Cervical Impairments in Subjects with Chronic Migraine: An Observational Study
Abstract
:1. Introduction
2. Methods
2.1. Study Design
2.2. Participants
2.3. Procedure
2.4. Outcomes
2.5. Characteristics of Migraine Episodes
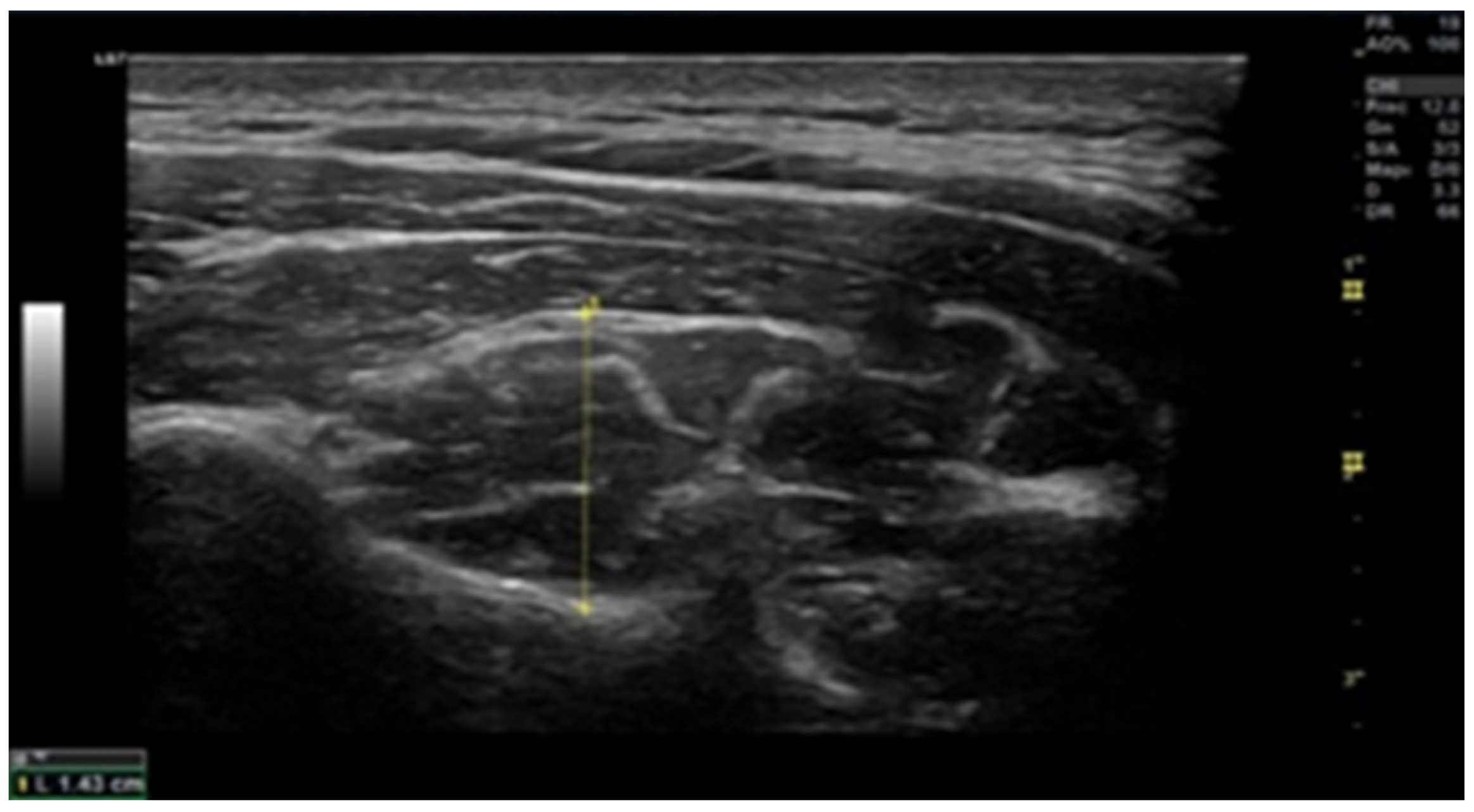
2.6. Muscle Thickness
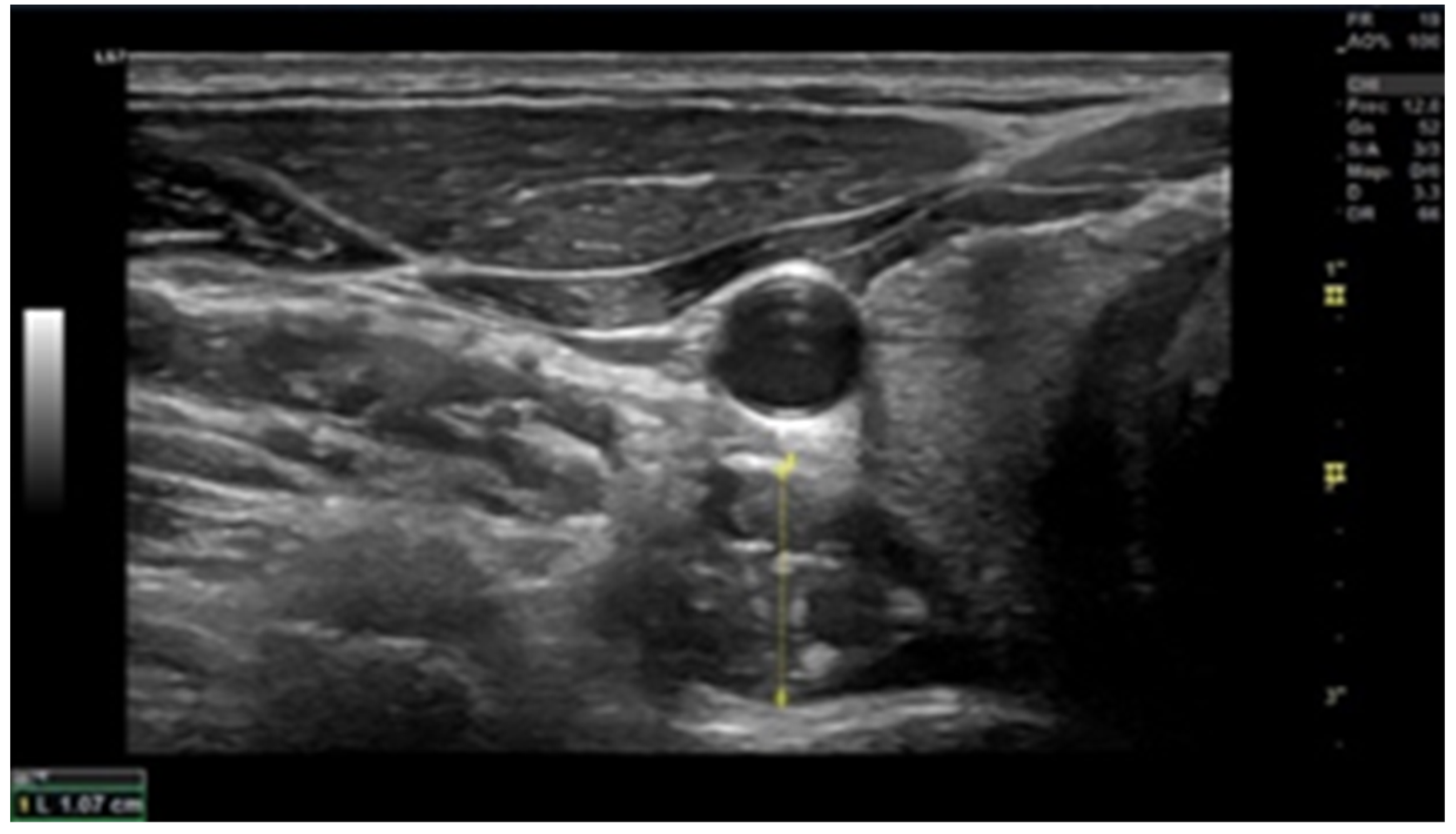
2.7. Pressure Pain Threshold (PPT)
- -
- Trapezius: Midway between C7 and the acromion.
- -
- Masseter: 1 cm above the insertion point of the superficial portion at the angle of the jaw.
- -
- Temporalis: The middle fibers in the bone depression approximately 2 cm lateral to the outer edge of the eyebrow.
- -
- Anterior tibialis: upper one third of the muscle belly.
- -
- Median nerve: in the flexion crease of the elbow, located from the biceps brachii tendon at the elbow.
