An Overview of E-Cigarette Impact on Reproductive Health
Abstract
1. Introduction
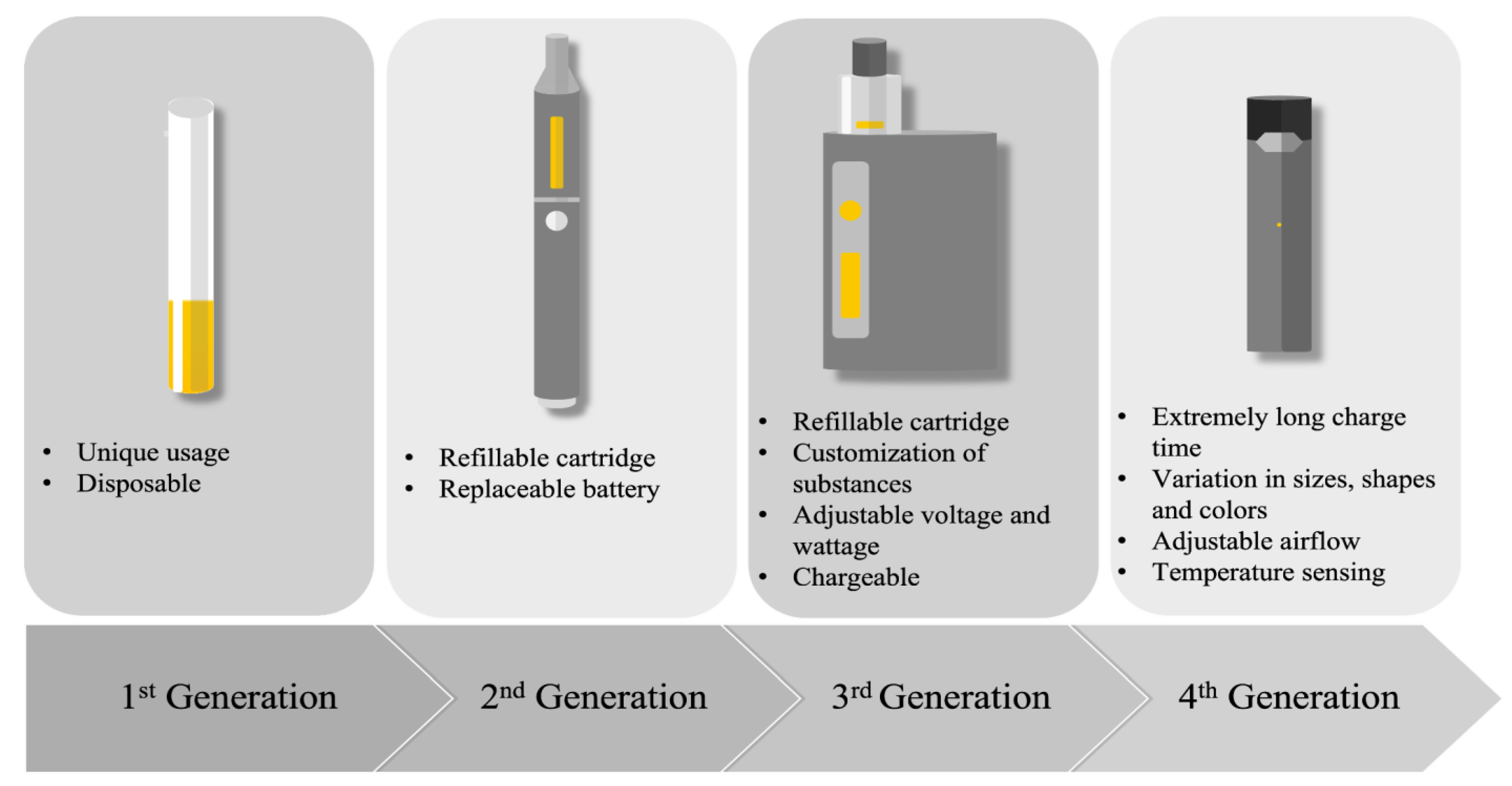
2. E-Cigarettes: Types, Usage
3. Composition of E-Cigarettes
3.1. Glycols
3.2. Nicotine
3.3. Particles
3.4. Metals
3.5. Tobacco-Specific Nitrosamines (TSNAs)
3.6. Carbonyls
3.7. Volatile Organic Compounds (VOCs)
3.8. Hydrocarbons and Polycyclic Aromatic Hydrocarbons (PAHs)
3.9. Phenols
4. E-Cigarette and Conventional Cigarette: Compositional Comparisons
5. Conventional Cigarette Smoking and Reproduction
6. Reprotoxicological Profile of E-Cigarette Components
6.1. Nicotine
6.2. Flavouring Compounds
6.3. Heavy Metals
6.4. E-Cigarette Vapour
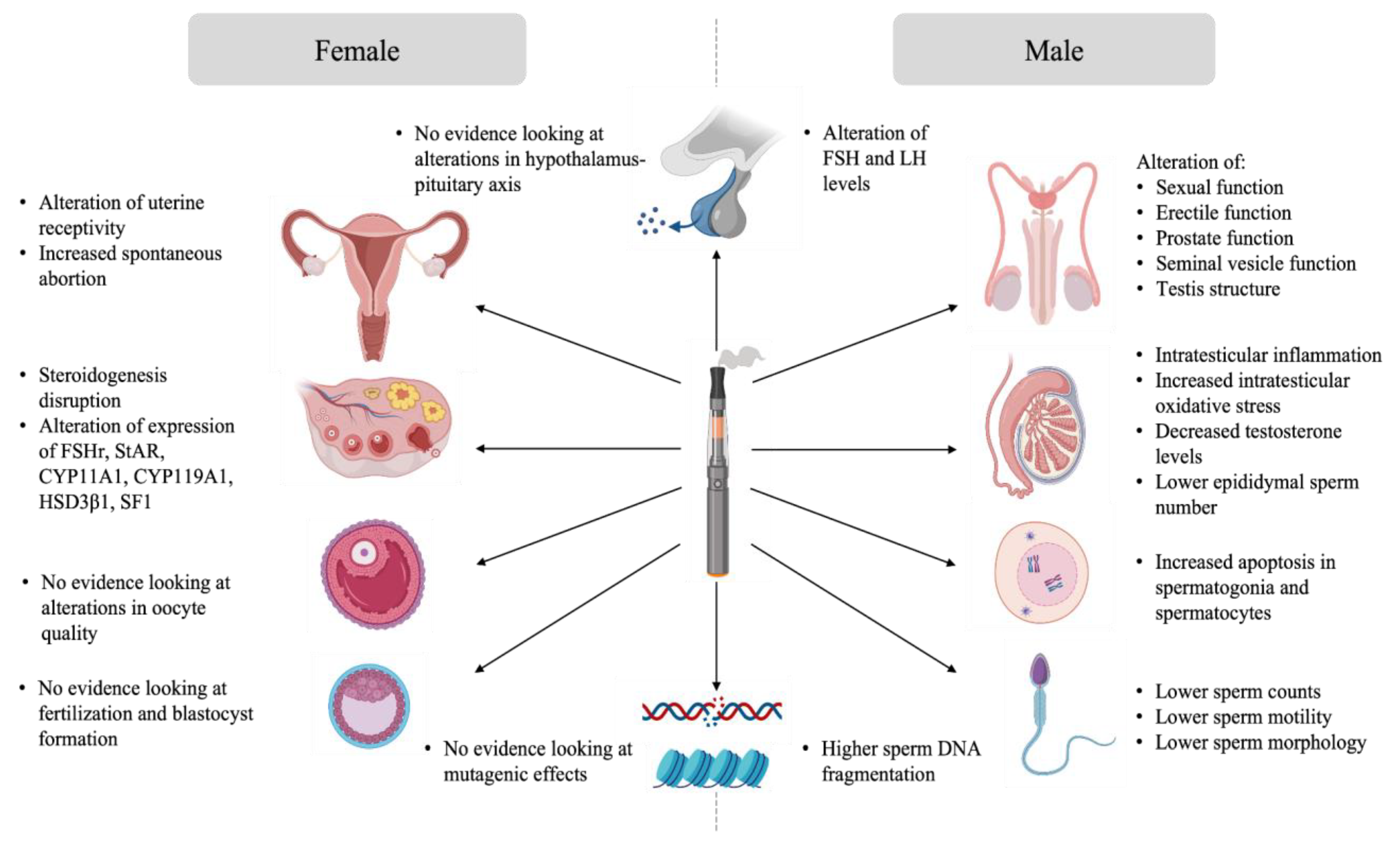
7. Evidence of the Impact of E-Cigarette Exposure on Reproduction
7.1. Evidence Analysis of the Impact of E-Cigarette on Male Reproduction
7.2. Evidence Analysis of the Impact of E-Cigarette on Female Reproduction
7.3. Evidence Analysis of the Impact of E-Cigarette on ART Outcomes
8. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- World Health Organization (WHO). International Classification of Diseases, 11th Revision (ICD-11); WHO: Geneva, Switzerland, 2018. [Google Scholar]
- Mascarenhas, M.N.; Flaxman, S.R.; Boerma, T.; Vanderpoel, S.; Stevens, G.A. National, regional, and global trends in infertility prevalence since 1990: A systematic analysis of 277 health surveys. PLoS Med. 2012, 9, e1001356. [Google Scholar] [CrossRef] [PubMed]
- Boivin, J.; Bunting, L.; Collins, J.A.; Nygren, K.G. International estimates of infertility prevalence and treatment-seeking: Potential need and demand for infertility medical care. Hum. Reprod. 2007, 22, 1506–1512. [Google Scholar] [CrossRef]
- Rutstein, S.O.; Shah, I.H. Infecundity Infertility and Childlessness in Developing Countries; World Health Organization: Geneva, Switzerland, 2004. [Google Scholar]
