Induction of Mutations in Veronica Species by Colchicine Treatment
Abstract
1. Introduction
2. Materials and Methods
2.1. Plant Materials
2.2. Mutation Induction by Chemical Mutagens
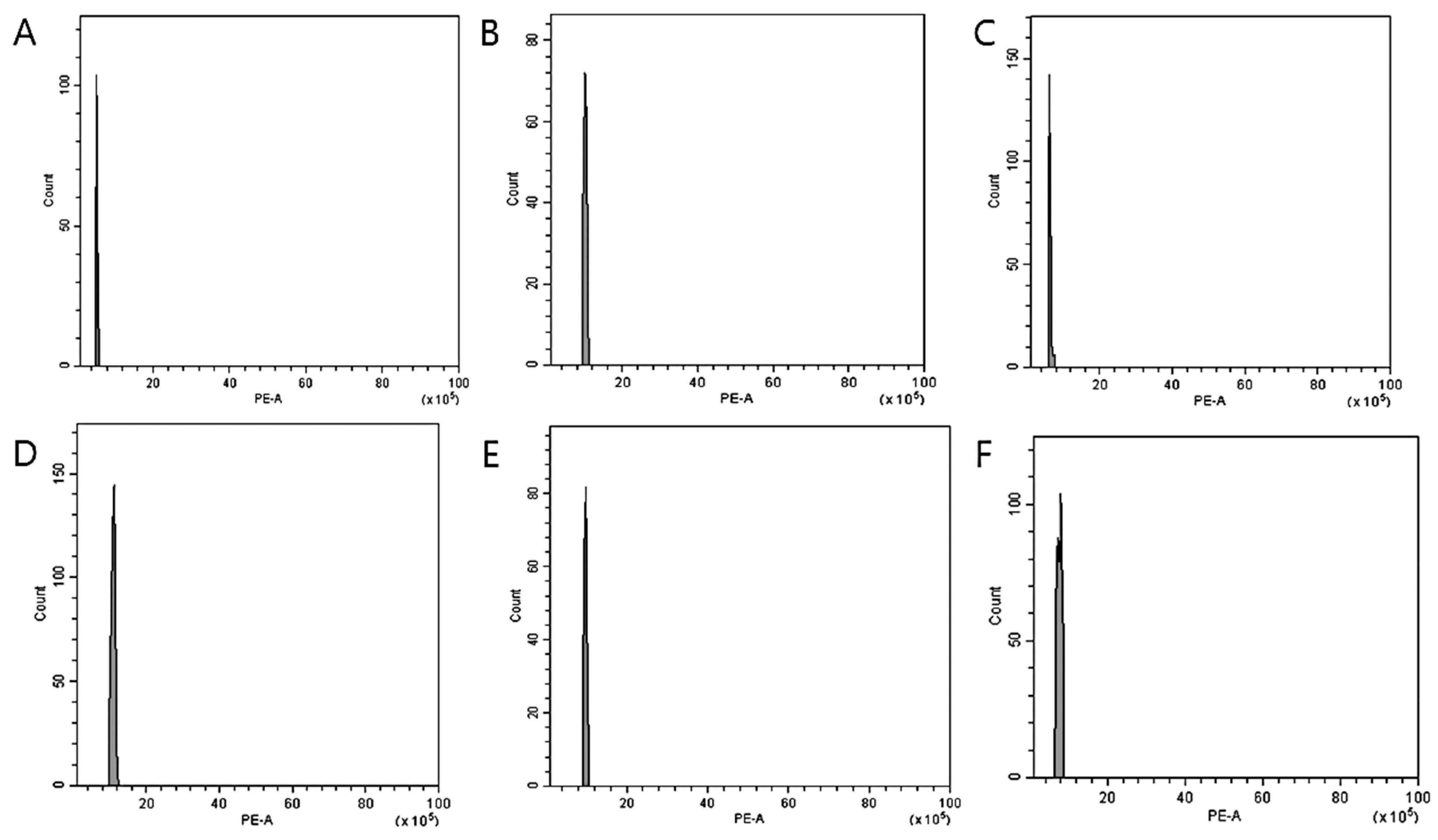
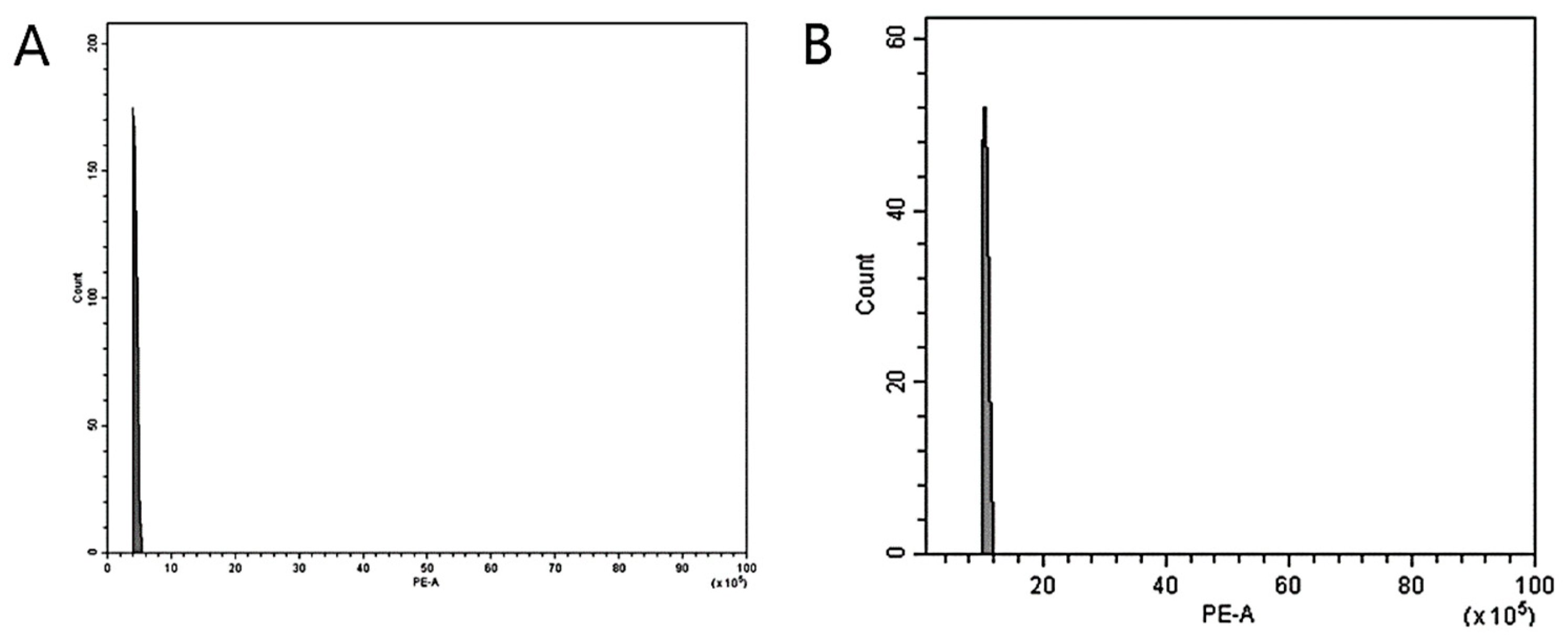
2.3. Flow Cytometry (FCM) Analysis
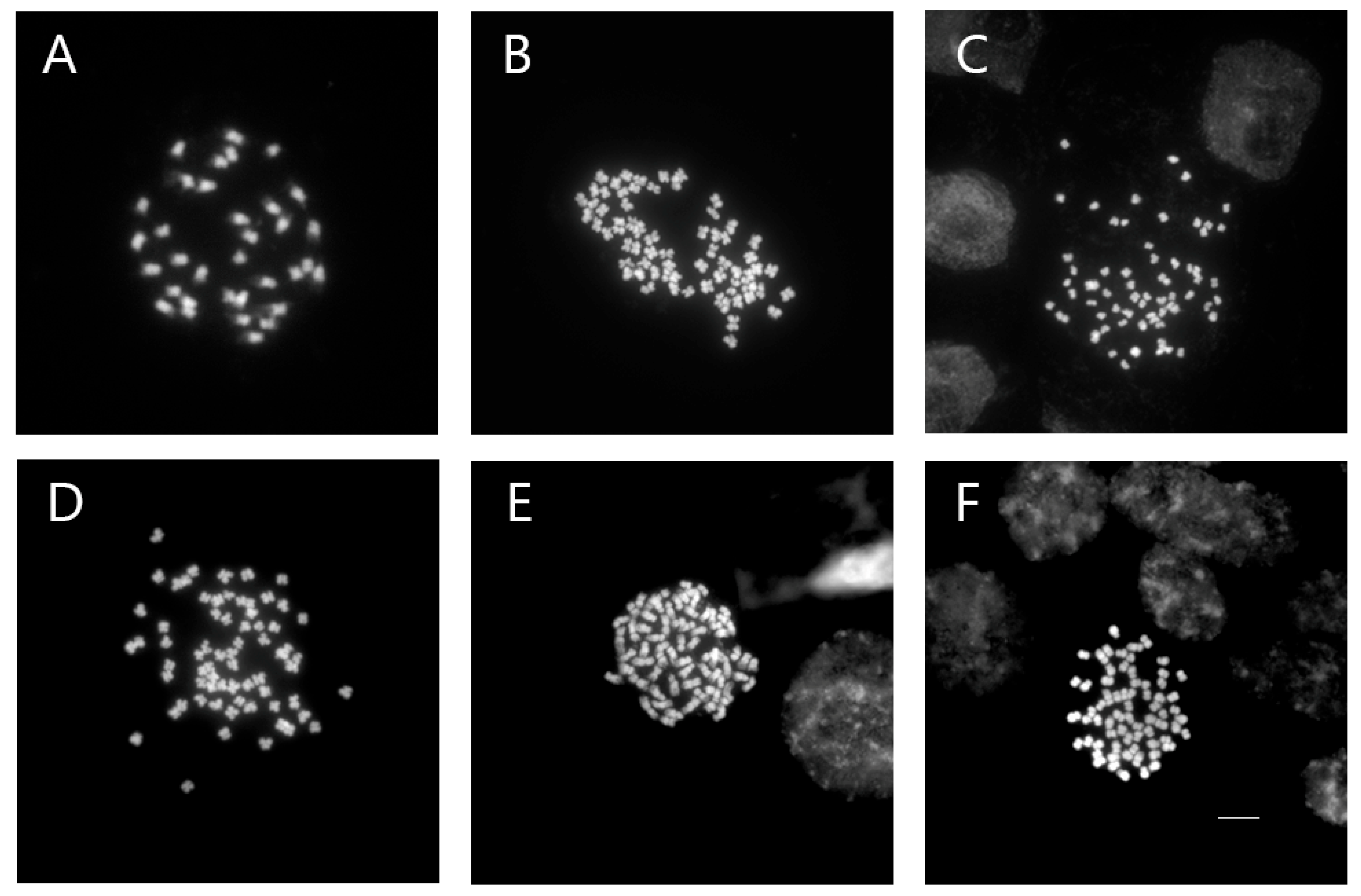
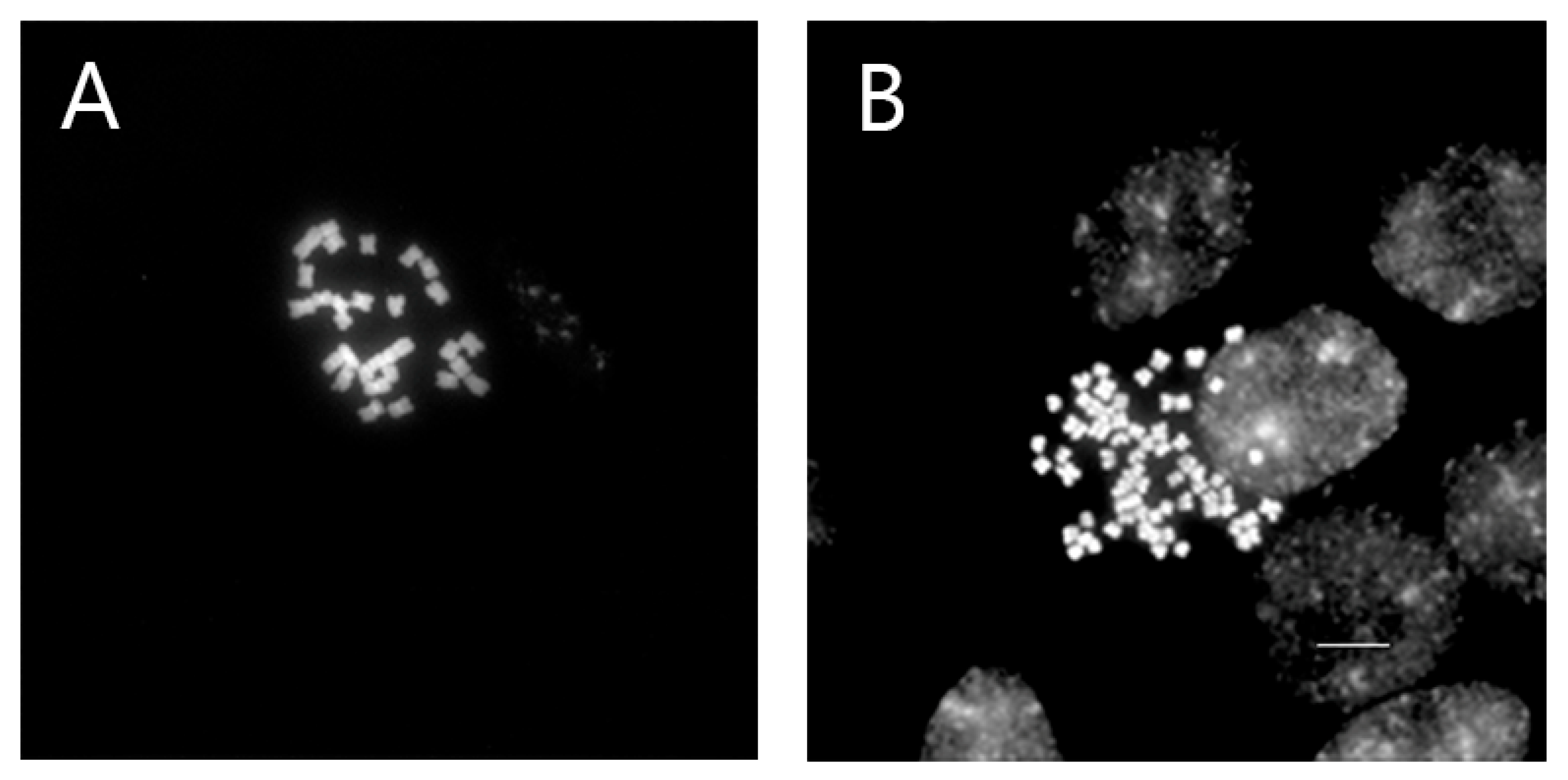
2.4. Chromosome Preparation
2.5. Preparation for Fixation Methods
