Abstract
The genus Kalanchoe Adans. (Crassulaceae) is native to Madagascar and comprises 145 species, being naturalized in the tropics and cultivated worldwide. In addition to having ornamental value, several Kalanchoe species are commonly used in popular medicine for the treatment of inflammatory conditions, wounds, gastric ulcers, and other diseases. The great importance of the genus is reflected on its acknowledgment by traditional and alternative health systems and organizations, as well as on the growing number of papers reporting pharmacological properties of extracts and isolated compounds from Kalanchoe. Among these properties, we highlight anti-inflammatory, antitumor, wound healing, antiulcer, and muscle relaxing properties. These activities are attributed mostly to flavonoids and bufadienolides, the main secondary metabolites reported in Kalanchoe extracts. While bufadienolides are generally related to cytotoxic and muscle relaxing activities, flavonoids are commonly reported as anti-inflammatory and wound healing agents. This review provides up to date information and perspectives on bioactive compounds from the Kalanchoe genus that are potentially useful for the development of new drugs. It includes not only a discussion on the advantages of the Kalanchoe species as source of bioactive compounds, but also the gaps, opportunities, and challenges to translate the acquired knowledge into innovation for drug development.
1. Introduction
The Kalanchoe Adans. (Syn.: Bryophyllum Salisb.) genus comprises succulent perennial plants intensively used for ornamental and medicinal purposes [1,2]. It belongs to Crassulaceae family and currently encompasses 145 species [3], among which some are largely known, such as Kalanchoe pinnata (Syn. Bryophyllum pinnatum), K. daigremontiana (B. daigremontianum), K. blossfeldiana, and K. delagoensis (Syn. K. tubiflora, B. delagoense, B. tubiflorum). The genus is native to Madagascar, although its species can be found worldwide, especially in the tropics due to easy propagation and acclimation of the plants [2,4].
Plants of the genus are widely used in traditional medicine in several countries, including India, China, South Africa and other African nations, and Brazil, as well as in alternative medicine systems. Indeed, in some of these countries, we can find reports of the use of Kalanchoe plants in Ayurveda, Chinese Traditional Medicine, and anthroposophic medicine [1,5,6,7]. In particular, in Brazil, K. pinnata is part of a list of medicinal plants to be used in the national public health system (SUS–Sistema Único de Saúde) [8]. Among the ethnomedicinal uses, plants of the genus are reputed therapeutics in the treatment of inflammatory conditions, wounds, gastric ulcers, genito-urinary disorders, and other illnesses [1,9].
The multiple medicinal uses of Kalanchoe plants stimulated several pharmacological studies by research groups around the world. In fact, the importance of the genus is corroborated by the increasing number of papers regarding medicinal uses and properties of Kalanchoe species in the last 30 years, according to the PubMed database (Figure 1).
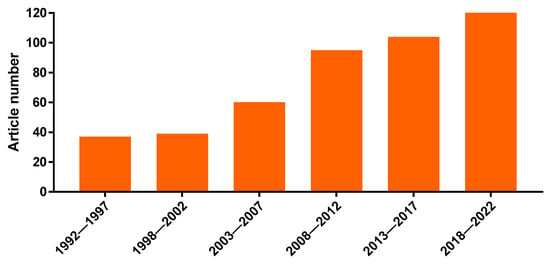
Figure 1.
Evolution of the number of papers in indexed journals regarding medicinal uses and properties of Kalanchoe plants, distributed in 5-year periods (from 1992 to 2022). Source: PubMed, consulted on 15 December 2022 with the keywords “Kalanchoe” and “Bryophyllum”.
Several biological activities have been shown for extracts, fractions, and some isolated compounds of Kalanchoe plants. The most cited activities are anti-inflammatory, antimicrobial, wound-healing, muscle relaxing, and antitumor activities [1,6,9,10,11]. Bufadienolides and flavonoids are the most reported compounds in plant extracts and stand out as bioactive molecules, being potentially useful for the development of new drugs [1,6,12]. Despite this knowledge, a review focused on the importance and pharmacological properties of both bufadienolides and flavonoids is unprecedented. Therefore, this paper is a critical review of the literature (1989–2023) and provides up to date information and perspectives on these bioactive compounds, also including a discussion on the advantages of the Kalanchoe genus, as well as the gaps, opportunities, and challenges to translate the acquired knowledge into innovation for drug development.
Three decades of pioneering multidisciplinary studies conducted by our group and collaborators described a broad pharmacological profile of extracts and bioactive compounds, mainly flavonoids, of Kalanchoe species (including K. brasiliensis, K. pinnata, K. daigremotiana, K. gastonis-bonnieri, and K. thyrsiflora, among others) demonstrating, for example, antioxidant, anti-inflammatory, antileishmaniasis, antiviral, immunomodulatory, and anti-allergic activities [1,11,13,14,15,16,17,18]. We have also worked on biotechnological aspects of plants of this genus, demonstrating the elicitation of flavonoids production and the optimization of their extraction [19,20,21].
The papers considered in this review were obtained from PubMed, Scopus, and Google Scholar databases, resulting from the search with the following keywords combined: “Kalanchoe”, “Bryophyllum”, “flavonoids”, and “bufadienolides”. Papers reporting only the biological activity of Kalanchoe extracts, without identifying the bioactive compound responsible for the described activity, were not considered in detail, being only cited in some parts of the text. Additionally, some criteria were taken into account for the choice of the papers here cited, such as the quality of the information presented (based on the analytical studies, chemical characterization of the compounds, and biological assays conducted) and the indexation of the journal in which they were published.
2. Main Classes of Bioactive Compounds in Kalanchoe Species
2.1. Bufadienolides
Bufadienolides are C-24 steroids characterized by a six-membered lactone (α-pyrone) ring located at C-17β. They can be found in the form of glycosides and aglycones in animals (mainly toads from the genus Bufo) and in several plant families such as Crassulaceae, Iridaceae, and Melianthaceae. The biological activities most reported for this class of compounds are cardiotonic and antitumor activities [12,22]. Species of the genus Kalanchoe are known to produce bufadienolides with diverse structural features, including glycosides and orthoacetate forms. These compounds have been reported mainly in K. daigremontiana, K. delagoensis (syn.: Kalanchoe tubiflora), K. pinnata, K. ceratophylla (syn.: Kalanchoe gracilis), K. tomentosa, and K. lanceolata [12].
The great majority of pharmacological studies carried out with Kalanchoe bufadienolides are related to their antitumor properties. Bufadienolides seem to be the main substances responsible for the antitumor activity of Kalanchoe extracts, as evidenced by bioassay-guided studies [23,24,25,26]. Table 1 summarizes the data on antitumor properties of bufadienolides from Kalanchoe species in human cell lines. Their structures (1–18) are shown in Figure 2. Among these substances, bryophyllin A (7), also known as bryotoxin C, stands out. This compound, found in K. ceratophylla, K. delagoensis, and K. pinnata, has evidenced antitumor activity in eleven different cancer cell lines. Bersaldegenin-1,3,5-ortoacetate (9), also found in different Kalanchoe species, is also worth mentioning as well, having evidenced activity against nine distinct cell lines. The action mechanism of this substance in HeLa cells, a cervical cancer cell line, was recently evaluated in detail by Stefanowicz-Hajduk [27]. Bersaldegenin-1,3,5-ortoacetate induced DNA damages, overproduction of reactive oxygen species, and cell cycle arrest in the G2/M phase. The DNA damages were possibly associated with the overexpression of NF-κB inhibitor genes, whose pathways take part in the regulation of gene transcription [27].

Table 1.
Antitumor activity of bufadienolides from Kalanchoe species.
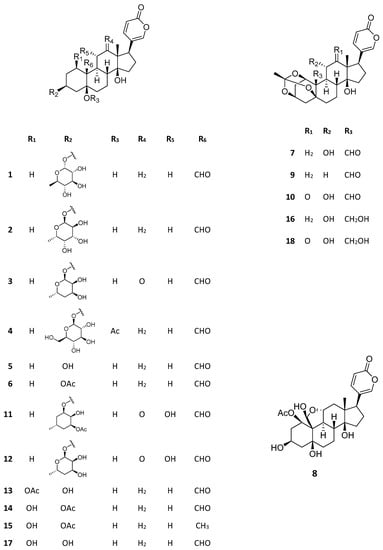
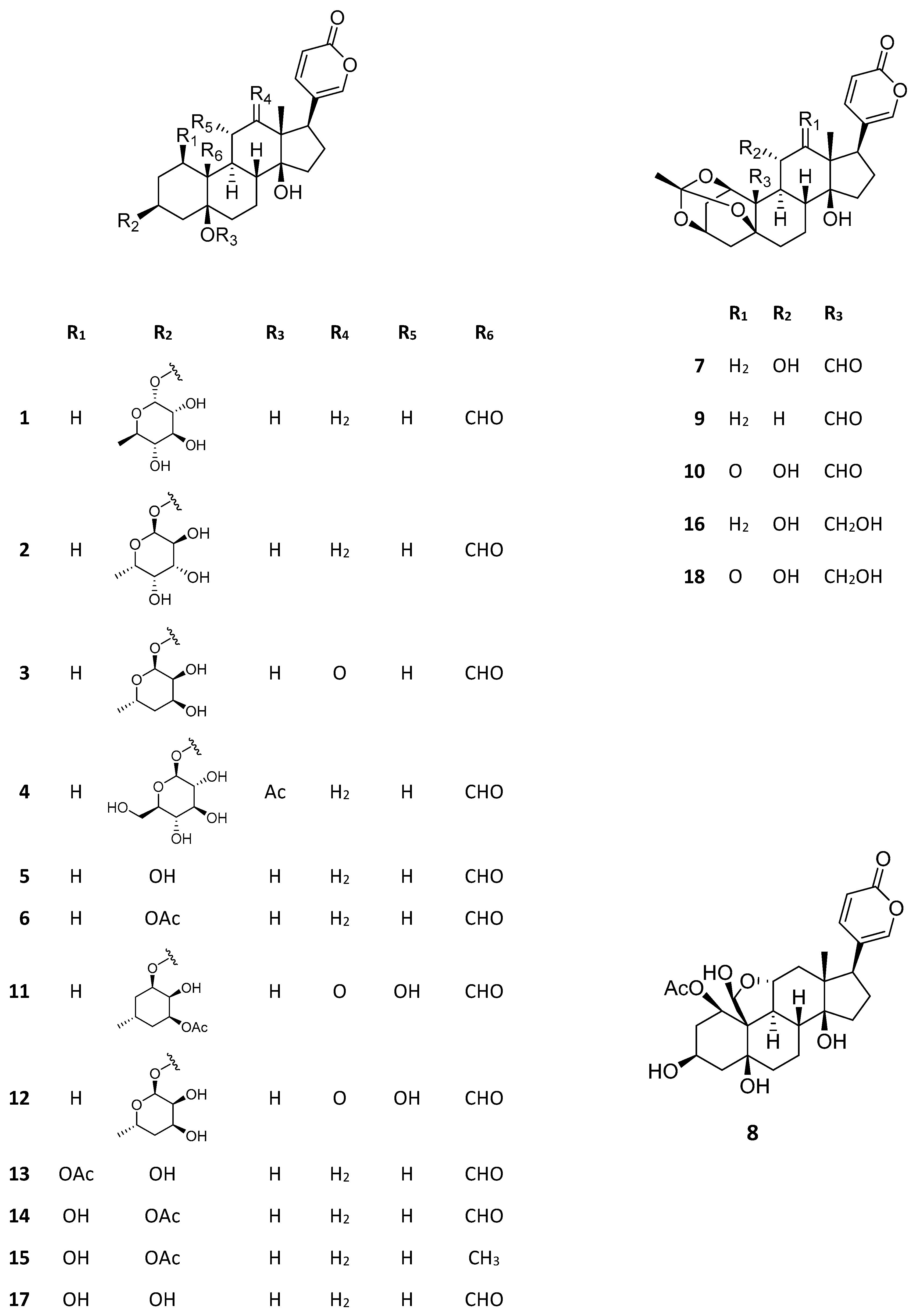
Figure 2.
Structures of Kalanchoe bioactive bufadienolides. Kalanchoside A (1), kalanchoside B (2), kalanchoside C (3), thesiuside (4), hellebrigenin (5), hellebrigenin-3-acetate (6), bryophyllin A (7), bryophyllin B (8), bersaldegenin-1,3,5-orthoacetate (9), daigremontianin (10), kalantuboside A (11), kalantuboside B (12), bersaldegenin-1-acetate (13), bersaldegenin-3-acetate (14), daigredorigenin-3-acetate (15), bryophyllin C (16), bersaldegenin (17), bryotoxin B (18).
Bryophyllin A, along with other two bufadienolides (kalantubosides A and B—11 and 12) from K. delagoensis, also had their activity in CL1-5 human lung cancer cells evaluated at the molecular level [28]. The substances produced no significant induction of apoptosis or cell cycle arrest and their main cell death mechanism was shown to be the activation of autophagy pathways [28].
Interestingly, in another study, kalantuboside B (12) was shown to induce apoptosis of A2058 melanoma cells, probably via mitochondria, death-receptor-mediated, and ER stress pathways, and the observed autophagy was probably not involved in cell death [29]. The authors also conducted further in vivo studies with A2058-xenografted mice. The in vivo experiments confirmed the previous in vitro results: kalantuboside B (0.4 mg/kg i.p. every two days for 8 weeks) was able to suppress tumor development by apoptosis promotion with no toxicity signs [29].
Other pharmacological activities are attributed to bufadienolides. A bufadienolide-enriched fraction from K. pinnata leaf juice revealed relaxing activity in bladder porcine muscle strips (0.1–1 mg/mL), while a flavonoid-enriched fraction from the same extract showed no significant activity [33]. In another study carried out by the same group, both bufadienolides and flavonoid-enriched fractions reduced the contractility of human myometrium [34]. Nevertheless, the former was the most active (40% decrease at 1 µg/mL), evidencing that bufadienolides are the main factor responsible for K. pinnata activity on muscle tissue, an important pharmacological effect in the context of the anthroposophic medicinal use of the plant for overreactive bladder and premature labor (see Section 3.1). The main bufadienolides present in the enriched fraction in both studies were bersaldegenin-1-acetate (13), bersaldegenin-3-acetate (14), bersaldegenin-1,3,5-orthoacetate (9), and bryophyllin A (7) [33,34]. A more recent study of the authors evaluated the effects of bioactive compounds from K. pinnata in the oxytocin signaling pathways in human myometrial cell cultures and showed that both bufadienolides and flavonoids take part in the cell events that lead to the inhibition of oxytocin signaling by K. pinnata leaf extracts [35]. The authors evaluated a bufadienolide-enriched fraction, a flavonoid-enriched fraction, and bersaldegenin-1,3,5-orthoacetate. All of them showed to inhibit the oxytocin-induced increase in intracellular calcium concentration, but none of them were as active as the whole leaf extract.
In addition to the studies of bufadienolides from K. pinnata extracts, there is also evidence for the activity of these compounds from K. daigremontiana. A bufadienolide-enriched fraction from a root aqueous extract of the plant showed to be active on hemostasis, being able to inhibit the thrombin activity in vitro in the concentration range of 1–50 µg/mL. A molecular docking study showed that the most probable thrombin inhibitors in the tested fraction were the bufadienolides bersaldegenin (17), bersaldegenin 1-acetate (13), bersaldegenin 1,3,5-orthoacetate (9), and daigredorigenin 3-acetate (15), as well as the lignoids hovetrichoside C and schisandriside [36]. Thrombin is a critical plasma coagulation factor and the capacity of bufadienolides to inhibit its enzymatic activity points to a possible anti-thrombotic effect [36]. Additionally, the same group evaluated the activity of a similar bufadienolide-enriched fraction from K. daigremontiana roots on the plasmin activity. Plasmin is part of the fibrinolytic system, which regulates the extent of formed thrombi. The type of effect exerted by the fraction on the enzymatic properties of plasmin was dependent on the concentration range: lower concentrations (0.05–2.5 µg/mL) enhanced its activity, while higher concentrations (25–50 µg/mL) resulted in inhibition. As stated by the authors, the effect observed probably resulted from an allosteric regulation of plasmin activity. According to the docking studies, the most probable bioactive compounds from the fraction were the bufadienolides bersaldegenin 1-acetate (14) and bryotoxin B (18), as well as hovitrichoside C [37].