- Gore, A.C.; Chappell, V.A.; Fenton, S.E.; Flaws, J.A.; Nadal, A.; Prins, G.S.; Toppari, J.; Zoeller, R.T. EDC-2: The Endocrine Society’s Second Scientific Statement on Endocrine-Disrupting Chemicals. Endocr. Rev. 2015, 36, E1–E150. [Google Scholar] [CrossRef]
- Segal, T.R.; Giudice, L.C. Before the beginning: Environmental exposures and reproductive and obstetrical outcomes. Fertil. Steril. 2019, 112, 613–621. [Google Scholar] [CrossRef] [PubMed]
- Chen, R.; Pierce, J.P.; Leas, E.C.; Benmarhnia, T.; Strong, D.R.; White, M.M.; Stone, M.; Trinidad, D.R.; McMenamin, S.B.; Messer, K. Effectiveness of e-cigarettes as aids for smoking cessation: Evidence from the PATH Study cohort, 2017–2019. Tob. Control 2022. [Google Scholar] [CrossRef]
- Hutzler, C.; Paschke, M.; Kruschinski, S.; Henkler, F.; Hahn, J.; Luch, A. Chemical hazards present in liquids and vapors of electronic cigarettes. Arch. Toxicol. 2014, 88, 1295–1308. [Google Scholar] [CrossRef] [PubMed]
- Pisinger, C.; Døssing, M. A systematic review of health effects of electronic cigarettes. Prev. Med. 2014, 69, 248–260. [Google Scholar] [CrossRef] [PubMed]
- Thirión-Romero, I.; Pérez-Padilla, R.; Zabert, G.; Barrientos-Gutiérrez, I. Respiratory impacy of electronic cigarettes and “low-risk” tobacco. Rev. Investig. Clin. 2019, 71, 17–27. [Google Scholar] [PubMed]
- Bertholon, J.F.; Becquemin, M.H.; Annesi-Maesano, I.; Dautzenberg, B. Electronic cigarettes: A short review. Respiration 2013, 86, 433–438. [Google Scholar] [CrossRef]
- Ali, F.R.M.; Dave, D.M.; Colman, G.J.; Wang, X.; Saffer, H.; Marynak, K.L.; Dench, D.; Grossman, M. Association of e-cigarette advertising with e-cigarette and cigarette use among US adults. Addiction 2021, 116, 1212–1223. [Google Scholar] [CrossRef]
- Jun, J.; Kim, J.K. Do state regulations on e-cigarettes have impacts on the e-cigarette prevalence? Tob. Control 2021, 30, 221–226. [Google Scholar] [CrossRef]
- Sapru, S.; Vardhan, M.; Li, Q.; Guo, Y.; Li, X.; Saxena, D. E-cigarettes use in the United States: Reasons for use, perceptions, and effects on health. BMC Public Health 2020, 20, 1518. [Google Scholar] [CrossRef] [PubMed]