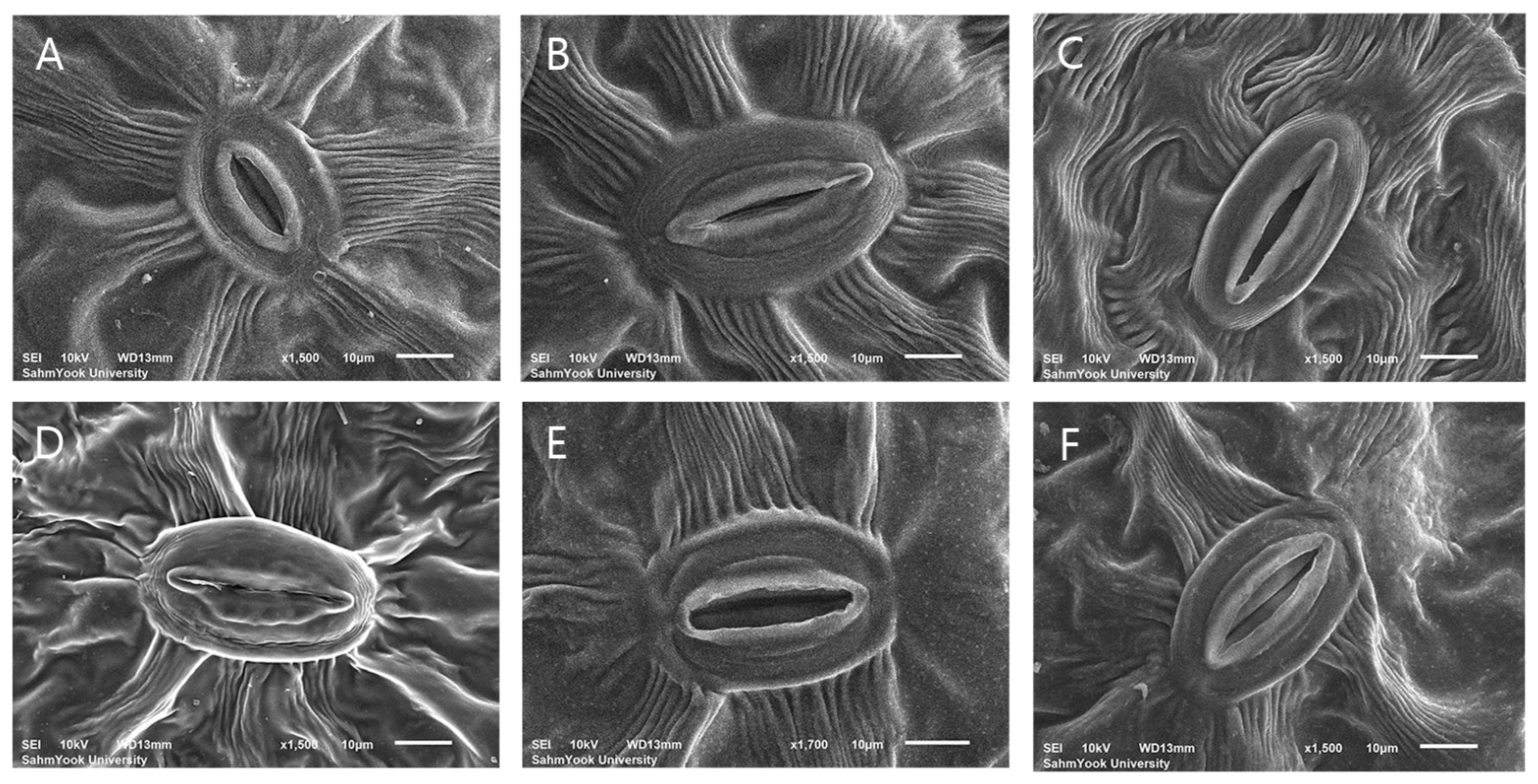
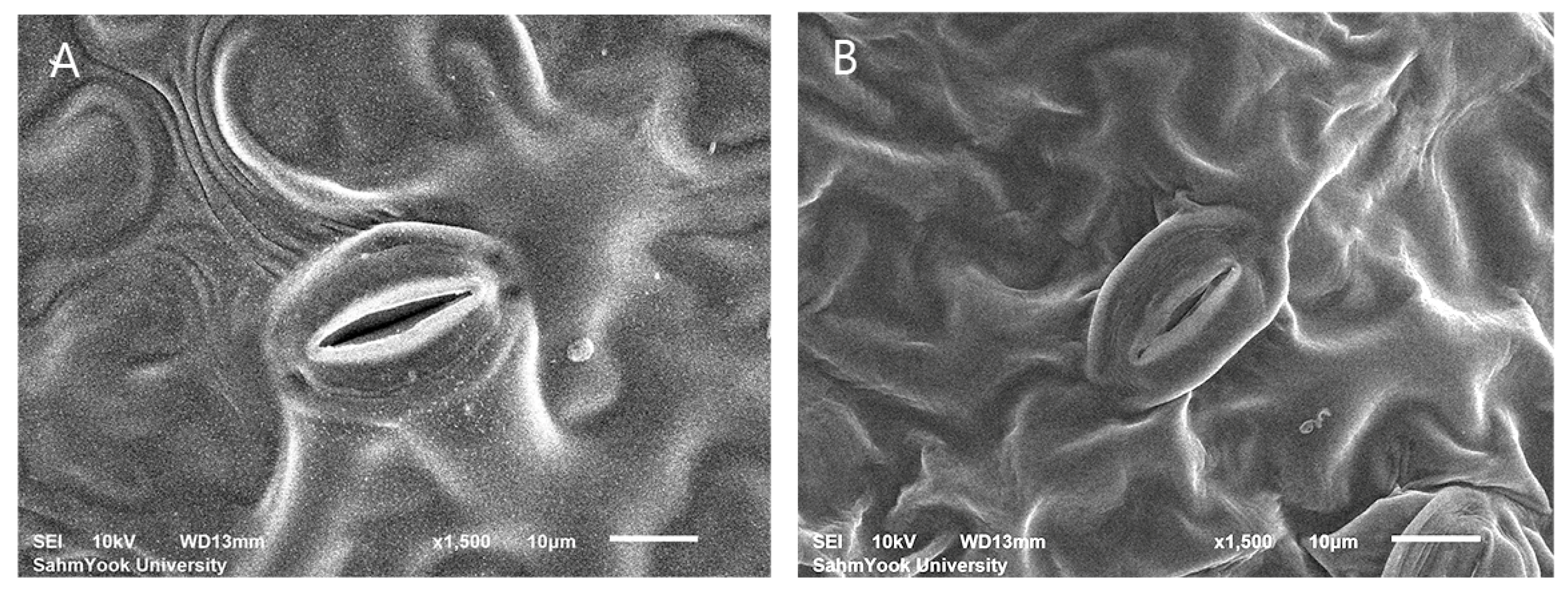
2.6. Stomata Characteristics
2.7. Leaf Morphological Evaluation
2.8. Data Collection and Analysis
3. Results
3.1. Survival and Mutation Rate
3.2. Nuclear DNA Genome Size Estimation
3.3. Chromosome Number Confirmation
3.4. Evaluation of Stomatal Characteristics
3.5. Phenotypic Traits
4. Discussion
4.1. Analysis of Survival and Mutation Rates
4.2. Significance of Cytogenetic Findings
4.3. Stomatal Characteristics
4.4. Leaf Phenotypic Characteristics
5. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
| ANOVA | Analysis of Variance |
| CPD | Critical Point Drying |
| DAPI | 4’, 6-diamidino-2-phenylindole |
| DMRT | Duncan’s Multiple Range Test |
| DNA | Deoxyribonucleic Acid |
| FAA | Formalin Aceto Alcohol |
| FCM | Flow Cytometry |
| n | Haploid Genome |
| PI | Propidium Iodide |
| rDNA | Ribosomal Deoxyribonucleic Acid |
| rpm | Revolutions per Minute |
| RT | Room Temperature |
| SEM | Single Nucleotide Polymorphism |
| SSC | Saline Sodium Citrate |
References
- Hawke, R.G. Comparative Studies of Veronica and Veronicastrum. Plant Evol. Notes 2010, 33, 1–8. Available online: https://citeseerx.ist.psu.edu/document?repid=rep1&type=pdf&doi=3473917bbc02e2e9afd4964f54dd5ebddf38ef97 (accessed on 21 September 2023).
