Adaptation of Plants to UV-B Radiation with Altitude in Tuha Basin: Synergistic Regulation of Epidermal Structure, Secondary Metabolites, and Organic Element Allocation
Abstract
1. Introduction
2. Materials and Methods
2.1. Study Area and Sample Collection
2.2. Chlorophyll Content Analysis
2.3. Analysis of Anthocyanin Content
2.4. Flavonoid Content Analysis
2.5. Analysis of Cuticle Thickness
2.6. Statistics of Epidermal Hair Density
2.7. Analysis of Stomatal Features
2.8. Analysis of H2O2 and O2− Contents
2.9. Determination of Organic ELEMENT Content
2.10. Comprehensive Evaluation Based on Membership Function
2.11. Statistical Analysis
3. Results
3.1. Changes in UV-B Absorbing Substances and Chloroplast Pigments in Plants Across Altitudes in the Tuha Basin
3.2. Principal Component Analysis (PCA) on the Physiological Indicators in Plant Tolerance to UV-B Under Different Altitudes
3.3. Comprehensive Evaluation of UV-B Tolerance in Tuha Plants Under Different Altitudes Based on Membership Function Analysis
3.4. Variations in Epidermal Hairs and Cuticles in Various Plant Species from Different Altitudes of the Tuha Basin
3.5. Stomatal Morphology in Various Plant Species from Different Altitudes of the Tuha Basin
3.6. Difference in ROS Contents in PLANT Species from Different Altitudes of the Tuha Basin
3.7. Changes in C, N, H, and S Contents in Leaves of Plants from Different Altitudes
4. Discussion
4.1. Plants Adapt to the Altitude-Dependent UV-B Radiation Through Differentially Regulating the Levels of Photo-Protective and UV-B Absorbing Substances in the Tuha Basin
4.2. The Epidermal Structure and Stomatal Characteristics Coordinately Regulate Plants in Adaptation to the High-Altitude Environments in the Tuha Basin
4.3. Plants in the Tuha Basin Reconfigure Element Allocation to Cope with HA Environments
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| Ant | Anthocyanin |
| C | Carbon |
| Car | Carotenoid |
| Chl | Chlorophyll |
| Fla | Flavonoid |
| HA | High altitude |
| H2O2 | Hydrogen oxide |
| LA | Low altitude |
| MA | Middle altitude |
| N | Nitrogen |
| O2− | Superoxide anion |
| PCA | Principal component analysis |
| ROS | Reactive oxygen species |
| S | Sulfur |
| Tuha | Turpan-Hami |
| UV-B | Ultraviolet B |
References
- Domros, M.; Peng, G.; Zhang, S. The characteristics of the desert climate at Turpan, China. Erdkunde 1992, 119, 217–233. [Google Scholar] [CrossRef]
- Zhang, Q.; Yan, M.; Zhang, L.; Shao, W.; Chen, Y.; Dong, Y. Three decades of oasis transition and its driving factors in Turpan–Hami Basin in Xinjiang, China: A complex network approach. Remote Sens. 2024, 16, 465. [Google Scholar] [CrossRef]
- Zhang, S.Q.; Pu, Z.C.; Li, J.L. The spatial-temporal variation of sunshine duration in Xinjiang during 1961–2010. Acta Geogr. Sin. 2013, 68, 1481–1492. [Google Scholar]
- Kirschner, G.K.; Xiao, T.T.; Blilou, I. Rooting in the desert: A developmental overview on desert plants. Genes 2021, 12, 709. [Google Scholar] [CrossRef] [PubMed]
