Preliminary Survey of Pathogens in the Asian Honey Bee (Apis cerana) in Thailand
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection
2.2. DNA Extraction and DNA Analyses for Species Identification
2.3. RNA Extraction and cDNA Synthesis for Viral Detection
2.4. DNA Extraction for Non-Virus Analysis
2.5. PCR Conditions
2.6. Phylogenetic Tree Analysis
3. Results
3.1. COI-Sequence-Based Characterization of A. cerana Samples
3.2. N. ceranae, P. larvae, and A. apis Frequencies in A. cerana Colonies
3.3. Virus Frequencies in A. cerana Colonies
3.4. Phylogenetic Analysis
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Chantawannakul, P.; Williams, G.; Neumann, P. Asian Beekeeping in the 21st Century; Springer: Berlin/Heidelberg, Germany, 2018; ISBN 978-981-10-8221-4. [Google Scholar]
- Sylvester, H.A.; Limbipichai, K.; Wongsiri, S.; Rinderer, T.E.; Mardan, M. Morphometric Studies of Apis cerana in Thailand and the Malaysian Peninsula. J. Apic. Res. 1998, 37, 137–145. [Google Scholar] [CrossRef]
- Potts, S.G.; Biesmeijer, J.C.; Kremen, C.; Neumann, P.; Schweiger, O.; Kunin, W.E. Global Pollinator Declines: Trends, Impacts and Drivers. Trends Ecol. Evol. 2010, 25, 345–353. [Google Scholar] [CrossRef] [PubMed]
- Gallai, N.; Salles, J.-M.; Settele, J.; Vaissière, B.E. Economic Valuation of the Vulnerability of World Agriculture Confronted with Pollinator Decline. Ecol. Econ. 2009, 68, 810–821. [Google Scholar] [CrossRef]
- Liu, Z.; Chen, C.; Niu, Q.; Qi, W.; Yuan, C.; Su, S.; Liu, S.; Zhang, Y.; Zhang, X.; Ji, T. Survey Results of Honey Bee (Apis mellifera) Colony Losses in China (2010–2013). J. Apic. Res. 2016, 55, 29–37. [Google Scholar] [CrossRef]
- Vanengelsdorp, D.; Meixner, M.D. A Historical Review of Managed Honey Bee Populations in Europe and the United States and the Factors That May Affect Them. J. Invertebr. Pathol. 2010, 103, S80–S95. [Google Scholar] [CrossRef]
- Gisder, S.; Genersch, E. Special Issue: Honey Bee Viruses. Viruses 2015, 7, 5603–5608. [Google Scholar] [CrossRef]
- Higes, M.; Martín-Hernández, R.; Botías, C.; Bailón, E.G.; González-Porto, A.V.; Barrios, L.; del Nozal, M.J.; Bernal, J.L.; Jiménez, J.J.; Palencia, P.G.; et al. How Natural Infection by Nosema ceranae Causes Honeybee Colony Collapse. Environ. Microbiol. 2008, 10, 2659–2669. [Google Scholar] [CrossRef]
- Higes, M.; Martín, R.; Meana, A. Nosema ceranae, a New Microsporidian Parasite in Honeybees in Europe. J. Invertebr. Pathol. 2006, 92, 93–95. [Google Scholar] [CrossRef]
- Forsgren, E.; Wei, S.; Guiling, D.; Zhiguang, L.; Van Tran, T.; Tang, P.T.; Truong, T.A.; Dinh, T.Q.; Fries, I. Preliminary Observations on Possible Pathogen Spill-over from Apis mellifera to Apis cerana. Apidologie 2015, 46, 265–275. [Google Scholar] [CrossRef]
