Genetic Association and Differential RNA Expression of Histone (De)Acetylation-Related Genes in Pemphigus Foliaceus—A Possible Epigenetic Effect in the Autoimmune Response
Abstract
:1. Introduction
2. Materials and Methods
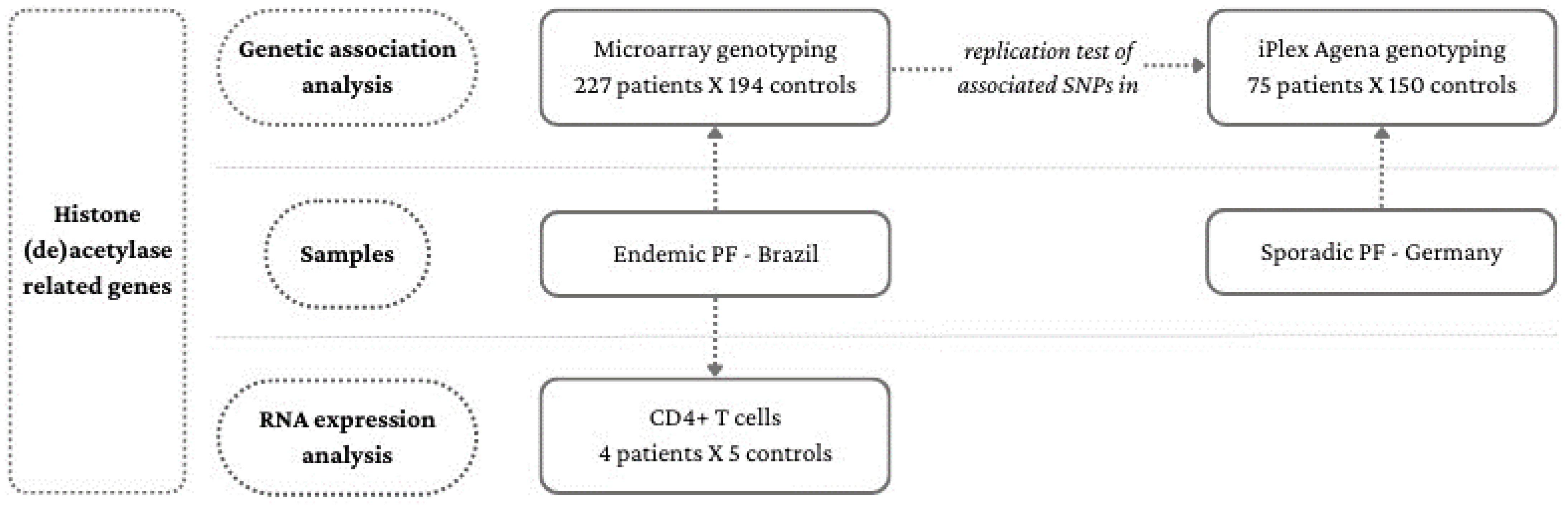
2.1. Association Analysis Samples
2.2. RNA Expression Analysis Samples
2.3. SNP Selection and Genotyping
2.4. Association Analysis
2.5. In Silico Analysis
2.6. RNAseq
3. Results
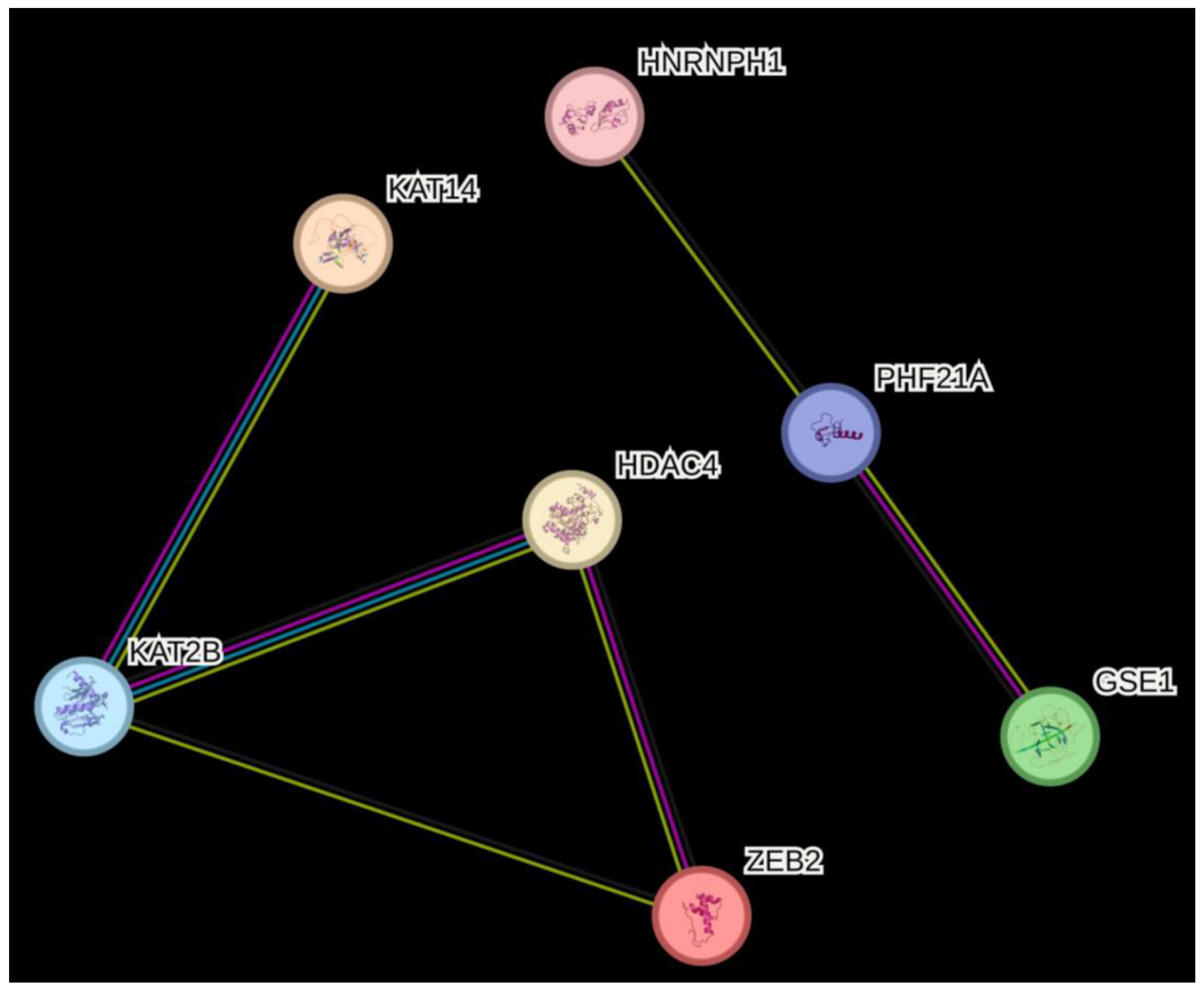
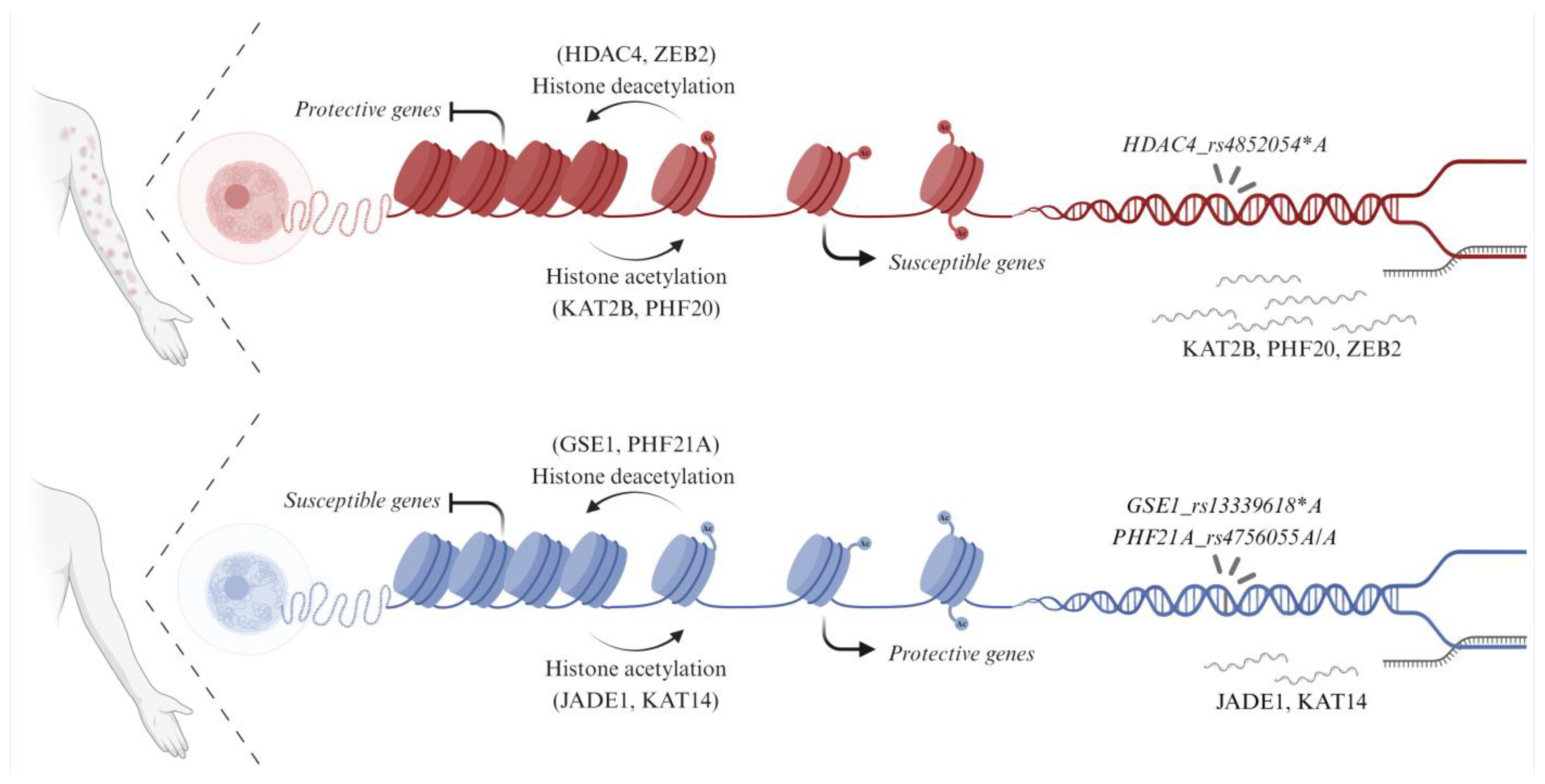
3.1. Genetic Association of Histone (De)Acetylation-Related Genes in EPF
3.2. Predicted Consequences of SNPs in Histone (De)Acetylation-Related Genes Associated with EPF
3.3. Differential RNA Expression of Histone (De)Acetylation-Related Genes in EPF
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Kasperkiewicz, M.; Ellebrecht, C.T.; Takahashi, H.; Yamagami, J.; Zillikens, D.; Payne, A.S.; Amagai, M. Pemphigus. Nat. Rev. Dis. Primers 2017, 3, 17026. [Google Scholar] [CrossRef] [PubMed]
- Schmidt, E.; Kasperkiewicz, M.; Joly, P. Pemphigus. Lancet 2019, 394, 882–894. [Google Scholar] [CrossRef] [PubMed]
- Qian, Y.; Jeong, J.S.; Maldonado, M.; Valenzuela, J.G.; Gomes, R.; Teixeira, C.; Evangelista, F.; Qaqish, B.; Aoki, V.; Hans, G.; et al. Cutting Edge: Brazilian Pemphigus Foliaceus Anti-Desmoglein 1 Autoantibodies Cross-React with Sand Fly Salivary LJM11 Antigen. J. Immunol. 2012, 189, 1535–1539. [Google Scholar] [CrossRef] [PubMed]
- Petzl-Erler, M.L. Beyond the HLA Polymorphism: A Complex Pattern of Genetic Susceptibility to Pemphigus. Genet. Mol. Biol. 2020, 43, e20190369. [Google Scholar] [CrossRef] [PubMed]
- Spadoni, M.B.; Bumiller-Bini, V.; Petzl-Erler, M.L.; Augusto, D.G.; Boldt, A.B.W. First Glimpse of Epigenetic Effects on Pemphigus Foliaceus. J. Investig. Dermatol. 2020, 140, 488–491.e1. [Google Scholar] [CrossRef] [PubMed]
- Petzl-Erler, M.L.; Santamaria, J. Are HLA Class II Genes Controlling Susceptibility and Resistance to Brazilian Pemphigus Foliaceus (Fogo Selvagem)? Tissue Antigens 1988, 33, 408–414. [Google Scholar] [CrossRef] [PubMed]
- Lobo-Alves, S.C.; Augusto, D.G.; Magalhães, W.C.S.; Tarazona-Santos, E.; Lima-Costa, M.F.; Barreto, M.L.; Horta, B.L.; de Almeida, R.C.; Petzl-Erler, M.L. Long Noncoding RNA Polymorphisms Influence Susceptibility to Endemic Pemphigus Foliaceus. Br. J. Dermatol. 2019, 181, 324–331. [Google Scholar] [CrossRef]
- Salviano-Silva, A.; Farias, T.D.J.; Bumiller-Bini, V.; Castro, M.D.S.; Lobo-Alves, S.C.; Busch, H.; Pföhler, C.; Worm, M.; Goebeler, M.; van Beek, N.; et al. Genetic Variability of Immune-Related LncRNAs: Polymorphisms in LINC-PINT and LY86-AS1 Are Associated with Pemphigus Foliaceus Susceptibility. Exp. Dermatol. 2021, 30, 831–840. [Google Scholar] [CrossRef]
- Salviano-Silva, A.; Becker, M.; Augusto, D.G.; Busch, H.; Adelman Cipolla, G.; Farias, T.D.J.; Bumiller-Bini, V.; Calonga-Solís, V.; Munz, M.; Franke, A.; et al. Genetic Association and Differential Expression of HLA Complex Group LncRNAs in Pemphigus. J. Autoimmun. 2021, 123, 102705. [Google Scholar] [CrossRef]
- Allis, C.D.; Caparros, M.-L.; Jenuwein, T.; Lachner, M.; Reinberg, D. Overview and Concepts. In Epigenetics; Allis, C.D., Caparros, M.-L., Jenuwein, T., Reinberg, D., Lachner, M., Eds.; Cold Spring Harbor Laboratory Press: New York, NY, USA, 2015. [Google Scholar]
- Marmorstein, R.; Zhou, M.M. Writers and Readers of Histone Acetylation: Structure, Mechanism, and Inhibition. Cold Spring Harb. Perspect. Biol. 2014, 6, a018762. [Google Scholar] [CrossRef]
- Zhao, M.; Wang, Z.; Yung, S.; Lu, Q. Epigenetic Dynamics in Immunity and Autoimmunity. Int. J. Biochem. Cell Biol. 2015, 67, 65–74. [Google Scholar] [CrossRef] [PubMed]
- Zhou, Y.; Su, Y.; Lu, Q.; Hu, N.; Qiu, X.; Luo, Y.; Yuan, J.; Li, Y.; Lei, W.; Zhang, G. Abnormal Histone Modification Patterns in Lupus CD4+ T Cells. J. Rheumatol. 2008, 35, 804–810. [Google Scholar]