- Chen, Z.; Dong, Y.; Huang, X. Plant responses to UV-B radiation: Signaling, acclimation and stress tolerance. Stress Biol. 2022, 2, 51. [Google Scholar] [CrossRef]
- Fu, W.D. The spatiotemporal distribution patterns of infrared and ultraviolet radiation in Xinjiang. Arid Land Geogr. 2000, 23, 116–121. [Google Scholar]
- Podolec, R.; Demarsy, E.; Ulm, R. Perception and signaling of ultraviolet-B radiation in plants. Annu Rev. Plant Biol. 2021, 72, 793–822. [Google Scholar] [CrossRef]
- Yao, J.W.; Ma, Z.; Ma, Y.Q.; Zhu, Y.; Lei, M.Q.; Hao, C.Y.; Chen, L.Y.; Xu, Z.Q.; Huang, X. Role of melatonin in UV-B signaling pathway and UV-B stress resistance in Arabidopsis thaliana. Plant Cell Environ. 2021, 44, 114–129. [Google Scholar] [CrossRef]
- Jansen, M.A.K.; Gaba, V.; Greenberg, B.M. Higher plants and UV-B radiation: Balancing damage, repair and acclimation. Trends Plant Sci. 1998, 3, 131–135. [Google Scholar] [CrossRef]
- Christopher, L.G.; Keara, A.F. UV RESISTANCE LOCUS 8 signalling enhances photosynthetic resilience to herbicide-induced damage in Arabidopsis thaliana. New Phytol. 2025, 247, 1763–1776. [Google Scholar]
- León-Chan, R.G.; López-Meyer, M.; Osuna-Enciso, T.; Sañudo-Barajas, J.A.; Heredia, J.B.; León-Félix, J. Low temperature and ultraviolet-B radiation affect chlorophyll content and induce the accumulation of UV-B-absorbing and antioxidant compounds in bell pepper (Capsicum annuum) plants. Environ. Exp. Bot. 2017, 139, 143–151. [Google Scholar] [CrossRef]
- Sztatelman, O.; Grzyb, J.; Gabrys, H.; Banaś, A.K. The effect of UV-B on Arabidopsis leaves depends on light conditions after treatment. BMC Plant Biol. 2015, 15, 281. [Google Scholar] [CrossRef] [PubMed]
- Laakso, K.; Sullivan, J.; Huttunen, S. The effects of UV-B radiation on epidermal anatomy in loblolly pine (Pinus taeda L.) and Scots pine (Pinus sylvestris L.). Plant Cell Environ. 2000, 23, 461–472. [Google Scholar] [CrossRef]
- Müller-Xing, R.; Xing, Q. Goodrich J Footprints of the sun: Memory of UV and light stress in plants. Front. Plant Sci. 2014, 5, 474. [Google Scholar]
- Britt, A.B. Repair of DNA damage induced by ultraviolet radiation. Plant Physiol. 1995, 108, 891–896. [Google Scholar] [CrossRef]
- Li, N.; Teranishi, M.; Yamaguchi, H.; Matsushita, T.; Watahiki, M.K.; Tsuge, T.; Li, S.S.; Hidema, J. UV-B-induced CPD photolyase gene expression is regulated by UVR8-dependent and -independent pathways in Arabidopsis. Plant Cell Physiol. 2015, 56, 2014–2023. [Google Scholar] [CrossRef]
- Wada, M.; Kagawa, T.; Sato, Y. Chloroplast movement. Annu. Rev. Plant Biol. 2003, 54, 455–468. [Google Scholar] [CrossRef]
- Takahashi, S.; Badger, M.R. Photoprotection in plants: A new light on photosystem II damage. Trends Plant Sci. 2011, 16, 53–60. [Google Scholar] [CrossRef]
- Kalidhasan, N.; Bhagavan, N.B.; Kannan, N.D. Ultraviolet B (280-320 nm) enhanced radiation induced changes in secondary metabolites and photosystem-II of medicinal plant Withania somnifera Dunal. Acad. J. 2013, 7, 3112–3120. [Google Scholar]