- Lin, Z.; Page, P.; Li, L.; Qin, Y.; Zhang, Y.; Hu, F.; Neumann, P.; Zheng, H.; Dietemann, V. Go East for Better Honey Bee Health: Apis cerana Is Faster at Hygienic Behavior than A. mellifera. PLoS ONE 2016, 11, e0162647. [Google Scholar] [CrossRef]
- Jensen, A.B.; Aronstein, K.; Flores, J.M.; Vojvodic, S.; Palacio, M.A.; Spivak, M. Standard Methods for Fungal Brood Disease Research. J. Apic. Res. 2013, 52, 1–20. [Google Scholar] [CrossRef]
- Pedersen, A.B.; Altizer, S.; Poss, M.; Cunningham, A.A.; Nunn, C.L. Patterns of Host Specificity and Transmission among Parasites of Wild Primates. Int. J. Parasitol. 2005, 35, 647–657. [Google Scholar] [CrossRef]
- Woolhouse, M.E.; Taylor, L.H.; Haydon, D.T. Population Biology of Multihost Pathogens. Science 2001, 292, 1109–1112. [Google Scholar] [CrossRef]
- Ai, H.; Yan, X.; Han, R. Occurrence and Prevalence of Seven Bee Viruses in Apis mellifera and Apis cerana Apiaries in China. J. Invertebr. Pathol. 2012, 109, 160–164. [Google Scholar] [CrossRef]
- Choe, S.E.; Nguyen, L.T.K.; Noh, J.H.; Koh, H.B.; Jean, Y.H.; Kweon, C.H.; Kang, S.W. Prevalence and Distribution of Six Bee Viruses in Korean Apis cerana Populations. J. Invertebr. Pathol. 2012, 109, 330–333. [Google Scholar] [CrossRef]
- Li, J.; Qin, H.; Wu, J.; Sadd, B.M.; Wang, X.; Evans, J.D.; Peng, W.; Chen, Y. The Prevalence of Parasites and Pathogens in Asian Honeybees Apis cerana in China. PLoS ONE 2012, 7, e47955. [Google Scholar] [CrossRef]
- Peng, W.; Li, J.; Boncristiani, H.; Strange, J.P.; Hamilton, M.; Chen, Y. Host Range Expansion of Honey Bee Black Queen Cell Virus in the Bumble Bee, Bombus huntii. Apidologie 2011, 42, 650–658. [Google Scholar] [CrossRef]
- Singh, R.; Levitt, A.L.; Rajotte, E.G.; Holmes, E.C.; Ostiguy, N.; Vanengelsdorp, D.; Lipkin, W.I.; Depamphilis, C.W.; Toth, A.L.; Cox-Foster, D.L. RNA Viruses in Hymenopteran Pollinators: Evidence of Inter-Taxa Virus Transmission via Pollen and Potential Impact on Non-Apis Hymenopteran Species. PLoS ONE 2010, 5, e14357. [Google Scholar] [CrossRef]
- Klee, J.; Besana, A.M.; Genersch, E.; Gisder, S.; Nanetti, A.; Tam, D.Q.; Chinh, T.X.; Puerta, F.; Ruz, J.M.; Kryger, P.; et al. Widespread Dispersal of the Microsporidian Nosema ceranae, an Emergent Pathogen of the Western Honey Bee, Apis mellifera. J. Invertebr. Pathol. 2007, 96, 1–10. [Google Scholar] [CrossRef]
- Carreck, N.L.; Ball, B.V.; Martin, S.J. Honey Bee Colony Collapse and Changes in Viral Prevalence Associated With Varroa destructor. J. Apic. Res. 2010, 49, 93–94. [Google Scholar] [CrossRef]
- Genersch, E. Honey Bee Pathology: Current Threats to Honey Bees and Beekeeping. Appl. Microbiol. Biotechnol. 2010, 87, 87–97. [Google Scholar] [CrossRef] [PubMed]
