Electroconvulsive Therapy and Hyperventilation: A Narrative Review
Abstract
1. Introduction
1.1. Aim
1.2. Materials and Methods
2. Narrative Review
2.1. Beginnings of Electroconvulsive Therapy and Evaluation of ECT with Concomitant Hyperventilation
2.2. Innovations and New Research Directions
3. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| BDNF | brain-derived neurotrophic factor |
| CNS | central nervous system |
| COPD | chronic obstructive pulmonary disease |
| DBS | deep brain stimulation |
| ECT | electroconvulsive therapy |
| EtCO2 | end-tidal CO2 |
| MR | magnetic resonance |
| MRSI | spectroscopic imaging |
| MST | Magnetic Seizure Therapy |
| NAA | N-acetylaspartate |
| O2 | oxygen saturation |
| rHV | reflex hyperventilation |
| STABLE | Symptom-Titrated, Algorithm-Based Longitudinal ECT |
| TMS | Transcranial Magnetic Stimulation |
References
- Baghai, T.C.; Möller, H.-J. Electroconvulsive therapy and its different indications. Dialogues Clin. Neurosci. 2008, 10, 105–117. [Google Scholar] [CrossRef]
- D’Afflitti, J.P. Electroconvulsive Therapy, 4th ed., edited by Richard Abrams. Gen. Hosp. Psychiatry 2004, 26, 331–332. [Google Scholar] [CrossRef]
- Hese, R.; Zyss, T.; Gorczyca, P. Modern requirements of electroshock therapy. Psychiatr. Pol. 2014, 48, 1283–1286. [Google Scholar] [CrossRef] [PubMed]
- Trevino, K.; McClintock, S.M.; Husain, M.M. A review of continuation electroconvulsive therapy. J. ECT 2010, 26, 186–195. [Google Scholar] [CrossRef] [PubMed]
- Bolwig, T.G. How does electroconvulsive therapy work? Theories on its mechanism. Can. J. Psychiatry 2011, 56, 13–18. [Google Scholar] [CrossRef]
- Bouckaert, F.; Sienaert, P.; Obbels, J.; Dols, A.; Vandenbulcke, M.; Stek, M.; Bolwig, T. ECT: Its brain enabling effects: A review of electroconvulsive therapy–induced structural brain plasticity. J. ECT 2014, 30, 143–151. [Google Scholar] [CrossRef]
- Wciórka, J.; Pużyński, S.; Rybakowski, J. Psychiatria; Edra Urban & Partner: Wrocław, Poland, 2017. [Google Scholar]
- Rose, D. Patients’ perspectives on electroconvulsive therapy: Systematic review. BMJ 2003, 326, 1363. [Google Scholar] [CrossRef]
- Cristancho, M.A.; Alici, Y.; Augoustides, J.G.; O’Reardon, J.P. Uncommon but serious complications associated with electroconvulsive therapy: Recognition and management for the clinician. Curr. Psychiatry Rep. 2008, 10, 474–480. [Google Scholar] [CrossRef]
