Advancement in Solubilization Approaches: A Step towards Bioavailability Enhancement of Poorly Soluble Drugs
Abstract
1. Introduction
Review Highlights
- The aqueous solubility of a drug plays a crucial role in drug dissolution and release, absorption, and, consequently, bioavailability.
- Conventional approaches, which include particle size reduction, solid dispersion, co-crystallization, prodrug approach, supercritical fluid technology, and inclusion complex, have been in use for decades for the enhancement of the aqueous solubility of poorly soluble drugs.
- Nanotechnology has the potential to revive poorly performing marketed drugs and many of those pre-clinically promising candidates that were “shelved” due to inadequate water-solubility.
- A variety of nanocarriers have been utilized and are still at the development stage. These include the dendrimers, micelles, SLNs, MOFs, CNTs, nanogels, and mesoporous silica nanoparticles used to increase the bioavailability of poorly soluble drugs; they could be useful for the future formulation of development research.
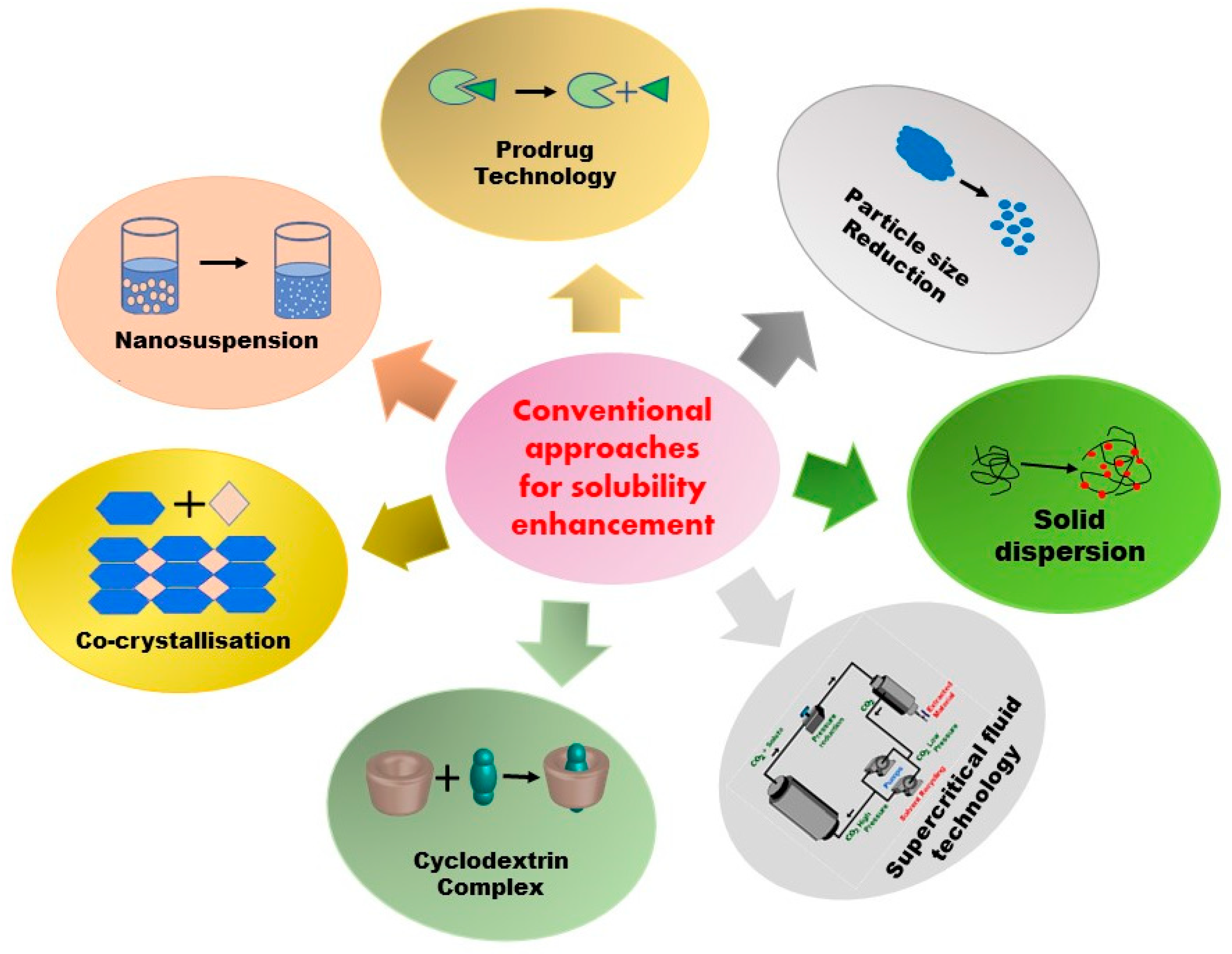
2. Conventional Approaches
2.1. Particle Size Reduction
2.2. Cyclodextrin Inclusion Complexes
2.3. Solid Dispersions
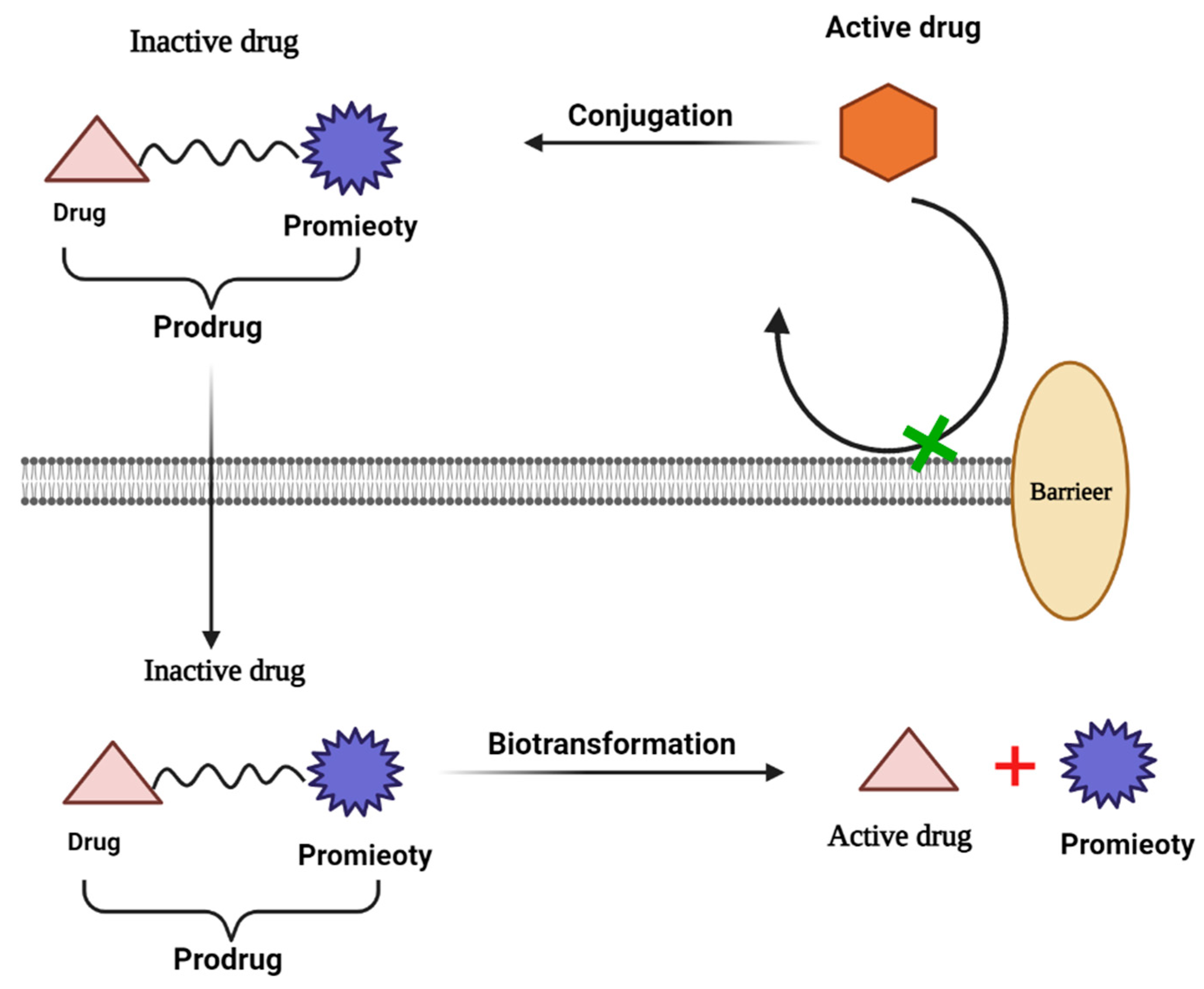
2.4. Prodrugs
2.5. Co-Crystallization
2.6. Supercritical Fluid Technology (SCF)
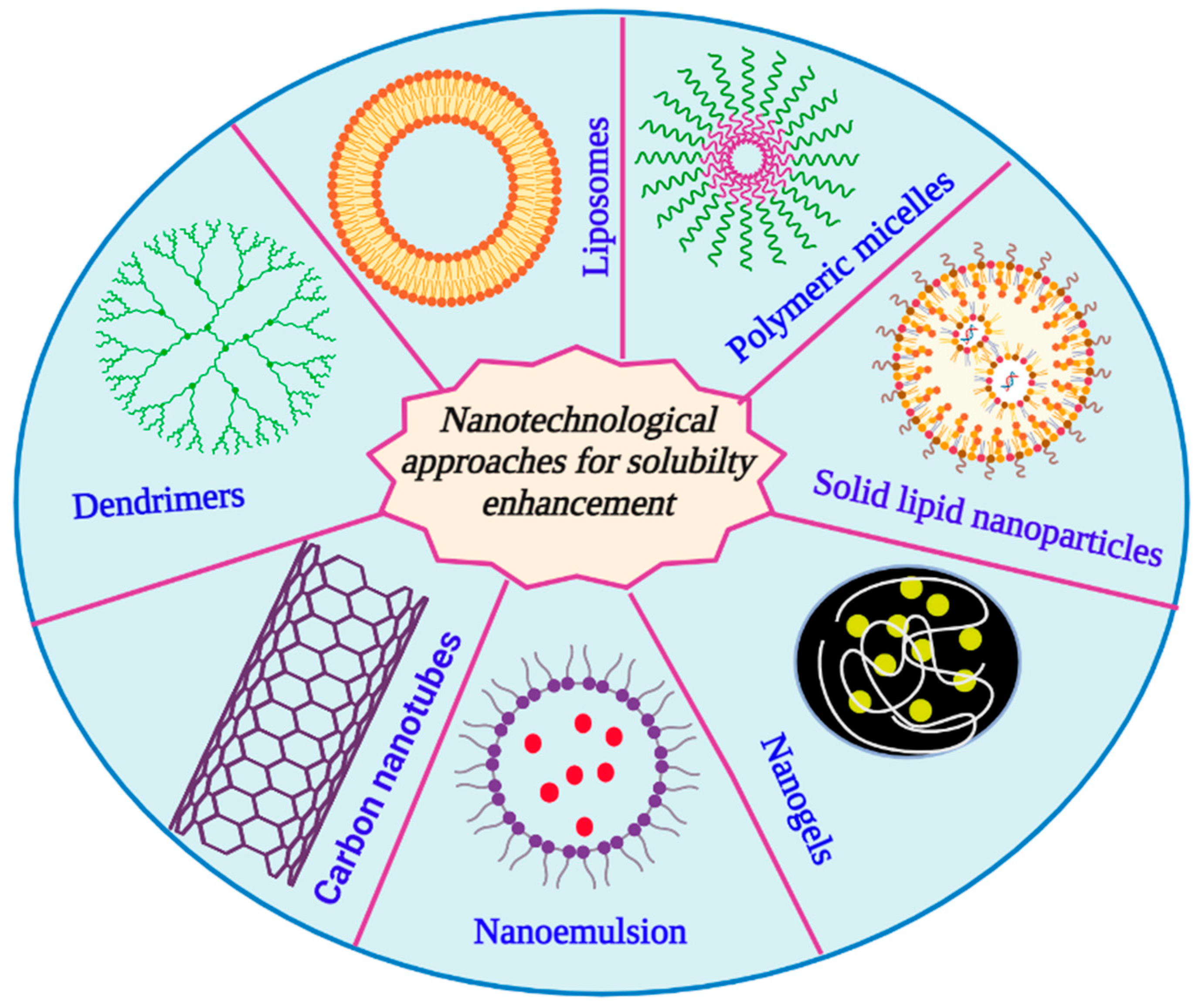
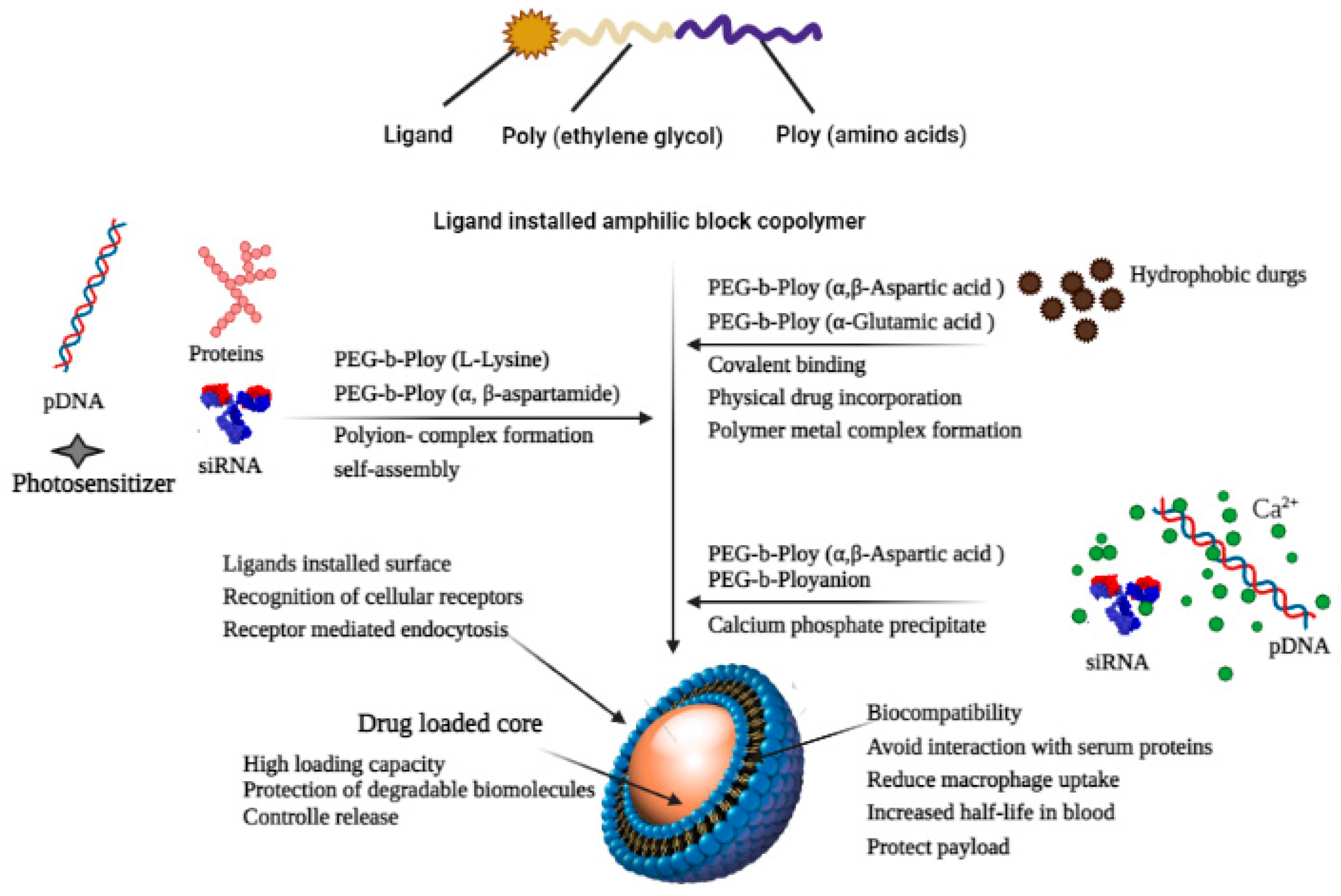
3. Nanotechnological Approaches for Solubility Enhancement
3.1. Liposomes
3.2. Dendrimers
3.3. Nanosuspensions
3.4. Micelles
3.5. Solid Lipid Nanoparticles and Nanostructured Lipid Carriers
3.6. Supercritical Antisolvent (SAS)
3.7. Nanoemulsions
3.8. Nanogels
3.9. Metal Organic Frameworks (MOFs)
3.10. Carbon Nanotubes
3.11. Mesoporous Silica
| Delivery System/Method Employed | Polymer Used | Drugs/API | Structure | Details | References |
|---|---|---|---|---|---|
| PAMAM Dendrimer | Amine and ester-terminated PAMAM Dendrimers | Nifedipine | 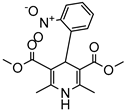 | Dendrimers composed of poly (amidoamine), or PAMAM, can improve the solubility of insoluble drugs in water at pH 7. | [163] |
| Dendrimers made of polyamidoamine(PAMAM) G3.5 and PAMAM G4.5. | Oxaliplatin |  | The solubility of oxaliplatin increases roughly linearly with dendrimer concentration. | [164] | |
| Dendrimers made from PAMAM | Temozolomid | 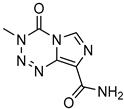 | TMZ solubility was shown to be enhanced in some solvent systems, with dendrimer, ethanol, and tween-20 showed construction and related in solubility. | [165] | |
| PAMAM dendrimers with pyrrolidone modification | Indomethacin | 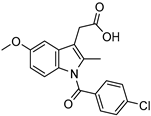 | The drug’s solubility and intracellular delivery are being improved | [166] | |
| PAMAM dendrimers | Nicotinic acid | 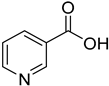 | PAMAM dendrimers of different generations (G1–G4) have the ability to dramatically improve nicotinic acid solubility. | [167] | |
| Polyether dendrimer | Artemether | 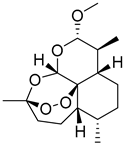 | Due to their excellent water solubility, non-immunogenicity, and increased biocompatibility, they are used as drug carriers. | [168] | |
| PAMAM dendrimers | Ibuprofen |  | PAMAM dendrimers improve ibuprofen solubility much more than SDS micelles. | [169] | |
| PAMAM and Lauryl PAMAM dendrimer | Propranolol | 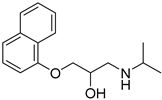 | Propranolol’s solubility has been improved | [170] | |
| Silica | Cur-fls & Cur-sls | Curcumin | 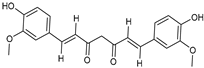 | Improved solubility with enhanced oral bioavailability up to 7-fold high than convectional suspensions. | [171] |
| Thin film hydration sonication | Glycol, Eudragit S100 | Sorafenib | 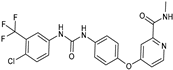 | It improved systemic exposure of about four-fold. | [172] |
| Thin-film hydration sonication | Lecithin | Cefotaxime | 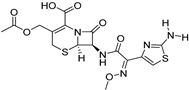 | About five-fold increase of in oral bioavailability and improved solubility | [173] |
| Thin-film hydration sonication | Soy lecithin | Capsaicin | 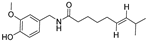 | Oral bioavailability and improved solubility increase about three-fold. | [174] |
| Film deposition on the carrier | HSPC | Lopinavir | 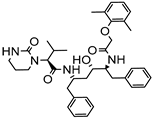 | Improved solubility with enhanced oral bioavailability up to 2-fold. | [175] |
| Thin-film hydration sonicate | DSPC | Asinine maleate | 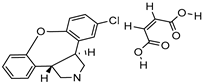 | About one-fold increase in oral bioavailability and improved solubility | [176] |
| Thin-film hydration sonication-freeze thawing | SPC | Spironolactone | 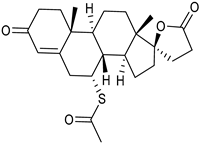 | Enhanced oral bioavailability with improved solubility up to 2-fold. | [177] |
| High-pressure homogenization | Poly Na styrene sulfonate | Paclitaxel | 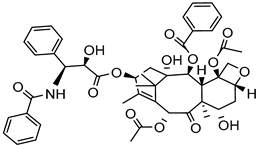 | About 14 -fold increase in oral bioavailability and improved solubility and drug dissolution: 20% (120 min). | [178] |
| Antisolvent precipitation | Pluronic® F68 | Puerarin | 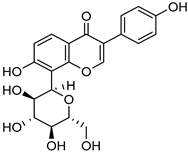 | Enhanced oral bioavailability with improved solubility up to 4-fold | [179] |
| Spray drying | SDS | Alisertib isoproxil |  | Drug dissolution: ~14% (1/2 min) enhanced oral bioavailability with improved solubility up to 4-fold. | [180] |
| Antisolvent precipitation | Ethyl cellulose | Domperidone | 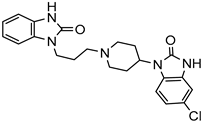 | Fifty percent (30 min) and 65 percent (60 min) drug dissolution and enhanced oral bioavailability with improved solubility up to 2-fold. | [181] |
| Precipitation-sonication | PVA | Cinnarizine | 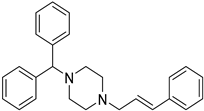 | One hundred percent drug in dissolution (240 s) enhanced oral bioavailability with improved solubility up to 2-fold. | [182] |
| Magnetic stirring-milling | PVP-K30 | Glyburide | 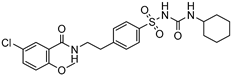 | 100 percent drug in dissolution (30 min) increased oral bioavailability with improved solubility up to four-fold. | [183] |
| Hot homogenization sonication | Stearic acid | Rosuvastatin | 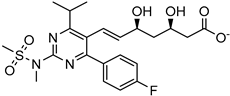 | Drug release: ~45 percent (120 min) and ~80 percent (10 h) Improved oral bioavailability with improved solubility up to 8-fold. | [184] |
| Micro emulsification | Compritol | Rifampicin | 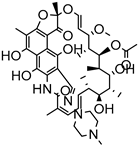 | Enhanced oral bioavailability with increased solubility up to 8-fold. | [185] |
| Emulsification sonication | Precirol® ATO-5, palmitic acid, Gelucire® 50/13, N | Resveratrol |  | Enhanced oral bioavailability and having a higher level of solubility up to 7-fold. | [186] |
4. Conclusions
5. Challenges and Future Perspectives
Author Contributions
Funding
Conflicts of Interest
References
- Rathi, P.B.; Kale, M.; Soleymani, J.; Jouyban, A. Solubility of etoricoxib in aqueous solutions of glycerin, methanol, polyethylene glycols 200, 400, 600, and propylene glycol at 298.2 K. J. Chem. Eng. Data 2018, 63, 321–330. [Google Scholar] [CrossRef]