- Tsurunaga, Y.; Takahashi, T.; Katsube, T.; Kudo, A.; Kuramitsu, O.; Ishiwata, M.; Matsumoto, S. Effects of UV-B irradiation on the levels of anthocyanin, rutin and radical scavenging activity of buckwheat sprouts. Food Chem. 2013, 141, 552–556. [Google Scholar] [CrossRef]
- Agarwal, S. Increased antioxidant activity in Cassia seedlings under UV-B radiation. Biol. Plantarum 2007, 51, 157–160. [Google Scholar] [CrossRef]
- Wang, H.; Gui, M.; Tian, X.; Xin, X.; Wang, T.; Li, J. Effects of UV-B on vitamin C, phenols, flavonoids and their related enzyme activities in mung bean sprouts (Vigna radiata). Int. J. Food Sci. Technol. 2017, 52, 827–833. [Google Scholar] [CrossRef]
- Manetas, Y. The importance of being hairy: The adverse effects of hair removal on stem photosynthesis of Verbascum speciosum are due to solar UV-B radiation. New Phytol. 2003, 158, 503–508. [Google Scholar] [CrossRef]
- Yeats, H.; Rose, J.K.C. The Formation and Function of Plant Cuticles. Plant Physiol. 2013, 163, 5–20. [Google Scholar] [CrossRef] [PubMed]
- Shepherd, T.; Griffiths, D.W. The effects of stress on plant cuticular waxes. New Phytol. 2006, 171, 469–499. [Google Scholar] [CrossRef] [PubMed]
- Riederer, M. Biology of the plant cuticle. Ann. Plant Rev. 2007, 23, 1–10. [Google Scholar]
- Nawrath, C.; Schreiber, L.; Franke, R.B.; Geldner, N.; Reina-Pinto, J.J.; Kunst, L. Apoplastic diffusion barriers in Arabidopsis. Arab. Book 2013, 11, e0167. [Google Scholar] [CrossRef]
- González Moreno, A.; de Cózar, A.; Prieto, P.; Domínguez, E.; Heredia, A. Radiationless mechanism of UV deactivation by cuticle phenolics in plants. Nat. Commun. 2022, 13, 1786. [Google Scholar] [CrossRef]
- Zhou, X.; Gong, F.; Cao, K.; Xu, H.; Zhou, X. Calcium signaling regulates the accumulation of phenolic acids in response to UV-B stress in Rhododendron chrysanthum Pall. Plant Cell Rep. 2024, 43, 224. [Google Scholar] [CrossRef]
- Jiang, N.; García Navarrete, T.; Lee, Y.S.; Pawlowski, E.G.; Park, S.; Grotewold, E. Flavonoid pathway intermediates implicate UVR8 in functions beyond canonical UV-B signaling. Nat. Commun. 2025, 16, 7810. [Google Scholar] [CrossRef]
- Zeng, H.; Li, S.; Wang, K.; Dai, Y.; Sun, L.; Gao, Y.; Yi, S.; Li, J.; Xu, S.; Xie, G.; et al. BvCGT1-mediated differential distribution of flavonoid C-glycosides contributes to plant’s response to UV-B stress. Plant J. 2024, 120, 354–369. [Google Scholar] [CrossRef]
- Wen, J.W.; Chen, H.X.; Teng, Y.P.; Zhang, S.X.; Wang, R.L. Variation of leaf stomatal traits in Quercus species along the altitudinal gradient in Taibai Mountain, China. Acta Ecol. Sin. 2018, 38, 6712–6721. [Google Scholar]
- Dai, L.; Li, J.; Harmens, H.; Zhang, C. Melatonin enhances drought resistance by regulating leaf stomatal behaviour, root growth and catalase activity in two contrasting rapeseed (Brassica napus L.) genotypes. Plant Physiol. Biochem. 2020, 149, 86–95. [Google Scholar] [CrossRef]