- Hajak, V.L.; Hajak, G.; Ziegelmayer, C.; Grimm, S.; Trapp, W. Risk assessment of electroconvulsive therapy in clinical routine: A 3-year analysis of life-threatening events in more than 3000 treatment sessions. Front. Psychol. 2021, 12, 767915. [Google Scholar] [CrossRef]
- Leiknes, K.A.; Cooke, M.J.; Jarosch-von Schweder, L.; Harboe, I.; Høie, B. Electroconvulsive therapy during pregnancy: A systematic review of case studies. Arch. Women’s Ment. Health 2013, 18, 1–39. [Google Scholar] [CrossRef]
- de Arriba-Arnau, A.; Dalmau, A.; Soria, V.; Salvat-Pujol, N.; Ribes, C.; Sánchez-Allueva, A.; Menchón, J.M.; Urretavizcaya, M. Protocolized hyperventilation enhances electroconvulsive therapy. J. Affect. Disord. 2017, 217, 225–232. [Google Scholar] [CrossRef]
- Räsänen, J.; Martin, D.J.; Downs, J.B.; Hodges, M.R. Oxygen supplementation during electroconvulsive therapy. Br. J. Anaesth. 1988, 61, 593–597. [Google Scholar] [CrossRef] [PubMed]
- Chater, S.N.; Simpson, K.H. Effect of passive hyperventilation on seizure duration in patients undergoing electroconvulsive therapy. Br. J. Anaesth. 1988, 60, 70–73. [Google Scholar] [CrossRef] [PubMed]
- Sawayama, E.; Takahashi, M.; Inoue, A.; Nakajima, K.; Kano, A.; Sawayama, T.; Okutomi, T.; Miyaoka, H. Moderate Hyperventilation Prolongs Electroencephalogram Seizure Duration of the First Electroconvulsive Therapy. J. ECT 2008, 24, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Di Pauli, J.; Conca, A. Impact of Seizure Duration in Maintenance Electroconvulsive Therapy. Psychiatry Clin. Neurosci. 2009, 63, 769–771. [Google Scholar] [CrossRef]
- Mayur, P.; Bray, A.; Fernandes, J.; Bythe, K.; Gilbett, D. Impact of Hyperventilation on Stimulus Efficiency During the Early Phase of an Electroconvulsive Therapy Course. J. ECT 2010, 26, 91–94. [Google Scholar] [CrossRef]
- Haeck, M.; Gillmann, B.; Janouschek, H.; Grözinger, M. Electroconvulsive therapy can benefit from controlled hyperventilation using a laryngeal mask. Eur. Arch. Psychiatry Clin. Neurosci. 2011, 261 (Suppl. S2), 172–176. [Google Scholar] [CrossRef]
- de Arriba-Arnau, A.; Dalmau, A.; Salvat-Pujol, N.; Soria, V.; Bocos, J.; Menchón, J.M.; Urretavizcaya, M. Hypocapnia and hyperoxia induction using a hyperventilation protocol in electroconvulsive therapy. Rev. Psiquiatr. Salud Ment. Engl. Ed. 2017, 10, 21–27. [Google Scholar] [CrossRef]
- Suleman, R. A brief history of electroconvulsive therapy. Am. J. Psychiatry Resid. J. 2020, 16, 6. [Google Scholar] [CrossRef]