- Murdande, S.B.; Pikal, M.J.; Shanker, R.M.; Bogner, R.H. Solubility advantage of amorphous pharmaceuticals: II. Application of quantitative thermodynamic relationships for prediction of solubility enhancement in structurally diverse insoluble pharmaceuticals. Pharm. Res. 2010, 27, 2704–2714. [Google Scholar] [CrossRef] [PubMed]
- MS, A.K.; RAJESH, M.; SUBRAMANIAN, L. Solubility enhancement techniques: A comprehensive review. World J. Biol. Pharm. Health Sci. 2023, 13, 141–149. [Google Scholar] [CrossRef]
- Jain, S.; Patel, N.; Lin, S. Solubility and dissolution enhancement strategies: Current understanding and recent trends. Drug Dev. Ind. Pharm. 2015, 41, 875–887. [Google Scholar] [CrossRef]
- Mantri, R.; Sanghvi, R. Solubility of pharmaceutical solids. In Developing Solid Oral Dosage Forms; Elsevier: Amsterdam, The Netherlands, 2017; pp. 3–22. [Google Scholar]
- Saal, C.; Petereit, A.C. Optimizing solubility: Kinetic versus thermodynamic solubility temptations and risks. Eur. J. Pharm. Sci. 2012, 47, 589–595. [Google Scholar] [CrossRef]
- Lu, J.X.; Tupper, C.; Murray, J. Biochemistry, Dissolution and Solubility; StatPearls: Treasure Island, FL, USA, 2022. [Google Scholar]
- Gabor, F.; Fillafer, C.; Neutsch, L.; Ratzinger, G.; Wirth, M. Improving Oral Delivery. In Drug Delivery; Springer: Berlin/Heidelberg, Germany, 2010; pp. 345–398. [Google Scholar] [CrossRef]
- Bhalani, D.V.; Nutan, B.; Kumar, A.; Singh Chandel, A.K. Bioavailability Enhancement Techniques for Poorly Aqueous Soluble Drugs and Therapeutics. Biomedicines 2022, 10, 2055. [Google Scholar] [CrossRef]
- Joshi, J.; Nainwal, N.; Saharan, V.A. Review on hydrotropy: A potential approach for the solubility enhancement of poorly soluble drug. Asian J. Pharm. Clin. Res. 2019, 12, 19–26. [Google Scholar] [CrossRef]
- Tsume, Y.; Mudie, D.M.; Langguth, P.; Amidon, G.E.; Amidon, G.L. The Biopharmaceutics Classification System: Subclasses for in vivo predictive dissolution (IPD) methodology and IVIVC. Eur. J. Pharm. Sci. 2014, 57, 152–163. [Google Scholar] [CrossRef]
- Khadka, P.; Ro, J.; Kim, H.; Kim, I.; Kim, J.T.; Kim, H.; Cho, J.M.; Yun, G.; Lee, J. Pharmaceutical particle technologies: An approach to improve drug solubility, dissolution and bioavailability. Asian J. Pharm. Sci. 2014, 9, 304–316. [Google Scholar] [CrossRef]
- Kalepu, S.; Nekkanti, V. Insoluble drug delivery strategies: Review of recent advances and business prospects. Acta Pharm. Sin. B 2015, 5, 442–453. [Google Scholar] [CrossRef]
- Khan, A.D.; Tabish, M.; Kaushik, R.; Saxena, V.; Kesharwani, P.; Gupta, S.; Alam, M.N.; Sharma, V. Hydrotropy: Recent Advancements in Enhancement of Drug Solubility and Formulation Development. Int. J. Drug Deliv. Technol. 2021, 11. [Google Scholar] [CrossRef]
- Savjani, K.T.; Gajjar, A.K.; Savjani, J.K. Drug solubility: Importance and enhancement techniques. Int. Sch. Res. Not. 2012, 2012, 195727. [Google Scholar] [CrossRef] [PubMed]
- Gupta, J.; Devi, A. Solubility Enhancement Techniques for Poorly Soluble Pharmaceuticals: A Review. Indian J. Pharm. Biol. Res. 2019, 7, 09–16. [Google Scholar] [CrossRef]
- Patel, V.R.; Agrawal, Y.K. Nanosuspension: An approach to enhance solubility of drugs. J. Adv. Pharm. Technol. Res. 2011, 2, 81–87. [Google Scholar] [CrossRef] [PubMed]
- Lakshmi, P.; Kumar, G.A. Nanosuspension technology: A review. Int. J. Pharm. Sci. 2010, 2, 35–40. [Google Scholar]
- Chauhan, A.S. Dendrimers for Drug Delivery. Molecules 2018, 23, 938. [Google Scholar] [CrossRef]
- Alfei, S.; Schito, A.M.; Zuccari, G. Considerable Improvement of Ursolic Acid Water Solubility by Its Encapsulation in Dendrimer Nanoparticles: Design, Synthesis and Physicochemical Characterization. Nanomaterials 2021, 11, 2196. [Google Scholar] [CrossRef]
- Bansal, K.K.; Kakde, D.; Gupta, U.; Jain, N.K. Development and Characterization of Triazine Based Dendrimers for Delivery of Antitumor Agent. J. Nanosci. Nanotechnol. 2010, 10, 8395–8404. [Google Scholar] [CrossRef]
- Clulow, A.J.; Barber, B.; Salim, M.; Ryan, T.; Boyd, B.J. Synergistic and antagonistic effects of non-ionic surfactants with bile salt + phospholipid mixed micelles on the solubility of poorly water-soluble drugs. Int. J. Pharm. 2020, 588, 119762. [Google Scholar] [CrossRef]
- Lin, Q.; Liang, R.; Zhong, F.; Ye, A.; Hemar, Y.; Yang, Z.; Singh, H. Self-Assembled Micelles Based on OSA-Modified Starches for Enhancing Solubility of beta-Carotene: Effect of Starch Macromolecular Architecture. J. Agric. Food Chem. 2019, 67, 6614–6624. [Google Scholar] [CrossRef]
- Bansal, K.K.; Lariya, N. Block Copolymer Micelles in Drug Delivery and Cancer Therapy. Chron. Pharm. Sci. 2018, 2, 534–544. [Google Scholar]
- Bansal, K.K.; Gupta, J.; Rosling, A.; Rosenholm, J.M. Renewable poly(delta-decalactone) based block copolymer micelles as drug delivery vehicle: In vitro and in vivo evaluation. Saudi Pharm. J. 2018, 26, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Valdes, S.A.; Alzhrani, R.F.; Rodriguez, A.; Lansakara, P.D.; Thakkar, S.G.; Cui, Z. A solid lipid nanoparticle formulation of 4-(N)-docosahexaenoyl 2′, 2′-difluorodeoxycytidine with increased solubility, stability, and antitumor activity. Int. J. Pharm. 2019, 570, 118609. [Google Scholar] [CrossRef] [PubMed]
- Mishra, V.; Bansal, K.K.; Verma, A.; Yadav, N.; Thakur, S.; Sudhakar, K.; Rosenholm, J.M. Solid Lipid Nanoparticles: Emerging Colloidal Nano Drug Delivery Systems. Pharmaceutics 2018, 10, 191. [Google Scholar] [CrossRef] [PubMed]
- Yin, X.; Luo, L.; Li, W.; Yang, J.; Zhu, C.; Jiang, M.; Qin, B.; Yuan, X.; Yin, H.; Lu, Y.; et al. A cabazitaxel liposome for increased solubility, enhanced antitumor effect and reduced systemic toxicity. Asian J. Pharm. Sci. 2019, 14, 658–667. [Google Scholar] [CrossRef]
- Zhu, Q.; Guo, T.; Xia, D.; Li, X.; Zhu, C.; Li, H.; Ouyang, D.; Zhang, J.; Gan, Y. Pluronic F127-modified liposome-containing tacrolimus-cyclodextrin inclusion complexes: Improved solubility, cellular uptake and intestinal penetration. J. Pharm. Pharmacol. 2013, 65, 1107–1117. [Google Scholar] [CrossRef] [PubMed]
- Wang, Y.; Zhang, Z.; Zheng, C.; Zhao, X.; Zheng, Y.; Liu, Q.; Liu, Y.; Shi, L. Multistage Adaptive Nanoparticle Overcomes Biological Barriers for Effective Chemotherapy. Small 2021, 17, e2100578. [Google Scholar] [CrossRef]
- Ogden, J.; Parry-Billings, M. Nanotechnology approaches to solving the problems of poorly water-soluble drugs. Drug Discov. 2005, 6, 71–76. [Google Scholar]
- Bulbake, U.; Doppalapudi, S.; Kommineni, N.; Khan, W. Liposomal Formulations in Clinical Use: An Updated Review. Pharmaceutics 2017, 9, 12. [Google Scholar] [CrossRef]
- Tenchov, R.; Bird, R.; Curtze, A.E.; Zhou, Q. Lipid Nanoparticles horizontal line From Liposomes to mRNA Vaccine Delivery, a Landscape of Research Diversity and Advancement. ACS Nano 2021, 15, 16982–17015. [Google Scholar] [CrossRef]
- Deshmukh, A.S.; Tiwari, K.J.; Mahajan, V.R. Solubility enhancement techniques for poorly water-soluble drugs. Int. J. Pharm. Sci. Nanotechnol. 2017, 10, 3701–3708. [Google Scholar] [CrossRef]
