The Endangered Sardinian Grass Snake: Distribution Update, Bioclimatic Niche Modelling, Dorsal Pattern Characterisation, and Literature Review
Abstract
1. Introduction
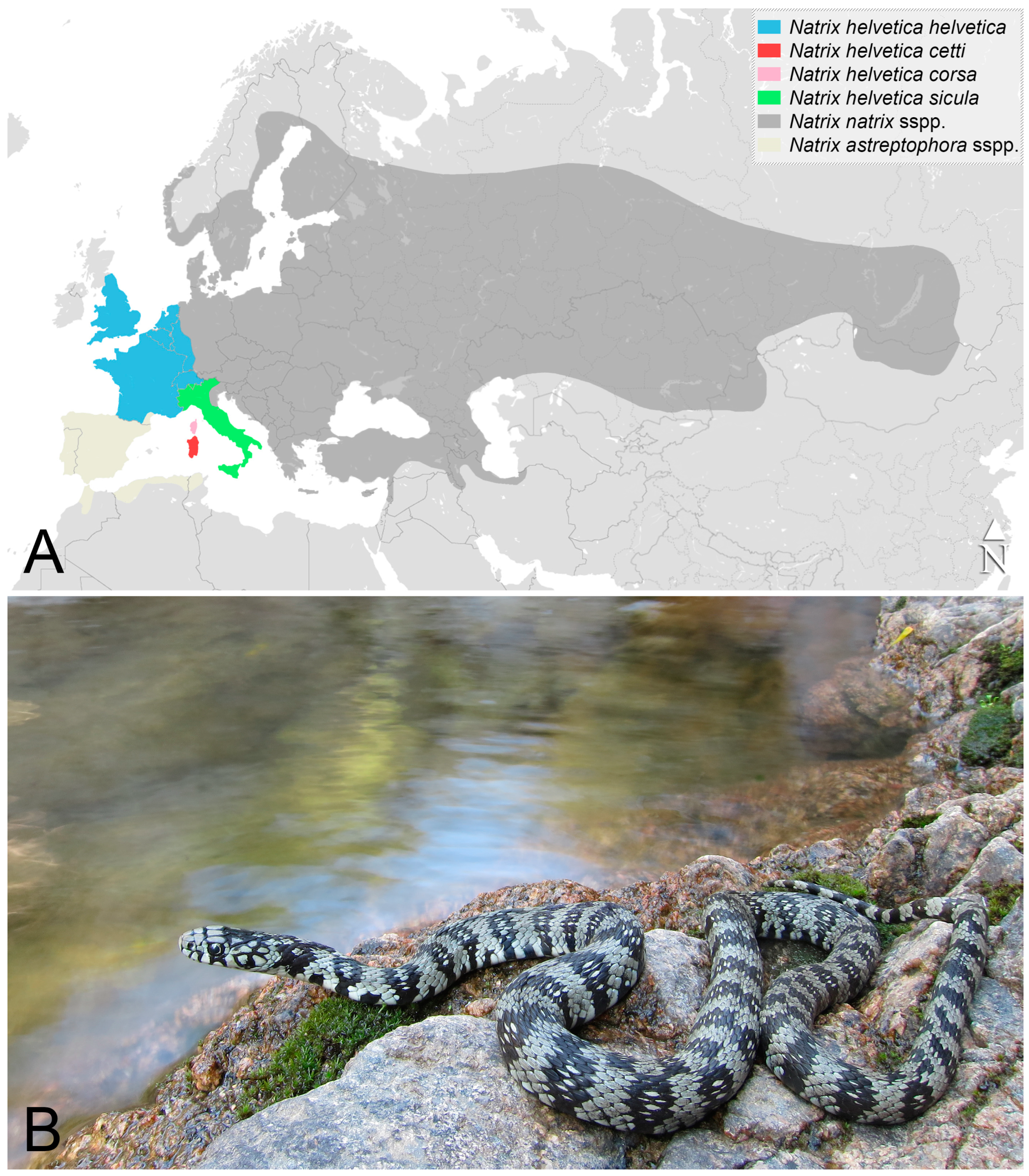
2. Materials and Methods
2.1. Natural History Review
2.2. Distribution Update
2.3. Habitat Suitability Estimation
2.4. Dorsal Pattern Characterisation
2.5. Principal Component Analysis
3. Results
3.1. Natural History Review
3.1.1. Taxonomy and Hypothesis on the Origins
3.1.2. Morphology
3.1.3. Ecology
3.1.4. Conservation
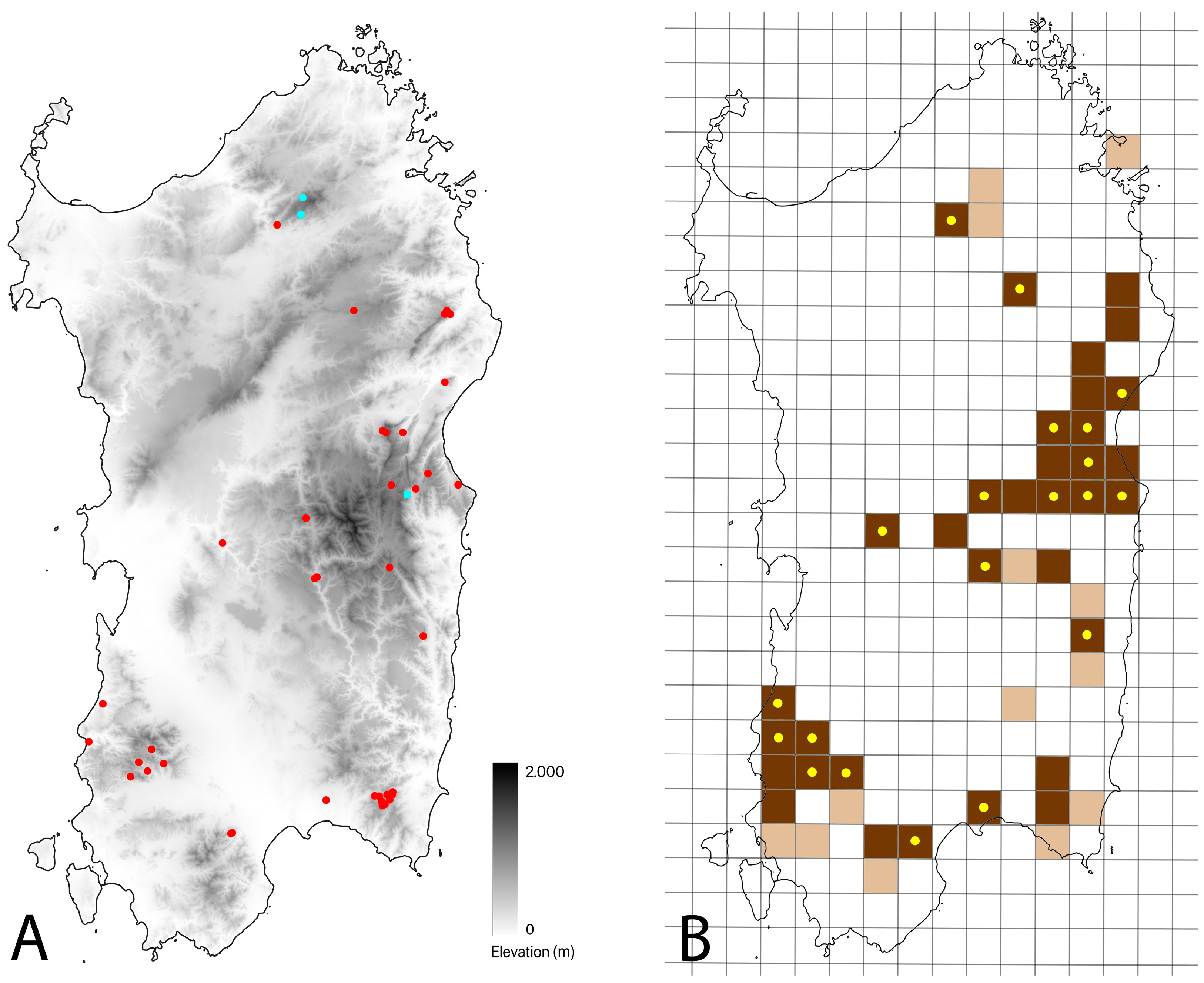
3.2. Distribution Update
3.3. Habitat Suitability Estimation
3.4. Dorsal Pattern Characterisation
3.5. Principal Component Analysis
4. Discussion
4.1. Natural History Review
4.2. Distribution Update
4.3. Habitat Suitability Estimation
4.4. Geographic Distribution of Potential Competitors and Trophic Resources
4.5. Geographic and Environmental Influence on Dorsal Pattern Variation
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Schmeller, D.S.; Gruber, B.; Budrys, E.; Framsted, E.; Lengyel, S.; Henle, K. National Responsibilities in European Species Conservation: A Methodological Review. Conserv. Biol. 2008, 22, 593–601. [Google Scholar] [CrossRef] [PubMed]
- Burlakova, L.E.; Karatayev, A.Y.; Karatayev, V.A.; May, M.E.; Bennett, D.L.; Cook, M.J. Endemic Species: Contribution to Community Uniqueness, Effect of Habitat Alteration, and Conservation Priorities. Biol. Conserv. 2011, 144, 155–165. [Google Scholar] [CrossRef]
- Le Galliard, J.F.; Massot, M.; Baron, J.-P.; Clobert, J. Ecological Effects of Climate Change on European Reptiles. Wildl. Conserv. Chang. Clim. 2012, 179, 203. [Google Scholar]
- Maiorano, L.; Amori, G.; Capula, M.; Falcucci, A.; Masi, M.; Montemaggiori, A.; Pottier, J.; Psomas, A.; Rondinini, C.; Russo, D.; et al. Threats from Climate Change to Terrestrial Vertebrate Hotspots in Europe. PLoS ONE 2013, 8, e74989. [Google Scholar] [CrossRef]
- Manes, S.; Costello, M.J.; Beckett, H.; Debnath, A.; Devenish-Nelson, E.; Grey, K.-A.; Jenkins, R.; Khan, T.M.; Kiessling, W.; Krause, C.; et al. Endemism Increases Species’ Climate Change Risk in Areas of Global Biodiversity Importance. Biol. Conserv. 2021, 257, 109070. [Google Scholar] [CrossRef]
- Syfert, M.M.; Joppa, L.; Smith, M.J.; Coomes, D.A.; Bachman, S.P.; Brummitt, N.A. Using Species Distribution Models to Inform IUCN Red List Assessments. Biol. Conserv. 2014, 177, 174–184. [Google Scholar] [CrossRef]
- Mota-Vargas, C.; Rojas-Soto, O.R. The Importance of Defining the Geographic Distribution of Species for Conservation: The Case of the Bearded Wood-Partridge. J. Nat. Conserv. 2012, 20, 10–17. [Google Scholar] [CrossRef]
- Marcer, A.; Sáez, L.; Molowny-Horas, R.; Pons, X.; Pino, J. Using Species Distribution Modelling to Disentangle Realised versus Potential Distributions for Rare Species Conservation. Biol. Conserv. 2013, 166, 221–230. [Google Scholar] [CrossRef]
- Guisan, A.; Tingley, R.; Baumgartner, J.B.; Naujokaitis-Lewis, I.; Sutcliffe, P.R.; Tulloch, A.I.T.; Regan, T.J.; Brotons, L.; McDonald-Madden, E.; Mantyka-Pringle, C.; et al. Predicting Species Distributions for Conservation Decisions. Ecol. Lett. 2013, 16, 1424–1435. [Google Scholar] [CrossRef]
- Mizsei, E.; Üveges, B.; Vági, B.; Szabolcs, M.; Lengyel, S.; Pfliegler, W.P.; Nagy, Z.T.; Tóth, J.P. Species Distribution Modelling Leads to the Discovery of New Populations of One of the Least Known European Snakes, Vipera ursinii graeca, in Albania. Amphibia-Reptilia 2016, 37, 55–68. [Google Scholar] [CrossRef]
- Camarda, I.; Falchi, S.; Nudda, G. Aspetti Geografici Della Sardegna Nell’ambito Del Mediterraneo. In L’ambiente Naturale in Sardegna; Libreria Della Natura: Milano, Italy, 1988; pp. 35–36. ISBN 978-88-7138-131-2. [Google Scholar]
- Corti, C.; Biaggini, M.; Nulchis, V.; Cogoni, R.; Cossu, I.M.; Frau, S.; Mulargia, M.; Lunghi, E.; Bassu, L. Species Diversity and Distribution of Amphibians and Reptiles in Sardinia, Italy. Acta Herpetol. 2022, 17, 125–133. [Google Scholar] [CrossRef]
