scholar] [CrossRef]
- Giuliano, B.M.; Martín-Doménech, R.; Escribano, R.M.; Manzano-Santamaría, J.; Muñoz Caro, G.M. Interstellar ice analogs: H2O ice mixtures with CH3OH and NH3 in the far-IR region. Astron. Astrophys. 2016, 592, A81. [Google Scholar] [CrossRef][Green Version]
- Muñoz Caro, G.M.; Dartois, E. A tracer of organic matter of prebiotic interest in space, made from UV and thermal processing of ice mantles. Astron. Astrophys. 2009, 494, 109–115. [Google Scholar] [CrossRef]
- Martín-Doménech, R.; Muñoz Caro, G.M.; Bueno, J.; Goesmann, F. Thermal desorption of circumstellar and cometary ice analogs. Astron. Astrophys. 2014, 564, A8. [Google Scholar] [CrossRef]
- Cazaux, S.; Martín-Doménech, R.; Chen, Y.J.; Caro, G.M.M.; Díaz, C.G. CO Depletion: A microscopic perspective. Astrophys. J. 2017, 849, 80. [Google Scholar] [CrossRef]
- Goesmann, F.; McKenna-Lawlor, S.; Roll, R.; Bredehöft, J.H.; Meierhenrich, U.; Raulin, F.; Thiemann, W.; Muñoz Caro, G.M.; Szopa, C. Interpretation of COSAC mass spectrometer data acquired during Rosetta’s lutetia fly-by 10 July 2010. Planet. Space Sci. 2012, 66, 187–191. [Google Scholar] [CrossRef]
- Goesmann, F.; Raulin, F.; Bredehöft, J.H.; Cabane, M.; Ehrenfreund, P.; MacDermott, A.J.; McKenna-Lawlor, S.; Meierhenrich, U.J.; Muñoz Caro, G.M.; Szopa, C.; et al. COSAC prepares for sampling and in situ analysis of cometary matter from comet 67P/Churyumov–Gerasimenko. Planet. Space Sci. 2014, 103, 318–330. [Google Scholar] [CrossRef]
- Goesmann, F.; Rosenbauer, H.; Bredehoft, J.H.; Cabane, M.; Ehrenfreund, P.; Gautier, T.; Giri, C.; Kruger, H.; Le Roy, L.; MacDermott, A.J.; et al. Organic compounds on comet 67P/Churyumov-Gerasimenko revealed by COSAC mass spectrometry. Science 2015, 349, 6247. [Google Scholar] [CrossRef] [PubMed]
- Muñoz Caro, G.M.; Dartois, E. Prebiotic chemistry in icy grain mantles in space: An experimental and observational approach. Chem. Soc. Rev. 2013, 42, 2173–2185. [Google Scholar] [CrossRef] [PubMed]
- Muñoz Caro, G.M.; Escribano, R. (Eds.) Laboratory Astrophysics: Astrophysics and Space Science Library; Springer: Basel, Switzerland, 2018; Volume 451. [Google Scholar]
- Muñoz Caro, G.; Mateo-Martí, E.; Martínez-Frías, J. Near-UV transmittance of basalt dust as an analog of the martian regolith: Implications for sensor calibration and astrobiology. Sensors 2006, 6, 688–696. [Google Scholar] [CrossRef]
- Zorzano, M.P.; Mateo-Martí, E.; Prieto-Ballesteros, O.; Osuna, S.; Renno, N. Stability of liquid saline water on present day Mars. Geophys. Res. Lett. 2009, 36. [Google Scholar] [CrossRef]
- Sanchez-Arenillas, M.; Mateo-Marti, E. Spectroscopic study of cystine adsorption on pyrite surface: From vacuum to solution conditions. Chem. Phys. 2015, 458, 92–98. [Google Scholar] [CrossRef]
- Sanchez-Arenillas, M.; Mateo-Marti, E. Pyrite surface environment drives molecular adsorption: Cystine on pyrite (100) investigated by X-ray photoemission spectroscopy and low energy electron diffraction. Phys. Chem. Chem. Phys. 2016, 18, 27219–27225. [Google Scholar] [CrossRef] [PubMed]
- Sanchez-Arenillas, M.; Galvez-Martinez, S.; Mateo-Marti, E. Sulfur amino acids and alanine on pyrite (100) by X-ray photoemission spectroscopy: Surface or molecular role? Appl. Surf. Sci. 2017, 414, 303–312. [Google Scholar] [CrossRef]