- Rasouli, F.; Kiani-Pouya, A.; Li, L.; Zhang, H.; Chen, Z.; Hedrich, R.; Wilson, R.; Shabala, S. Sugar beet (Beta vulgaris) guard cells responses to salinity stress: A Proteomic Analysis. Int. J. Mol. Sci. 2020, 21, 2331. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Yu, G.; He, N.; Wang, Q.; Xia, F.; Zhao, N.; Xu, Z.; Ge, J. Elevation-related variation in leaf stomatal traits as a function of plant functional type: Evidence from Changbai Mountain, China. PLoS ONE 2014, 9, e115395. [Google Scholar] [CrossRef] [PubMed]
- Yang, X.X.; Yang, Y.; Ji, C.J.; Feng, T.; Shi, Y.; Lin, L.; Ma, J.J.; He, J.S. Large-scale patterns of stomatal traits in Tibetan and Mongolian grassland species. Basic Appl. Ecol. 2014, 15, 122–132. [Google Scholar] [CrossRef]
- Sun, H.T.; Jiang, S.; Liu, J.M.; Guo, Y.J.; Shen, G.S.; Gu, S. Structure and ecological adaptability of the leaves of three Asteraceae species at different altitudes on the Qinghai-Tibet Plateau. Acta Ecol. Sin. 2016, 36, 1559–1570. [Google Scholar]
- Korner, C.; Bannister, P.; Mark, A. Altitudinal variation in stomatal conductance, nitrogen content and leaf anatomy in different plant life forms in New Zealand. Oecologia 1986, 69, 577–588. [Google Scholar] [CrossRef]
- Kessler, M.; Siorak, Y.; Wunderlich, M.; Wegner, C. Patterns of morphological leaf traits among pteridophytes along humidity and temperature gradients in the Bolivian Andes. Funct. Plant Biol. 2007, 34, 963–971. [Google Scholar] [CrossRef]
- Sartory, D.P.; Grobbelaar, J.U. Extraction of chlorophyll a from freshwater phytoplankton for spectrophotometric analysis. Hydrobiologia 1984, 114, 177–187. [Google Scholar] [CrossRef]
- Vandenbussche, F.; Habricot, Y.; Condiff, A.S. HY5 is a point of convergence between cryptochrome and cytokinin signalling path-ways in Arabidopsis thaliana. Plant J. 2007, 49, 428–441. [Google Scholar] [CrossRef]
- Tanaka, T.; Tanaka, H.; Machida, C.; Watanabe, M.; Machida, Y. A new method for rapid visualization of defects in leaf cuticle reveals five intrinsic patterns of surface defects in Arabidopsis. Plant J. 2004, 37, 139–146. [Google Scholar] [CrossRef]
- Yang, S.; Bai, L.; Guo, H.C.; Miu, Y.S.; Li, J. Morphological Characteristics, Types, and Developmental Process of Leaf Trichomes in Potato (Solanum tuberosum). Acta Agron. Sin. 2025, 51, 1582–1598. [Google Scholar]
- Han, C.; Liu, Y.; Shi, W.; Qiao, Y.; Wang, L.; Tian, Y.; Fan, M.; Deng, Z.; Lau, O.S.; De Jaeger, G.; et al. KIN10 promotes stomatal development through stabilization of the SPEECHLESS transcription factor. Nat. Commun. 2020, 11, 4214. [Google Scholar] [CrossRef]
- Wang, H.; Hou, J.; Li, Y.; Zhang, Y.; Huang, J.; Liang, W. Nitric oxide-mediated cytosolic glucose-6-phosphate dehydrogenase is involved in aluminum toxicity of soybean under high aluminum concentration. Plant Soil. 2017, 416, 39–52. [Google Scholar] [CrossRef]
