Abstract
Complementary alternative medicine approaches are growing treatments of diseases to standard medicine practice. Many of these concepts are being adopted into standard practice and orthomolecular medicine. Age-related diseases, in particular neurodegenerative disorders, are particularly difficult to treat and a cure is likely a distant expectation for many of them. Shifting attention from pharmaceuticals to phytoceuticals and “bugs as drugs” represents a paradigm shift and novel approaches to intervention and management of age-related diseases and downstream effects of aging. Although they have their own unique pathologies, a growing body of evidence suggests Alzheimer’s disease (AD) and vascular dementia (VaD) share common pathology and features. Moreover, normal metabolic processes contribute to detrimental aging and age-related diseases such as AD. Recognizing the role that the cerebral and cardiovascular pathways play in AD and age-related diseases represents a common denominator in their pathobiology. Understanding how prosaic foods and medications are co-metabolized with the gut microbiota (GMB) would advance personalized medicine and represents a paradigm shift in our view of human physiology and biochemistry. Extending that advance to include a new physiology for the advanced age-related diseases would provide new treatment targets for mild cognitive impairment, dementia, and neurodegeneration and may speed up medical advancements for these particularly devastating and debilitating diseases. Here, we explore selected foods and their derivatives and suggest new dementia treatment approaches for age-related diseases that focus on reexamining the role of the GMB.
1. Introduction
Natural alternatives are being explored for high-risk medications treating age-related diseases. Age-related neurodegenerative diseases have few treatments and no effective cures to date. During aging, several diseases become more prevalent. Alzheimer’s disease is the most common neurodegenerative disease and vascular dementia is the second most common related dementia in America and their prevalence is increasing in other developed countries [1,2]. Due to its protracted and progressive nature, AD and other neurodegenerative conditions increase with age [3]. AD is a devastating disease of the elderly, which begins before prodromal signs, leads to a decline in cognitive and behavioral capacity, dwindles functional ability, hampers independence, and eventually results in death. The gut microbiota, prebiotics, probiotics, symbiotics, nutritional, or other approaches have the potential for modifying recalcitrant and debilitating conditions and may help diminish the deleterious effects of aging [4]. AD leads to progressive cognitive and behavioral decline in the elderly and with advancing age. Although there are treatments, the disappointing fact is that there are no real promising cures on the immediate horizon and thus, it is extremely important to focus on basic research and novel evidence-based therapeutics with proven potential benefits to delay the onset of this protracted disease. Alzheimer’s Disease International [5,6] estimates that the number of people living with dementia worldwide will increase to 65.7 million by 2030 and 115.4 million by 2050 [7] provided we live into later decades of life. DeKosky and colleagues estimated that even a delay of disease onset by 5 years could reduce the number of dementia cases by approximately 50% after several decades [8,9]. Nevertheless, DeKosky and colleagues did not find, in their work with Ginkgo 120 mg alone, which was less than the 240 in other trials, that it reduced cognitive decline in those aged 72–96 years of age [10]. However, even the slightest progress toward new treatments or discovery would be a welcome advance and one must not exclude the role of the MGB in brain health.
We know that the multifactorial nature of cognitive decline and AD or VaD from our work and that of many others can involve mitochondrial dysfunction and propagators, such as cytokine stress or oxidative stress in the cardiovascular system, brain, and CNS, including brain microvasculature and parenchyma, which results in the accumulation of damaging reactive oxygen species (ROS) [11] and reactive nitrogen species (RNS) in the aging brain [12]. These reactive species in cerebrovascular and other diseases promote leukocyte and immune cell adhesion and increase endothelial and other cellular compartment permeability, which results in chronic injury stimulus cascading in a host of downstream events [12]. One event is mitochondrial damage, which feeds forward oxidative stress and other propagators of downstream chemical modification [11,13]. Other cascades include changes in mitochondrial enzymes in the vascular wall and in brain parenchymal cells and imbalances in the activity of vasoactive substances, such as different isoforms of nitric oxide synthase, endothelin-1, various oxidative stress markers, and mitochondrial DNA damage [12,14].
When damage occurs to the vascular networks of the brain, oxygen and nutrients are no longer able to provide sustenance to vital cells in the brain, including glial cells and neurons. This damage rearranges the natural supporting framework for neurons and associated cells, causes irreversible neuronal changes, leads to dysfunction in the cerebrovascular networks, and eventually contributes to vascular dementia [15]. It is this vascular pathology that causes the detrimental effects in vascular dementia and, given that it often coexists with AD, targeting treatments at the vascular level may help ward off dementia and AD. Progressive cognitive decline and eventual death is still unavoidable at this time, once the disease has begun its course. Considering how much these deleterious changes affect a person’s life and that of the caregiver, the urgency to find an effective treatment is essential and not to be understated. However, as the pathogenesis of AD is still not certain, research is ongoing for new ways to tackle this disease, and, as frightening as it may be, there are few promising treatments or cures to date for AD and related dementias. Animal studies centering on tolerance to hypoxia and aglycemia have revealed that neuroprotection mechanisms through hibernation for AD and VaD are possible [16]. Perhaps hibernation mimetics could be established to offer the same protection in human subjects.
In examining the link between the heart, brain, and the vasculature, we find common potential treatment targets for age-related diseases. When we add the role the microbiota–gut–brain axis plays, as both a novel treatment target and as a contributor and metabolic modulator of disease, we build a case for a new physiology of the elderly. The elderly often cannot tolerate or metabolize drugs well, so we consider safe, natural, inexpensive, and effective alternatives to pharmaceuticals and current dementia treatments. If one turns to alternative treatments such as natural products, prosaic foods, exercise, and even higher education, we find these modalities are associated with protection against AD [17]. Prosaic foods and natural products have been used to treat age-related diseases in ancient and recent medicine. One natural product that has had a lot of interest is Ginkgo biloba (G. biloba). Ginkgo biloba, and its main constituent compounds, the ginkgolides, have become something of a celebrity as far as natural drug candidates go. This treatment strategy stems from extensive research that supports the neuroprotective properties of the ginkgolides and their ability to enhance cognitive function and show promise against anxiety in dementia and other psychiatric disorders [18]. The role of the gut microbiota in cardiovascular disease and aortic stiffness is highlighted by trimethylamine-N-oxide, or TMANO, which is linked to specific microbiota and induces aortic stiffening and increases systolic blood pressure with aging in mice and humans in atherosclerosis [19]. TMANO is an oxidation product of trimethylamine, which is transformed from the metabolism of choline by the gut microbiota through dietary phosphatidylcholine. TMANO plasma concentrations depend on the gut microbial composition, but mostly on diet and the activity of the liver flavin monooxygenase [20,21,22].
2. Pathogenesis of Alzheimer’s Disease/Vascular Dementia and Effects of Ginkgo biloba
Alzheimer’s disease is characterized by the formation of neurofibrillary tangles [23] and the accumulation of abnormal amyloid beta (Aβ) peptide or smaller soluble Aβ oligomers and their deposition in senile and neuritic plaques [24] and in brain parenchyma and cerebral capillaries [25]. AD is largely a protracted disease, but the exact etiology is largely unknown. Nevertheless, age is implicated to be the strongest risk factor and the presence of a first-order family member also increases risk significantly for developing AD [9].
However, these hallmark lesions are mostly end-stage changes and their protracted nature is indicative of the disease, but are not sufficient and necessary for dementia to occur, which suggests earlier mechanisms are involved well before any prodromal stages [17,26]. The role of natural compounds such as Ginkgo biloba on immunomodulation, inflammation, and cardiovascular homeostasis have been demonstrated to include the antagonist activity of the platelet activating factor, by reducing the infiltration of eosinophils and neutrophils and alleviating neuroinflammatory injury by inhibiting astrocytic Lipocalin-2 expression after ischemic brain injury and stroke [27]. Taken together, this provides more evidence to support AD as a vascular disorder [28].
