Subcutaneous Implantable Cardioverter Defibrillator: A Contemporary Overview
Abstract
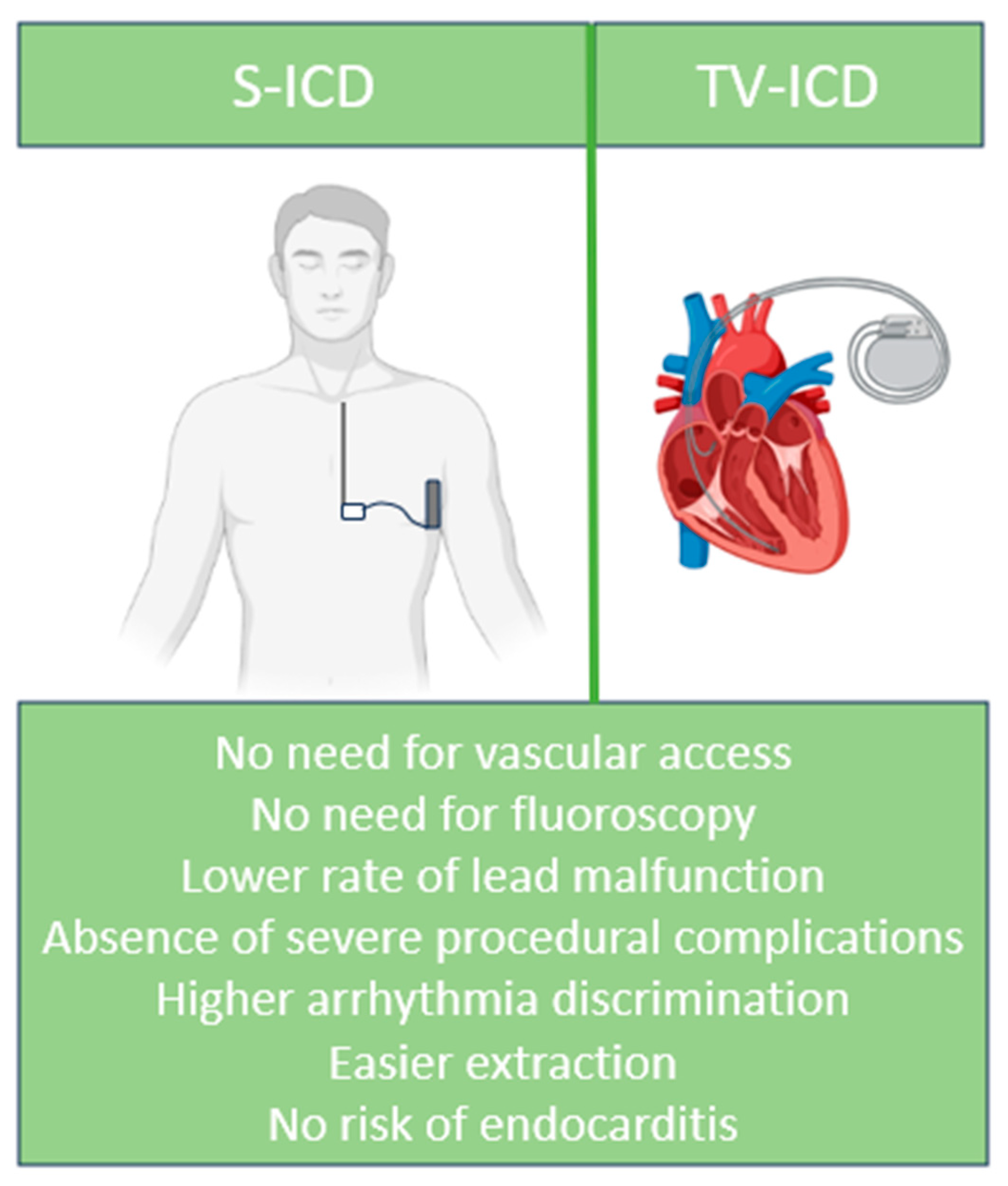
1. Introduction
2. Subcutaneous ICD: What We Know So Far
2.1. Pre-Implant Screening
2.2. Implant Technique
2.3. Inappropriate Shocks
2.4. Infections
2.5. Lead Complications
2.6. Appropriate Therapies
3. Indications for S-ICD Implant
4. Particular Set of Patients according to the Underlying Cardiomyopathy
4.1. Hypertrophic Cardiomyopathy
4.2. Brugada Syndrome, Long QT Syndrome and Arrhythmogenic Right Ventricular Cardiomyopathy
4.3. Congenital Heart Disease
5. Future Perspectives
6. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Alter, P.; Waldhans, S.; Plachta, E.; Moosdorf, R.; Grimm, W. Complications of implantable cardioverter defibrillator therapy in 440 consecutive patients. Pacing Clin. Electrophysiol. 2005, 28, 926–932. [Google Scholar] [CrossRef] [PubMed]
- Bardy, G.H.; Smith, W.M.; Hood, M.A.; Crozier, I.G.; Melton, I.C.; Jordaens, L.; Theuns, D.; Park, R.E.; Wright, D.J.; Connelly, D.T.; et al. An Entirely Subcutaneous Implantable Cardioverter–Defibrillator. N. Engl. J. Med. 2010, 363, 36–44. [Google Scholar] [CrossRef] [PubMed]
- Dabiri Abkenari, L.; Theuns, D.A.; Valk, S.D.; Van Belle, Y.; de Groot, N.M.; Haitsma, D.; Muskens-Heemskerk, A.; Szili-Torok, T.; Jordaens, L. Clinical experience with a novel subcutaneous implantable defibrillator system in a single center. Clin. Res. Cardiol. 2011, 100, 737–744. [Google Scholar] [CrossRef] [PubMed]
- De Filippo, P.; Migliore, F.; Palmisano, P.; Nigro, G.; Ziacchi, M.; Rordorf, R.; Pieragnoli, P.; Di Grazia, A.; Ottaviano, L.; Francia, P.; et al. Procedure, management, and outcome of subcutaneous implantable cardioverter–defibrillator extraction in clinical practice. EP Eur. 2023, 25, euad158. [Google Scholar] [CrossRef]
- Kuschyk, J.; Müller-Leisse, J.; Duncker, D.; Tülümen, E.; Fastenrath, F.; Fastner, C.; Kruska, M.; Akin, I.; Liebe, V.; Borggrefe, M.; et al. Comparison of transvenous vs subcutaneous defibrillator therapy in patients with cardiac arrhythmia syndromes and genetic cardiomyopathies. Int. J. Cardiol. 2021, 323, 100–105. [Google Scholar] [CrossRef]
- Migliore, F.; Pelliccia, F.; Autore, C.; Bertaglia, E.; Cecchi, F.; Curcio, A.; Bontempi, L.; Curnis, A.; De Filippo, P.; D’Onofrio, A.; et al. Subcutaneous implantable cardioverter defibrillator in cardiomyopathies and channelopathies. J. Cardiovasc. Med. 2018, 19, 633–642. [Google Scholar] [CrossRef]
- Weinstock, J.; Bader, Y.H.; Maron, M.S.; Rowin, E.J.; Link, M.S. Subcutaneous Implantable Cardioverter Defibrillator in Patients With Hypertrophic Cardiomyopathy: An Initial Experience. J. Am. Heart Assoc. 2016, 5, e002488. [Google Scholar] [CrossRef]
- Willy, K.; Doldi, F.; Reinke, F.; Rath, B.; Wolfes, J.; Wegner, F.K.; Leitz, P.; Ellermann, C.; Lange, P.S.; Köbe, J.; et al. Bradycardia in Patients with Subcutaneous Implantable Defibrillators—An Overestimated Problem? Experience from a Large Tertiary Centre and a Review of the Literature. RCM 2022, 23, 352. [Google Scholar] [CrossRef]
- Burke, M.C.; Gold, M.R.; Knight, B.P.; Barr, C.S.; Theuns, D.; Boersma, L.V.A.; Knops, R.E.; Weiss, R.; Leon, A.R.; Herre, J.M.; et al. Safety and Efficacy of the Totally Subcutaneous Implantable Defibrillator: 2-Year Results From a Pooled Analysis of the IDE Study and EFFORTLESS Registry. J. Am. Coll. Cardiol. 2015, 65, 1605–1615. [Google Scholar] [CrossRef]
- Gold, M.R.; Lambiase, P.D.; El-Chami, M.F.; Knops, R.E.; Aasbo, J.D.; Bongiorni, M.G.; Russo, A.M.; Deharo, J.-C.; Burke, M.C.; Dinerman, J.; et al. Primary Results From the Understanding Outcomes With the S-ICD in Primary Prevention Patients With Low Ejection Fraction (UNTOUCHED) Trial. Circulation 2021, 143, 7–17. [Google Scholar] [CrossRef]
- Knops, R.E.; Olde Nordkamp, L.R.A.; Delnoy, P.-P.H.M.; Boersma, L.V.A.; Kuschyk, J.; El-Chami, M.F.; Bonnemeier, H.; Behr, E.R.; Brouwer, T.F.; Kääb, S.; et al. Subcutaneous or Transvenous Defibrillator Therapy. N. Engl. J. Med. 2020, 383, 526–536. [Google Scholar] [CrossRef]
- Rordorf, R. The ATLAS Randomised Clinical Trial: What do the Superiority Results Mean for Subcutaneous ICD Therapy and Sudden Cardiac Death Prevention as a Whole? Arrhythm Electrophysiol. Rev. 2022, 11. [Google Scholar] [CrossRef]
- Boersma, L.; Barr, C.; Knops, R.; Theuns, D.; Eckardt, L.; Neuzil, P.; Scholten, M.; Hood, M.; Kuschyk, J.; Jones, P.; et al. Implant and Midterm Outcomes of the Subcutaneous Implantable Cardioverter-Defibrillator Registry: The EFFORTLESS Study. J. Am. Coll. Cardiol. 2017, 70, 830–841. [Google Scholar] [CrossRef]