2.2. Flavonoids
Flavonoids are probably the best known and most active molecules found in Kalanchoe species. For these phenolic substances, a broad profile of pharmacological activity in diverse targets has been reported [1]. These compounds, ubiquitously distributed in plants, are commonly found in fruits, leaves, seeds, stems, and flowers of angiosperms [38]. In Kalanchoe, flavonoids from leaves are the most studied [1,39]. Flavonoids comprise the largest class of polyphenols and mainly consist of two aromatic rings (A and B), united to a central heterocyclic ring (C), resulting in a basic unit C6-C3-C6. These substances are derived from mixed biosynthetic pathways: while the A-ring originates from the acetic acid pathway, with characteristic hydroxylation at 5′ and 3′ positions, the B-ring originates from the shikimic acid pathway [38]. Flavonoids comprise different structural subclasses such as flavonols, flavones, flavanonols, flavanones, flavanols, and anthocyanidins, differentiated mainly by the degree of oxidation and substituents of the C ring [40]. These polyphenolic compounds have well known biological activities, several of them related to their ability to scavenge free radicals, resulting in antioxidant activity. These include the prevention of cardiovascular and neurodegenerative diseases, cancer, inflammatory processes, atherosclerosis, and in their action in diabetes and autoimmune diseases, as well as antimicrobial and antiviral properties [41]. These molecules are also known for their immunomodulatory activity [42].
Flavonoids have been extensively reported in different Kalanchoe species, including K. brasiliensis, K. ceratophyla (syn.: K. gracilis), K. spathulata, K. pinnata, K. daigremontiana, K. blossfeldiana, K. gastonis-bonnieri, and others [1,17,39]. These compounds were firstly identified in the genus in the 1970s with the studies of Gain and Gupta [43], who showed the presence of quercetin and kaempferol glycosides in K. pinnata leaf extracts. Indeed, studies developed in the last 50 years have shown that flavonoids in this genus are mainly represented by flavonol and flavone glycosides [1,43,44]. Despite the growing number of studies describing pharmacological actions of Kalanchoe extracts, and the knowledge about flavonoids as important chemical constituents in this genus, most of the studies have just reported the presence of these molecules, without properly investigating their bioactive behavior. Indeed, several of these studies only suggest the potential role of flavonoids in a given activity [45,46,47,48,49,50,51,52], without assessing the isolated compound to confirm its bioactivity. The characterization of bioactive compounds is important not only to define the molecules responsible for a given biological property, but also for the development of phytomedicines. The use of such substances as biomarkers (active marker) for standardization and quality control guarantees the safety and reproducibility of the pharmacological effects of these formulations, being preferable to the use of analytical markers, unrelated to the therapeutic activity [53].
Due to the importance of identifying the bioactive compound, the number of papers tentatively attributing the biological activity of Kalanchoe extracts to flavonoids or to flavonoid-enriched fractions is increasing nowadays. In a study on the activity of K. pinnata in a mice model of lupus arthritis, the authors observed that the ethyl acetate fraction (rich in quercetin glycosylated flavonoids), obtained from a leaf aqueous extract, inhibited the progression of the disease [54]. This fraction also produced positive effects on anti-Smith antibody and T reg in lupus mice [55]. Still regarding K. pinnata, ethyl acetate fractions enriched in quercetin, luteolin, isorhamnetin, and luteolin-7-glucoside ameliorated oxidative imbalance in neurotoxicity caused by aluminum chloride in rats, showing a neuroprotective effect [56]. Antioxidant properties and anticholinesterase activity have also been described for this fraction [57]. A chloroform fraction of stem methanolic extracts from K. ceratophyla (syn. K. gracilis) presenting a high content of flavonoids showed antioxidant, anti-inflammatory, and antiproliferative activities [58]. In some of these studies, the effect of the whole extract is higher than that of the fractions [35,58], indicating a synergistic effect of these compounds.
Several examples of bioactivity for flavonoids isolated from extracts of Kalanchoe plants are presented in Table 2. Their chemical structures can be seen in Figure 3. In particular, previous studies developed by our group demonstrated several activities of flavonoids isolated from Kalanchoe species, such as leishmanicidal, anti-allergic, antinociceptive, antiedematogenic, anti-inflammatory, antiviral, antitumor, immunosuppressive, and wound healing activities [1,11,15,16,59,60,61,62].

Table 2.
Flavonoids from Kalanchoe species and their biological activities.
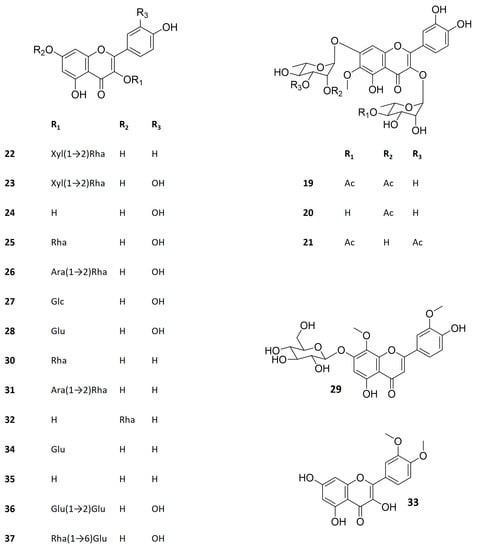
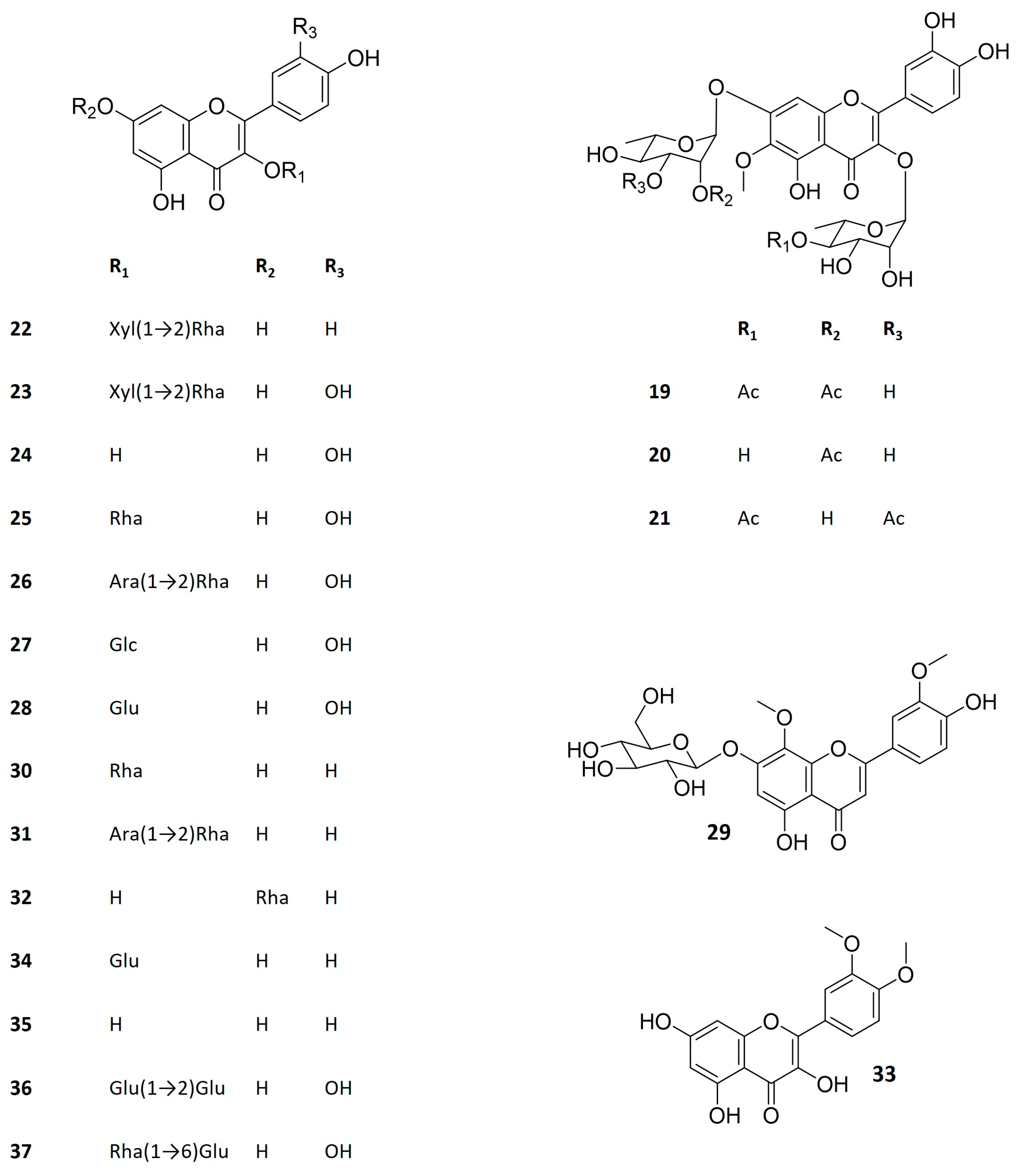
Figure 3.
Structure of Kalanchoe bioactive flavonoids. Kalambroside A (19), kalambroside B (20), patuletin 3-O-(4″-O-acetyl-α-L-rhamnopyranosyl)-7-O-(3‴-O-acetyl-α-L-rhamnopyranoside) (21), kaempferol 3-O-β-D-xylopyranosyl (1→2) α-L-rhamnopyranoside (22), quercetin 3-O-β-D-xylopyranosyl (1→2) α-L-rhamnopyranoside (23), quercetin (24), quercitrin (25), quercetin 3-O-α-L-arabinopyranosyl (1→2) α-L-rhamnopyranoside (26), miquelianin (27), isoquercitrin (28), 4′,5-dihydroxy-3′,8-dimethoxyflavone-7-O-D-glucopyranioside (29), afzelin (30), kapinnatoside (31), α-rhamnoisorobin (32), 3′,4′-dimethoxy quercetin (33), astragalin (34), kaempferol (35), quercetin-3-O-sophoroside (36), rutin (37). Ac–acetate, Ara–arabinose, Glc–glucuronic acid, Glu–glucose, Rha–rhamnose, Xyl–xylose.
Among the flavonoids described in Kalanchoe genus, the glycosyl derivatives of quercetin, kaempferol, eupafolin, and patuletin are the most common [1,39]. Considering the bioactive flavonoids in the genus, quercetin glycosides are the most cited (Table 2). Indeed, several Kalanchoe species have quercetin derivatives as the compounds responsible for antimicrobial (including antiprotozoal), antiviral, anti-inflammatory, antioxidant, gastroprotective, immunomodulatory, and wound healing activities (Table 2). Quercetin (24), quercetin 3-O-ramnopyranonoside (hereafter called quercitrin, 25), and quercetin 3-O-α-L-arabinopyranosyl (1→2) α-L-rhamnopyranoside (hereafter referred as QAR, 26) occurring in K. pinnata, the most studied species of the genus, are intensively described as bioactive compounds for several biological effects, some of them proven by in vivo assays (Table 2).
In fact, K. pinnata is the species for which we have the largest number of in vivo studies.
Quercetin (24) is a flavonol widely present in plants. Several quercetin-derived glycosides can be found in nature, varying from very common compounds, such as rutin and quercitrin, to unusual ones. The glycosylation usually occurs in the -OH at the C-3 position, but it can occur on 4′-OH and 7-OH as well [74,75]. Studies with K. pinnata reported that quercetin has shown antiviral, antileishmanial, immunomodulatory, and immunossupressive activities [16,60,61,63]. In addition to these effects, quercetin and other flavonoids obtained from K. prolifera showed antitumor activity against murine leukemic cells [71]. Studies have already revealed the wide range of quercetin activity in inhibiting the proliferation of cancer cells through different mechanisms of action [76]. Regarding human leukemia, quercetin has shown activity using a xenograft model of HL60 human leukemic cells. There is evidence that quercetin induces signaling at the levels of apoptosis, cell cycle, and autophagy culminating in the inhibition of HL60 tumor growth [77]. A pilot clinical study carried out in patients with chronic lymphocytic leukemia/small lymphocytic lymphoma to evaluate the antitumor effect of quercetin (500 mg/twice daily; 3 months) showed promising results despite the small sample size [78].
There is a huge number of studies supporting the antiviral properties of quercetin. In particular, this flavonoid showed inhibition against adenovirus, herpes simplex virus, Japanese encephalitis virus, and respiratory syncytial virus [74,79]. The mechanisms of action have been also described for several viruses. For instance, quercetin interacts with the NS3 helicase, NS5B polymerase, and p7 proteins of hepatitis C virus (HCV), suggesting that the anti-HCV activity of this flavonoid occurs through inhibition of NS3 helicase and heat-shock proteins [74,80,81]. Quercetin may inhibit the viral infection of the influenza virus in different ways: (i) blocking the endocytosis of the virus by the host cell, (ii) hampering the replication and translation of the virus, and (iii) increasing the viral clearance. It is worth mentioning that, recently, quercetin has been also investigated for its effect on coronavirus (Coronaviridae) [74].
Quercetin, along with some of its glycosides, also plays a significant role in the immunomodulatory activity of K. pinnata. This flavonoid, compared with its glycosides quercitrin, QAR, miquelianin, and isoquercitrin, exhibited the highest in vitro inhibitory activity (IC50 = 2.5 μg/mL) of lymphocyte proliferation in mice, suggesting that the increased glycosylation reduces the inhibitory activity under in vitro conditions [16]. In addition, another study with quercetin obtained from K. pinnata showed that this flavonoid inhibited degranulation and cytokine production of bone marrow-derived mast cells in in vitro assays [61]. Quercetin also exhibited a decrease in the development of airway hyperresponsiveness, airway inflammation, goblet cell metaplasia, and production of the cytokines IL-5, IL-13, and TNF in an in vivo model [61].
Quercetin has also been shown to be a molecule with antileishmanial properties. Oral treatment with this flavonoid suppressed the burden of Leishmania amazonensis by 76% compared with untreated animals, a result superior to that of the positive control [60]. The effect of quercetin on cutaneous leishmaniasis was compatible with its effectiveness in the visceral form of this disease [82]. Previous studies showed that quercetin has in vitro leishmanicidal activity against promastigote forms of L. amazonensis as well as amastigote forms of L. amazonensis and L. donovani [83,84]. Recently, it was reported that quercetin is also active against the proliferation of L. braziliensis promastigotes [83]. The mode of action of this flavonoid against leishmaniasis and other protozoal parasites is the matter of several studies. It is believed that quercetin acts in different ways: disrupting the mitochondria function; inhibiting heat shock proteins, DNA topoisomerases, and kinases; increasing ROS and RNS production; causing DNA degradation; or by stimulating nitric oxide and cytokines production [85].
The in vitro antileishmanial activity observed for K. pinnata extracts was not only attributed to quercetin. Other leishmanicidal flavonoids were isolated from the aqueous leaf extract of this Kalanchoe species and tested both in vitro [59,66] and in vivo [60]. These flavonoids include quercitrin (25), QAR (26), kapinnatoside (31), afzelin (30), and 4′,5-dihydroxy-3′,8-dimethoxyflavone-7-O-D-glycopyranoside (29). Kapinnatoside and afzelin, both kaempferol derivates, showed to be less active than quercetin glycosides in this study, suggesting the importance of the B ring hydroxylation feature for antileishmanial activity [59].