- Kosmider, L.; Sobczak, A.; Fik, M.; Knysak, J.; Zaciera, M.; Kurek, J.; Goniewicz, M. Carbonyl Compounds in Electronic Cigarette Vapors: Effects of Nicotine Solvent and Battery Output Voltage. Nicotine Tob. Res. 2014, 16, 1319–1326. [Google Scholar] [CrossRef]
- Goniewicz, M.L.; Kuma, T.; Gawron, M.; Knysak, J.; Kosmider, L. Nicotine Levels in Electronic Cigarettes. Nicotine Tob. Res. 2012, 15, 158–166. [Google Scholar] [CrossRef]
- Fuoco, F.; Buonanno, G.; Stabile, L.; Vigo, P. Influential parameters on particle concentration and size distribution in the mainstream of e-cigarettes. Environ. Pollut. 2014, 184, 523–529. [Google Scholar] [CrossRef]
- Kim, H.-J.; Shin, H.-S. Determination of tobacco-specific nitrosamines in replacement liquids of electronic cigarettes by liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 2013, 1291, 48–55. [Google Scholar] [CrossRef]
- Hoffmann, D.; Rivenson, A.; Chung, F.-L.; Hecht, S.S. Nicotine-Derived N-Nitrosamines (TSNA) and their Relevance in Tobacco Carcinogenesis. Crit. Rev. Toxicol. 1991, 21, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Cheah, N.P.; Chong, N.W.L.; Tan, J.; Morsed, F.A.; Yee, S.K. Electronic nicotine delivery systems: Regulatory and safety challenges: Singapore perspective. Tob. Control 2012, 23, 119–125. [Google Scholar] [CrossRef] [PubMed]
- Etter, J.-F.; Zäther, E.; Svensson, S. Analysis of refill liquids for electronic cigarettes. Addiction 2013, 108, 1671–1679. [Google Scholar] [CrossRef]
- Kubica, P.; Wasik, A.; Kot-Wasik, A.; Namiesnik, J. An evaluation of sucrose as a possible contaminant in e-liquids for electronic cigarettes by hydrophilic interaction liquid chromatography–tandem mass spectrometry. Anal. Bioanal. Chem. 2014, 406, 3013–3018. [Google Scholar] [CrossRef]
- Office on Smoking and Health, National Center for Chronic Disease Prevention and Health Promotion (US). 2016.