- Burke, M.C.; Aasbo, J.D.; El-Chami, M.F.; Weiss, R.; Dinerman, J.; Hanon, S.; Kalahasty, G.; Bass, E.; Gold, M.R. 1-Year Prospective Evaluation of Clinical Outcomes and Shocks: The Subcutaneous ICD Post Approval Study. JACC Clin. Electrophysiol. 2020, 6, 1537–1550. [Google Scholar] [CrossRef]
- Weiss, R.; Knight, B.P.; Gold, M.R.; Leon, A.R.; Herre, J.M.; Hood, M.; Rashtian, M.; Kremers, M.; Crozier, I.; Lee, K.L.; et al. Safety and efficacy of a totally subcutaneous implantable-cardioverter defibrillator. Circulation 2013, 128, 944–953. [Google Scholar] [CrossRef]
- Gold, M.R.; Theuns, D.A.; Knight, B.P.; Sturdivant, J.L.; Sanghera, R.; Ellenbogen, K.A.; Wood, M.A.; Burke, M.C. Head-to-head comparison of arrhythmia discrimination performance of subcutaneous and transvenous ICD arrhythmia detection algorithms: The START study. J. Cardiovasc. Electrophysiol. 2012, 23, 359–366. [Google Scholar] [CrossRef]
- Chang, S.C.; Patton, K.K.; Robinson, M.R.; Poole, J.E.; Prutkin, J.M. Subcutaneous ICD screening with the Boston Scientific ZOOM programmer versus a 12-lead ECG machine. Pacing Clin. Electrophysiol. 2018, 41, 511–516. [Google Scholar] [CrossRef]
- Groh, C.A.; Sharma, S.; Pelchovitz, D.J.; Bhave, P.D.; Rhyner, J.; Verma, N.; Arora, R.; Chicos, A.B.; Kim, S.S.; Lin, A.C.; et al. Use of an electrocardiographic screening tool to determine candidacy for a subcutaneous implantable cardioverter-defibrillator. Heart Rhythm 2014, 11, 1361–1366. [Google Scholar] [CrossRef]
- Olde Nordkamp, L.R.A.; Warnaars, J.L.F.; Kooiman, K.M.; de Groot, J.R.; Rosenmöller, B.; Wilde, A.A.M.; Knops, R.E. Which patients are not suitable for a subcutaneous ICD: Incidence and predictors of failed QRS-T-wave morphology screening. J. Cardiovasc. Electrophysiol. 2014, 25, 494–499. [Google Scholar] [CrossRef]
- Randles, D.A.; Hawkins, N.M.; Shaw, M.; Patwala, A.Y.; Pettit, S.J.; Wright, D.J. How many patients fulfil the surface electrocardiogram criteria for subcutaneous implantable cardioverter-defibrillator implantation? Europace 2014, 16, 1015–1021. [Google Scholar] [CrossRef]
- Ziacchi, M.; Corzani, A.; Diemberger, I.; Martignani, C.; Marziali, A.; Mazzotti, A.; Massaro, G.; Rapezzi, C.; Biffi, M.; Boriani, G. Electrocardiographic Eligibility for Subcutaneous Implantable Cardioverter Defibrillator: Evaluation during Bicycle Exercise. Heart Lung. Circ. 2016, 25, 476–483. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Afzal, M.R.; Evenson, C.; Badin, A.; Patel, D.; Godara, H.; Essandoh, M.; Okabe, T.; Tyler, J.; Houmsse, M.; Augostini, R.; et al. Role of exercise electrocardiogram to screen for T-wave oversensing after implantation of subcutaneous implantable cardioverter-defibrillator. Heart Rhythm 2017, 14, 1436–1439. [Google Scholar] [CrossRef] [PubMed]
- Dunn, A.J.; ElRefai, M.H.; Roberts, P.R.; Coniglio, S.; Wiles, B.M.; Zemkoho, A.B. Deep learning methods for screening patients’ S-ICD implantation eligibility. Artif. Intell. Med. 2021, 119, 102139. [Google Scholar] [CrossRef] [PubMed]
- Wiles, B.M.; Morgan, J.M.; Allavatam, V.; ElRefai, M.; Roberts, P.R. S-ICD screening revisited: Do passing vectors sometimes fail? Pacing Clin. Electrophysiol. 2022, 45, 182–187. [Google Scholar] [CrossRef] [PubMed]
- Srinivasan, N.T.; Patel, K.H.; Qamar, K.; Taylor, A.; Bacà, M.; Providência, R.; Tome-Esteban, M.; Elliott, P.M.; Lambiase, P.D. Disease Severity and Exercise Testing Reduce Subcutaneous Implantable Cardioverter-Defibrillator Left Sternal ECG Screening Success in Hypertrophic Cardiomyopathy. Circ. Arrhythmia Electrophysiol. 2017, 10, e004801. [Google Scholar] [CrossRef]
- El-Chami, M.; Weiss, R.; Burke, M.C.; Gold, M.R.; Prutkin, J.M.; Kalahasty, G.; Shen, S.; Mirro, M.J.; Carter, N.; Aasbo, J.D. Outcomes of two versus three incision techniques: Results from the subcutaneous ICD post-approval study. J. Cardiovasc. Electrophysiol. 2021, 32, 792–801. [Google Scholar] [CrossRef]
- Knops, R.E.; Olde Nordkamp, L.R.; de Groot, J.R.; Wilde, A.A. Two-incision technique for implantation of the subcutaneous implantable cardioverter-defibrillator. Heart Rhythm 2013, 10, 1240–1243. [Google Scholar] [CrossRef]
- Francia, P.; Biffi, M.; Adduci, C.; Ottaviano, L.; Migliore, F.; De Bonis, S.; Dello Russo, A.; De Filippo, P.; Viani, S.; Bongiorni, M.G.; et al. Implantation technique and optimal subcutaneous defibrillator chest position: A Praetorian score-based study. Europace 2020, 22, 1822–1829. [Google Scholar] [CrossRef]