Despite the importance of the various biological activities demonstrated for these flavonoids obtained from K. pinnata, maybe the most prominent bioactive flavonoid is QAR (26). It is the major component of the aqueous leaf extracts of K. pinnata, equivalent to about 65% of the total area of chromatographic signals corresponding to phenolics detected by high performance liquid chromatography (HPLC) [19,21,67,69]. Although this compound occurs in such high content in K. pinnata leaf extracts, it is not a common molecule in nature (see Section 3.2), and for this reason it can be proposed as a chemical marker of this Kalanchoe species [21]. It is also reported in extracts of flowers of K. pinnata and K. blossfeldiana [16,86], but in lesser amounts. Considering the several biological activities of K. pinnata extracts attributed to this flavonoid in the last 15 years (Table 2), this compound might be proposed as bioactive marker of the species, in addition to the already attributed role of a chemical marker.
In addition to the antileishmanial activity [59,60], QAR has demonstrated several other biological activities both in vitro and in vivo [15,16,19,67,68,69]. It has exhibited a potent antioxidant activity and the inhibition of T-cell proliferation [16,19]. Furthermore, this flavonoid has shown strong anti-inflammatory (carrageenan-induced leukocyte migration test, ID50 2.0 mg/kg), antiedematogenic (in croton oil-induced ear edema, ID50 0.76 mg/kg), and antinociceptive (in the acetic acid-induced writhing model, ID50 9.4 mg/kg) activities in mice [15]. The evaluation of the mechanism of action involved in these effects revealed that QAR inhibits the activity of both COX-1 and COX-2, with preferential inhibition to COX-1, besides reducing the TNF-α concentration in pleural exudates. These results indicate that the anti-inflammatory effects of QAR involve COX-1/COX-2 and TNF-α synthesis/release inhibition [15].
Also regarding the anti-inflammatory activity, QAR has recently shown in vitro inhibition and high selectivity of the PDE4B enzyme (phosphodiesterase 4B), being proposed as an interesting candidate for the design of other PDE4B inhibitors [67]. In fact, PDE4B has lately been identified as a major cyclic AMP (cAMP)-metabolizing enzyme in inflammatory cells. Therefore, the inhibition of PDE4B decreases the inflammatory responses in multiple cell types [87]. QAR is also referred to as a modulator of Organic Anion Transporting Polypeptides (OATPs) [88,89], sodium-dependent membrane carriers present in several human organs responsible for mediating the uptake of various anticancer drugs, including those used in the treatment of liver cancer [89].
Studies on the activity of K. pinnata in an animal model of gastric ulcer revealed the beneficial effect of ethyl acetate fractions of leaf hydroethanolic extracts. Gastroprotective activity was attributed to QAR and quercitrin [49,52]. Recently, De Araújo et al. [69] demonstrated the gastroprotective property of QAR against gastric lesions induced by ethanol and indomethacin in mice. The authors observed that the oral pretreatment with QAR (5 mg/kg) reduced the gastric lesions of the animals. However, it was supposed that this compound acts synergistically with other compounds increasing the gastroprotection [69]. It is also important to highlight that QAR and quercitrin have shown inhibitory activity in α-glucosidases, being proposed as candidates for the treatment of type II diabetes [65]. Indeed, inhibitors of α-glucosidase stand out as a noninvasive treatment for this metabolic disorder, and quercetin and its glucosides are being studied as potential drugs [90,91].
In a recent study developed by our group, a cream formulated with QAR showed a wound healing effect in a rat model of an excisional wound [68]. Creams obtained with K. pinnata leaf extracts and the isolated flavonoid (QAR) exhibited 95.3 ± 1.2% and 97.5 ± 0.8% of healing on the 12th day of experiment, respectively, both showing to be as effective as the positive control (a commercial cream containing neomycin and bacitracin, 96.7 ± 0.8%). Based on these results, QAR can be considered as the bioactive substance for the wound healing effect, since it plays a fundamental role in this activity [68]. Although the mode of action of the cream containing QAR has not been described, several previous works supported the wound healing effect of K. pinnata extracts rich in this flavonoid [92,93]. The already proven anti-inflammatory activity of QAR, in addition to the antioxidant and antimicrobial activities, can also favorably contribute to the healing stages [68]. In addition, a gel formulated with leaf extracts of K. pinnata and having QAR as the main compound also stimulated skin wound healing in rats in a very recent study [94]. The authors evaluated the stability of the gel by checking the content of QAR for 30 days, using this flavonoid as a chemical and quality control marker [94]. All these findings show the importance of QAR as a bioactive molecule in the wound healing effect. Quercetin and its glycosides are intensively studied as wound healing compounds and the ability of this flavonoid to improve the healing process seems to be associated with its well-established anti-inflammatory and antioxidant activities. Some other examples of quercetin derivatives with wound healing effects include quercetin-3-O-glucoside (isoquercitrin), quercetin-3-O-rhamnosyl-(1→6)-glucoside, quercetin-3-O-galactoside, and quercetin-3-O-rhamnoside (quercitrin), some of them also described in extracts of K. pinnata [95,96,97].
Compared to quercetin derivatives, kaempferol glycosides are less described as bioactive constituents of K. pinnata extracts, despite being also reported in this species and others belonging to the same genus. For instance, afzelin (30), a kaempferol rhamnoside found in many Kalanchoe species, has presented antitumor and antileishmanial activities. In the study of Aisyah et al. [71], the cytotoxic activity of afzelin was comparable with that of other kaempferol and quercetin monoglycosides, as depicted at Table 2. The in vitro effect of the flavonoid on Leishmania amazonensis amastigotes, on the other hand, was weaker than that presented by quercetin glycosides (QAR and quercitrin) [59]. Afzelin is a rather common flavonoid, which has been reported in a variety of species such as Sedum dendroideum (Crassulaceae) [98], Bauhinia forficata (Fabaceae) [99], Ficus palmata (Moraceae), and Nymphaea odorata (Nymphaeaceae) [100], among others. Possibly for that reason, the pharmacological properties of the flavonoid have been extensively studied. Besides the above-mentioned activities, other examples include in vivo anti-asthmatic [100] and diuretic effects [99]. The antitumor activity of the flavonoid was also demonstrated in vivo in Ehrlich’s ascites carcinoma [101] and in vitro in AGS gastric cancer cells [102], as well as in androgen-independent PC-3 and androgen-sensitive LNCaP prostate cancer cells [103]. Contrarily to afzelin, kapinnatoside (31) is an uncommon kaempferol derivative, which bears the same glycosylation pattern of QAR. It has also shown antileishmanial activity, but here again, the effect was weaker than that of its quercetin counterpart (QAR) [59].
3. Why Choose Kalanchoe? Singular Aspects of the Genus as a Source of Bioactive Compounds
After presenting the detailed aspects of the main bioactive compounds detected in Kalanchoe species, in this section, we will present the main reasons why plants of this genus can be considered a prominent source of bioactive molecules for drug development.
3.1. Established Uses and Traditional Knowledge: Kalanchoe Species Are Widely Used around the World
It is a fact that Kalanchoe plants have several medicinal uses around the world. Species of this genus are applied to treat a wide range of diseases and heath conditions. The uses of Kalanchoe plants include mostly infusions, cataplasms, juices, or even compresses from crushed leaves. In addition, some Kalanchoe species are also consumed as salads [1,39,104]. Kalanchoe species are intensively cultivated in home gardens, and are also found in medicinal herb markets, which can also explain their large use [1].
Among the several medicinal Kalanchoe species, the most used in traditional medicine is K. pinnata. Undoubtedly, this fact justifies the greater interest of researchers and, consequently, the greatest number of studies of this medicinal species. For the treatment of wounds, people use local leaf compresses, while oral administration is used for treating other health problems. Some Kalanchoe species have had their pharmacological properties evidenced in in vitro studies and even in animal models, reinforcing the traditional knowledge.
While Kalanchoe plants have been widely used in traditional medicine in India, Sri Lanka, Pakistan, China, African countries, and Central and South America for a long time [48,105,106,107,108,109], their use in Europe is more restricted to anthroposophic medicine. K. pinnata and K. daigremontiana (B. pinnatum and B. daigremontianum) are used in this alternative medicinal system, and both species are listed in the German Homeopathic Pharmacopoeia (HAB) 2014 [6]. In particular, K. pinnata has been prescribed by practitioners of anthroposophic medicine for a wide range of health problems such as insomnia and emotional disturbances, and to prevent premature labor since 1970 [6,110]. Recently, this practice has also been adopted in perinatal centers in Switzerland [6,111,112,113], which is important, because new tocolytic agents without side effects are strongly needed. For this reason, we can observe significant advances in preclinical and clinical studies of K. pinnata extract regarding its tocolytic effect [35,114,115,116]. In vitro studies have shown that flavonoids and especially bufadienolides are implicated in the tocolytic effect of K. pinnata, as mentioned in Section 2.1 [34,35].
K. pinnata also plays a prominent role as a medicinal plant in Brazil due to its range of therapeutic properties. The species was included in a list of 71 medicinal plants elaborated by the Brazilian Ministry of Health in 2009: the National List of Plants of Interest to the Unified Health System (RENISUS) [8]. This list aims to guide studies and research on medicinal plants already used in the Brazilian primary care of the Unified Health System (Sistema Único de Saúde–SUS). This Brazilian Health policy intends to provide advances in knowledge of their safety and efficacy, as well as to promote the development of herbal medicines by the pharmaceutical sector. In fact, besides the intensive popular use, there is already a great amount of knowledge about the therapeutic properties of K. pinnata, as well as about the mechanisms of pharmacological action in several targets, and the safety profile of extracts and formulations [15,61,62,68,117,118,119], which can, at least partially, explain the interest in this species by the Brazilian government. The set of pharmacological data about K. pinnata shows the potential of this medicinal plant for the development of phytomedicines as a strategic source of new bioactive substances.
3.2. Presence of Unusual Compounds: Kalanchoe Is a Source of Rare Compounds in Nature
As discussed throughout this review, the pharmacological activities of Kalanchoe species are mainly attributed to flavonoids and bufadienolides [1,6,12] (Table 1 and Table 2). Bufadienolides are compounds with limited distribution among plants [12,120]. Thus, not surprisingly, many of the bioactive bufadienolides reported in Kalanchoe were, up to now, described exclusively in species belonging to the genus, according to Scifinder and PubChem databases. This is the case of kalanchosides A-C (1–3), bryophyllins A and B (7 and 8), daigremontianin (10), kalantubosides A and B (11 and 12), daigredorigenin-3-acetate (15), bryophyllin C (16), and bryotoxin B (18).
On the other hand, flavonoids are secondary metabolites with a wide occurrence in plant kingdom, as previously mentioned. As discussed here, several studies on different Kalanchoe species showed that flavonoids (mainly quercetin skeleton), usually glycosylated at positions 3 and/or 7 with glucose and rhamnose residues, play an important role on several therapeutic targets (Table 2, Section 2.2). Among the bioactive flavonoids of Kalanchoe species (Table 2), most of them occur in other botanical genera, in greater or lesser amounts (e.g., quercetin, rutin, quercitrin, isoquercitrin, miquelianin, quercetin-3-O-sophoroside, 3′,4′-dimethoxy quercetin, kaempferol, afzelin, astragalin, and α-rhamnoisorobin). However, some other flavonoids seem to be restricted to this genus or occur in a greater amount in Kalanchoe plants when compared with other species, at least according to the information found in the literature so far.
Glycosylation of common aglycones with diverse sugar moieties through different inter-glycosidic linkages increases the variety and complexity of glycosylated flavonoid structures. Therefore, from a same aglycone, a less common flavonoid can be obtained depending on the uniqueness of the sugar in the glycosylation and, if there is a second or a third sugar in the structure, on the type of inter-glycosidic bond between these units. For this reason, several bioactive flavonoids found in Kalanchoe species have a more restricted occurrence, and some of them can be considered rare, especially when pentose units (xylose and arabinose) are the sugars linked to the aglycone. This is the case of QAR (quercetin 3-O-α-L-arabinopyranoside-(1→2)α-L-rhamnopyranoside—26), the most abundant flavonoid in aqueous extracts from K. pinnata with several in vitro and in vivo biological activities [15,60,68,69] (see Section 2.2).
Although QAR has a quercetin skeleton, which is quite common, the glycosylation pattern with an arabinosyl residue at position 3 of this flavonoid could explain its sparse frequency in nature. This glycosylation motif (3-O-arabinosyl) in a flavonol is less frequent than the glycosylation with glucosyl or rhamnosyl moieties. In fact, the enzyme flavonol-3-O-arabinosyltransferase contributes less to the glycosylation of flavonols than 3-O-glucosyl and 3-O-rhamnosyltransferases, as reported in Arabidopsis thaliana leaves [121]. This rare flavonoid has been reported in a few other plant species such as Alphitonia philippinensis (Rhamnaceae), Zizyphus jujuba (Rhamnaceae), and Rollinia emarginata (Annonaceae) [88,89,122]. As discussed previously here, the restricted occurrence in the plant kingdom and its relative abundance in K. pinnata make this flavonoid a good chemical marker for this plant species with high therapeutic potential [66].
A new and minoritarian kaempferol diglycoside–kapinnatoside (31), bearing the same carbohydrate moieties at the 3 position as in QAR, was also isolated from K. pinnata, along with another unusual flavone glycoside, 4′,5-dihydroxy-3′,8-dimethoxyflavone 7-O-beta-D-glucopyranoside [66]. Both compounds are also not commonly found in other plant species.
Besides flavonoids and bufadienolides, there are other molecules that can be cited as unusual in Kalanchoe. For instance, a glycosylated aryltetralin lignan presenting an unprecedented structure and exhibiting inhibitory activity against HSV-1 and vaccinia viruses was reported in K. pinnata [123,124]. The isolation of this polyphenol in this species seems to be the only report in the genus, although lignans have been detected in the aqueous extract of K. gastonis-bonnieri, potentially useful to treat primary benign prostatic hyperplasia [125]. Lignans are more common in Rhodiola species, another medicinal genus of Crassulaceae [126].
3.3. Kalanchoe Plants: The “Mother of Thousands” Genus Is Becoming Biotechnological
About 25% of the annual global market of medicines is based on plant molecules [127], which stimulates scientists to research and describe new bioactive ingredients obtained from this natural source [128,129]. To successfully develop phytomedicines, it is necessary not only to choose the plant source based on its activity (supported by the traditional use and scientific research) but also based on the easiness of access to the plant material. In fact, accessing sufficient material to isolate, characterize, and apply the bioactive compounds is challenging [130]. Nowadays, diverse biotechnological and cultivation techniques are being developed and applied to ensure the access to the raw material, as well as the best ways to produce, extract, and isolate their bioactive ingredients [130]. Regarding the cultivation and biotechnological knowledge for triggering the production of bioactive compounds, Kalanchoe plants are moving to promising places.
Apart from the sexual reproduction commonly found in superior plants (by means of flower production, cross-pollination, and seed dispersal), Kalanchoe has evolved a wide range of asexual reproductive strategies, mainly based on both organogenesis and embryogenesis processes localized on the leaf margin [131]. These leaf areas commonly have buds, which contribute to the easiness and wide spreading of Kalanchoe species in several regions, mainly in mild-climate parts of the world. Even the name of the genus refers to its facility of propagation, representing the phonetic transcription of the term in Chinese “Kalan Chauhuy” that means “which falls and grows” [132]. Indeed, Kalanchoe plants are often propagated from seed, leaf-cuttings, buds, and even from detached/injured leaves [132]. Plants of these genus also require low agricultural input for their cultivation, needing neither a specific type of soil nor high water availability [132]. In addition, as succulents, Kalanchoe plants are able to survive for long periods under environmental stresses, by accumulating water in their leaves and by producing secondary metabolites that contribute as antioxidants, radiation-protectors, and/or light-catchers [20,132,133,134,135].