One of the major potential therapeutic indications for the experimental use of Ginkgo biloba extract EGb761 is its ability to protect against Aβ-induced neurotoxicity and neuroinflammation, a key to pathology in Alzheimer’s disease, which also includes cerebral amyloid angiopathy [29]. Moreover, there is a vascular and cerebrovascular–brain axis in AD including VaD since vascular dementia and cardiovascular disease implicate shared vascular mechanisms in the development and/or progression of AD [30,31,32,33]. In the development of this disease, the Aβ deposition in the brain causes a reactionary stimulation of the body’s defense system. In this defense, resident microglia/infiltration macrophages become activated to clear the deposition and concomitantly produce a range of inflammatory mediators that are potentially neurotoxic, which contributes to the pathogenesis of AD [34,35,36]. In a study analyzing the abnormal amyloid beta peptide formation, it was found that Ginkgo biloba has an anti-amyloid aggregation effect due to its effect on increasing transthyretin RNA levels, an amyloid beta peptide transporter that decreases amyloid brain deposition [9]. Moreover, amyloid oligomers and downstream fibril formation can be affected or arrested in many ways that are not immediately evident in a clinical study, due to our inability to image such changes in vivo [37]. Many studies in rats and in vitro reveal an alternate method of fighting Aβ accumulation by modulating alpha-secretase, the enzyme responsible for cutting the amyloid beta peptide, and preventing the final fragments from being made [38]. Further, in human subjects, Ginkgo biloba was found to have utility in the treatment of cerebral insufficiency due to inflammatory neurotoxicity-induced dementia [39], whether due to degenerative processes or vascular insufficiency [40].
Another hallmark lesion in AD is the hyperphosphorylation of the microtubule-associated protein tau [41]. To mimic AD-like pathological conditions and memory deficits, hyperhomocysteinemia (HHcy) rat models were employed. Low folate raises blood HHcy concentrations, which are associated with poor cognitive performance in the general population. Elevated HHcy has neurotoxic and vasotoxic effects in AD and dementia and can be considered an early marker of cognitive decline [42]. HHcy is implicated in many age-related diseases, including neurologic diseases AD and VaD, cardiovascular disease, and ocular disorders such as macular degeneration and diabetic retinopathy, but has a less defined role in vascular dementia. Further, HHcy induces inflammatory-mediated blood–brain barrier (BBB) dysfunction through the activation of glutamate receptor N-methyl-d-Aspartate receptor [43]. HHcy-induced brain inflammation is a mechanism of BBB dysfunction and other immune-privileged barriers [43]. HHcy can also induce oxidative stress, which contributes to endothelial and arterial damage, increases tau microtubule-associated protein phosphorylation, and promotes blood clot formation, all of which are comorbidities of aging. While these models do not result in senile plaque deposition or intracellular inclusions or recapitulate the human condition, rodent models are the best approximation for in vivo changes. DNA damage with apoptosis is another protracted hallmark of AD pathobiology [44,45,46].
A hallmark lesion in AS is the hyperphosphorylation of tau protein. To explore the phosphorylation condition, rats were injected with EGb761 or saline as a sham control [47] and the status of oxidative damage, spatial/learning memory, level of memory-related proteins, tau phosphorylation, level/activity of tau kinase (GSK-β), and protein phosphatase 2A (PP2A) were all measured and reported. This particular group [47] showed that EGb761 could significantly antagonize HHcy-induced oxidative damage and recover PP2Ac and GSK3 β activities deregulated by HHcy. Not only did EGb761 protect against Aβ-induced neurotoxicity, but this study gave promising results with EGb761 for treating AD by decreasing hyperphosphorylation and demonstrating antioxidative activity [47]. When analyzing the abnormal amyloid beta peptide formation, it was found that Ginkgo biloba has an anti-amyloid aggregation effect due to its ability to increase transthyretin RNA levels, an amyloid beta peptide transporter that decreases amyloid brain deposition [9,23]. In vitro, Ginkgo biloba extract (GLE) provided protection from abnormal Aβ peptide accumulation with deposition in the brain parenchyma and cerebral capillaries [25]. However, native amyloid precursor protein is not sufficient or necessary for AD pathology alone [48] and the microtubule-associated protein tau is another mechanism believed to cause AD pathology [49]. The effects of Ginkgo biloba on beta secretase or the cholinergic system as well as increasing neurotransmitters should be further explored as they are both associated with AD [50,51]. In that regard, in studies aimed at improving cognitive function and control, Ginkgo biloba extracts were found to increase levels of acetylcholine, dopamine, and 5-hydroxytryptamine as well as to inhibit the activation of acetylcholinesterase, which is responsible for degrading acetylcholine [52]. The use of cholinergic agents, such as acetylcholinesterase (AChE) inhibitors, is currently being explored and these agents have shown efficacy and considerable benefit in AD and VaD therapy [53]. AChE inhibitors include donepezil, rivastigmine, and galantamine, as well as active compounds from the herb huperzine [53]. This accumulating evidence reveals that cholinergic deficiency contributes to vascular dementia and disease, which can be modulated by drugs such as Z-ligustilide [54,55]. Other natural products such as Hwangryunhaedok-tang (HT) [56], black tea, green tea, and coffee contain compounds that inhibit acetylcholinesterase and green tea metabolites also inhibit beta secretase, which could prevent the release of Aβ peptides from amyloid precursor protein. However, coffee is a less effective inhibitor of acetylcholinesterase, having no butyrylcholinesterase- or beta secretase-inhibiting activity [57].
The popularity for the use of Ginkgo biloba stems in part from the finding that it largely has no major adverse events or reactions or known side effects, to date, other than those due to the antagonism of clotting mechanisms, which should be a contraindication in some trials. Moreover, one study showed that when compared to donepezil monotherapy, the adverse event rate was lower in the Ginkgo biloba group and even under the combination group when compared to a placebo [58]. Contrary to the expectation that taking two drugs simultaneously would increase adverse events, this study had the opposite effect. In fact, improvement in adverse reactions with donepezil was observed when taken in conjunction with Ginkgo biloba extracts. These findings would be expected to lead to renewed interest in this area toward developing Ginkgo-derived compounds as therapeutics for the treatment of dementia. Nutrition companies looking towards commercializing this new discovery can help fund clinical trials and help this newly developed research accumulate more evidence-based research to support the claims of treatment success.
Amyloid β deposition is associated with the pathogenesis of AD, in particular tissue damage in the brain and associated parenchymal cells and neurons. Oxidative stress and neuroinflammation are associated with the damage caused by this pathology, which contributes significantly to dementia and the deleterious symptoms of AD. One of the cytokines found to increase with AD is CD38. This cytokine, expressed by neurons, astrocytes, and microglial cells, is responsible for regulating the repair and inflammatory processes within the brain [24], among other actions, degrades nicotine adenine dinucleotide (NAD), and regulates the migration of inflammatory cells during neuroinflammation [59,60]. One study examined CD38 expression on AD pathology in CD38-deficient mice and in vitro cultures. All treatments decreased secretions of Aβ from neuronal cultures, decreased the activity of β- and γ-secretase, the enzymes that cleave amyloid precursor protein, and significantly reduced Aβ plaque load and levels of insoluble Aβ [24]. This resulted in the attenuation of AD pathology and improved spatial learning and learning performance. The neuroprotective effects of CD38 inhibition suggest a novel therapeutic approach for AD with GLE. One mechanism of action for GLE and other flavonoids in AD involves attenuating CD38 neuroinflammation. Studies examining select flavonoids reported that, at low molecular levels, CD38 was inhibited by flavonoids, such as luteolinidin, kuromanin, and luteolin [61]. Other studies exploring kuromanin and luteolin found it could inhibit CD38 directly by affecting NAD levels in glycolysis [62]. Taken together, these findings suggest alternative roles for flavanols, such as those found in GLE, which have alternative mechanisms in modifying the hallmark lesions associated with AD pathology, including effects on neuronal metabolism and glyco-oxidative stress. Moreover, the microbiota also plays a key role in flavanol metabolism and must be considered when assigning any effect of GLE on the experimental model.
Also implicated in AD are mitogen-activated protein kinase (MAPK) signaling pathways. MAPK pathways represent a promising therapeutic target as they are implicated in inflammatory and apoptotic processes during cerebral ischemia and reperfusion injury as well [63,64]. Inhibitors of MAPK pathways are being explored as a therapeutic strategy for ischemic stroke. Moreover, bilobalide, a predominant sesquiterpene trilactone constituent of Ginkgo biloba leaves, has been shown to exert powerful neuroprotective properties, which are closely related to both anti-inflammatory and anti-apoptotic pathways [63]. This group investigated the neuroprotective roles of bilobalide in middle cerebral artery occlusion and reperfusion and oxygen–glucose deprivation and reoxygenation models of cerebral ischemia/reperfusion injury. They attempted to confirm the hypothesis that the protective effects of bilobalide were through the modulation of pro-inflammatory mediators and MAPK pathways. Their data indicated that the neuroprotective effects of bilobalide on their cerebral injury model are associated with the inhibition of pro-inflammatory mediator production and the downregulation of JNK1/2 and p38 MAPK activation [63].