- Migliore, F.; Mattesi, G.; De Franceschi, P.; Allocca, G.; Crosato, M.; Calzolari, V.; Fantinel, M.; Ortis, B.; Facchin, D.; Daleffe, E.; et al. Multicentre experience with the second-generation subcutaneous implantable cardioverter defibrillator and the intermuscular two-incision implantation technique. J. Cardiovasc. Electrophysiol. 2019, 30, 854–864. [Google Scholar] [CrossRef]
- Ferrari, P.; Giofrè, F.; De Filippo, P. Intermuscular pocket for subcutaneous implantable cardioverter defibrillator: Single-center experience. J. Arrhythm 2016, 32, 223–226. [Google Scholar] [CrossRef]
- Smietana, J.; Frankel, D.S.; Serletti, J.M.; Arkles, J.; Pothineni, N.V.K.; Marchlinski, F.E.; Schaller, R.D. Subserratus implantation of the subcutaneous implantable cardioverter-defibrillator. Heart Rhythm 2021, 18, 1799–1804. [Google Scholar] [CrossRef]
- Heist, E.K.; Belalcazar, A.; Stahl, W.; Brouwer, T.F.; Knops, R.E. Determinants of Subcutaneous Implantable Cardioverter-Defibrillator Efficacy: A Computer Modeling Study. JACC Clin. Electrophysiol. 2017, 3, 405–414. [Google Scholar] [CrossRef]
- Knops, R.E.; Brouwer, T.F.; Barr, C.S.; Theuns, D.A.; Boersma, L.; Weiss, R.; Neuzil, P.; Scholten, M.; Lambiase, P.D.; Leon, A.R.; et al. The learning curve associated with the introduction of the subcutaneous implantable defibrillator. Europace 2016, 18, 1010–1015. [Google Scholar] [CrossRef]
- Brewster, J.; Sexton, T.; Dhaliwal, G.; Charnigo, R.; Morales, G.; Parrott, K.; Darrat, Y.; Gurley, J.; Smyth, S.; Elayi, C.S. Acute Effects of Implantable Cardioverter-Defibrillator Shocks on Biomarkers of Myocardial Injury, Apoptosis, Heart Failure, and Systemic Inflammation. Pacing Clin. Electrophysiol. 2017, 40, 344–352. [Google Scholar] [CrossRef]
- MacIntyre, C.J.; Sapp, J.L.; Abdelwahab, A.; Al-Harbi, M.; Doucette, S.; Gray, C.; Gardner, M.J.; Parkash, R. The Effect of Shock Burden on Heart Failure and Mortality. CJC Open 2019, 1, 161–167. [Google Scholar] [CrossRef]
- Moss, A.J.; Hall, W.J.; Cannom, D.S.; Klein, H.; Brown, M.W.; Daubert, J.P.; Estes, N.A.M.; Foster, E.; Greenberg, H.; Higgins, S.L.; et al. Cardiac-Resynchronization Therapy for the Prevention of Heart-Failure Events. N. Engl. J. Med. 2009, 361, 1329–1338. [Google Scholar] [CrossRef]
- Li, A.; Kaura, A.; Sunderland, N.; Dhillon, P.S.; Scott, P.A. The Significance of Shocks in Implantable Cardioverter Defibrillator Recipients. Arrhythm Electrophysiol. Rev. 2016, 5, 110–116. [Google Scholar] [CrossRef]
- Sears, S.F.; Hauf, J.D.; Kirian, K.; Hazelton, G.; Conti, J.B. Posttraumatic Stress and the Implantable Cardioverter-Defibrillator Patient. Circ. Arrhythmia Electrophysiol. 2011, 4, 242–250. [Google Scholar] [CrossRef] [PubMed]
- Fleeman, B.E.; Aleong, R.G. Optimal Strategies to Reduce Inappropriate Implantable Cardioverter-defibrillator Shocks. J. Innov. Card Rhythm Manag. 2019, 10, 3623–3632. [Google Scholar] [CrossRef]
- Auricchio, A.; Hudnall, J.H.; Schloss, E.J.; Sterns, L.D.; Kurita, T.; Meijer, A.; Fagan, D.H.; Rogers, T. Inappropriate shocks in single-chamber and subcutaneous implantable cardioverter-defibrillators: A systematic review and meta-analysis. Europace 2017, 19, 1973–1980. [Google Scholar] [CrossRef]
- Auricchio, A.; Schloss, E.J.; Kurita, T.; Meijer, A.; Gerritse, B.; Zweibel, S.; AlSmadi, F.M.; Leng, C.T.; Sterns, L.D. Low inappropriate shock rates in patients with single- and dual/triple-chamber implantable cardioverter-defibrillators using a novel suite of detection algorithms: PainFree SST trial primary results. Heart Rhythm 2015, 12, 926–936. [Google Scholar] [CrossRef] [PubMed]
- Olde Nordkamp, L.R.; Brouwer, T.F.; Barr, C.; Theuns, D.A.; Boersma, L.V.; Johansen, J.B.; Neuzil, P.; Wilde, A.A.; Carter, N.; Husby, M.; et al. Inappropriate shocks in the subcutaneous ICD: Incidence, predictors and management. Int. J. Cardiol. 2015, 195, 126–133. [Google Scholar] [CrossRef] [PubMed]
- Noel, A.; Ploux, S.; Bulliard, S.; Strik, M.; Haeberlin, A.; Welte, N.; Marchand, H.; Klotz, N.; Ritter, P.; Haïssaguerre, M.; et al. Oversensing issues leading to device extraction: When subcutaneous implantable cardioverter-defibrillator reached a dead-end. Heart Rhythm 2020, 17, 66–74. [Google Scholar] [CrossRef] [PubMed]
- Fong, K.Y.; Ng, C.J.R.; Wang, Y.; Yeo, C.; Tan, V.H. Subcutaneous Versus Transvenous Implantable Defibrillator Therapy: A Systematic Review and Meta-Analysis of Randomized Trials and Propensity Score–Matched Studies. J. Am. Heart Assoc. 2022, 11, e024756. [Google Scholar] [CrossRef] [PubMed]