- Zhang, Z.; Song, L.; Maurer, K.; Petri, M.A.; Sullivan, K.E. Global H4 Acetylation Analysis by ChIP-Chip in Systemic Lupus Erythematosus Monocytes. Genes. Immun. 2010, 11, 124–133. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Liang, G.; Wu, X.; Wang, S.; Zhang, P.; Su, Y.; Yin, H.; Tan, Y.; Zhang, J.; Lu, Q. Abnormal Epigenetic Modifications in Peripheral Blood Mononuclear Cells from Patients with Alopecia Areata. Br. J. Dermatol. 2012, 166, 266–273. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Huang, W.; Zhang, Q.; Gao, F.; Wang, L.; Zhang, G.; Su, Y.; Xiao, R.; Zhang, J.; Tang, M.; et al. Aberrant Epigenetic Modifications in Peripheral Blood Mononuclear Cells from Patients with Pemphigus Vulgaris. Br. J. Dermatol. 2012, 167, 523–531. [Google Scholar] [CrossRef] [PubMed]
- Calonga-Solís, V.; Amorim, L.M.; Farias, T.D.J.; Petzl-Erler, M.L.; Malheiros, D.; Augusto, D.G. Variation in Genes Implicated in B-Cell Development and Antibody Production Affects Susceptibility to Pemphigus. Immunology 2021, 162, 58–67. [Google Scholar] [CrossRef] [PubMed]
- Hoch, V.B.B.; Kohler, A.F.; Augusto, D.G.; Lobo-Alves, S.C.; Malheiros, D.; Cipolla, G.A.; Boldt, A.B.W.; Braun-Prado, K.; Wittig, M.; Franke, A.; et al. Genetic Associations and Differential MRNA Expression Levels of Host Genes Suggest a Viral Trigger for Endemic Pemphigus Foliaceus. Viruses 2022, 14, 879. [Google Scholar] [CrossRef] [PubMed]
- Augusto, D.G.; de Almeida, R.C.; Farias, T.D.J.; Magalhães, W.C.S.; Malheiros, D.; Lima-Costa, M.F.; Barreto, M.L.; Horta, B.L.; Kumar, V.; Wittig, M.; et al. Unsuspected Associations of Variants within the Genes NOTCH4 and STEAP2-AS1 Uncovered by a GWAS in Endemic Pemphigus Foliaceus. J. Investig. Dermatol. 2021, 141, 2741–2744. [Google Scholar] [CrossRef]
- Maglott, D.; Ostell, J.; Pruitt, K.D.; Tatusova, T. Entrez Gene: Gene-centered information at NCBI. Nucleic Acids Res. 2005, 33, D54–D58. [Google Scholar] [CrossRef]
- Benjamin, D.J.; Berger, J.O.; Johannesson, M.; Nosek, B.A.; Wagenmakers, E.J.; Berk, R.; Bollen, K.A.; Brembs, B.; Brown, L.; Camerer, C.; et al. Redefine Statistical Significance. Nat. Hum. Behav. 2018, 2, 6–10. [Google Scholar] [CrossRef]
- Kent, W.J.; Sugnet, C.W.; Furey, T.S.; Roskin, K.M.; Pringle, T.H.; Zahler, A.M.; Haussler, A.D. The Human Genome Browser at UCSC. Genome Res. 2002, 12, 996–1006. [Google Scholar] [CrossRef] [PubMed]
- Yates, A.D.; Achuthan, P.; Akanni, W.; Allen, J.; Allen, J.; Alvarez-Jarreta, J.; Amode, M.R.; Armean, I.M.; Azov, A.G.; Bennett, R.; et al. Ensembl 2020. Nucleic Acids Res. 2020, 48, D682–D688. [Google Scholar] [CrossRef] [PubMed]
- Uhlén, M.; Fagerberg, L.; Hallström, B.M.; Lindskog, C.; Oksvold, P.; Mardinoglu, A.; Sivertsson, Å.; Kampf, C.; Sjöstedt, E.; Asplund, A.; et al. Tissue-Based Map of the Human Proteome. Science 2015, 347, 1260419. [Google Scholar] [CrossRef] [PubMed]
- Monaco, G.; Lee, B.; Xu, W.; Mustafah, S.; Hwang, Y.Y.; Carré, C.; Burdin, N.; Visan, L.; Ceccarelli, M.; Poidinger, M.; et al. RNA-Seq Signatures Normalized by MRNA Abundance Allow Absolute Deconvolution of Human Immune Cell Types. Cell Rep. 2019, 26, 1627–1640. [Google Scholar] [CrossRef] [PubMed]
- Schmiedel, B.J.; Singh, D.; Madrigal, A.; Valdovino-Gonzalez, A.G.; White, B.M.; Zapardiel-Gonzalo, J.; Ha, B.; Altay, G.; Greenbaum, J.A.; McVicker, G.; et al. Impact of Genetic Polymorphisms on Human Immune Cell Gene Expression. Cell 2018, 175, 1701–1715. [Google Scholar] [CrossRef] [PubMed]
- Ward, L.D.; Kellis, M. HaploReg: A Resource for Exploring Chromatin States, Conservation, and Regulatory Motif Alterations within Sets of Genetically Linked Variants. Nucleic Acids Res. 2012, 40, D930–D934. [Google Scholar] [CrossRef] [PubMed]
- Oscanoa, J.; Sivapalan, L.; Gadaleta, E.; Dayem Ullah, A.Z.; Lemoine, N.R.; Chelala, C. SNPnexus: A Web Server for Functional Annotation of Human Genome Sequence Variation (2020 Update). Nucleic Acids Res. 2020, 48, W185–W192. [Google Scholar] [CrossRef] [PubMed]
- Roadmap Epigenomics Consortium; Kundaje, A.; Meuleman, W.; Ernst, J.; Bilenky, M.; Yen, A.; Heravi-Moussavi, A.; Kheradpour, P.; Zhang, Z.; Wang, J.; et al. Integrative Analysis of 111 Reference Human Epigenomes. Nature 2015, 518, 317–329. [Google Scholar] [CrossRef]
