The Stereochemical Basis of the Genetic Code and the (Mostly) Autotrophic Origin of Life
Abstract
:1. Introduction
2. Origin of the Genetic Code and the First Peptides
2.1. Theories about Codeless Peptide Synthesis and the Code Appearing before Translation
2.2. Where Did the First Peptides’ Amino Acids Come from?
2.3. The “Stereochemical Era” of Code Evolution
| Amino Acid | Miller-Urey | Johnson-Wang |
|---|---|---|
| A | + | |
| C | N/D | |
| D | + | + |
| E | + | |
| F * | + | |
| G | + | |
| H * | + | |
| I * | + | + |
| K | + | |
| L * | + | + |
| M | + | |
| N | ||
| P | + | |
| Q | + | |
| R * | + | |
| S | + | |
| T | + | |
| V | + | |
| W * | + | |
| Y * | + |
3. Discussion
3.1. Abiotic Amino Acids as unlikely Components of the First Peptides
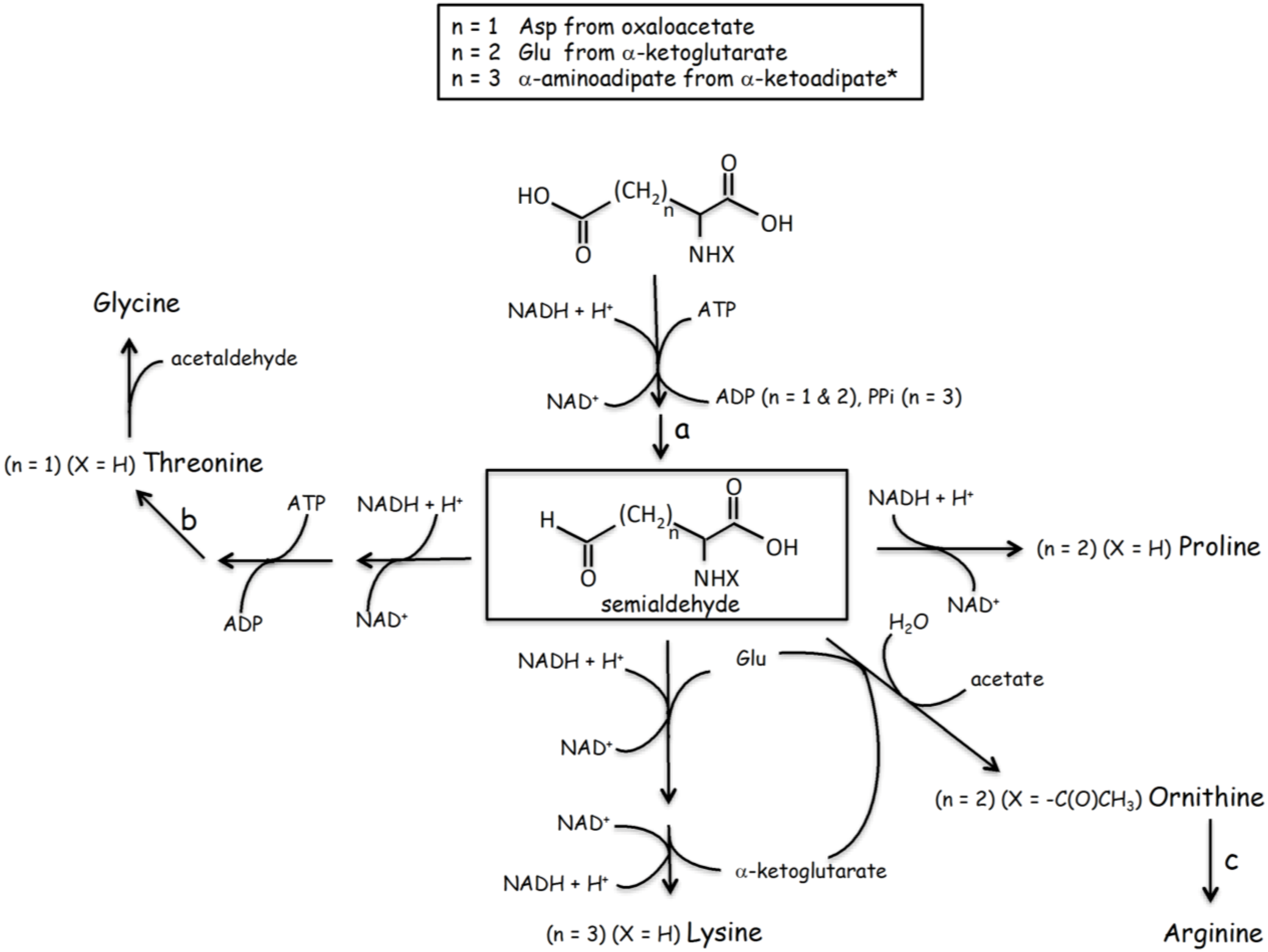
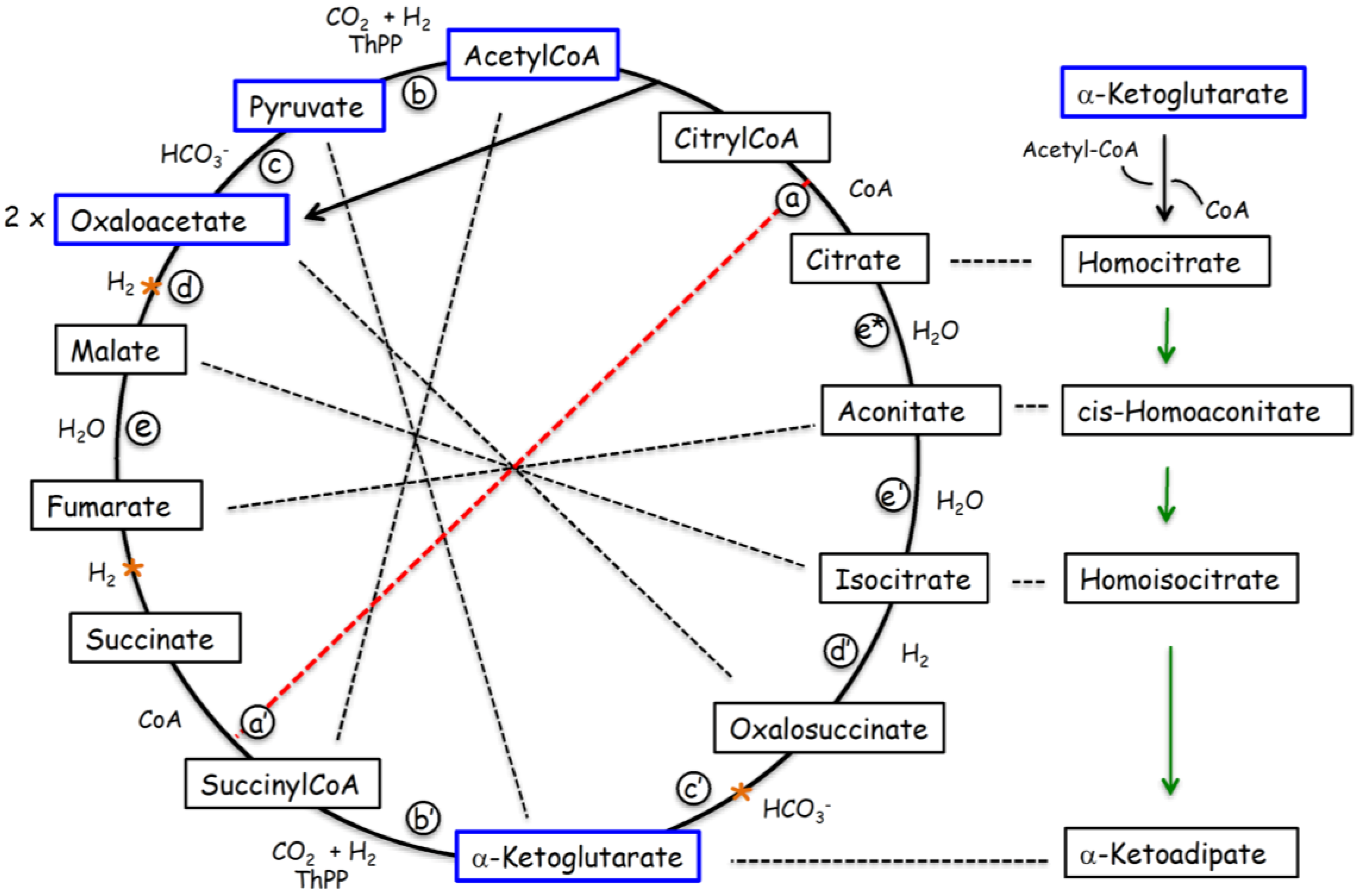
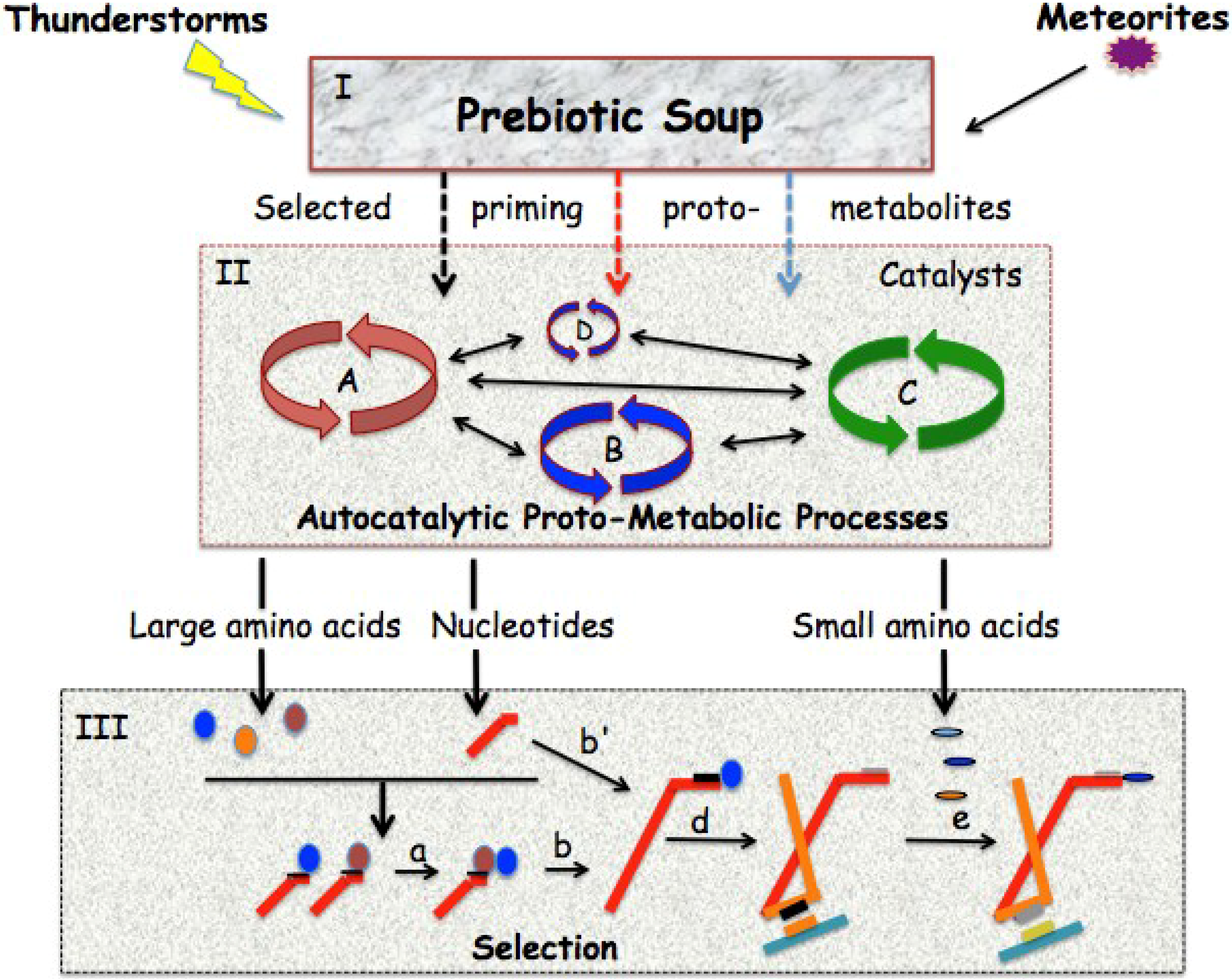
3.2. Gene Duplication of Primordial RNA Adaptors and Expansion of the Genetic Code
4. Conclusions
Acknowledgments
Conflicts of Interest
References
- Oparin, A.I. The Origin of Life; MacMillan: New York, NY, USA, 1938. [Google Scholar]
- Miller, S.L.; Urey, H.C. Organic compound synthesis on the primitive earth. Science 1959, 130, 245–251. [Google Scholar]
- Lazcano, A.; Bada, J.L. The 1953 Stanley L. Miller experiment: Fifty years of prebiotic organic chemistry. Orig. Life Evol. Biosph. 2004, 33, 235–242. [Google Scholar]
- Parker, E.T.; Cleaves, H.J.; Dworkin, J.P.; Glavin, D.P.; Callahan, M.; Aubrey, A.; Lazcano, A.; Bada, J.L. Primordial synthesis of amines and amino acids in a 1958 Miller H2S-rich spark discharge experiment. Proc. Natl. Acad. Sci. USA 2011, 108, 5526–5531. [Google Scholar] [PubMed]
- Kvenvolden, K.; Lawless, J.; Pering, K.; Peterson, E.; Flores, J.; Ponnamperuma, C.; Kaplan, I.R.; Moore, C. Evidence for extraterrestrial amino-acids and hydrocarbons in the Murchison meteorite. Nature 1970, 228, 923–926. [Google Scholar] [PubMed]