- Conte, G.; Cattaneo, F.; de Asmundis, C.; Berne, P.; Vicentini, A.; Namdar, M.; Scalone, A.; Klersy, C.; Caputo, M.L.; Demarchi, A.; et al. Impact of SMART Pass filter in patients with ajmaline-induced Brugada syndrome and subcutaneous implantable cardioverter-defibrillator eligibility failure: Results from a prospective multicentre study. Europace 2022, 24, 845–854. [Google Scholar] [CrossRef]
- Theuns, D.; Brouwer, T.F.; Jones, P.W.; Allavatam, V.; Donnelley, S.; Auricchio, A.; Knops, R.E.; Burke, M.C. Prospective blinded evaluation of a novel sensing methodology designed to reduce inappropriate shocks by the subcutaneous implantable cardioverter-defibrillator. Heart Rhythm 2018, 15, 1515–1522. [Google Scholar] [CrossRef]
- Madhavan, M.; Friedman, P.A. Optimal Programming of Implantable Cardiac-Defibrillators. Circulation 2013, 128, 659–672. [Google Scholar] [CrossRef]
- Gold, M.R.; Aasbo, J.D.; Weiss, R.; Burke, M.C.; Gleva, M.J.; Knight, B.P.; Miller, M.A.; Schuger, C.D.; Carter, N.; Leigh, J.; et al. Infection in patients with subcutaneous implantable cardioverter-defibrillator: Results of the S-ICD Post Approval Study. Heart Rhythm 2022, 19, 1993–2001. [Google Scholar] [CrossRef]
- Rordorf, R.; Casula, M.; Pezza, L.; Fortuni, F.; Sanzo, A.; Savastano, S.; Vicentini, A. Subcutaneous versus transvenous implantable defibrillator: An updated meta-analysis. Heart Rhythm 2021, 18, 382–391. [Google Scholar] [CrossRef]
- Su, L.; Guo, J.; Hao, Y.; Tan, H. Comparing the safety of subcutaneous versus transvenous ICDs: A meta-analysis. J. Interv. Card Electrophysiol. 2021, 60, 355–363. [Google Scholar] [CrossRef]
- Knops, R.E.; Pepplinkhuizen, S.; Delnoy, P.; Boersma, L.V.A.; Kuschyk, J.; El-Chami, M.F.; Bonnemeier, H.; Behr, E.R.; Brouwer, T.F.; Kaab, S.; et al. Device-related complications in subcutaneous versus transvenous ICD: A secondary analysis of the PRAETORIAN trial. Eur. Heart J. 2022, 43, 4872–4883. [Google Scholar] [CrossRef]
- Bongiorni, M.G.; Kennergren, C.; Butter, C.; Deharo, J.C.; Kutarski, A.; Rinaldi, C.A.; Romano, S.L.; Maggioni, A.P.; Andarala, M.; Auricchio, A.; et al. The European Lead Extraction ConTRolled (ELECTRa) study: A European Heart Rhythm Association (EHRA) Registry of Transvenous Lead Extraction Outcomes. Eur. Heart J. 2017, 38, 2995–3005. [Google Scholar] [CrossRef]
- Behar, N.; Galand, V.; Martins, R.P.; Jacon, P.; Badenco, N.; Blangy, H.; Alonso, C.; Guy-Moyat, B.; El Bouazzaoui, R.; Lebon, A.; et al. Subcutaneous Implantable Cardioverter-Defibrillator Lead Extraction: First Multicenter French Experience. JACC Clin. Electrophysiol. 2020, 6, 863–870. [Google Scholar] [CrossRef]
- Kusumoto, F.M.; Schoenfeld, M.H.; Wilkoff, B.L.; Berul, C.I.; Birgersdotter-Green, U.M.; Carrillo, R.; Cha, Y.M.; Clancy, J.; Deharo, J.C.; Ellenbogen, K.A.; et al. 2017 HRS expert consensus statement on cardiovascular implantable electronic device lead management and extraction. Heart Rhythm 2017, 14, e503–e551. [Google Scholar] [CrossRef]
- Alt, E.; Völker, R.; Blömer, H. Lead fracture in pacemaker patients. Thorac. Cardiovasc. Surg. 1987, 35, 101–104. [Google Scholar] [CrossRef]
- Kleemann, T.; Becker, T.; Doenges, K.; Vater, M.; Senges, J.; Schneider, S.; Saggau, W.; Weisse, U.; Seidl, K. Annual rate of transvenous defibrillation lead defects in implantable cardioverter-defibrillators over a period of >10 years. Circulation 2007, 115, 2474–2480. [Google Scholar] [CrossRef]
- Occhetta, E.; Bortnik, M.; Magnani, A.; Francalacci, G.; Marino, P. Inappropriate implantable cardioverter-defibrillator discharges unrelated to supraventricular tachyarrhythmias. EP Eur. 2006, 8, 863–869. [Google Scholar] [CrossRef]
- Pfeiffer, D.; Jung, W.; Fehske, W.; Korte, T.; Manz, M.; Moosdorf, R.; Lüderitz, B. Complications of pacemaker-defibrillator devices: Diagnosis and management. Am. Heart J. 1994, 127, 1073–1080. [Google Scholar] [CrossRef]
- Knops, R.E.; Stuijt, W.v.d.; Delnoy, P.P.H.M.; Boersma, L.V.A.; Kuschyk, J.; El-Chami, M.F.; Bonnemeier, H.; Behr, E.R.; Brouwer, T.F.; Kääb, S.; et al. Efficacy and Safety of Appropriate Shocks and Antitachycardia Pacing in Transvenous and Subcutaneous Implantable Defibrillators: Analysis of All Appropriate Therapy in the PRAETORIAN Trial. Circulation 2022, 145, 321–329. [Google Scholar] [CrossRef]