- Dunham, I.; Kundaje, A.; Aldred, S.F.; Collins, P.J.; Davis, C.A.; Doyle, F.; Epstein, C.B.; Frietze, S.; Harrow, J.; Kaul, R.; et al. An Integrated Encyclopedia of DNA Elements in the Human Genome. Nature 2012, 489, 57–74. [Google Scholar] [CrossRef]
- Myers, T.A.; Chanock, S.J.; Machiela, M.J. LDlinkR: An R Package for Rapidly Calculating Linkage Disequilibrium Statistics in Diverse Populations. Front. Genet. 2020, 11, 157. [Google Scholar] [CrossRef]
- Munz, M.; Tönnies, S.; Balke, W.T.; Simon, E. Multidimensional Gene Search with Genehopper. Nucleic Acids Res. 2015, 43, W98–W103. [Google Scholar] [CrossRef] [PubMed]
- Westra, H.J.; Peters, M.J.; Esko, T.; Yaghootkar, H.; Schurmann, C.; Kettunen, J.; Christiansen, M.W.; Fairfax, B.P.; Schramm, K.; Powell, J.E.; et al. Systematic Identification of Trans EQTLs as Putative Drivers of Known Disease Associations. Nat. Genet. 2013, 45, 1238–1243. [Google Scholar] [CrossRef] [PubMed]
- Fehrmann, R.S.N.; Jansen, R.C.; Veldink, J.H.; Westra, H.J.; Arends, D.; Bonder, M.J.; Fu, J.; Deelen, P.; Groen, H.J.M.; Smolonska, A.; et al. Trans-Eqtls Reveal That Independent Genetic Variants Associated with a Complex Phenotype Converge on Intermediate Genes, with a Major Role for the Hla. PLoS Genet. 2011, 7, e1002197. [Google Scholar] [CrossRef] [PubMed]
- Lonsdale, J.; Thomas, J.; Salvatore, M.; Phillips, R.; Lo, E.; Shad, S.; Hasz, R.; Walters, G.; Garcia, F.; Young, N.; et al. The Genotype-Tissue Expression (GTEx) Project. Nat. Genet. 2013, 45, 580–585. [Google Scholar] [CrossRef] [PubMed]
- Garnier, S.; Truong, V.; Brocheton, J.; Zeller, T.; Rovital, M.; Wild, P.S.; Ziegler, A.; Munzel, T.; Tiret, L.; Blankenberg, S.; et al. Genome-Wide Haplotype Analysis of Cis Expression Quantitative Trait Loci in Monocytes. PLoS Genet. 2013, 9, e1003240. [Google Scholar] [CrossRef] [PubMed]
- Codoni, V.; Blum, Y.; Civelek, M.; Proust, C.; Franzén, O.; Consortium, C.; Björkegren, J.L.M.; Le Goff, W.; Cambien, F.; Lusis, A.J.; et al. Preservation Analysis of Macrophage Gene Coexpression between Human and Mouse Identifies PARK2 as a Genetically Controlled Master Regulator of Oxidative Phosphorylation in Humans. G3 Genes. Genomes Genet. 2016, 6, 3361–3371. [Google Scholar] [CrossRef]
- Zeller, T.; Wild, P.; Szymczak, S.; Rotival, M.; Schillert, A.; Castagne, R.; Maouche, S.; Germain, M.; Lackner, K.; Rossmann, H.; et al. Genetics and beyond—the Transcriptome of Human Monocytes and Disease Susceptibility. PLoS ONE 2010, 5, e10693. [Google Scholar] [CrossRef]
- Landrum, M.J.; Chitipiralla, S.; Brown, G.R.; Chen, C.; Gu, B.; Hart, J.; Hoffman, D.; Jang, W.; Kaur, K.; Liu, C.; et al. ClinVar: Improvements to Accessing Data. Nucleic Acids Res. 2020, 48, D835–D844. [Google Scholar] [CrossRef]
- Meda, F.; Folci, M.; Baccarelli, A.; Selmi, C. The Epigenetics of Autoimmunity. Cell Mol. Immunol. 2011, 8, 226–236. [Google Scholar] [CrossRef]
- Bumiller-Bini, V.; Cipolla, G.A.; de Almeida, R.C.; Petzl-Erler, M.L.; Augusto, D.G.; Boldt, A.B.W. Sparking Fire under the Skin? Answers from the Association of Complement Genes with Pemphigus Foliaceus. Front. Immunol. 2018, 9, 695. [Google Scholar] [CrossRef]
- Grozinger, C.M.; Hassig, C.A.; Schreiber, S.L. Three Proteins Define a Class of Human Histone Deacetylases Related to Yeast Hda1p. Proc. Natl. Acad. Sci. USA 1999, 96, 4868–4873. [Google Scholar] [CrossRef] [PubMed]
- Miska, E.A.; Karlsson, C.; Langley, E.; Nielsen, S.J.; Pines, J.; Kouzarides, T. HDAC4 Deacetylase Associates with and Represses the MEF2 Transcription Factor. EMBO J. 1999, 18, 5099–5107. [Google Scholar] [CrossRef] [PubMed]
- Wang, A.H.; Bertos, N.R.; Vezmar, M.; Pelletier, N.; Crosato, M.; Heng, H.H.; Th’ng, J.; Han, J.; Yang, X.-J. HDAC4, a Human Histone Deacetylase Related to Yeast HDA1, Is a Transcriptional Corepressor. Mol. Cell Biol. 1999, 19, 7816–7827. [Google Scholar] [CrossRef] [PubMed]
- Youn, H.D.; Grozinger, C.M.; Liu, J.O. Calcium Regulates Transcriptional Repression of Myocyte Enhancer Factor 2 by Histone Deacetylase 4. J. Biol. Chem. 2000, 275, 22563–22567. [Google Scholar] [CrossRef] [PubMed]