- Pande, A.C.; Shea, J.; Shettar, S.; Grunhaus, L.J.; Haskett, R.F. Effect of Hyperventilation on Seizure Length During Electroconvulsive Therapy. Biol. Psychiatry 1990, 27, 799–801. [Google Scholar] [CrossRef]
- Gündoğdu, O. The Effects of Hyperventilation on Seizure Length and Cerebral Oxygenation during Electroconvulsive Therapy. North. Clin. Istanb. 2019, 7, 246–254. [Google Scholar] [CrossRef]
- Guerrier, G.; Gianni, M.A. The effectiveness of BIS monitoring during electro-convulsive therapy: A systematic review and meta-analysis. J. Clin. Anesth. 2019, 58, 100–104. [Google Scholar] [CrossRef]
- Coshal, S.; Jones, K.; Coverdale, J.; Livingston, R. An overview of reviews on the safety of electroconvulsive therapy administered during pregnancy. J. Psychiatr. Pract. 2019, 25, 2–6. [Google Scholar] [CrossRef] [PubMed]
- Shimamoto, Y.; Michiyoshi Sanuki Kurita, S.; Ueki, M.; Kuwahara, Y. Two-handed facemask technique effectively causes hyperventilation in electroconvulsive therapy: An observational study. BMC Anesthesiol. 2022, 22, 376. [Google Scholar] [CrossRef] [PubMed]
- Luccarelli, J.; Fernandez-Robles, C.; Fernandez-Robles, C.; Horvath Ryan, J.; Berg, S.; McCoy Thomas, H.; Seiner, S.J.; Henry, M.E. Modified Anesthesia Protocol for Electroconvulsive Therapy Permits Reduction in Aerosol-Generating Bag-Mask Ventilation during the COVID-19 Pandemic. Psychother. Psychosom. 2020, 89, 314–319. [Google Scholar] [CrossRef] [PubMed]
- Ferrier, I.N.; Waite, J.; Sivasanker, V. Recent advances in electroconvulsive therapy and physical treatments for depression. BJPsych Adv. 2021, 9, 295–302. [Google Scholar] [CrossRef]
- Singh, S.; Jolly, A. Does electroconvulsive therapy cause brain damage: An update. Indian J. Psychiatry 2020, 62, 339. [Google Scholar] [CrossRef]
- Ende, G.; Braus, D.F.; Walter, S.; Wolfgang Weber-Fahr Henn, F.A. The Hippocampus in Patients Treated with Electroconvulsive Therapy. Arch. Gen. Psychiatry 2000, 57, 937–943. [Google Scholar] [CrossRef]
- Michael, N.; Erfurth, A.; Ohrmann, P.; Arolt, V.; Heindel, W.; Pfleiderer, B. Metabolic changes within the left dorsolateral prefrontal cortex occurring with electroconvulsive therapy in patients with treatment resistant unipolar depression. Psychol. Med. 2003, 33, 1277–1284. [Google Scholar] [CrossRef]
- Jorgensen, A.; Magnusson, P.; Hanson, L.G.; Kirkegaard, T.; Benveniste, H.; Lee, H.; Svarer, C.; Mikkelsen, J.D.; Fink-Jensen, A.; Knudsen, G.M.; et al. Regional brain volumes, diffusivity, and metabolite changes after electroconvulsive therapy for severe depression. Eur. Neuropsychopharmacol. 2016, 26, S304–S305. [Google Scholar] [CrossRef]