- Wolman, Y.; Haverland, W.J.; Miller, S.L. Nonprotein amino acids from spark discharges and their comparison with the Murchison meteorite amino acids. Proc. Natl. Acad. Sci. USA 1972, 69, 809–811. [Google Scholar] [PubMed]
- Mojzsis, S.J.; Arrhenius, G.; McKeegan, K.D.; Harrison, T.M.; Nutman, A.P.; Friend, C.R.L. Evidence for life on Earth before 3800 million years ago. Nature 1996, 384, 55–59. [Google Scholar] [CrossRef] [PubMed]
- Unrau, P.J.; David, P.; Bartel, D.P. RNA-catalyzed nucleotide synthesis. Nature 1998, 395, 260–263. [Google Scholar] [PubMed]
- Gilbert, W. Origin of life: The RNA world. Nature 1986, 319, 618. [Google Scholar] [CrossRef]
- Yarus, M. Darwinian behavior in a cold, sporadically fed pool of ribonucleotides. Astrobiology 2012, 12, 1–14. [Google Scholar] [PubMed]
- Noller, H.F. The driving force for molecular evolution of translation. RNA 2004, 10, 1833–1837. [Google Scholar] [PubMed]
- Krupkin, M.; Matzov, D.; Tang, H.; Metz, M.; Kalaora, R.; Belousoff, M.J.; Zimmerman, E.; Bashan, A.; Yonath, A. A vestige of a prebiotic bonding machine is functioning within the contemporary ribosome. Phil. Trans. R. Soc. B 2011, 366, 2972–2978. [Google Scholar] [CrossRef] [PubMed]
- Fox, G.E. Origin and evolution of the ribosome. Cold Spring Harb. Perspect. Biol. 2010, 2. [Google Scholar] [CrossRef]
- Johnson, D.B.F.; Wang, L. Imprints of the genetic code in the ribosome. Proc. Natl. Acad. Sci. USA 2010, 107, 8298–8203. [Google Scholar] [CrossRef] [PubMed]
- Yarus, M.; Widmann, J.J.; Knight, R. RNA–amino acid binding: A stereochemical era for the genetic code. J. Mol. Evol. 2009, 69, 406–429. [Google Scholar] [CrossRef]
- Rodin, A.S.; Szathmáry, E.; Rodin, S.N. On origin of genetic code and tRNA before translation. Biol. Direct 2011, 6. [Google Scholar] [CrossRef]