- Higes, M.; Martín-Hernández, R.; Garrido-Bailón, E.; González-Porto, A.V.; García-Palencia, P.; Meana, A.; Del Nozal, M.J.; Mayo, R.; Bernal, J.L. Honeybee Colony Collapse Due to Nosema ceranae in Professional Apiaries. Environ. Microbiol. Rep. 2009, 1, 110–113. [Google Scholar] [CrossRef] [PubMed]
- Phokasem, P.; Liuhao, W.; Panjad, P.; Yujie, T.; Li, J.; Chantawannakul, P. Differential Viral Distribution Patterns in Reproductive Tissues of Apis mellifera and Apis cerana Drones. Front. Vet. Sci. 2021, 8, 608700. [Google Scholar] [CrossRef] [PubMed]
- Holly, T.-J.; Kaspar, T.-J. The Asian Honey Bee (Apis cerana) Is Significantly in Decline. Bee World 2016, 93, 90–97. [Google Scholar]
- Nanetti, A.; Bortolotti, L.; Cilia, G. Pathogens Spillover from Honey Bees to Other Arthropods. Pathogens 2021, 10, 1044. [Google Scholar] [CrossRef]
- Nazaruddin, D.A. Granite Landforms of Samui Island (Southern Thailand) from Geoheritage, Geoconservation and Geotourism Perspectives. Int. J. Geoheritage Park. 2020, 8, 75–86. [Google Scholar] [CrossRef]
- James, R.R.; Skinner, J.S. PCR Diagnostic Methods for Ascosphaera Infections in Bees. J. Invertebr. Pathol. 2005, 90, 98–103. [Google Scholar] [CrossRef]
- Dobbelaere, W.; de Graaf, D.C.; Peeters, J.E. Development of a Fast and Reliable Diagnostic Method for American Foulbrood Disease (Paenibacillus larvae Subsp. Larvae) Using a 16S RRNA Gene Based PCR. Apidologie 2001, 32, 363–370. [Google Scholar] [CrossRef]
- Klee, J.; Tek Tay, W.; Paxton, R.J. Specific and Sensitive Detection of Nosema Bombi (Microsporidia: Nosematidae) in Bumble Bees (Bombus Spp.; Hymenoptera: Apidae) by PCR of Partial RRNA Gene Sequences. J. Invertebr. Pathol. 2006, 91, 98–104. [Google Scholar] [CrossRef]
- Tay, W.T.; O’mahony, E.M.; Paxton, R.J. Complete RRNA Gene Sequences Reveal That the Microsporidium Nosema Bombi Infects Diverse Bumblebee (Bombus Spp.) Hosts and Contains Multiple Polymorphic Sites. J. Eukaryot. Microbiol. 2005, 52, 505–513. [Google Scholar] [CrossRef]
- Ravoet, J.; Maharramov, J.; Meeus, I.; De Smet, L.; Wenseleers, T.; Smagghe, G.; de Graaf, D.C. Comprehensive Bee Pathogen Screening in Belgium Reveals Crithidia mellificae as a New Contributory Factor to Winter Mortality. PLoS ONE 2013, 8, e72443. [Google Scholar] [CrossRef]
- Runckel, C.; Flenniken, M.L.; Engel, J.C.; Ruby, J.G.; Ganem, D.; Andino, R.; DeRisi, J.L. Temporal Analysis of the Honey Bee Microbiome Reveals Four Novel Viruses and Seasonal Prevalence of Known Viruses, Nosema, and Crithidia. PLoS ONE 2011, 6, e20656. [Google Scholar] [CrossRef]
- Gauthier, L.; Tentcheva, D.; Tournaire, M.; Dainat, B.; Cousserans, F.; Colin, M.E.; Bergoin, M. Viral Load Estimation in Asymptomatic Honey Bee Colonies Using the Quantitative RT-PCR Technique. Apidologie 2007, 38, 426–435. [Google Scholar] [CrossRef]