- Hildebrand, J.H. Solubility. J. Am. Chem. Soc. 1916, 38, 1452–1473. [Google Scholar] [CrossRef]
- Vadlamudi, M.K.; Dhanaraj, S. Disparate practical way of doing solubility enhancement study to improve the bioavailability of poorly soluble drugs. J. Chem. Pharm. Res. 2016, 8, 208–235. [Google Scholar]
- Chaudhary, A.; Nagaich, U.; Gulati, N.; Sharma, V.; Khosa, R.; Partapur, M.U. Enhancement of solubilization and bioavailability of poorly soluble drugs by physical and chemical modifications: A recent review. J. Adv. Pharm. Educ. Res. 2012, 2, 32–67. [Google Scholar]
- Jagtap, S.; Magdum, C.; Jadge, D.; Jagtap, R. Research. Solubility enhancement technique: A review. J. Pharm. Sci. Res. 2018, 10, 2205–2211. [Google Scholar]
- Ghumre, P.B.; Bote, S.S.; Kotgir, S.R.; Korde, A.B.; Bhosale, B.S.; Chaudhari, R.B. Solubility Enhancement Technique—A Review. World J. Pharm. Res. 2021, 10, 571–589. [Google Scholar]
- Blagden, N.; de Matas, M.; Gavan, P.T.; York, P. Crystal engineering of active pharmaceutical ingredients to improve solubility and dissolution rates. Adv. Drug Deliv. Rev. 2007, 59, 617–630. [Google Scholar] [CrossRef]
- Kumar, A.; Sahoo, S.K.; Padhee, K.; Kochar, P.S.; Sathapathy, A.; Pathak, N.J.P.G. Review on solubility enhancement techniques for hydrophobic drugs. Int. J. Compr. Pharm. 2011, 3, 001–007. [Google Scholar]
- Vandana, K.; Raju, Y.P.; Chowdary, V.H.; Sushma, M.; Kumar, N.V. An overview on in situ micronization technique—An emerging novel concept in advanced drug delivery. Saudi Pharm. J. 2014, 22, 283–289. [Google Scholar] [CrossRef]
- Sherje, A.P.; Jadhav, M. beta-Cyclodextrin-based inclusion complexes and nanocomposites of rivaroxaban for solubility enhancement. J. Mater. Sci. Mater. Med. 2018, 29, 186. [Google Scholar] [CrossRef]
- Semcheddine, F.; Guissi, N.E.I.; Liu, X.; Wu, Z.; Wang, B. Effects of the Preparation Method on the Formation of True Nimodipine SBE-beta-CD/HP-beta-CD Inclusion Complexes and Their Dissolution Rates Enhancement. AAPS PharmSciTech 2015, 16, 704–715. [Google Scholar] [CrossRef] [PubMed]
- Argade, P.; Magar, D.; Saudagar, R.B. Solid dispersion: Solubility enhancement technique for poorly water soluble drugs. J. Adv. Pharm. Edu. Res. 2013, 3, 427–439. [Google Scholar]
- Dhirendra, K.; Lewis, S.; Udupa, N.; Atin, K. Solid dispersions: A review. Pak. J. Pharm. Sci. 2009, 22, 234–246. [Google Scholar]
- Cid, A.G.; Simonazzi, A.; Palma, S.D.; Bermudez, J.M. Solid dispersion technology as a strategy to improve the bioavailability of poorly soluble drugs. Ther. Deliv. 2019, 10, 363–382. [Google Scholar] [CrossRef] [PubMed]
- Thakur, N.; Goswami, M.; Mittal, P.; Goyal, P.; Sood, A. Solid Dispersion: A Novel Approach for Bioavailability Enhancement. Int. J. Pharm. Sci. Rev. Res. 2016, 3, 265–272. [Google Scholar]
- Devhare, L.; Kore, P.K. A Recent Review on Bioavailability and Solubility Enhancement of Poorly Soluble Drugs by Physical and Chemical Modifications. Res. Chron. Health Sci. 2016, 2, 299–308. [Google Scholar]
- Muniandy, A.; Lee, C.S.; Lim, W.H.; Pichika, M.R. Investigation of hyperbranched Poly (glycerol esteramide) as potential drug carrier in solid dispersion for solubility enhancement of lovastatin. J. Drug Deliv. Sci. Technol. 2021, 61, 102237. [Google Scholar] [CrossRef]
- Jatwani, S.; Rana, A.; Singh, G.; Aggarwal, G. Solubility and dissolution enhancement of simvastatin using synergistic effect of hydrophilic carriers. Der. Pharm. Lett. 2011, 3, 280–293. [Google Scholar]
- Gao, N.; Guo, M.; Fu, Q.; He, Z. Application of hot melt extrusion to enhance the dissolution and oral bioavailability of oleanolic acid. Asian J. Pharm. Sci. 2017, 12, 66–72. [Google Scholar] [CrossRef]
- Tran, P.; Pyo, Y.-C.; Kim, D.-H.; Lee, S.-E.; Kim, J.-K.; Park, J.-S. Overview of the manufacturing methods of solid dispersion technology for improving the solubility of poorly water-soluble drugs and application to anticancer drugs. Pharmaceutics 2019, 11, 132. [Google Scholar] [CrossRef]
- Chen, Y.; Shi, Q.; Chen, Z.; Zheng, J.; Xu, H.; Li, J.; Liu, H. Preparation and characterization of emulsified solid dispersions containing docetaxel. Arch. Pharmacal Res. 2011, 34, 1909–1917. [Google Scholar] [CrossRef] [PubMed]
- Herbrink, M.; Schellens, J.H.; Beijnen, J.H.; Nuijen, B. Improving the solubility of nilotinib through novel spray-dried solid dispersions. Int. J. Pharm. 2017, 529, 294–302. [Google Scholar] [CrossRef] [PubMed]
- Chamsai, B.; Limmatvapirat, S.; Sungthongjeen, S.; Sriamornsak, P. Enhancement of solubility and oral bioavailability of manidipine by formation of ternary solid dispersion with d-alpha-tocopherol polyethylene glycol 1000 succinate and copovidone. Drug Dev. Ind. Pharm. 2017, 43, 2064–2075. [Google Scholar] [CrossRef]
- Vo, C.L.; Park, C.; Lee, B.J. Current trends and future perspectives of solid dispersions containing poorly water-soluble drugs. Eur. J. Pharm. Biopharm. 2013, 85, 799–813. [Google Scholar] [CrossRef] [PubMed]
- Ita, K. Chapter 7—Prodrugs. In Transdermal Drug Delivery; Ita, K., Ed.; Academic Press: Cambridge, MA, USA, 2020; pp. 123–141. [Google Scholar]
- Rodriguez-Aller, M.; Guillarme, D.; Veuthey, J.-L.; Gurny, R. Strategies for formulating and delivering poorly water-soluble drugs. J. Drug Deliv. Sci. Technol. 2015, 30, 342–351. [Google Scholar] [CrossRef]
- Jornada, D.H.; dos Santos Fernandes, G.F.; Chiba, D.E.; de Melo, T.R.; dos Santos, J.L.; Chung, M.C. The Prodrug Approach: A Successful Tool for Improving Drug Solubility. Molecules 2015, 21, 42. [Google Scholar] [CrossRef]
- Najjar, A.; Karaman, R. The prodrug approach in the era of drug design. Expert Opin. Drug Deliv. 2019, 16, 1–5. [Google Scholar] [CrossRef]
- Beaulieu, P.L.; De Marte, J.; Garneau, M.; Luo, L.; Stammers, T.; Telang, C.; Wernic, D.; Kukolj, G.; Duan, J. A prodrug strategy for the oral delivery of a poorly soluble HCV NS5B thumb pocket 1 polymerase inhibitor using self-emulsifying drug delivery systems (SEDDS). Bioorganic Med. Chem. Lett. 2015, 25, 210–215. [Google Scholar] [CrossRef]
- Ain, S.; Ain, Q.; Parveen, S. An overview on various approaches used for solubilization of poorly soluble drugs. Int. J. Pharm. Investig. 2009, 2, 84–104. [Google Scholar]
- Karagianni, A.; Malamatari, M.; Kachrimanis, K. Pharmaceutical Cocrystals: New Solid Phase Modification Approaches for the Formulation of APIs. Pharmaceutics 2018, 10, 18. [Google Scholar] [CrossRef]
- Sugandha, K.; Kaity, S.; Mukherjee, S.; Isaac, J.; Ghosh, A. Solubility enhancement of ezetimibe by a cocrystal engineering technique. Cryst. Growth Des. 2014, 14, 4475–4486. [Google Scholar] [CrossRef]
- Kankala, R.K.; Zhang, Y.S.; Wang, S.B.; Lee, C.H.; Chen, A.Z. Supercritical Fluid Technology: An Emphasis on Drug Delivery and Related Biomedical Applications. Adv. Health Mater. 2017, 6, 1700433. [Google Scholar] [CrossRef] [PubMed]
- Deshpande, P.B.; Kumar, G.A.; Kumar, A.R.; Shavi, G.V.; Karthik, A.; Reddy, M.S.; Udupa, N. Supercritical fluid technology: Concepts and pharmaceutical applications. PDA J. Pharm. Sci. Technol. 2011, 65, 333–344. [Google Scholar] [CrossRef] [PubMed]