- Olmstead, R.G. Whatever happened to the Scrophulariaceae. Fremontia 2002, 30, 13–22. [Google Scholar]
- Albach, D.C.; Martínez-Ortega, M.M.; Delgado, L.; Weiss-Schneeweiss, H.; Özgökce, F.; Fischer, M.A. Chromosome numbers in Veroniceae (Plantaginaceae): Review and several new Counts. Ann. Mo. Bot. Gard. 2008, 95, 543–566. [Google Scholar] [CrossRef]
- Albach, D.C.; Martínez-Ortega, M.M.; Chase, M.W. Veronica: Parallel morphological evolution and phylogeography in the Mediterranean. Plant Syst. Evol. 2004, 246, 177–194. [Google Scholar] [CrossRef]
- Fischer, E. Scrophulariaceae. In Flowering Plants Dicotyledons: Lamiales (Except Acanthaceae Including Avicenniaceae); Kadereit, J.W., Ed.; Springer: Berlin/Heidelberg, Germany, 2004; Volume 7, pp. 333–432. [Google Scholar] [CrossRef]
- Albach, D.C.; Martínez-Ortega, M.M.; Fischer, M.A.; Chase, M.W. A new classification of the tribe Veroniceae—Problems and a possible solution. Taxon 2004, 53, 429–452. [Google Scholar] [CrossRef]
- Albach, D.C.; Chase, M.W. Paraphyly of Veronica (Veroniceae; Scrophulariaceae): Evidence from the internal transcribed spacer (ITS) sequences of nuclear ribosomal DNA. J. Plant Res. 2001, 114, 9–18. [Google Scholar] [CrossRef]
- Oh, H.J.; Shin, U.S.; Song, S.J.; Kim, J.H.; Kim, S.Y.; Suh, G.U. Growth and flowering characteristics of 20 Veronica species. Flower Res. J. 2019, 27, 308–317. [Google Scholar] [CrossRef]
- Jung, S.Y.; Park, S.H.; Nam, C.H.; Lee, H.J.; Lee, Y.M.; Chang, K.S. The distribution of vascular plants in Ulleungdo and nearby island regions (Gwaneumdo, Jukdo), Korea. J. Asia Pac. Biodivers. 2013, 6, 123–156. [Google Scholar] [CrossRef][Green Version]
- Kwon, H.H.; Oh, H.J.; Cho, W. Germination characteristics and seedling growth of Pseudolysimachion nakaianum (Ohwi) T. Yamaz. seeds according to light quality type. J. People Plants Environ. 2023, 26, 477–485. [Google Scholar] [CrossRef]
- Shin, H.T.; Yi, M.H.; Shin, J.S.; Lee, B.C.; Yoon, J.W. Distribution of rare plants-Ulsan, Busan, Yangsan. J. Korean Nat. 2012, 5, 145–153. [Google Scholar] [CrossRef][Green Version]
- Lee, M.H.; Lim, J.H.; Park, C.H.; Kim, J.H.; Na, C.S. Effect of different temperature regimes on the germination of Pseudolysimachion pusanensis (Y.N.Lee) Y.N.Lee seeds. Horticulturae 2021, 7, 577. [Google Scholar] [CrossRef]
- Lee, S.I.; Yeon, S.H.; Cho, J.S.; Jeong, M.J.; Lee, C.H. Optimization of cultivation conditions on effective seedlings of Veronica rotunda var. subintegra (Nakai) T. Yamaz. J. Bio-Environ. Control 2020, 29, 181–188. [Google Scholar] [CrossRef]
- Park, S.H.; Lee, J.H.; Nam, S.Y. An analysis of the growth and photosynthetic responses of potted Veronica pusanensis Y.N.Lee according to the shading levels. J. People Plants Environ. 2023, 26, 219–231. [Google Scholar] [CrossRef]
- Chung, Y.K.; Kim, Y.J. Analyze the Status of Native Plants in Korea Flower Market and Uses of Floral Decoration. Korean Soc. Flor. Art Des. 2019, 40, 77–96. Available online: https://www.koreascience.kr/article/JAKO201915963899904.page (accessed on 15 February 2019).
- Kim, W.H.; Park, P.H.; Jung, J.A.; Park, K.Y.; Suh, J.N.; Kwon, O.K.; Yoo, B.S.; Lee, S.Y.; Park, P.M.; Choi, Y.J.; et al. Achievement of flower breeding in Korea and its prospects. Korean J. Breed. Sci. 2020, 52, 161–169. [Google Scholar] [CrossRef]
- Pathirana, R. Plant mutation breeding in agriculture. CAB Rev. 2011, 6, 1–20. [Google Scholar] [CrossRef]
- Roychowdhury, R.; Tah, J. Mutagenesis—A potential approach for crop improvement. In Crop Improvement: New Approaches and Modern Techniques; Hakeem, K.R., Ahmad, P., Ozturk, M., Eds.; Springer: Boston, MA, USA, 2013; pp. 149–187. [Google Scholar] [CrossRef]
- Ascough, G.D.; Van Staden, J.; Erwin, J.E. Effectiveness of colchicine and oryzalin at inducing polyploidy in Watsonia lepida N.E. Brown. Hort. Sci. 2008, 43, 2248–2251. [Google Scholar] [CrossRef]
- Chaudhary, J.; Alisha, A.; Bhatt, V.; Chandanshive, S.; Kumar, N.; Mir, Z.; Kumar, A.; Yadav, S.K.; Shivaraj, S.M.; Sonah, H.; et al. Mutation breeding in tomato: Advances, applicability and challenges. Plants 2019, 8, 128. [Google Scholar] [CrossRef] [PubMed]
- Oladosu, Y.; Rafii, M.Y.; Abdullah, N.; Hussin, G.; Ramli, A.; Rahim, H.A.; Miah, G.; Usman, M. Principle and application of plant mutagenesis in crop improvement: A review. Biotechnol. Biotechnol. Equip. 2016, 30, 1–16. [Google Scholar] [CrossRef]
- Kumar, M.K.; Rani, M.U. Colchiploidy in Fruit Breeding-A review. Int. J. Sci. Res. 2013, 2, 325–326. [Google Scholar]
- Sattler, M.C.; Carvalho, C.R.; Clarindo, W.R. The polyploidy and its key role in plant breeding. Planta 2016, 243, 281–296. [Google Scholar] [CrossRef] [PubMed]