Some genetic studies and breeding programs have been developed with Kalanchoe plants in the recent years [132,136]. It can be attributed to the horticultural importance of some species of the genus (e.g., K. blossfeldiana) and to the interest of plant physiologists in understanding and manipulating the Crassulacean Acid Metabolism (CAM) as well as its asexual reproduction [137,138]. The scientists are also excited to understand the evolutionary relationship between the different species of the genus. As a matter of fact, DNA markers of K. blossfeldiana genotypes have been recently assessed [139], allowing the selection of varieties to be used in breeding strategies. Moreover, the complete plastomes (chloroplast genomes) of five horticultural Kalanchoe species, K. daigremontiana, K. delagoensis K. fedtschenkoi, K. longiflora, and K. pinnata, have also been recently assembled [140]. In addition, the genome of K. fedtschenkoi, a species intensively used as ornamental around the world and known as a CAM model, can be currently found in the NCBI database (https://www.ncbi.nlm.nih.gov/assembly/GCA_002312845.1/ Last accessed on 25 February 2023). All these aspects allow biotechnological approaches with plants of this genus. Additionally, regarding breeding programs, new cultivars with different characteristics of flower aspects (such as color, number and shape) are being created not only by means of conventional breeding, but also by using biotechnological methods. For example, gene transformation methods have been used in Kalanchoe, producing plants with shorter internodes, longer-lasting flowers, and late blooming (for a deeper reading, see [141]).
Nevertheless, the knowledge and interest in the genome of Kalanchoe plants can also be attributed to the medicinal and commercial interest in them. In fact, understanding the enzymes involved in the biosynthesis pathway of bioactive compounds as well as the genes implicated in their biosynthesis opens new opportunities to obtain bioactive molecules at a large scale. Several recent studies have described how biotechnological approaches can be applied to increase the content of secondary metabolites in plants (in situ) or how plant cells, tissues, and organelles, as well as microorganisms (e.g., Escherichia coli), can be used as platforms to produce these compounds in vitro [142]. Indeed, in vitro cultures of Kalanchoe plants are well established. Nutrient requirements, disinfection and micropropagation protocols, as well as the establishment of suspension-cultured cells, are well known for several Kalanchoe species, being recently revised by García-Pérez et al. [143].
The progressive development of biotechnology led to the expansion of new strategies to produce phytopharmaceuticals and herbal medicines, creating alternatives for obtaining and producing bioactive substances [144]. In this context, plant biotechnology offers possibilities to change, in an innovative way, the metabolism of plants, increasing the production of prominent chemicals, and producing them in cells and tissues of other species [144]. Some of the main techniques used to obtain higher contents of bioactive molecules are DNA manipulation and metabolic engineering. For Kalanchoe plants, Fujimoto and co-workers produced novel colored-leaf plants of K. blossfeldiana by the ectopic expression of R2R3 MYB genes from Tricyrtis sp. and Arabidopsis thaliana, which activated the flavonoid biosynthetic pathway and led to anthocyanin accumulation [137].
Considering the most prominent bioactive molecules in Kalanchoe, flavonoids have more studies aiming at increasing yield or at reconstructing their biosynthetic pathways, if compared with bufadienolides. These works are interested in scaling up the obtaining of flavonoids under controlled conditions, by means of cell suspension cultures, hairy root cultures, transgenics, and microbial hosts utilization [145,146,147]. The increasing understanding of biosynthetic pathways and molecular genetics of different plants led to the higher application of metabolic engineering to enhance the production of compounds in plants and microorganisms. These achievements, however, need complete information about the biosynthetic pathway of the desired compound. The absence of such data is a barrier for several plant species [148], including Kalanchoe. Therefore, genomics, proteomics, and metabolomics studies in Kalanchoe plants are still needed and must be stimulated.
If on one hand the genetic engineering is still little applied for Kalanchoe plants, on the other hand, the elicitation process of the bioactive compounds in plants of the genus has been more employed and is a topic of interest in recent research. Indeed, different environmental factors can trigger the production of bioactive compounds [149], being biotechnologically applied in the development of products, including herbal and phytopharmaceuticals. For Kalanchoe, several elicitors have been studied and used for the enhancement of flavonoid production. In this sense, our group was the pioneer in the application of different light qualities on Kalanchoe. In 2010, Leal-Costa and col. demonstrated that the supplementation of blue light in the in vitro cultivation of K. pinnata increased the phenolic idioblasts in leaf tissues, suggesting that this light quality might stimulate the accumulation of these compounds [150]. In fact, few years later, Nascimento et al. [19] showed the effects not only of blue light but also of UV-A radiation in the enhancement of phenolics and flavonoids content as well as the antioxidant activity of K. pinnata extracts. The same authors also showed the elicitation of quercitrin and other flavonoids production by UV-B radiation in this same species [20]. Later, UV-B radiation was also used as elicitation strategy for the enhancement of bioactive compounds in K. pinnata leaves by another group, together with the biotic elicitation through natural transformation of the plants by Agrobacterium rhizogenes [151]. The authors observed that the combination of both factors resulted in an increase of more than 100% of the total flavonoid content [151]. Lastly, nutrient stress, salicylic acid, and cyclodextrin have also been applied as elicitors in Kalanchoe plants and cell suspension cultures, with the prediction of the best conditions by using new tools of machine learning [152,153,154].
4. Gaps of Knowledge and Challenges for Drug Development with Kalanchoe Compounds
Despite presenting all the potential characteristics that make Kalanchoe plants (and especially K. pinnata) important sources of bioactive molecules, some gaps of knowledge, challenges, and limitations must be presented. These are specially based on the few in vivo studies conducted with the bioactive compounds, as well as the lack of evaluation of their action mechanisms and possible toxicity. In addition, concerns about the bioavailability of these compounds and the available technological solutions ought to be mentioned. In this section, a brief critical analysis of these aspects is presented in order to contribute to future research in the field.
Generally, studies investigating bioactive substances in medicinal plants begin by demonstrating the pharmacological effects of the extracts, using in vitro and in vivo assays. While demonstrating the pharmacological activity of a plant extract is relatively simple, identifying the key constituents for this observed pharmacological effect is a challenge for researchers. The bioassay-guided fractionation of extracts focusing on a given pharmacological activity has been used as a strategy to recognize the bioactive substances, among other possibilities. Once this stage has been reached, it is essential to understand how these substances act on a given pharmacological target.
Even though the pharmacological potential of Kalanchoe flavonoids and bufadienolides has been evidenced by multiple studies, there is still a long way to go. For instance, only a limited number of these substances have been the object of further investigation to determine their modes of action. This is the case of the bufadienolide bryophyllin A and kalantubosides A and B, that have had their cytotoxic mechanisms assessed (Section 2.1). Regarding flavonoids, quercetin and QAR stand out due to their more in-depth mode of action studies (Section 2.2). A thorough understanding of the targets and mechanisms of action of other flavonoids and bufadienolides from Kalanchoe would be essential for exploring their pharmacological potential.
Regarding the type of studies conducted with Kalanchoe species, we can observe that most of them are in vitro assays, while in vivo studies are scarce (Table 1 and Table 2). Even though various studies have been carried out to evaluate the antitumor potential of Kalanchoe bufadienolides in multiple cell lineages, only one in vivo study was retrieved in the literature [29]. The same is true for the other pharmacological activities reported for these bufadienolides. There are comparatively more in vivo studies for flavonoids, but they represent only a small fraction of the research works.
Considering the use of K. pinnata in the treatment of wounds and inflammatory processes, there has been an important advance in the knowledge of the pharmacological role of QAR proven by in vivo assays. The antileishmanial, anti-inflammatory, antinociceptive, antiedematogenic, wound healing, and gastroprotective effects of this flavonoid were demonstrated in mice and rats [15,59,60,68,69], although there is still much to be done to clarify the intricacies of its in vivo behavior. For example, the immunomodulatory profile of QAR deserves to be further investigated considering other therapeutic targets in which the inflammatory process plays an important role. Moreover, for many other Kalanchoe bioactive flavonoids, there are no in vivo studies. The in vivo studies are, undoubtedly, a very important step to assess the potential of drug candidates. Promising in vitro results do not always translate into the desired in vivo effects, since the metabolism of bioactive substances is not taken into account in in vitro assays [155,156,157].
Therefore, despite the greater complexity and cost of in vivo assays, as well as the necessity to abide to stringent rules imposed by ethical considerations and regulations, animal models are still essential for the evaluation and validation of drugs [156]. In fact, assays with different animal species are required by regulatory agencies, such as the Food and Drug Administration (FDA, USA), the European Medicine Agency (EMA), and the Brazilian Health Regulatory Agency (ANVISA), before human trials are considered [156,158]. It is also important to mention that animal models frequently do not adequately predict the effects of a drug in humans. Technologies such as organ-on chip and microfluidic integrated systems, based on human cell cultures, are promising alternatives to assess the effects of bioactive compounds in complex systems [159] and could also be applied in the future for the study of flavonoids and bufadienolides from Kalanchoe.
Another important and often overlooked aspect regarding Kalanchoe bioactive compounds is toxicity. This is a concern especially for bufadienolides, which have been associated with cases of livestock intoxication after the ingestion of Kalanchoe plants [1,160,161]. The majority of studies with bufadienolides from Kalanchoe species are about their cytotoxicity against cancer cell lines; however, only in a few of them were assays also carried out with normal cell lines to compare the selectivity of the effects [25,29]. Thus, the potential toxicity risks of these bufadienolides are poorly understood.
Concerning flavonoids, even though this class of compounds has been extensively studied, toxicological assessments seem to represent just a small portion of this research. Most of the available data concerns flavonoids with a wide distribution among plants. This is the case of quercetin and kaempferol, both found in Kalanchoe. These flavonol aglycones have had their toxic effects assessed in multiple in vitro and in vivo studies. Although some in vitro assays point out the possible toxicity mainly due to prooxidant activity, these effects have not been confirmed in vivo. In most of these in vivo studies, quercetin and kaempferol have shown low toxicity [162,163]. On the other hand, data on the toxicity of flavonol glycosides are scarce. Many of these compounds are present in food (beverages, fruits, and vegetables) and nutritional studies have evidenced that their consumption is safe [164,165]. In fact, in vitro studies carried out by our group have shown that flavonoids from K. pinnata have low or even no cytotoxicity in normal cell lines [16,66]. However, studies to specifically assess the toxicological profile of Kalanchoe flavonol glycosides are important if these compounds are to be considered candidates for drug development.
We must also consider that bioactive substances belonging to the class of secondary metabolites are not synthesized on a large scale by plants. Their low production in plants is a challenge for the processes of extracting and purifying these compounds in order to obtain sufficient amounts to carry out pharmacological tests, especially in vivo, whether for assessing toxicity or for studying mechanisms of action in preclinical research [156]. This partially justifies the lack of studies focusing on the toxicity of the most bioactive substances already described for the Kalanchoe genus.
Another aspect that deserves attention is the bioavailability of the bioactive compounds. This is one of the main reasons for the failure of bioactive molecules evaluated in complex organisms, such as animal models and human individuals. In particular, bioactive substances in their original structures, although effective in the studied animal models, need to present good properties of absorption, distribution, metabolism, excretion, and low toxicity. If they do not meet these requirements, it will certainly be necessary to invest in technological formulations and/or chemical modifications, which will increase costs and time for the development of a new drug.
Bufadienolides, especially in the form of aglycones, are poorly soluble in water and have a low bioavailability [166]. Recently, Shao et al. [166] reviewed strategies to enhance these parameters, such as structural modifications, cyclodextrin inclusion, and nanodrug delivery systems. The latter were considered the most promising. Studies to better understand the bioavailability of Kalanchoe bufadienolides and possible strategies to improve their bioavailability are essential for their future development as drugs. Oral bioavailability is also a concern for flavonoids. It is known that flavonoid O-glycosides, which are major in Kalanchoe species, are hydrolyzed by enzymes from the intestinal tract and from the colonic microbiota, generating their aglycone counterparts, which diffuse through the intestinal epithelium [167,168]. This can represent a major drawback for glycosylated flavonoids that have shown in vitro effects. It is therefore important to evaluate the effect of their aglycone forms and/or their in vivo effects. Flavonoids also undergo hepatic metabolism, creating conjugated compounds, mostly glucuronides and sulfates. Muzitano et al. [44] evidenced that QAR and quercitrin from K. pinnata, after oral intake by mice, have quercetin and quercetin glucuronide as the main plasmatic metabolites. The gut microbiota can also extensively transform flavonoids, creating phenolic acid derivatives [167,168,169]. The bioavailability of Kalanchoe flavonoids as well as the possible effects of their metabolites are important objects for future studies. Bioavailability issues can be mitigated using multiple strategies. Examples include permeation enhancers, development of prodrugs, and nanotechnology-based formulations [167]. This topic has been extensively reviewed elsewhere [165,167,168,170,171].
Notwithstanding, the process of drug development based on bioactive substances of natural origin also requires the collaboration between the academic research sector and pharmaceutical industries, which constitutes an enormous challenge. An additional challenge is the transfer of knowledge generated in academia to the industrial sector, including the registration of intellectual property rights.
5. Final Considerations and Perspectives
We presented here an up to date and critical review of the most prominent bioactive compounds found in Kalanchoe species. Some bioactive substances present in this genus, especially in K. pinnata, the most studied species, are interesting from a pharmacological point of view. These compounds could be applied isolated as drugs, inspire new drugs, or even constitute part of their original plant extracts, composing a phytomedicine. In this case, knowledge of the bioactive compounds could guide and ensure the quality and efficacy of these medicines, an aspect required by some regulatory agencies worldwide.
Kalanchoe species are mainly a source of bufadienolides, with antitumor and muscle relaxing activities, and flavonoids, especially recognized as anti-inflammatory and wound healing agents. Among the bioactive molecules of Kalanchoe, the flavonoid quercetin 3-O-α-L-arabinopyranosyl (1→2) α-L-rhamnopyranoside (QAR), a major compound from K. pinnata, stands out compared to all other molecules. This flavonoid has a wide range of pharmacological evidence, including the reduction of inflammatory processes, wound healing action, and protection against gastric ulcers, all of these already proven in animal models. QAR gathers promising evidence that makes this flavonoid an excellent drug candidate for the therapy of these conditions. Moreover, being an unusual molecule present in a high quantity, QAR can be considered as the chemical and the bioactive marker of extracts and products prepared with K. pinnata. A significant part of the preclinical research with this flavonoid has already been carried out, but there are still important gaps to be overcome. Furthermore, taking into account the large content of QAR in the plant and the knowledge and the biotechnological tools available for the production and extraction of this molecule [20,21], the sustainable access to QAR in a sufficient quantity to develop clinical studies does not seem to be difficult to achieve.
Except for QAR, most of the other bioactive molecules described in Kalanchoe were only tested by in vitro assays. The few studies focusing on their in vivo effect, mode of action, and toxicity are gaps of knowledge in this field. Thus, these aspects should be considered in further studies with plants of this genus. In summary, considering the richness in bioactive compounds (some of them uncommon in nature), the easiness of propagation and cultivation of Kalanchoe, and the new biotechnological aspects concerning these plants, the development of new drugs with Kalanchoe species can be envisaged in the near future.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Data Availability Statement
This review generated no new data. All consulted data are available on cited literature.