- Benjeddou, M.; Leat, N.; Allsopp, M.; Davison, S. Detection of Acute Bee Paralysis Virus and Black Queen Cell Virus from Honeybees by Reverse Transcriptase PCR. Appl. Environ. Microbiol. 2001, 67, 2384–2387. [Google Scholar] [CrossRef]
- Ribière, M.; Triboulot, C.; Mathieu, L.; Aurières, C.; Faucon, J.-P.; Pépin, M. Molecular Diagnosis of Chronic Bee Paralysis Virus Infection. Apidologie 2002, 33, 339–351. [Google Scholar] [CrossRef]
- Chen, Y.; Smith, I.; Collins, A.; Pettis, J.S.; Feldlaufer, M. Detection of Deformed Wing Virus Infection in Honey Bees, Apis mellifera L., in the United States. Am. Bee J. 2004, 144, 557–559. [Google Scholar]
- Zioni, N.; Soroker, V.; Chejanovsky, N. Replication of Varroa Destructor Virus 1 (VDV-1) and a Varroa Destructor Virus 1–Deformed Wing Virus Recombinant (VDV-1–DWV) in the Head of the Honey Bee. Virology 2011, 417, 106–112. [Google Scholar] [CrossRef]
- Yang, B.; Peng, G.; Li, T.; Kadowaki, T. Molecular and Phylogenetic Characterization of Honey Bee Viruses, Nosema Microsporidia, Protozoan Parasites, and Parasitic Mites in China. Ecol. Evol. 2013, 3, 298–311. [Google Scholar] [CrossRef]
- Stoltz, D.; Shen, X.-R.; Boggis, C.; Sisson, G. Molecular Diagnosis of Kashmir Bee Virus Infection. J. Apic. Res. 1995, 34, 153–160. [Google Scholar] [CrossRef]
- Chen, Y.; Zhao, Y.; Hammond, J.P.; Hsu, H.; Evans, J.D.; Feldlaufer, M.F. Multiple Virus Infections in the Honey Bee and Genome Divergence of Honey Bee Viruses. J. Invertebr. Pathol. 2004, 87, 84–93. [Google Scholar] [CrossRef]
- Manley, R.; Boots, M.; Wilfert, L. Condition-Dependent Virulence of Slow Bee Paralysis Virus in Bombus terrestris: Are the Impacts of Honeybee Viruses in Wild Pollinators Underestimated? Oecologia 2017, 184, 305–315. [Google Scholar] [CrossRef] [PubMed]
- Evans, J.D.; Pettis, J.S. Colony-Level Impacts of Immune Responsiveness in Honey Bees, Apis mellifera. Evolution 2005, 59, 2270–2274. [Google Scholar] [CrossRef]
- Simone, M.; Evans, J.D.; Spivak, M. Resin Collection and Social Immunity in Honey Bees. Evolution 2009, 63, 3016–3022. [Google Scholar] [CrossRef] [PubMed]
- Folmer, O.; Black, M.; Hoeh, W.; Lutz, R.; Vrijenhoek, R. DNA Primers for Amplification of Mitochondrial Cytochrome c Oxidase Subunit I from Diverse Metazoan Invertebrates. Mol. Mar. Biol. Biotechnol. 1994, 3, 294–299. [Google Scholar] [PubMed]
- Hall, T.A. BioEdit: A User-Friendly Biological Sequence Alignment Editor and Analysis Program for Windows 95/98/NT. Nucleic Acids Symp. Ser. 1999, 41, 95–98. [Google Scholar]
- Kumar, S.; Stecher, G.; Li, M.; Knyaz, C.; Tamura, K. MEGA X: Molecular Evolutionary Genetics Analysis across Computing Platforms. Mol. Biol. Evol. 2018, 35, 1547–1549. [Google Scholar] [CrossRef]