- de Arriba-Arnau, A.; Dalmau Llitjos, A.; Soria, V.; Labad, J.; Menchón, J.M.; Urretavizcaya, M. Ventilation Adjustment in ECT During COVID-19: Voluntary Hyperventilation is an Effective Strategy. Neuropsychiatr. Dis. Treat. 2021, 17, 1563–1569. [Google Scholar] [CrossRef]
- Gálvez, V.; Hadzi-Pavlovic, D.; Smith, D.; Loo, C.K. Predictors of Seizure Threshold in Right Unilateral Ultrabrief Electroconvulsive Therapy: Role of Concomitant Medications and Anaesthesia Used. Brain Stimul. 2015, 8, 486–492. [Google Scholar] [CrossRef]
- Lisanby, S.H.; Sampson, S.; Husain, M.M.; Petrides, G.; Knapp, R.G.; McCall, V.; Young, R.C.; Prudic, J.; Kellner, C.H. Toward Individualized Post-Electroconvulsive Therapy Care. J. ECT 2008, 24, 179–182. [Google Scholar] [CrossRef]
- Chen, T.; Su, H.; Li, R.; Jiang, H.; Li, X.; Wu, Q.; Tian, H.; Zhang, J.; Zhong, N.; Du, J.; et al. The exploration of optimized protocol for repetitive transcranial magnetic stimulation in the treatment of methamphetamine use disorder: A randomized sham-controlled study. EBioMedicine 2020, 60, 103027. [Google Scholar] [CrossRef]
- Cretaz, E.; Brunoni, A.R.; Lafer, B. Magnetic seizure therapy for unipolar and Bipolar depression: A systematic review. Neural Plast. 2015, 2015, 521398. [Google Scholar] [CrossRef]
- Kamel, L.Y.; Xiong, W.; Gott, B.M.; Kumar, A.; Conway, C.R. Vagus nerve stimulation: An update on a novel treatment for treatment-resistant depression. J. Neurol. Sci. 2022, 434, 120171. [Google Scholar] [CrossRef] [PubMed]
- Borrione, L.; Moffa, A.H.; Martin, D.; Loo, C.K.; Brunoni, A.R. Transcranial Direct Current Stimulation in the Acute Depressive Episode. J. ECT 2018, 34, 153–163. [Google Scholar] [CrossRef] [PubMed]
- Dougherty, D.D.; Rezai, A.R.; Carpenter, L.L.; Howland, R.H.; Bhati, M.T.; O’Reardon, J.P.; Eskandar, E.N.; Baltuch, G.H.; Machado, A.D.; Kondziolka, D.; et al. A randomized sham-controlled trial of deep brain stimulation of the ventral capsule/ventral striatum for chronic treatment-resistant depression. Biol. Psychiatry 2015, 78, 240–248. [Google Scholar] [CrossRef] [PubMed]
- Semkovska, M.; Landau, S.; Dunne, R.; Kolshus, E.; Kavanagh, A.; Jelovac, A.; Noone, M.; Carton, M.; Lambe, S.; McHugh, C.; et al. Bitemporal Versus High-Dose Unilateral Twice-Weekly Electroconvulsive Therapy for Depression (EFFECT-Dep): A Pragmatic, Randomized, Non-Inferiority Trial. Am. J. Psychiatry 2016, 173, 408–417. [Google Scholar] [CrossRef]