- Jha, R.K.; Mishra, A. Introgression of SbERD4 gene encodes an early-responsive dehydration-stress protein that confers tolerance against different types of abiotic stresses in transgenic tobacco. Cells 2022, 11, 62. [Google Scholar] [CrossRef]
- Zhao, L.Q.; Zhang, F.; Guo, J.k.; Yang, Y.L.; Li, B.B.; Zhang, L.X. Nitric oxide functions as a signal in salt resistance in the calli from two ecotypes of reed. Plant Physiol. 2004, 134, 849–857. [Google Scholar] [CrossRef] [PubMed]
- Cao, X.; Jiang, F.; Wang, X.; Zang, Y.; Wu, Z. Comprehensive evaluation and screening for chilling-tolerance in tomato lines at the seedling stage. Euphytica 2015, 205, 569–584. [Google Scholar] [CrossRef]
- Long, L.M.; Patel, H.P.; Cory, W.C.; Stapleton, A.E. The maize epicuticular wax layer provides UV protection. Funct. Plant Biol. 2003, 30, 75–81. [Google Scholar] [CrossRef] [PubMed]
- Wang, S.; Zhou, Z.; Rahiman, R.; Lee, G.S.Y.; Yeo, Y.K.; Yang, X.; Lau, O.S. Light regulates stomatal development by modulating paracrine signaling from inner tissues. Nat. Commun. 2021, 12, 3403. [Google Scholar] [CrossRef]
- Tan, Y.; Duan, Y.; Chi, Q.; Wang, R.; Yin, Y.; Cui, D.; Li, S.; Wang, A.; Ma, R.; Li, B. The Role of Reactive Oxygen Species in Plant Response to Radiation. Int. J. Mol. Sci. 2023, 24, 3346. [Google Scholar] [CrossRef]
- Kopriva, S.; Malagoli, M.; Takahashi, H. Sulfur nutrition: Impacts on plant development, metabolism.; and stress responses. J. Exp. Bot. 2019, 70, 4069–4073. [Google Scholar] [CrossRef] [PubMed]
- Cheng, X.; Ma, T.; Wang, P.; Liang, Y.; Zhang, J.; Zhang, A.; Chen, Q.; Li, W.; Ge, Q.; Sun, X.; et al. Foliar nitrogen application from veraison to preharvest improved flavonoids, fatty acids and aliphatic volatiles composition in grapes and wines. Food Res. Int. 2020, 137, 109566. [Google Scholar] [CrossRef] [PubMed]
- Li, Z.; Jiang, H.; Yan, H.; Jiang, X.; Ma, Y.; Qin, Y. Carbon and nitrogen metabolism under nitrogen variation affects flavonoid accumulation in the leaves of Coreopsis tinctoria. Peer J. 2021, 9, e12152. [Google Scholar] [CrossRef]
- Cha, O.K.; Yang, S.; Lee, H. Transcriptomics using the enriched Arabidopsis shoot apex reveals developmental priming genes Involved in plastic plant growth under salt stress conditions. Plants 2022, 11, 2546. [Google Scholar] [CrossRef] [PubMed]
- Stracke, R.; Favory, J.J.; Gruber, H.; Bartelniewoehner, L.; Bartels, S.; Binkert, M.; Funk, M.; Weisshaar, B.; Ulm, R. The Arabidopsis bZIP transcription factor HY5 regulates expression of the PFG1/MYB12 gene in response to light and ultraviolet-B radiation. Plant Cell Environ. 2010, 33, 88–103. [Google Scholar]
- Tissot, N.; Ulm, R. Cryptochrome-mediated blue-light signalling modulates UVR8 photoreceptor activity and contributes to UV-B tolerance in Arabidopsis. Nat. Commun. 2020, 11, 1323. [Google Scholar] [CrossRef]
- Ueda, T.; Nakamura, C. Ultraviolet-defense mechanisms in higher plants. Biotechnol. Biotec. Eq. 2011, 25, 2177–2182. [Google Scholar] [CrossRef]