- Han, S.; Chen, H.; Zhang, X.; Liu, T.; Fu, Y. Levels of Selected Groups of Compounds in Refill Solutions for Electronic Cigarettes. Nicotine Tob. Res. 2015, 18, 708–714. [Google Scholar] [CrossRef] [PubMed]
- Dawkins, L.; Turner, J.; Hasna, S.; Soar, K. The electronic-cigarette: Effects on desire to smoke, withdrawal symptoms and cognition. Addict. Behav. 2012, 37, 970–973. [Google Scholar] [CrossRef]
- Dawkins, L.; Turner, J.; Crowe, E. Nicotine derived from the electronic cigarette improves time-based prospective memory in abstinent smokers. Psychopharmacology 2013, 227, 377–384. [Google Scholar] [CrossRef] [PubMed]
- Dicpinigaitis, P.V.; Lee, C.A.; Dicpinigaitis, A.J.; Negassa, A. Effect of electronic cigarette use on cough reflex sensitivity. Chest 2015, 149, 161–165. [Google Scholar] [CrossRef] [PubMed]
- Palamidas, A.; Tsikrika, S.; Katsaounou, P.A.; Vakali, S.; Gennimata, S.-A.; Kaltsakas, G.; Gratziou, C.; Koulouris, N. Acute effects of short term use of e-cigarettes on airways physiology and respiratory symptoms in smokers with and without airways obstructive diseases and in healthy non smokers. Tob. Prev. Cessat. 2017, 3, 5. [Google Scholar] [CrossRef] [PubMed]
- Pellegrino, R.M.; Tinghino, B.; Mangiaracina, G.; Marani, A.; Vitali, M.; Protano, C.; Osborn, J.F.; Cattaruzza, M.S. Electronic cigarettes: An evaluation of exposure to chemicals and fine particulate matter (PM). Ann. Ig. 2012, 24, 279–288. [Google Scholar]
- Williams, M.; Villarreal, A.; Bozhilov, K.; Lin, S.; Talbot, P. Metal and silicate particles including nanoparticles are present in electronic cigarette cartomizer fluid and aerosol. PLoS ONE 2013, 8, e57987. [Google Scholar] [CrossRef] [PubMed]
- Lerner, C.A.; Sundar, I.K.; Watson, R.M.; Elder, A.; Jones, R.; Done, D.; Kurtzman, R.; Ossip, D.J.; Robinson, R.; McIntosh, S.; et al. Environmental health hazards of e-cigarettes and their components: Oxidants and copper in e-cigarette aerosols. Environ. Pollut. 2015, 198, 100–107. [Google Scholar] [CrossRef]
- McAuley, T.R.; Hopke, P.; Zhao, J.; Babaian, S. Comparison of the effects of e-cigarette vapor and cigarette smoke on indoor air quality. Inhal. Toxicol. 2012, 24, 850–857. [Google Scholar] [CrossRef]
- Goniewicz, M.L.; Knysak, J.; Gawron, M.; Kosmider, L.; Sobczak, A.; Kurek, J.; Prokopowicz, A.; Jablonska-Czapla, M.; Rosik-Dulewska, C.; Havel, C.; et al. Levels of selected carcinogens and toxicants in vapour from electronic cigarettes. Tob. Control 2014, 23, 133–139. [Google Scholar] [CrossRef] [PubMed]
- Weisberg, E. Smoking and reproductive health. Clin. Reprod. Fertil. 1985, 3, 175–186. [Google Scholar]
- Jandíková, H.; Dušková, M.; Stárka, L. The influence of smoking and cessation on the human reproductive hormonal balance. Physiol. Res. 2017, 66, S323–S331. [Google Scholar] [CrossRef] [PubMed]
- Ochedalski, T.; Lachowicz-Ochedalska, A.; Dec, W.; Czechowski, B. Examining the effects of tobacco smoking on levels of certain hormones in serum of young men. Ginekol. Pol. 1994, 65, 87–93. [Google Scholar] [PubMed]
- Vine, M.F. Smoking and male reproduction: A review. Int. J. Androl. 1996, 19, 323–337. [Google Scholar] [CrossRef]
- Harlev, A.; Agarwal, A.; Gunes, S.O.; Shetty, A.; du Plessis, S.S. Smoking and Male Infertility: An Evidence-Based Review. World J. Men Health 2015, 33, 143–160. [Google Scholar] [CrossRef]
- Pakrashi, A.; Chatterjee, S. Effect of tobacco consumption on the function of male accessory sex glands. Int. J. Androl. 1995, 18, 232–236. [Google Scholar] [CrossRef] [PubMed]