- Tamura, K.; Nei, M. Estimation of the Number of Nucleotide Substitutions in the Control Region of Mitochondrial DNA in Humans and Chimpanzees. Mol. Biol. Evol. 1993, 10, 512–526. [Google Scholar] [CrossRef]
- Yang, S.; Gayral, P.; Zhao, H.; Wu, Y.; Jiang, X.; Wu, Y.; Bigot, D.; Wang, X.; Yang, D.; Herniou, E.A.; et al. Occurrence and Molecular Phylogeny of Honey Bee Viruses in Vespids. Viruses 2020, 12, 6. [Google Scholar] [CrossRef]
- Dolezal, A.G.; Hendrix, S.D.; Scavo, N.A.; Carrillo-Tripp, J.; Harris, M.A.; Wheelock, M.J.; O’Neal, M.E.; Toth, A.L. Honey Bee Viruses in Wild Bees: Viral Prevalence, Loads, and Experimental Inoculation. PLoS ONE 2016, 11, e0166190. [Google Scholar] [CrossRef]
- Šimenc, L.; Kuhar, U.; Jamnikar-Ciglenečki, U.; Toplak, I. First Complete Genome of Lake Sinai Virus Lineage 3 and Genetic Diversity of Lake Sinai Virus Strains from Honey Bees and Bumble Bees. J. Econ. Entomol. 2020, 113, 1055–1061. [Google Scholar] [CrossRef]
- Malfroy, S.F.; Roberts, J.M.K.; Perrone, S.; Maynard, G.; Chapman, N. A Pest and Disease Survey of the Isolated Norfolk Island Honey Bee (Apis mellifera) Population. J. Apic. Res. 2016, 55, 202–211. [Google Scholar] [CrossRef]
- Čukanová, E.; Moutelíková, R.; Prodělalová, J. First Detection of Lake Sinai Virus in the Czech Republic: A Potential Member of a New Species. Arch. Virol. 2022, 167, 2213–2222. [Google Scholar] [CrossRef]
- Shojaei, A.; Nourian, A.; Khanjani, M.; Mahmoodi, P. The First Molecular Characterization of Lake Sinai Virus in Honey Bees (Apis mellifera) and Varroa destructor Mites in Iran. J. Apic. Res. 2021, 1–7. [Google Scholar] [CrossRef]
- Bigot, D.; Dalmon, A.; Roy, B.; Hou, C.; Germain, M.; Romary, M.; Deng, S.; Diao, Q.; Weinert, L.A.; Cook, J.M.; et al. The Discovery of Halictivirus Resolves the Sinaivirus Phylogeny. J. Gen. Virol. 2017, 98, 2864–2875. [Google Scholar] [CrossRef]
- Ravoet, J.; De Smet, L.; Wenseleers, T.; de Graaf, D.C. Genome Sequence Heterogeneity of Lake Sinai Virus Found in Honey Bees and Orf1/RdRP-Based Polymorphisms in a Single Host. Virus Res. 2015, 201, 67–72. [Google Scholar] [CrossRef]
- Holloway, J.D. Lepidoptera of Norfolk Island. Their Biogeography and Ecology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 1977; ISBN 978-90-6193-124-9. [Google Scholar]
- Ribière, M.; Olivier, V.; Blanchard, P. Chronic Bee Paralysis: A Disease and a Virus like No Other? J. Invertebr. Pathol. 2010, 103, S120–S131. [Google Scholar] [CrossRef]
- Daughenbaugh, K.F.; Martin, M.; Brutscher, L.M.; Cavigli, I.; Garcia, E.; Lavin, M.; Flenniken, M.L. Honey Bee Infecting Lake Sinai Viruses. Viruses 2015, 7, 3285–3309. [Google Scholar] [CrossRef]