Acknowledgments
The authors sincerely express their gratitude to all researchers, especially from our Brazilian research group, that have contributed to the knowledge regarding the therapeutic potential of Kalanchoe species in the last 30 years, by the incessant conduction of quality studies with seriousness and commitment.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Costa, S.S.; Muzitano, M.F.; Camargo, L.M.M.; Coutinho, M.A.S. Therapeutic Potential of Kalanchoe Species: Flavonoids and Other Secondary Metabolites. Nat. Prod. Commun. 2008, 3, 2151–2164. [Google Scholar] [CrossRef]
- Eggli, U. Illustrated Handbook of Succulent Plants: Crassulaceae; Springer: Berlin, Heidelberg, 2003; ISBN 978-3-642-62629-6. [Google Scholar]
- Stevens, P.F. Angiosperm Phylogeny Website, Version 14. 2017. Available online: http://www.mobot.org/MOBOT/research/APweb/ (accessed on 28 December 2022).
- Boiteau, P.; Alorge-Boiteau, L. Kalanchoe (Crassulaceae) de Madagascar, Systématique, Écophysiology et Phytochimie; Karthala: Paris, France, 1995. [Google Scholar]
- Wang, C.-Y.; Huang, S.-C.; Zhang, Y.; Lai, Z.-R.; Kung, S.-H.; Chang, Y.-S.; Lin, C.-W. Antiviral Ability of Kalanchoe gracilis Leaf Extract against Enterovirus 71 and Coxsackievirus A16. Evid. Based Complementary Altern. Med. 2012, 2012, 503165. [Google Scholar] [CrossRef]
- Fürer, K.; Simões-Wüst, A.; von Mandach, U.; Hamburger, M.; Potterat, O. Bryophyllum pinnatum and Related Species Used in Anthroposophic Medicine: Constituents, Pharmacological Activities, and Clinical Efficacy. Planta Med. 2016, 82, 930–941. [Google Scholar] [CrossRef] [PubMed]
- Biswas, K. Literature Review on Pharmacological Potentials of Kalanchoe pinnata (Crassulaceae). Afr. J. Pharm. Pharmacol. 2011, 5, 1258–1262. [Google Scholar] [CrossRef]
- Ministério da Saúde RENISUS—Relação Nacional de Plantas Medicinais de Interesse ao SUS 2009. Available online: https://www.gov.br/saude/pt-br/composicao/sectics/daf/pnpmf/ppnpmf/arquivos/2014/renisus.pdf (accessed on 23 December 2022).
- Fernandes, J.M.; Cunha, L.M.; Azevedo, E.P.; Lourenço, E.M.G.; Fernandes-Pedrosa, M.F.; Zucolotto, S.M. Kalanchoe laciniata and Bryophyllum pinnatum: An Updated Review about Ethnopharmacology, Phytochemistry, Pharmacology and Toxicology. Braz. J. Pharmacog. 2019, 29, 529–558. [Google Scholar] [CrossRef]
- Hsieh, Y.-J.; Yang, M.-Y.; Leu, Y.-L.; Chen, C.; Wan, C.-F.; Chang, M.-Y.; Chang, C.-J. Kalanchoe tubiflora Extract Inhibits Cell Proliferation by Affecting the Mitotic Apparatus. BMC Complement Altern. Med. 2012, 12, 149. [Google Scholar] [CrossRef] [PubMed]
- Ürményi, F.G.G.; Saraiva, G.D.N.; Casanova, L.M.; Matos, A.D.S.; de Magalhães Camargo, L.M.; Romanos, M.T.V.; Costa, S.S. Anti-HSV-1 and HSV-2 Flavonoids and a New Kaempferol Triglycoside from the Medicinal Plant Kalanchoe daigremontiana. Chem. Biodivers. 2016, 13, 1707–1714. [Google Scholar] [CrossRef]
- Kolodziejczyk-Czepas, J.; Stochmal, A. Bufadienolides of Kalanchoe Species: An Overview of Chemical Structure, Biological Activity and Prospects for Pharmacological Use. Phytochem. Rev. 2017, 16, 1155–1171. [Google Scholar] [CrossRef] [PubMed]
- Da Silva, S.A.G.; Costa, S.S.; Mendonça, S.C.F.; Silva, E.M.; Moraes, V.L.G.; Rossi-Bergmann, B. Therapeutic Effect of Oral Kalanchoe pinnata Leaf Extract in Murine Leishmaniasis. Acta Tropica 1995, 60, 201–210. [Google Scholar] [CrossRef] [PubMed]
- Rossi-Bergmann, B.; Costa, S.S.; Borges, M.B.S.; da Silva, S.A.; Noleto, G.R.; Souza, M.L.M.; Moraes, V.L.G. Immunosuppressive Effect of the Aqueous Extract of Kalanchoe pinnata in Mice. Phytother. Res. 1994, 8, 399–402. [Google Scholar] [CrossRef]
- Ferreira, R.T.; Coutinho, M.A.S.; Malvar, D.D.C.; Costa, E.A.; Florentino, I.F.; Costa, S.S.; Vanderlinde, F.A. Mechanisms Underlying the Antinociceptive, Antiedematogenic, and Anti-Inflammatory Activity of the Main Flavonoid from Kalanchoe pinnata. Evid. Based Complement. Altern. Med. 2014, 2014, 429256. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, M.A.S.; Muzitano, M.F.; Cruz, E.A.; Bergonzi, M.C.; Kaiser, C.R.; Tinoco, L.W.; Bilia, A.R.; Vincieri, F.F.; Rossi-Bergmann, B.; Costa, S.S. Flowers from Kalanchoe pinnata Are a Rich Source of T Cell-Suppressive Flavonoids. Nat. Prod. Commun. 2012, 7, 175–178. [Google Scholar] [CrossRef]
- Costa, S.S.; Corrêa, M.F.P.; Casanova, L.M. A New Triglycosyl Flavonoid Isolated from Leaf Juice of Kalanchoe gastonis-bonnieri (Crassulaceae). Nat. Prod. Commun. 2015, 10, 433–436. [Google Scholar] [CrossRef] [PubMed]
- Costa, S.S.; Jossang, A.; Bodo, B.; Souza, M.L.M.; Moraes, V.L.G. Patuletin Acetylrhamnosides from Kalanchoe brasiliensis as Inhibitors of Human Lymphocyte Proliferative Activity. J. Nat. Prod. 1994, 57, 1503–1510. [Google Scholar] [CrossRef]
- Nascimento, L.B.S.; Leal-Costa, M.V.; Coutinho, M.A.S.; Moreira, N.D.S.; Lage, C.L.S.; Barbi, N.D.S.; Costa, S.S.; Tavares, E.S. Increased Antioxidant Activity and Changes in Phenolic Profile of Kalanchoe pinnata (Lamarck) Persoon (Crassulaceae) Specimens Grown Under Supplemental Blue Light. Photochem. Photobiol. 2013, 89, 391–399. [Google Scholar] [CrossRef]
- Dos Santos Nascimento, L.B.; Leal-Costa, M.V.; Menezes, E.A.; Lopes, V.R.; Muzitano, M.F.; Costa, S.S.; Tavares, E.S. Ultraviolet-B Radiation Effects on Phenolic Profile and Flavonoid Content of Kalanchoe pinnata. J. Photochem. Photobiol. B Biol. 2015, 148, 73–81. [Google Scholar] [CrossRef]
- Dos Santos Nascimento, L.B.; Aguiar, P.F.; Leal-Costa, M.V.; Coutinho, M.A.S.; Borsodi, M.P.G.; Rossi-Bergmann, B.; Tavares, E.S.; Costa, S.S. Optimization of Aqueous Extraction from Kalanchoe pinnata Leaves to Obtain the Highest Content of an Anti-Inflammatory Flavonoid Using a Response Surface Model: Optimization of a Bioactive Flavonoid Extraction from K. pinnata. Phytochem. Anal. 2018, 29, 308–315. [Google Scholar] [CrossRef] [PubMed]
- Gao, H.; Popescu, R.; Kopp, B.; Wang, Z. Bufadienolides and Their Antitumor Activity. Nat. Prod. Rep. 2011, 28, 953–969. [Google Scholar] [CrossRef]
- Yamagishi, T.; Haruna, M.; Yan, X.-Z.; Chang, J.-J.; Lee, K.-H. Antitumor Agents, 110, Bryophyllin B, a Novel Potent Cytotoxic Bufadienolide from Bryophyllum pinnatum. J. Nat. Prod. 1989, 52, 1071–1079. [Google Scholar] [CrossRef] [PubMed]
- Kuo, P.-C.; Kuo, T.-H.; Su, C.-R.; Liou, M.-J.; Wu, T.-S. Cytotoxic Principles and α-Pyrone Ring-Opening Derivatives of Bufadienolides from Kalanchoe hybrida. Tetrahedron 2008, 64, 3392–3396. [Google Scholar] [CrossRef]
- Stefanowicz-Hajduk, J.; Asztemborska, M.; Krauze-Baranowska, M.; Godlewska, S.; Gucwa, M.; Moniuszko-Szajwaj, B.; Stochmal, A.; Ochocka, J.R. Identification of Flavonoids and Bufadienolides and Cytotoxic Effects of Kalanchoe daigremontiana Extracts on Human Cancer Cell Lines. Planta Med. 2020, 86, 239–246. [Google Scholar] [CrossRef]
- Wu, P.-L.; Hsu, Y.-L.; Wu, T.-S.; Bastow, K.F.; Lee, K.-H. Kalanchosides A−C, New Cytotoxic Bufadienolides from the Aerial Parts of Kalanchoe gracilis. Org. Lett. 2006, 8, 5207–5210. [Google Scholar] [CrossRef]
- Stefanowicz-Hajduk, J.; Gucwa, M.; Moniuszko-Szajwaj, B.; Stochmal, A.; Kawiak, A.; Ochocka, J.R. Bersaldegenin-1,3,5-Orthoacetate Induces Caspase-Independent Cell Death, DNA Damage and Cell Cycle Arrest in Human Cervical Cancer HeLa Cells. Pharm. Biol. 2021, 59, 54–65. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-C.; Chang, W.-T.; Lee, M.-S.; Chen, H.-Y.; Chen, Y.-H.; Lin, C.-C.; Lin, M.-K. Three Bufadienolides Induce Cell Death in the Human Lung Cancer Cell Line CL1-5 Mainly through Autophagy. Bioorg. Med. Chem. Lett. 2021, 31, 127715. [Google Scholar] [CrossRef] [PubMed]
- Hseu, Y.-C.; Cho, H.-J.; Gowrisankar, Y.V.; Thiyagarajan, V.; Chen, X.-Z.; Lin, K.-Y.; Huang, H.-C.; Yang, H.-L. Kalantuboside B Induced Apoptosis and Cytoprotective Autophagy in Human Melanoma A2058 Cells: An in Vitro and in Vivo Study. Free Radic. Biol. Med. 2019, 143, 397–411. [Google Scholar] [CrossRef] [PubMed]
- Supratman, U.; Fujita, T.; Akiyama, K.; Hayashi, H.; Murakami, A.; Sakai, H.; Koshimizu, K.; Ohigashi, H. Anti-Tumor Promoting Activity of Bufadienolides from Kalanchoe pinnata and K. daigremontiana × tubiflora. Biosci. Biotechnol. Biochem. 2001, 65, 947–949. [Google Scholar] [CrossRef] [PubMed]
- Huang, H.-C.; Lin, M.-K.; Yang, H.-L.; Hseu, Y.-C.; Liaw, C.-C.; Tseng, Y.-H.; Tsuzuki, M.; Kuo, Y.-H. Cardenolides and Bufadienolide Glycosides from Kalanchoe tubiflora and Evaluation of Cytotoxicity. Planta Med. 2013, 79, 1362–1369. [Google Scholar] [CrossRef] [PubMed]
- Yamagashi, T.; Yan, X.-Z.; Wu, R.-Y.; Mcphail, D.R.; Mcphail, A.T.; Lee, K.-H. Structure and Stereochemistry of Bryophyllin-A, a Novel Potent Cytotoxic Bufadienolide Orthoacetate from Bryophyllum pinnatum. Chem. Pharm. Bull. 1988, 36, 1615–1617. [Google Scholar] [CrossRef]
- Bachmann, S.; Betschart, C.; Gerber, J.; Fürer, K.; Mennet, M.; Hamburger, M.; Potterat, O.; von Mandach, U.; Simões-Wüst, A. Potential of Bryophyllum pinnatum as a Detrusor Relaxant: An in Vitro Exploratory Study. Planta Med. 2017, 83, 1274–1280. [Google Scholar] [CrossRef] [PubMed]
- Santos, S.; Haslinger, C.; Klaic, K.; Faleschini, M.T.; Mennet, M.; Potterat, O.; von Mandach, U.; Hamburger, M.; Simões-Wüst, A.P. A Bufadienolide-Enriched Fraction of Bryophyllum pinnatum Inhibits Human Myometrial Contractility In Vitro. Planta Med. 2019, 85, 385–393. [Google Scholar] [CrossRef]
- Santos, S.; Zurfluh, L.; Mennet, M.; Potterat, O.; von Mandach, U.; Hamburger, M.; Simões-Wüst, A.P. Bryophyllum pinnatum Compounds Inhibit Oxytocin-Induced Signaling Pathways in Human Myometrial Cells. Front. Pharmacol. 2021, 12, 632986. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejczyk-Czepas, J.; Sieradzka, M.; Moniuszko-Szajwaj, B.; Pecio, Ł.; Ponczek, M.B.; Nowak, P.; Stochmal, A. Bufadienolides from Kalanchoe daigremontiana as Thrombin Inhibitors—In Vitro and in Silico Study. Int. J. Biol. Macromol. 2017, 99, 141–150. [Google Scholar] [CrossRef] [PubMed]
- Kolodziejczyk-Czepas, J.; Pasiński, B.; Ponczek, M.B.; Moniuszko-Szajwaj, B.; Kowalczyk, M.; Pecio, Ł.; Nowak, P.; Stochmal, A. Bufadienolides from Kalanchoe daigremontiana Modulate the Enzymatic Activity of Plasmin—In Vitro and in Silico Analyses. Int. J. Biol. Macromol. 2018, 120, 1591–1600. [Google Scholar] [CrossRef] [PubMed]
- Tohge, T.; de Souza, L.P.; Fernie, A.R. Fernie On the Natural Diversity of Phenylacylated-Flavonoid and Their in Planta Function under Conditions of Stress. Phytochem. Rev. 2018, 17, 279–290. [Google Scholar] [CrossRef]
- Milad, R. Genus Kalanchoe (Crassulaceae): A Review of Its Ethnomedicinal, Botanical, Chemical and Pharmacological Properties. Eur. J. Med. Phys. 2014, 4, 86–104. [Google Scholar] [CrossRef]
- Tohge, T.; de Souza, L.P.; Fernie, A.R. Current Understanding of the Pathways of Flavonoid Biosynthesis in Model and Crop Plants. J. Exp. Bot. 2017, 68, 4013–4028. [Google Scholar] [CrossRef] [PubMed]
- Dini, I.; Grumetto, L. Recent Advances in Natural Polyphenol Research. Molecules 2022, 27, 8777. [Google Scholar] [CrossRef]
- Hosseinzade, A.; Sadeghi, O.; Naghdipour Biregani, A.; Soukhtehzari, S.; Brandt, G.S.; Esmaillzadeh, A. Immunomodulatory Effects of Flavonoids: Possible Induction of T CD4+ Regulatory Cells Through Suppression of MTOR Pathway Signaling Activity. Front. Immunol. 2019, 10, 51. [Google Scholar] [CrossRef]
- Gaind, K.N.; Gupta, R.L. Flavonoid Glycosides from Kalanchoe pinnata. Planta Med. 1971, 20, 368–373. [Google Scholar] [CrossRef]