- El-Nashar, Y.I.; Ammar, M.H. Mutagenic influences of colchicine on phenological and molecular diversity of Calendula officinalis L. Genet. Mol. Res. 2016, 15, 1–15. [Google Scholar] [CrossRef]
- Cabahug, R.A.M.; Khanh, H.T.T.M.; Lim, K.B.; Hwang, Y.J. Phenotype and ploidy evaluation of colchicine-induced Echeveria ‘Peerless’. Toxicol. Environ. Health Sci. 2021, 13, 17–24. [Google Scholar] [CrossRef]
- Doležel, J.; Greilhuber, J.; Suda, J. Estimation of nuclear DNA content in plants using flow cytometry. Nat. Protoc. 2007, 2, 2233–2244. [Google Scholar] [CrossRef]
- Ha Tran, M.K.T.; Cabahug-Braza, R.A.M.; Hwang, Y.J. Echeveria Leaf morpho-anatomical analysis and its implications for environmental stress conditions. Horticulturae 2024, 10, 308. [Google Scholar] [CrossRef]
- Park, I.S.; Cho, K.J.; Kim, J.; Cho, J.Y.; Lim, T.J.; Oh, W. Growth and flowering responses of petunia to various artificial light sources with different light qualities. Hortic. Sci. Technol. 2016, 34, 55–66. [Google Scholar] [CrossRef]
- Lee, J.H.; Nam, S.Y. Effects of temperature treatments on the growth and flowering characteristics of Kalanchoe pumila Baker. Flower Res. J. 2023, 31, 92–100. [Google Scholar] [CrossRef]
- Yun, J.H.; Jeong, H.W.; Hwang, S.Y.; Yu, J.; Hwang, H.S.; Hwang, S.J. Growth of cucumber and tomato seedlings by different light intensities and CO2 concentrations in closed-type plant production system. J. Bio-Env. Con. 2023, 32, 257–266. [Google Scholar] [CrossRef]
- Encina, C.L.; Regalado, J.J. Aspects of in vitro plant tissue culture and breeding of Asparagus: A review. Horticulturae 2022, 8, 439. [Google Scholar] [CrossRef]
- EFSA Panel on Genetically Modified Organisms (GMO); Mullins, E.; Bresson, J.L.; Dalmay, T.; Dewhurst, I.C.; Epstein, M.M.; Firbank, L.G.; Guerche, P.; Hejatko, J.; Moreno, F.J.; et al. In vivo and in vitro random mutagenesis techniques in plants. EFSA J. 2021, 19, e06611. [Google Scholar] [CrossRef]
- Schum, A.; Preil, W. Induced Mutations in Ornamental Plants, Somaclonal Variation and Induced Mutations in Crop Improvement; Springer: Berlin/Heidelberg, Germany, 1998; pp. 333–366. [Google Scholar] [CrossRef]
- Yue, F.; Yunhui, R.; Xiuyu, L.; Yuhan, S.; Yongping, D.; Jihong, Z.; Hong, Y.; Yun, L. Artificial induction of polyploid Robinia pseudoacacia L. and identification of its ploidy. Mol. Plant Breed. 2018, 16, 7124–7131. [Google Scholar] [CrossRef]
- Curry, H.A. Making marigolds: Colchicine, Mutation Breeding, and Ornamental Horticulture, 1937–1950. Identifying Mutat. 1938, 29, 259. [Google Scholar]
- Omidbaigi, R.; Mirzaee, M.; Hassani, M.E.; Sedghi Moghadam, M. Induction and identification of polyploidy in basil (Ocimum basilicum L.) medicinal plant by colchicine treatment. Int. J. Plant Prod. 2010, 4, 87–98. [Google Scholar] [CrossRef]
- Urwin, N.A.R. Generation and characterisation of colchicine-induced polyploid Lavandula × intermedia. Euphytica 2014, 197, 331–339. [Google Scholar] [CrossRef]
- Mo, L.; Chen, J.; Lou, X.; Xu, Q.; Dong, R.; Tong, Z.; Huang, H.; Lin, E. Colchicine-induced polyploidy in Rhododendron fortunei Lindl. Plants 2020, 9, 424. [Google Scholar] [CrossRef]
- Cabahug, R.A.M.; Tran, M.K.T.H.; Ahn, Y.J.; Hwang, Y.J. Retention of mutations in colchicine-induced ornamental succulent Echeveria “Peerless”. Plants 2022, 11, 3420. [Google Scholar] [CrossRef]
- Mahna, S.K.; Garg, R.E.K.H.A.; Parvateesam, M. Mutagenic Effects of Sodium Azide on Black Gram (Phaseolus mungo L.). Curr. Sci. 1989, 58, 582–584. Available online: https://www.cabidigitallibrary.org/doi/full/10.5555/19911621028 (accessed on 15 June 1991).
- Singh, B.; Yun, S.; Gil, Y.; Park, M.H. The Role of Colchicine in Plant Breeding. Int. J. Mol. Sci. 2025, 26, 6743. [Google Scholar] [CrossRef]
- Corneillie, S.; De Storme, N.; Van Acker, R.; Fangel, J.U.; De Bruyne, M.; De Rycke, R.; Geelen, D.; Willats, W.G.T.; Vanholme, B.; Boerjan, W. Polyploidy affects plant growth and alters cell wall composition. Plant Physiol. 2019, 179, 74–87. [Google Scholar] [CrossRef]
- Eeckhaut, T.; Leus, L.; Van Huylenbroeck, J. Exploitation of flow cytometry for plant breeding. Acta Physiol. Plant. 2005, 27, 743–750. [Google Scholar] [CrossRef]
- Guerra, M. Chromosome numbers in plant cytotaxonomy: Concepts and implications. Cytogenet. Genome Res. 2008, 120, 339–350. [Google Scholar] [CrossRef] [PubMed]
- Van Duren, M.; Morpurgo, R.; Dolezel, J.; Afza, R. Induction and verification of autotetraploids in diploid banana (Musa acuminata) by in vitro techniques. Euphytica 1996, 88, 25–34. [Google Scholar] [CrossRef]