- Poole, A.M.; Jeffares, D.C.; Penny, D. The path from the RNA world. J. Mol. Evol. 1998, 46, 1–17. [Google Scholar] [CrossRef]
- Szathmáry, E. The origin of the genetic code: Amino acids as cofactors in an RNA World. Trends Genet. 1999, 15, 223–229. [Google Scholar] [CrossRef] [PubMed]
- Wolf, Y.I.; Koonin, E.V. On the origin of the translation system and the genetic code in the RNA world by means of natural selection, exaptation, and subfunctionalization. Biol. Direct 2007, 2. [Google Scholar] [CrossRef]
- Suga, H.; Hayashi, G.; Terasaka, N. The RNA origin of transfer RNA aminoacylation and beyond. Phil. Trans. R. Soc. B 2011, 366, 2959–2964. [Google Scholar] [CrossRef]
- Wong, J.T.-F. Emergence of life: From functional RNA selection to natural selection and beyond. Front. Biosci. 2014, 19, 1117–1150. [Google Scholar]
- Wong, J.T.-F. A coevolution theory of the genetic code. Proc. Natl. Acad. Sci. USA 1975, 72, 1909–1912. [Google Scholar] [CrossRef]
- Lehmann, J.; Cibils, M.; Libchaber, A. Emergence of a code in the polymerization of amino acids along RNA templates. PLoS One 2009, 4. [Google Scholar] [CrossRef] [PubMed]
- Longo, L.M.; Blaber, M. Protein design at the interface of the pre-biotic and biotic worlds. Arch. Biochem. Biophys. 2012, 526, 16–21. [Google Scholar] [PubMed]
- Weber, A.L.; Miller, S.L. Reasons for the occurrence of the twenty coded protein amino acid. J. Mol. Evol. 1981, 17, 273–284. [Google Scholar] [PubMed]
- Yarus, M.; Caporaso, J.G.; Knight, R. Origins of the genetic code: The escaped triplet theory. Annu. Rev. Biochem. 2005, 74, 179–198. [Google Scholar] [CrossRef] [PubMed]
- Bada, J.L. New insights into prebiotic chemistry from Stanley Miller’s spark discharge experiments. Chem. Soc. Rev. 2013, 42, 2186–2196. [Google Scholar] [CrossRef] [PubMed]