- Paroni, G.; Mizzau, M.; Henderson, C.; Giannino; Sal, D.; Schneider, C.; Brancolini, C. Caspase-Dependent Regulation of Histone Deacetylase 4 Nuclear-Cytoplasmic Shuttling Promotes Apoptosis. Mol. Biol. Cell 2004, 15, 2804–2818. [Google Scholar] [CrossRef]
- Liu, Z.-G.; Smith, S.W.; McLaughlin, K.A.; Schwartz, L.M.; Osborne, B.A. Apoptotic Signals Delivered through the T-Cell Receptor of a T-Cell Hybrid Require the Immediate–Early Gene Nur77. Nature 1994, 367, 281–284. [Google Scholar] [CrossRef] [PubMed]
- Kumar, A.; Lin, Z.; SenBanerjee, S.; Jain, M.K. Tumor Necrosis Factor Alpha-Mediated Reduction of KLF2 Is Due to Inhibition of MEF2 by NF-ΚB and Histone Deacetylases. Mol. Cell Biol. 2005, 25, 5893–5903. [Google Scholar] [CrossRef]
- Jha, P.; Das, H. KLF2 in Regulation of NF-ΚB-Mediated Immune Cell Function and Inflammation. Int. J. Mol. Sci. 2017, 18, 2383. [Google Scholar] [CrossRef]
- Bumiller-Bini, V.; Cipolla, G.A.; Spadoni, M.B.; Augusto, D.G.; Petzl-Erler, M.L.; Beltrame, M.H.; Boldt, A.B.W. Condemned or Not to Die? Gene Polymorphisms Associated with Cell Death in Pemphigus Foliaceus. Front. Immunol. 2019, 10, 2416. [Google Scholar] [CrossRef]
- Hoch, V.B.B.; Schneider, L.; Pumpe, A.E.; Lüders, E.; Hundt, J.E.; Boldt, A.B.W. Marked to Die-Cell Death Mechanisms for Keratinocyte Acantholysis in Pemphigus Diseases. Life 2022, 12, 329. [Google Scholar] [CrossRef]
- Hatzi, K.; Philip Nance, J.; Kroenke, M.A.; Bothwell, M.; Haddad, E.K.; Melnick, A.; Crotty, S. BCL6 Orchestrates Tfh Cell Differentiation via Multiple Distinct Mechanisms. J. Exp. Med. 2015, 212, 539–553. [Google Scholar] [CrossRef] [PubMed]
- Lemercier, C.; Brocard, M.P.; Puvion-Dutilleul, F.; Kao, H.Y.; Albagli, O.; Khochbin, S. Class II Histone Deacetylases Are Directly Recruited by BCL6 Transcriptional Repressor. J. Biol. Chem. 2002, 277, 22045–22052. [Google Scholar] [CrossRef] [PubMed]
- Li, Q.; Liu, Z.; Dang, E.; Jin, L.; He, Z.; Yang, L.; Shi, X.; Wang, G. Follicular Helper T Cells (Tfh) and IL-21 Involvement in the Pathogenesis of Bullous Pemphigoid. PLoS ONE 2013, 8, e68145. [Google Scholar] [CrossRef] [PubMed]
- Molineros, J.E.; Yang, W.; Zhou, X.J.; Sun, C.; Okada, Y.; Zhang, H.; Chua, K.H.; Lau, Y.L.; Kochi, Y.; Suzuki, A.; et al. Confirmation of Five Novel Susceptibility Loci for Systemic Lupus Erythematosus (SLE) and Integrated Network Analysis of 82 SLE Susceptibility Loci. Hum. Mol. Genet. 2017, 26, 1205–1216. [Google Scholar] [CrossRef] [PubMed]
- McKinsey, T.A.; Kuwahara, K.; Bezprozvannaya, S.; Olson, E.N. Class II Histone Deacetylases Confer Signal Responsiveness to the Ankyrin-Repeat Proteins ANKRA2 and RFXANK. Mol. Biol. Cell 2006, 17, 438–447. [Google Scholar] [CrossRef] [PubMed]
- Piovezan, B.Z.; Petzl-Erler, M.L. Both Qualitative and Quantitative Genetic Variation of MHC Class II Molecules May Influence Susceptibility to Autoimmune Diseases: The Case of Endemic Pemphigus Foliaceus. Hum. Immunol. 2013, 74, 1134–1140. [Google Scholar] [CrossRef] [PubMed]
- Han, S.; Lu, J.; Zhang, Y.; Cheng, C.; Han, L.; Wang, X.; Li, N.; Liu, C.; Huang, B. Recruitment of Histone Deacetylase 4 by Transcription Factors Represses Interleukin-5 Transcription. Biochem. J. 2006, 400, 439–448. [Google Scholar] [CrossRef] [PubMed]
- Warren, S.J.P.; Arteaga, L.A.; Rivitti, E.A.; Aoki, V.; Hans-Filho, G.; Qaqish, B.F.; Lin, M.S.; Giudice, G.J.; Diaz, L.A. The Role of Subclass Switching in the Pathogenesis of Endemic Pemphigus Foliaceus. J. Invest. Dermatol. 2003, 120, 1–5. [Google Scholar] [CrossRef]
- Aoki, V.; Millikan, R.C.; Rivitti, E.A.; Hans-Filho, G.; Eaton, D.P.; Warren, S.J.P.; Li, N.; Hilario-Vargas, J.; Hoffmann, R.G.; Diaz, L.A. Environmental Risk Factors in Endemic Pemphigus Foliaceus (Fogo Selvagem). JID Symp. Proc. 2004, 9, 34–40. [Google Scholar] [CrossRef]
- Pan, J.; Zhao, L. Long Non-Coding RNA Histone Deacetylase 4 Antisense RNA 1 (HDAC4-AS1) Inhibits HDAC4 Expression in Human ARPE-19 Cells with Hypoxic Stress. Bioengineered 2021, 12, 2228–2237. [Google Scholar] [CrossRef]
- Hakimi, M.A.; Dong, Y.; Lane, W.S.; Speicher, D.W.; Shiekhattar, R. A Candidate X-Linked Mental Retardation Gene Is a Component of a New Family of Histone Deacetylase-Containing Complexes. J. Biol. Chem. 2003, 278, 7234–7239. [Google Scholar] [CrossRef]