- Blondel, J.; Aronson, J.; Bodiou, J.-Y.; Boeuf, G. (Eds.) The Mediterranean Region: Biological Diversity in Space and Time, 2nd ed.; Oxford Biology; Oxford University Press: Oxford, UK; New York, NY, USA, 2010; ISBN 978-0-19-955799-8. [Google Scholar]
- Salvi, D.; Bombi, P. Reptiles of Sardinia: Updating the Knowledge on Their Distribution. Acta Herpetol. 2010, 5, 161–177. [Google Scholar] [CrossRef]
- Di Nicola, M.R.; Mezzadri, S. Anfibi e Rettili Di Sardegna: Guida Fotografica; Libreria Della Natura: Milano, Italy, 2018; ISBN 88-909788-7-2. [Google Scholar]
- Sindaco, R.; Razzetti, E. An Updated Check-List of Italian Amphibians and Reptiles. Nat. Hist. Sci. 2021, 8, 35–46. [Google Scholar] [CrossRef]
- Faraone, F.P.; Melfi, R.; Di Nicola, M.R.; Giacalone, G.; Lo Valvo, M. Phylogenetic Relationships of the Italian Populations of Horseshoe Whip Snake Hemorrhois hippocrepis (Serpentes, Colubridae). Acta Herpetol. 2020, 15, 129–135. [Google Scholar] [CrossRef]
- Guicking, D.; Joger, U.; Wink, M. Molecular Phylogeography of the Viperine Snake Natrix maura (Serpentes: Colubridae): Evidence for Strong Intraspecific Differentiation. Org. Divers. Evol. 2008, 8, 130–145. [Google Scholar] [CrossRef][Green Version]
- Mezzasalma, M.; Dall’Asta, A.; Loy, A.; Cheylan, M.; Lymberakis, P.; Zuffi, M.A.L.; Tomović, L.; Odierna, G.; Guarino, F.M. A Sisters’ Story: Comparative Phylogeography and Taxonomy of Hierophis viridiflavus and H. gemonensis (Serpentes, Colubridae). Zool. Scr. 2015, 44, 495–508. [Google Scholar] [CrossRef]
- Fritz, U.; Corti, C.; Päckert, M. Mitochondrial DNA Sequences Suggest Unexpected Phylogenetic Position of Corso-Sardinian Grass Snakes (Natrix cetti) and Do Not Support Their Species Status, with Notes on Phylogeography and Subspecies Delineation of Grass Snakes. Org. Divers. Evol. 2012, 12, 71–80. [Google Scholar] [CrossRef]
- Schöneberg, Y.; Winter, S.; Arribas, O.; Di Nicola, M.R.; Master, M.; Owens, J.B.; Rovastos, M.; Wuster, W.; Janke, A.; Fritz, U. Genomics Reveals Broad Hybridization in Deeply Divergent Palearctic Grass and Water Snakes (Natrix spp.). Mol. Phylogenet. Evol. 2023, 184, 107787. [Google Scholar] [CrossRef]
- Schultze, N.; Spitzweg, C.; Corti, C.; Delaugerre, M.; Di Nicola, M.R.; Geniez, P.; Lapini, L.; Liuzzi, C.; Lunghi, E.; Novarini, N.; et al. Mitochondrial Ghost Lineages Blur Phylogeography and Taxonomy of Natrix helvetica and N. natrix in Italy and Corsica. Zool. Scr. 2020, 49, 395–411. [Google Scholar] [CrossRef]
- Fritz, U.; Schmidtler, J.F. The Fifth Labour of Heracles: Cleaning the Linnean Stable of Names for Grass Snakes (Natrix astreptophora, N helvetica, N. natrix Sensu Stricto). Vertebr. Zool. 2020, 70, 621–665. [Google Scholar] [CrossRef]
- Vanni, S.; Cimmaruta, R. Natrix cetti. In Fauna d’Italia Reptilia; Corti, C., Capula, M., Luiselli, L., Sindaco, R., Razzetti, E., Eds.; Calderini: Bologna, Italy, 2010; Volume XLV, pp. 538–545. [Google Scholar]
- Di Nicola, M.R.; Cavigioli, L.; Luiselli, L.; Andreone, F. Anfibi & Rettili d’Italia. Edizione Aggiornata; Edizioni Belvedere: Latina, Italy, 2021. [Google Scholar]
- Rondinini, C.; Battistoni, A.; Teofili, C. Lista Rossa IUCN Dei Vertebrati Italiani 2022; Comitato Italiano IUCN e Ministero dell’Ambiente e della Sicurezza Energetica: Roma, Italy, 2022. [Google Scholar]
- Luiselli, L. Resource Partitioning and Interspecific Competition in Snakes: The Search for General Geographical and Guild Patterns. Oikos 2006, 114, 193–211. [Google Scholar] [CrossRef]
- Lunghi, E.; Giachello, S.; Mulargia, M.; Dore, P.P.; Cogoni, R.; Corti, C. Variability in the Dorsal Pattern of the Sardinian Grass Snake (Natrix natrix Cetti) with Notes on Its Ecology. Acta Herpetol. 2019, 14, 141–145. [Google Scholar] [CrossRef]
- Kindler, C.; Graciá, E.; Fritz, U. Extra-Mediterranean Glacial Refuges in Barred and Common Grass Snakes (Natrix helvetica, N. natrix). Sci. Rep. 2018, 8, 1821. [Google Scholar] [CrossRef] [PubMed]
