First-in-Human Application of Very High-Power Short-Duration RF Ablation for Refractory AVNRT: A Case Report
Abstract
1. Introduction
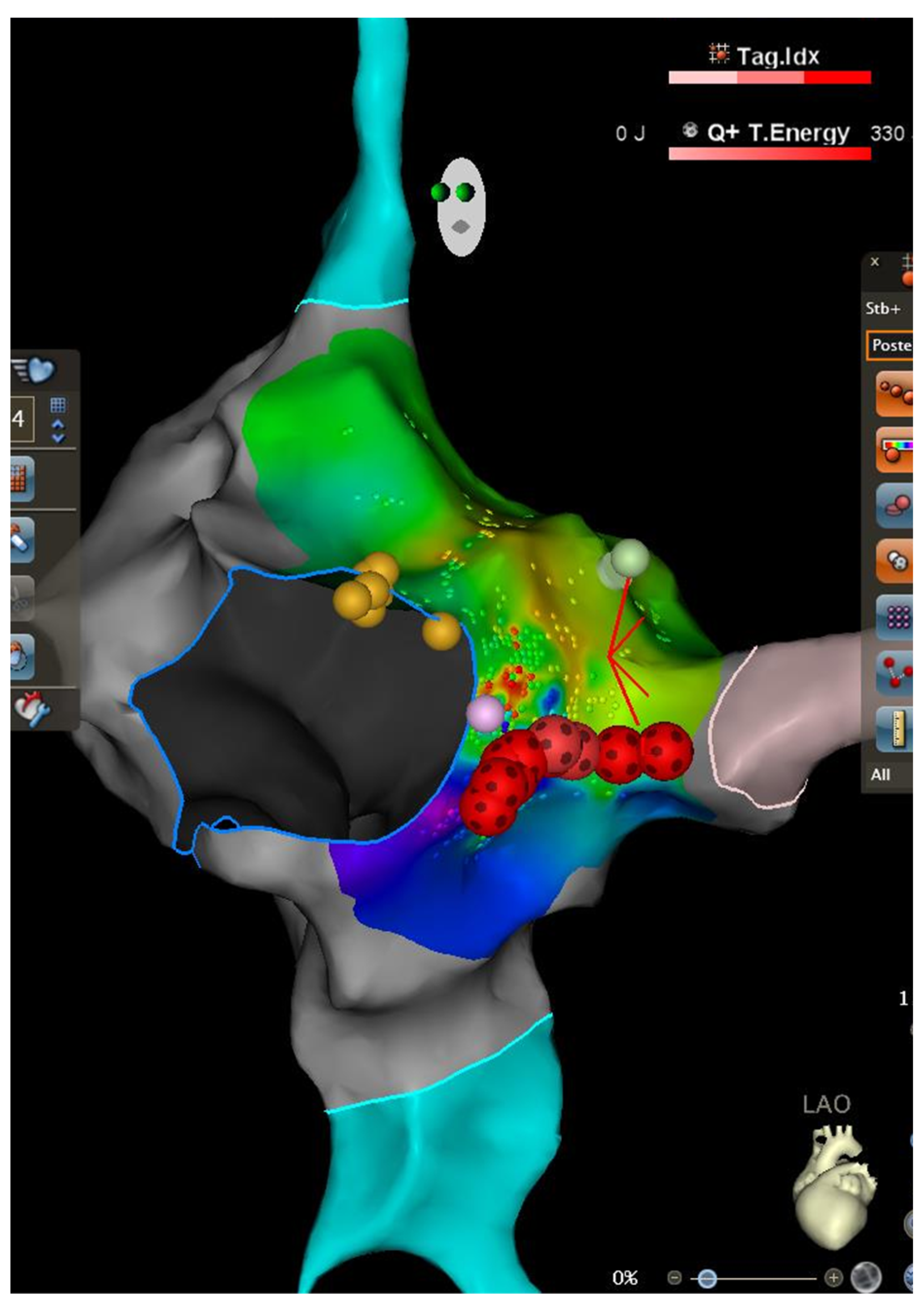
2. Case Description
3. Discussion
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Blomström-Lundqvist, C.; Scheinman, M.M.; Smith, S.C., Jr.; Priori, S.G. ACC/AHA/ESC guidelines for management of supraventricular arrhythmias. Circulation 2003, 108, 1871–1909. [Google Scholar] [CrossRef] [PubMed]
- Katritsis, D.G.; Anderson, R.H. New insights into the mechanisms of fast and slow conduction in the atrioventricular node. Heart Rhythm. 2023, 20, 627–630. [Google Scholar] [CrossRef] [PubMed]
- Do, U.; Nam, G.B.; Cho, M.S.; Kim, J.; Choi, K.J. Role of electrograms recorded from mini electrodes in catheter ablation of atrioventricular nodal reentrant tachycardia: A retrospective study. BMC Cardiovasc. Disord. 2025, 25, 461. [Google Scholar] [CrossRef] [PubMed]
- Chrispin, J.; Misra, S.; Marine, J.E.; Rickard, J.; Barth, A.; Kolandaivelu, A.; Ashikaga, H.; Tandri, H.; Spragg, D.D.; Crosson, J.; et al. Current management and clinical outcomes for catheter ablation of atrioventricular nodal reentrant tachycardia. Europace 2018, 20, e51–e59. [Google Scholar] [CrossRef] [PubMed]
- Hanninen, M.; Yeung-Lai-Wah, N.; Massel, D.; Gula, L.J.; Skanes, A.C.; Yee, R.; Klein, G.J.; Manlucu, J.; Leong-Sit, P. Cryoablation versus RF ablation for AVNRT: A meta-analysis and systematic review. J. Cardiovasc. Electrophysiol. 2013, 24, 1354–1360. [Google Scholar] [CrossRef] [PubMed]
- Katritsis, D.G.; John, R.M.; Latchamsetty, R.; Zografos, T.; Katritsis, G.D.; Stevenson, W.G.; Efimov, I.R.; Morady, F. Left Septal Slow Pathway Ablation for Atrioventricular Nodal Reentrant Tachycardia. Circ. Arrhythm. Electrophysiol. 2018, 11, e005907. [Google Scholar] [CrossRef] [PubMed]
- Wang, N.C. Catheter ablation via the left atrium for atrioventricular nodal reentrant tachycardia: A narrative review. Heart Rhythm. O2 2021, 2, 187–200. [Google Scholar] [CrossRef] [PubMed]
- Schwagten, B.; Van Belle, Y.; Jordaens, L. Cryoablation: How to improve results in atrioventricular nodal reentrant tachycardia ablation? Europace 2010, 12, 1522–1525. [Google Scholar] [CrossRef] [PubMed]
- Insulander, P.; Bastani, H.; Braunschweig, F.; Drca, N.; Kennebäck, G.; Schwieler, J.; Tapanainen, J.; Jensen-Urstad, M. Cryoablation of atrioventricular nodal reentrant tachycardia: 7-year follow-up in 515 patients—Confirmed safety but very late recurrences occur. Europace 2017, 19, 1038–1042. [Google Scholar] [CrossRef] [PubMed]
- Leshem, E.; Zilberman, I.; Tschabrunn, C.M.; Barkagan, M.; Contreras-Valdes, F.M.; Govari, A.; Anter, E. High-Power and Short-Duration Ablation for Pulmonary Vein Isolation: Biophysical Characterization. JACC Clin. Electrophysiol. 2018, 4, 467–479. [Google Scholar] [CrossRef] [PubMed]