3. Safety of Ginkgo biloba, GLE Extracts, and Similar Compounds
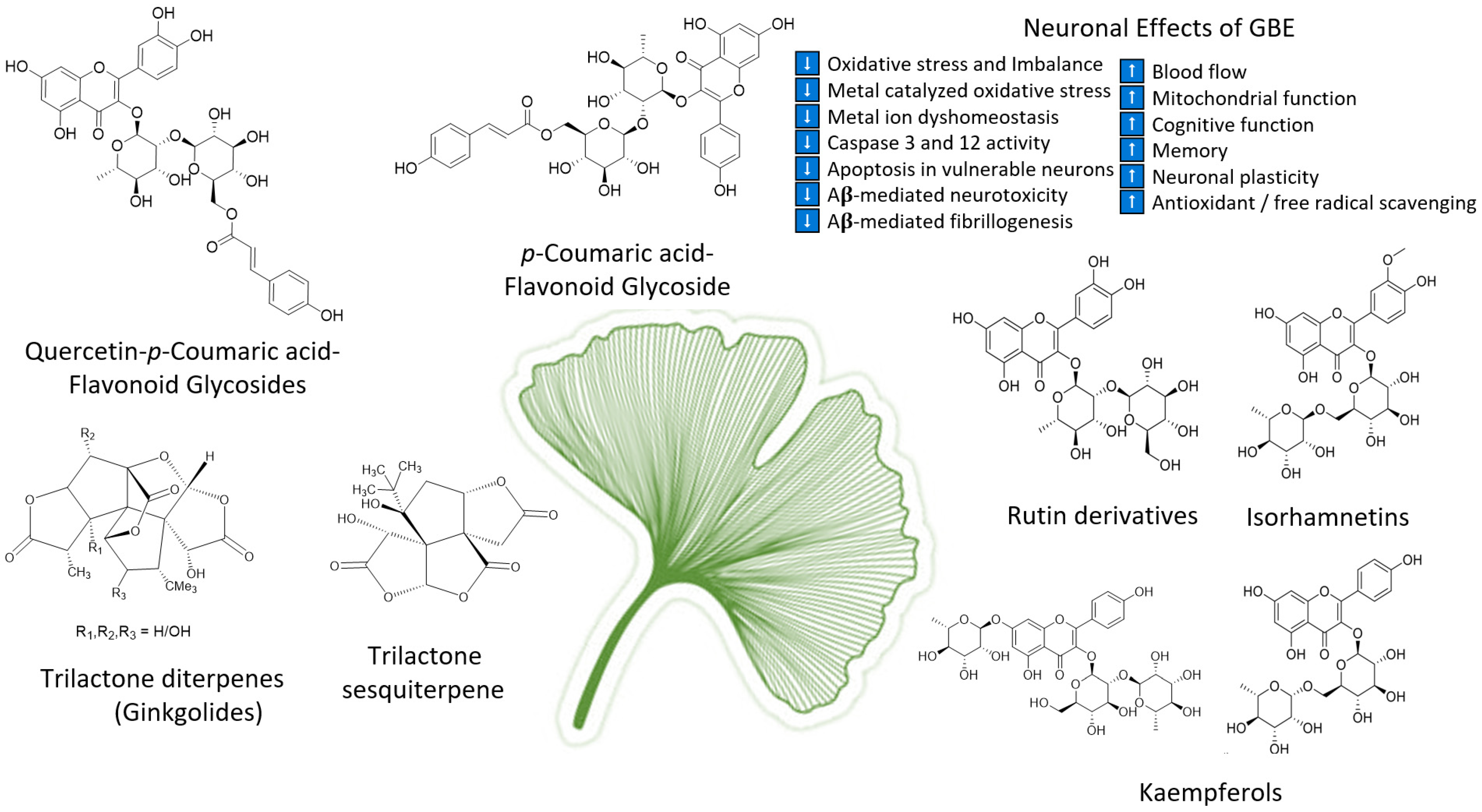
As the oldest extant deciduous tree, Ginkgo biloba, with its bilobed fan-shaped leaves, is known for its beauty, longevity, and resistance to pathogens [65]. In terms of longevity, it can live for over 1000 years [66]. Ginkgo biloba has a rich history for medicinal use dating back 5000 years [65]. Currently, it is a treatment option for acute ischemic stroke in China [39]. It was not until the mid-1960s that it was introduced into Western medicine by German physician–pharmacist Dr. Willmar Schwabe [65]. Standardized Ginkgo biloba leaf extract (EGb761) is typically prepared from whole dried green leaves [65], which contain many bioactive compounds. Two main components stand out and are largely responsible for GLE’s putative pharmacological effects (see Figure 1), namely, flavonoid glycosides and terpene trilactones, which represent 24% and 6% of the overall plant content, respectively [23,39]. Other potentially beneficial small molecules and phenolic acids, including aging, are present in Ginkgo biloba extracts, which are useful for the treatment of vascular cognitive impairment, vertigo, tinnitus, early diabetic retinopathy, and senile macular degeneration [39,65]. Ginkgo biloba leaf extract is also taken for the treatment of inflammation, asthma, and bronchitis [67,68].
Figure 1.
Structures of ginkgolides A, B, C (diterpenes), and bilobalide (sesquiterpene) and function of compounds found in G. biloba extract and EGb 761; proanthocyanidins, quercetins, coumarins, flavonoid glycosides, ginkgolides A, B, C-J, isorhamnetins, bilobalide, and rutin derivatives.
An important consideration for all of the superfood-related treatments in this work is bleeding risk, which is often associated with potential adverse interactions between some of the used herbal supplements and analgesic drugs with natural and prosaic foods, derivatives, and some of the so-called superfoods—in particular, ginkgo, garlic, ginger, bilberry, dong quai, ginseng, turmeric, and willow bark [69], as well as with those containing coumarin, i.e., chamomile, horse chestnut, fenugreek, and red clover. That said, taking the herbal or whole food extracts can limit the risk. Nevertheless, the whole food of Ginkgo biloba and its GLE extracts together with the interaction of Ginkgo biloba with non-steroidal anti-inflammatories could complicate the bleeding risk, since the combined use may induce increased bleeding risk to sensitive patients. However, these risks were not exclusory in the studies elucidated. Further, it is known that the use of the whole Ginkgo biloba extract and not simply the fractions can be the better form for consumption due to the synergic effect that occurs when consuming the different fractions together. Interestingly, the hibernating ground squirrel has a defect in clotting, which may offer clues into the neuroprotective mechanisms or mimic natural compounds such as those discussed [70,71].
Commercial Ginkgo biloba standardized extracts are available in the US market as coated tablets for oral administration [65] with a recommended daily dose of 240 mg, either taken in one or two doses. One of the most important contraindications when supplementing orally with Ginkgo biloba is counter indicated for anyone receiving anticoagulation, antiplatelet therapy, or for those with bleeding disorders [65]. This is due to the ability of Ginkgo biloba to form free platelets by antagonizing platelet-activating factors [39] and inhibiting platelet aggregation [65]. Administration is contraindicated in children and during pregnancy and nursing because of the potential adverse effects [65].
The safety and efficacy of ginkgolides in AD and VaD can be found in a Ginkgo biloba randomized 400 patient clinical trial of 50 years or older with VaD or AD who were given a special extract EGb761 or placebo for 22 weeks [72]. Patients who scored below 36 on the test for the early detection of dementia, with discrimination from depression, and who also scored between 9 and 23 on the Short Syndrome Test battery and at least 5 on the Neuropsychiatric Inventory were chosen for this particular trial. The drug was well tolerated and the adverse events were no greater than those of the placebo treatment according to this clinical trial, which focused on safety. Moreover, the treatment was found to improve cognitive functioning and behavior symptoms in patients with neuropsychiatric features and age-associated cognitive impairment or mild to moderate dementia. Since the treatment results were essentially similar for the AD and VaD subgroups, the data supports the safety and efficacy of EGb761 in the treatment of cognitive and non-cognitive symptoms of dementia overall, notwithstanding any interference with clotting [72]. A clinical trial examined the efficacy of Ginkgo biloba on the neuropsychological functioning of cognitively intact adults aged 60 years or older assigned randomly to receive either 180 mg/day of GLE or a placebo daily for 6 weeks. Compared to the placebo group, the group taking GLE rated their overall abilities as “improved”. This study harmonizes evidence for the potential efficacy of GLE.