- Kumar, M.; Sharma, Y.; Chahar, K.; Kumari, L.; Mishra, L.; Patel, P.; Singh, D.; Kurmi, B.D. Validation of a Novel Supercritical Fluid Extractor/Dryer Combo Instrument. Assay Drug Dev. Technol. 2023, 21, 126–136. [Google Scholar] [CrossRef] [PubMed]
- Pasquali, I.; Bettini, R.; Giordano, F. Supercritical fluid technologies: An innovative approach for manipulating the solid-state of pharmaceuticals. Adv. Drug Deliv. Rev. 2008, 60, 399–410. [Google Scholar] [CrossRef]
- Girotra, P.; Singh, S.K.; Nagpal, K. Supercritical fluid technology: A promising approach in pharmaceutical research. Pharm. Dev. Technol. 2013, 18, 22–38. [Google Scholar] [CrossRef]
- Misra, S.K.; Pathak, K. Supercritical fluid technology for solubilization of poorly water soluble drugs via micro- and naonosized particle generation. ADMET DMPK 2020, 8, 355–374. [Google Scholar] [CrossRef]
- Jia, J.; Wang, J.; Zhang, K.; Zhou, D.; Ge, F.; Zhao, Y. Aescin nanoparticles prepared using SEDS: Composition stability and dissolution enhancement. J. Supercrit. Fluids 2017, 130, 267–272. [Google Scholar] [CrossRef]
- Aguiar, G.P.S.; Arcari, B.D.; Chaves, L.M.; Dal Magro, C.; Boschetto, D.L.; Piato, A.L.; Lanza, M.; Oliveira, J.V. Micronization of trans-resveratrol by supercritical fluid: Dissolution, solubility and in vitro antioxidant activity. Ind. Crop. Prod. 2018, 112, 1–5. [Google Scholar] [CrossRef]
- Kumar, S.; Dilbaghi, N.; Saharan, R.; Bhanjana, G. Nanotechnology as emerging tool for enhancing solubility of poorly water-soluble drugs. Bionanoscience 2012, 2, 227–250. [Google Scholar] [CrossRef]
- Singh, A.; Worku, Z.A.; Van den Mooter, G. Oral formulation strategies to improve solubility of poorly water-soluble drugs. Expert Opin. Drug Deliv. 2011, 8, 1361–1378. [Google Scholar] [CrossRef] [PubMed]
- Elder, D.P. Solubility: Effective Formulation Development Strategies for Poorly Soluble Active Pharmaceutical Ingredients (APIs). Am. Pharm. Rev. 2010, 13, 28. [Google Scholar]
- Kahraman, E.; Gungor, S.; Ozsoy, Y. Potential enhancement and targeting strategies of polymeric and lipid-based nanocarriers in dermal drug delivery. Ther. Deliv. 2017, 8, 967–985. [Google Scholar] [CrossRef] [PubMed]
- Elsabahy, M.; Wooley, K.L. Design of polymeric nanoparticles for biomedical delivery applications. Chem. Soc. Rev. 2012, 41, 2545–2561. [Google Scholar] [CrossRef]
- Sundar, S.; Chakravarty, J. Liposomal amphotericin B and leishmaniasis: Dose and response. J. Glob. Infect. Dis. 2010, 2, 159–166. [Google Scholar] [CrossRef]
- Shastri, D.H. Effective Delivery Routes And Strategies For Solid Lipid Nanoparticles (Sln) And Nanostructured Lipid Carriers (Nlc). Curr. Pharm. Des. 2017, 23, 6592–6601. [Google Scholar] [CrossRef]
- Poonia, M.; Ramalingam, K.; Goyal, S.; Sidhu, S.K. Nanotechnology in oral cancer: A comprehensive review. J. Oral Maxillofac. Pathol. 2017, 21, 407–414. [Google Scholar] [CrossRef]
- Patra, J.K.; Das, G.; Fraceto, L.F.; Campos, E.V.R.; Rodriguez-Torres, M.D.P.; Acosta-Torres, L.S.; Diaz-Torres, L.A.; Grillo, R.; Swamy, M.K.; Sharma, S.; et al. Nano based drug delivery systems: Recent developments and future prospects. J. Nanobiotechnol. 2018, 16, 71. [Google Scholar] [CrossRef]
- Jia, L. Nanoparticle Formulation Increases Oral Bioavailability of Poorly Soluble Drugs: Approaches Experimental Evidences and Theory. Curr. Nanosci. 2005, 1, 237–243. [Google Scholar] [CrossRef]
- Gao, L.; Liu, G.; Ma, J.; Wang, X.; Zhou, L.; Li, X.; Wang, F. Application of drug nanocrystal technologies on oral drug delivery of poorly soluble drugs. Pharm. Res. 2013, 30, 307–324. [Google Scholar] [CrossRef]
- Seedher, N.; Kanojia, M. Micellar solubilization of some poorly soluble antidiabetic drugs: A technical note. AAPS PharmSciTech 2008, 9, 431–436. [Google Scholar] [CrossRef] [PubMed]
- Vinarov, Z.; Katev, V.; Radeva, D.; Tcholakova, S.; Denkov, N.D. Micellar solubilization of poorly water-soluble drugs: Effect of surfactant and solubilizate molecular structure. Drug Dev. Ind. Pharm. 2018, 44, 677–686. [Google Scholar] [CrossRef] [PubMed]
- Junyaprasert, V.B.; Morakul, B. Nanocrystals for enhancement of oral bioavailability of poorly water-soluble drugs. Asian J. Pharm. Sci. 2015, 10, 13–23. [Google Scholar] [CrossRef]
- Zhang, L.; Wang, S.; Zhang, M.; Sun, J. Nanocarriers for oral drug delivery. J. Drug Target. 2013, 21, 515–527. [Google Scholar] [CrossRef] [PubMed]
- Zhao, L.; Feng, S.S. Enhanced oral bioavailability of paclitaxel formulated in vitamin E-TPGS emulsified nanoparticles of biodegradable polymers: In vitro and in vivo studies. J. Pharm. Sci. 2010, 99, 3552–3560. [Google Scholar] [CrossRef]
- Chen, J. Preparation of Doxorubicin Liposomes by Remote Loading Method. Methods Mol. Biol. 2023, 2622, 95–101. [Google Scholar] [CrossRef]
- Mufamadi, M.S.; Pillay, V.; Choonara, Y.E.; Du Toit, L.C.; Modi, G.; Naidoo, D.; Ndesendo, V.M. A review on composite liposomal technologies for specialized drug delivery. J. Drug Deliv. 2011, 2011, 939851. [Google Scholar] [CrossRef]
- Torchilin, P.V.; Torchilin, V.; Torchilin, V.; Weissig, V. Liposomes: A Practical Approach; Oxford University Press: Oxford, UK, 2003. [Google Scholar]
- Jin, G.-Z.; Chakraborty, A.; Lee, J.-H.; Knowles, J.C.; Kim, H.-W. Targeting with nanoparticles for the therapeutic treatment of brain diseases. J. Tissue Eng. 2020, 11, 2041731419897460. [Google Scholar] [CrossRef]
- Lee, M.K. Liposomes for Enhanced Bioavailability of Water-Insoluble Drugs: In Vivo Evidence and Recent Approaches. Pharmaceutics 2020, 12, 264. [Google Scholar] [CrossRef]
- Chaudhary, S.; Garg, T.; Murthy, R.S.; Rath, G.; Goyal, A.K. Recent approaches of lipid-based delivery system for lymphatic targeting via oral route. J. Drug Target. 2014, 22, 871–882. [Google Scholar] [CrossRef]
- Rao, M.R.; Babrekar, L.S. Liposomal Drug Delivery for Solubility and Bioavailability Enhancement of Efavirenz. J. Pharm. Sci. 2018, 80, 1115–1124. [Google Scholar]
- Telange, D.R.; Patil, A.T.; Pethe, A.M.; Fegade, H.; Anand, S.; Dave, V.S. Formulation and characterization of an apigenin-phospholipid phytosome (APLC) for improved solubility, in vivo bioavailability, and antioxidant potential. Eur. J. Pharm. Sci. 2017, 108, 36–49. [Google Scholar] [CrossRef] [PubMed]
- Choudhary, S.; Gupta, L.; Rani, S.; Dave, K.; Gupta, U. Impact of Dendrimers on Solubility of Hydrophobic Drug Molecules. Front. Pharmacol. 2017, 8, 261. [Google Scholar] [CrossRef] [PubMed]
- D’Emanuele, A.; Attwood, D. Dendrimer–drug interactions. Adv. Drug Deliv. Rev. 2005, 57, 2147–2162. [Google Scholar] [CrossRef] [PubMed]
- Mignani, S.; El Kazzouli, S.; Bousmina, M.; Majoral, J.P. Expand classical drug administration ways by emerging routes using dendrimer drug delivery systems: A concise overview. Adv. Drug Deliv. Rev. 2013, 65, 1316–1330. [Google Scholar] [CrossRef] [PubMed]
- Yellepeddi, V.K.; Ghandehari, H. Poly(amido amine) dendrimers in oral delivery. Tissue Barriers 2016, 4, e1173773. [Google Scholar] [CrossRef] [PubMed]