- Hakimi, M.-A.; Bochar, D.A.; Chenoweth, J.; Lane, W.S.; Mandel, G.; Shiekhattar, R.; Rosenfeld, M.G. A Core-BRAF35 Complex Containing Histone Deacetylase Mediates Repression of Neuronal-Specific Genes. PNAS 2002, 99, 7420–7425. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Yan, S.; Yang, B.; Wang, Y.; Zhou, H.; Lian, Q.; Sun, B. TRIM35 Negatively Regulates TLR7- and TLR9-Mediated Type I Interferon Production by Targeting IRF7. FEBS Lett. 2015, 589, 1322–1330. [Google Scholar] [CrossRef] [PubMed]
- Hall, J.C.; Rosen, A. Type I Interferons: Crucial Participants in Disease Amplification in Autoimmunity. Nat. Rev. Rheumatol. 2010, 6, 40–49. [Google Scholar] [CrossRef] [PubMed]
- Meier-Soelch, J.; Jurida, L.; Weber, A.; Newel, D.; Kim, J.; Braun, T.; Lienhard Schmitz, M.; Kracht, M. RNAi-Based Identification of Gene-Specific Nuclear Cofactor Networks Regulating Interleukin-1 Target Genes. Front. Immunol. 2018, 9, 775. [Google Scholar] [CrossRef] [PubMed]
- Meier, K.; Brehm, A. Chromatin Regulation: How Complex Does It Get? Epigenetics 2014, 9, 1485–1495. [Google Scholar] [CrossRef] [PubMed]
- Xiong, Y.; Wang, L.; Giorgio, E.D.; Akimova, T.; Beier, U.H.; Han, R.; Trevisanut, M.; Kalin, J.H.; Cole, P.A.; Hancock, W.W. Inhibiting the Coregulator CoREST Impairs Foxp3+ Treg Function and Promotes Antitumor Immunity. J. Clin. Investig. 2020, 130, 1830–1842. [Google Scholar] [CrossRef]
- Weintraub, Y.; Cohen, S.; Yerushalmy-Feler, A.; Chapnik, N.; Tsameret, S.; Anafy, A.; Damari, E.; Ben-Tov, A.; Shamir, R.; Froy, O. Circadian Clock Gene Disruption in White Blood Cells of Patients with Celiac Disease. Biochimie 2023, 219, 51–54. [Google Scholar] [CrossRef]
- Cao, Q.; Zhao, X.; Bai, J.; Gery, S.; Sun, H.; Lin, D.C.; Chen, Q.; Chen, Z.; Mack, L.; Yang, H.; et al. Circadian Clock Cryptochrome Proteins Regulate Autoimmunity. Proc. Natl. Acad. Sci. USA 2017, 114, 12548–12553. [Google Scholar] [CrossRef]
- Tanu, T.; Taniue, K.; Imamura, K.; Onoguchi-Mizutani, R.; Han, H.; Jensen, T.H.; Akimitsu, N. HnRNPH1-MTR4 Complex-Mediated Regulation of NEAT1v2 Stability Is Critical for IL8 Expression. RNA Biol. 2021, 18, 537–547. [Google Scholar] [CrossRef]
- Kowalski, E.H.; Kneibner, D.; Kridin, K.; Amber, K.T. Serum and Blister Fluid Levels of Cytokines and Chemokines in Pemphigus and Bullous Pemphigoid. Autoimmun. Rev. 2019, 18, 526–534. [Google Scholar] [CrossRef] [PubMed]
- Verschueren, K.; Remacle, J.E.; Collart, C.; Kraft, H.; Baker, B.S.; Tylzanowski, P.; Nelles, L.; Wuytens, G.; Su, M.T.; Bodmer, R.; et al. SIP1, a Novel Zinc Finger/Homeodomain Repressor, Interacts with Smad Proteins and Binds to 5′-CACCT Sequences in Candidate Target Genes. J. Biol. Chem. 1999, 274, 20489–20498. [Google Scholar] [CrossRef] [PubMed]
- Postigo, A.A.; Dean, D.C.; Kipnis, D.M. Differential Expression and Function of Members of the Zfh-1 Family of Zinc Fingerhomeodomain Repressors. Proc. Natl. Acad. Sci. USA 2000, 97, 6391–6396. [Google Scholar] [CrossRef] [PubMed]
- Goossens, S.; Janzen, V.; Bartunkova, S.; Yokomizo, T.; Drogat, B.; Crisan, M.; Haigh, K.; Seuntjens, E.; Umans, L.; Riedt, T.; et al. The EMT Regulator Zeb2/Sip1 Is Essential for Murine Embryonic Hematopoietic Stem/Progenitor Cell Differentiation and Mobilization. Blood J. Am. Soc. Hematol. 2011, 117, 5620–5630. [Google Scholar] [CrossRef] [PubMed]
- Omilusik, K.D.; Adam Best, J.; Yu, B.; Goossens, S.; Weidemann, A.; Nguyen, J.V.; Seuntjens, E.; Stryjewska, A.; Zweier, C.; Roychoudhuri, R.; et al. Transcriptional Repressor ZEB2 Promotes Terminal Differentiation of CD8+ Effector and Memory T Cell Populations during Infection. J. Exp. Med. 2015, 212, 2027–2039. [Google Scholar] [CrossRef]
- Dominguez, C.X.; Amezquita, R.A.; Guan, T.; Marshall, H.D.; Joshi, N.S.; Kleinstein, S.H.; Kaech, S.M. The Transcription Factors ZEB2 and T-Bet Cooperate to Program Cytotoxic T Cell Terminal Differentiation in Response to LCMV Viral Infection. J. Exp. Med. 2015, 212, 2041–2056. [Google Scholar] [CrossRef]