However, the findings on Ginkgo are mixed. A larger study of three thousand and sixty-nine community volunteers 75 years or older conducted from a total of five academic medical centers in the United States revealed that 120 mg of GLE twice a day was not effective in reducing the overall incidence of dementia. In this study, five hundred and twenty-three individuals developed dementia with 92% classified as possible or probable AD or VaD with evidence of vascular disease of the brain, offering hope for the vasculopathies in AD and VaD [73]. We do not know enough about why the outcomes were mixed, but poor diet in the elderly and the co-metabolism of the host with select gut microbiota can contribute or modulate the pathogenesis of AD and VaD, which could explain any variant results. Nevertheless, an association was found between gut microbial composition and arterial stiffness and the GM has an influence on the aging of the vasculature [74]. Therefore, one must consider the impact a poor diet has on the gut microbiota composition and overall health in the elderly [75]. It was observed that in the elderly population over 100 years of age, centenarians, the structure of their human gut microbiota greatly differs from that of young adults and people 70 years of age. Centenarians were described as having an increased number of facultative anaerobes leading to an increased inflammatory status [76].
4. Blood Flow and a Leaky Gut, Leaky Brain in AD, VaD, and Aging
We have shown a clear connection for VaD and AD centering on G-protein-coupled receptor kinase 2 (GRK2) and AD [31], which was the basis for the concept of the heart–brain connection in vascular AD, vascular dementia, and other forms of Alzheimer’s disease [26]. Moreover, there are several metabolite-sensing G-protein-coupled receptors that bind with short-chain fatty organic acids (SCFAs), which are important for overall gut health and immune response regulation [77].
The mechanism of action of Ginkgo is largely to improve brain perfusion or blood flow. Central to blood flow is the integrity and sensitivity of blood-privileged immune barriers to the environment around it. The BBB and/or gastrointestinal barrier can experience disruption and increased permeability due to surrounding inflammation or other stressors, including oxidative stress, possibly through mitochondrial dysfunction or changes [26,78]. Each immune-privileged barrier is important for the protection of vital organs from pathogens and other assaults; a natural immune barrier maintains this separation from the rest of the body. These immune barriers also function in cellular communication and movement of nutrients and other factors. Moreover, the immune-privileged barriers, including the gastrointestinal tract epithelial barrier and blood–brain barrier, become significantly more permeable with advancing age [79]. As previously stated, this can may make the CNS more susceptible to neurotoxins generated by pathogenic resident or environmental microbiota (45), especially during chronic infection. Aging and inflammation progressively alter BBB permeability and can facilitate increased cerebral viral infection and opportunistic pathogen colonization as we age [80]. This is not surprising given that vascular-type dementias are the second most common form of dementia after Alzheimer’s disease [52,81] and both AD and VaD are age-related diseases. Moreover, cerebrovascular changes and associated amyloid beta peptide deposition in the brain, and elsewhere, are due, in part, to the leakiness of the blood–brain barrier [78,82]. Moreover, the role of the gut in dysbiosis or the production of xenobiotic molecules could affect the CNS and brain at both the blood–brain barrier interface [83] and from gut dysbiosis, which could be a cause and a consequence of increased levels of oxidative stress, since anaerobes thrive in the presence of electron acceptors [84].
The clear role of mitochondria in cerebrovascular hypoperfusion and models demonstrate that dysfunction is characterized by cerebral hypoperfusion and loss of the BBB integrity, and these occur before any signs of frank AD and in the prodromal stages [31]. After this stage, the pathogenesis is associated with increased Aβ deposition in the cerebral vessel walls and elsewhere. Concomitantly, or due to hypoperfusion injury, mitochondrial damage ensues along with increased oxidative stressors and the production of ROS/RNS and the release of mitochondrial apoptotic proteins and consequent induction of brain endothelial cell degeneration and death [85]. These changes contribute to the downstream induction and activation of nitric oxide [86] and of the transcription factor hypoxia-inducible factor-1α [87], which regulates the response to hypoxic conditions, which are involved in amyloid processing [88] and amyloidogenic mechanisms involving amyloid-β precursor protein and eventually aberrant Aβ production [89].
In order to fully understand how Ginkgo biloba can play a role in the treatment of AD and VaD, it is important to understand how blood flow and our gut epithelial and blood–brain barriers may be affected. When there is a disruption of blood flow to any area in the body through occlusive ischemia, such as thromboembolism or strangulation, the affected area either perishes or experiences a reintroduction of blood flow when the disruption is corrected. However, this reperfusion causes injury when the reintroduction of blood flow leads to the production of reactive oxygen species (ROS) and reactive nitrogen species (RNS) and free radical formation and propagation. As blood flow decreases in high oxygen-requiring tissues, especially the brain, ROS and RNS accelerate the pathological changes in some diseases including AD [9] and damages protein, nucleic acids, and lipids, particularly the vulnerable hippocampal neurons, the vasculature, and the BBB [78,90,91]. These are all, in part, also involved in the breakdown of the blood–brain barrier and/or gastrointestinal blood barrier, which contributes to the so-called leaky gut and leaky brain [78]. Interestingly, research shows that Ginkgo biloba enhances cerebral blood flow [39], scavenges free radicals [92], dilates blood vessels [9], decreases blood viscosity and erythrocyte deformability [9], and can improve BBB integrity, likely by quenching oxidative stress, since Ginkgo biloba has demonstrated antioxidant properties [93,94]. Targeting oxidative stress, one of the many components in the dementia-developing brain, suggests additional support for Ginkgo biloba by affecting the underlying mechanisms of pathology in AD and VAD.
During systemic inflammation, often due to infection, not unlike neurologic disease, the changes in the blood–brain barrier include abnormal sensitivity to the effects of systemic inflammation [95]. The effects of systemic inflammation are modulated by cytokines, interleukins (IL), neutrophils, leucocytes, complement cascade activation, antibody complexes, T cells, and/or other cell-mediated immune responses. Together, the gut–blood and blood–brain barriers provide a diffusion-selective gate that helps monitor and prevent the entry of select substances and bacteria into host organs and tissues. Inflammation from cytokine stressors, such as lipopolysaccharide, can eventually affect the integrity of these barriers, which can affect brain health [96,97,98]. For example, interleukin-1 and IL-6 can increase cortisol release by the stimulation of the hypothalamic pituitary arm of the microbiota–gut–brain (MGB) axis, which is often exhibited in stressed or depressed patients [99]. In addition to the microbiome, the mycobiome participates in modulating the cytokine production [100]. Thus, cytokines produced at the gut level do reach the brain via the bloodstream. For example, in autism, it was found some molecules may cross the blood–brain barrier and modulate brain area stimulation [101]. This systemic inflammation has been associated with antibiotic use, depression [97], and psychiatric comorbidities that occur in irritable bowel disease [78]. As the disruption of the blood–brain barrier occurs due to surrounding inflammatory reactions, this so-called leaky brain now leads to increased cerebrospinal fluid (CSF) protein production and its translocation. As the leakiness continues, so does the movement of small molecules, antibiotics, and phagocytes into the brain [78,102]. Also linked to the gut microbiota is their access to peripheral blood, for example during sepsis and infections, which can lead to serious illness.
5. Barrier Breaches and the Microbiota
Natural immune barriers are important for the protection of our brain, where they maintain separation of the immune-privileged environments from pathogens and the rest of the body. With assaults, the BBB can break down or become leaky, resulting in bacterial translocation and infiltration [103]. Vascular-type dementias are the second most common form of dementia after Alzheimer’s disease [52,81]. AD and VaD share cerebrovascular changes and with (Aβ) peptide deposition in the brain. The importance of breaching the tight barriers in the gastrointestinal system and blood–brain barrier is their link to alterations in the gut microbiota and the association with neurodegenerative diseases [104]. We suggest that a leaky gut can predispose the individual to increased blood–brain barrier permeability and promote neuroinflammation. Evidence now suggests this is due, in part, to leakiness of the BBB [78,82]. Further, we have seen the translocation of Aβ, proteins, and cytokines, which are found distal to the brain and, at the same time, can enter the brain to cause damage to neurons through the disrupted BBB [105]. Moreover, alterations or breaches in the gut microbiota composition can increased gut–blood barrier permeability and lead to immune-activated downstream systemic inflammation. Examples of bacteria translocating in AD include Porphyromonas gingivalis, which has been observed passing from oral sites of infection via olfactory or trigeminal cranial nerves into the brains of neurodegenerative disease patients [106,107]. This example supports the notion that neurological diseases have a relationship with the microbiota [106]. Moreover, it is not simply bacteria, as the intracellular protozoan, Toxoplasma gondii, can cross the BBB and cause neurological dysfunction and encephalitis, which suggests that the microbiota may be influencing neurological function over time to contribute to pathogenesis [78,106].