- Mateo-Marti, E.; Pradier, C.M. UV irradiation study of a tripeptide isolated in an argon matrix: A tautomerism process evidenced by infrared and X-ray photoemission spectroscopies. Spectrochim. Acta Part. A Mol. Biomol. Spectrosc. 2013, 109, 247–252. [Google Scholar] [CrossRef] [PubMed]
- Galvez-Martinez, S.; Mateo-Marti, E. Ultraviolet irradiation on a pyrite surface improves triglycine adsorption. Life 2018, 8, 50. [Google Scholar] [CrossRef] [PubMed]
- Mateo-Martí, E.; Pradier, C.M.; Martín-Gago, J.A. Ultraviolet photostability of adenine on gold and silicon surfaces. Astrobiology 2009, 9, 573–579. [Google Scholar] [CrossRef] [PubMed]
- Lopez-Reyes, G.; Pilorget, C.; Moral, A.G.; Manrique, J.A.; Sanz, A.; Berrocal, A.; Veneranda, M.; Rull, F.; Medina, J.; Hamm, V.; et al. RLS calibration target design to allow onboard combined science between RLS and MicrOmega instruments on the ExoMars rover. J. Raman Spectrosc. 2019, in press. [Google Scholar]
- Gil-Lozano, C.; Mateo-Martí, E.; Gago-Duport, L.; Losa-Adams, E.; Fernández Sampedro, M.; Chevrier, V.; Bishop, J.L.; Fairén, A.G. Sulfide mineral paragenesis triggered oxidations reactions in a cold and wet early mars. EPSL 2018. under review. [Google Scholar]
- Gil-Lozano, C.; Mateo-Martí, E.; Gago-Duport, L.; Losa-Adams, E.; Chevrier, V.; Fairen, A.G. Exploring the mineral sequences that can be formed from a disulfide-rich soil on early mars. In Proceedings of the 48th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 20–24 March 2017. [Google Scholar]
- Fairén, A.G.; Mateo-Martí, E.; Robas, C.; Muñoz-Iglesias, V.; Carrizo, D.; Fernández Sampedro, M.; Prieto-Ballesteros, O.; Gago-Duport, L.; Losa-Adams, E.; Gil-Lozano, C. Preservation of organic matter on mars: The role of the mineral matrix. In Proceedings of the Agu Fall Meeting, Washington, DC, USA, 10–14 December 2018. [Google Scholar]
- Gómez, F.; Mateo-Martí, E.; Prieto-Ballesteros, O.; Martín-Gago, J.; Amils, R. Protection of chemolithoautotrophic bacteria exposed to simulated Mars environmental conditions. Icarus 2010, 209, 482–487. [Google Scholar] [CrossRef]
- Sánchez, F.J.; Mateo-Martí, E.; Raggio, J.; Meeßen, J.; Martínez-Frías, J.; Sancho, L.G.; Ott, S.; de la Torre, R. The resistance of the lichen circinaria gyrosa (nom. provis.) towards simulated Mars conditions—a model test for the survival capacity of a eukaryotic extremophile. Planet. Space Sci. 2012, 72, 102–110. [Google Scholar] [CrossRef]
- Gomez Gomez, J.M.; Estebanez, B. Survival of moss reproductive structures under simulated martian environmental conditions and extreme thermal stress: Vibrational spectroscopic study and astrobiological implications. J. Astrobiol. Outreach 2016, 4, 2. [Google Scholar] [CrossRef]
- Prieto-Ballesteros, O.; Vorobyova, E.; Parro, V.; Rodriguez Manfredi, J.A.; Gómez, F. Strategies for detection of putative life on Europa. Adv. Space Res. 2011, 48, 678–688. [Google Scholar] [CrossRef]
- Hand, K.P.; Murray, A.E.; Garvin, J.B.; Brinckerhoff, W.B.; Christner, B.C.; Edgett, K.S.; Ehlmann, B.L.; German, C.R.; Hayes, A.G.; Hoehler, T.M.; et al. Report of the Europa Lander Science Definition Team 2017; National Aeronautics and Space Administration: Washington, DC, USA, 2017.