- Patel, J.; Garala, K.; Basu, B.; Raval, M.; Dharamsi, A. Solubility of aceclofenac in polyamidoamine dendrimer solutions. Int. J. Pharm. Investig. 2011, 1, 135–138. [Google Scholar] [CrossRef]
- Gautam, S.P.; Verma, A. PAMAM dendrimers: Novel polymeric nanoarchitectures for solubility enhancement of candesartan cilexetil. J. Pharm. Sci. 2012, 1, 1–4. [Google Scholar]
- Kulhari, H.; Pooja, D.; Prajapati, S.K.; Chauhan, A.S. Performance evaluation of PAMAM dendrimer based simvastatin formulations. Int. J. Pharm. 2011, 405, 203–209. [Google Scholar] [CrossRef]
- Jacob, S.; Nair, A.B.; Shah, J. Emerging role of nanosuspensions in drug delivery systems. Biomater. Res. 2020, 24, 3. [Google Scholar] [CrossRef]
- Bhakay, A.; Rahman, M.; Dave, R.N.; Bilgili, E. Bioavailability Enhancement of Poorly Water-Soluble Drugs via Nanocomposites: Formulation(-)Processing Aspects and Challenges. Pharmaceutics 2018, 10, 86. [Google Scholar] [CrossRef] [PubMed]
- Saddam, H.; Abdul Baquee, A.; Jiban, D. Nanosuspension: A promising drug delivery system for poorly water soluble drug and enhanced bioavailability. Int. J. Pharm. Sci. Res. 2020, 10, 4822–4832. [Google Scholar]
- Aghrbi, I.; Fülöp, V.; Jakab, G.; Kállai-Szabó, N.; Balogh, E.; Antal, I. Nanosuspension with improved saturated solubility and dissolution rate of cilostazol and effect of solidification on stability. J. Drug Deliv. Sci. Technol. 2021, 61, 102165. [Google Scholar] [CrossRef]
- Rao, M.R.P.; Godbole, R.V.; Borate, S.G.; Mahajan, S.; Gangwal, T. Nanosuspension coated multiparticulates for controlled delivery of albendazole. Drug Dev. Ind. Pharm. 2021, 47, 367–376. [Google Scholar] [CrossRef]
- Hanafy, N.A.N.; El-Kemary, M.; Leporatti, S. Micelles Structure Development as a Strategy to Improve Smart Cancer Therapy. Cancers 2018, 10, 238. [Google Scholar] [CrossRef]
- Xu, W.; Ling, P.; Zhang, T. Polymeric micelles, a promising drug delivery system to enhance bioavailability of poorly water-soluble drugs. J. Drug Deliv. 2013, 2013, 340315. [Google Scholar] [CrossRef]
- Jhaveri, A.M.; Torchilin, V.P. Multifunctional polymeric micelles for delivery of drugs and siRNA. Front. Pharmacol. 2014, 5, 77. [Google Scholar] [CrossRef]
- Aliabadi, H.M.; Lavasanifar, A. Polymeric micelles for drug delivery. Front. Cell Dev. Biol. 2006, 3, 139–162. [Google Scholar] [CrossRef]
- Bansal, K.K.; Ali, A.A.; Rahman, M.; Sjöholm, E.; Wilén, C.-E.; Rosenholm, J.M. Evaluation of solubilizing potential of functional poly (jasmine lactone) micelles for hydrophobic drugs: A comparison with commercially available polymers. Int. J. Polym. Mater. Polym. Biomater. 2022, 1–9. [Google Scholar] [CrossRef]
- Ali, A.; Bhadane, R.; Asl, A.A.; Wilén, C.-E.; Salo-Ahen, O.; Rosenholm, J.M.; Bansal, K.K. Functional block copolymer micelles based on poly (jasmine lactone) for improving the loading efficiency of weakly basic drugs. RSC Adv. 2022, 12, 26763–26775. [Google Scholar] [CrossRef]
- Zhou, Z.; Forbes, R.T.; D’Emanuele, A. Preparation of core-crosslinked linear-dendritic copolymer micelles with enhanced stability and their application for drug solubilisation. Int. J. Pharm. 2017, 523, 260–269. [Google Scholar] [CrossRef]
- Naseri, N.; Valizadeh, H.; Zakeri-Milani, P. Solid lipid nanoparticles and nanostructured lipid carriers: Structure, preparation and application. Adv. Pharm. Bull. 2015, 5, 305–313. [Google Scholar] [CrossRef] [PubMed]
- Das, S.; Chaudhury, A. Recent advances in lipid nanoparticle formulations with solid matrix for oral drug delivery. AAPS PharmSciTech 2011, 12, 62–76. [Google Scholar] [CrossRef] [PubMed]
- Jaiswal, P.; Gidwani, B.; Vyas, A. Nanostructured lipid carriers and their current application in targeted drug delivery. Artif. Cells Nanomed. Biotechnol. 2016, 44, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Hu, L.; Tang, X.; Cui, F. Solid lipid nanoparticles (SLNs) to improve oral bioavailability of poorly soluble drugs. J. Pharm. Pharmacol. 2004, 56, 1527–1535. [Google Scholar] [CrossRef] [PubMed]
- Khan, S.; Shaharyar, M.; Fazil, M.; Hassan, M.Q.; Baboota, S.; Ali, J. Tacrolimus-loaded nanostructured lipid carriers for oral delivery-in vivo bioavailability enhancement. Eur. J. Pharm. Biopharm. 2016, 109, 149–157. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.; Puna, J.; Gomes, J.J.E. A Brief Review of the Supercritical Antisolvent (SAS) Technique for the Preparation of Nanocatalysts to Be Used in Biodiesel Production. Energies 2022, 15, 9355. [Google Scholar] [CrossRef]
- Abuzar, S.M.; Hyun, S.M.; Kim, J.H.; Park, H.J.; Kim, M.S.; Park, J.S.; Hwang, S.J. Enhancing the solubility and bioavailability of poorly water-soluble drugs using supercritical antisolvent (SAS) process. Int. J. Pharm. 2018, 538, 1–13. [Google Scholar] [CrossRef]
- Sinha, B.; Müller, R.H.; Möschwitzer, J.P. Bottom-up approaches for preparing drug nanocrystals: Formulations and factors affecting particle size. Int. J. Pharm. 2013, 453, 126–141. [Google Scholar] [CrossRef] [PubMed]
- Franco, P.; De Marco, I. Supercritical antisolvent process for pharmaceutical applications: A review. J. Process. 2020, 8, 938. [Google Scholar] [CrossRef]
- Park, J.; Cho, W.; Cha, K.H.; Ahn, J.; Han, K.; Hwang, S.J. Solubilization of the poorly water soluble drug, telmisartan, using supercritical anti-solvent (SAS) process. Int. J. Pharm. 2013, 441, 50–55. [Google Scholar] [CrossRef] [PubMed]
- Park, H.J.; Kim, M.S.; Lee, S.; Kim, J.S.; Woo, J.S.; Park, J.S.; Hwang, S.J. Recrystallization of fluconazole using the supercritical antisolvent (SAS) process. Int. J. Pharm. 2007, 328, 152–160. [Google Scholar] [CrossRef] [PubMed]
- Tayeb, H.H.; Felimban, R.; Almaghrabi, S.; Hasaballah, N. Nanoemulsions: Formulation, characterization, biological fate, and potential role against COVID-19 and other viral outbreaks. Colloid Interface Sci. Commun. 2021, 45, 100533. [Google Scholar] [CrossRef] [PubMed]
- Pandey, P.; Gulati, N.; Makhija, M.; Purohit, D.; Dureja, H. Nanoemulsion: A novel drug delivery approach for enhancement of bioavailability. Recent Patents Nanotechnol. 2020, 14, 276–293. [Google Scholar] [CrossRef] [PubMed]
- Kanke, P.K.; Pathan, I.B.; Jadhav, A.; Usman, M.R.M. Formulation and evaluation of febuxostat nanoemulsion for transdermal drug delivery. J. Pharm. BioSci. 2019, 7, 1–7. [Google Scholar]
- Wik, J.; Bansal, K.K.; Assmuth, T.; Rosling, A.; Rosenholm, J.M. Facile methodology of nanoemulsion preparation using oily polymer for the delivery of poorly soluble drugs. Drug Deliv. Transl. Res. 2020, 10, 1228–1240. [Google Scholar] [CrossRef]
- Pyrhönen, J.; Bansal, K.K.; Bhadane, R.; Wilén, C.-E.; Salo-Ahen, O.M.; Rosenholm, J.M. Molecular Dynamics Prediction Verified by Experimental Evaluation of the Solubility of Different Drugs in Poly (decalactone) for the Fabrication of Polymeric Nanoemulsions. Adv. NanoBiomed Res. 2022, 2, 2100072. [Google Scholar] [CrossRef]
- Kendre, P.; Satav, T.J.P.B. Current trends and concepts in the design and development of nanogel carrier systems. Polym. Bull. 2019, 76, 1595–1617. [Google Scholar] [CrossRef]