In order to understand how systemic changes in immune-privileged organ breaches occur, we consider mechanisms of host barrier integrity [108]. The gut can influence the blood–brain barrier permeability by means of oxidative stress, cytokine stress, gastrointestinal-derived hormonal secretion, small molecules and metabolic cofactor production, and other inflammatory mechanisms [108]. It was this BBB pathology coupled with the expression of the transmembrane receptor for advanced glycation end-products (RAGE) that a mechanism for translocation and the Maillard reaction was found in AD patients. Moreover, Vienna and colleagues show that the accumulation of AGEs and glycation crosslinking of intimal arterial structural proteins and oxidative stress together with novel mechanisms mediated trimethylamine N-oxide-induced arterial stiffening [19]. For AD pathology, and working through a positive feedback loop, it was discovered that as amyloid beta peptide accumulated, RAGE expression increased, and as RAGE expression increased, so did the breakdown of the BBB’s integrity [25]. Aging and age-related diseases are inextricably linked to metabolism and caloric restriction is the only proven anti-aging intervention to date. What we have not considered fully is the role that the microbiota, also referred to as our second genome, has on our metabolism of superfoods, prosaic foods, or nutrition. The author has long maintained a role for the Maillard reaction, glycation, and oxidation coupled with metal catalyzed oxidation, called glycol-oxidative stress, components in AD pathogenesis [109] and there is now a form of AD called diabetes type IIIc. This form of AD inextricably links metabolism, pancreatogenic diabetes, and the exocrine and digestive properties of the pancreas to age-related diseases. In that regard, we can also add that the microbiota can play a role as part of the host microbiome interaction, namely, the microbiota–gut–brain–endocrine metabolic interactome [99].
Ginkgo biloba is gaining interest not only from a nutritional and biochemical perspective, but from a microbiota–gut–brain axis and leaky gut–leaky brain perspective as well. A potential role for Ginkgo biloba in RAGE receptor activation and expression and in BBB permeability was illustrated in an in vitro study that mimicked the BBB conditions found in Alzheimer’s disease patients. Examining the effects of EGb761, it was found that the upregulation of RAGE expression was significantly reversed with EGb761 [110]. Further dosing with EGb761 attenuated cell injury, apoptosis, generation of intracellular ROS, BBB leakiness, and RAGE expression [25]. This not only suggests an important progression in the steps to bring about change for Alzheimer’s patients cognitively, but suggests that these modalities may help stabilize the BBB and decrease the accumulation of Aβ peptides in the brain parenchyma. This finding suggests that Ginkgo may affect AD and possible type III-c diabetes through the RAGE receptor and may affect the brain, through decreasing mechanisms of glyco-oxidative stress, glycation, or glycotoxins [111]. However, what is not well characterized, and warrants further study, is the effect of prosaic foods and so-called superfoods, and the natural products they represent, such as the effects Ginkgo biloba has when co-metabolized within our dynamic gut and its colonizing flora [17].
It is important to note that other prosaic foods and supplements have similar results in vitro. Histopathological alterations that induce deficits in the respiratory chain complex function beget mitochondrial dysfunction, which is intrinsically tied to oxidative stress. Using the selective mitochondrial antioxidants acetyl-L-carnitine and R-lipoic acid, the author published support for improved cerebral blood flow and a trend in improved cognitive function on human ApoE4 transgenic mice and rats in an age-dependent manner [112], and we suggest Ginkgo may have similar neuroprotective effects as a phytoceutical and mitochondrial protectant. Although the data are quite heterogeneous, Ginkgo biloba extract seems to be of therapeutic benefit in the treatment of mild to moderate dementia of different etiology including mitochondrial dysfunction. These events are found in aging, age-related disease, and in the pathogenesis of aging-related neurodegeneration, and mitochondrial protection and the subsequent reduction of oxidative stress are important components of the neuroprotective activity of GLE extract [39,113].
Moreover, when monoclonal antibodies were made against recombinant α-synuclein protein or α-synuclein epitope 118–123 and then applied to 180 prosaic food product antigens, the antibody–antigen reaction and inhibition or cross-reactivity was studied [114]. This group found that food proteins containing peptides most homologous with α-synuclein showed the highest cross-reactivity, which suggests an autoimmune aspect to Parkinson’s disease and likely other brain diseases that involve proteinopathies. Further, molecular similarity with brain antigens involved in synucleinopathy was demonstrated between food peptides with cross-reactive epitopes with human α-synuclein. The converse could also be true for the conformational diseases, where cross-reactivity to components of superfoods could bind and inhibit aberrant forms of proteins and the structural cross-β structure such as amyloid [115] or paired helical filaments [116]. The author suggests this mechanism could be protective for neurodegenerative diseases such as AD and prion disease [117] and suggests that compounds such as curcumin could bind cross-beta or beta-pleated sheet or barrel structures found in many aberrant proteins, which can occur during disease pathogenesis. The natural product or so-called superfood, curcumin, which is a member of the diarylheptanoid compounds, undergoes microbial transformation. Recently, the bioactivity of the isolated metabolites were studied, as they are potentially therapeutic for several diseases, including neurodegenerative diseases [118].
Interestingly, Ginkgo biloba shares biologically active secondary metabolites with other prosaic foods. Many compounds isolated from Ginkgo biloba were evaluated for their antioxidant activity and compared to ascorbic acid [119]. Ginkgolides and bilobalide (31.3 μg/mL), which are unique constituents of Ginkgo biloba, were not very potent inhibitors of the respiratory burst with a range of inhibition between 28% and 37.3%. However, strong antioxidant activity was observed with flavonoids, but not bioflavonoids, and these researchers found that the hydroxyl group in the C ring (3-OH) conferred more antioxidant activity for compounds including kaempferol, quercetin, myricetin, and tamarixetin than if the functional group was absent, e.g., bioflavonoids, apigenin, and 4′-O-methylapigenin. This finding contradicts others, who suggested that antioxidant potency increases with the increasing number of hydroxyl groups [119].
Finally, the coadministration of GLE with other drugs or treatment modalities would be helpful. For instance, in the modulation of this type of diabetes from the standpoint of oxidative stress, improved vascular blood flow and metabolism could be examined with the insulin sensitizer metformin, which has been attributed anti-aging and to many positive health effects. In diabetic mice, metformin was able to correct memory impairment and the abnormal transport of Aβ across the BBB [120]. Further, GLE (EGB 761) and hyperbaric oxygen therapy, administered together, were shown to ameliorate cognitive and memory impairment in AD via the nuclear transcription factor kappa-B pathway [121]. Another point of the manuscript was to consider the role the microbiota has on oxidative stress and the metabolic products from co-metabolism with the host’s systems, whether diabetic or not.
6. Altered Microbiota in the Processing of Prosaic Foods and Antibiotics
With increased curiosity and movement to discover novel, innovative, and safe alternative treatments for our overall health, including mental and behavioral health, abnormal gut microbiota and/or loss of the intestinal blood barrier are now being linked to abnormalities in memory, depression, mood, anxiety [78,104], and even Alzheimer’s disease [104,122]. In that regard, natural products and so-called superfoods are key targets in future exploration, together with the MGB, for AD and VaD. The importance of this overreaching consideration is to focus on the role of the microbiota in assessing the etiology of a wide range of neurodegenerative and neuropsychiatric diseases and disorders. This support is strengthened by the fact that prosaic foods are co-metabolized with our microbiota together with our own metabolic systems. Moreover, trace amines, 5-hydroxyindoleacetic acid, homovanillic acid, serotonin, and oxitriptan are implicated in this process as they are found in the CNS of said affected patients, which incidentally can also be produced by bacteria [104]. The same holds for phytoceuticals that have suspect affect or putative effects on AD and VaD.