- Noctor, G.; Veljovic-Jovanovic, S.; Driscoll, S.; Novitskaya, L.; Foyer, C.H. Drought and oxidative load in the leaves of C3 plants: A predominant role for photorespiration. Ann. Bot. 2002, 89, 841–850. [Google Scholar] [CrossRef]
- Mittler, R.; Zandalinas, S.I.; Fichman, Y.; Van Breusegem, F. Reactive oxygen species signalling in plant stress responses. Nat. Rev. Mol. Cell Bio. 2022, 23, 663–679. [Google Scholar] [CrossRef]
- Karaourniotis, G.; Liakopoulos, G.; Nikolopoulos, D.; Bresta, P. Protective and defensive roles of non-glandular trichomes against multiple stresses: Structure–function coordination. J. For. Res. 2020, 31, 1–12. [Google Scholar] [CrossRef]
- Schuurink, R.; Tissier, A. Glandular trichomes: Micro-organs with model status? New. Phytol. 2020, 225, 2251–2266. [Google Scholar] [CrossRef] [PubMed]
- Schonherr, J. Water permeability of isolated cuticular membranes: The effect of cuticular waxes on diffusion of water. Planta 1976, 131, 159–164. [Google Scholar] [CrossRef]
- Seo, P.J.; Park, C.M. Cuticular wax biosynthesis as a way of inducing drought resistance. Plant Signal. Behav. 2011, 6, 1043–1045. [Google Scholar] [CrossRef]
- Liu, Y.; Wang, J.; Liu, X.T.; Liao, T.; Ren, H.; Liu, L.; Huang, X. The UV-B photoreceptor UVR8 interacts with the LOX1 enzyme to promote stomatal closure through the LOX-derived oxylipin pathway. Plant Cell 2025, 37, koaf060. [Google Scholar] [CrossRef]
- Nolke, G.; Barsoum, M.; Houdelet, M.; Arcalís, E.; Kreuzaler, F.; Fischer, R.; Schillberg, S. The integration of algal carbon concentration mechanism components into tobacco chloroplasts increases photosynthetic efficiency and biomass. Biotechnol. J. 2018, 14, 1800170. [Google Scholar] [CrossRef]
- Larbat, R.; Robin, C.; Lillo, C.; Drengstig, T.; Ruoff, P. Modeling the diversion of primary carbon flux into secondary metabolism under variable nitrate and light/dark conditions. J. Theor. Biol. 2016, 402, 144–157. [Google Scholar] [CrossRef]
- Liu, M.; Gong, J.; Li, Y.; Li, X.; Yang, B.; Zhang, Z.; Yang, L.; Hou, X. Growth–defense trade-off regulated by hormones in grass plants growing under different grazing intensities. Physiol. Plantarum 2018, 166, 553–569. [Google Scholar] [CrossRef]
- Nunes-Nesi, A.; Fernie, A.R.; Stitt, M. Metabolic and signaling aspects underpinning the regulation of plant carbon nitrogen interactions. Mol. Plant 2010, 3, 973–996. [Google Scholar] [CrossRef]
- Delgado, E.; Mitchell, R.A.C.; Parry, M.A.; Driscoll, S.P.; Mitchell, V.J.; Lawlor, D.W. Interacting effects of CO2 concentration, temperature and nitrogen supply on the photosynthesis and composition of winter wheat leaves. Plant Cell Environ. 1994, 17, 1205–1213. [Google Scholar] [CrossRef]
- Gong, J.; Zhang, Z.; Wang, B.; Shi, J.; Zhang, W.; Dong, Q.; Song, L.; Li, Y.; Liu, Y. N addition rebalances the carbon and nitrogen metabolisms of Leymus chinensis through leaf N investment. Plant Physiol. Biochem. 2022, 185, 221–232. [Google Scholar] [CrossRef]