- Zhang, Y.; Andrén, O.C.; Nordström, R.; Fan, Y.; Malmsten, M.; Mongkhontreerat, S.; Malkoch, M. Off-Stoichiometric Thiol-Ene Chemistry to Dendritic Nanogel Therapeutics. Adv. Funct. Mater. 2019, 29, 1806693. [Google Scholar] [CrossRef]
- Sharma, A.; Garg, T.; Aman, A.; Panchal, K.; Sharma, R.; Kumar, S.; Markandeywar, T. Nanogel—An advanced drug delivery tool: Current and future. Artif. Cells Nanomed. Biotechnol. 2016, 44, 165–177. [Google Scholar] [CrossRef]
- Kaewruethai, T.; Laomeephol, C.; Pan, Y.; Luckanagul, J.A. Multifunctional Polymeric Nanogels for Biomedical Applications. Gels 2021, 7, 228. [Google Scholar] [CrossRef] [PubMed]
- Soni, G.; Yadav, K.S. Nanogels as potential nanomedicine carrier for treatment of cancer: A mini review of the state of the art. Saudi Pharm. J. 2016, 24, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Yao, Y.; Xia, M.; Wang, H.; Li, G.; Shen, H.; Ji, G.; Meng, Q.; Xie, Y. Preparation and evaluation of chitosan-based nanogels/gels for oral delivery of myricetin. Eur. J. Pharm. Sci. 2016, 91, 144–153. [Google Scholar] [CrossRef] [PubMed]
- Khan, K.U.; Akhtar, N.; Minhas, M.U. Poloxamer-407-Co-Poly (2-Acrylamido-2-Methylpropane Sulfonic Acid) Cross-linked Nanogels for Solubility Enhancement of Olanzapine: Synthesis, Characterization, and Toxicity Evaluation. AAPS PharmSciTech 2020, 21, 141. [Google Scholar] [CrossRef]
- He, S.; Wu, L.; Li, X.; Sun, H.; Xiong, T.; Liu, J.; Huang, C.; Xu, H.; Sun, H.; Chen, W.; et al. Metal-organic frameworks for advanced drug delivery. Acta Pharm. Sin. B 2021, 11, 2362–2395. [Google Scholar] [CrossRef]
- Lawson, S.; Newport, K.; Pederniera, N.; Rownaghi, A.A.; Rezaei, F. Curcumin Delivery on Metal-Organic Frameworks: The Effect of the Metal Center on Pharmacokinetics within the M-MOF-74 Family. ACS Appl. Bio Mater. 2021, 4, 3423–3432. [Google Scholar] [CrossRef]
- Zhou, H.C.; Long, J.R.; Yaghi, O.M. Introduction to metal-organic frameworks. Chem. Rev. 2012, 112, 673–674. [Google Scholar] [CrossRef]
- Wang, Z.; Ma, Y.; Jiang, Y.; Zhou, F.; Wu, Y.; Jiang, H.; Wang, R.; Xu, Q.; Hua, C. Encapsulating quercetin in cyclodextrin metal-organic frameworks improved its solubility and bioavailability. J. Sci. Food Agric. 2022, 102, 3887–3896. [Google Scholar] [CrossRef]
- Chen, X.; Guo, T.; Zhang, K.; Chen, J.; Wang, C.; Ren, X.; Wang, Q.; Yang, Y.; Liu, C.; Tan, W.; et al. Simultaneous improvement to solubility and bioavailability of active natural compound isosteviol using cyclodextrin metal-organic frameworks. Acta Pharm. Sin. B 2021, 11, 2914–2923. [Google Scholar] [CrossRef]
- He, Y.; Zhang, W.; Guo, T.; Zhang, G.; Qin, W.; Zhang, L.; Wang, C.; Zhu, W.; Yang, M.; Hu, X.; et al. Drug nanoclusters formed in confined nano-cages of CD-MOF: Dramatic enhancement of solubility and bioavailability of azilsartan. Acta Pharm. Sin. B 2019, 9, 97–106. [Google Scholar] [CrossRef]
- Dubey, R.; Dutta, D.; Sarkar, A.; Chattopadhyay, P. Functionalized carbon nanotubes: Synthesis, properties and applications in water purification, drug delivery, and material and biomedical sciences. Nanoscale Adv. 2021, 3, 5722–5744. [Google Scholar] [CrossRef] [PubMed]
- Mahor, A.; Singh, P.P.; Bharadwaj, P.; Sharma, N.; Yadav, S.; Rosenholm, J.M.; Bansal, K.K. Carbon-based nanomaterials for delivery of biologicals and therapeutics: A cutting-edge technology. J. Cancer 2021, 7, 19. [Google Scholar] [CrossRef]
- Gomez-Gualdron, D.A.; Burgos, J.C.; Yu, J.; Balbuena, P.B. Carbon nanotubes: Engineering biomedical applications. Prog. Mol. Biol. Transl. Sci. 2011, 104, 175–245. [Google Scholar] [CrossRef] [PubMed]
- Hasnain, M.S.; Nayak, A.K.; Hasnain, M.S.; Nayak, A.K. Applications of carbon nanotubes. In Carbon Nanotubes Targeteted Drug Delivery; Springer: Berlin/Heidelberg, Germany, 2019; pp. 33–36. [Google Scholar]
- Chen, K.; Mitra, S. Incorporation of functionalized carbon nanotubes into hydrophobic drug crystals for enhancing aqueous dissolution. Colloids Surf. B Biointerfaces 2019, 173, 386–391. [Google Scholar] [CrossRef] [PubMed]
- Zhu, W.; Huang, H.; Dong, Y.; Han, C.; Sui, X.; Jian, B. Multi-walled carbon nanotube-based systems for improving the controlled release of insoluble drug dipyridamole. Exp. Med. 2019, 17, 4610–4616. [Google Scholar] [CrossRef]
- Vialpando, M.; Martens, J.A.; Van den Mooter, G. Potential of ordered mesoporous silica for oral delivery of poorly soluble drugs. Ther. Deliv. 2011, 2, 1079–1091. [Google Scholar] [CrossRef]
- Laine, A.L.; Price, D.; Davis, J.; Roberts, D.; Hudson, R.; Back, K.; Bungay, P.; Flanagan, N. Enhanced oral delivery of celecoxib via the development of a supersaturable amorphous formulation utilising mesoporous silica and co-loaded HPMCAS. Int. J. Pharm. 2016, 512, 118–125. [Google Scholar] [CrossRef]
- Maleki, A.; Kettiger, H.; Schoubben, A.; Rosenholm, J.M.; Ambrogi, V.; Hamidi, M. Mesoporous silica materials: From physico-chemical properties to enhanced dissolution of poorly water-soluble drugs. J. Control. Release 2017, 262, 329–347. [Google Scholar] [CrossRef]
- Bremmell, K.E.; Prestidge, C.A. Enhancing oral bioavailability of poorly soluble drugs with mesoporous silica based systems: Opportunities and challenges. Drug Dev. Ind. Pharm. 2019, 45, 349–358. [Google Scholar] [CrossRef]
- Rengarajan, G.; Enke, D.; Steinhart, M.; Beiner, M. Stabilization of the amorphous state of pharmaceuticals in nanopores. J. Mater. Chem. 2008, 18, 2537–2539. [Google Scholar] [CrossRef]
- Sen Karaman, D.; Patrignani, G.; Rosqvist, E.; Smatt, J.H.; Orlowska, A.; Mustafa, R.; Preis, M.; Rosenholm, J.M. Mesoporous silica nanoparticles facilitating the dissolution of poorly soluble drugs in orodispersible films. Eur. J. Pharm. Sci. 2018, 122, 152–159. [Google Scholar] [CrossRef] [PubMed]
- Jadhav, V. Formulation and Evaluation of Mesoporous Silica Nanoparticle Loaded Fast Dissolving Tablet of Tamoxifen. Indian J. Pharm. Sci. 2021, 83, 32–38. [Google Scholar] [CrossRef]
- Ibrahim, A.H.; Rosqvist, E.; Smatt, J.H.; Ibrahim, H.M.; Ismael, H.R.; Afouna, M.I.; Samy, A.M.; Rosenholm, J.M. Formulation and optimization of lyophilized nanosuspension tablets to improve the physicochemical properties and provide immediate release of silymarin. Int. J. Pharm. 2019, 563, 217–227. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, A.H.; Smått, J.-H.; Govardhanam, N.P.; Ibrahim, H.M.; Ismael, H.R.; Afouna, M.I.; Samy, A.M.; Rosenholm, J.M. Formulation and optimization of drug-loaded mesoporous silica nanoparticle-based tablets to improve the dissolution rate of the poorly water-soluble drug silymarin. Eur. J. Pharm. Sci. 2020, 142, 105103. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.; Wang, J.; Bai, X.; Jiang, T.; Zhang, Q.; Wang, S. Mesoporous silica nanoparticles for increasing the oral bioavailability and permeation of poorly water soluble drugs. Mol. Pharm. 2012, 9, 505–513. [Google Scholar] [CrossRef]