- Natali, A.; Mondaini, N.; Lombardi, G.; Del Popolo, G.; Rizzo, M. Heavy smoking is an important risk factor for erectile dysfunction in young men. Int. J. Impot. Res. 2005, 17, 227–230. [Google Scholar] [CrossRef] [PubMed]
- Cao, S.; Yin, X.; Wang, Y.; Zhou, H.; Song, F.; Lu, Z. Smoking and Risk of Erectile Dysfunction: Systematic Review of Observational Studies with Meta-Analysis. PLoS ONE 2013, 8, e60443. [Google Scholar] [CrossRef]
- Sharma, R.; Harlev, A.; Agarwal, A.; Esteves, S.C. Cigarette Smoking and Semen Quality: A New Meta-analysis Examining the Effect of the 2010 World Health Organization Laboratory Methods for the Examination of Human Semen. Eur. Urol. 2016, 70, 635–645. [Google Scholar] [CrossRef]
- Pacifici, R.; Altieri, I.; Gandini, L.; Lenzi, A.; Pichini, S.; Rosa, M.; Zuccaro, P.; Dondero, F. Nicotine, cotinine, and trans-3-hydroxycotinine levels in seminal plasma of smokers: Effects on sperm parameters. Ther. Drug Monit. 1993, 15, 358–363. [Google Scholar] [CrossRef]
- El Mulla, K.F.; Köhn, F.M.; El Beheiry, A.H.; Schill, W.B. The effect of smoking and varicocele on human sperm acrosin activity and acrosome reaction. Hum. Reprod. 1995, 10, 3190–3194. [Google Scholar] [CrossRef] [PubMed]
- Practice Committee of the American Society for Reproductive Medicine; Penzias, A.; Bendikson, K.; Butts, S.; Coutifaris, C.; Falcone, T.; Gitlin, S.; Gracia, C.; Hansen, K.; Jindal, S.; et al. Smoking and infertility: A committee opinion. Fertil Steril 2018, 110, 611–618. [Google Scholar]
- La Maestra, S.; De Flora, S.; Micale, T. Effect of cigarette smoke on DNA damage, oxidative stress, and morphological alterations in mouse testis and spermatozoa. Int. J. Hyg. Environ. Health 2015, 2018, 117–122. [Google Scholar] [CrossRef] [PubMed]
- Firns, S.; Cruzat, V.; Keane, K.N.; Joesbury, K.A.; Lee, A.H.; Newsholme, P.; Yovich, J.L. The effect of cigarette smoking, alcohol consumption and fruit and vegetable consumption on IVF outcomes: A review and presentation of original data. Reprod. Biol. Endocrinol. 2015, 13, 134. [Google Scholar] [CrossRef] [PubMed]
- Klonoff-Cohen, H. Female and male lifestyle habits and IVF: What is known and unknown. Hum. Reprod. Update 2005, 11, 179–203. [Google Scholar] [CrossRef]
- Håkonsen, L.B.; Ernst, A.; Ramlau-Hansen, C.H. Maternal cigarette smoking during pregnancy and reproductive health in children: A review of epidemiological studies. Asian J. Androl. 2014, 16, 39–49. [Google Scholar] [CrossRef]
- Ozbakir, B.; Tulay, P. Does cigarette smoking really have a clinical effect on folliculogenesis and oocyte maturation? Zygote 2020, 28, 318–321. [Google Scholar] [CrossRef]
- Lyngsø, J.; Kesmodel, U.S.; Bay, B.; Ingerslev, H.J.; Pisinger, C.H.; Ramlau-Hansen, C.H. Female cigarette smoking and successful fertility treatment: A Danish cohort study. Acta Obstet. Gynecol. Scand. 2021, 100, 58–66. [Google Scholar] [CrossRef]
- Harte, C.B.; Meston, C.M. Acute Effects of Nicotine on Physiological and Subjective Sexual Arousal in Nonsmoking Men: A Randomized, Double-Blind, Placebo-Controlled Trial. J. Sex. Med. 2008, 5, 110–121. [Google Scholar] [CrossRef]
- El Golli, N.; Rahali, D.; Jrad-Lamine, A.; Dallagi, Y.; Jallouli, M.; Bdiri, Y.; Ba, N.; Lebret, M.; Rosa, J.; El May, M.; et al. Impact of electronic-cigarette refill liquid on rat testis. Toxicol. Mech. Methods 2016, 26, 417–424. [Google Scholar] [CrossRef]
- Rahali, D.; Jrad-Lamine, A.; Dallagi, Y.; Bdiri, Y.; Ba, N.; El May, M.; El Fazaa, S.; El Golli, N. Semen Parameter Alteration, Histological Changes and Role of Oxidative Stress in Adult Rat Epididymis on Exposure to Electronic Cigarette Refill Liquid. Chin. J. Physiol. 2018, 61, 75–84. [Google Scholar] [CrossRef] [PubMed]