2.8. Craniocervical Flexion Test (CCFT)
2.9. Data Collection Methods
2.10. Statistical Analysis
3. Results
3.1. Characteristics of the Enrolled Participants
3.2. Headache Characteristics
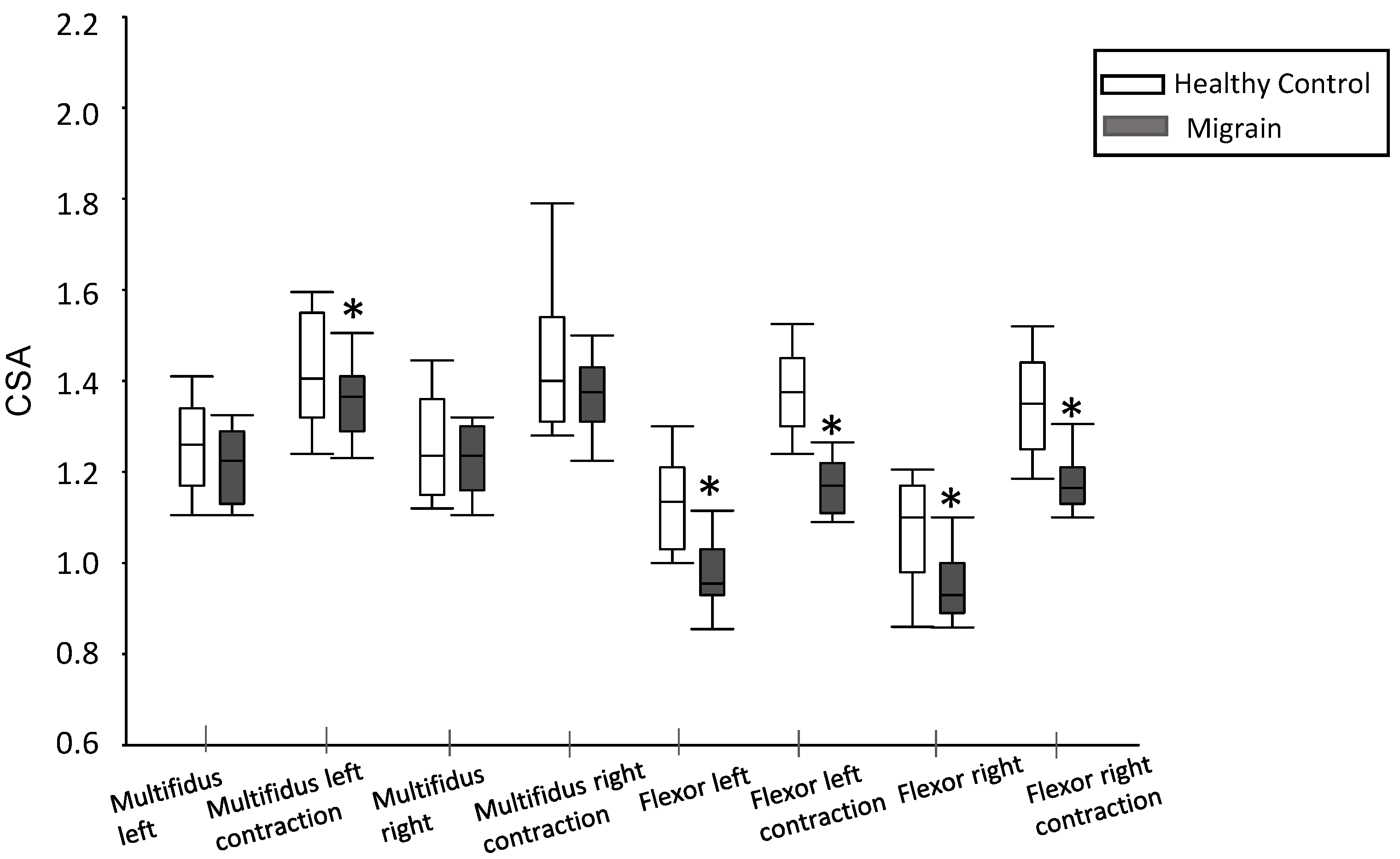
3.3. Muscle Thickness
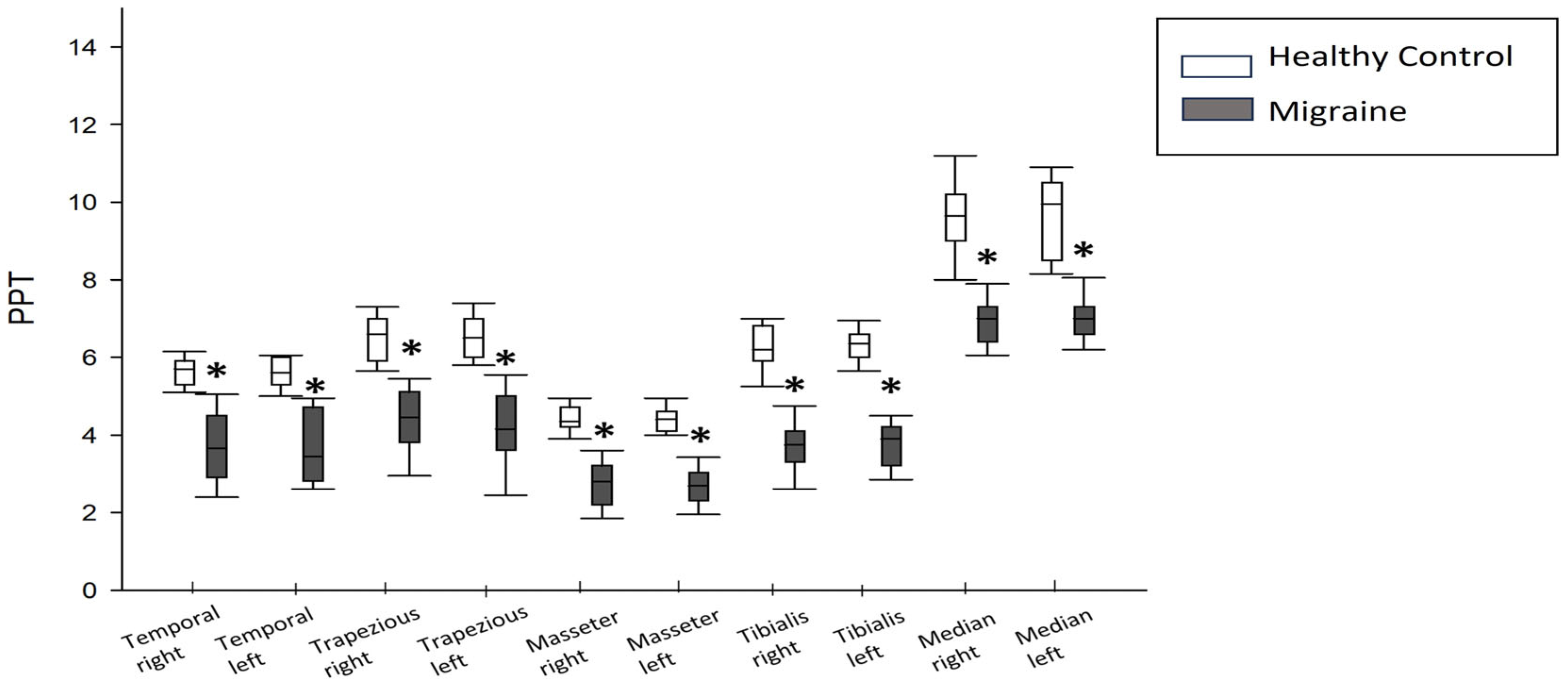
3.4. Pain Pressure Threshold
3.5. Craniocervical Flexion
3.6. Correlations
4. Discussion
5. Conclusions
6. Clinical Implications
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Almazán-Polo, J.; López-López, D.; Romero-Morales, C.; Rodríguez-Sanz, D.; Becerro-De-Bengoa-Vallejo, R.; Losa-Iglesias, M.E.; Bravo-Aguilar, M.; Calvo-Lobo, C. Quantitative Ultrasound Imaging Differences in Multifidus and Thoracolumbar Fasciae between Athletes with and without Chronic Lumbopelvic Pain: A Case-Control Study. J. Clin. Med. 2020, 9, 2647. [Google Scholar] [CrossRef] [PubMed]
- Anarte-Lazo, E.; Carvalho, G.F.; Schwarz, A.; Luedtke, K.; Falla, D. Differentiating migraine, cervicogenic headache and asymptomatic individuals based on physical examination findings: A systematic review and meta-analysis. BMC Musculoskelet. Disord. 2021, 22, 1–18. [Google Scholar] [CrossRef]
- Ashina, S.; Bendtsen, L.; Lyngberg, A.C.; Lipton, R.B.; Hajiyeva, N.; Jensen, R. Prevalence of neck pain in migraine and tension-type headache: A population study. Cephalalgia Int. J. Headache 2014, 35, 211–219. [Google Scholar] [CrossRef] [PubMed]
- Berman, G.; Croop, R.; Kudrow, D.; Halverson, P.; Lovegren, M.; Thiry, A.C.; Conway, C.M.; Coric, V.; Lipton, R.B. Safety of Rimegepant, an Oral CGRP Receptor Antagonist, Plus CGRP Monoclonal Antibodies for Migraine. Headache 2020, 60, 1734–1742. [Google Scholar] [CrossRef]
- Burch, R.; Rizzoli, P.; Loder, E. The prevalence and impact of migraine and severe headache in the United States: Updated age, sex, and socioeconomic-specific estimates from government health surveys. Headache 2020, 61, 60–68. [Google Scholar] [CrossRef] [PubMed]
- Cameron, C.; Kelly, S.; Hsieh, S.-C.; Murphy, M.; Chen, L.; Kotb, A.; Peterson, J.; Coyle, D.; Skidmore, B.; Gomes, T.; et al. Triptans in the Acute Treatment of Migraine: A Systematic Review and Network Meta-Analysis. Headache 2015, 55 (Suppl. 4), 221–235. [Google Scholar] [CrossRef]