- Prieto Ballesteros, O.; Muñoz-Iglesias, V.; Bonales, L.J. Interiors of icy moons from astrobiology perspective. In An Introduction to High Pressure Science and Technology; Recio, J.M., Menendez, J.M., Otero de la Roza, A., Eds.; CRC Press: Boca Raton, FL, USA, 2014; pp. 459–488. [Google Scholar]
- Muñoz-Iglesias, V.; Bonales, L.J.; Prieto-Ballesteros, O. pH and salinity evolution of europa’s brines: Raman spectroscopy study of fractional precipitation at 1 and 300 bar. Astrobiology 2013, 13, 693–702. [Google Scholar] [CrossRef] [PubMed]
- Bonales, L.J.; Muñoz-Iglesias, V.; Prieto-Ballesteros, O. Raman spectroscopy as a tool to study the solubility of CO2 in magnesium sulphate brines: Application to the fluids of Europa’s cryomagmatic reservoirs. Eur. J. Mineral. 2014, 25, 735–743. [Google Scholar] [CrossRef]
- Muñoz-Iglesias, V.; Prieto-Ballesteros, O.; Bonales, L.J. Conspicuous assemblages of hydrated minerals from the H2O–MgSO4–CO2 system on Jupiter’s Europa satellite. Geochim. Cosmochim. Acta 2014, 125, 466–475. [Google Scholar] [CrossRef]
- Méndez, A.S.J.; Muñoz-Iglesias, V.; Izquierdo-Ruíz, F.; Prieto-Ballesteros, O. Salting-out phenomenon induced by the clathrate hydrates formation at high-pressure. J. Phys. Conf. Ser. 2017, 950, 42. [Google Scholar] [CrossRef]
- Izquierdo-Ruiz, F.; Otero-de-la-Roza, A.; Contreras-García, J.; Menéndez, J.M.; Prieto-Ballesteros, O.; Recio, J.M. Guest–host interactions in gas clathrate hydrates under pressure. High. Press. Res. 2015, 35, 49–56. [Google Scholar] [CrossRef]
- Izquierdo-Ruiz, F.; Otero-de-la-Roza, A.; Contreras-García, J.; Prieto-Ballesteros, O.; Recio, J. Effects of the CO2 guest molecule on the Si clathrate hydrate structure. Materials 2016, 9, 777. [Google Scholar] [CrossRef] [PubMed]
- McCord, T.B.; Hansen, G.B.; Fanale, F.P.; Carlson, R.W.; Matson, D.L.; Johnson, T.V.; Smythe, W.D.; Crowley, J.K.; Martin, P.D.; Ocampo, A.; et al. Salts on Europa’s surface detected by Galileo’s near infrared mapping spectrometer. Science 1998, 280, 1242–1245. [Google Scholar] [CrossRef] [PubMed]
- McCord, T.B.; Hansen, G.B.; Matson, D.L.; Johnson, T.V.; Crowley, J.K.; Fanale, F.P.; Carlson, R.W.; Smythe, W.D.; Martin, P.D.; Hibbitts, C.A.; et al. Hydrated salt minerals on Europa’s surface from the Galileo near-infrared mapping spectrometer (NIMS) investigation. J. Geophys. Res. Planets 1999, 104, 11827–11851. [Google Scholar] [CrossRef]
- Roth, L.; Saur, J.; Retherford, K.D.; Strobel, D.F.; Feldman, P.D.; McGrath, M.A.; Nimmo, F. Transient water vapor at Europa’s south pole. Science 2014, 343, 171–174. [Google Scholar] [CrossRef] [PubMed]
- Geissler, P. Cryovolcanism in the Outher Solar system. In The Encyclopedia of Volcanoes, 2nd ed.; Academic Press: Cambridge, MA, USA, 2015; pp. 763–776. [Google Scholar]
- Jia, X.; Kivelson, M.G.; Khurana, K.K.; Kurth, W.S. Evidence of a plume on Europa from Galileo magnetic and plasma wave signatures. Nat. Astron. 2018, 2, 459–464. [Google Scholar] [CrossRef]
- Méndez, A.S.J.; Izquierdo-Ruíz, F.; Prieto-Ballesteros, O. Textural evolution of clathrate-salt hydrates-water ice assemblage and the induced geological activity in icy moons. In Proceedings of the 47th Lunar and Planetary Science Conference, The Woodlands, TX, USA, 21–25 March 2016. [Google Scholar]


© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mateo-Marti, E.; Prieto-Ballesteros, O.; Muñoz Caro, G.; González-Díaz, C.; Muñoz-Iglesias, V.; Gálvez-Martínez, S. Characterizing Interstellar Medium, Planetary Surface and Deep Environments by Spectroscopic Techniques Using Unique Simulation Chambers at Centro de Astrobiologia (CAB). Life 2019, 9, 72. https://doi.org/10.3390/life9030072
Mateo-Marti E, Prieto-Ballesteros O, Muñoz Caro G, González-Díaz C, Muñoz-Iglesias V, Gálvez-Martínez S. Characterizing Interstellar Medium, Planetary Surface and Deep Environments by Spectroscopic Techniques Using Unique Simulation Chambers at Centro de Astrobiologia (CAB). Life. 2019; 9(3):72. https://doi.org/10.3390/life9030072
Chicago/Turabian StyleMateo-Marti, Eva, Olga Prieto-Ballesteros, Guillermo Muñoz Caro, Cristobal González-Díaz, Victoria Muñoz-Iglesias, and Santos Gálvez-Martínez. 2019. "Characterizing Interstellar Medium, Planetary Surface and Deep Environments by Spectroscopic Techniques Using Unique Simulation Chambers at Centro de Astrobiologia (CAB)" Life 9, no. 3: 72. https://doi.org/10.3390/life9030072
APA StyleMateo-Marti, E., Prieto-Ballesteros, O., Muñoz Caro, G., González-Díaz, C., Muñoz-Iglesias, V., & Gálvez-Martínez, S. (2019). Characterizing Interstellar Medium, Planetary Surface and Deep Environments by Spectroscopic Techniques Using Unique Simulation Chambers at Centro de Astrobiologia (CAB). Life, 9(3), 72. https://doi.org/10.3390/life9030072