- Gentilli, A.; Scali, S. Natrix natrix. In Atlante Degli Anfibi e Dei Rettili d’Italia/Atlas of Italian Amphibians and Reptiles. Societas Herpetologica Italica; Sindaco, R., Doria, G., Razzetti, E., Bernini, F., Eds.; Edizioni Polistampa: Firenze, Italy, 2006; pp. 560–565. [Google Scholar]
- Bassu, L.; Nulchis, V.; Satta, M.G.; Fresi, C.; Corti, C. Atlas of Amphibians and Reptiles of Sardinia—State of the Art and General Considerations. In Herpetologia Sardiniae; Corti, C., Ed.; Edizioni Belvedere: Latina, Italy, 2008; pp. 52–58. [Google Scholar]
- Corti, C.; Nistri, A.; Vanni, S.; Lanza, B. Atlante Erpetologico Della Sardegna, Risultati Preliminari. In Proceedings of the Atti I Congresso Nazionale Societas Herpetologica Italica, Turin, Italy, 2–6 October 1996; Bollettino Museo Regionale di Scienze Naturali: Turin, Italy, 2000; pp. 573–576. [Google Scholar]
- Bassu, L.; Nulchis, V.; Satta, M.G.; Fresi, C.; Corti, C. Atlas of Amphibians and Reptiles of Sardinia Part III, Reptilia. Anfibi e Rettili di Sardegna III, Reptilia. In Proceedings of the Atti IX Congresso Nazionale Della Societas Herpetologica Italica, Rende-Cosenza, Italy, 1–5 October 2018; Tipolitografia Pineta: Bari, Italy, 2013; pp. 108–113. [Google Scholar]
- de Pous, P.; Speybroeck, J.; Bogaerts, S.; Pasmans, F.; Beukema, W. A Contribution to the Atlas of the Terrestrial Herpetofauna of Sardinia. Herpetol. Notes 2012, 5, 391–405. [Google Scholar]
- Mulargia, M.; Corti, C.; Lunghi, E. The Herpetofauna of the Monte Albo, Sardinia (Italy). Russ. J. Herpetol. 2018, 25, 172. [Google Scholar] [CrossRef]
- Phillips, S.J.; Anderson, R.P.; Dudík, M.; Schapire, R.E.; Blair, M.E. Opening the Black Box: An Open-Source Release of Maxent. Ecography 2017, 40, 887–893. [Google Scholar] [CrossRef]
- Fick, S.E.; Hijmans, R.J. WorldClim 2: New 1-km Spatial Resolution Climate Surfaces for Global Land Areas. Int. J. Climatol. 2017, 37, 4302–4315. [Google Scholar] [CrossRef]
- Fielding, A.H.; Bell, J.F. A Review of Methods for the Assessment of Prediction Errors in Conservation Presence/Absence Models. Environ. Conserv. 1997, 24, 38–49. [Google Scholar] [CrossRef]
- Boria, R.A.; Olson, L.E.; Goodman, S.M.; Anderson, R.P. Spatial Filtering to Reduce Sampling Bias Can Improve the Performance of Ecological Niche Models. Ecol. Model. 2014, 275, 73–77. [Google Scholar] [CrossRef]
- R Development Core Team. R: A Language and Environment for Statistical Computing; R Foundation for Statistical Computing: Vienna, Austria, 2019. [Google Scholar]
- QGIS Development Team. QGIS Geographic Information System. Open Source Geospatial Foundation Project. 2020. Available online: http://qgis.osgeo.org (accessed on 5 June 2023).
- Sau, S.; Smolinský, R.; Martínková, N. Environment Drives Color Pattern Polymorphism in Sand Lizards beyond the Gloger’s Rule. J. Zool. 2023. [Google Scholar] [CrossRef]
- Storniolo, F.; Mangiacotti, M.; Zuffi, M.A.L.; Scali, S.; Sacchi, R. Large Scale Phenotypic Characterisation of Hierophis viridiflavus (Squamata: Serpentes): Climatic and Environmental Drivers Suggest the Role of Evolutionary Processes in a Polymorphic Species. Evol. Ecol. 2023, 37, 419–434. [Google Scholar] [CrossRef]
- Broennimann, O.; Ursenbacher, S.; Meyer, A.; Golay, P.; Monney, J.-C.; Schmocker, H.; Guisan, A.; Dubey, S. Influence of Climate on the Presence of Colour Polymorphism in Two Montane Reptile Species. Biol. Lett. 2014, 10, 20140638. [Google Scholar] [CrossRef] [PubMed]
- Thorpe, R.S. Multivariate Analysis of the Population Systematics of the Ringed Snake, Natrix natrix (L). Proc. R. Soc. Edinb. Sect. B Biol. Sci. 1979, 78, 1–62. [Google Scholar] [CrossRef]
- Lê, S.; Josse, J.; Husson, F. FactoMineR: An R Package for Multivariate Analysis. J. Stat. Softw. 2008, 25, 1–18. [Google Scholar] [CrossRef]
- Kassambara, A.; Mundt, F. Factoextra: Extract and Visualize the Results of Multivariate Data Analyses. R Package Version 1.0.7. 2020. Available online: https://cran.r-project.org/package=factoextra (accessed on 5 June 2023).