- Békési, L.; Ball, B.V.; Dobos-Kovács, M.; Bakonyi, T.; Rusvai, M. Occurrence of Acute Paralysis Virus of the Honey Bee (Apis mellifera) in a Hungarian Apiary Infested with the Parasitic Mite Varroa jacobsoni. Acta Vet. Hung. 1999, 47, 319–324. [Google Scholar] [CrossRef]
- Kojima, Y.; Toki, T.; Morimoto, T.; Yoshiyama, M.; Kimura, K.; Kadowaki, T. Infestation of Japanese Native Honey Bees by Tracheal Mite and Virus from Non-Native European Honey Bees in Japan. Microb. Ecol. 2011, 62, 895–906. [Google Scholar] [CrossRef]
- Sanpa, S.; Chantawannakul, P. Survey of Six Bee Viruses Using RT-PCR in Northern Thailand. J. Invertebr. Pathol. 2009, 100, 116–119. [Google Scholar] [CrossRef]
- Chantawannakul, P.; de Guzman, L.I.; Li, J.; Williams, G.R. Parasites, Pathogens, and Pests of Honeybees in Asia. Apidologie 2016, 47, 301–324. [Google Scholar] [CrossRef]
- de Miranda, J.R.; Cordoni, G.; Budge, G. The Acute Bee Paralysis Virus–Kashmir Bee Virus–Israeli Acute Paralysis Virus Complex. J. Invertebr. Pathol. 2010, 103, S30–S47. [Google Scholar] [CrossRef] [PubMed]
- Mookhploy, W.; Kimura, K.; Disayathanoowat, T.; Yoshiyama, M.; Hondo, K.; Chantawannakul, P. Capsid Gene Divergence of Black Queen Cell Virus Isolates in Thailand and Japan Honey Bee Species. J. Econ. Entomol. 2015, 108, 1460–1464. [Google Scholar] [CrossRef] [PubMed]
- Yuan, C.; Jiang, X.; Liu, M.; Yang, S.; Deng, S.; Hou, C. An Investigation of Honey Bee Viruses Prevalence in Managed Honey Bees (Apis mellifera and Apis cerana) Undergone Colony Decline. Open Microbiol. J. 2021, 15, 58–66. [Google Scholar] [CrossRef]
- Hong, I.-P.; Woo, S.-O.; Choi, Y.-S.; Han, S.-M.; Kim, N.-S.; Kim, H.-K.; Han, S.-H.; Lee, M.-Y.; Lee, M.-L.; Byeon, K.-H. Prevalence of Nosema and Virus in Honey Bee (Apis mellifera L.) Colonies on Flowering Period of Acacia in Korea. Mycobiology 2011, 39, 317–320. [Google Scholar] [CrossRef]
- Han, B.; Weiss, L.M. Microsporidia: Obligate Intracellular Pathogens Within the Fungal Kingdom. Microbiol. Spectr. 2017, 5. [Google Scholar] [CrossRef]
- Fries, I.; Feng, F.; da Silva, A.; Slemenda, S.B.; Pieniazek, N.J. Nosema ceranae n. Sp. (Microspora, Nosematidae), Morphological and Molecular Characterization of a Microsporidian Parasite of the Asian Honey Bee Apis cerana (Hymenoptera, Apidae). Eur. J. Protistol. 1996, 32, 356–365. [Google Scholar] [CrossRef]
- Paxton, R.J.; Klee, J.; Korpela, S.; Fries, I. Nosema ceranae Has Infected Apis mellifera in Europe since at Least 1998 and May Be More Virulent than Nosema apis. Apidologie 2007, 38, 558–565. [Google Scholar] [CrossRef]
- Fries, I. Nosema ceranae in European Honey Bees (Apis mellifera). J. Invertebr. Pathol. 2010, 103, S73–S79. [Google Scholar] [CrossRef]
- Natsopoulou, M.E.; McMahon, D.P.; Doublet, V.; Bryden, J.; Paxton, R.J. Interspecific Competition in Honeybee Intracellular Gut Parasites Is Asymmetric and Favours the Spread of an Emerging Infectious Disease. Proc. R. Soc. B Biol. Sci. 2015, 282, 20141896. [Google Scholar] [CrossRef]