- Killingsworth, C.R.; Melnick, S.B.; Litovsky, S.H.; Ideker, R.E.; Walcott, G.P. Evaluation of acute cardiac and chest wall damage after shocks with a subcutaneous implantable cardioverter defibrillator in Swine. Pacing Clin. Electrophysiol. 2013, 36, 1265–1272. [Google Scholar] [CrossRef]
- Al-Khatib, S.M.; Stevenson, W.G.; Ackerman, M.J.; Bryant, W.J.; Callans, D.J.; Curtis, A.B.; Deal, B.J.; Dickfeld, T.; Field, M.E.; Fonarow, G.C.; et al. 2017 AHA/ACC/HRS guideline for management of patients with ventricular arrhythmias and the prevention of sudden cardiac death: Executive summary: A Report of the American College of Cardiology/American Heart Association Task Force on Clinical Practice Guidelines and the Heart Rhythm Society. Heart Rhythm 2018, 15, e190–e252. [Google Scholar] [CrossRef] [PubMed]
- Quast, A.B.E.; Brouwer, T.F.; Tjong, F.V.Y.; Wilde, A.A.M.; Knops, R.E. Clinical parameters to optimize patient selection for subcutaneous and transvenous implantable defibrillator therapy. Pacing Clin. Electrophysiol. 2018, 41, 990–995. [Google Scholar] [CrossRef] [PubMed]
- Boersma, L.; Burke, M.C.; Neuzil, P.; Lambiase, P.; Friehling, T.; Theuns, D.A.; Garcia, F.; Carter, N.; Stivland, T.; Weiss, R. Infection and mortality after implantation of a subcutaneous ICD after transvenous ICD extraction. Heart Rhythm 2016, 13, 157–164. [Google Scholar] [CrossRef] [PubMed]
- Lewis, G.F.; Gold, M.R. Safety and Efficacy of the Subcutaneous Implantable Defibrillator. J. Am. Coll. Cardiol. 2016, 67, 445–454. [Google Scholar] [CrossRef]
- Sarubbi, B.; Colonna, D.; Correra, A.; Romeo, E.; D’Alto, M.; Palladino, M.T.; Virno, S.; D’Onofrio, A.; Russo, M.G. Subcutaneous implantable cardioverter defibrillator in children and adolescents: Results from the S-ICD “Monaldi care” registry. J. Interv. Card Electrophysiol. 2022, 63, 283–293. [Google Scholar] [CrossRef] [PubMed]
- Olgun, H.; Karagoz, T.; Celiker, A.; Ceviz, N. Patient- and lead-related factors affecting lead fracture in children with transvenous permanent pacemaker. Europace 2008, 10, 844–847. [Google Scholar] [CrossRef]
- Saad, T.F.; Hentschel, D.M.; Koplan, B.; Wasse, H.; Asif, A.; Patel, D.V.; Salman, L.; Carrillo, R.; Hoggard, J. Cardiovascular implantable electronic device leads in CKD and ESRD patients: Review and recommendations for practice. Semin. Dial. 2013, 26, 114–123. [Google Scholar] [CrossRef]
- Brunner, M.P.; Cronin, E.M.; Duarte, V.E.; Yu, C.; Tarakji, K.G.; Martin, D.O.; Callahan, T.; Cantillon, D.J.; Niebauer, M.J.; Saliba, W.I.; et al. Clinical predictors of adverse patient outcomes in an experience of more than 5000 chronic endovascular pacemaker and defibrillator lead extractions. Heart Rhythm 2014, 11, 799–805. [Google Scholar] [CrossRef]
- Koman, E.; Gupta, A.; Subzposh, F.; Saltzman, H.; Kutalek, S.P. Outcomes of subcutaneous implantable cardioverter-defibrillator implantation in patients on hemodialysis. J. Interv. Card Electrophysiol. 2016, 45, 219–223. [Google Scholar] [CrossRef]
- Francia, P.; Olivotto, I.; Lambiase, P.D.; Autore, C. Implantable cardioverter-defibrillators for hypertrophic cardiomyopathy: The Times They Are a-Changin’. Europace 2022, 24, 1384–1394. [Google Scholar] [CrossRef]
- Lambiase, P.D.; Gold, M.R.; Hood, M.; Boersma, L.; Theuns, D.; Burke, M.C.; Weiss, R.; Russo, A.M.; Kääb, S.; Knight, B.P. Evaluation of subcutaneous ICD early performance in hypertrophic cardiomyopathy from the pooled EFFORTLESS and IDE cohorts. Heart Rhythm 2016, 13, 1066–1074. [Google Scholar] [CrossRef]
- Elliott, P.M.; Anastasakis, A.; Borger, M.A.; Borggrefe, M.; Cecchi, F.; Charron, P.; Hagege, A.A.; Lafont, A.; Limongelli, G.; Mahrholdt, H.; et al. 2014 ESC Guidelines on diagnosis and management of hypertrophic cardiomyopathy: The Task Force for the Diagnosis and Management of Hypertrophic Cardiomyopathy of the European Society of Cardiology (ESC). Eur. Heart. J. 2014, 35, 2733–2779. [Google Scholar] [CrossRef]
- Jankelson, L.; Garber, L.; Sherrid, M.; Massera, D.; Jones, P.; Barbhaiya, C.; Holmes, D.; Knotts, R.; Bernstein, S.; Spinelli, M.; et al. Subcutaneous versus transvenous implantable defibrillator in patients with hypertrophic cardiomyopathy. Heart Rhythm 2022, 19, 759–767. [Google Scholar] [CrossRef]
- Lambiase, P.D.; Eckardt, L.; Theuns, D.A.; Betts, T.R.; Kyriacou, A.L.; Duffy, E.; Knops, R. Evaluation of subcutaneous implantable cardioverter-defibrillator performance in patients with ion channelopathies from the EFFORTLESS cohort and comparison with a meta-analysis of transvenous ICD outcomes. Heart Rhythm O2 2020, 1, 326–335. [Google Scholar] [CrossRef]