- Hernández-Montes, G.; Díaz-Mejía, J.J.; Pérez-Rueda, E.; Segovia, L. The hidden universal distribution of amino acid biosynthetic networks: A genomic perspective on their origins and evolution. Genome Biol. 2008, 9. [Google Scholar] [CrossRef]
- Braakman, R.; Smith, E. Metabolic evolution of a deep-branching hyperthermophilic chemoautotrophic bacterium. PLoS One 2014, 9. [Google Scholar] [CrossRef] [PubMed]
- Wächtershäusser, G. Before enzymes and templates: Theory of surface metabolism. Microbiol. Rev. 1988, 52, 452–484. [Google Scholar] [PubMed]
- Martin, W.; Russell, M.J. On the origins of cells: A hypothesis for the evolutionary transitions from abiotic geochemistry to chemoautotrophic prokaryotes, and from prokaryotes to nucleated cells. Philos. Trans. R. Soc. Lond. Ser. B 2003, 358, 59–83. [Google Scholar] [CrossRef]
- Jadhav, V.R.; Yarus, M. Coenzymes as coribozymes. Biochimie 2002, 84, 877–888. [Google Scholar]
- Yarus, M. Getting past the RNA world: The initial darwinian ancestor. Cold Spring Harb. Perspect. Biol. 2011, 3. [Google Scholar] [CrossRef]
- Zhang, X.V.; Martin, S.T. Driving parts of the Krebs cycle in reverse through mineral photochemistry. J. Am. Chem. Soc. 2006, 128, 16032–16033. [Google Scholar] [CrossRef] [PubMed]
- Zhang, X.V.; Ellery, S.P.; Cynthia, M.; Friend, C.M.; Holland, H.D.; Michel, F.M.; Martin, A.A.; Schoonen, M.A.A.; Martin, S.T. Photodriven reduction and oxidation reactions on colloidal semiconductor particles: Implications for prebiotic synthesis. J. Photochem. Photobiol. A Chem. 2007, 18, 301–311. [Google Scholar] [CrossRef]
- Guzman, M.I.; Martin, S.T. Prebiotic metabolism: Production by mineral photoelectrochemistry of α-Ketocarboxylic acids in the reductive tricarboxylic acid cycle. Astrobiology 2009, 9, 833–842. [Google Scholar] [CrossRef] [PubMed]
- Cooper, G.; Reed, C.; Nguyen, D.; Carter, M.; Wang, Y. Detection and formation scenario of citric acid, pyruvic acid, and other possible metabolism precursors in carbonaceous meteorites. Proc. Natl. Acad. Sci. USA 2011, 108, 14015–14020. [Google Scholar] [CrossRef] [PubMed]