- Luchini, F.; Medda, P.; Mariani, M.G.; Mauri, M.; Toni, C.; Perugi, G. Electroconvulsive therapy in catatonic patients: Efficacy and predictors of response. World J. Psychiatry 2015, 5, 182. [Google Scholar] [CrossRef]
- Sienaert, P.; Lambrichts, L.; Dols, A.; De Fruyt, J. Evidence-based treatment strategies for treatment-resistant bipolar depression: A systematic review. Bipolar Disord. 2012, 15, 61–69. [Google Scholar] [CrossRef]
- Read, J.; Bentall, R. The effectiveness of electroconvulsive therapy: A literature review. Epidemiol. Psychiatr. Sci. 2010, 19, 333–347. [Google Scholar] [CrossRef]
- Somnuke, P. An Innovative Design of Siriraj Collapsible Paper Box Set for Electroconvulsive Therapy in Psychiatric Patients during COVID-19 Outbreak. Thai J. Anesthesiol. 2020, 46, 165–170. [Google Scholar]

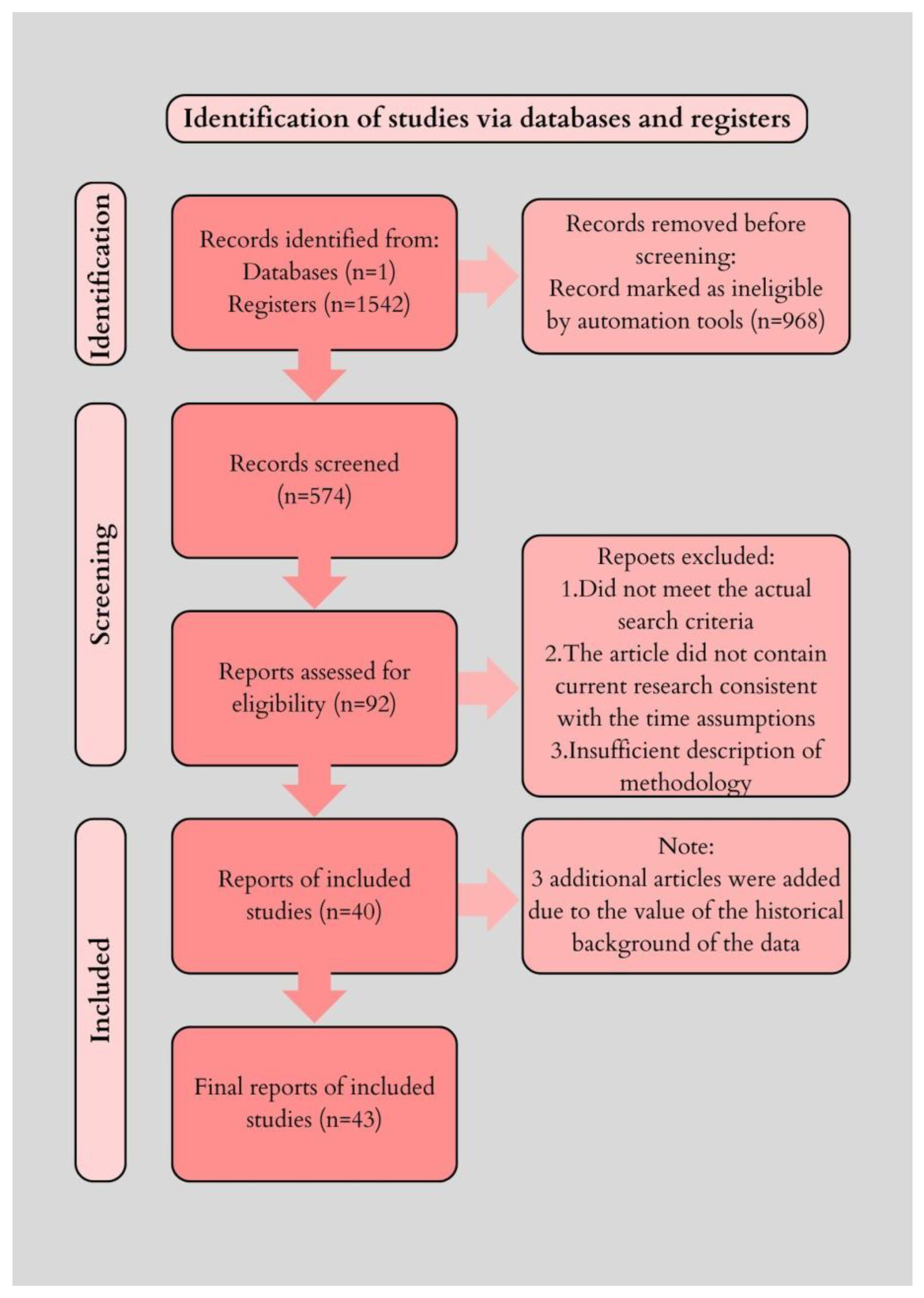
| Author, Year | Study Design | Sample Size | Centre Type | Main Limitations | Risk of Bias |
|---|---|---|---|---|---|
| Räsänen, 1988 [13] | Prospective, controlled | 12 | Single-centre | Very small sample, old ECT protocols | High |
| Chater & Simpson, 1988 [14] | Prospective | 30 | Single-centre | No randomisation, limited monitoring technology | Moderate |
| Sawayama, 2008 [15] | Prospective | 40 | Single-centre | Small sample, heterogeneous patient group | Moderate |
| Di Pauli, 2009 [16] | Prospective | 36 | Single-centre | Small sample, non-randomised | Moderate |
| Mayur, 2010 [17] | Prospective | 32 | Single-centre | Small sample, limited seizure quality assessment | Moderate |
| Haeck, 2011 [18] | Observational | 114 | Single-centre | Non-randomised, heterogeneous psychiatric diagnoses | Moderate |
| Arriba-Arnau, 2017 [19] | Protocolised intervention | 52 | Multi-centre | Modest sample size, limited follow-up | Low/Moderate |
| Author, Year | Sample Size | Population | Ventilation Protocol | Target EtCO2 (mmHg) | O2 Saturation (%) | Change in Seizure Duration | Key Findings |
|---|---|---|---|---|---|---|---|
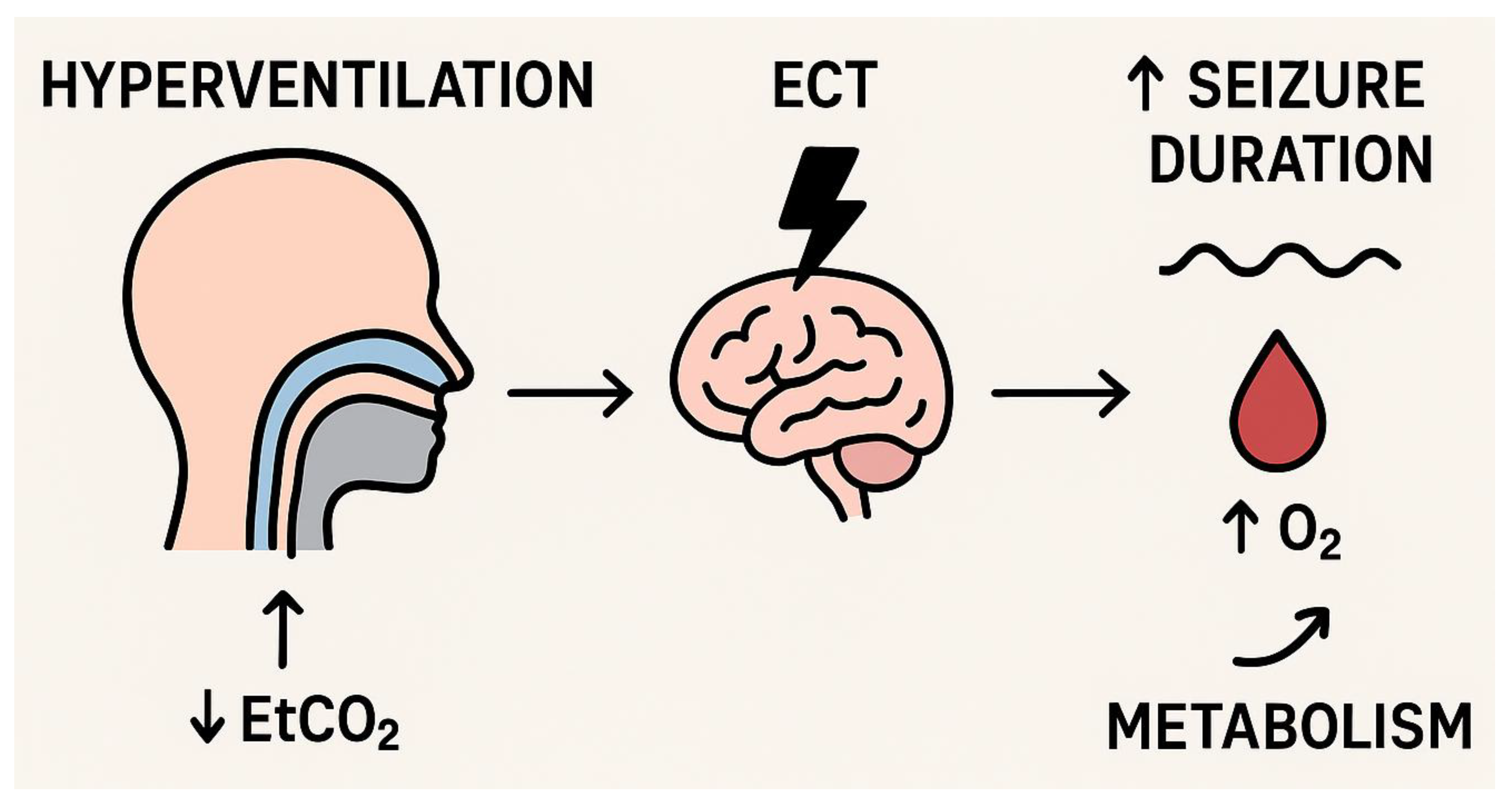
| Pande et al., 1990 [21] | 15 | Patients with depression | Varying hyperventilation levels during ECT | ~20 | Not reported | Increased seizure duration during first session | Lowering pCO2 increased seizure duration in the first session without major adverse effects |
| Chater & Simpson, 1988 [14] | 30 | Patients with depression undergoing ECT | Passive hyperventilation, EtCO2 measured pre- and postictal | 25–30 (reduced postictal) | Continuously monitored | Significant prolongation | Hyperventilation significantly prolonged seizure duration and reduced EtCO2. |