- Charles, A. The pathophysiology of migraine: Implications for clinical management. Lancet Neurol. 2018, 17, 174–182. [Google Scholar] [CrossRef]
- Demarquay, G.; Mawet, J.; Guégan-Massardier, E.; de Gaalon, S.; Donnet, A.; Giraud, P.; Lantéri-Minet, M.; Lucas, C.; Moisset, X.; Roos, C.; et al. Revised guidelines of the French headache society for the diagnosis and management of migraine in adults. Part 3: Non-pharmacological treatment. Rev. Neurol. 2021, 177, 753–759. [Google Scholar] [CrossRef]
- Fleckenstein, J. Diagnostic requirements are necessary before dry needling in the treatment of chronic pain. Pain 2015, 156, 1827. [Google Scholar] [CrossRef]
- Florencio, L.L.; De Oliveira, A.S.; Carvalho, G.F.; de Almeida Tolentino, G.; Dach, F.; Bigal, M.E.; Fernández-De-Las-Peñas, C.; Bevilaqua-Grossi, D. Cervical Muscle Strength and Muscle Coactivation During Isometric Contractions in Patients with Migraine: A Cross-Sectional Study. Headache 2015, 55, 1312–1322. [Google Scholar] [CrossRef]
- Gago-Veiga, A.B.; Camiña Muñiz, J.; García-Azorín, D.; González-Quintanilla, V.; Ordás, C.M.; Torres-Ferrus, M.; Santos-Lasaosa, S.; Viguera-Romero, J.; Pozo-Rosich, P. Headache: What to ask, how to examine, and which scales to use. Recommendations of the Spanish Society of Neurology’s Headache Study Group. Neurologia 2019, 37, 564–574. [Google Scholar] [CrossRef] [PubMed]
- Grossi, D.B.; Chaves, T.C.; Gonçalves, M.C.; Moreira, V.C.; Canonica, A.C.; Florencio, L.L.; Bordini, C.A.; Speciali, J.G.; Bigal, M.E. Pressure pain threshold in the craniocervical muscles of women with episodic and chronic migraine: A controlled study. Arq. Neuro-Psiquiatr. 2011, 69, 607–612. [Google Scholar] [CrossRef] [PubMed]
- Hides, J.; Gilmore, C.; Stanton, W.; Bohlscheid, E. Multifidus size and symmetry among chronic LBP and healthy asymptomatic subjects. Man. Ther. 2008, 13, 43–49. [Google Scholar] [CrossRef] [PubMed]
- Headache Classification Committee of the International Headache Society (IHS). The International Classification of Headache Disorders, 3rd ed.; Cephalalgia Int. J. Headache 2018, 38, 1–211. [CrossRef]