- Lin, X.; Zhou, Y.; Zhang, J.; Lu, X.; Zhang, F.; Shen, Q.; Wu, S.; Chen, Y.; Wang, T.; Tang, K. Enhancement of artemisinin content in tetraploid Artemisia annua plants by modulating the expression of genes in artemisinin biosynthetic pathway. Biotechnol. Appl. Biochem. 2011, 58, 50–57. [Google Scholar] [CrossRef] [PubMed]
- Masterson, J. Stomatal size in fossil plants: Evidence for polyploidy in majority of angiosperms. Science 1994, 264, 421–424. [Google Scholar] [CrossRef]
- Münzbergová, Z. Colchicine application significantly affects plant performance in the second generation of synthetic polyploids and its effects vary between populations. Ann. Bot. 2017, 120, 329–339. [Google Scholar] [CrossRef]
- Moghbel, N.; Borujeni, M.K.; Bernard, F. Colchicine effect on the DNA content and stomata size of Glycyrrhiza glabra var. glandulifera and Carthamus tinctorius L. cultured in vitro. J. Genet. Eng. Biotechnol. 2015, 13, 1–6. [Google Scholar] [CrossRef]
- Yang, X.M.; Cao, Z.Y.; An, L.Z.; Wang, Y.M.; Fang, X.W. In vitro tetraploid induction via colchicine treatment from diploid somatic embryos in grapevine (Vitis vinifera L.). Euphytica 2006, 152, 217–224. [Google Scholar] [CrossRef]
- Yeung, E.C.; Chan, C.K.W. The glycol methacrylate embedding resins—Technovit 7100 and 8100. In Plant Microtechniques and Protocols; Yeung, E.C.T., Stasolla, C., Sumner, M.J., Huang, B.Q., Eds.; Springer International Publishing: Cham, Switzerland, 2015; pp. 67–82. [Google Scholar] [CrossRef]
- Huang, B.Q.; Yeung, E.C. Chemical and physical fixation of cells and tissues: An overview. In Plant Microtechniques and Protocols; Yeung, E.C.T., Stasolla, C., Sumner, M.J., Huang, B.Q., Eds.; Springer: Cham, Switzerland, 2015; Volume 1, pp. 23–43. [Google Scholar] [CrossRef]
- Yuan, J.; Wang, X.; Zhou, H.; Li, Y.; Zhang, J.; Yu, S.; Wang, M.; Hao, M.; Zhao, Q.; Liu, L.; et al. Comparison of sample preparation techniques for inspection of leaf epidermises using light microscopy and scanning electronic microscopy. Front. Plant Sci. 2020, 11, 133. [Google Scholar] [CrossRef]
- Pathan, A.K.; Bond, J.; Gaskin, R.E. Sample preparation for SEM of plant surfaces. Mater. Today 2010, 12, 32–43. [Google Scholar] [CrossRef]
- Leshcheniuk, O.; Chipilyak, T. Morpho-anatomical adaptation of the leaves of certain Veronica species to arid conditions. Plant Introduction 2020, 87–88, 47–53. [Google Scholar] [CrossRef]
- Neinhuis, C.; Edelmann, H.G. Methanol as a rapid fixative for the investigation of plant surfaces by SEM. J. Microsc. 1996, 184, 14–16. [Google Scholar] [CrossRef]
- Ha Tran, M.K.T.H.; Cabahug-Braza, R.A.M.; Lim, K.B.; Hwang, Y.J. Leaf morpho-anatomical evaluation of echeveria cultivars using different fixation methods. Hortic. Sci. Technol. 2024, 42, 127–142. [Google Scholar] [CrossRef]
- Chung, M.Y.; Kim, C.Y.; Min, J.S.; Lee, D.J.; Naing, A.H.; Chung, J.D.; Kim, C.K. In vitro induction of tetraploids in an interspecific hybrid of Calanthe (Calanthe discolor × Calanthe sieboldii) through colchicine and oryzalin treatments. Plant Biotechnol. Rep. 2014, 8, 251–257. [Google Scholar] [CrossRef]
- Hassanzadeh, F.; Zakaria, R.A.; Azad, N.H. Polyploidy induction in Salvia officinalis L. and its effects on some morphological and physiological characteristics. Cytologia 2020, 85, 157–162. [Google Scholar] [CrossRef]
- Wei, K.H.; Xu, J.P.; Li, L.X.; Cai, J.Y.; Miao, J.H.; Li, M.H. In vitro induction and generation of tetraploid plants of Sophora tonkinensis Gapnep. Pharmacogn. Mag. 2018, 14, 149–154. [Google Scholar] [CrossRef]
- Seneviratn, K.A.C.N.; Wijesundar, D.S.A. First African violets (Saintpaulia ionantha, H. Wendl.) with a changing colour pattern induced by mutation. Am. J. Plant Physiol. 2007, 2, 233–236. [Google Scholar] [CrossRef][Green Version]
- Kushwah, K.S.; Verma, R.C.; Patel, S.; Jain, N.K. Colchicine induced polyploidy in Chrysanthemum carinatum L. J. Phylogenet. Evol. Biol. 2018, 6, 2. [Google Scholar] [CrossRef]
- Pan, I.C.; Lu, Y.F.; Wen, P.J.; Chen, Y.M. Using colchicine to create poinsettia (Euphorbia pulcherrima × Euphorbia cornastra) mutants with various morphological traits. HortScience 2019, 54, 1667–1672. [Google Scholar] [CrossRef]
- Samatadze, T.E.; Yurkevich, O.Y.; Khazieva, F.M.; Basalaeva, I.V.; Konyaeva, E.A.; Burova, A.E.; Zoshchuk, S.A.; Morozov, A.I.; Amosova, A.V.; Muravenko, O.V. Agro-morphological and cytogenetic characterization of colchicine-induced tetraploid plants of Polemonium caeruleum L. (Polemoniaceae). Plants 2022, 11, 2585. [Google Scholar] [CrossRef]




| Code | Treatment | V. nakaiana Ohwi | V. pusanensis Y.N.Lee | ||