| Sawayama et al., 2008 [15] | 38 | Patients undergoing ECT | Hyperventilation (manual ventilation with 100% O2) | <30 | 99–100 | Prolonged | Hyperventilation prolonged seizure duration compared to normoventilation. |
| Di Pauli et al., 2009 [16] | 42 | Patients undergoing ECT | Controlled hyperventilation | <30 | Not specified | Prolonged | Hyperventilation increased seizure duration significantly. |
| Mayur, 2010 [17] | 40 | Patients undergoing ECT | Hyperventilation vs. normoventilation | 25–30 | Not reported | Prolonged | Hyperventilation improved seizure efficiency and duration. |
| Haeck et al., 2011 [18] | 114 | Patients with depression or psychosis (first-time ECT) | Controlled hyperventilation with laryngeal mask and mechanical ventilation | 25–30 | Not reported | Prolonged; lower electrical charge required | Hyperventilation increased seizure energy and reduced charge requirement. |
| de Arriba-Arnau & Dalmau, 2017 [19] | 51 | Patients undergoing ECT | Protocolised hyperventilation with EtCO2 and O2 monitoring | 25–30 | Correlated with seizure quality | Prolonged | rHV prolonged seizure duration; O2 correlated with seizure quality, not duration. |
| Gündoğdu, 2019 [18] | 62 | Patients undergoing ECT | Hyperventilation vs. normoventilation | 25–30 | 98–100 | Prolonged | Hyperventilation prolonged seizure length and improved oxygenation. |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Smolarczyk, J.; Piłat, P.; Blanch, J.; Cetnarowska, A.; Dębski, P.; Torrent, A.; Batalla, I.; Piegza, M. Electroconvulsive Therapy and Hyperventilation: A Narrative Review. Life 2025, 15, 1368. https://doi.org/10.3390/life15091368
Smolarczyk J, Piłat P, Blanch J, Cetnarowska A, Dębski P, Torrent A, Batalla I, Piegza M. Electroconvulsive Therapy and Hyperventilation: A Narrative Review. Life. 2025; 15(9):1368. https://doi.org/10.3390/life15091368
Chicago/Turabian StyleSmolarczyk, Joanna, Patrycja Piłat, Jordi Blanch, Aleksandra Cetnarowska, Paweł Dębski, Aurora Torrent, Iolanda Batalla, and Magdalena Piegza. 2025. "Electroconvulsive Therapy and Hyperventilation: A Narrative Review" Life 15, no. 9: 1368. https://doi.org/10.3390/life15091368
APA StyleSmolarczyk, J., Piłat, P., Blanch, J., Cetnarowska, A., Dębski, P., Torrent, A., Batalla, I., & Piegza, M. (2025). Electroconvulsive Therapy and Hyperventilation: A Narrative Review. Life, 15(9), 1368. https://doi.org/10.3390/life15091368