- Vivarelli, F.; Canistro, D.; Cirillo, S.; Cardenia, V.; Rodriguez-Estrada, M.T.; Paolini, M. Impairment of testicular function in electronic cigarette (e-cig, e-cigs) exposed rats under low-voltage and nicotine-freeconditions. Life Sci. 2019, 228, 53–65. [Google Scholar] [CrossRef]
- Wawryk-Gawda, E.; Zarobkiewicz, M.K.; Chłapek, K.; Chylinska-Wrzos, P.; Jodłowska-Jedrych, B. Histological changes in the reproductive system of male rats exposed to cigarette smoke or electronic cigarette vapor. Toxicol. Environ. Chem. 2019, 101, 404–419. [Google Scholar] [CrossRef]
- Vine, M.F.; Tse, C.K.; Hu, P.; Truong, K.Y. Cigarette smoking and semen quality. Fertil. Steril. 1996, 65, 835–842. [Google Scholar] [CrossRef] [PubMed]
- Soares, S.; Simón, C.; Remohi, J.; Pellicer, A. Cigarette smoking affects uterine receptiveness. Hum. Reprod. 2006, 22, 543–547. [Google Scholar] [CrossRef]
- Heger, A.; Sator, M.; Walch, K.; Pietrowski, D. Smoking Decreases Endometrial Thickness in IVF/ICSI Patients. Geburtshilfe Und Frauenheilkd. 2018, 78, 78–82. [Google Scholar] [CrossRef]
- Orzabal, M.R.; Lunde-Young, E.R.; Ramirez, J.I.; Howe, S.Y.; Naik, V.D.; Lee, J.; Heaps, C.L.; Threadgill, D.W.; Ramadoss, J. Chronic exposure to e-cig aerosols during early development causes vascular dysfunction and offspring growth deficits. Transl. Res. 2019, 207, 70–82. [Google Scholar] [CrossRef]
- Szumilas, K.; Szumilas, P.; Grzywacz, A.; Wilk, A. The Effects of E-Cigarette Vapor Components on the Morphology and Function of the Male and Female Reproductive Systems: A Systematic Review. Int. J. Environ. Res. Public Health 2020, 17, 6152. [Google Scholar] [CrossRef]
- Akar, Y.; Ahmad, N.; Khalıd, M. The effect of cadmium on the bovine in vitro oocyte maturation and early embryo development. Int. J. Veter Sci. Med. 2018, 6, S73–S77. [Google Scholar] [CrossRef] [PubMed]
- O’Neill, H.; Nutakor, A.; Magnus, E.; Bracey, E.; Williamson, E.; Harper, J. Effect of Electronic-Cigarette Flavourings on (I) Human Sperm Motility, Chromatin Integrity in Vitro and (II) Mice Testicular Function in Vivo. 2017. Available online: http://srf-reproduction.org/wp-content/uploads/2017/01/Fertility-2017-Final-Programme-and-Abstracts.pdf (accessed on 20 January 2023).
- Holden, L.L.; Truong, L.; Simonich, M.T.; Tanguay, R.L. Assessing the hazard of E-Cigarette flavor mixtures using zebrafish. Food Chem. Toxicol. 2020, 136, 110945. [Google Scholar] [CrossRef]
- Marzec-Wróblewska, U.; Kamiński, P.; Lakota, P.; Szymański, M.; Wasilow, K.; Ludwikowski, G.; Kuligowska-Prusińska, M.; Odrowąż-Sypniewska, G.; Stuczyński, T.; Michałkiewicz, J. Zinc and iron concentration and SOD activity in human semen and seminal plasma. Biol. Trace Elem. Res. 2011, 143, 167–177. [Google Scholar] [CrossRef]
- Marzec-Wróblewska, U.; Kamiński, P.; Łakota, P.; Szymański, M.; Wasilow, K.; Ludwikowski, G.; Jerzak, L.; Stuczyński, T.; Woźniak, A.; Buciński, A. Human Sperm Characteristics with Regard to Cobalt, Chromium, and Lead in Semen and Activity of Catalase in Seminal Plasma. Biol. Trace Elem. Res. 2019, 188, 251–260. [Google Scholar] [CrossRef] [PubMed]
- Shi, X.; Chan, C.P.S.; Man, G.K.Y.; Chan, D.Y.L.; Wong, M.H.; Li, T.C. Associations between blood metal/ metalloid concentration and human semen quality and sperm function: A cross-sectional study in Hong Kong. J. Trace Elem. Med. Biol. 2021, 65, 126735. [Google Scholar] [CrossRef]
- Zhang, X.F.; Gurunathan, S.; Kim, J.H. Effects of silver nanoparticles on neonatal testis development in mice. Int. J. Nanomed. 2015, 10, 6243–6256. [Google Scholar]