- Wickham, H. Ggplot2: Elegant Graphics for Data Analysis, 2nd ed.; Use R! Springer International Publishing: Cham, Switzerland, 2016; ISBN 978-3-319-24277-4. [Google Scholar]
- Geniez, P. Snakes of Europe, North Africa and the Middle East. A Photographic Guide; Princeton University Press: Princeton, NJ, USA, 2018. [Google Scholar]
- Zaher, H.; Murphy, R.W.; Arredondo, J.C.; Graboski, R.; Machado-Filho, P.R.; Mahlow, K.; Montingelli, G.G.; Quadros, A.B.; Orlov, N.L.; Wilkinson, M.; et al. Large-Scale Molecular Phylogeny, Morphology, Divergence-Time Estimation, and the Fossil Record of Advanced Caenophidian Snakes (Squamata: Serpentes). PLoS ONE 2019, 14, e0216148. [Google Scholar] [CrossRef]
- Pokrant, F.; Kindler, C.; Ivanov, M.; Cheylan, M.; Geniez, P.; Böhme, W.; Fritz, U. Integrative Taxonomy Provides Evidence for the Species Status of the Ibero-Maghrebian Grass Snake Natrix astreptophora. Biol. J. Linn. Soc. 2016, 118, 873–888. [Google Scholar] [CrossRef]
- Kindler, C.; Chèvre, M.; Ursenbacher, S.; Böhme, W.; Hille, A.; Jablonski, D.; Vamberger, M.; Fritz, U. Hybridization Patterns in Two Contact Zones of Grass Snakes Reveal a New Central European Snake Species. Sci. Rep. 2017, 7, 7378. [Google Scholar] [CrossRef]
- Schultze, N.; Laufer, H.; Kindler, C.; Fritz, U. Distribution and Hybridisation of Barred and Common Grass Snakes (Natrix helvetica, N. natrix) in Baden-Württemberg, South-Western Germany. Herpetozoa 2019, 32, 229–236. [Google Scholar] [CrossRef]
- Asztalos, M.; Schultze, N.; Ihlow, F.; Geniez, P.; Berroneau, M.; Delmas, C.; Guiller, G.; Legentilhomme, J.; Kindler, C.; Fritz, U. How Often Do They Do It? An in-Depth Analysis of the Hybrid Zone of Two Grass Snake Species (Natrix astreptophora and Natrix helvetica). Biol. J. Linn. Soc. 2020, 131, 756–773. [Google Scholar] [CrossRef]
- Asztalos, M.; Glaw, F.; Franzen, M.; Kindler, C.; Fritz, U. Transalpine Dispersal: Italian Barred Grass Snakes in Southernmost Bavaria—This Far but No Further! J. Zool. Syst. Evol. Res. 2021, 59, 1136–1148. [Google Scholar] [CrossRef]
- Speybroeck, J.; Beukema, W.; Dufresnes, C.; Fritz, U.; Jablonski, D.; Lymberakis, P.; Martínez-Solano, I.; Razzetti, E.; Vamberger, M.; Vences, M.; et al. Species List of the European Herpetofauna—2020 Update by the Taxonomic Committee of the Societas Europaea Herpetologica. Amphib. Reptil. 2020, 41, 139–189. [Google Scholar] [CrossRef]
- Fritz, U.; Ihlow, F. Citizen Science, Taxonomy and Grass Snakes: INaturalist Helps to Clarify Variation of Coloration and Pattern in Natrix natrix Subspecies. Vertebr. Zool. 2022, 72, 533–549. [Google Scholar] [CrossRef]
- Gené, C.G. Synopsis Reptilium: Sardiniae Indigenorum; Reale Academia Delle Scienze: Torino, Italy, 1839. [Google Scholar]
- Mertens, R.; Müller, L. Liste der Amphibien und Reptilien Europas. Senckenberg. Naturforschenden Ges. 1928, 41, 1–62. [Google Scholar]
- Leunis, J. Synopsis der Drei Naturreiche. I. Zoologie. Zweite Auflage; Hahn’sche Hofbuchhandlung: Hannover, Italy, 1860. [Google Scholar]
- Jan, G. Elenco Sistematico Degli Ofidi Descritti e Disegnati per L’iconografia Generale Edita Dal Prof. G. Jan Direttore Del Museo Civico Di Milano; Tip. di A. Lombardi: Milano, Italy, 1863. [Google Scholar]
- Camerano, L. Monografia Degli Ofidi Italiani. Parte Seconda. Colubridi e Monografia Dei Cheloni Italiani. Mem. R. Accad. Sci. Fis. Mat. Torino Ser. 1891, 2, 403–481. [Google Scholar]
- Hecht, G. Systematik, Ausbreitungsgeschichte und Ökologie der Europäischen Arten der Gattung Tropidonotus (Kuhl) H. Boie. Mitt. Zool. Mus. Berl. 1930, 16, 244–393. [Google Scholar]
- Mertens, R. Studien zur Eidonomie und Taxonomie der Ringelnatter-(Natrix natrix). Senckenberg. Naturforschenden Ges. 1947, 476, 1–38. [Google Scholar]
- Mertens, R. Die Amphibien und Reptilien Korsikas. Senckenberg. Biol. 1957, 38, 175–192. [Google Scholar]
- Thorpe, R.S. Intraspecific Variation of the Ringed Snake, Natrix natrix (L.). Ph. D. Thesis, Council for National Academic Awards, London, UK, 1973. [Google Scholar]
- Thorpe, R.S. Biometric Analysis of Incipient Speciation in the Ringed Snake, Natrix natrix (L.). Experientia 1975, 31, 180–182. [Google Scholar] [CrossRef]
- Thorpe, R.S. Quantitative Handling of Characters Useful in Snake Systematics with Particular Reference to Intraspecific Variation in the Ringed Snake Natrix natrix (L.). Biol. J. Linn. Soc. 1975, 7, 27–43. [Google Scholar] [CrossRef]
- Thorpe, R.S. Microevolution and Taxonomy of European Reptiles with Particular Reference to the Grass Snake Natrix natrix and the Wall Lizards Podarcis sicula and P. melisellensis. Biol. J. Linn. Soc. 1980, 14, 215–233. [Google Scholar] [CrossRef]
- Lanza, B. Guide per il Riconoscimento Delle Specie Animali Nelle Acque Interne Italiane. 27. Anfibi, Rettili (Amphibia, Reptilia); Collana del Progetto Finalizzato “Promozione della Qualità dell’Ambiente” AQ/1/205; Consiglio Nazionale delle Ricerche: Roma, Italy, 1983; pp. 156–185. [Google Scholar]
- Aprea, G.; Odierna, G.; Capriglione, T.; Caputo, V.; Guarino, F.M. Analisi Cromosomica in Tre Specie del Genere Natrix Duméril (Reptilia, Squamata). In Atti del I Congresso Nazionale della Societas Herpetologica Italica, Torino, 1996; Museo Regionale di Scienze Nataturali: Turin, Italy, 2000; pp. 423–428. [Google Scholar]
- Kindler, C.; Böhme, W.; Corti, C.; Gvoždík, V.; Jablonski, D.; Jandzik, D.; Metallinou, M.; Široký, P.; Fritz, U. Mitochondrial Phylogeography, Contact Zones and Taxonomy of Grass Snakes (Natrix natrix, N. megalocephala). Zool. Scr. 2013, 42, 458–472. [Google Scholar] [CrossRef]
- Burbrink, F.T.; Grazziotin, F.G.; Pyron, R.A.; Cundall, D.; Donnellan, S.; Irish, F.; Keogh, J.S.; Kraus, F.; Murphy, R.W.; Noonan, B.; et al. Interrogating Genomic-Scale Data for Squamata (Lizards, Snakes, and Amphisbaenians) Shows No Support for Key Traditional Morphological Relationships. Syst. Biol. 2020, 69, 502–520. [Google Scholar] [CrossRef]
- Deepak, V.; Cooper, N.; Poyarkov, N.A.; Kraus, F.; Burin, G.; Das, A.; Narayanan, S.; Streicher, J.W.; Smith, S.-J.; Gower, D.J. Multilocus Phylogeny, Natural History Traits and Classification of Natricine Snakes (Serpentes: Natricinae). Zool. J. Linn. Soc. 2022, 195, 279–298. [Google Scholar] [CrossRef]
- Lanza, B. Ipotesi Sulle Origini Del Popolamento Erpetologico della Sardegna. Lavori della Società Italiana di Biogeografia. Biogeogr. J. Integr. Biogeogr. 1983, 8, 723–744. [Google Scholar] [CrossRef]
- Lanza, B. Hypotheses on the Origins of Corsican Herpetofauna (p. 2). In Proceedings of the Colloque International sur les Vertébrés Terrestres et Dulçaquicoles des Îles Méditerranéennes, Evisa, France, 10–16 October 1983; p. 53. [Google Scholar]