- Leonardi, M.; Raggi, A. Burden of migraine: International perspectives. Neurol. Sci. Off. J. Ital. Neurol. Soc. Ital. Soc. Clin. Neurophysiol. 2013, 34 (Suppl 1), 117–118. [Google Scholar] [CrossRef]
- Liang, Z.; Galea, O.; Thomas, L.; Jull, G.; Treleaven, J. Cervical musculoskeletal impairments in migraine and tension type headache: A systematic review and meta-analysis. Musculoskelet. Sci. Pract. 2019, 42, 67–83. [Google Scholar] [CrossRef]
- Luedtke, K.; Allers, A.; Schulte, L.H.; May, A. Efficacy of interventions used by physiotherapists for patients with headache and migraine—Systematic review and meta-analysis. Cephalalgia 2015, 36, 474–492. [Google Scholar] [CrossRef]
- Luedtke, K.; May, A. Stratifying migraine patients based on dynamic pain provocation over the upper cervical spine. J. Headache Pain 2017, 18, 97. [Google Scholar] [CrossRef]
- Matias-Guiu, J.; Fernandez, C.; Porta-Etessam, J.; Mateos, V.; Diaz-Insa, S. Factors Associated with the Differences in Migraine Prevalence Rates between Spanish Regions. Sci. World J. 2014, 2014, 323084. [Google Scholar] [CrossRef]
- Moore, C.S.; Sibbritt, D.W.; Adams, J. A critical review of manual therapy use for headache disorders: Prevalence, profiles, motivations, communication and self-reported effectiveness. BMC Neurol. 2017, 17, 61. [Google Scholar] [CrossRef]
- Muñoz-Gómez, E.; Inglés, M.; Serra-Añó, P.; Espí-López, G.V. Effectiveness of a manual therapy protocol based on articulatory techniques in migraine patients. A randomized controlled trial. Musculoskelet. Sci. Pract. 2021, 54, 102386. [Google Scholar] [CrossRef]
- Øverås, C.K.; Myhrvold, B.L.; Røsok, G.; Magnesen, E. Musculoskeletal diagnostic ultrasound imaging for thickness measurement of four principal muscles of the cervical spine -a reliability and agreement study. Chiropr. Man. Ther. 2017, 25, 2. [Google Scholar] [CrossRef] [PubMed]
- Pilar Navarro-Pérez, M.; Marín-Gracia, M.; Bellosta-Diago, E.; Santos-Lasaosa, S. Epidemiology of migraine in Spain and Latin America. Rev. Neurol. 2020, 71, 110–118. [Google Scholar] [CrossRef]
- Shephard, R.J. Qualified Fitness and Exercise as Professionals and Exercise Prescription: Evolution of the PAR-Q and Canadian Aerobic Fitness Test. J. Phys. Act. Health 2015, 12, 454–461. [Google Scholar] [CrossRef]