- Griffith, E.C.; Shoemaker, R.K.; Vaida, V. Sunlight-initiated chemistry of aqueous pyruvic acid: Building complexity in the origin of life. Orig. Life Evol. Biosph. 2013, 43, 341–352. [Google Scholar] [CrossRef] [PubMed]
- Di Giulio, M. Permuted tRNA genes of Cyanidioschyzon merolae, the origin of the tRNA molecule and the root of the Eukarya domain. J. Theor. Biol. 2008, 253, 587–592. [Google Scholar] [CrossRef] [PubMed]
- Sugahara, J.; Fujishima, K.; Morita, K.; Tomita, M.; Kanai, A. Disrupted tRNA gene diversity and possible evolutionary scenarios. J. Mol. Evol. 2009, 69, 497–504. [Google Scholar] [PubMed]
- Kim, S.H.; Sussman, J.L.; Suddath, F.L.; Quigley, G.J.; McPherson, A.; Wang, A.H.; Seeman, N.C.; Rich, A. The general structure of transfer RNA molecules. Proc. Natl. Acad. Sci. USA 1974, 71, 4970–4974. [Google Scholar] [CrossRef] [PubMed]
- Hopfield, J.H. Origin of the genetic code: A testable hypothesis based on tRNA structure, sequence, and kinetic proofreading. Proc. Natl. Acad. Sci. USA 1978, 75, 4334–4338. [Google Scholar] [CrossRef]
- Erives, A. A Model of proto-anti-codon RNA enzymes requiring l-amino acid homochirality. J. Mol. Evol. 2011, 73, 10–22. [Google Scholar] [CrossRef] [PubMed]
- Schimmel, P.; Henderson, B. Possible role of aminoacyl-RNA complexes in noncoded peptide synthesis and origin of coded synthesis. Proc. Natl. Acad. Sci. USA 1994, 91, 11283–11286. [Google Scholar] [CrossRef] [PubMed]
- Nissen, P.; Hansen, J.; Ban, N.; Moore, P.B.; Steitz, T.A. The structural basis of ribosome activity in peptide bond synthesis. Science 2000, 289, 920–930. [Google Scholar] [PubMed]
- Schimmel, P.; Giegé, R.; Moras, D.; Yokoyama, S. An operational RNA code for amino acids and possible relationship to genetic code. Proc. Natl. Acad. Sci. USA 1993, 90, 8763–8768. [Google Scholar] [CrossRef] [PubMed]