- Wang, W.; Gasperetti, A.; Sears, S.F.; Tichnell, C.; Murray, B.; Tandri, H.; James, C.A.; Calkins, H. Subcutaneous and Transvenous Defibrillators in Arrhythmogenic Right Ventricular Cardiomyopathy: A Comparison of Clinical and Quality-of-Life Outcomes. JACC Clin. Electrophysiol. 2022, 9, 394–402. [Google Scholar] [CrossRef]
- Mizusawa, Y.; Wilde, A.A.M. Brugada Syndrome. Circ. Arrhythmia Electrophysiol. 2012, 5, 606–616. [Google Scholar] [CrossRef]
- Passman, R.; Kadish, A. Polymorphic ventricular tachycardia, long Q-T syndrome, and torsades de pointes. Med. Clin. North. Am. 2001, 85, 321–341. [Google Scholar] [CrossRef]
- Towbin, J.A.; McKenna, W.J.; Abrams, D.J.; Ackerman, M.J.; Calkins, H.; Darrieux, F.C.C.; Daubert, J.P.; de Chillou, C.; DePasquale, E.C.; Desai, M.Y.; et al. 2019 HRS expert consensus statement on evaluation, risk stratification, and management of arrhythmogenic cardiomyopathy. Heart Rhythm 2019, 16, e301–e372. [Google Scholar] [CrossRef]
- Link, M.S.; Laidlaw, D.; Polonsky, B.; Zareba, W.; McNitt, S.; Gear, K.; Marcus, F.; Estes, N.A., 3rd. Ventricular arrhythmias in the North American multidisciplinary study of ARVC: Predictors, characteristics, and treatment. J. Am. Coll. Cardiol. 2014, 64, 119–125. [Google Scholar] [CrossRef]
- Corrado, D.; van Tintelen, P.J.; McKenna, W.J.; Hauer, R.N.W.; Anastastakis, A.; Asimaki, A.; Basso, C.; Bauce, B.; Brunckhorst, C.; Bucciarelli-Ducci, C.; et al. Arrhythmogenic right ventricular cardiomyopathy: Evaluation of the current diagnostic criteria and differential diagnosis. Eur. Heart J. 2019, 41, 1414–1429. [Google Scholar] [CrossRef]
- Watanabe, H.; Chinushi, M.; Izumi, D.; Sato, A.; Okada, S.; Okamura, K.; Komura, S.; Hosaka, Y.; Furushima, H.; Washizuka, T.; et al. Decrease in amplitude of intracardiac ventricular electrogram and inappropriate therapy in patients with an implantable cardioverter defibrillator. Int. Heart. J. 2006, 47, 363–370. [Google Scholar] [CrossRef]
- Von Hafe, P.; Faria, B.; Dias, G.; Cardoso, F.; Alves, M.J.; Alves, A.; Rodrigues, B.; Ribeiro, S.; Sanfins, V.; Lourenço, A. Brugada syndrome: Eligibility for subcutaneous implantable cardioverter-defibrillator after exercise stress test. Rev. Port. Cardiol. 2021, 40, 33–38. [Google Scholar] [CrossRef] [PubMed]
- D’Souza, B.A.; Epstein, A.E.; Garcia, F.C.; Kim, Y.Y.; Agarwal, S.C.; Belott, P.H.; Burke, M.C.; Leon, A.R.; Morgan, J.M.; Patton, K.K.; et al. Outcomes in Patients With Congenital Heart Disease Receiving the Subcutaneous Implantable-Cardioverter Defibrillator: Results From a Pooled Analysis From the IDE Study and the EFFORTLESS S-ICD Registry. JACC Clin. Electrophysiol. 2016, 2, 615–622. [Google Scholar] [CrossRef] [PubMed]
- Bordachar, P.; Marquié, C.; Pospiech, T.; Pasquié, J.L.; Jalal, Z.; Haissaguerre, M.; Thambo, J.B. Subcutaneous implantable cardioverter defibrillators in children, young adults and patients with congenital heart disease. Int. J. Cardiol. 2016, 203, 251–258. [Google Scholar] [CrossRef] [PubMed]
- Mondésert, B.; Dubuc, M.; Khairy, P.; Guerra, P.G.; Gosselin, G.; Thibault, B. Combination of a leadless pacemaker and subcutaneous defibrillator: First in-human report. HearthRhytm Case Rep. 2015, 1, 469–471. [Google Scholar] [CrossRef]
- Tjong, F.V.Y.; Brouwer, T.F.; Koop, B.; Soltis, B.; Shuros, A.; Schmidt, B.; Swackhamer, B.; Quast, A.E.B.; Wilde, A.A.M.; Burke, M.C.; et al. Acute and 3-Month Performance of a Communicating Leadless Antitachycardia Pacemaker and Subcutaneous Implantable Defibrillator. JACC Clin. Electrophysiol. 2017, 3, 1487–1498. [Google Scholar] [CrossRef]
- Tjong, F.V.Y.; Brouwer, T.F.; Kooiman, K.M.; Smeding, L.; Koop, B.; Soltis, B.; Shuros, A.; Wilde, A.A.M.; Burke, M.; Knops, R.E. Communicating Antitachycardia Pacing-Enabled Leadless Pacemaker and Subcutaneous Implantable Defibrillator. J. Am. Coll. Cardiol. 2016, 67, 1865–1866. [Google Scholar] [CrossRef]
- Breeman, K.T.N.; Swackhamer, B.; Brisben, A.J.; Quast, A.B.E.; Carter, N.; Shuros, A.; Soltis, B.; Koop, B.E.; Burke, M.C.; Wilde, A.A.M.; et al. Long-term performance of a novel communicating antitachycardia pacing-enabled leadless pacemaker and subcutaneous implantable cardioverter-defibrillator system: A comprehensive preclinical study. Heart Rhythm 2022, 19, 837–846. [Google Scholar] [CrossRef]
- Schneider, A.E.; Burkhart, H.M.; Ackerman, M.J.; Dearani, J.A.; Wackel, P.; Cannon, B.C. Minimally invasive epicardial implantable cardioverter-defibrillator placement for infants and children: An effective alternative to the transvenous approach. Heart Rhythm 2016, 13, 1905–1912. [Google Scholar] [CrossRef]