- Tabatabaiee, A.; Takamjani, I.E.; Sarrafzadeh, J.; Salehi, R.; Ahmadi, M. Pressure Pain Threshold in Subjects With Piriformis Syndrome: Test-Retest, Intrarater, and Interrater Reliability, and Minimal Detectible Changes. Arch. Phys. Med. Rehabilitation 2020, 101, 781–788. [Google Scholar] [CrossRef] [PubMed]
- Thongprasert, C.; Kanlayanaphotporn, R. Abnormal performance of cervical stabilizer muscles in individuals with low back pain. J. Man. Manip. Ther. 2019, 27, 215–221. [Google Scholar] [CrossRef]
- Solanki, V.D.; Soni, N. Interrater and intrarater reliability of pressure biofeedback unit in measurement of transverses abdominis muscle activation in asymptomatic adults. Int. J. Adv. Res. 2021, 9, 664–670. [Google Scholar] [CrossRef]
- Vázquez-Justes, D.; Yarzábal-Rodríguez, R.; Doménech-García, V.; Herrero, P.; Bellosta-López, P. Effectiveness of dry needling for headache: A systematic review. Neurologia 2020, 37, 806–815. [Google Scholar] [CrossRef]
- Von Elm, E.; Altman, D.G.; Egger, M.; Pocock, S.J.; Gøtzsche, P.C.; Vandenbroucke, J.P. The Strengthening the Reporting of Observational Studies in Epidemiology (STROBE) statement: Guidelines for reporting observational studies. J. Clin. Epidemiol. 2008, 61, 344–349. [Google Scholar] [CrossRef]
- Vos, T.; Abajobir, A.A.; Abbafati, C.; Abbas, K.M.; Abate, K.H.; Abd-Allah, F.; Abdulle, A.M.; Abebo, T.A.; Abera, S.F.; Aboyans, V.; et al. Global, regional, and national incidence, prevalence, and years lived with disability for 328 diseases and injuries for 195 countries, 1990–2016: A systematic analysis for the Global Burden of Disease Study 2016. Lancet 2017, 390, 1211–1259. [Google Scholar] [CrossRef]
- Williams, A.E.; Miller, M.M.; Bartley, E.J.; McCabe, K.M.; Kerr, K.L.; Rhudy, J.L. Impairment of Inhibition of Trigeminal Nociception via Conditioned Pain Modulation in Persons with Migraine Headaches. Pain Med. 2019, 20, 1600–1610. [Google Scholar] [CrossRef] [PubMed]
- Woldeamanuel, Y.W.; Oliveira, A.B.D. What is the efficacy of aerobic exercise versus strength training in the treatment of migraine? A systematic review and network meta-analysis of clinical trials. J. Headache Pain 2022, 23, 134. [Google Scholar] [CrossRef] [PubMed]