- Sweetlove, L.J.; Lytovchenko, A.; Morgan, M. Mitochondrial uncoupling protein is required for efficient photosynthesis. Proc. Natl. Acad. Sci. USA 2006, 103, 19587–19592. [Google Scholar] [CrossRef] [PubMed]
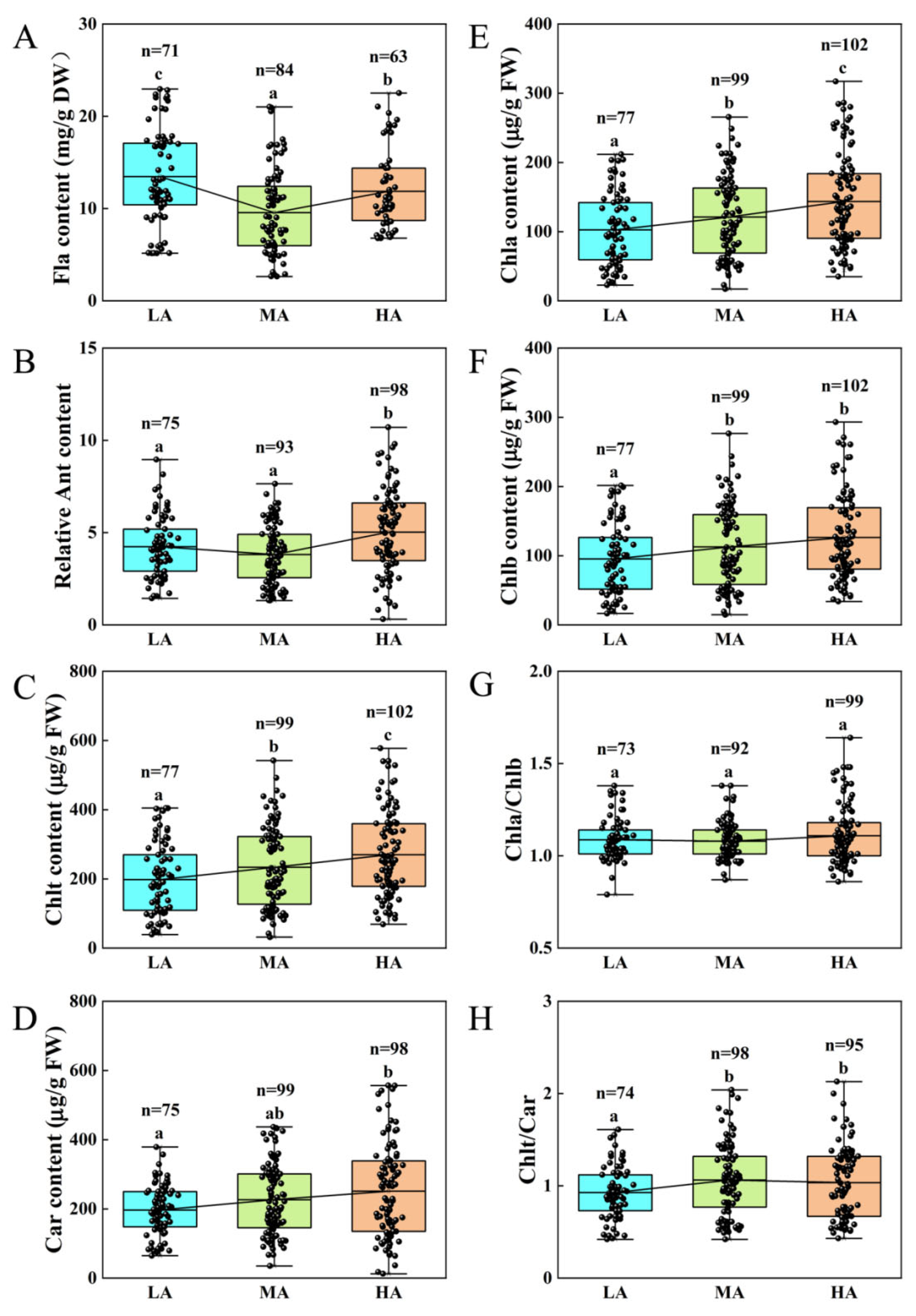
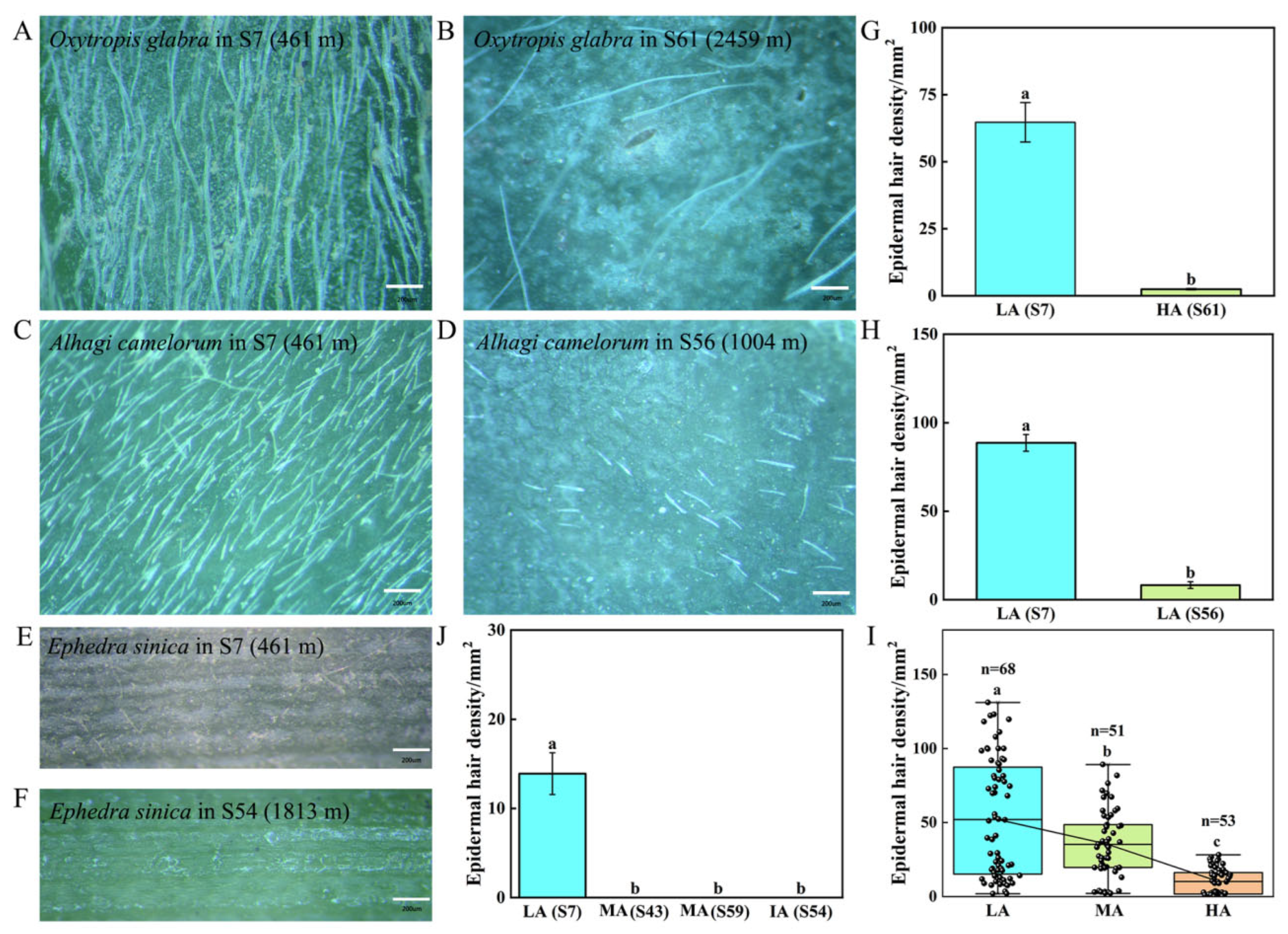
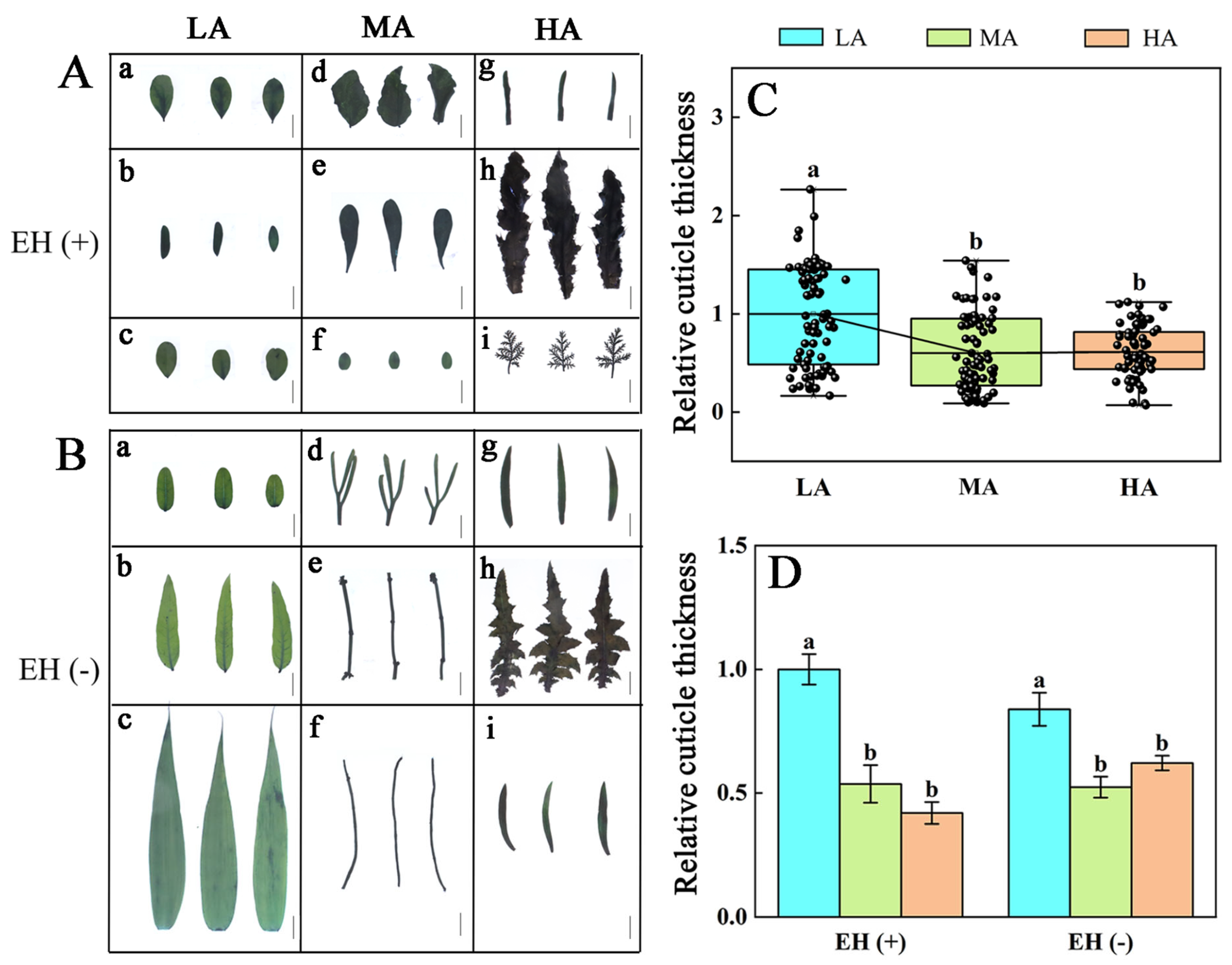

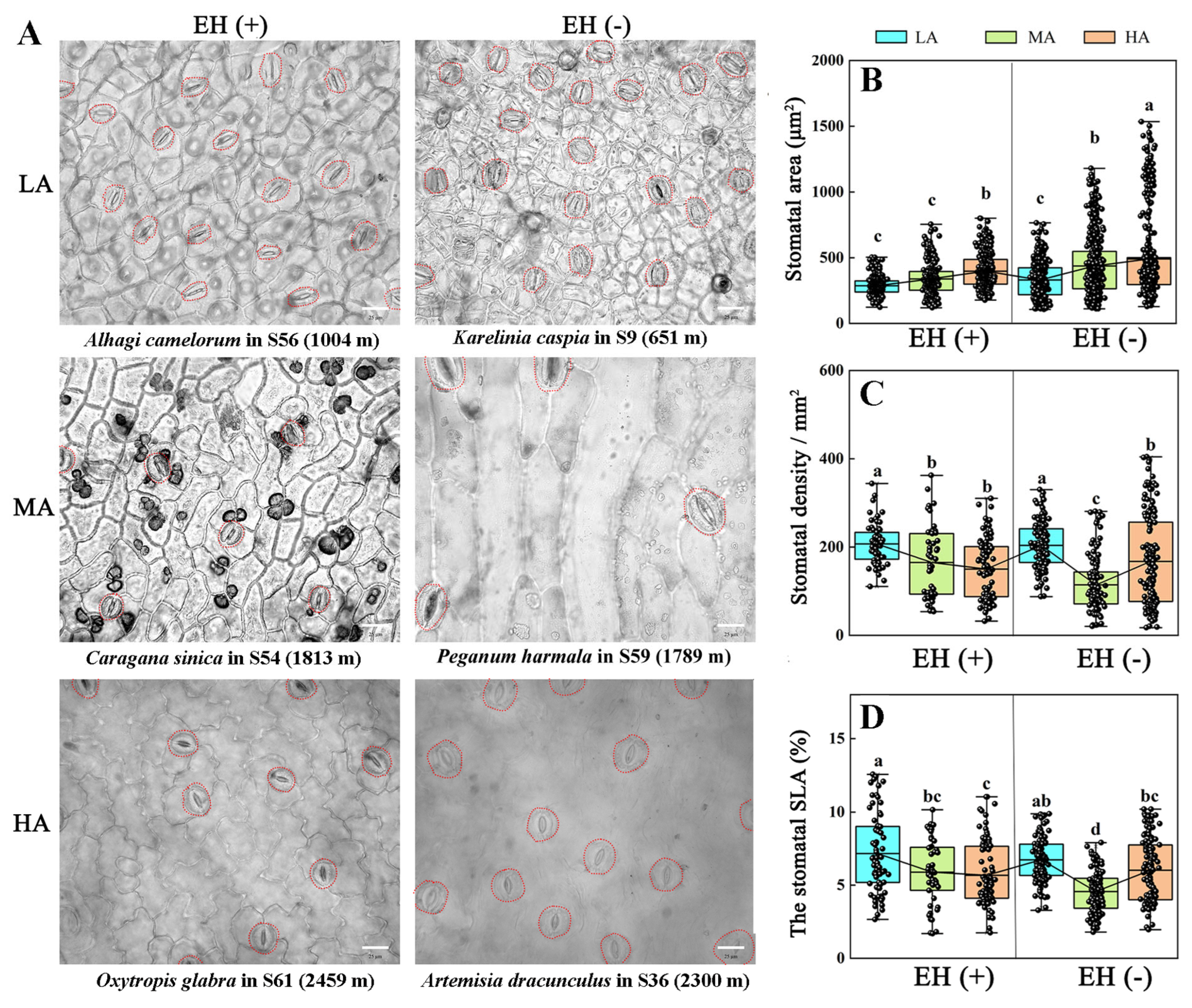
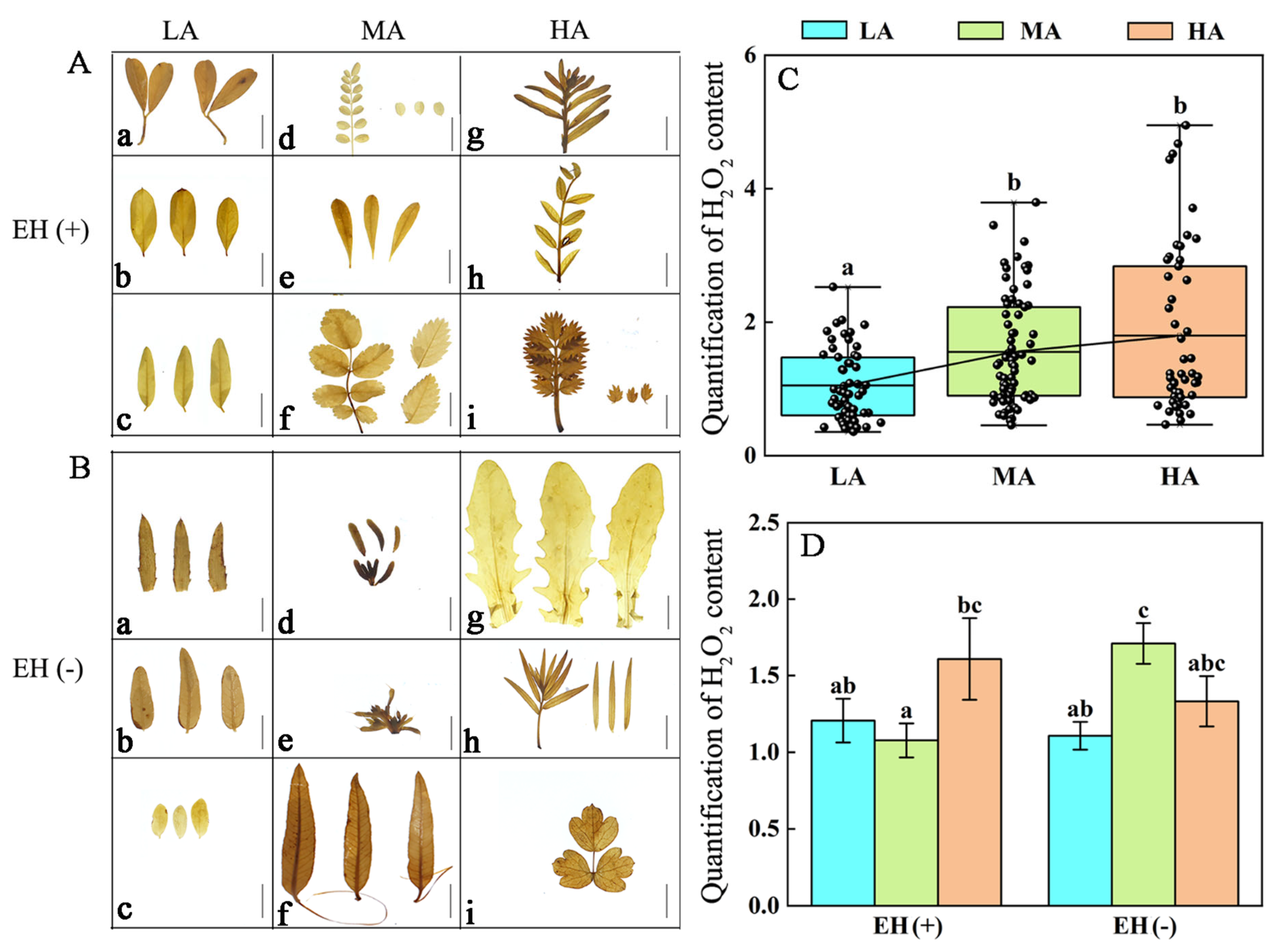
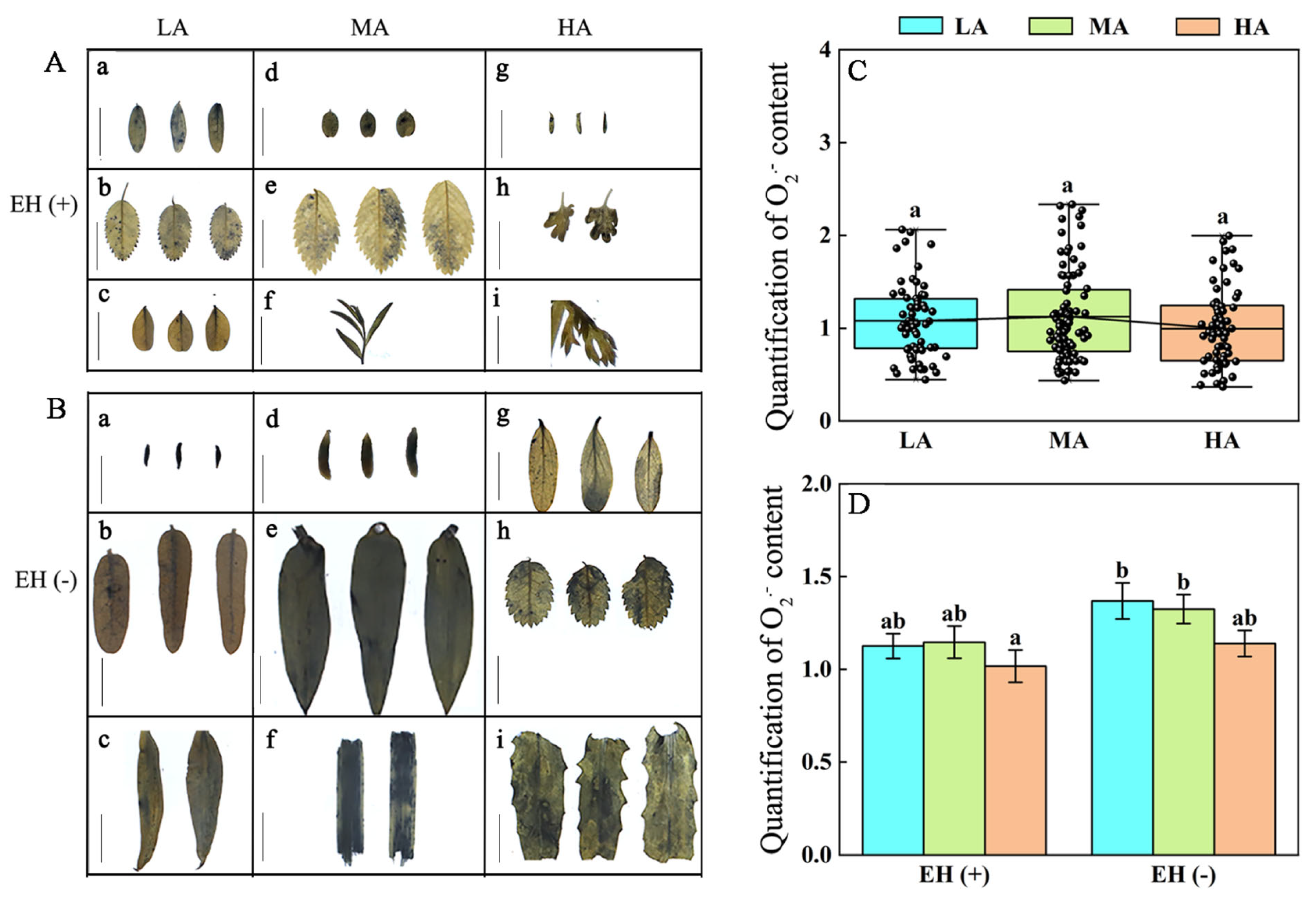

| Altitude Type | Principal Components | PC1 | PC2 | PC3 |
|---|---|---|---|---|
| Low altitude | Fla (n = 71) | 0.306 | 0.227 | 0.695 |
| Ant (n = 75) | 0.357 | 0.419 | 0.581 | |
| Chl a (n = 77) | 0.977 | 0.116 | −0.170 | |
| Chl b (n = 77) | 0.991 | −0.016 | −0.116 | |
| Car (n = 75) | 0.732 | 0.610 | −0.217 | |
| Chl a/Chl b (n = 73) | 0.483 | 0.714 | −0.348 | |
| Chl t/Car (n = 74) | 0.696 | −0.673 | −0.008 | |
| Chl t (n = 77) | 0.987 | 0.050 | −0.144 | |
| Characteristic root (λ) | 4.385 | 1.579 | 1.052 | |
| Contribution rate (%) | 54.807 | 19.732 | 13.145 | |
| Cumulative rate of contribution (%) | 54.807 | 74.540 | 87.685 | |
| Middle altitude | Fla (n = 84) | −0.486 | −0.190 | 0.744 |
| Ant (n = 93) | 0.539 | 0.260 | 0.522 | |
| Chl a (n = 99) | 0.975 | 0.138 | 0.025 | |
| Chl b (n = 99) | 0.985 | −0.039 | 0.030 | |
| Car (n = 99) | 0.575 | 0.772 | 0.005 | |
| Chl a/Chl b (n = 92) | −0.476 | 0.828 | −0.057 | |
| Chl t/Car (n = 98) | 0.539 | −0.791 | −0.057 | |
| Chl t (n = 99) | 0.985 | 0.049 | 0.028 | |
| Characteristic root (λ) | 4.265 | 2.034 | 0.834 | |
| Contribution rate (%) | 53.314 | 25.421 | 10.430 | |
| Cumulative rate of contribution (%) | 53.314 | 78.735 | 89.165 | |
| High altitude | Fla (n = 63) | 0.207 | −0.428 | 0.806 |
| Ant (n = 98) | 0.749 | −0.160 | 0.126 | |
| Chl a (n = 102) | 0.877 | 0.467 | −0.014 | |
| Chl b (n = 102) | 0.905 | −0.110 | −0.266 | |
| Car (n = 98) | 0.749 | −0.338 | 0.177 | |
| Chl a/Chl b (n = 99) | −0.003 | 0.929 | 0.309 | |
| Chl t/Car (n = 95) | −0.042 | 0.981 | 0.147 | |
| Chl t (n = 102) | 0.946 | 0.206 | −0.142 | |
| Characteristic root (λ) | 3.651 | 2.420 | 0.906 | |
| Contribution rate (%) | 45.635 | 30.247 | 11.321 | |
| Cumulative rate of contribution (%) | 45.635 | 75.882 | 87.203 |
| Altitude Type | Species | Altitude (m) | D Value | The Resistance to UV |
|---|---|---|---|---|
| High altitude | Oxytropis glabra | 2212 | 0.947 | I |
| Potentilla chinensis | 2212 | 0.933 | I | |
| Neotrinia splendens | 1907 | 0.870 | II | |
| Medicago sativa | 1907 | 0.859 | II | |
| Agropyron cristatum | 2212 | 0.730 | II | |
| Chenopodium album | 1907 | 0.670 | II | |
| Astragalus scaberrimus | 2212 | 0.597 | II | |
| Iris tectorum | 2212 | 0.555 | II | |
| Oxytropis aciphylla | 2212 | 0.547 | II | |
| Echinops sphaerocephalus | 2212 | 0.501 | II | |
| Takhtajaniantha austriaca | 2212 | 0.492 | III | |
| Halogeton glomeratus | 1907 | 0.484 | III | |
| Glaucium squamigerum | 1907 | 0.460 | III | |
| Braya humilis | 1907 | 0.293 | IV | |
| Middle altitude | Phragmites australis | 1144 | 0.973 | I |