- Hata, H.; Sumitomo, N.; Nakai, T.; Amano, A. Retrosternal Implantation of the Cardioverter-Defibrillator Lead in an Infant. Ann. Thorac. Surg. 2017, 103, e449–e451. [Google Scholar] [CrossRef]
- Boersma, L.V.A.; Merkely, B.; Neuzil, P.; Crozier, I.G.; Akula, D.N.; Timmers, L.; Kalarus, Z.; Sherfesee, L.; DeGroot, P.J.; Thompson, A.E.; et al. Therapy From a Novel Substernal Lead: The ASD2 Study. JACC Clin. Electrophysiol. 2019, 5, 186–196. [Google Scholar] [CrossRef]
- Stiles, M.K.; Fauchier, L.; Morillo, C.A.; Wilkoff, B.L. 2019 HRS/EHRA/APHRS/LAHRS focused update to 2015 expert consensus statement on optimal implantable cardioverter-defibrillator programming and testing. Europace 2019, 21, 1442–1443. [Google Scholar] [CrossRef]
- Birnie, D.; Tung, S.; Simpson, C.; Crystal, E.; Exner, D.; Ayala Paredes, F.A.; Krahn, A.; Parkash, R.; Khaykin, Y.; Philippon, F.; et al. Complications associated with defibrillation threshold testing: The Canadian experience. Heart Rhythm 2008, 5, 387–390. [Google Scholar] [CrossRef]
- Forleo, G.B.; Gasperetti, A.; Breitenstein, A.; Laredo, M.; Schiavone, M.; Ziacchi, M.; Vogler, J.; Ricciardi, D.; Palmisano, P.; Piro, A.; et al. Subcutaneous implantable cardioverter-defibrillator and defibrillation testing: A propensity-matched pilot study. Heart Rhythm 2021, 18, 2072–2079. [Google Scholar] [CrossRef]
- Quast, A.B.E.; Baalman, S.W.E.; Brouwer, T.F.; Smeding, L.; Wilde, A.A.M.; Burke, M.C.; Knops, R.E. A novel tool to evaluate the implant position and predict defibrillation success of the subcutaneous implantable cardioverter-defibrillator: The PRAETORIAN score. Heart Rhythm 2019, 16, 403–410. [Google Scholar] [CrossRef]
- Quast, A.B.E.; Baalman, S.W.E.; Betts, T.R.; Boersma, L.V.A.; Bonnemeier, H.; Boveda, S.; Brouwer, T.F.; Burke, M.C.; Delnoy, P.; El-Chami, M.; et al. Rationale and design of the PRAETORIAN-DFT trial: A prospective randomized CompArative trial of SubcutanEous ImplanTable CardiOverter-DefibrillatoR ImplANtation with and without DeFibrillation testing. Am. Heart J. 2019, 214, 167–174. [Google Scholar] [CrossRef]
- Ganeshan, R.; Enriquez, A.D.; Freeman, J.V. Remote monitoring of implantable cardiac devices: Current state and future directions. Curr. Opin. Cardiol. 2018, 33, 20–30. [Google Scholar] [CrossRef]
- De Filippo, P.; Luzi, M.; D’Onofrio, A.; Bongiorni, M.G.; Giammaria, M.; Bisignani, G.; Menardi, E.; Ferrari, P.; Bianchi, V.; Viani, S.; et al. Remote monitoring of subcutaneous implantable cardioverter defibrillators. J. Interv. Card Electrophysiol. 2018, 53, 373–381. [Google Scholar] [CrossRef]
| Study | Year | Type | Aim of Study | Primary Endpoints | Secondary Endpoints | Results |
|---|---|---|---|---|---|---|
| IDE (Investigational Device Exemption) Trial [15] | 2013 | Prospective, non-randomized, multicenter clinical study | Safety and effectiveness of S-ICD | -Shock effectiveness in converting induced VF in conversion test -Complication-free Rate at 180 days | // | -100% VF conversion rate at 180 days -92–99% complications-free rate at 180 days |
| EFFORTLESS (Evaluation of factors impacting clinical outcome and cost effectiveness of the S-ICD) Registry [13] | 2017 | Prospective, non-randomized, multicenter observational registry | Early, mid- and long-term clinical effectiveness | -Complication-free rate at 30 days -Complication-free rate at 360 days -Inappropriate shocks-free rate for AF/SVT | // | -97% complication-free rate at 30 days -94% complication-free rate at 360 days -7% inappropriate shock rate (94% oversensed episodes) |
| S-ICD post approval Study [14] | 2017 | Prospective, non-randomized, multicenter registry | Safety and effectiveness of S-ICD | -Complication-free rate at 60 months -Shock effectiveness in converting spontaneous VT/VF at 60 months | -Electrode-related complications-free rate at 60 months -First shock effectiveness i converting induced and spontaneous VT/VF at 60 months | -96.2% complication- free rate at 30 days -98.7% successful conversion rate of induced VT/VF at 60 months |