|---|---|---|---|---|---|
| Survival Rate (%) | Mutant Rate (%) | Survival Rate (%) | Mutant Rate (%) | ||
| T0 | Control | 40 (40.0) | - | 20 (20.0) | - |
| T1 | 0.2% + 1 h | 30 (30.0) | 2 (2.0) | 43 (43.0) | 1 (1.0) |
| T2 | 0.2% + 2 h | 19 (19.0) | 1 (1.0) | 43 (43.0) | 1 (1.0) |
| T3 | 0.2% + 3 h | 29 (29.0) | 1 (1.0) | 33 (33.0) | 1 (1.0) |
| T4 | 0.2% + 4 h | 25 (25.0) | 2 (2.0) | 26 (26.0) | 0 (0.0) |
| T5 | 0.2% + 5 h | 14 (14.0) | 2 (2.0) | 29 (29.0) | 1 (1.0) |
| T6 | 0.4% + 1 h | 29 (29.0) | 1 (1.0) | 35 (35.0) | 2 (2.0) |
| T7 | 0.4% + 2 h | 26 (26.0) | 2 (2.0) | 14 (14.0) | 0 (0.0) |
| T8 | 0.4% + 3 h | 19 (19.0) | 3 (3.0) | 22 (22.0) | 0 (0.0) |
| T9 | 0.4% + 4 h | 18 (18.0) | 2 (2.0) | 27 (27.0) | 0 (0.0) |
| T10 | 0.4% + 5 h | 15 (15.0) | 0 (0.0) | 17 (17.0) | 0 (0.0) |
| T11 | 0.6% + 1 h | 16 (16.0) | 2 (2.0) | 35 (35.0) | 2 (2.0) |
| T12 | 0.6% + 2 h | 11 (11.0) | 1 (1.0) | 31 (31.0) | 0 (0.0) |
| T13 | 0.6% + 3 h | 15 (15.0) | 2 (2.0) | 15 (15.0) | 0 (0.0) |
| T14 | 0.6% + 4 h | 11 (11.0) | 0 (0.0) | 21 (21.0) | 0 (0.0) |
| T15 | 0.6% + 5 h | 11 (11.0) | 0 (0.0) | 16 (16.0) | 0 (0.0) |
| T16 | 0.8% + 1 h | 14 (14.0) | 1 (1.0) | 18 (18.0) | 0 (0.0) |
| T17 | 0.8% + 2 h | 14 (14.0) | 1 (1.0) | 21 (21.0) | 0 (0.0) |
| T18 | 0.8% + 3 h | 9 (9.0) | 0 (0.0) | 9 (9.0) | 0 (0.0) |
| T19 | 0.8% + 4 h | 14 (14.0) | 1 (1.0) | 18 (18.0) | 0 (0.0) |
| T20 | 0.8% + 5 h | 6 (6.0) | 1 (1.0) | 15 (15.0) | 0 (0.0) |
| T21 | 1.0% + 1 h | 10 (10.0) | 0 (0.0) | 10 (10.0) | 0 (0.0) |
| T22 | 1.0% + 2 h | 16 (16.0) | 0 (0.0) | 37 (37.0) | 0 (0.0) |
| T23 | 1.0% + 3 h | 7 (7.0) | 1 (1.0) | 19 (19.0) | 1 (1.0) |
| T24 | 1.0% + 4 h | 14 (14.0) | 1 (1.0) | 18 (18.0) | 0 (0.0) |
| T25 | 1.0% + 5 h | 5 (5.0) | 0 (0.0) | 24 (24.0) | 0 (0.0) |
| Total | 397 (15.9) | 27 (6.8) | 596 (23.8) | 9 (1.5) | |
| Colchicine Treatments | Chromosome Number |
|---|---|
| V. nakaiana Ohwi | |
| Control | 2n = 34 |
| T8-P1 (0.4% + 3 h) | 2n = 68 |
| T8-P2 (0.4% + 3 h) | 2n = 67 |
| T8-P3 (0.4% + 3 h) | 2n = 68 |
| T11-P5 (0.6% + 1 h) | 2n = 68 |
| T23-P2 (1.0% + 3 h) | 2n = 68 |
| V. pusanensis Y.N.Lee | |
| Control | 2n = 34 |
| T11-P4 (0.6% + 1 h) | 2n = 66 |
| Colchicine Treatments | Length of Stomata (µm) | Diameter of Stomata (µm) | Stomatal Density (mm2) |
|---|---|---|---|
| V. nakaiana Ohwi | |||
| Control | 30.59 ± 1.52 x a y | 22.97 ± 1.68 bc | 151.33 ± 16.53 b |
| T8-P1 (0.4% + 3 h) | 46.53 ± 2.90 e | 28.78 ± 1.65 e | 87.07 ± 15.73 a |
| T8-P2 (0.4% + 3 h) | 34.30 ± 3.88 b | 18.96 ± 1.43 a | 182.42 ± 33.06 d |
| T8-P3 (0.4% + 3 h) | 36.14 ± 3.12 b | 22.27 ± 2.38 b | 178.28 ± 33.06 bc |
| T11-P5 (0.6% + 1 h) | 38.63 ± 2.04 c | 24.49 ± 2.25 cd | 95.36 ± 18.72 a |
| T23-P2 (1.0% + 3 h) | 43.28 ± 1.59 d | 25.06 ± 1.92 d | 70.48 ± 10.15 a |
| F-test | ** | ** | ** |
| V. pusanensis Y.N.Lee | |||
| Control | 28.36 ± 3.51 | 19.00 ± 2.29 | 145.11 ± 12.84 |
| T11-P4 (0.6% + 1 h) | 28.87 ± 1.93 | 19.85 ± 1.76 | 190.71 ± 18.72 |
| T-test | NS | NS | ** |
| Colchicine Treatments | Leaf Measurement | ||
|---|---|---|---|
| Length (mm) | Width (mm) | Area (mm2) | |
| Control | 35.53 ± 0.14 x a y | 46.75 ± 0.26 a | 1661.43 ± 13.54 a |
| T8-P1 (0.4% + 3 h) | 62.30 ± 0.12 c | 63.75 ± 0.12 b | 3971.62 ± 0.17 c |
| T8-P2 (0.4% + 3 h) | 58.05 ± 0.19 b | 64.23 ± 0.53 b | 3728.88 ± 21.54 b |
| T8-P3 (0.4% + 3 h) | 71.99 ± 0.95 d | 90.17 ± 0.50 c | 6491.88 ± 115.50 d |
| F-test | ** | ** | ** |
| Colchicine Treatments | Leaf Measurement | ||
|---|---|---|---|
| Length (mm) | Width (mm) | Area (mm2) | |
| Control | 38.36 ± 0.09 x | 47.90 ± 0.34 | 1837.60 ± 13.04 |
| T11-P4 (0.6% + 1 h) | 45.30 ± 0.40 | 48.15 ± 0.42 | 2181.29 ± 37.00 |
| T-test | ** | NS | ** |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Park, H.-W.; Sevilleno, S.S.; Yi, J.-H.; Cho, W.; Kim, Y.-J.; Hwang, Y.-J. Induction of Mutations in Veronica Species by Colchicine Treatment. Life 2025, 15, 1367. https://doi.org/10.3390/life15091367
Park H-W, Sevilleno SS, Yi J-H, Cho W, Kim Y-J, Hwang Y-J. Induction of Mutations in Veronica Species by Colchicine Treatment. Life. 2025; 15(9):1367. https://doi.org/10.3390/life15091367
Chicago/Turabian StylePark, Hye-Wan, Samantha Serafin Sevilleno, Ji-Hun Yi, Wonwoo Cho, Young-Jae Kim, and Yoon-Jung Hwang. 2025. "Induction of Mutations in Veronica Species by Colchicine Treatment" Life 15, no. 9: 1367. https://doi.org/10.3390/life15091367
APA StylePark, H.-W., Sevilleno, S. S., Yi, J.-H., Cho, W., Kim, Y.-J., & Hwang, Y.-J. (2025). Induction of Mutations in Veronica Species by Colchicine Treatment. Life, 15(9), 1367. https://doi.org/10.3390/life15091367