Moreover, the disruption of the blood–brain barrier allows for microbial access to occur either by transcytosis, crossing microvascular endothelial cells, pericytes, or by parasitosis between cells or by hiding inside a host cell [123], which can increase inflammation. Conversely, of particular interest are microbes that access the peripheral blood or affect the BBB, which can allow amyloid beta proteins and cytokines to pass into the periphery or enter the brain to cause damage to neurons through this disruption [105,124]. Lending support to this link, the study of other neurodegenerative disorders, such as Parkinson’s disease and even autism, has identified microbial-driven components in their pathogenesis within the gut–brain–endocrine interplay [104,125]. For example, in autism, it was found that some compounds do cross the BBB and modulate or stimulate brain areas [101].
Communication between the gut microbiota and the brain is now gaining popularity in its significant role in a broad spectrum of diseases [104]. For example, after microbial colonization with short-chain fatty acid-producing bacteria or after short-chain fatty acid administration or supplementation after C. difficile infection in a germ-free animal experiment with rodents, it was found that the presence of tight junctions in the digestive tract of rodents can be improved, thereby implicating the role in strengthening the gastrointestinal barrier [126]. These findings add to a growing body of evidence that show that disruptions in the BBB are linked to a disruption of the gastrointestinal barrier and that probiotics could potentially prevent this disruption by improving BBB integrity and potentially restoring the colonization of microbes that contribute to the barrier [127]. Therefore, one cannot ignore the role for microbial metabolism and biotransformation in modulating Ginkgo biloba and any superfood or co-metabolized compounds with the host. Moreover, exploring which microbes are the “right microbes”, or the “eumicrobial milieu”, could improve outcomes for the administration of any natural product or their derivatives.
7. Bioavailability, Bioactivity of GLE Components, and Antibiotic Use
Ginkgo biloba leaf contains many components, but most of the bioactivity is attributed to the terpene lactones and flavonols. Many constituents are largely unabsorbed in parent forms, such as the terpene lactones [128,129,130], flavonols, and glycosides, some of which must be broken down into readily absorbable compounds. This is where the gut microbiota may play a significant role, providing enzymes such as glycosidase and metabolism to break down the glycosides and release the absorbable forms and aglycones [131,132,133].
Our diet, lifestyle, and medications, particularly antibiotics, influence and shape the gut microbiome throughout our life. In that regard, Ginkgo biloba is known for its resistance to serious plant diseases and pathogens [134]. The findings show that small amounts of crude GLE extracts (7.8 ug/mL) possess inhibitory activity against Gram-positive and Gram-negative bacteria [134]. One study investigated the antibacterial activity and potency of different solvent fractions from a Himalayan gymnosperm Ginkgo biloba longa, collected at high altitude in Kumaun Himalaya, India. The leaf extracts, solvated in methanol, ethanol, chloroform, and hexane, were assessed against the animal and plant bacterial strains Agrobacterium tumefaciens, Bacillus subtilis, Escherichia coli, Erwinia chrysanthemi, and Xanthomonas phaseoli. The zone of inhibition, minimum inhibitory concentration, and minimum bactericidal concentration values were utilized to quantitatively assess the antibacterial activity and potency of the fractions. The methanol extract showed the highest activity as in zones of inhibition in millimeters, which was 15–21 mm, followed by ethanol and hexanes (14–19 mm) and chloroform (15–20 mm), which indicated that the methanol extract inhibited the growth of all of the bacterial strains. It was also found that all of the GLE extracts were more active than the used standard antibiotics, erythromycin, ampicillin, and gentamycin. Taken together, these findings reinforce the power Ginkgo biloba holds in influencing the human microbiota and altering the mycobiome.
One study aimed at further investigating how fluctuations in the gut microbiota composition were undertaken with ciprofloxacin-treated mice to investigate the pharmacokinetics of bioactive extracts and specific terpene lactones including bilobalide, ginkgolide A, ginkgolide B, and ginkgolide C as well as specific Ginkgo flavonols, isorhamnetin, kaempferol, and quercetin [135]. The study found that the operational taxonomic unit value and the phylogenetic diversity was lower in the ciprofloxacin-treated mice than in the control mice. Further, the number of total cultured colonies was suppressed by ciprofloxacin treatment when mouse stools were cultured in blood, liver, and deoxycholate hydrogen sulfide lactose agar plates. Ciprofloxacin administration led to alterations in the pharmacological potency of leaf extracts by diminishing the taxonomic richness after only 5 days of exposure and impacted the levels of about 33% of the bacteria taxa in the gut [136]. In the antibacterial-treated mice, the maximum plasma concentration of isorhamnetin was significantly (p < 0.05) increased 1.7-fold in the ciprofloxacin-treated mice [135], which indicated that the uptake of isorhamnetin was increased by antibacterial treatment and that the antibacterial consumption may increase the bioavailability of isorhamnetin by suppressing gut microbial metabolic activity. The results indicate that ciprofloxacin administration decreased the number and diversity of the gut microbiota. This indicated that the uptake of isorhamnetin was increased by antibacterial treatment and that the antibacterial consumption may increase the bioavailability of isorhamnetin by suppressing gut microbial metabolic activities. There are other studies into the fluctuations in diversity and enzyme activity of the gut microbiota in response to external factors such antibiotics and probiotics. Another study found that the treatment of rats with antibiotics caused alterations in the metabolic activities of the gut microbiota, which led to alterations of the pharmacokinetics of hesperidin [137].
Many other studies have looked deeper into the fluctuations in diversity and enzyme activity the gut microbiota undergoes in response to external factors, such antibiotics and probiotics. Another study looked at ampicillin-treated mice to find that alterations of the gut microbiota composition modulated the pharmacokinetics of aspirin in rats [131,138,139] and to their biotransformation into novel or new compounds with co-metabolism. Taken together, the active components of Ginkgo biloba leaf extracts, prosaic foods, or natural products undergo biotransformation by the gut microbiota to allow for its absorption through the digestive tract. This study demonstrates that the gut microbes play a role in the biotransformation of orally administered phytochemicals and contributes to the activation or detoxification of phytochemicals that are orally ingested [131]. This finding lends support to the fact that the active components of Ginkgo biloba leaf extracts can endure microbial biotransformation in the gut, which allows for metabolite absorption through the digestive tract. It also shows that with antibacterial treatment, changes occur to the GLE extracts, leading to alterations in the pharmacological potency of GLE extracts and to the formation of important secondary metabolites.
8. Comparison of Ginkgo biloba to Common Alzheimer Medications
Going head-to-head with the palliative medications currently used in the treatment of dementia, which include memantine, donepezil, galantamine, and rivastigmine [140], is a standard approach. However, there are few drugs that can be considered the gold standard when comparing the effectiveness of natural products and all drugs have drawbacks and exhibit adverse effects; added to that is their high cost. These are factors partly responsible for the popularity of Ginkgo biloba in comparison to currently recommended dementia medications. Considerable overlap occurs between VaD and AD as both show decreases in neurotransmitters in several studies with AD and VaD patients. The significance of neurotransmitter imbalance to the pathobiology of vascular dementia and AD is through cholinergic deficits, which occur in the brain and CSF in both diseases [53]. Injury to cholinergic fibers leads to the decrease in several neurotransmitters [53]. Improving cognitive decline and the symptoms of AD and VaD with cholinergic therapies, including acetylcholinesterase inhibitors, have shown promising effects in patients [141,142]. Acetylcholinesterase inhibitors are currently being explored and have shown promising effects on cognitive improvement in AD and VaD patients [53,143]. Cholinergic agents have been proposed for improving cognitive decline and the symptoms of AD and VaD, including AChE inhibitors as well as natural products such as active compounds from the herb huperzine [53].