| PRAETORIAN (Prospective randomized comparison of subcutaneous and transvenous implantable cardioverter defibrillator therapy) Study [11] | 2020 | Prospective, randomized, international, controlled trial | Comparison of safety and effectiveness in TV-ICD and S-ICD (non-inferiority) | -Adverse event rate at 48 months | -MACE, appropriate and inappropriate shocks, time to successful therapy, first shock conversion efficacy, implant procedure time, hospitalization rate, fluoroscopy time, cardiac (pre)-syncope events, cross over to the other arm, cardiac decompensation at 48 months -Quality of life at 30 months | -No difference in overall and arrhythmic mortality -Four times lead-related complications rate in TV -ICD -Two times infection rate in TV-ICD -No difference in complications rate in 4 years -No difference in inappropriate shock rate |
| UNTOUCHED (Understanding outcomes with the S-ICD in primary prevention patients with low ejection fraction) Study [10] | 2021 | Prospective, non-randomized, multinational trial | Safety and effectiveness of S-ICD | -Inappropriate shocks free rate at 18 months | -Freedom from system and procedure related complication at 30 days -All cause shock free rate at 18 months | -95.9% inappropriate shock-free rate at 18 months -90.6% all-cause shock-free rate at 18 months -92.7% complications-free rate at 18 months |
| ATLAS (Avoid transvenous leads in appropriate subjects) Trial [12] | 2022 | Prospective, randomized, multicenter controlled study | Comparison of safety and effectiveness in TV-ICD and S-ICD (superiority) | -Lead-related complications at 6 months -Other complications at 6 months | -Late device-related complications after 6 months -Arrhythmic deaths, visits, inappropriate shocks, all-cause mortality, economic analysis, patients acceptance after 6 months | -12 times lead-related complications in TV-ICD |
| TV-ICD | S-ICD | |
|---|---|---|
| Pre-implant Screening | Not needed | Needed |
| Implant Technique | Transvenous | Subcutaneous |
| Sedation | Local | Deep/general anesthesia |
| Fluoroscopy | Needed | Not needed |
| Electrocardiogram | Intracavitary ECG | 12-lead ECG |
| Inappropriate shocks | SVT | T oversensing, myopotential, discrimination error |
| Anti-tachycardia pacing | Possible | Not possible |
| SHOCK threshold | 5–30 J | 80 J |
| Infections | Systemic infections | Pocket infections |
| Lead complications | Dislocations/fractures; tricuspid regurgitation, pericardial effusion or pericarditis, cardiac perforation | Lead movement/suboptimal lead position |
| Indications | Pitfalls |
|---|---|
| Indication for ICD when pacing for bradycardia, cardiac resynchronization or ATP is not needed | Disease progression with need for anti-bradycardia pacing, cardiac resynchronization or enhancement of antiarrhythmic medical therapy |
| Congenital heart disease | Frequent development of conduction system disfunction overtime |
| Anatomical barriers to IV-ICD implantation (i.e., venous occlusion) | Aesthetic defect in thin women |
| History of IV lead infection | Only defibrillation therapy provided |
| Immunocompromised individuals | Large surgical wound, need for large disinfection area |
| Hemodialysis | \\ |
| Young patients | Aesthetic defect, contact sports forbidden |
| Ion channelopathies | Polymorphic or monomorphic VTs not treatable |
| Hypertrophic cardiomyopathy | Increased risk of T oversensing |
| Dilated cardiomyopathy | Usually manifested with VTs of variable cardiac frequency (slower VTs not treated by the device) |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Guarracini, F.; Preda, A.; Bonvicini, E.; Coser, A.; Martin, M.; Quintarelli, S.; Gigli, L.; Baroni, M.; Vargiu, S.; Varrenti, M.; et al. Subcutaneous Implantable Cardioverter Defibrillator: A Contemporary Overview. Life 2023, 13, 1652. https://doi.org/10.3390/life13081652
Guarracini F, Preda A, Bonvicini E, Coser A, Martin M, Quintarelli S, Gigli L, Baroni M, Vargiu S, Varrenti M, et al. Subcutaneous Implantable Cardioverter Defibrillator: A Contemporary Overview. Life. 2023; 13(8):1652. https://doi.org/10.3390/life13081652
Chicago/Turabian StyleGuarracini, Fabrizio, Alberto Preda, Eleonora Bonvicini, Alessio Coser, Marta Martin, Silvia Quintarelli, Lorenzo Gigli, Matteo Baroni, Sara Vargiu, Marisa Varrenti, and et al. 2023. "Subcutaneous Implantable Cardioverter Defibrillator: A Contemporary Overview" Life 13, no. 8: 1652. https://doi.org/10.3390/life13081652
APA StyleGuarracini, F., Preda, A., Bonvicini, E., Coser, A., Martin, M., Quintarelli, S., Gigli, L., Baroni, M., Vargiu, S., Varrenti, M., Forleo, G. B., Mazzone, P., Bonmassari, R., Marini, M., & Droghetti, A. (2023). Subcutaneous Implantable Cardioverter Defibrillator: A Contemporary Overview. Life, 13(8), 1652. https://doi.org/10.3390/life13081652