- Zhang, T.; Ru, Y.F.; Bin Wu, B.; Dong, H.; Chen, L.; Zheng, J.; Li, J.; Wang, X.; Wang, Z.; Wang, X.; et al. Effects of low lead exposure on sperm quality and sperm DNA methylation in adult men. Cell Biosci. 2021, 30, 110–150. [Google Scholar] [CrossRef] [PubMed]
- Chen, C.; Li, B.; Huang, R.; Dong, S.; Zhou, Y.; Song, J.; Zeng, X.; Zhang, X. Involvement of Ca2+ and ROS signals in nickel-impaired human sperm function. Ecotoxicol. Environ. Saf. 2022, 231, 113181. [Google Scholar] [CrossRef]
- Zhao, L.L.; Ru, Y.F.; Liu, M.; Tang, J.N.; Zheng, J.F.; Wu, B.; Gu, Y.H.; Shi, H.J. Reproductive effects of cadmium on sperm function and early embryonic development in vitro. PLoS ONE 2017, 12, e0186727. [Google Scholar] [CrossRef]
- Roychoudhury, S.; Nath, S.; Massanyi, P.; Stawarz, R.; Kacaniova, M.; Kolesarova, A. Copper-induced changes in reproductive functions: In vivo and in vitro effects. Physiol. Res. 2016, 65, 11–22. [Google Scholar] [CrossRef]
- Kong, L.; Tang, M.; Zhang, T.; Wang, D.; Hu, K.; Lu, W.; Wei, C.; Liang, G.; Pu, Y. Nickel Nanoparticles Exposure and Reproductive Toxicity in Healthy Adult Rats. Int. J. Mol. Sci. 2014, 15, 21253–21269. [Google Scholar] [CrossRef] [PubMed]
- Yiqin, C.; Yan, S.; Peiwen, W.; Yiwei, G.; Qi, W.; Qian, X.; Panglin, W.; Sunjie, Y.; Wenxiang, W. Copper exposure disrupts ovarian steroidogenesis in human ovarian granulosa cells via the FSHR/CYP19A1 pathway and alters methylation patterns on the SF-1 gene promoter. Toxicol. Lett. 2022, 356, 11–20. [Google Scholar] [CrossRef]
- Anttila, A.; Sallmén, M. Effects of parental occupational exposure to lead and other metals on spontaneous abortion. J. Occup. Environ. Med. 1995, 37, 915–921. [Google Scholar] [CrossRef] [PubMed]
- Vosoughi, S.; Khavanin, A.; Salehnia, M.; Mahabadi, H.A.; Shahverdi, A.; Esmaeili, V. Adverse Effects of Formaldehyde Vapor on Mouse Sperm Parameters and Testicular Tissue. Int. J. Fertil. Steril. 2013, 6, 250–267. [Google Scholar]
- Zang, Z.-J.; Fang, Y.-Q.; Ji, S.-Y.; Gao, Y.; Zhu, Y.-Q.; Xia, T.-T.; Jiang, M.-H.; Zhang, Y.-N. Formaldehyde Inhibits Sexual Behavior and Expression of Steroidogenic Enzymes in the Testes of Mice. J. Sex. Med. 2017, 14, 1297–1306. [Google Scholar] [CrossRef]
- Wang, H.-X.; Wang, X.-Y.; Zhou, D.-X.; Zheng, L.-R.; Zhang, J.; Huo, Y.-W.; Tian, H. Effects of low-dose, long-term formaldehyde exposure on the structure and functions of the ovary in rats. Toxicol. Ind. Heal. 2012, 29, 609–615. [Google Scholar] [CrossRef] [PubMed]
- Wang, H.-X.; Li, H.-C.; Lv, M.-Q.; Zhou, D.-X.; Bai, L.-Z.; Du, L.-Z.; Xue, X.; Lin, P.; Qiu, S.-D. Associations between occupation exposure to Formaldehyde and semen quality, a primary study. Sci. Rep. 2015, 5, 15874. [Google Scholar] [CrossRef]
- Balabanič, D.; Rupnik, M.S.; Klemenčič, A.K. Negative impact of endocrine-disrupting compounds on human reproductive health. Reprod. Fertil. Dev. 2011, 23, 403–416. [Google Scholar] [CrossRef]
- Longo, V.; Forleo, A.; Ferramosca, A.; Notari, T.; Pappalardo, S.; Siciliano, P.; Capone, S.; Montano, L. Blood, urine and semen Volatile Organic Compound (VOC) pattern analysis for assessing health environmental impact in highly polluted areas in Italy. Environ. Pollut. 2021, 286, 117410. [Google Scholar] [CrossRef]
- Yang, P.; Wang, Y.-X.; Chen, Y.-J.; Sun, L.; Li, J.; Liu, C.; Huang, Z.; Lu, W.-Q.; Zeng, Q. Urinary Polycyclic Aromatic Hydrocarbon Metabolites and Human Semen Quality in China. Environ. Sci. Technol. 2017, 51, 958–967. [Google Scholar] [CrossRef] [PubMed]
- Khoudja, R.Y.; Xu, Y.; Li, T.; Zhou, C.J. Better IVF outcomes following improvements in laboratory air quality. J. Assist. Reprod. Genet. 2013, 30, 69–76. [Google Scholar] [CrossRef]