If dopamine has a role in neurodegeneration and can covalently modify proteins such as parkin [144], for example, and Ginkgo biloba can act at the level of neurotransmission, we could venture to conclude that one mechanism of Ginkgo’s action is by increasing dopamine levels. Animal studies with rats led to the discovery that with chronic EGb761 administration, dopamine levels in the prefrontal cortex, a region involved in executive function, memory, intelligence, language, and visual search and gaze control [52,145], were increased by the flavonol glycosides and ginkgolide fractions improved cognitive function [81]. Another study with Ginkgo biloba extracts explored improving cognitive function and control by modulating the levels of acetylcholine, dopamine, and 5-hydroxytryptamine (serotonin). These researchers found that Ginkgo biloba extracts increased select neurotransmitters, while concomitantly inhibited the activation of acetylcholinesterase, an enzyme responsible for degrading the neurotransmitter acetylcholine [52,142]. The finding supports the case that Ginkgo biloba activity and current acetylcholine drugs, used for dementia, both work to increase important neurotransmitters. The accumulating evidence suggests that decreasing cholinergic deficiency can contribute to delayed brain damage in VaD and AD [54,55] and that there is a potential role for Ginkgo biloba in this mechanism. The use of ginkgolide and related compounds complements findings centered around the French paradox, as both wine compounds and ginkgolide share mechanistic features in common with age-related brain diseases. Importantly, both Ginkgo biloba and red wine constituents have acetyl cholinesterase-inhibiting activity [142]. What is not known is whether there is a role for bacterial co-metabolism and the microbiota–gut–brain axis that may contribute to the beneficial findings [142].
There are several other trials worth mentioning that advocate the success of Ginkgo biloba in randomized placebo-controlled double-blind studies (see Table 1), which compare Ginkgo biloba (160 mg daily dose) vs. donepezil (5 mg daily dose) vs. placebo for 6 months. The results showed that the effect of Ginkgo biloba were comparable to donepezil in its improvement of the patients’ attention, memory, and cognitive performance after 6 months of treatment [146]. In addition, another randomized double-blind exploratory trial also compared Ginkgo biloba (240 mg/day) vs. donepezil (initially 5 mg after 4 weeks 10 mg/day) vs. combined (same doses) for 22 weeks, and the results revealed that there was no difference between taking Ginkgo biloba alone or donepezil alone and, in fact, combined therapy was superior to monotherapy with donepezil because of the decreased side effects and improvement in the patients’ cognitive performance and neuropsychiatric symptoms [58]. Among other clinical trial studies looking at both dementia to due Alzheimer’s or vascular etiology, the multi-center, double-blind, randomized placebo-controlled trials found that treatment with 240 mg of EGb761 daily resulted in significant and clinically relevant improvement in cognition and quality of life of patients and caregivers when compared to placebo [147]. A meta-analysis of eight randomized, double-blind, placebo-controlled clinical trials assessed the efficacy Ginkgo biloba in the symptomatic treatment of cerebral insufficiency [39,65]. Treatment with an average dose of 150 mg of EGb761 or placebo for 6 to 12 weeks showed superior results with EGb761 in terms of treating memory deficits, disturbances in concentration, depressive emotional conditions, dizziness, tinnitus, and headache [65]. To further provide credibility to Ginkgo biloba and its pharmacological effects, in a study of elderly patients with mild to moderate dementia, the oral administration of EGb761 produced similar EEG profiles of anti-dementia drugs [65]. Moreover, healthy male subjects taking Ginkgo biloba exhibited increased alpha activity and cognitive activating-type response, which was more apparent when compared to placebo [65] and demonstrated the credibility and effectiveness of Ginkgo biloba when compared to currently used modern drugs, such as donepezil.
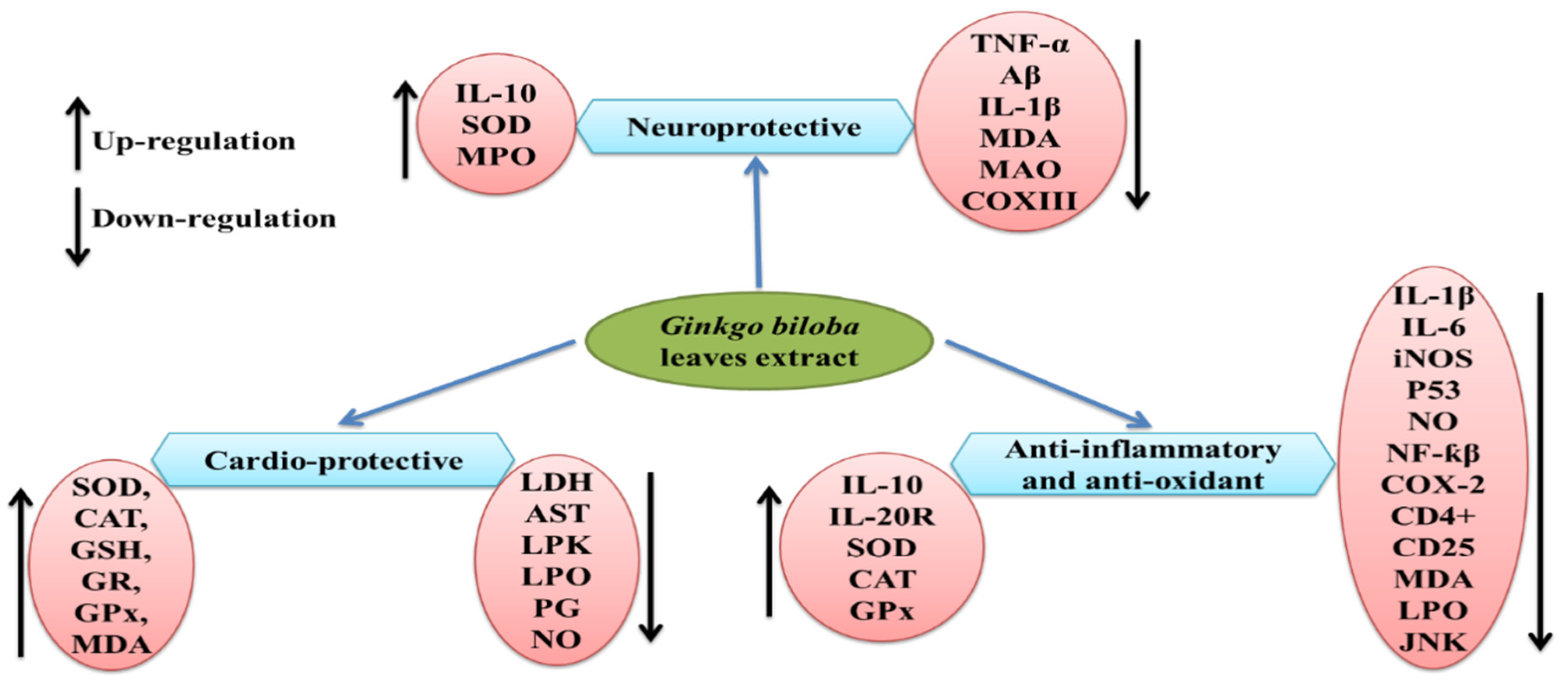
G. biloba extract is one of the most important and popular dietary supplements that can prevent neurological damage and help in memory enhancement [148]. Previous studies reported the neuroprotective properties of the leaf extract EGb 761 in Alzheimer’s disease at a dose of 240 mg/kg/day [148,149], which may be due to its antioxidant properties and ability to inhibit Aβ-induced neurotoxicity and apoptosis [150,151,152]. Similarly, isolated polysaccharides from G. biloba leaf extract were found to be effective against ischemia/reperfusion injury in rat brains when given 7 days before the procedure [153], which resulted in progress in neurological deficits and improved motor function by a reduction in proinflammatory cytokines (TNF-α and IL-1β) and MDA content, while the elevation in the activity of superoxide dismutase, myeloperoxidase, and anti-inflammatory cytokine IL-10 have also been observed (see Figure 2). A Ginkgo biloba leaf extract was tested against 6-hydroxydopamine-induced neurotoxicity in rats and was found effective in reducing the behavioral deficit [154]. The underlying mechanism for the neuroprotective effect was highlighted by CA1 neuron protection from death and the downregulation of COX III mRNA encoded by mitochondrial DNA. Ginkgo biloba extract was proven to have neuroprotective properties, as well as neurorestorative properties, when studied in mice [155].
Figure 2.
Regulation of various molecular targets and therapeutic effects of Ginkgo biloba. Aβ = amyloid beta; AST = aminotransferase; BCl-2 = B-cell lymphoma 2; CAT = catalase; CD4+ = cluster of differentiation 4; COX III = cyclooxygenase 3; GPx = glutathione peroxidase; GR = glutathione reductase; GSH = glutathione; IL-1β = interleukin 1 beta; IL-10 = interleukin 10; IL-20R = interleukin 20R; iNOS = nitric oxide synthase; JNK = c-Jun N-terminal kinase; LDH = lactate dehydrogenase; LPK = L-pyruvate kinase; LPO = lipid hydroperoxide; MAO = monoamino oxidase; MDA = malondialdehyde; MPO = myeloperoxidase; NO = nitric oxide; p53 = tumor protein p53; SOD = superoxide dismutase; TNF-α = tumor necrosis factor α.