- García-Pérez, P.; Barreal, M.E.; Rojo-De Dios, L.; Cameselle-Teijeiro, J.F.; Gallego, P.P. Bioactive Natural Products From the Genus Kalanchoe as Cancer Chemopreventive Agents: A Review. In Studies in Natural Products Chemistry; Elsevier: Amsterdam, The Netherlands, 2019; Volume 61, ISBN 978-0-444-64183-0. [Google Scholar]
- Pandey, B.; Pradhan, S.P.; Adhikari, K. LC-ESI-QTOF-MS for the Profiling of the Metabolites and in Vitro Enzymes Inhibition Activity of Bryophyllum pinnatum and Oxalis corniculata Collected from Ramechhap District of Nepal. Chem. Biodivers. 2020, 17, e2000155. [Google Scholar] [CrossRef]
- Báez, M.; Torres, E.I.; Gruszycki, A.E.; Alba, D.A.; Valenzuela, M.; Gruszycki, M.R. Actividad antioxidante y antiinflamatoria en extractos hidroalcohólicos de Kalanchoe daigremontiana Raym. -Hamet & H. Perrier. Rev. Colomb. Cienc. Quim. Farm. 2021, 50, 86–99. [Google Scholar] [CrossRef]
- Pereira, K.M.F.; De Carvalho, A.C.; Veiga, T.A.M.; Melgoza, A.; Hernández, R.; dos Santos Grecco, S.; Nakamura, M.U.; Guo, S. The Psychoactive Effects of Bryophyllum pinnatum (Lam.) Oken Leaves in Young Zebrafish. PLoS ONE 2022, 17, e0264987. [Google Scholar] [CrossRef]
- Hewagama, S.P.; Hewawasam, R.P. Antiurolithiatic Potential of Three Sri Lankan Medicinal Plants by the Inhibition of Nucleation, Growth, and Aggregation of Calcium Oxalate Crystals In Vitro. Sci. World J. 2022, 2022, 8657249. [Google Scholar] [CrossRef] [PubMed]
- Sobreira, F.; Hernandes, L.S.; Vetore-Neto, A.; Díaz, I.E.C.; de Santana, F.C.; Mancini-Filho, J.; Bacchi, E.M. Gastroprotective Activity of the Hydroethanolic Extract and Ethyl Acetate Fraction from Kalanchoe pinnata (Lam.) Pers. Braz. J. Pharm. Sci. 2017, 53, e16027. [Google Scholar] [CrossRef]
- Chibli, L.A.; Rodrigues, K.C.M.; Gasparetto, C.M.; Pinto, N.C.C.; Fabri, R.L.; Scio, E.; Alves, M.S.; Del-Vechio-Vieira, G.; Sousa, O.V. Anti-Inflammatory Effects of Bryophyllum pinnatum (Lam.) Oken Ethanol Extract in Acute and Chronic Cutaneous Inflammation. J. Ethnopharmacol. 2014, 154, 330–338. [Google Scholar] [CrossRef] [PubMed]
- Mayorga, O.A.S.; da Costa, Y.F.G.; da Silva, J.B.; Scio, E.; Ferreira, A.L.P.; de Sousa, O.V.; Alves, M.S. Kalanchoe brasiliensis Cambess., a Promising Natural Source of Antioxidant and Antibiotic Agents against Multidrug-Resistant Pathogens for the Treatment of Salmonella Gastroenteritis. Oxid. Med. Cell. Longev. 2019, 2019, 9245951. [Google Scholar] [CrossRef]
- Sobreira, F.; Sobreira, G.; Díaz, I.E.C.; Corazza, F.G.; Santoro, L.H.; Gama, P.; Lourenço, F.R.; Mancini-Filho, J.; dos Santos Ramos, M.A.; Bauab, T.M.; et al. Study of the Gastroprotective Action and Healing Effects of Kalanchoe pinnata (Lam.) against Acidified Ethanol- and Acetic Acid-Induced Gastric Ulcers in Rats. Braz. J. Dev. 2022, 8, 39105–39131. [Google Scholar] [CrossRef]
- Ruiz, G.G.; Nelson, E.O.; Kozin, A.F.; Turner, T.C.; Waters, R.F.; Langland, J.O. A Lack of Bioactive Predictability for Marker Compounds Commonly Used for Herbal Medicine Standardization. PLoS ONE 2016, 11, e0159857. [Google Scholar] [CrossRef]
- Indriyanti, N.; Soeroso, J.; Khotib, J. The Benefits of Active Compounds in Kalanchoe pinnata (Lmk) Ethyl Acetate Fractions on Lupus Arthritis Mice. Asian J. Pharm. Clin. Res. 2017, 10, 199. [Google Scholar] [CrossRef]
- Indriyanti, N.; Khotib, J.; Soeroso, J. Positive Impact of Ethyl Acetate Fraction of Kalanchoe pinnata on Anti-Smith Antibody and T Reg in Lupus Mice. Ind. J. Pharm. Sci. 2017, 15, 57–62. [Google Scholar]
- Ogidigo, J.O.; Anosike, C.A.; Joshua, P.E.; Ibeji, C.U.; Nwanguma, B.C.; Nwodo, O.F.C. Neuroprotective Effect of Bryophyllum pinnatum Flavonoids against Aluminum Chloride-Induced Neurotoxicity in Rats. Toxicol. Mech. Methods. 2022, 32, 243–258. [Google Scholar] [CrossRef] [PubMed]
- Ogidigo, J.O.; Anosike, C.A.; Joshua, P.E.; Ibeji, C.U.; Ekpo, D.E.; Nwanguma, B.C.; Nwodo, O.F.C. UPLC-PDA-ESI-QTOF-MS/MS Fingerprint of Purified Flavonoid Enriched Fraction of Bryophyllum pinnatum; Antioxidant Properties, Anticholinesterase Activity and in Silico Studies. Pharm. Biol. 2021, 59, 442–454. [Google Scholar] [CrossRef]
- Lai, Z.-R.; Ho, Y.-L.; Huang, S.-C.; Huang, T.-H.; Lai, S.-C.; Tsai, J.-C.; Wang, C.-Y.; Huang, G.-J.; Chang, Y.-S. Antioxidant, Anti-Inflammatory and Antiproliferative Activities of Kalanchoe gracilis (L.) DC Stem. Am. J. Chin. Med. 2011, 39, 1275–1290. [Google Scholar] [CrossRef] [PubMed]
- Muzitano, M.F.; Tinoco, L.W.; Guette, C.; Kaiser, C.R.; Rossi-Bergmann, B.; Costa, S.S. The Antileishmanial Activity Assessment of Unusual Flavonoids from Kalanchoe pinnata. Phytochemistry 2006, 67, 2071–2077. [Google Scholar] [CrossRef] [PubMed]
- Muzitano, M.; Falcão, C.; Cruz, E.; Bergonzi, M.; Bilia, A.; Vincieri, F.; Rossi-Bergmann, B.; Costa, S. Oral Metabolism and Efficacy of Kalanchoe pinnata Flavonoids in a Murine Model of Cutaneous Leishmaniasis. Planta Med. 2009, 75, 307–311. [Google Scholar] [CrossRef] [PubMed]
- Cruz, E.A.; Reuter, S.; Martin, H.; Dehzad, N.; Muzitano, M.F.; Costa, S.S.; Rossi-Bergmann, B.; Buhl, R.; Stassen, M.; Taube, C. Kalanchoe pinnata Inhibits Mast Cell Activation and Prevents Allergic Airway Disease. Phytomedicine 2012, 19, 115–121. [Google Scholar] [CrossRef] [PubMed]
- Cruz, E.A.; Da-Silva, S.A.G.; Muzitano, M.F.; Silva, P.M.R.; Costa, S.S.; Rossi-Bergmann, B. Immunomodulatory Pretreatment with Kalanchoe pinnata Extract and Its Quercitrin Flavonoid Effectively Protects Mice against Fatal Anaphylactic Shock. Int. Immunopharmacol. 2008, 8, 1616–1621. [Google Scholar] [CrossRef]
- Aoki, C.; Hartati, S.; Santi, M.R.; Firdaus, R.; Hanafi, M.; Kardono, L.B.S.; Shimizu, Y.; Sudarmono, P.; Hotta, H. Isolation and Identification of Substances with Anti-Hepatitis C Virus Activities from Kalanchoe pinnata. Int. J. Pharm. Pharm. Sci. 2014, 6, 211–215. [Google Scholar]
- Fernandes, J.M.; Ortiz, S.; Tavares, R.P.M.; Mandova, T.; Araújo, E.R.D.; Andrade, A.W.L.; Michel, S.; Grougnet, R.; Zucolotto, S.M. Bryophyllum pinnatum Markers: CPC Isolation, Simultaneous Quantification by a Validated UPLC-DAD Method and Biological Evaluations. J. Pharm. Biomed. Anal. 2021, 193, 113682. [Google Scholar] [CrossRef] [PubMed]
- Sofa, F.; Akhmad, D.; Megawati, M.; Muhammad, H. Isolation and Identification of Quercetin Derivatives and Their α-Glucosidase Inhibitory Acitivities From. Res. J. Chem. Environ. 2018, 22, 114–119. [Google Scholar]
- Muzitano, M.F.; Cruz, E.A.; de Almeida, A.P.; Da Silva, S.A.; Kaiser, C.R.; Guette, C.; Rossi-Bergmann, B.; Costa, S.S. Quercitrin: An Antileishmanial Flavonoid Glycoside from Kalanchoe pinnata. Planta Med. 2006, 72, 81–83. [Google Scholar] [CrossRef]
- Lourenço, E.M.G.; Fernandes, J.M.; Carvalho, V.D.F.; Grougnet, R.; Martins, M.A.; Jordão, A.K.; Zucolotto, S.M.; Barbosa, E.G. Identification of a Selective PDE4B Inhibitor From Bryophyllum pinnatum by Target Fishing Study and In Vitro Evaluation of Quercetin 3-O-α-L-Arabinopyranosyl-(1→2)-O-α-L-Rhamnopyranoside. Front. Pharmacol. 2020, 10, 1582. [Google Scholar] [CrossRef] [PubMed]
- Coutinho, M.A.S.; Casanova, L.M.; Nascimento, L.B.D.S.; Leal, D.; Palmero, C.; Toma, H.K.; dos Santos, E.P.; Nasciutti, L.E.; Costa, S.S. Wound Healing Cream Formulated with Kalanchoe pinnata Major Flavonoid Is as Effective as the Aqueous Leaf Extract Cream in a Rat Model of Excisional Wound. N. Prod. Res. 2021, 35, 6034–6039. [Google Scholar] [CrossRef]
- De Araújo, E.R.D.; Guerra, G.C.B.; Andrade, A.W.L.; Fernandes, J.M.; Da Silva, V.C.; De Aragão Tavares, E.; De Araújo, A.A.; de Araújo Júnior, R.F.; Zucolotto, S.M. Gastric Ulcer Healing Property of Bryophyllum pinnatum Leaf Extract in Chronic Model In Vivo and Gastroprotective Activity of Its Major Flavonoid. Front. Pharmacol. 2021, 12, 744192. [Google Scholar] [CrossRef] [PubMed]
- Tatsimo, S.J.N.; Tamokou, J.D.D.; Havyarimana, L.; Csupor, D.; Forgo, P.; Hohmann, J.; Kuiate, J.-R.; Tane, P. Antimicrobial and Antioxidant Activity of Kaempferol Rhamnoside Derivatives from Bryophyllum pinnatum. BMC Res. Notes 2012, 5, 158. [Google Scholar] [CrossRef]
- Aisyah, L.S.; Yun, Y.F.; Herlina, T.; Julaeha, E.; Zainuddin, A.; Nurfarida, I.; Hidayat, A.T.; Supratman, U.; Shiono, Y. Flavonoid Compounds from the Leaves of Kalanchoe prolifera and Their Cytotoxic Activity against P-388 Murine Leukimia Cells. Nat. Prod. Sci. 2017, 23, 139–145. [Google Scholar] [CrossRef]
- Aisyah, L.S.; Yun, Y.F.; Julaeha, E.; Herlina, T.; Zainuddin, A.; Hermawan, W.; Supratman, U.; Hayashi, H. Flavonoids from the Fresh Leaves of Kalanchoe tomentosa (Crassulaceae). Open Chem. J. 2015, 2, 36–39. [Google Scholar] [CrossRef]
- Aisyah, L.S.; Ilfani, D.; Lestari, F.P.; Yun, Y.F. α-Amylase Inhibition Activities by Flavonoid Compounds from Panda Plants (Kalanchoe tomentosa). J. Kim. Sains Apl. 2020, 23, 96–101. [Google Scholar] [CrossRef]
- Agrawal, P.K.; Agrawal, C.; Blunden, G. Quercetin: Antiviral Significance and Possible COVID-19 Integrative Considerations. Nat. Prod. Commun. 2020, 15, 1934578X20976293. [Google Scholar] [CrossRef]
- Yang, D.; Wang, T.; Long, M.; Li, P. Quercetin: Its Main Pharmacological Activity and Potential Application in Clinical Medicine. Oxid. Med. Cell. Longev. 2020, 2020, 8825387. [Google Scholar] [CrossRef] [PubMed]
- Torello, C.O.; Alvarez, M.C.; Olalla Saad, S.T. Polyphenolic Flavonoid Compound Quercetin Effects in the Treatment of Acute Myeloid Leukemia and Myelodysplastic Syndromes. Molecules 2021, 26, 5781. [Google Scholar] [CrossRef] [PubMed]
- Calgarotto, A.K.; Maso, V.; Junior, G.C.F.; Nowill, A.E.; Filho, P.L.; Vassallo, J.; Saad, S.T.O. Antitumor Activities of Quercetin and Green Tea in Xenografts of Human Leukemia HL60 Cells. Sci Rep 2018, 8, 3459. [Google Scholar] [CrossRef]
- Baron, B.W.; Thirman, M.J.; Giurcanu, M.C.; Baron, J.M. Quercetin Therapy for Selected Patients with PIM1 Kinase-Positive Chronic Lymphocytic Leukemia/Small Lymphocytic Lymphoma: A Pilot Study. Acta Haematol. 2018, 139, 132–139. [Google Scholar] [CrossRef]
- Anand David, A.; Arulmoli, R.; Parasuraman, S. Overviews of Biological Importance of Quercetin: A Bioactive Flavonoid. Pharmacog. Rev. 2016, 10, 84–89. [Google Scholar] [CrossRef]
- Rojas, Á.; Del Campo, J.A.; Clement, S.; Lemasson, M.; García-Valdecasas, M.; Gil-Gómez, A.; Ranchal, I.; Bartosch, B.; Bautista, J.D.; Rosenberg, A.R.; et al. Effect of Quercetin on Hepatitis C Virus Life Cycle: From Viral to Host Targets. Sci. Rep. 2016, 6, 31777. [Google Scholar] [CrossRef] [PubMed]
- Bachmetov, L.; Gal-Tanamy, M.; Shapira, A.; Vorobeychik, M.; Giterman-Galam, T.; Sathiyamoorthy, P.; Golan-Goldhirsh, A.; Benhar, I.; Tur-Kaspa, R.; Zemel, R. Suppression of Hepatitis C Virus by the Flavonoid Quercetin Is Mediated by Inhibition of NS3 Protease Activity: Quercetin Suppresses HCV through Inhibition of NS3. J. Viral Hepat. 2012, 19, e81–e88. [Google Scholar] [CrossRef] [PubMed]
- Sen, G.; Mandal, S.; Saharoy, S.; Mukhopadhyay, S.; Biswas, T. Therapeutic Use of Quercetin in the Control of Infection and Anemia Associated with Visceral Leishmaniasis. Free Radic. Biol. Med. 2005, 38, 1257–1264. [Google Scholar] [CrossRef]
- Cataneo, A.H.D.; Tomiotto-Pellissier, F.; Miranda-Sapla, M.M.; Assolini, J.P.; Panis, C.; Kian, D.; Yamauchi, L.M.; Colado Simão, A.N.; Casagrande, R.; Pinge-Filho, P.; et al. Quercetin Promotes Antipromastigote Effect by Increasing the ROS Production and Anti-Amastigote by Upregulating Nrf2/HO-1 Expression, Affecting Iron Availability. Biomed. Pharmacother. 2019, 113, 108745. [Google Scholar] [CrossRef]