- Bloch, D.P.; McArthur, B.; Widdowson, R.; Specter, D.; Guimaraes, R.C.; Smith, J. tRNA-rRNA sequence homologies: Evidence for a common evolutionary origin? J. Mol. Evol. 1983, 19, 420–428. [Google Scholar] [CrossRef] [PubMed]
- Nazarea, A.D.; Bloch, D.P.; Semrau, A.C. Detection of a fundamental modular format common to transfer and ribosomal RNAs: Second-order spectral analysis. Proc. Natl. Acad. Sci. USA 1985, 82, 5337–5341. [Google Scholar] [CrossRef] [PubMed]
- Eigen, M.; Winkler-Oswatitsch, R. Transfer-RNA, an early gene? Naturwissenschaften 1981, 68, 282–292. [Google Scholar] [CrossRef] [PubMed]
- Illangasekare, M.; Yarus, M. A tiny RNA that catalyzes both aminoacyl-RNA and peptidyl-RNA synthesis. RNA 1999, 5, 1482–1489. [Google Scholar] [CrossRef]
- Hsiao, C.; Mohan, S.; Kalahar, B.K.; Williams, L.D. Peeling the onion: Ribosomes are ancient molecular fossils. Mol. Biol. Evol. 2009, 26, 2415–2425. [Google Scholar] [CrossRef] [PubMed]
© 2014 by the authors; licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Fontecilla-Camps, J.C. The Stereochemical Basis of the Genetic Code and the (Mostly) Autotrophic Origin of Life. Life 2014, 4, 1013-1025. https://doi.org/10.3390/life4041013
Fontecilla-Camps JC. The Stereochemical Basis of the Genetic Code and the (Mostly) Autotrophic Origin of Life. Life. 2014; 4(4):1013-1025. https://doi.org/10.3390/life4041013
Chicago/Turabian StyleFontecilla-Camps, Juan C. 2014. "The Stereochemical Basis of the Genetic Code and the (Mostly) Autotrophic Origin of Life" Life 4, no. 4: 1013-1025. https://doi.org/10.3390/life4041013
APA StyleFontecilla-Camps, J. C. (2014). The Stereochemical Basis of the Genetic Code and the (Mostly) Autotrophic Origin of Life. Life, 4(4), 1013-1025. https://doi.org/10.3390/life4041013