| Pennisetum alopecuroides | 1473 | 0.959 | I | |
| Carex ovatispioulata | 1473 | 0.951 | I | |
| Oxytropis glabra | 1500 | 0.782 | II | |
| Caragana sinica | 1500 | 0.771 | II | |
| Kali collinum | 1473 | 0.704 | II | |
| Sophora alopecuroides | 1144 | 0.665 | II | |
| Lepidium latifolium | 1473 | 0.643 | II | |
| Krascheninnikovia ceratoides | 1473 | 0.638 | II | |
| Takhtajaniantha austriaca | 1473 | 0.629 | II | |
| Apocynum pictum | 1144 | 0.501 | II | |
| Sympegma regelii | 1473 | 0.472 | III | |
| Krascheninnikovia ceratoides | 1500 | 0.454 | III | |
| Braya humilis | 1473 | 0.424 | III | |
| Ephedra sinica | 1473 | 0.400 | III | |
| Halogeton glomeratus | 1473 | 0.386 | III | |
| Peganum harmala | 1500 | 0.364 | III | |
| Zygophyllum fabago | 1473 | 0.332 | III | |
| Zygophyllum xanthoxylum | 1473 | 0.322 | III | |
| Neotrinia splendens | 1500 | 0.279 | IV | |
| Anabasis brevifolia | 1473 | 0.253 | IV | |
| Lycium ruthenicum | 1114 | 0.224 | IV | |
| Anabasis aphylla | 1500 | 0.152 | IV | |
| Low altitude | Sophora alopecuroides | 615 | 0.913 | I |
| Phragmites australis | 461 | 0.839 | II | |
| Populus euphratica | 615 | 0.645 | II | |
| Apocynum venetum | 510 | 0.622 | II | |
| Tamarix chinensis | 510 | 0.525 | II | |
| Capparis spinosa | 510 | 0.503 | II | |
| Alhagi camelorum | 980 | 0.467 | III | |
| Cynanchum acutum subsp | 510 | 0.368 | III | |
| Peganum harmala | 980 | 0.303 | III | |
| Lycium ruthenicum | 461 | 0.283 | IV | |
| Haloxylon ammodendron | 626 | 0.243 | IV | |
| Karelinia caspia | 510 | 0.217 | IV |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Wang, X.-M.; Zhao, G.-Q.; Chen, J.; Jing, F.-Z.; Li, L.; Wang, Y.-Y.; Ma, P.; Wu, Y.-H.; Xu, S.-J.; He, W.-L. Adaptation of Plants to UV-B Radiation with Altitude in Tuha Basin: Synergistic Regulation of Epidermal Structure, Secondary Metabolites, and Organic Element Allocation. Life 2025, 15, 1375. https://doi.org/10.3390/life15091375
Wang X-M, Zhao G-Q, Chen J, Jing F-Z, Li L, Wang Y-Y, Ma P, Wu Y-H, Xu S-J, He W-L. Adaptation of Plants to UV-B Radiation with Altitude in Tuha Basin: Synergistic Regulation of Epidermal Structure, Secondary Metabolites, and Organic Element Allocation. Life. 2025; 15(9):1375. https://doi.org/10.3390/life15091375
Chicago/Turabian StyleWang, Xiao-Min, Guo-Qiang Zhao, Jie Chen, Fang-Zheng Jing, Li Li, Yu-Ying Wang, Ping Ma, Yu-Hang Wu, Shi-Jian Xu, and Wen-Liang He. 2025. "Adaptation of Plants to UV-B Radiation with Altitude in Tuha Basin: Synergistic Regulation of Epidermal Structure, Secondary Metabolites, and Organic Element Allocation" Life 15, no. 9: 1375. https://doi.org/10.3390/life15091375
APA StyleWang, X.-M., Zhao, G.-Q., Chen, J., Jing, F.-Z., Li, L., Wang, Y.-Y., Ma, P., Wu, Y.-H., Xu, S.-J., & He, W.-L. (2025). Adaptation of Plants to UV-B Radiation with Altitude in Tuha Basin: Synergistic Regulation of Epidermal Structure, Secondary Metabolites, and Organic Element Allocation. Life, 15(9), 1375. https://doi.org/10.3390/life15091375