- Hall, J.; Gilligan, A.; Schimmel, T.; Cecchi, M.; Cohen, J. The origin, effects and control of air pollution in laboratories used for human embryo culture. Hum. Reprod. 1998, 13 (Suppl. S4), 146–155. [Google Scholar] [CrossRef] [PubMed]
- Chen, T.; Wu, M.; Dong, Y.; Kong, B.; Cai, Y.; Hei, C.; Wu, K.; Zhao, C.; Chang, Q. Effect of e-cigarette refill liquid on follicular development and estrogen secretion in rats. Tob. Induc. Dis. 2022, 20, 36. [Google Scholar] [CrossRef] [PubMed]
- Wetendorf, M.; Randall, L.T.; Lemma, M.T.; Hurr, S.H.; Pawlak, J.B.; Tarran, R.; Doerschuk, C.M.; Caron, K.M. E-Cigarette Exposure Delays Implantation and Causes Reduced Weight Gain in Female Offspring Exposed In Utero. J. Endocr. Soc. 2019, 3, 1907–1916. [Google Scholar] [CrossRef] [PubMed]
- McGrath-Morrow, S.A.; Hayashi, M.; Aherrera, A.; Lopez, A.; Malinina, A.; Collaco, J.M.; Neptune, E.; Klein, J.D.; Winickoff, J.P.; Breysse, P.; et al. The Effects of Electronic Cigarette Emissions on Systemic Cotinine Levels, Weight and Postnatal Lung Growth in Neonatal Mice. PLoS ONE 2015, 10, e0118344. [Google Scholar] [CrossRef]
- Smith, D.; Aherrera, A.; Lopez, A.; Neptune, E.; Winickoff, J.P.; Klein, J.D.; Chen, G.; Lazarus, P.; Collaco, J.M.; McGrath-Morrow, S.A. Adult Behavior in Male Mice Exposed to E-Cigarette Nicotine Vapors during Late Prenatal and Early Postnatal Life. PLoS ONE 2015, 10, e0137953. [Google Scholar] [CrossRef]
- Greene, R.M.; Pisano, M.M. Developmental toxicity of e-cigarette aerosols. Birth Defects Res. 2019, 111, 1294–1301. [Google Scholar] [CrossRef]
- Freour, T.; Masson, D.; Mirallie, S.; Jean, M.; Bach, K.; Dejoie, T.; Barriere, P. Active smoking compromises IVF outcome and affects ovarian reserve. Reprod. Biomed. Online 2008, 16, 96–102. [Google Scholar] [CrossRef] [PubMed]
- Klonoff-Cohen, H.; Natarajan, L.; Marrs, R.; Yee, B. Effects of female and male smoking on success rates of IVF and gamete intra-Fallopian transfer. Hum. Reprod. 2001, 16, 1382–1390. [Google Scholar] [CrossRef]
- St. Helen, G.; Havel, C.; Dempsey, D.A.; Jacob, P., 3rd; Benowitz, N.L. Nicotine delivery, retention and pharmacokinetics from various electronic cigarettes. Addiction 2016, 111, 535–544. [Google Scholar] [CrossRef] [PubMed]

| Outcome | Evidence | References |
|---|---|---|
| Conventional cigarette and male reproduction |
| [34,35] |
| [35,36,37,38] | |
| [38] | |
| [39] | |
| [40,41] | |
| [37,38,42,43,44] | |
| [37,38,45,46] | |
| [45] | |
| [37] | |
| Conventional cigarette smoking and female reproduction |
| [47,48] |
| [35,49] | |
| Conventional cigarette smoking and ART outcomes |
| [50] |
| [35,51] | |
| [48] | |
| [35] | |
| [47] |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Montjean, D.; Godin Pagé, M.-H.; Bélanger, M.-C.; Benkhalifa, M.; Miron, P. An Overview of E-Cigarette Impact on Reproductive Health. Life 2023, 13, 827. https://doi.org/10.3390/life13030827
Montjean D, Godin Pagé M-H, Bélanger M-C, Benkhalifa M, Miron P. An Overview of E-Cigarette Impact on Reproductive Health. Life. 2023; 13(3):827. https://doi.org/10.3390/life13030827
Chicago/Turabian StyleMontjean, Debbie, Marie-Hélène Godin Pagé, Marie-Claire Bélanger, Moncef Benkhalifa, and Pierre Miron. 2023. "An Overview of E-Cigarette Impact on Reproductive Health" Life 13, no. 3: 827. https://doi.org/10.3390/life13030827
APA StyleMontjean, D., Godin Pagé, M.-H., Bélanger, M.-C., Benkhalifa, M., & Miron, P. (2023). An Overview of E-Cigarette Impact on Reproductive Health. Life, 13(3), 827. https://doi.org/10.3390/life13030827