- Lanza, B. Hypothèses Sur Les Origines de la Faune Herpétologique Corse. Actes du Colloque International Sur Les Vertébrés Terrestres et Dulçaquicoles des Îles Méditerranéennes, Evisa (Corse), 10–16 October 1983. Bull. Ecol. 1988, 19, 163–170. [Google Scholar]
- Garcia-Castellanos, D.; Estrada, F.; Jiménez-Munt, I.; Gorini, C.; Fernàndez, M.; Vergés, J.; De Vicente, R. Catastrophic Flood of the Mediterranean after the Messinian Salinity Crisis. Nature 2009, 462, 778–781. [Google Scholar] [CrossRef] [PubMed]
- Lambeck, K.; Antonioli, F.; Purcell, A.; Silenzi, S. Sea-Level Change along the Italian Coast for the Past 10,000 yr. Quat. Sci. Rev. 2004, 23, 1567–1598. [Google Scholar] [CrossRef]
- Marchetti, M.; Soldati, M.; Vandelli, V. The Great Diversity of Italian Landscapes and Landforms: Their Origin and Human Imprint. In Landscapes and Landforms of Italy; Soldati, M., Marchetti, M., Eds.; Springer International Publishing: Cham, Switzerland, 2017; pp. 7–20. ISBN 978-3-319-26192-8. [Google Scholar]
- Lunghi, E.; Deschandol, F.; Cornago, L.; Cogoni, R. Dark Coloration in Sardinian Grass Snakes (Natrix natrix cetti). Herpetol. Bull. 2016, 137, 28–29. [Google Scholar]
- Bruni, G.; Di Nicola, M.R.; Banfi, F.; Faraone, F.P. Distribution and Characterization of Melanism in Grass Snakes from Italy. Nat. Sicil. 2022, 46, 41–48. [Google Scholar] [CrossRef]
- Capula, M.; Rugiero, L.; Luiselli, L. Ecological Observations on the Sardinian Grass Snake, Natrix natrix cetti. Amphib. Reptil. 1994, 15, 221–224. [Google Scholar] [CrossRef]
- Scali, S.; Gentilli, A.; Lanza, B. Natrix natrix. In Fauna d’Italia, Reptilia; Corti, C., Capula, M., Luiselli, L., Sindaco, R., Razzetti, E., Eds.; Calderini: Bologna, Italy, 2010; Volume 45, pp. 552–562. [Google Scholar]
- Lunghi, E.; Mascia, C.; Mulargia, M.; Corti, C. Is the Sardinian Grass Snake (Natrix natrix cetti) an Active Hunter in Underground Environments. Spixiana 2018, 41, 160. [Google Scholar]
- Stefani, R. La Natrice Del Cetti. Biogeogr. J. Integr. Biogeogr. 1983, 8, 745–755. [Google Scholar] [CrossRef]
- Lanza, B.; Camarda, I.; Falchi, S.; Nudda, G. (Eds.) I Rettili e Gli Anfibi. In L’ambiente Naturale in Sardegna (Elementi di Base per la Conoscenza e la Gestione del Territorio); Carlo Delfino Editore: Sassari, Italy, 1986. [Google Scholar]
- Mebert, K.; Trapp, B.; Kreiner, G.; Billing, H.; Speybroeck, J.; Henggeler, M. Nocturnal Activity in Natrix tessellata, a Neglected Aspect of Its Behavioral Repetoire. Mertensiella 2011, 18, 234–236. [Google Scholar]
- Luiselli, L.; Filippi, E.; Capula, M. Geographic Variation in Diet Composition of the Grass Snake (Natrix natrix) along the Mainland and an Island of Italy: The Effects of Habitat Type and Interference with Potential Competitors. Herpetol. J. 2005, 15, 221–230. [Google Scholar]
- Filippi, E.; Capula, M.; Luiselli, L.; Agrimi, U. The Prey Spectrum of Natrix natrix (Linnaeus, 1758) and Natrix tessellata (Laurenti, 1768) in Sympatric Populations. Herpetozoa 1996, 8, 155–164. [Google Scholar]
- Reading, C.J.; Davies, J.L. Predation by Grass Snakes (Natrix natrix) at a Site in Southern England. J. Zool. 1996, 239, 73–82. [Google Scholar] [CrossRef]
- Gregory, P.T.; Isaac, L.A. Food Habits of the Grass Snake in Southeastern England: Is Natrix natrix a Generalist Predator? J. Herpetol. 2004, 38, 88–95. [Google Scholar] [CrossRef]
- Faraone, F.P.; Giacalone, G.; Lo Valvo, M. Dati Preliminari Sulla Biometria, Il Cromatismo e la Dieta di una Popolazione di Natrix natrix della Sicilia Occidentale. In Proceedings of the Atti VIII Congresso Nazionale Societas Herpetologica Italica, Chieti, Italy, 22–26 September 2010; pp. 247–252. [Google Scholar]
- Šukalo, G.; Đorđević, S.; Gvozdenović, S.; Simović, A.; Anđelković, M.; Blagojević, V.; Tomović, L. Intra-and Inter-Population Variability of Food Preferences of Two Natrix Species on the Balkan Peninsula. Herpetol. Conserv. Biol. 2014, 9, 123–136. [Google Scholar]
- Di Nicola, M.R.; Bruni, G. A Case of Active Predation of Natrix helvetica (Serpentes: Colubridae) on Sturnus vulgaris (Passeriformes: Sturnidae). Herpetol. Notes 2020, 13, 461–462. [Google Scholar]
- Di Nicola, M.R.; Zabbia, T. Natrix helvetica (Barred Grass Snake). Diet. Herpetol. Rev. 2021, 52, 877–878. [Google Scholar]
- Luiselli, L.; Rugiero, L. Food Niche Partitioning by Water Snakes (Genus Natrix) at a Freshwater Environment in Central Italy. J. Freshw. Ecol. 1991, 6, 439–444. [Google Scholar] [CrossRef]
- Kaczmarski, M. Arboreal Foraging and Ambush by Grass Snakes Natrix natrix on European Treefrogs Hyla Arborea. Herpetol. Bull. 2020, 154, 39–40. [Google Scholar] [CrossRef]
- Puddu, F.; Viarengo, M.; Erminio, C. Animali di Sardegna. Gli Anfibi e i Rettili; Edizioni Della Torre: Cagliari, Italy, 1988; ISBN 0-01-870169-8. [Google Scholar]
- Lanza, B.; Pastorelli, C.; Laghi, P.; Cimmaruta, R. A Review of Systematics, Taxonomy, Genetics, Biogeography and Natural History of the Genus Speleomantes Dubois, 1984 (Amphibia Caudata Plethodontidae). Atti Mus. Civ. Stor. Nat. Trieste 2006, 52, 5–135. [Google Scholar]
- Van Rooy, P.T.; Stumpel, A.H. Ecological Impact of Economic Development on Sardinian Herpetofauna. Conserv. Biol. 1995, 9, 263–269. [Google Scholar] [CrossRef]
- Luiselli, L. Individual Success in Mating Balls of the Grass Snake, Natrix natrix: Size Is Important. J. Zool. 1996, 239, 731–740. [Google Scholar] [CrossRef]
- Cauli, F.; Di Nicola, M.R.; Audisio, P.; Petretti, F.; Faraone, F.P. Feeding Habits of the Short-Toed Eagle Circaetus gallicus during the Breeding Period in Central Italy. Avocetta 2022, 46, 1. [Google Scholar] [CrossRef]
- Filippi, E.; Luiselli, L. Negative Effect of the Wild Boar (Sus scrofa) on the Populations of Snakes at a Protected Mountainous Forest in Central Italy. Ecol. Mediterr. 2002, 28, 93–98. [Google Scholar] [CrossRef]
- Schoenfeld, M.; Yom-Tov, Y. The Biology of Two Species of Hedgehogs, Erinaceus europaeus Concolor and Hemiechinus auritus aegyptius, in Israel. Mammalia 1985, 49, 339–356. [Google Scholar] [CrossRef]
- Di Nicola, M.R.; Costa, W.; Mori, E. An Evidence of Asp Viper (Vipera aspis) Consumption by a Western European Hedgehog (Erinaceus europaeus) on Elba Island (Italy). Nat. Hist. Sci. 2021, 9, 35–38. [Google Scholar] [CrossRef]
- Petrilla, V.; Flešárová, S.; Šulla, I. Poison Apparatus in Snakes. Folia Vet. 2008, 52, 22–23. [Google Scholar]
- Di Nicola, M.R.; Chiara, R.; Colnaghi, S.; Valvo, M.L.; Faraone, F.P. First Documented Cases of Defensive Biting Behaviour towards Humans in the Italian Barred Grass Snake Natrix helvetica sicula (Cuvier, 1829). Herpetol. Notes 2023, 16, 229–232. [Google Scholar]
- European Reptile & Amphibians Specialist Group. Natrix natrix ssp. Cetti. The IUCN Red List of Threatened Species. 1996, e.T14364A4436077. Available online: https://doi.org/10.2305/IUCN.UK.1996.RLTS.T14364A4436077.en (accessed on 17 August 2023).