- Scott, C.L.; Soen, B.; Martens, L.; Skrypek, N.; Saelens, W.; Taminau, J.; Blancke, G.; Van Isterdael, G.; Huylebroeck, D.; Haigh, J.; et al. The Transcription Factor Zeb2 Regulates Development of Conventional and Plasmacytoid DCs by Repressing Id2. J. Exp. Med. 2016, 213, 897–911. [Google Scholar] [CrossRef]
- Ghasemi, A.; Farazmand, A.; Hassanzadeh, V.; Poursani, S.; Soltani, S.; Akhtari, M.; Akhlaghi, M.; Farhadi, E.; Jamshidi, A.; Mahmoudi, M. Upregulation of KAT2B and ESCO2 Gene Expression Level in Patients with Rheumatoid Arthritis. Clin. Rheumatol. 2023, 42, 253–259. [Google Scholar] [CrossRef]
- Brockmann, D.; Lehmkühler, O.; Schmücker, U.; Esche, H. The Histone Acetyltransferase Activity of PCAF Cooperates with the Brahma/SWI2-Related Protein BRG-1 in the Activation of the Enhancer A of the MHC Class I Promoter. Gene 2001, 277, 111–120. [Google Scholar] [CrossRef]
- Liu, Y.; Bao, C.; Wang, L.; Han, R.; Beier, U.H.; Akimova, T.; Cole, P.A.; Dent, S.Y.R.; Hancock, W.W. Complementary Roles of Gcn5 and Pcaf in Foxp3+ T-Regulatory Cells. Cancers 2019, 11, 554. [Google Scholar] [CrossRef]
- de Jong, A.; de Jong, R.C.M.; Peters, E.A.; Arens, R.; Jukema, J.W.; de Vries, M.R.; Quax, P.H.A. P300/CBP Associated Factor (PCAF) Deficiency Enhances Diet-Induced Atherosclerosis in ApoE3*Leiden Mice via Systemic Inhibition of Regulatory T Cells. Front. Cardiovasc. Med. 2021, 7, 604821. [Google Scholar] [CrossRef] [PubMed]
- Bastiaansen, A.J.N.M.; Ewing, M.M.; De Boer, H.C.; Van Der Pouw Kraan, T.C.; De Vries, M.R.; Peters, E.A.B.; Welten, S.M.J.; Arens, R.; Moore, S.M.; Faber, J.E.; et al. Lysine Acetyltransferase PCAF Is a Key Regulator of Arteriogenesis. Arter. Thromb. Vasc. Biol. 2013, 33, 1902–1910. [Google Scholar] [CrossRef] [PubMed]
- Cleary, J.; Sitwala, K.V.; Khodadoust, M.S.; Kwok, R.P.S.; Mor-Vaknin, N.; Cebrat, M.; Cole, P.A.; Markovitz, D.M. P300/CBP-Associated Factor Drives DEK into Interchromatin Granule Clusters. J. Biol. Chem. 2005, 280, 31760–31767. [Google Scholar] [CrossRef] [PubMed]
- Sierakowska, H.; Williams, K.R.; Szer, I.S.; Szer, W. The Putative Oncoprotein DEK, Part Ofa Chimera Protein Associated with Acute Myeloid Leukaemia, Is an Autoantigen in Juvenile Rheumatoid Arthritis. Clin. Exp. Immunol. 1993, 94, 435–439. [Google Scholar] [CrossRef] [PubMed]
- Dong, X.; Wang, J.; Kabir, F.N.; Shaw, M.; Reed, A.M.; Stein, L.; Andrade, L.E.C.; Trevisani, V.F.M.; Miller, M.L.; Fujii, T.; et al. Autoantibodies to Dek Oncoprotein in Human Inflammatory Disease. Arthritis Rheum. Off. J. Am. Coll. Rheumatol. 2000, 43, 85–93. [Google Scholar] [CrossRef]
- Cai, Y.; Jin, J.; Swanson, S.K.; Cole, M.D.; Choi, S.H.; Florens, L.; Washburn, M.P.; Conaway, J.W.; Conaway, R.C. Subunit Composition and Substrate Specificity of a MOF-Containing Histone Acetyltransferase Distinct from the Male-Specific Lethal (MSL) Complex. J. Biol. Chem. 2010, 285, 4268–4272. [Google Scholar] [CrossRef] [PubMed]
- Badeaux, A.I.; Yang, Y.; Cardenas, K.; Vemulapalli, V.; Chen, K.; Kusewitt, D.; Richie, E.; Li, W.; Bedford, M.T. Loss of the Methyl Lysine Effector Protein PHF20 Impacts the Expression of Genes Regulated by the Lysine Acetyltransferase MOF. J. Biol. Chem. 2012, 287, 429–437. [Google Scholar] [CrossRef]
- Adams-Cioaba, M.A.; Li, Z.; Tempel, W.; Guo, Y.; Bian, C.; Li, Y.; Lam, R.; Min, J. Crystal Structures of the Tudor Domains of Human PHF20 Reveal Novel Structural Variations on the Royal Family of Proteins. FEBS Lett. 2012, 586, 859–865. [Google Scholar] [CrossRef]
- Klein, B.J.; Wang, X.; Cui, G.; Yuan, C.; Botuyan, M.V.; Lin, K.; Lu, Y.; Wang, X.; Zhao, Y.; Bruns, C.J.; et al. PHF20 Readers Link Methylation of Histone H3K4 and P53 with H4K16 Acetylation. Cell Rep. 2016, 17, 1158–1170. [Google Scholar] [CrossRef]
- Cui, G.; Park, S.; Badeaux, A.I.; Kim, D.; Lee, J.; Thompson, J.R.; Yan, F.; Kaneko, S.; Yuan, Z.; Botuyan, M.V.; et al. PHF20 Is an Effector Protein of P53 Double Lysine Methylation That Stabilizes and Activates P53. Nat. Struct. Mol. Biol. 2012, 19, 916–924. [Google Scholar] [CrossRef]