- Martín-Hernández, R.; Botías, C.; Bailón, E.G.; Martínez-Salvador, A.; Prieto, L.; Meana, A.; Higes, M. Microsporidia Infecting Apis mellifera: Coexistence or Competition. Is Nosema ceranae Replacing Nosema apis? Environ. Microbiol. 2012, 14, 2127–2138. [Google Scholar] [CrossRef]
- Chaimanee, V.; Warrit, N.; Chantawannakul, P. Infections of Nosema ceranae in Four Different Honeybee Species. J. Invertebr. Pathol. 2010, 105, 207–210. [Google Scholar] [CrossRef]
- Chaimanee, V.; Chen, Y.; Pettis, J.S.; Scott Cornman, R.; Chantawannakul, P. Phylogenetic Analysis of Nosema ceranae Isolated from European and Asian Honeybees in Northern Thailand. J. Invertebr. Pathol. 2011, 107, 229–233. [Google Scholar] [CrossRef]
- Chaimanee, V.; Chantawannakul, P.; Chen, Y.; Evans, J.D.; Pettis, J.S. Differential Expression of Immune Genes of Adult Honey Bee (Apis mellifera) after Inoculated by Nosema ceranae. J. Insect Physiol. 2012, 58, 1090–1095. [Google Scholar] [CrossRef]
- Chaimanee, V.; Pettis, J.S.; Chen, Y.; Evans, J.D.; Khongphinitbunjong, K.; Chantawannakul, P. Susceptibility of Four Different Honey Bee Species to Nosema ceranae. Vet. Parasitol. 2013, 193, 260–265. [Google Scholar] [CrossRef]
- Sinpoo, C.; Disayathanoowat, T.; Williams, P.H.; Chantawannakul, P. Prevalence of Infection by the Microsporidian Nosema Spp. in Native Bumblebees (Bombus spp.) in Northern Thailand. PLoS ONE 2019, 14, e0213171. [Google Scholar] [CrossRef]
- Suwannapong, G.; Maksong, S.; Yemor, T.; Junsuri, N.; Benbow, M.E. Three Species of Native Thai Honey Bees Exploit Overlapping Pollen Resources: Identification of Bee Flora from Pollen Loads and Midguts from Apis cerana, A. dorsata and A. florea. J. Apic. Res. 2013, 52, 196–201. [Google Scholar] [CrossRef]
- Suraporn, S.; Natsopoulou, M.E.; Doublet, V.; McMahon, D.P.; Paxton, R.J. Nosema ceranae Is Not Detected in Honey Bees (Apis spp.) of Northeast Thailand. J. Apic. Res. 2013, 52, 259–261. [Google Scholar] [CrossRef]

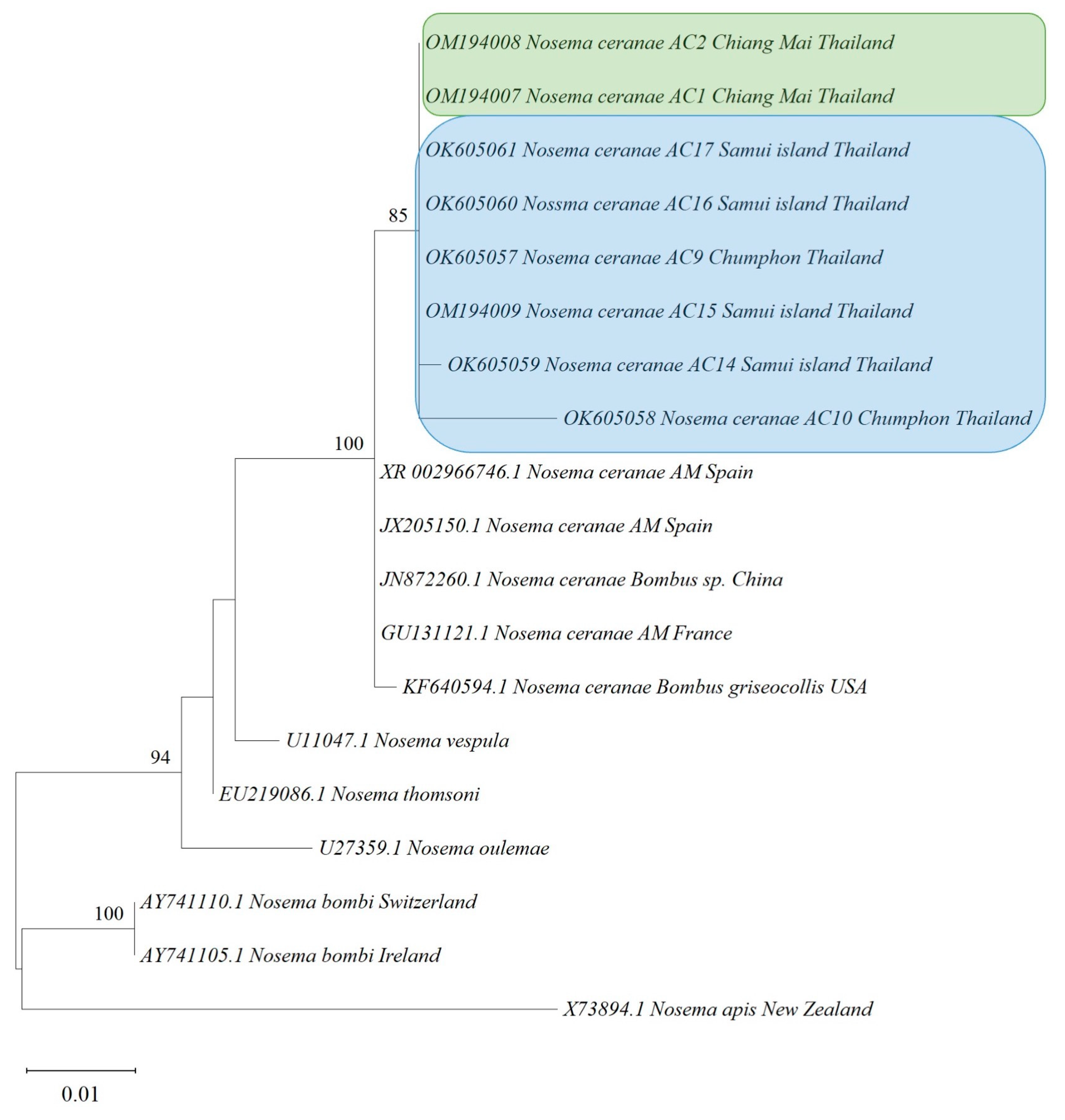
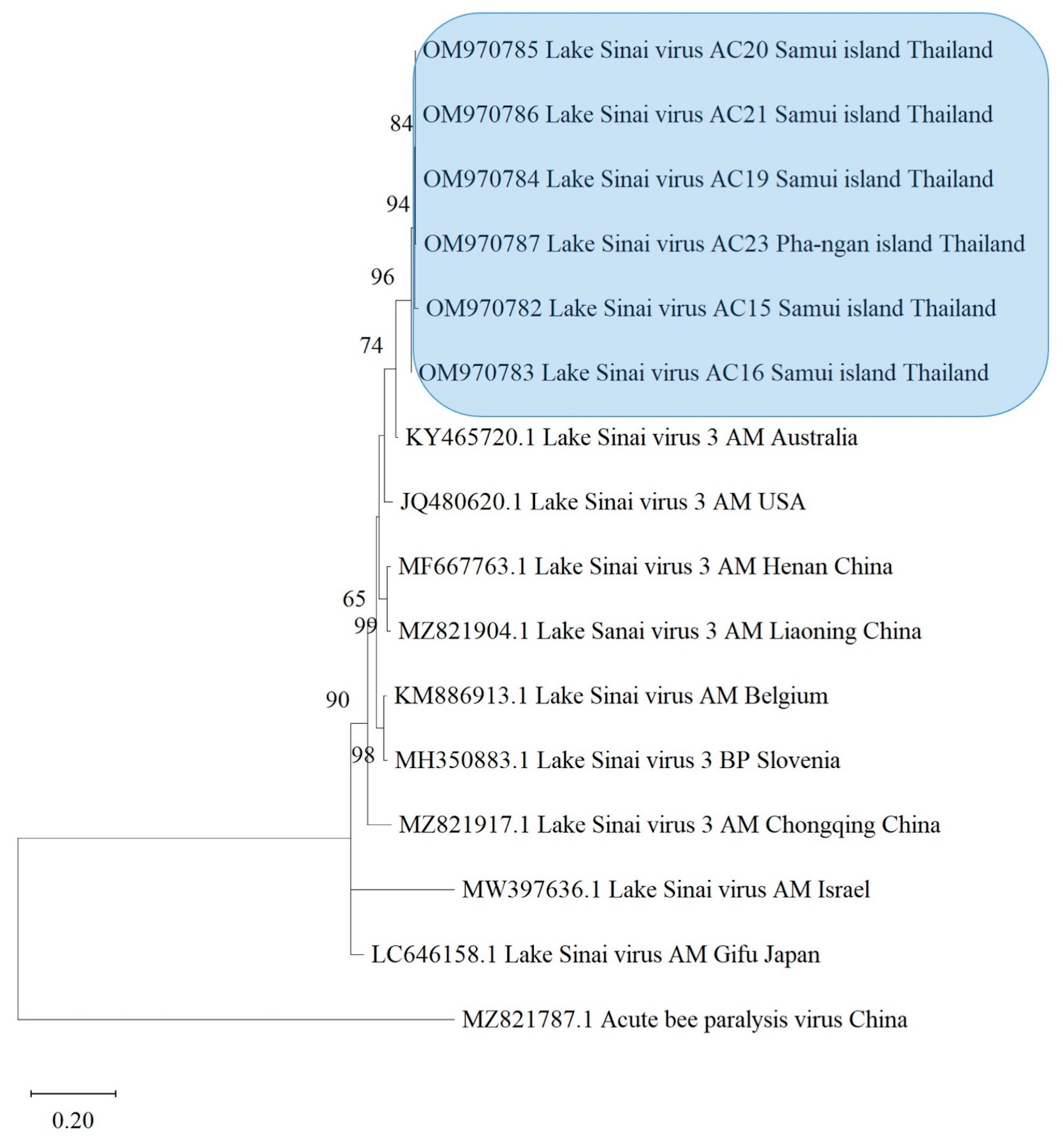
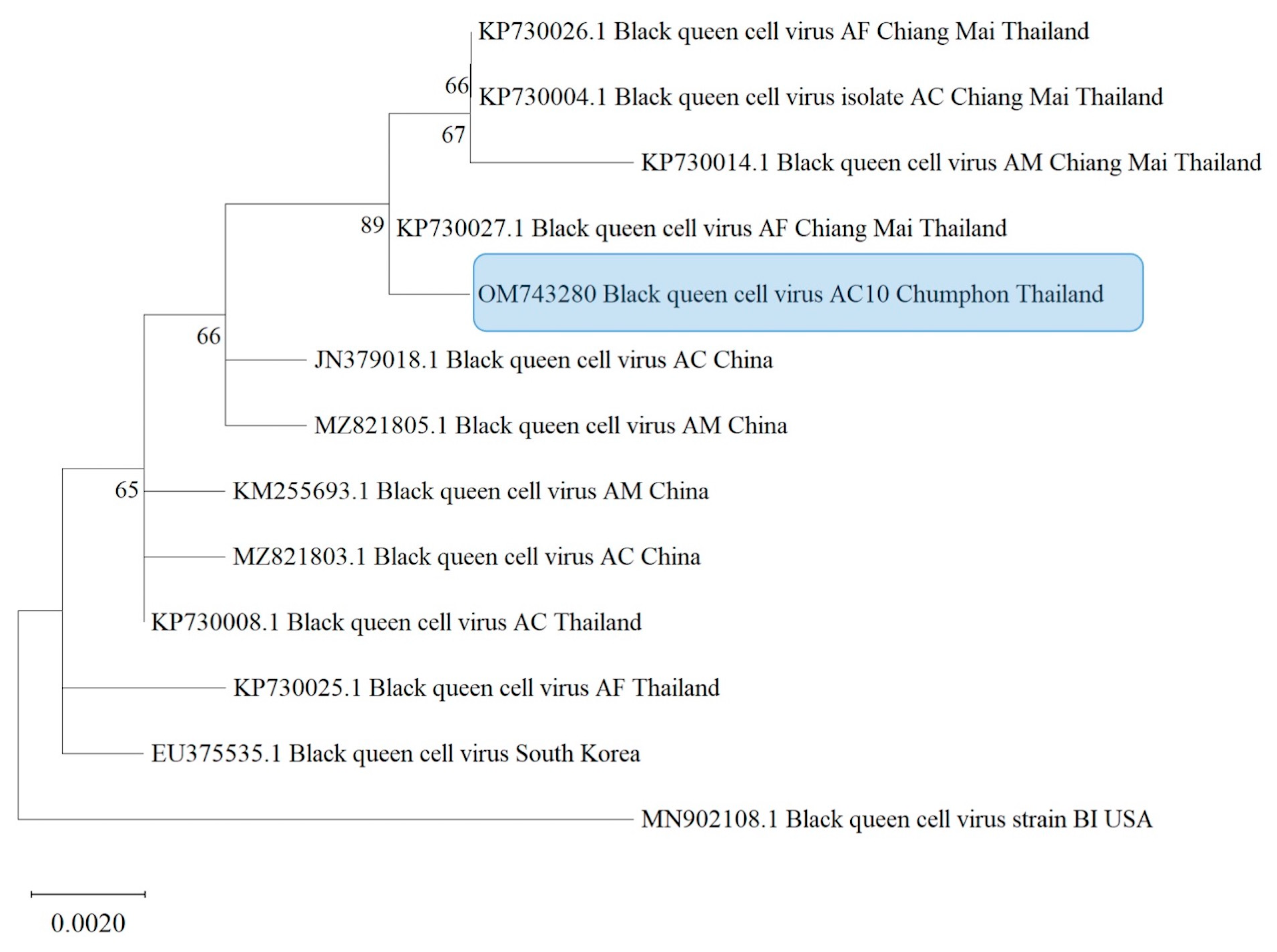
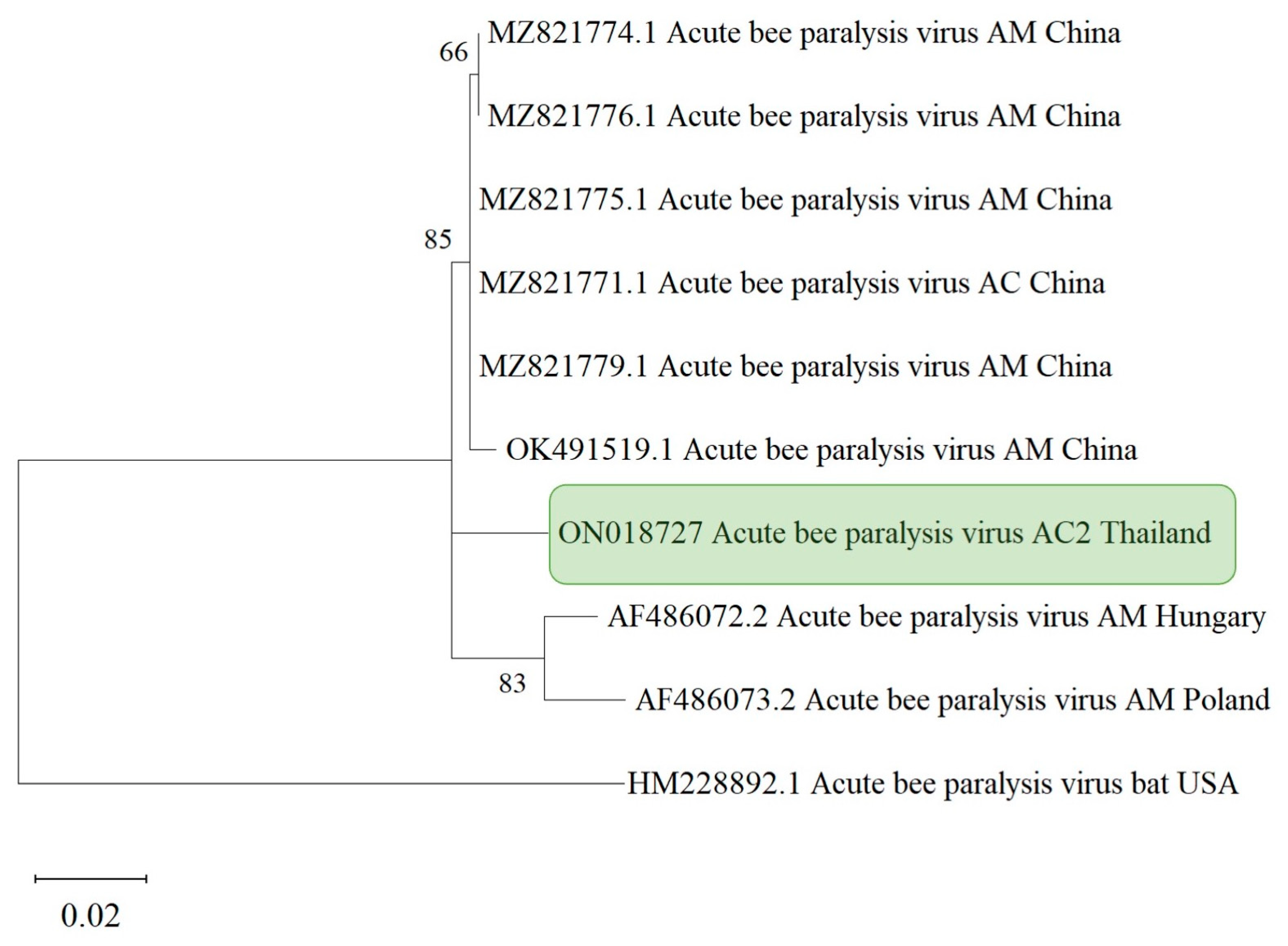
| Sample Code | Bacteria | Fungi | Microsporidians | Viruses | ||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| P. larvae | A. apis | N. apis | N. ceranae | N. bobi | C. bobi | LSV | ALPV | BSRV | ABPV | BQCV | CBPV | DWVA | DWVB | IAPV | KBV | SBV | SBPV | |
| AC1 | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC2 | - | - | - | + | - | - | - | - | - | + | - | - | - | - | - | - | - | - |