| A | Headache (tension-type or migraine-like) occurring for a period of ≥15 days/month for >3 months, fulfilling criteria B and C. |
| B | It occurs in patients who have experienced at least five attacks fulfilling criteria B-D for 1.1, migraine without aura and/or criteria B and C for 1.2, migraine with aura |
| C | During a period of ≥8 days/month for >3 months, fulfilling any of the following:
|
| D | Not attributable to another diagnosis in the ICHD-III. |
| Healthy Controls (n = 30) | Migraine Patients (n = 30) | p Value | |||
|---|---|---|---|---|---|
| Sex n (%) | Men | 13 (43.3%) | Men | 10 (33.3%) | 0.426 |
| Women | 17 (56.7%) | Women | 20 (66.7%) | ||
| Age (years) | 29 [46–23] | 28 [43–23] | 0.90 | ||
| Weight (Kg) | 68 [74–57] | 63 [71–61] | 0.66 | ||
| Height (m) | 1.67 [1.75–1.60] | 1.65 [1.74–1.61] | 0.71 | ||
| Mean (SD) | |
|---|---|
| Frequency (days in four weeks) | 11.00 (8.78) |
| Intensity (VAS 0–10) | 7.22 (1.33) |
| Duration (h/day) | 10.91 (8.23) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
del-Blanco-Muñiz, J.A.; Martín-Vera, D.; Sosa-Reina, M.D.; Trinidad-Morales, A.; de-la-Plaza-San-Frutos, M.; Sánchez-Sierra, A. Cervical Impairments in Subjects with Chronic Migraine: An Observational Study. Life 2023, 13, 1773. https://doi.org/10.3390/life13081773
del-Blanco-Muñiz JA, Martín-Vera D, Sosa-Reina MD, Trinidad-Morales A, de-la-Plaza-San-Frutos M, Sánchez-Sierra A. Cervical Impairments in Subjects with Chronic Migraine: An Observational Study. Life. 2023; 13(8):1773. https://doi.org/10.3390/life13081773
Chicago/Turabian Styledel-Blanco-Muñiz, José Angel, Daniel Martín-Vera, Maria Dolores Sosa-Reina, Alfonso Trinidad-Morales, Marta de-la-Plaza-San-Frutos, and Alberto Sánchez-Sierra. 2023. "Cervical Impairments in Subjects with Chronic Migraine: An Observational Study" Life 13, no. 8: 1773. https://doi.org/10.3390/life13081773
APA Styledel-Blanco-Muñiz, J. A., Martín-Vera, D., Sosa-Reina, M. D., Trinidad-Morales, A., de-la-Plaza-San-Frutos, M., & Sánchez-Sierra, A. (2023). Cervical Impairments in Subjects with Chronic Migraine: An Observational Study. Life, 13(8), 1773. https://doi.org/10.3390/life13081773