- Park, S.W.; Kim, J.; Oh, S.; Lee, J.; Cha, J.; Lee, H.S.; Kim, K.I.; Park, D.; Baek, S.H. PHF20 Is Crucial for Epigenetic Control of Starvation-Induced Autophagy through Enhancer Activation. Nucleic Acids Res. 2022, 50, 7856–7872. [Google Scholar] [CrossRef] [PubMed]
- Guelman, S.; Kozuka, K.; Mao, Y.; Pham, V.; Solloway, M.J.; Wang, J.; Wu, J.; Lill, J.R.; Zha, J. The Double-Histone-Acetyltransferase Complex ATAC Is Essential for Mammalian Development. Mol. Cell Biol. 2009, 29, 1176–1188. [Google Scholar] [CrossRef] [PubMed]
- Tzouanacou, E.; Tweedie, S.; Wilson, V. Identification of Jade1, a Gene Encoding a PHD Zinc Finger Protein, in a Gene Trap Mutagenesis Screen for Genes Involved in Anteroposterior Axis Development. Mol. Cell Biol. 2003, 23, 8553–8562. [Google Scholar] [CrossRef] [PubMed]
- Panchenko, M.V.; Zhou, M.I.; Cohen, H.T. Von Hippel-Lindau Partner Jade-1 Is a Transcriptional Co-Activator Associated with Histone Acetyltransferase Activity. J. Biol. Chem. 2004, 279, 56032–56041. [Google Scholar] [CrossRef]
- Foy, R.L.; Ihn, Y.S.; Chitalia, V.C.; Cohen, H.T.; Saksouk, N.; Cayrou, C.; Vaziri, C.; Côté, J.; Panchenko, M.V. Role of Jade-1 in the Histone Acetyltransferase (HAT) HBO1 Complex. J. Biol. Chem. 2008, 283, 28817–28826. [Google Scholar] [CrossRef]
- Avvakumov, N.; Lalonde, M.-E.; Saksouk, N.; Paquet, E.; Glass, K.C.; Landry, A.-J.; Doyon, Y.; Cayrou, C.; Robitaille, G.A.; Richard, D.E.; et al. Conserved Molecular Interactions within the HBO1 Acetyltransferase Complexes Regulate Cell Proliferation. Mol. Cell Biol. 2012, 32, 689–703. [Google Scholar] [CrossRef]
- Zhou, M.I.; Foy, R.L.; Chitalia, V.C.; Zhao, J.; Panchenko, M.V.; Wang, H.; Cohen, H.T. Jade-1, a Candidate Renal Tumor Suppressor That Promotes Apoptosis. Proc. Natl. Acad. Sci. USA 2005, 102, 11035–11040. [Google Scholar] [CrossRef]
| Gene | SNP | Endemic Pemphigus Foliaceus | Sporadic Pemphigus Foliaceus | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| MAF (%) | Model | Control | Patients | OR | 95% CI | p | MAF (%) | Model | Control | Patients | OR | 95% CI | p | ||||
| Controls | Patients | Controls | Patients | ||||||||||||||
| HDAC4 | rs4852054 | 30.67 | 39.82 | ADD | 119/269 | 180/272 | 1.525 | [1.14–2.05] | 0.0051 | 18.71 | 23.03 | ADD | 52/226 | 35/117 | 1.299 | [0.80–2.11] | 0.2891 |
| 2q37.3 | G > a | REC | 19/175 | 32/194 | 1.536 | [0.84–2.81] | 0.1647 | REC | 05/134 | 04/72 | 1.489 | [0.39–5.72] | 0.5621 | ||||
| intron 1 | DOM | 78/148 | 94/100 | 1.794 | [1.21–2.67] | 0.0038 | DOM | 47/92 | 31/45 | 1.348 | [0.76–2.40] | 0.3097 | |||||
| GSE1 | rs13339618 | 29.64 | 19.42 | ADD | 115/273 | 87/361 | 0.571 | [0.41–0.80] | 0.0011 | 19.06 | 23.65 | ADD | 53/225 | 35/133 | 1.257 | [0.81–1.96] | 0.3108 |
| 16q24.1 | G > a | REC | 16/178 | 5/219 | 0.261 | [0.09–0.73] | 0.0102 | REC | 09/130 | 07/67 | 1.509 | [0.54–4.23] | 0.4339 | ||||
| regulatory region downstream | DOM | 99/95 | 82/142 | 0.578 | [0.39–0.86] | 0.0064 | DOM | 44/95 | 28/46 | 1.314 | [0.73–2.37] | 0.3643 | |||||
| PHF21A | rs4756055 | 38.05 | 42.82 | ADD | 186/198 | 172/280 | 0.671 | [0.50–0.90] | 0.0079 | 45.32 | 37.50 | ADD | 126/152 | 57/95 | 0.740 | [0.50–1.10] | 0.1322 |
| 11p11.2 | g > A | REC | 50/142 | 26/200 | 0.391 | [0.23–0.67] | 0.0007 | REC | 31/108 | 12/64 | 0.653 | [0.38–1.36] | 0.2559 | ||||
| intron 1 | DOM | 136/56 | 146/80 | 0.792 | [0.52–1.21] | 0.2831 | DOM | 95/44 | 45/31 | 0.672 | [0.38–1.20] | 0.1801 | |||||
| SNPs | r2 | eQTL | Tissues and Cells |
|---|---|---|---|
| HDAC4_rs4852054*A (Susceptibility) | - | HDAC4-AS1 | Blood (ES = 0.15; p = 1.5 × 10−4) |
| Skin not sun-exposed (ES = 0.17; p = 3.4 × 10−3) | |||
| HDAC4_rs4852053*C | 1.0 | HDAC4-AS1 | Blood (ES = 0.17, p = 4.7 × 10−5) |
| Skin sun-exposed (ES = 0.16; p = 5.0 × 10−3) | |||
| HDAC4_rs55932933*T | 0.99 | HDAC4-AS1 | Blood (ES = 0.17; p = 6.3 × 10−5) |
| Skin sun-exposed (ES = 0.16; p = 5.0 × 10−3) | |||
| HDAC4_rs56064197*T | 0.99 | HDAC4-AS1 | Blood (ES = 0.17; p = 5.8 × 10−5) |
| Skin sun-exposed (ES = 0.16; p = 5.0 × 10−3) | |||
| HDCA4_rs1476321*T | 0.99 | HDAC4-AS1 | Blood (ES = 0.15; p = 1.9 × 10−4) |
| Skin not sun-exposed (ES = 0.17; p = 3.4 × 10−3) | |||
| HDCA4_rs10182344*A | 0.99 | HDAC4-AS1 | Blood (ES = 0.14; p = 3.5 × 10−4) |