- Bukara, K.; Schueller, L.; Rosier, J.; Martens, M.A.; Daems, T.; Verheyden, L.; Eelen, S.; Van Speybroeck, M.; Libanati, C.; Martens, J.A.; et al. Ordered mesoporous silica to enhance the bioavailability of poorly water-soluble drugs: Proof of concept in man. Eur. J. Pharm. Biopharm. 2016, 108, 220–225. [Google Scholar] [CrossRef]
- Devarakonda, B.; Hill, R.A.; de Villiers, M.M. The effect of PAMAM dendrimer generation size and surface functional group on the aqueous solubility of nifedipine. Int. J. Pharm. 2004, 284, 133–140. [Google Scholar] [CrossRef]
- Nazlı, H.; Gedik, G. In-vitro evaluation of dendrimeric formulation of oxaliplatin. Pharm. Dev. Technol. 2021, 26, 750–764. [Google Scholar] [CrossRef]
- Sharma, A.K.; Gupta, L.; Sahu, H.; Qayum, A.; Singh, S.K.; Nakhate, K.T.; Ajazuddin; Gupta, U. Chitosan Engineered PAMAM Dendrimers as Nanoconstructs for the Enhanced Anti-Cancer Potential and Improved In vivo Brain Pharmacokinetics of Temozolomide. Pharm. Res. 2018, 35, 9. [Google Scholar] [CrossRef]
- Gorzkiewicz, M.; Janaszewska, A.; Ficker, M.; Svenningsen, S.W.; Christensen, J.B.; Klajnert-Maculewicz, B. Pyrrolidone-modified PAMAM dendrimers enhance anti-inflammatory potential of indomethacin in vitro. Colloids Surf. B Biointerfaces 2019, 181, 959–962. [Google Scholar] [CrossRef]
- Yiyun, C.; Tongwen, X. Solubility of nicotinic acid in polyamidoamine dendrimer solutions. Eur. J. Med. Chem. 2005, 40, 1384–1389. [Google Scholar] [CrossRef] [PubMed]
- Bhadra, D.; Bhadra, S.; Jain, N.K. PEGylated peptide-based dendritic nanoparticulate systems for delivery of artemether. J. Drug Deliv. Sci. Technol. 2005, 15, 65–73. [Google Scholar] [CrossRef]
- Milhem, O.; Myles, C.; McKeown, N.; Attwood, D.; D’Emanuele, A. Polyamidoamine Starburst® dendrimers as solubility enhancers. Int. J. Pharm. 2000, 197, 239–241. [Google Scholar] [CrossRef]
- D’Emanuele, A.; Jevprasesphant, R.; Penny, J.; Attwood, D. The use of a dendrimer-propranolol prodrug to bypass efflux transporters and enhance oral bioavailability. J. Control. Release Off. J. Control. Release Soc. 2004, 95, 447–453. [Google Scholar] [CrossRef]
- Li, C.; Zhang, Y.; Su, T.; Feng, L.; Long, Y.; Chen, Z. Silica-coated flexible liposomes as a nanohybrid delivery system for enhanced oral bioavailability of curcumin. Int. J. Nanomed. 2012, 7, 5995–6002. [Google Scholar] [CrossRef] [PubMed]
- Zhao, M.; Lee, S.H.; Song, J.G.; Kim, H.Y.; Han, H.K. Enhanced oral absorption of sorafenib via the layer-by-layer deposition of a pH-sensitive polymer and glycol chitosan on the liposome. Int. J. Pharm. 2018, 544, 14–20. [Google Scholar] [CrossRef]
- Arafat, M.; Kirchhoefer, C.; Mikov, M.; Sarfraz, M.; Löbenberg, R. Nanosized Liposomes Containing Bile Salt: A Vesicular Nanocarrier for Enhancing Oral Bioavailability of BCS Class III Drug. J. Pharm. Pharm. Sci. A Publ. Can. Soc. Pharm. Sci. Soc. Can. Des Sci. Pharm. 2017, 20, 305–318. [Google Scholar] [CrossRef]
- Zhu, Y.; Wang, M.; Zhang, J.; Peng, W.; Firempong, C.K.; Deng, W.; Wang, Q.; Wang, S.; Shi, F.; Yu, J.; et al. Improved oral bioavailability of capsaicin via liposomal nanoformulation: Preparation, in vitro drug release and pharmacokinetics in rats. Arch. Pharmacal Res. 2015, 38, 512–521. [Google Scholar] [CrossRef]
- Patel, G.M.; Shelat, P.K.; Lalwani, A.N. QbD based development of proliposome of lopinavir for improved oral bioavailability. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2017, 108, 50–61. [Google Scholar] [CrossRef]
- Managuli, R.S.; Wang, J.T.; Faruqu, F.M.; Pandey, A.; Jain, S.; Al-Jamal, K.T.; Mutalik, S. Surface engineered nanoliposomal platform for selective lymphatic uptake of asenapine maleate: In vitro and in vivo studies. Mater. Sci. Eng. C Mater. Biol. Appl. 2020, 109, 110620. [Google Scholar] [CrossRef]
- Ji, W.J.; Ma, Y.Q.; Zhang, X.; Zhang, L.; Zhang, Y.D.; Su, C.C.; Xiang, G.A.; Zhang, M.P.; Lin, Z.C.; Wei, L.Q.; et al. Inflammatory monocyte/macrophage modulation by liposome-entrapped spironolactone ameliorates acute lung injury in mice. Nanomedicine 2016, 11, 1393–1406. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.; Verma, A.; Teja, B.V.; Shukla, P.; Mishra, P.R. Development of stabilized Paclitaxel nanocrystals: In-vitro and in-vivo efficacy studies. Eur. J. Pharm. Sci. Off. J. Eur. Fed. Pharm. Sci. 2015, 69, 51–60. [Google Scholar] [CrossRef] [PubMed]
- Xiong, S.; Liu, W.; Li, D.; Chen, X.; Liu, F.; Yuan, D.; Pan, H.; Wang, Q.; Fang, S.; Chen, T. Oral Delivery of Puerarin Nanocrystals To Improve Brain Accumulation and Anti-Parkinsonian Efficacy. Mol. Pharm. 2019, 16, 1444–1455. [Google Scholar] [CrossRef] [PubMed]
- Hou, Y.; Shao, J.; Fu, Q.; Li, J.; Sun, J.; He, Z. Spray-dried nanocrystals for a highly hydrophobic drug: Increased drug loading, enhanced redispersity, and improved oral bioavailability. Int. J. Pharm. 2017, 516, 372–379. [Google Scholar] [CrossRef] [PubMed]
- Ndlovu, S.T.; Ullah, N.; Khan, S.; Ramharack, P.; Soliman, M.; de Matas, M.; Shahid, M.; Sohail, M.; Imran, M.; Shah, S.W.A.; et al. Domperidone nanocrystals with boosted oral bioavailability: Fabrication, evaluation and molecular insight into the polymer-domperidone nanocrystal interaction. Drug Deliv. Transl. Res. 2019, 9, 284–297. [Google Scholar] [CrossRef]
- Mishra, B.; Sahoo, J.; Dixit, P.K. Enhanced bioavailability of cinnarizine nanosuspensions by particle size engineering: Optimization and physicochemical investigations. Mater. Sci. Eng. C Mater. Biol. Appl. 2016, 63, 62–69. [Google Scholar] [CrossRef] [PubMed]
- Yang, W.; Wang, Y.; Fu, Q.; Guo, Z.; Sun, B.; Liu, W.; Liu, Y.; Mu, S.; Guo, M.; Li, J.; et al. The role of particle size of glyburide crystals in improving its oral absorption. Drug Deliv. Transl. Res. 2017, 7, 428–438. [Google Scholar] [CrossRef]
- Al-Heibshy, F.N.S.; Başaran, E.; Arslan, R.; Öztürk, N.; Erol, K.; Demirel, M. Physicochemical characterization and pharmacokinetic evaluation of rosuvastatin calcium incorporated solid lipid nanoparticles. Int. J. Pharm. 2020, 578, 119106. [Google Scholar] [CrossRef]
- Singh, H.; Jindal, S.; Singh, M.; Sharma, G.; Kaur, I.P. Nano-formulation of rifampicin with enhanced bioavailability: Development, characterization and in-vivo safety. Int. J. Pharm. 2015, 485, 138–151. [Google Scholar] [CrossRef]
- Ramalingam, P.; Ko, Y.T. Improved oral delivery of resveratrol from N-trimethyl chitosan-g-palmitic acid surface-modified solid lipid nanoparticles. Colloids Surf. B Biointerfaces 2016, 139, 52–61. [Google Scholar] [CrossRef]
- Fagerholm, U.; Hellberg, S.; Spjuth, O. Advances in Predictions of Oral Bioavailability of Candidate Drugs in Man with New Machine Learning Methodology. Molecules 2021, 26, 2572. [Google Scholar] [CrossRef] [PubMed]
- Cabrera-Perez, M.A.; Pham-The, H. Computational modeling of human oral bioavailability: What will be next? Expert Opin. Drug Discov. 2018, 13, 509–521. [Google Scholar] [CrossRef] [PubMed]
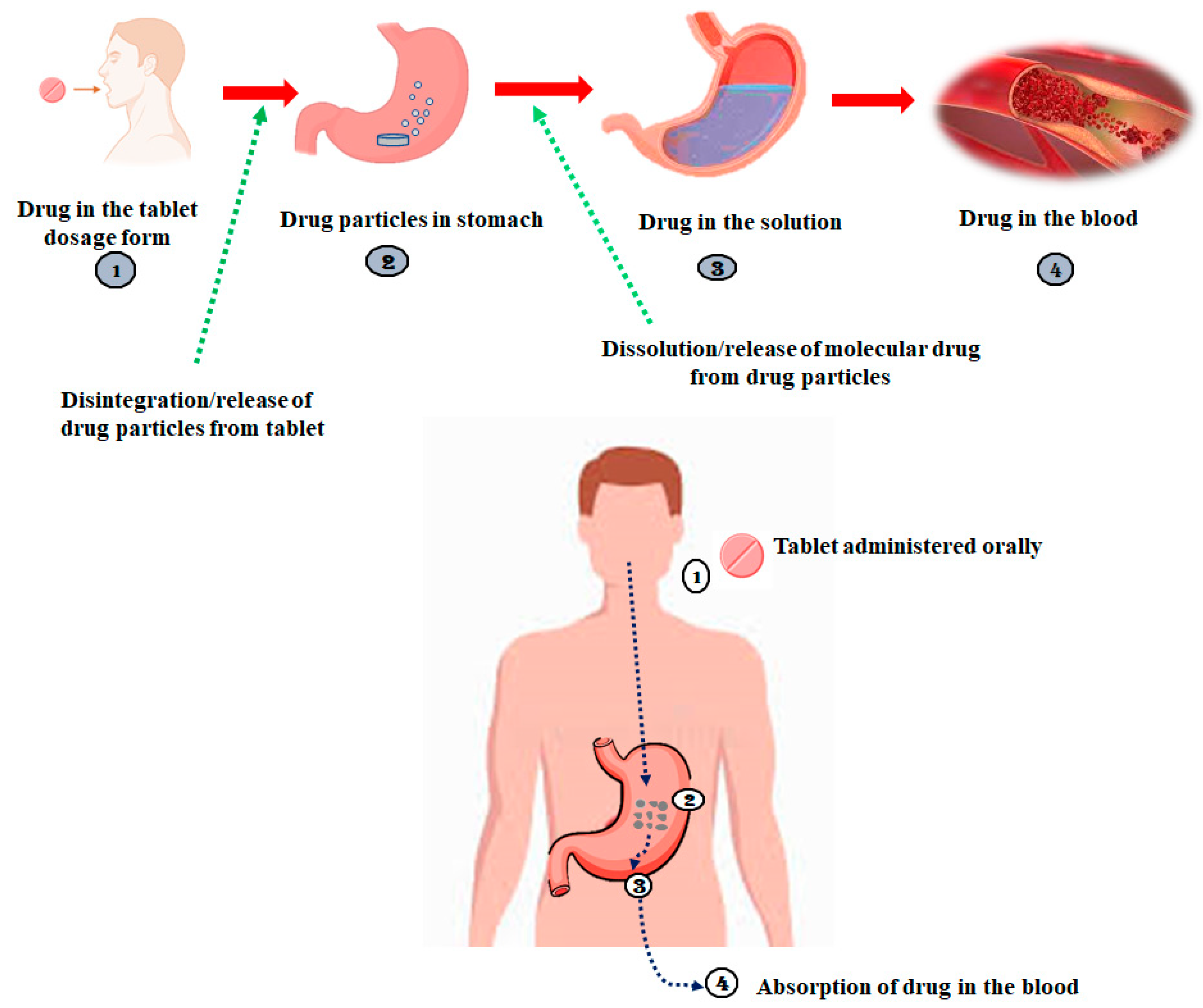
| BCS Class | Solubility | Permeability | Drug Molecule Examples |
|---|---|---|---|
| I | High | High | Mefoquine hydrochloride, Nelfnavir mesylate, Quinine sulfate, Clomiphene citrate |