- Teumer, Y.; Ziemssen, H.; Katov, L.; Bothner, C.; Mayer, B.; Rottbauer, W.; Weinmann-Emhardt, K. Comparative lesion metrics analysis of very high-power and high-power short-duration radiofrequency ablation in a Porcine ex vivo model. Sci. Rep. 2025, 15, 20215. [Google Scholar] [CrossRef] [PubMed]
- Chen, S. Very high-power short duration 90 W/4 s (vHPSD) vs. vHPSD-combined ablation index–guided 50 W ablation (hybrid) approach for pulmonary vein isolation in treating atrial fibrillation: Have we found the best radiofrequency recipe? J. Interv. Card. Electrophysiol. 2024, 67, 1483–1485. [Google Scholar] [CrossRef] [PubMed]
- Reddy, V.Y.; Grimaldi, M.; De Potter, T.; Vijgen, J.M.; Bulava, A.; Duytschaever, M.F.; Martinek, M.; Natale, A.; Knecht, S.; Neuzil, P.; et al. Pulmonary Vein Isolation with Very High Power, Short Duration, Temperature-Controlled Lesions: The QDOT-FAST Trial. JACC Clin. Electrophysiol. 2019, 5, 778–786. [Google Scholar] [CrossRef] [PubMed]
- Nakagawa, H.; Ikeda, A.; Sharma, T.; Govari, A.; Ashton, J.; Maffre, J.; Lifshitz, A.; Fuimaono, K.; Yokoyama, K.; Wittkampf, F.H.M.; et al. Comparison of in vivo tissue temperature profile and lesion geometry for radiofrequency ablation with high power-short duration and moderate power-moderate duration effects of thermal latency and contact force on lesion formation. Circ. Arrhythm. Electrophysiol. 2021, 14, e009899. [Google Scholar] [CrossRef] [PubMed]

Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2025 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Babic, M.; Tomovic, M.; Vukajlovic, D.; Zugic, V.; Grbovic, A.; Petrovic, M.; Bojic, M.; Nikolic, A. First-in-Human Application of Very High-Power Short-Duration RF Ablation for Refractory AVNRT: A Case Report. Life 2025, 15, 1834. https://doi.org/10.3390/life15121834
Babic M, Tomovic M, Vukajlovic D, Zugic V, Grbovic A, Petrovic M, Bojic M, Nikolic A. First-in-Human Application of Very High-Power Short-Duration RF Ablation for Refractory AVNRT: A Case Report. Life. 2025; 15(12):1834. https://doi.org/10.3390/life15121834
Chicago/Turabian StyleBabic, Milos, Milosav Tomovic, Dejan Vukajlovic, Vasko Zugic, Aleksandra Grbovic, Masa Petrovic, Milovan Bojic, and Aleksandra Nikolic. 2025. "First-in-Human Application of Very High-Power Short-Duration RF Ablation for Refractory AVNRT: A Case Report" Life 15, no. 12: 1834. https://doi.org/10.3390/life15121834
APA StyleBabic, M., Tomovic, M., Vukajlovic, D., Zugic, V., Grbovic, A., Petrovic, M., Bojic, M., & Nikolic, A. (2025). First-in-Human Application of Very High-Power Short-Duration RF Ablation for Refractory AVNRT: A Case Report. Life, 15(12), 1834. https://doi.org/10.3390/life15121834