- Ozinga, W.A.; de Heer, M.; Hennekens, S.M.; van Opstal, A.; Schaminee, J.H.J.; Sierdsema, H.; Smits, N.A.C.; Stumpel, A.H.P.; van Swaay, C. Target Species-Species of European Concern; a Database Driven Selection of Plant and Animal Species for the Implementation of the Pan European Ecological Network; Alterra: Wageningen, The Netherlands, 2005. [Google Scholar]
- Andreone, F.; Corti, C.; Ficetola, G.F.; Razzetti, E.; Romano, A.; Sindaco, R. Natrix natrix ssp. cetti; IUCN Comitato Italiano: Grand, Switzerland, 2013. [Google Scholar]
- Krijgsman, W.; Hilgen, F.J.; Raffi, I.; Sierro, F.J.; Wilson, D.S. Chronology, Causes and Progression of the Messinian Salinity Crisis. Nature 1999, 400, 652–655. [Google Scholar] [CrossRef]
- Schätti, B. Bemerkungen zur Ökologie, Verbreitung und Intraspezifischen Variation der Vipernatter, Natrix maura (Linné, 1758) (Reptilia, Serpentes). Rev. Suisse Zool. 1982, 89, 521–542. [Google Scholar] [CrossRef]
- Poggesi, M.; Agnelli, P.; Borri, M.; Corti, C.; Finotello, P.; Lanza, B.; Tosini, G. Erpetologia delle Isole Circumsarde. Biogeogr. J. Integr. Biogeogr. 1996, 18, 583–618. [Google Scholar] [CrossRef]
- Guicking, D.; Griffiths, R.A.; Moore, R.D.; Joger, U.; Wink, M. Introduced Alien or Persecuted Native? Resolving the Origin of the Viperine Snake (Natrix maura) on Mallorca. Biodivers. Conserv. 2006, 15, 3045–3054. [Google Scholar] [CrossRef]
- Rugiero, L.; Capula, M.; Persichetti, D.; Luiselli, L.; Angelici, F.M. Life-History and Diet of Two Populations of Natrix maura (Reptilia, Colubridae) from Contrasted Habitats in Sardinia. Miscel Lània Zool. 2000, 23, 41–51. [Google Scholar]
- European Environment Agency. Natrix natrix cetti; European Environment Agency: Copenhagen, Denmark, 2012. [Google Scholar]
- Falcucci, A.; Maiorano, L.; Boitani, L. Changes in Land-Use/Land-Cover Patterns in Italy and Their Implications for Biodiversity Conservation. Landsc. Ecol. 2007, 22, 617–631. [Google Scholar] [CrossRef]
- Puddu, G.; Falcucci, A.; Maiorano, L. Forest Changes over a Century in Sardinia: Implications for Conservation in a Mediterranean Hotspot. Agrofor. Syst. 2012, 85, 319–330. [Google Scholar] [CrossRef]
- Fiorini, L.; Zullo, F.; Romano, B. Urban Development of the Coastal System of the Italian Largest Islands: Sicily and Sardinia. Ocean Coast. Manag. 2017, 143, 184–194. [Google Scholar] [CrossRef]
- Reed, D.H.; Frankham, R. Correlation between Fitness and Genetic Diversity. Conserv. Biol. 2003, 17, 230–237. [Google Scholar] [CrossRef]
- Charlesworth, D.; Willis, J.H. The Genetics of Inbreeding Depression. Nat. Rev. Genet. 2009, 10, 783–796. [Google Scholar] [CrossRef] [PubMed]
- Frankham, R. Genetic Rescue of Small Inbred Populations: Meta-Analysis Reveals Large and Consistent Benefits of Gene Flow. Mol. Ecol. 2015, 24, 2610–2618. [Google Scholar] [CrossRef] [PubMed]
- Phillipsen, I.C.; Kirk, E.H.; Bogan, M.T.; Mims, M.C.; Olden, J.D.; Lytle, D.A. Dispersal Ability and Habitat Requirements Determine Landscape-Level Genetic Patterns in Desert Aquatic Insects. Mol. Ecol. 2015, 24, 54–69. [Google Scholar] [CrossRef] [PubMed]
- Di Nicola, M.R.; Ortiz-Santaliestra, M.E.; Bosch, J.; Pasmans, F.; Martel, A.; Focks, A.; Dorne, J.L.C.M.; Marini, D. Indagine Sull’ofidiomicosi Nei Serpenti Italiani Nell’ambito del Progetto EFSA “AMPHIDEB”. In Proceedings of the XIV Congresso Nazionale Societas Herpetologica Italica, Turin, Italy, 13–17 September 2022. [Google Scholar]
- Di Nicola, M.R.; Coppari, L.; Notomista, T.; Marini, D. Ophidiomyces ophidiicola Detection and Infection: A Global Review on a Potential Threat to the World’s Snake Populations. Eur. J. Wildl. Res. 2022, 68, 64. [Google Scholar] [CrossRef]
- Marini, D.; Di Nicola, M.R.; Crocchianti, V.; Notomista, T.; Iversen, D.; Coppari, L.; Di Criscio, M.; Brouard, V.; Dorne, J.-L.C.M.; Rüegg, J.; et al. Pilot Survey Reveals Ophidiomycosis in Dice Snakes Natrix tessellata from Lake Garda, Italy. Vet. Res. Commun. 2023. Online ahead of print. [Google Scholar] [CrossRef]
- Mace, G.M.; Collar, N.J.; Gaston, K.J.; Hilton-Taylor, C.; Akçakaya, H.R.; Leader-Williams, N.; Milner-Gulland, E.J.; Stuart, S.N. Quantification of Extinction Risk: IUCN’s System for Classifying Threatened Species. Conserv. Biol. 2008, 22, 1424–1442. [Google Scholar] [CrossRef]
- Gaston, K.J.; Fuller, R.A. The Sizes of Species’ Geographic Ranges. J. Appl. Ecol. 2009, 46, 1–9. [Google Scholar] [CrossRef]
- Margules, C.R.; Pressey, R.L. Systematic Conservation Planning. Nature 2000, 405, 243–253. [Google Scholar] [CrossRef]
- Trindade-Filho, J.; de Carvalho, R.A.; Brito, D.; Loyola, R.D. How Does the Inclusion of Data Deficient Species Change Conservation Priorities for Amphibians in the Atlantic Forest? Biodivers. Conserv. 2012, 21, 2709–2718. [Google Scholar] [CrossRef]
- Royle, J.A.; Link, W.A. Generalized Site Occupancy Models Allowing for False Positive and False Negative Errors. Ecology 2006, 87, 835–841. [Google Scholar] [CrossRef]
- Durso, A.M.; Willson, J.D.; Winne, C.T. Needles in Haystacks: Estimating Detection Probability and Occupancy of Rare and Cryptic Snakes. Biol. Conserv. 2011, 144, 1508–1515. [Google Scholar] [CrossRef]
- McCarthy, M.A.; Moore, J.L.; Morris, W.K.; Parris, K.M.; Garrard, G.E.; Vesk, P.A.; Rumpff, L.; Giljohann, K.M.; Camac, J.S.; Bau, S.S. The Influence of Abundance on Detectability. Oikos 2013, 122, 717–726. [Google Scholar] [CrossRef]
- Pacifici, K.; Reich, B.J.; Dorazio, R.M.; Conroy, M.J. Occupancy Estimation for Rare Species Using a Spatially-adaptive Sampling Design. Methods Ecol. Evol. 2016, 7, 285–293. [Google Scholar] [CrossRef]
- Gu, W.; Swihart, R.K. Absent or Undetected? Effects of Non-Detection of Species Occurrence on Wildlife–Habitat Models. Biol. Conserv. 2004, 116, 195–203. [Google Scholar] [CrossRef]
- Dyugmedzhiev, A.; Naumov, B.; Tzankov, N. Sex-and Age-Related Variations in Seasonal and Circadian Activity of the Nose-Horned Viper Vipera ammodytes (Linnaeus, 1758). Belg. J. Zool. 2022, 152, 139–156. [Google Scholar] [CrossRef]
- Wilson, J.S.; Pan, A.D.; General, D.E.M.; Koch, J.B. More Eyes on the Prize: An Observation of a Very Rare, Threatened Species of Philippine Bumble Bee, Bombus irisanensis, on INaturalist and the Importance of Citizen Science in Conservation Biology. J. Insect Conserv. 2020, 24, 727–729. [Google Scholar] [CrossRef]
- Mesaglio, T.; Soh, A.; Kurniawidjaja, S.; Sexton, C. ‘First Known Photographs of Living Specimens’: The Power of INaturalist for Recording Rare Tropical Butterflies. J. Insect Conserv. 2021, 25, 905–911. [Google Scholar] [CrossRef]