| HDCA4_rs58332998*A | 0.93 | - | - |
| GSE1_rs13339618*A (Protection) | GSE1 | Blood (ES = 5.37; p = 7.9 × 10−8) | |
| GSE1_rs13339626*A | 1.0 | GSE1 | Blood (ES = 5.37; p = 7.9 × 10−8) |
| GSE1_rs13329722*C | 1.0 | GSE1 | Blood (ES = 5.40; p = 6.9 × 10−8) |
| TRIM35 | Monocytes (EF = -; p = 9.2 × 10−6) | ||
| GSE1_rs7498141*A | 1.0 | GSE1 | Blood (EF = 5.40; p = 6.9 × 10−8) |
| TRIM35 | Monocytes (ES = -; p = 9.0 × 10−6) | ||
| GSE1_rs4843505*G | 1.0 | GSE1 | Blood (ES = 5.40; p = 6.9 × 10−8) |
| GSE1_rs13332576*A | 0.99 | GSE1 | Blood (ES = 5.31; p = 1.1 × 10−7) |
| GSE1_rs8060638*A | 0.89 | - | - |
| GSE1_rs28671512*T | 0.99 | - | - |
| PHF21A_rs4756055*A/A (Protection) | - | CRY2 | Blood (ES = 3.64; p = 2.7 × 10−4) |
| PEX16 | Blood (ES = −3.16; p = 1.0 × 10−3) | ||
| PHF21A | Blood (ES = -; p = 2.8 × 10−4) | ||
| HNRNPH1 | Macrophages (ES = 0.08; p = 5.8 × 10−6) | ||
| PHF21A_rs1976182*A | 0.98 | HNRNPH1 | Macrophages (ES = 0.08; p = 5.4 × 10−6) |
| PHF21A_rs923530*T | 0.87 | CRY2 | Monocytes (ES = −0.04, p = 3.8 × 10−4) |
| HNRNPH1 | Macrophages (ES = 0.08, p = 7.6 × 10−6) | ||
| SLC37A1 | Macrophages (EF = −0.05, p = 2.3 × 10−6) | ||
| PHF21A_rs11374563*T | 0.87 | - | - |
| PHF21A_rs7109480*T | 0.93 | CRY2 | Blood (ES = 3.46, p = 5.4 × 10−4) |
| HNRNPH1 | Macrophages (EF = 0.08; p = 6.8 × 10−6) | ||
| PHF21A_rs7107550*C | 0.86 | CRY2 | Monocytes (ES = −0.04, p = 3.2 × 10−4) |
| HNRNPH1 | Macrophages (ES = 0.08, p = 9.3 × 10−6) | ||
| SLC37A1 | Macrophages (EF = −0.05, p = 4.3 × 10−6) | ||
| PHF21A_rs950105*A | 1.0 | CRY2 | Blood (ES = 3.41; p = 6.4 × 10−4) |
| HNRNPH1 | Macrophages (EF = 0.08; p = 9.1 × 10−6) | ||
| PHF21A_rs74366855*A | 0.87 | CRY2 | Monocytes (ES = −0.04, p = 5.8 × 10−4) |
| HNRNPH1 | Macrophages (ES = 0.08, p = 8.0 × 10−6) | ||
| SLC37A1 | Macrophages (EF = −0.05, p = 5.7 × 10−6) |
| Gene | Full Name | Fold Change | p-Value |
|---|---|---|---|
| ZEB2 | zinc finger E-box binding homeobox 2 | 1.08 | 6.2 × 10−6 |
| KAT2B | lysine acetyltransferase 2B | 0.55 | 2.8 × 10−5 |
| PHF20 | PHD finger protein 20 | 0.41 | 8.6 × 10−5 |
| KAT14 | lysine acetyltransferase 14 | −0.59 | 2.0 × 10−4 |
| JADE1 | jade family PHD finger 1 | −0.31 | 2.6 × 10−3 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sulzbach Denardin, M.; Bumiller-Bini Hoch, V.; Salviano-Silva, A.; Lobo-Alves, S.C.; Adelman Cipolla, G.; Malheiros, D.; Augusto, D.G.; Wittig, M.; Franke, A.; Pföhler, C.; et al. Genetic Association and Differential RNA Expression of Histone (De)Acetylation-Related Genes in Pemphigus Foliaceus—A Possible Epigenetic Effect in the Autoimmune Response. Life 2024, 14, 60. https://doi.org/10.3390/life14010060
Sulzbach Denardin M, Bumiller-Bini Hoch V, Salviano-Silva A, Lobo-Alves SC, Adelman Cipolla G, Malheiros D, Augusto DG, Wittig M, Franke A, Pföhler C, et al. Genetic Association and Differential RNA Expression of Histone (De)Acetylation-Related Genes in Pemphigus Foliaceus—A Possible Epigenetic Effect in the Autoimmune Response. Life. 2024; 14(1):60. https://doi.org/10.3390/life14010060
Chicago/Turabian StyleSulzbach Denardin, Maiara, Valéria Bumiller-Bini Hoch, Amanda Salviano-Silva, Sara Cristina Lobo-Alves, Gabriel Adelman Cipolla, Danielle Malheiros, Danillo G. Augusto, Michael Wittig, Andre Franke, Claudia Pföhler, and et al. 2024. "Genetic Association and Differential RNA Expression of Histone (De)Acetylation-Related Genes in Pemphigus Foliaceus—A Possible Epigenetic Effect in the Autoimmune Response" Life 14, no. 1: 60. https://doi.org/10.3390/life14010060
APA StyleSulzbach Denardin, M., Bumiller-Bini Hoch, V., Salviano-Silva, A., Lobo-Alves, S. C., Adelman Cipolla, G., Malheiros, D., Augusto, D. G., Wittig, M., Franke, A., Pföhler, C., Worm, M., van Beek, N., Goebeler, M., Sárdy, M., Ibrahim, S., Busch, H., Schmidt, E., Hundt, J. E., Petzl-Erler, M. L., & Beate Winter Boldt, A. (2024). Genetic Association and Differential RNA Expression of Histone (De)Acetylation-Related Genes in Pemphigus Foliaceus—A Possible Epigenetic Effect in the Autoimmune Response. Life, 14(1), 60. https://doi.org/10.3390/life14010060