- Tasdemir, D.; Kaiser, M.; Brun, R.; Yardley, V.; Schmidt, T.J.; Tosun, F.; Rüedi, P. Antitrypanosomal and Antileishmanial Activities of Flavonoids and Their Analogues: In Vitro, In Vivo, Structure-Activity Relationship, and Quantitative Structure-Activity Relationship Studies. Antimicrob. Agents Chemother. 2006, 50, 1352–1364. [Google Scholar] [CrossRef] [PubMed]
- Salehi, B.; Machin, L.; Monzote, L.; Sharifi-Rad, J.; Ezzat, S.M.; Salem, M.A.; Merghany, R.M.; El Mahdy, N.M.; Kılıç, C.S.; Sytar, O.; et al. Therapeutic Potential of Quercetin: New Insights and Perspectives for Human Health. ACS Omega 2020, 5, 11849–11872. [Google Scholar] [CrossRef] [PubMed]
- Nielsen, A.H.; Olsen, C.E.; Møller, B.L. Flavonoids in Flowers of 16 Kalanchoë blossfeldiana Varieties. Phytochemistry 2005, 66, 2829–2835. [Google Scholar] [CrossRef] [PubMed]
- Su, Y.; Ding, J.; Yang, F.; He, C.; Xu, Y.; Zhu, X.; Zhou, H.; Li, H. The Regulatory Role of PDE4B in the Progression of Inflammatory Function Study. Front. Pharmacol. 2022, 13, 982130. [Google Scholar] [CrossRef]
- Roth, M.; Araya, J.J.; Timmermann, B.N.; Hagenbuch, B. Isolation of Modulators of the Liver-Specific Organic Anion-Transporting Polypeptides (OATPs) 1B1 and 1B3 from Rollinia emarginata Schlecht (Annonaceae). J. Pharmacol. Exp. Ther. 2011, 339, 624–632. [Google Scholar] [CrossRef]
- Zhang, Y.; Hays, A.; Noblett, A.; Thapa, M.; Hua, D.H.; Hagenbuch, B. Transport by OATP1B1 and OATP1B3 Enhances the Cytotoxicity of Epigallocatechin 3- O -Gallate and Several Quercetin Derivatives. J. Nat. Prod. 2013, 76, 368–373. [Google Scholar] [CrossRef] [PubMed]
- Michala, A.-S.; Pritsa, A. Quercetin: A Molecule of Great Biochemical and Clinical Value and Its Beneficial Effect on Diabetes and Cancer. Diseases 2022, 10, 37. [Google Scholar] [CrossRef] [PubMed]
- Dirir, A.M.; Daou, M.; Yousef, A.F.; Yousef, L.F. A Review of Alpha-Glucosidase Inhibitors from Plants as Potential Candidates for the Treatment of Type-2 Diabetes. Phytochem. Rev. 2022, 21, 1049–1079. [Google Scholar] [CrossRef]
- Suprapto, A.K.; Tih, F.; Evacuasiany, E. Effect of Methanolic Extract in Ointment and Powder of Kalanchoe pinnata (Lamk) Leaf in Ointment towards Incision Wound Healing in Mice. J. Med. Health 2015, 1, 12–19. [Google Scholar] [CrossRef]
- Nayak, B.S.; Marshall, J.R.; Isitor, G. Wound Healing Potential of Ethanolic Extract of Kalanchoe pinnata Lam. Leaf—A Preliminary Study. Indian J. Exp. Biol. 2010, 49, 572–576. [Google Scholar]
- Araújo, E.R.D.; Xavier-Santos, J.B.; da Silva, V.C.; de Lima, J.B.F.; Schlamb, J.; Fernandes-Pedrosa, M.D.F.; da Silva Júnior, A.A.; de Araújo Júnior, R.F.; Rathinasabapathy, T.; Moncada, M.; et al. Gel Formulated with Bryophyllum pinnatum Leaf Extract Promotes Skin Wound Healing in Vivo by Increasing VEGF Expression: A Novel Potential Active Ingredient for Pharmaceuticals. Front. Pharmacol. 2023, 13, 1104705. [Google Scholar] [CrossRef]
- Krishnappa, P.; Venkatarangaiah, K.; Venkatesh; Shimoga Rajanna, S.K.; Kayattukandy Balan, R. Wound Healing Activity of Delonix elata Stem Bark Extract and Its Isolated Constituent Quercetin-3-Rhamnopyranosyl-(1-6) Glucopyranoside in Rats. J. Pharm. Anal. 2016, 6, 389–395. [Google Scholar] [CrossRef]
- Özbilgin, S.; Acıkara, Ö.B.; Akkol, E.K.; Süntar, I.; Keleş, H.; İşcan, G.S. In Vivo Wound-Healing Activity of Euphorbia characias Subsp. wulfenii: Isolation and Quantification of Quercetin Glycosides as Bioactive Compounds. J. Ethnopharmacol. 2018, 224, 400–408. [Google Scholar] [CrossRef] [PubMed]
- Süntar, I.P.; Akkol, E.K.; Yalçın, F.N.; Koca, U.; Keleş, H.; Yesilada, E. Wound Healing Potential of Sambucus ebulus L. Leaves and Isolation of an Active Component, Quercetin 3-O-Glucoside. J. Ethnopharmacol. 2010, 129, 106–114. [Google Scholar] [CrossRef]
- De Melo, G.O.; Malvar, D.D.C.; Vanderlinde, F.A.; Rocha, F.F.; Pires, P.A.; Costa, E.A.; de Matos, L.G.; Kaiser, C.R.; Costa, S.S. Antinociceptive and Anti-Inflammatory Kaempferol Glycosides from Sedum dendroideum. J. Ethnopharmacol. 2009, 124, 228–232. [Google Scholar] [CrossRef]
- Cechinel-Zanchett, C.C.; Bolda Mariano, L.N.; Boeing, T.; da Costa, J.D.C.; Da Silva, L.M.; Bastos, J.K.; Cechinel-Filho, V.; de Souza, P. Diuretic and Renal Protective Effect of Kaempferol 3-O-Alpha-l-Rhamnoside (Afzelin) in Normotensive and Hypertensive Rats. J. Nat. Prod. 2020, 83, 1980–1989. [Google Scholar] [CrossRef]
- Zhou, W.; Nie, X. Afzelin Attenuates Asthma Phenotypes by Downregulation of GATA3 in a Murine Model of Asthma. Mol. Med. Rep. 2015, 12, 71–76. [Google Scholar] [CrossRef] [PubMed]
- Akter, M.; Parvin, M.S.; Hasan, M.M.; Rahman, M.A.A.; Islam, M.E. Anti-Tumor and Antioxidant Activity of Kaempferol-3-O-Alpha-L-Rhamnoside (Afzelin) Isolated from Pithecellobium dulce Leaves. BMC Complement. Med. Ther. 2022, 22, 169. [Google Scholar] [CrossRef]
- Radziejewska, I.; Supruniuk, K.; Czarnomysy, R.; Buzun, K.; Bielawska, A. Anti-Cancer Potential of Afzelin towards AGS Gastric Cancer Cells. Pharmaceuticals 2021, 14, 973. [Google Scholar] [CrossRef]
- Zhu, K.-C.; Sun, J.-M.; Shen, J.-G.; Jin, J.-Z.; Liu, F.; Xu, X.-L.; Chen, L.; Liu, L.-T.; Lv, J.-J. Afzelin Exhibits Anti-Cancer Activity against Androgen-Sensitive LNCaP and Androgen-Independent PC-3 Prostate Cancer Cells through the Inhibition of LIM Domain Kinase 1. Oncol. Lett. 2015, 10, 2359–2365. [Google Scholar] [CrossRef]
- Casanova, J.M.; Nascimento, L.B.D.S.; Casanova, L.M.; Castricini, S.D.; De Souza, J.E.; Kao Yien, R.M.; Costa, S.S.; Tavares, E.S. Kalanchoe fedtschenkoi R. Hamet & H. Perrier, a Non-Conventional Food Plant in Brazil: HPLC-DAD-ESI-MS/MS Profile and Leaf Histochemical Location of Flavonoids. J. Appl. Bot. Food Qual. 2022, 95, 154–166. [Google Scholar] [CrossRef]
- Abebe, W. An Overview of Ethiopian Traditional Medicinal Plants Used for Cancer Treatment. Eur. J. Phys. 2016, 14, EJMP.25670. [Google Scholar] [CrossRef]
- Arias-González, I.; García-Carrancá, A.M.; Cornejo-Garrido, J.; Ordaz-Pichardo, C. Cytotoxic Effect of Kalanchoe flammea and Induction of Intrinsic Mitochondrial Apoptotic Signaling in Prostate Cancer Cells. J. Ethnopharmacol. 2018, 222, 133–147. [Google Scholar] [CrossRef] [PubMed]
- Joshi, B.; Panda, S.K.; Jouneghani, R.S.; Liu, M.; Parajuli, N.; Leyssen, P.; Neyts, J.; Luyten, W. Antibacterial, Antifungal, Antiviral, and Anthelmintic Activities of Medicinal Plants of Nepal Selected Based on Ethnobotanical Evidence. Evid. Based Complement. Altern. Med. 2020, 2020, 1043471. [Google Scholar] [CrossRef]
- Muñoz, V.; Sauvain, M.; Bourdy, G.; Callapa, J.; Rojas, I.; Vargas, L.; Tae, A.; Deharo, E. The Search for Natural Bioactive Compounds through a Multidisciplinary Approach in Bolivia. Part II. Antimalarial Activity of Some Plants Used by Mosetene Indians. J. Ethnopharmacol. 2000, 69, 139–155. [Google Scholar] [CrossRef]
- Patil, S.B.; Dongare, V.R.; Kulkarni, C.R.; Joglekar, M.M.; Arvindekar, A.U. Antidiabetic Activity of Kalanchoe pinnata in Streptozotocin-Induced Diabetic Rats by Glucose Independent Insulin Secretagogue Action. Pharm. Biol. 2013, 51, 1411–1418. [Google Scholar] [CrossRef] [PubMed]
- Hamre, H.J.; Witt, C.M.; Glockmann, A.; Tröger, W.; Willich, S.N.; Kiene, H. Use and Safety of Anthroposophic Medications in Chronic Disease: A 2-Year Prospective Analysis. Drug Safety 2006, 29, 1173–1189. [Google Scholar] [CrossRef] [PubMed]
- Plangger, N.; Rist, L.; Zimmermann, R.; von Mandach, U. Intravenous Tocolysis with Bryophyllum pinnatum Is Better Tolerated than Beta-Agonist Application. Eur. J. Obstet. Gynecol. Reprod. Biol. 2006, 124, 168–172. [Google Scholar] [CrossRef]
- Simões-Wüst, A.P.; Jeschke, E.; Mennet, M.; Schnelle, M.; Matthes, H.; von Mandach, U. Prescribing Pattern of Bryophyllum Preparations among a Network of Anthroposophic Physicians. Forsch Komplementmed. 2012, 19, 293–301. [Google Scholar] [CrossRef] [PubMed]
- Fürer, K.; Simões-Wüst, A.P.; Winkler, A.; Amsler, N.; Schnelle, M.; von Mandach, U. Die Anwendung von Bryophyllum pinnatum-Präparaten in Der Geburtshilfe Und Gynäkologie—Eine Multizentrische Prospektive Beobachtungsstudie. Complement. Med. Res 2015, 22, 231–236. [Google Scholar] [CrossRef]
- Hosomi, J.K.; da Silva Facina, A.; de Jesus Simões, M.; Nakamura, M.U. Effects of Bryophyllum pinnatum Administration on Wistar Rat Pregnancy: Biochemical and Histological Aspects. Complement. Med. Res. 2022, 29, 35–42. [Google Scholar] [CrossRef] [PubMed]
- Simões-Wüst, A.P.; Lapaire, O.; Hösli, I.; Wächter, R.; Fürer, K.; Schnelle, M.; Mennet-von Eiff, M.; Seifert, B.; von Mandach, U. Two Randomised Clinical Trials on the Use of Bryophyllum pinnatum in Preterm Labour: Results after Early Discontinuation. Complement. Med. Res. 2018, 25, 269–273. [Google Scholar] [CrossRef]
- Santos, S.; Haslinger, C.; Mennet, M.; von Mandach, U.; Hamburger, M.; Simões-Wüst, A.P. Bryophyllum pinnatum Enhances the Inhibitory Effect of Atosiban and Nifedipine on Human Myometrial Contractility: An in Vitro Study. BMC Complement. Altern. Med. 2019, 19, 292. [Google Scholar] [CrossRef] [PubMed]
- Torres-Santos, E.C.; Da Silva, S.A.G.; Costa, S.S.; Santos, A.P.P.T.; Almeida, A.P.; Rossi-Bergmann, B. Toxicological Analysis and Effectiveness of Oral Kalanchoe pinnata on a Human Case of Cutaneous Leishmaniasis. Phytother. Res. 2003, 17, 801–803. [Google Scholar] [CrossRef] [PubMed]
- Khan, M.; Patil, P.A.; Shobha, J.C. Influence of Bryophyllum pinnatum (Lam.) Leaf Extract on Wound Healing in Albino Rats. J. Nat. Rem. 2004, 4, 41–46. [Google Scholar]
- Rajsekhar, P.; Bharani, R.; Ramachandran, M.; Angel, K.; Rajsekhar, S. The “Wonder Plant” Kalanchoe pinnata (Linn.) Pers.: A Review. J. Appl. Pharm. Sci. 2016, 3, 151–158. [Google Scholar] [CrossRef]
- Steyn, P.S.; van Heerden, F.R. Bufadienolides of Plant and Animal Origin. Nat. Prod. Rep. 1998, 15, 397–413. [Google Scholar] [CrossRef]
- Yin, R.; Messner, B.; Faus-Kessler, T.; Hoffmann, T.; Schwab, W.; Hajirezaei, M.-R.; von Saint Paul, V.; Heller, W.; Schäffner, A.R. Feedback Inhibition of the General Phenylpropanoid and Flavonol Biosynthetic Pathways upon a Compromised Flavonol-3-O-Glycosylation. J. Exp. Bot. 2012, 63, 2465–2478. [Google Scholar] [CrossRef] [PubMed]
- Jou, S.-J.; Chen, C.-H.; Guh, J.-H.; Lee, C.-N.; Lee, S.-S. Flavonol Glycosides and Cytotoxic Triterpenoids from Alphitonia philippinensis. J. Chin. Chem. Soc. 2004, 51, 827–834. [Google Scholar] [CrossRef]
- Yan-Ying, Y.E. Chemical Constituents in Herbs of Bryophyllum pinnatum. Chin. Trad. Her. Drugs 2012, 24, 2642–2646. [Google Scholar]
- Cryer, M.; Lane, K.; Greer, M.; Cates, R.; Burt, S.; Andrus, M.; Zou, J.; Rogers, P.; Hansen, M.D.H.; Burgado, J.; et al. Isolation and Identification of Compounds from Kalanchoe pinnata Having Human Alphaherpesvirus and Vaccinia Virus Antiviral Activity. Pharm. Biol. 2017, 55, 1586–1591. [Google Scholar] [CrossRef] [PubMed]
- Palumbo, A.; Casanova, L.M.; Corrêa, M.F.P.; Da Costa, N.M.; Nasciutti, L.E.; Costa, S.S. Potential Therapeutic Effects of Underground Parts of Kalanchoe gastonis-bonnieri on Benign Prostatic Hyperplasia. Evid. Based Complement. Altern. Med. 2019, 2019, 6340757. [Google Scholar] [CrossRef] [PubMed]
- Zhou, J.-T.; Li, C.-Y.; Wang, C.-H.; Wang, Y.-F.; Wang, X.-D.; Wang, H.-T.; Zhu, Y.; Jiang, M.-M.; Gao, X.-M. Phenolic Compounds from the Roots of Rhodiola crenulata and Their Antioxidant and Inducing IFN-γ Production Activities. Molecules 2015, 20, 13725–13739. [Google Scholar] [CrossRef] [PubMed]