| II | Low | High | Ibuprofen, Nifedipine, Carbamazepine, Diazepam, Efavirenz |
| III | High | Low | Amiloride hydrochloride, Amoxicillin, Ethosuximide, Fluconazole, Isoniazid, Salbutamol |
| IV | Low | Low | Acetazolamide, Dapsone, Doxycycline, Nalidixic acid, Theophylline |
| S. No | Factors Affecting Solubility | Details | References |
|---|---|---|---|
| 1. | Particle size | As particle size is reduced, the surface area will increase, and the larger surface area will provide a greater interaction of the solute molecules with the solvent. | [34] |
| 2. | Temperature | Solubility will be increased when the temperature rises and the solution process absorbs energy; if the solution process generates energy, then solubility will decrease with an increase in temperature. | [35] |
| 3. | Pressure | Pressure will only affect the solubility of gaseous solutes and have no effect on solid and liquid solutes. A decrease in pressure causes a decrease in solubility, and an increase in pressure causes an increase in the solubility of gaseous solutes. | [35] |
| 4. | Nature of solute and solvent | Properties of solute, as well as the solvent, have drastic effects on solubility. | [36] |
| 5. | Polarity | Substances with the same type of polarity will be soluble in one another, “similia similibus solvuntur”. Polar solute molecules or ions will dissolve in polar solvents, while non-polar solute molecules will dissolve in non-polar solvents. | [37] |
| 6. | Polymorphism | Polymorphs differ in melting points. Different polymorphs have different solubilities as solubility and melting point are linked. | [38] |
| 7. | Stirring | Stirring ensures that new solvent components come into contact with the solid and liquid solutes, resulting in increasing solubility. | [39] |
| S. No | Trade Name | Therapeutic Agent | Manufacturer | Polymer Used in Formulation | Indication |
|---|---|---|---|---|---|
| 1. | Certican | Everolimus | Novartis | HPMC | Anti-cancer |
| 2. | Cesamet | Nabilone | Valeant Pharmaceuticals | PVP | Chemotherapy-induced nausea |
| 3. | Gris-PEG | Griseofulvin | Pedinol Pharmacal Inc. | PEG6000 | Antifungal |
| 4. | Intelence | Etravirin | Tibotec | HPMC | Antiviral (HIV infection) |
| 5. | Isoptin SR-E | Verapamil | Abbott | HPMC/HPC | Anti-Hypertensive |
| 6. | Nivadil | Nivalidipine | Fujisawa Pharmaceutical Co., Ltd. | HPMC | Anti-Hypertensive |
| 7. | Prograf | Tacrolimus | Fujisawa Pharmaceutical Co., Ltd. | HPMC | Immunosuppressant |
| 8. | Rezulin | Troglitazone | Pfizer, Inc. | PVP | Antihyperglycemic |
| 9. | Sporanox | Itraconazole | Jansen Pharmaceuticals, Inc. | HPMC | Antifungal |
| S. No | Techniques | Advantages | Disadvantages | References |
|---|---|---|---|---|
| 1. | Particle Size Reduction | Increases surface area volume ratio | Due to the high surface charge on discrete small particles, there is a strong tendency for particle agglomeration. Thermal stress may occur, which harms thermosensitive or unstable active compound. | [74] |
| 2. | Cyclodextrin Inclusion Complex | Cyclodextrin has high aqueous solubility and commensurately low viscosity. High API concentrations are achievable. Additionally, facilitates chemical stability. | Cyclodextrins demonstrates renal toxicity in most species, limiting their use in pre-clinical toxicology assessments. | [75,76] |
| 3. | Solid Dispersion | Dissolution rate and bioavailability are enhanced by keeping drug in more soluble amorphous state. | Not commonly used as a commercial product because of the conversion of the amorphous drug into the less soluble crystalline form on long-term storage and, consequently, increased drug mobility can lead to phase separation and instability. Large-scale production is limited due to expensive preparation methods. | [75] |
| 4. | Prodrug approach | Higher solubility in lipid membranes and improved oral or local absorption. Reduced toxicity and local irritation. Increases chemical or metabolic stability. | Not feasible for all drug formulation. | [74] |
| 5. | Supercritical fluid technology | Free from organic solvents and heavy metals. Green extraction techniques. | Expensive and complex equipment, operating at elevated pressure. High power consumption. | [68] |
| 6. | Polymeric Micelles | Ease of fabrication and chemical modification. Suitable for numerous hydrophobic drug candidates. Control and targeted drug release is possible. | The disintegration of micelles due to their dilution after oral administration, in vivo instability below the critical micellar concentration. Low drug loading. | [77] |
| 7. | Polymeric Nanoparticles | Enhanced drug stability, sustained drug delivery, shielding of the drug cargo from enzymatic activity, prolonged retention in the GI tract, and improved mucoadhesiveness. | Challenges in biocompatibility and safety of polymeric carriers. Toxicity is a result of the high tissue accumulation of non-biodegradable NPs. Difficulties in optimizing the process parameters and scaling up the production into a pharmaceutical product. | [78] |
| 8. | Liposomes | Non-immunogenic, biocompatible, and biodegradable. Ability to carry both hydrophilic as well as hydrophobic drugs. | Poor stability and short shelf life. | [79] |
| 9. | Solid lipid nanoparticles (SLNs) | Biocompatible. Easy scale-up. Protects drug against harsh environmental conditions. | Because of crystalline structure, low drug-loading efficacy and chance of drug expulsion during storage. | [80] |
| 10. | Dendrimers | Drug encapsulation and conjugation is possible. Tunable chemical and physical properties. | May cause cellular toxicity. Elimination and metabolism depending on the generation of the dendrimers. High synthetic cost. | [77] |
| 11. | Quantum dots | Multiple molecular targets simultaneously. | Toxicity effect of metal core. | [81] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Kumari, L.; Choudhari, Y.; Patel, P.; Gupta, G.D.; Singh, D.; Rosenholm, J.M.; Bansal, K.K.; Kurmi, B.D. Advancement in Solubilization Approaches: A Step towards Bioavailability Enhancement of Poorly Soluble Drugs. Life 2023, 13, 1099. https://doi.org/10.3390/life13051099
Kumari L, Choudhari Y, Patel P, Gupta GD, Singh D, Rosenholm JM, Bansal KK, Kurmi BD. Advancement in Solubilization Approaches: A Step towards Bioavailability Enhancement of Poorly Soluble Drugs. Life. 2023; 13(5):1099. https://doi.org/10.3390/life13051099
Chicago/Turabian StyleKumari, Lakshmi, Yash Choudhari, Preeti Patel, Ghanshyam Das Gupta, Dilpreet Singh, Jessica M. Rosenholm, Kuldeep Kumar Bansal, and Balak Das Kurmi. 2023. "Advancement in Solubilization Approaches: A Step towards Bioavailability Enhancement of Poorly Soluble Drugs" Life 13, no. 5: 1099. https://doi.org/10.3390/life13051099
APA StyleKumari, L., Choudhari, Y., Patel, P., Gupta, G. D., Singh, D., Rosenholm, J. M., Bansal, K. K., & Kurmi, B. D. (2023). Advancement in Solubilization Approaches: A Step towards Bioavailability Enhancement of Poorly Soluble Drugs. Life, 13(5), 1099. https://doi.org/10.3390/life13051099