- Roberts, C.J.; Vergés, A.; Callaghan, C.T.; Poore, A.G.B. Many Cameras Make Light Work: Opportunistic Photographs of Rare Species in INaturalist Complement Structured Surveys of Reef Fish to Better Understand Species Richness. Biodivers. Conserv. 2022, 31, 1407–1425. [Google Scholar] [CrossRef]
- Gaier, A.G.; Resasco, J. Does Adding Community Science Observations to Museum Records Improve Distribution Modeling of a Rare Endemic Plant? Ecosphere 2023, 14, e4419. [Google Scholar] [CrossRef]
- Balletto, G.; Borruso, G.; Ladu, M.; Milesi, A. Smart and Slow Tourism. Evaluation and Challenges in Sardinia (Italy). In Innovation in Urban and Regional Planning; La Rosa, D., Privitera, R., Eds.; Springer International Publishing: Cham, Switzerland, 2022; Volume 242, pp. 175–182. ISBN 978-3-030-96984-4. [Google Scholar]
- Bussu, E. Sardegna, Un Turismo Con Un Futuro Diverso. AMMENTU-Boll. Stor. Arch. Mediterr. Americhe 2022, 1, 283–296. [Google Scholar]
- Guisan, A.; Broennimann, O.; Engler, R.; Vust, M.; Yoccoz, N.G.; Lehmann, A.; Zimmermann, N.E. Using Niche-based Models to Improve the Sampling of Rare Species. Conserv. Biol. 2006, 20, 501–511. [Google Scholar] [CrossRef] [PubMed]
- Marino, J.; Bennett, M.; Cossios, D.; Iriarte, A.; Lucherini, M.; Pliscoff, P.; Sillero-Zubiri, C.; Villalba, L.; Walker, S. Bioclimatic Constraints to Andean Cat Distribution: A Modelling Application for Rare Species. Divers. Distrib. 2011, 17, 311–322. [Google Scholar] [CrossRef]
- Ferrer-Sánchez, Y.; Rodríguez-Estrella, R. How Rare Species Conservation Management Can Be Strengthened with the Use of Ecological Niche Modelling: The Case for Endangered Endemic Gundlach’s Hawk and Cuban Black-Hawk. Glob. Ecol. Conserv. 2016, 5, 88–99. [Google Scholar] [CrossRef]
- Pedrotta, T.; Gobet, E.; Schwörer, C.; Beffa, G.; Butz, C.; Henne, P.D.; Morales-Molino, C.; Pasta, S.; van Leeuwen, J.F.; Vogel, H. 8000 Years of Climate, Vegetation, Fire and Land-Use Dynamics in the Thermo-Mediterranean Vegetation Belt of Northern Sardinia (Italy). Veg. Hist. Archaeobot. 2021, 30, 789–813. [Google Scholar] [CrossRef]
- Caloiero, T.; Coscarelli, R.; Gaudio, R. Spatial and Temporal Variability of Daily Precipitation Concentration in the Sardinia Region (Italy). Int. J. Climatol. 2019, 39, 5006–5021. [Google Scholar] [CrossRef]
- Rodríguez, M.Á.; Belmontes, J.A.; Hawkins, B.A. Energy, Water and Large-Scale Patterns of Reptile and Amphibian Species Richness in Europe. Acta Oecologica 2005, 28, 65–70. [Google Scholar] [CrossRef]
- Detto, M.; Montaldo, N.; Albertson, J.D.; Mancini, M.; Katul, G. Soil Moisture and Vegetation Controls on Evapotranspiration in a Heterogeneous Mediterranean Ecosystem on Sardinia, Italy. Water Resour. Res. 2006, 42, W08419. [Google Scholar] [CrossRef]
- Montaldo, N.; Corona, R.; Albertson, J.D. On the Separate Effects of Soil and Land Cover on Mediterranean Ecohydrology: Two Contrasting Case Studies in Sardinia, Italy. Water Resour. Res. 2013, 49, 1123–1136. [Google Scholar] [CrossRef]
- Kobayashi, T.; Tateishi, R.; Alsaaideh, B.; Sharma, R.C.; Wakaizumi, T.; Miyamoto, D.; Bai, X.; Long, B.D.; Gegentana, G.; Maitiniyazi, A.; et al. Production of Global Land Cover Data—GLCNMO2013. J. Geogr. Geol. 2017, 9, 1. [Google Scholar] [CrossRef]
- Bird, D.N.; Benabdallah, S.; Gouda, N.; Hummel, F.; Koeberl, J.; La Jeunesse, I.; Meyer, S.; Prettenthaler, F.; Soddu, A.; Woess-Gallasch, S. Modelling Climate Change Impacts on and Adaptation Strategies for Agriculture in Sardinia and Tunisia Using AquaCrop and Value-at-Risk. Sci. Total Environ. 2016, 543, 1019–1027. [Google Scholar] [CrossRef]
- Lai, S.; Leone, F.; Zoppi, C. Spatial Distribution of Surface Temperature and Land Cover: A Study Concerning Sardinia, Italy. Sustainability 2020, 12, 3186. [Google Scholar] [CrossRef]
- Caloiero, T.; Guagliardi, I. Climate Change Assessment: Seasonal and Annual Temperature Analysis Trends in the Sardinia Region (Italy). Arab. J. Geosci. 2021, 14, 2149. [Google Scholar] [CrossRef]
- Cipolla, S.S.; Montaldo, N. On the Impacts of Historical and Future Climate Changes to the Sustainability of the Main Sardinian Forests. Remote Sens. 2022, 14, 4893. [Google Scholar] [CrossRef]
- Gentilli, A.; Scali, S. Natrix maura. In Atlante Degli Anfibi e dei Rettili D’italia/Atlas of Italian Amphibians and Reptiles. Societas Herpetologica Italica; Sindaco, R., Doria, G., Razzetti, E., Bernini, F., Eds.; Edizioni Polistampa: Firenze, Italy, 2006; pp. 556–559. [Google Scholar]
- Martínez-Freiría, F.; i de Lanuza, G.P.; Pimenta, A.A.; Pinto, T.; Santos, X. Aposematism and Crypsis Are Not Enough to Explain Dorsal Polymorphism in the Iberian Adder. Acta Oecologica 2017, 85, 165–173. [Google Scholar] [CrossRef]
- Souchet, J.; Bossu, C.; Darnet, E.; Le Chevalier, H.; Poignet, M.; Trochet, A.; Bertrand, R.; Calvez, O.; Martinez-Silvestre, A.; Mossoll-Torres, M.; et al. High Temperatures Limit Developmental Resilience to High-Elevation Hypoxia in the Snake Natrix maura (Squamata: Colubridae). Biol. J. Linn. Soc. 2021, 132, 116–133. [Google Scholar] [CrossRef]
- Wüster, W.; Allum, C.S.; Bjargardottir, I.B.; Bailey, K.L.; Dawson, K.J.; Guenioui, J.; Lewis, J.; McGurk, J.; Moore, A.G.; Niskanen, M. Do Aposematism and Batesian Mimicry Require Bright Colours? A Test, Using European Viper Markings. Proc. R. Soc. Lond. B Biol. Sci. 2004, 271, 2495–2499. [Google Scholar] [CrossRef] [PubMed]
- Valkonen, J.K.; Nokelainen, O.; Mappes, J. Antipredatory Function of Head Shape for Vipers and Their Mimics. PLoS ONE 2011, 6, e22272. [Google Scholar] [CrossRef]
- Allen, W.L.; Baddeley, R.; Scott-Samuel, N.E.; Cuthill, I.C. The Evolution and Function of Pattern Diversity in Snakes. Behav. Ecol. 2013, 24, 1237–1250. [Google Scholar] [CrossRef]
- Rajabizadeh, M.; Adriaens, D.; Kaboli, M.; Sarafraz, J.; Ahmadi, M. Dorsal Colour Pattern Variation in Eurasian Mountain Vipers (Genus Montivipera): A Trade-off between Thermoregulation and Crypsis. Zool. Anz. J. Comp. Zool. 2015, 257, 1–9. [Google Scholar] [CrossRef]
- Santos, X.; Azor, J.S.; Cortés, S.; Rodríguez, E.; Larios, J.; Pleguezuelos, J.M. Ecological Significance of Dorsal Polymorphism in a Batesian Mimic Snake. Curr. Zool. 2018, 64, 745–753. [Google Scholar] [CrossRef]
- Pizzigalli, C.; Banfi, F.; Ficetola, G.F.; Falaschi, M.; Mangiacotti, M.; Sacchi, R.; Zuffi, M.A.; Scali, S. Eco-Geographical Determinants of the Evolution of Ornamentation in Vipers. Biol. J. Linn. Soc. 2020, 130, 345–358. [Google Scholar] [CrossRef]
- Martínez-Freiría, F.; Toyama, K.S.; Freitas, I.; Kaliontzopoulou, A. Thermal Melanism Explains Macroevolutionary Variation of Dorsal Pigmentation in Eurasian Vipers. Sci. Rep. 2020, 10, 16122. [Google Scholar] [CrossRef] [PubMed]
- Andrén, C.; Nilson, G. Reproductive Success and Risk of Predation in Normal and Melanistic Colour Morphs of the Adder, Vipera berus. Biol. J. Linn. Soc. 1981, 15, 235–246. [Google Scholar] [CrossRef]