- Calixto, J.B. The Role of Natural Products in Modern Drug Discovery. An. Acad. Bras. Cienc. 2019, 91, e20190105. [Google Scholar] [CrossRef]
- Najmi, A.; Javed, S.A.; Al Bratty, M.; Alhazmi, H.A. Modern Approaches in the Discovery and Development of Plant-Based Natural Products and Their Analogues as Potential Therapeutic Agents. Molecules 2022, 27, 349. [Google Scholar] [CrossRef]
- Newman, D.J.; Cragg, G.M. Natural Products as Sources of New Drugs over the Nearly Four Decades from 01/1981 to 09/2019. J. Nat. Prod. 2020, 83, 770–803. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Zotchev, S.B.; Dirsch, V.M.; Supuran, C.T. Natural Products in Drug Discovery: Advances and Opportunities. Nat. Rev. Drug Discov. 2021, 20, 200–216. [Google Scholar] [CrossRef]
- Smith, G.F.; Shtein, R.; Klein, D.-P.; Parihar, B.; Almeida, A.; Rodewald, S.; Kadereit, G. Sexual and Asexual Reproduction in Kalanchoe (Crassulaceae): A Review of Known and Newly Recorded Strategies. Haseltonia 2022, 28, 2–20. [Google Scholar] [CrossRef]
- Smith, G.; Figueiredo, E.; Van Wyk, B. Kalanchoe (Crassulaceae) in Southern Africa: Classification, Biology, and Cultivation; Academic Press, Imprint of Elsevier: London, UK; San Diego, CA, USA, 2019; ISBN 978-0-12-814007-9. [Google Scholar]
- Cruz, B.P.; Chedier, L.M.; Fabri, R.L.; Pimenta, D.S. Chemical and Agronomic Development of Kalanchoe brasiliensis Camb. and Kalanchoe pinnata (Lamk.) Pers under Light and Temperature Levels. An. Acad. Bras. Cienc. 2011, 83, 1435–1442. [Google Scholar] [CrossRef] [PubMed]
- Cruz, B.P.; Chedier, L.M.; Peixoto, P.H.P.; Fabri, R.L.; Pimenta, D.S. Effects of Light Intensity on the Distribution of Anthocyanins in Kalanchoe brasiliensis Camb. and Kalanchoe pinnata (Lamk.) Pers. An. Acad. Bras. Cienc. 2012, 84, 211–217. [Google Scholar] [CrossRef] [PubMed]
- Muzitano, M.F.; Bergonzi, M.C.; De Melo, G.O.; Lage, C.L.S.; Bilia, A.R.; Vincieri, F.F.; Rossi-Bergmann, B.; Costa, S.S. Influence of Cultivation Conditions, Season of Collection and Extraction Method on the Content of Antileishmanial Flavonoids from Kalanchoe pinnata. J. Ethnopharmacol. 2011, 133, 132–137. [Google Scholar] [CrossRef] [PubMed]
- Smith, G.F.; Shtein, R. A Review of Horticulturally Desirable Characters in Kalanchoe (Crassulaceae Subfam. Kalanchooideae): Variable and Deviating Vegetative and Reproductive Morphologies Useful in Breeding Programmes. Haseltonia 2022, 28, 106–119. [Google Scholar] [CrossRef]
- Fujimoto, T.; Otani, M.; Nakano, M. Production of Colored Foliage Phenotypes in Kalanchoe blossfeldiana by Ectopic Expression of R2R3 MYB Genes. J. Plant Biochem. Biotechnol. 2022, 31, 665–672. [Google Scholar] [CrossRef]
- Wang, L.; Zhu, C.; Jin, L.; Xiao, A.; Duan, J.; Ma, L. A Novel Gene of Kalanchoe daigremontiana Confers Plant Drought Resistance. Sci. Rep. 2018, 8, 2547. [Google Scholar] [CrossRef]
- Al-Khayri, J.M.; Mahdy, E.M.B.; Taha, H.S.A.; Eldomiaty, A.S.; Abd-Elfattah, M.A.; Abdel Latef, A.A.H.; Rezk, A.A.; Shehata, W.F.; Almaghasla, M.I.; Shalaby, T.A.; et al. Genetic and Morphological Diversity Assessment of Five Kalanchoe Genotypes by SCoT, ISSR and RAPD-PCR Markers. Plants 2022, 11, 1722. [Google Scholar] [CrossRef]
- Tian, X.; Guo, J.; Zhou, X.; Ma, K.; Ma, Y.; Shi, T.; Shi, Y. Comparative and Evolutionary Analyses on the Complete Plastomes of Five Kalanchoe Horticultural Plants. Front. Plant Sci. 2021, 12, 705874. [Google Scholar] [CrossRef] [PubMed]
- Kahraman, M.U.; Mendi, Y.Y.; Karabıyık, Ş.; Lütken, H.V.; Favero, B.T. Kalanchoë Breeding: Past, Present and Future. Ornam. Hortic. 2022, 28, 19–35. [Google Scholar] [CrossRef]
- Tariq, H.; Asif, S.; Andleeb, A.; Hano, C.; Abbasi, B.H. Flavonoid Production: Current Trends in Plant Metabolic Engineering and De Novo Microbial Production. Metabolites 2023, 13, 124. [Google Scholar] [CrossRef] [PubMed]
- García-Pérez, P.; Lozano-Milo, E.; Landín, M.; Gallego, P.P. Combining Medicinal Plant In Vitro Culture with Machine Learning Technologies for Maximizing the Production of Phenolic Compounds. Antioxidants 2020, 9, 210. [Google Scholar] [CrossRef]
- Gandhi, S.G.; Mahajan, V.; Bedi, Y.S. Changing Trends in Biotechnology of Secondary Metabolism in Medicinal and Aromatic Plants. Planta 2015, 241, 303–317. [Google Scholar] [CrossRef]
- Gutierrez-Valdes, N.; Häkkinen, S.T.; Lemasson, C.; Guillet, M.; Oksman-Caldentey, K.-M.; Ritala, A.; Cardon, F. Hairy Root Cultures—A Versatile Tool With Multiple Applications. Front. Plant Sci. 2020, 11, 33. [Google Scholar] [CrossRef]
- Kowalczyk, T.; Merecz-Sadowska, A.; Picot, L.; Brčić Karačonji, I.; Wieczfinska, J.; Śliwiński, T.; Sitarek, P. Genetic Manipulation and Bioreactor Culture of Plants as a Tool for Industry and Its Applications. Molecules 2022, 27, 795. [Google Scholar] [CrossRef]
- Wang, Y.; Chen, S.; Yu, O. Metabolic Engineering of Flavonoids in Plants and Microorganisms. Appl. Microbiol. Biotechnol. 2011, 91, 949–956. [Google Scholar] [CrossRef] [PubMed]
- Khatri, S.; Saini, R.V.; Chhillar, A.K. Molecular Farming Approach Towards Bioactive Compounds. In Metabolic Engineering for Bioactive Compounds; Kalia, V.C., Saini, A.K., Eds.; Springer: Singapore, 2017; pp. 49–72. ISBN 978-981-10-5510-2. [Google Scholar]
- Baenas, N.; García-Viguera, C.; Moreno, D.A. Elicitation: A Tool for Enriching the Bioactive Composition of Foods. Molecules 2014, 19, 13541–13563. [Google Scholar] [CrossRef] [PubMed]
- Leal-Costa, M.V.; Nascimento, L.B.D.S.; Moreira, N.D.S.; Reinert, F.; Costa, S.S.; Lage, C.L.S.; Tavares, E.S. Influence of Blue Light on the Leaf Morphoanatomy of In Vitro Kalanchoe pinnata (Lamarck) Persoon (Crassulaceae). Microsc. Microanal. 2010, 16, 576–582. [Google Scholar] [CrossRef]
- Fkiara, A.; Barba-Espín, G.; El-Houri, R.; Müller, R.; Christensen, L.P.; Lütken, H. Elicitation of Flavonoids in Kalanchoe pinnata by Agrobacterium Rhizogenes-Mediated Transformation and UV-B Radiation. In Medicinal Plants: Biodiversity, Sustainable Utilization and Conservation; Khasim, S.M., Long, C., Thammasiri, K., Lutken, H., Eds.; Springer: Singapore, 2020; pp. 395–403. ISBN 9789811516351. [Google Scholar]
- García-Pérez, P.; Losada-Barreiro, S.; Gallego, P.P.; Bravo-Díaz, C. Cyclodextrin-Elicited Bryophyllum Suspension Cultured Cells: Enhancement of the Production of Bioactive Compounds. Int. J. Mol. Sci. 2019, 20, 5180. [Google Scholar] [CrossRef]
- García-Pérez, P.; Lozano-Milo, E.; Zhang, L.; Miras-Moreno, B.; Landin, M.; Lucini, L.; Gallego, P.P. Neurofuzzy Logic Predicts a Fine-Tuning Metabolic Reprogramming on Elicited Bryophyllum PCSCs Guided by Salicylic Acid. Front. Plant Sci. 2022, 13, 991557. [Google Scholar] [CrossRef]
- García-Pérez, P.; Lozano-Milo, E.; Landin, M.; Gallego, P.P. From Ethnomedicine to Plant Biotechnology and Machine Learning: The Valorization of the Medicinal Plant Bryophyllum Sp. Pharmaceuticals 2020, 13, 444. [Google Scholar] [CrossRef]
- Butterweck, V.; Nahrstedt, A. What Is the Best Strategy for Preclinical Testing of Botanicals? A Critical Perspective. Planta Med. 2012, 78, 747–754. [Google Scholar] [CrossRef]
- Atanasov, A.G.; Waltenberger, B.; Pferschy-Wenzig, E.-M.; Linder, T.; Wawrosch, C.; Uhrin, P.; Temml, V.; Wang, L.; Schwaiger, S.; Heiss, E.H.; et al. Discovery and Resupply of Pharmacologically Active Plant-Derived Natural Products: A Review. Biotechnol. Adv. 2015, 33, 1582–1614. [Google Scholar] [CrossRef] [PubMed]
- Houghton, P.J.; Howes, M.-J.; Lee, C.C.; Steventon, G. Uses and Abuses of in Vitro Tests in Ethnopharmacology: Visualizing an Elephant. J. Ethnopharmacol. 2007, 110, 391–400. [Google Scholar] [CrossRef] [PubMed]
- ANVISA Guia Para a Condução de Estudos Não Clínicos de Toxicologia e Segurança Farmacológica Necessários Ao Desenvolvimento de Medicamentos 2013. Available online: https://www.gov.br/anvisa/pt-br/centraisdeconteudo/publicacoes/medicamentos/pesquisa-clinica/manuais-e-guias/guia-para-a-conducao-de-estudos-nao-clinicos-de-toxicologia-e-seguranca-farmacologica-necessarios-ao-desenvolvimento-de-medicamentos-versao-2.pdf/view (accessed on 4 January 2023).
- Thomford, N.; Senthebane, D.; Rowe, A.; Munro, D.; Seele, P.; Maroyi, A.; Dzobo, K. Natural Products for Drug Discovery in the 21st Century: Innovations for Novel Drug Discovery. Int. J. Mol. Sci. 2018, 19, 1578. [Google Scholar] [CrossRef] [PubMed]
- Anderson, L.A.P.; Schultz, R.A.; Joubert, J.P.J.; Prozesky, L.; Kellerman, T.; Erasmus, G.L.; Procos, J. Krimpsiekte and Acute Cardiac Glycoside Poisoning in Sheep Caused by Bufadienolides from the Plant Kalanchoe lanceolata Forsk. Onderstepoort J. Vet. Res. 1983, 50, 295–300. [Google Scholar] [PubMed]
- Mckenzie, R.A.; Franke, F.P.; Dunster, P.J. The Toxicity to Cattle and Bufadienolide Content of Six Bryophyllum Species. Aust. Vet. J. 1987, 64, 298–301. [Google Scholar] [CrossRef] [PubMed]
- Wang, W.; Sun, C.; Mao, L.; Ma, P.; Liu, F.; Yang, J.; Gao, Y. The Biological Activities, Chemical Stability, Metabolism and Delivery Systems of Quercetin: A Review. Trends Food Sci. Technol. 2016, 56, 21–38. [Google Scholar] [CrossRef]
- Alam, W.; Khan, H.; Shah, M.A.; Cauli, O.; Saso, L. Kaempferol as a Dietary Anti-Inflammatory Agent: Current Therapeutic Standing. Molecules 2020, 25, 4073. [Google Scholar] [CrossRef] [PubMed]
- Corcoran, M.P.; McKay, D.L.; Blumberg, J.B. Flavonoid Basics: Chemistry, Sources, Mechanisms of Action, and Safety. J. Nutr. Gerontol. Geriatr. 2012, 31, 176–189. [Google Scholar] [CrossRef]
- Zhou, D.; Bai, Z.; Guo, T.; Li, J.; Li, Y.; Hou, Y.; Chen, G.; Li, N. Dietary Flavonoids and Human Top-Ranked Diseases: The Perspective of in Vivo Bioactivity and Bioavailability. Trends Food Sci. Technol. 2022, 120, 374–386. [Google Scholar] [CrossRef]
- Shao, H.; Li, B.; Li, H.; Gao, L.; Zhang, C.; Sheng, H.; Zhu, L. Novel Strategies for Solubility and Bioavailability Enhancement of Bufadienolides. Molecules 2021, 27, 51. [Google Scholar] [CrossRef]
- Naeem, A.; Ming, Y.; Pengyi, H.; Jie, K.Y.; Yali, L.; Haiyan, Z.; Shuai, X.; Wenjing, L.; Ling, W.; Xia, Z.M.; et al. The Fate of Flavonoids after Oral Administration: A Comprehensive Overview of Its Bioavailability. Crit. Rev. Food Sci. Nutr. 2022, 62, 6169–6186. [Google Scholar] [CrossRef]
- Vazhappilly, C.G.; Amararathna, M.; Cyril, A.C.; Linger, R.; Matar, R.; Merheb, M.; Ramadan, W.S.; Radhakrishnan, R.; Rupasinghe, H.P.V. Current Methodologies to Refine Bioavailability, Delivery, and Therapeutic Efficacy of Plant Flavonoids in Cancer Treatment. J. Nutr. Biochem. 2021, 94, 108623. [Google Scholar] [CrossRef]
- Gonzales, G.B.; Smagghe, G.; Grootaert, C.; Zotti, M.; Raes, K.; Camp, J.V. Flavonoid Interactions during Digestion, Absorption, Distribution and Metabolism: A Sequential Structure–Activity/Property Relationship-Based Approach in the Study of Bioavailability and Bioactivity. Drug Metab. Rev. 2015, 47, 175–190. [Google Scholar] [CrossRef]
- Zhao, J.; Yang, J.; Xie, Y. Improvement Strategies for the Oral Bioavailability of Poorly Water-Soluble Flavonoids: An Overview. Int. J. Pharm. 2019, 570, 118642. [Google Scholar] [CrossRef] [PubMed]
- Dos Santos Lima, B.; Shanmugam, S.; de Souza Quintans, J.S.; Quintans-Júnior, L.J.; de Souza Araújo, A.A. Inclusion Complex with Cyclodextrins Enhances the Bioavailability of Flavonoid Compounds: A Systematic Review. Phytochem. Rev. 2019, 18, 1337–1359. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).