| AC3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC4 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC5 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC6 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC7 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC8 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC9 | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC10 | - | - | - | + | - | - | - | - | - | - | + | - | - | - | - | - | - | - |
| AC11 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC12 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC13 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC14 | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC15 | - | - | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - |
| AC16 | - | - | - | + | - | - | + | - | - | - | - | - | - | - | - | - | - | - |
| AC17 | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC18 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC19 | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - |
| AC20 | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - |
| AC21 | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - |
| AC22 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| AC23 | - | - | - | - | - | - | + | - | - | - | - | - | - | - | - | - | - | - |
| AC24 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Percentage (%) | 0% | 0% | 0% | 33% | 0% | 0% | 25% | 0% | 0% | 4% | 4% | 0% | 0% | 0% | 0% | 0% | 0% | 0% |
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2023 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Phokasem, P.; Sinpoo, C.; Attasopa, K.; Krongdang, S.; Chantaphanwattana, T.; Ling, T.C.; Pettis, J.S.; Chantawannakul, P.; Chaimanee, V.; Disayathanoowat, T. Preliminary Survey of Pathogens in the Asian Honey Bee (Apis cerana) in Thailand. Life 2023, 13, 438. https://doi.org/10.3390/life13020438
Phokasem P, Sinpoo C, Attasopa K, Krongdang S, Chantaphanwattana T, Ling TC, Pettis JS, Chantawannakul P, Chaimanee V, Disayathanoowat T. Preliminary Survey of Pathogens in the Asian Honey Bee (Apis cerana) in Thailand. Life. 2023; 13(2):438. https://doi.org/10.3390/life13020438
Chicago/Turabian StylePhokasem, Patcharin, Chainarong Sinpoo, Korrawat Attasopa, Sasiprapa Krongdang, Thunyarat Chantaphanwattana, Tial C. Ling, Jeffery S. Pettis, Panuwan Chantawannakul, Veeranan Chaimanee, and Terd Disayathanoowat. 2023. "Preliminary Survey of Pathogens in the Asian Honey Bee (Apis cerana) in Thailand" Life 13, no. 2: 438. https://doi.org/10.3390/life13020438
APA StylePhokasem, P., Sinpoo, C., Attasopa, K., Krongdang, S., Chantaphanwattana, T., Ling, T. C., Pettis, J. S., Chantawannakul, P., Chaimanee, V., & Disayathanoowat, T. (2023). Preliminary Survey of Pathogens in the Asian Honey Bee (Apis cerana) in Thailand. Life, 13(2), 438. https://doi.org/10.3390/life13020438