- Bury, S.; Mazgajski, T.D.; Najbar, B.; Zając, B.; Kurek, K. Melanism, Body Size, and Sex Ratio in Snakes—New Data on the Grass Snake (Natrix natrix) and Synthesis. Sci. Nat. 2020, 107, 22. [Google Scholar] [CrossRef] [PubMed]
- Venczel, M.; Sanchiz, B. Lower Miocene Amphibians and Reptiles from Oschiri (Sardinia, Italy). Hantkeniana 2006, 5, 72–75. [Google Scholar]
- Delfino, M.; Bailon, S.; Pitruzzella, G. The Late Pliocene Amphibians and Reptiles from “Capo Mannu D1 Local Fauna”(Mandriola, Sardinia, Italy). Geodiversitas 2011, 33, 357–382. [Google Scholar] [CrossRef]
- Jackson, J.F.; Ingram, W., III; Campbell, H.W. The Dorsal Pigmentation Pattern of Snakes as an Antipredator Strategy: A Multivariate Approach. Am. Nat. 1976, 110, 1029–1053. [Google Scholar] [CrossRef]
- Ford, E.B. Genetic Polymorphism. Proc. R. Soc. Lond. B Biol. Sci. 1966, 164, 350–361. [Google Scholar]
- Cox, C.L.; Davis Rabosky, A.R. Spatial and Temporal Drivers of Phenotypic Diversity in Polymorphic Snakes. Am. Nat. 2013, 182, E40–E57. [Google Scholar] [CrossRef]
- Schielzeth, H. Phylogenetic, Geographic and Habitat Distribution of the Green-Brown Polymorphism in European Orthopterans. Evol. Biol. 2020. [Google Scholar] [CrossRef]
- Matthews, G.; Goulet, C.T.; Delhey, K.; Atkins, Z.S.; While, G.M.; Gardner, M.G.; Chapple, D.G. Avian Predation Intensity as a Driver of Clinal Variation in Colour Morph Frequency. J. Anim. Ecol. 2018, 87, 1667–1684. [Google Scholar] [CrossRef] [PubMed]
- Bittner, T.D.; King, R.B.; Kerfin, J.M. Effects of Body Size and Melanism on the Thermal Biology of Garter Snakes (Thamnophis sirtalis). Copeia 2002, 2002, 477–482. [Google Scholar] [CrossRef]
- Clusella Trullas, S.; van Wyk, J.H.; Spotila, J.R. Thermal Melanism in Ectotherms. J. Therm. Biol. 2007, 32, 235–245. [Google Scholar] [CrossRef]
- Hantak, M.M.; Guralnick, R.P.; Cameron, A.C.; Griffing, A.H.; Harrington, S.M.; Weinell, J.L.; Paluh, D.J. Colour Scales with Climate in North American Ratsnakes: A Test of the Thermal Melanism Hypothesis Using Community Science Images. Biol. Lett. 2022, 18, 20220403. [Google Scholar] [CrossRef] [PubMed]
- Madsen, T.; Stille, B.; Ujvari, B.; Bauwens, D.; Endler, J.A. Negative Frequency-Dependent Selection on Polymorphic Color Morphs in Adders. Curr. Biol. 2022, 32, 3385–3388.e3. [Google Scholar] [CrossRef]
- Osborne, J.W.; Costello, A.B. Sample Size and Subject to Item Ratio in Principal Components Analysis. Pract. Assess. Res. Eval. 2004, 9, 11. [Google Scholar]
- Huey, R.B. Temperature, Physiology, and the Ecology of Reptiles. Physiol. Ecol. 1982, 25–95. [Google Scholar]



| Species and Subspecies | Notes |
|---|---|
| Emys orbicularis (Linnaeus 1758) Emys orbicularis galloitalica Fritz, 1995 | |
| Trachemys scripta (Thunberg in Schoepff, 1792) Trachemys scripta elegans (Wied 1838) Trachemys scripta scripta (Thunberg in Schoepff 1792) | Recently introduced allochthonous species. |
| Testudo hermanni Gmelin 1789 Testudo hermanni hermanni Gmelin 1789 | |
| Testudo graeca Linnaeus 1758 Testudo graeca nabeulensis (Highfield 1990) | |
| Testudo marginata Schoepff, 1792 Testudo marginata marginata Schoepff, 1792 | |
| Caretta caretta (Linnaeus 1758) Caretta caretta caretta (Linnaeus 1758) | |
| Euleptes europaea (Gené, 1839) Euleptes europaea europaea (Gené 1839) | Species endemic to the Central-Western-Mediterranean. |
| Hemidactylus turcicus (Linnaeus, 1758) Hemidactylus turcicus turcicus (Linnaeus 1758) | |
| Tarentola mauritanica (Linnaeus, 1758) Tarentola mauritanica mauritanica (Linnaeus 1758) | |
| Algyroides fitzingeri (Wiegmann, 1834) Algyroides fitzingeri fitzingeri (Wiegmann 1834) | Species endemic to Sardinia and Corsica. |
| Archaeolacerta bedriagae (Camerano, 1885) Archaeolacerta bedriagae bedriagae (Camerano 1885) | Species endemic to Sardinia and Corsica. |
| Podarcis siculus (Rafinesque, 1810) Podarcis siculus siculus (Rafinesque-Schmaltz 1810) | |
| Podarcis tiliguerta (Gmelin, 1789) Podarcis tiliguerta tiliguerta (Gmelin, 1789) | Species endemic to Sardinia and Corsica. The validity of the subspecies P. t. ranzii and P. t. toro is not currently confirmed. |
| Chalcides chalcides (Linnaeus, 1758) Chalcides chalcides vittatus (Leuckart, 1828) | |
| Chalcides ocellatus (Forskål, 1775) Chalcides ocellatus tiligugu (Gmelin 1789) | |
| Hemorrhois hippocrepis (Linnaeus, 1758) Hemorrhois hippocrepis hippocrepis (Linnaeus 1758) | |
| Hierophis viridiflavus (Lacépède, 1789) Hierophis viridiflavus viridiflavus (Lacépède, 1789) | |
| Natrix helvetica (Lacépède 1789) Natrix helvetica cetti Gené, 1839 | Subspecies provisionally considered endemic to Sardinia (see Section 3.1.1). |
| Natrix maura (Linnaeus, 1758) Natrix maura (Linnaeus 1758) |
| Model | Variables | #Training Samples | #Test Samples | Test AUC | OR10 Threshold | OR10 Omission |
|---|---|---|---|---|---|---|
| 1 | Climate | 28 | 12 | 0.888 ± 0.042 | 0.165 | 0.071 |
| 2 | Climate Altitude | 28 | 12 | 0.912 ± 0.036 | 0.164 | 0.068 |
| 3 | Climate Altitude Land cover | 28 | 12 | 0.892 ± 0.041 | 0.141 | 0.071 |
| 4 | Climate, reduced | 28 | 12 | 0.839 ± 0.051 | 0.101 | 0.071 |
| 5 | Climate, reduced Altitude | 28 | 12 | 0.889 ± 0.042 | 0.130 | 0.064 |
| 6 | Climate, reduced Altitude Land cover | 28 | 12 | 0.879 ± 0.050 | 0.127 | 0.068 |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Di Nicola, M.R.; Pozzi, A.V.; Mezzadri, S.; Faraone, F.P.; Russo, G.; Dorne, J.L.M.C.; Minuti, G. The Endangered Sardinian Grass Snake: Distribution Update, Bioclimatic Niche Modelling, Dorsal Pattern Characterisation, and Literature Review. Life 2023, 13, 1867. https://doi.org/10.3390/life13091867
Di Nicola MR, Pozzi AV, Mezzadri S, Faraone FP, Russo G, Dorne JLMC, Minuti G. The Endangered Sardinian Grass Snake: Distribution Update, Bioclimatic Niche Modelling, Dorsal Pattern Characterisation, and Literature Review. Life. 2023; 13(9):1867. https://doi.org/10.3390/life13091867
Chicago/Turabian StyleDi Nicola, Matteo Riccardo, Andrea Vittorio Pozzi, Sergio Mezzadri, Francesco Paolo Faraone, Giorgio Russo, Jean Lou M. C. Dorne, and Gianmarco Minuti. 2023. "The Endangered Sardinian Grass Snake: Distribution Update, Bioclimatic Niche Modelling, Dorsal Pattern Characterisation, and Literature Review" Life 13, no. 9: 1867. https://doi.org/10.3390/life13091867
APA StyleDi Nicola, M. R., Pozzi, A. V., Mezzadri, S., Faraone, F. P., Russo, G., Dorne, J. L. M. C., & Minuti, G. (2023). The Endangered Sardinian Grass Snake: Distribution Update, Bioclimatic Niche Modelling, Dorsal Pattern Characterisation, and Literature Review. Life, 13(9), 1867. https://doi.org/10.3390/life13091867