Table 1.
List of selected clinical trials for the use of natural products of G. biloba in the treatment of vascular Alzheimer’s disease, vascular dementia, and AD.
Table 1.
List of selected clinical trials for the use of natural products of G. biloba in the treatment of vascular Alzheimer’s disease, vascular dementia, and AD.
| Clinical Trial | Objective | Conclusions |
|---|---|---|
| [156] | To assess the efficacy and safety of EGb in Alzheimer’s disease and multi-infarct dementia. | EGb was safe and appears capable of stabilizing and, in a substantial number of cases, improving the cognitive performance and the social functioning of dementia patients for 6 months to 1 year. Although modest, the changes induced by EGb were objectively measured by the ADAS-Cog and were of sufficient magnitude to be recognized by the caregivers in the GERRI. |
| [157] | This randomized, double-blind, exploratory trial was undertaken to compare treatment effects and tolerability of EGb 761(R), donepezil, and combined treatment in patients with AD and neuropsychiatric features. | These exploratory findings helped to develop three hypotheses that will have to be proven in further studies: (1) there is no significant difference in the efficiency between EGb 761(R) and donepezil, (2) a combination therapy will be superior to a mono-therapy with one of both substances, and (3) there will be fewer side effects under a combination therapy than under mono-therapy with donepezil. |
| [158] | The purpose of this research was to conduct the first known large-scale clinical trial of the efficacy of G. biloba extract (EGb 761) on the neuropsychological functioning of cognitively intact older adults. | Overall, the results from both objectives, standardized, neuropsychological tests and a subjective, follow-up self-report questionnaire provided complementary evidence of the potential efficacy of G. biloba EGb 761 in enhancing certain neuropsychological/memory processes of cognitively intact older adults, 60 years of age and over. |
| [159] | To determine effectiveness of G. biloba vs. placebo in reducing the incidence of all-cause dementia and Alzheimer’s disease in elderly individuals with normal cognition and those with mild cognitive impairment (MCI). | In this study, G. biloba at 120 mg twice a day was not effective in reducing either the overall incidence rate of dementia or AD incidence in elderly individuals with normal cognition or those with MCI. Trial Registration clinicaltrials.gov Identifier: NCT00010803. |
| [160] | The aim was to determine the effectiveness and the safety profile of G. biloba for treating early-stage dementia in a community setting. | We found no evidence that a standard dose of high purity G. biloba confers benefit in mild to moderate dementia over 6 months. |
| [161] | G. biloba may have a role in treating impairments in memory, cognitive speed, activities of daily living (ADL), edema, inflammation, and free-radical toxicity associated with traumatic brain injury (TBI), Alzheimer’s dementia, stroke, vaso-occlusive disorders, and aging. The purpose of this review was to provide a synthesis of the mechanisms of action, clinical indications, and safety of G. biloba extract. | Ginkgo shows promise in treating some of the neurologic sequelae associated with Alzheimer’s disease, TBI, stroke, normal aging, edema, tinnitus, and macular degeneration. Mechanisms of action may include antioxidant, neurotransmitter/receptor modulatory, and antiplatelet-activating factor properties. While safe, caution is advised when recommending Ginkgo to patients taking anticoagulants. Future studies should examine dose effects, component activity, mechanisms, and clinical applications. |
| [162] | To assess the cumulative evidence on the efficacy and effectiveness of G. biloba extract (GbE) in the treatment of dementia. | GbE has potentially beneficial effects for people with dementia when it is administered at doses greater than 200 mg/day for at least 5 months. Given the lower quality of the evidence, further rigorously designed, multicenter-based, large-scale RCTs are warranted. |
| [163] | Prospective cohort studies showed inverse associations between the intake of flavonoid-rich foods (cocoa and tea) and cardiovascular disease (CVD). Intervention studies showed protective effects on intermediate markers of CVD. This may be due to the protective effects of the flavonoids epicatechin (in cocoa and tea) and quercetin (in tea) were investigated for the effects of supplementation of pure epicatechin and quercetin on vascular function and cardiometabolic health. | Our results suggest that epicatechin may partially contribute to the cardioprotective effects of cocoa and tea by improving insulin resistance. It is unlikely that quercetin plays an important role in the cardioprotective effects of tea. This study was registered at clinicaltrials.gov as NCT01691404. |
| [164] | This group studied the effects of supplementation of pure epicatechin and quercetin on biomarkers of endothelial dysfunction and inflammation. | In (pre)hypertensive men and women, epicatechin may contribute to the cardioprotective effects of cocoa and tea through improvements in endothelial function. Quercetin may contribute to the cardioprotective effects of tea possibly by improving endothelial function and reducing inflammation. This trial was registered at clinicaltrials.gov as NCT01691404. |
9. Microbial Metabolism vs. Microbial Biotransformation and Engineering
Bacterial metabolism and biotransformation are distinct systems of molecular processing by microorganisms of sources such as plant material. Natural products and compounds with potential as phytoceuticals, or as drug candidates, mostly occur in nature at very low concentrations. Emerging alternatives to plant purification is to express biosynthetic genes in microbial systems such as bacteria and fungi [165]. More than 60% of antibiotics used in medical and other health practices are microorganism-derived natural products [166]. Primary metabolism centers around reactions associated with generating energy, biomass, and essential cellular components. Secondary metabolism, or xenobiotic metabolism, usually involves a non-survival role and adaptation to environmental conditions or offers some selective advantage. These compounds often have unusual structures and are normally synthesized later in the cell growth cycle and may have importance medicinally [167]. It is unquestioned that co-metabolism between the host and its resident microbiota produce naturally occurring secondary metabolites and any bioactivity owed to plant products must be considered against the backdrop of bacterial biotransformation. In order for proper evaluation and discovery, microbial biotransformation has emerged as an important player and a tool for obtaining novel structural drug analogs or to improve the pharmacokinetic parameters of other substances [168]. We have harnessed bacteria as programmable biochemical factories for diverse applications and have suggested the genetic engineering of bacteria for the production of valuable bioactive compounds through genome engineering CRISP-R and recombinant DNA technology [4]. These comprehensive engineering approaches are directed at improving the microbial production of natural products, proteins, and novel small molecules. Many of these bioactive compounds could be produced through engineering approaches to improve or augment the effectiveness or mechanism of action of Ginkgo biloba, as may be the case for the pathway engineering of primary bacterial metabolism [169].
Phenolic acids, polyphenols, and indigestible carbohydrates such as pectin, hemicellulose, and various polysaccharides are contained in many fruits and fruit juices. These dietary constituents support select microbial species and their colonization through co-metabolism. It is the co-metabolism through which the gut microbiota imparts many beneficial effects on health through health-promoting foods. The same premise holds for whole foods and Ginkgo or other so-called superfood constituents. For example, microbial fermentation by-products are SCFAs—in particular, acetate, propionate, and butyrate [170]. These prebiotics, in fact, act synergistically to modify colonic and intestinal microbiota, leading to a symbiosis with the human host that benefits health [171]. When fermented kiwifruit was added, SCFAs increased, while lactate and after 24 h succinate concentrations declined and most of the genera observed produced acetic acid [171]. Propionic acid is a SCFA product of Bacteroidetes fermentation and from Veillonella, members of the phyla Firmicutes. Butyrate, produced mainly by the Firmicutes subset, among others, features members of the Lachnospiraceae family [171].
Protective aspects of butyrate have been found, for example, from prebiotic potato starch with Faecalibacterium prausnitzii on Clostridioides difficile infection and damage [126]. Other studies found prebiotic effects, using 16S rRNA pyrosequencing of the microbial DNA prepared from stool co-incubated with select fruits when fermented with feces from 10 donors [171]. Additionally, in the aforementioned study by Juliet and colleagues, by-products of fermentation contributed to the first step in microbial colonization by modulating both microbial numbers and gut flora ecology through the adhesion of different bacteria to the gut wall [172]. Co-metabolism is a two-way street, and the same group found an immediate and long-lasting reduction in the abundance of all members of the Proteobacteria phyla as well as some members of the Firmicutes and Bacteroidetes phyla, which were originally present in the original fecal inoculum. Further, other abundances decreased by half, namely, Ruminococcaceae. In contrast, Bacteroidaceae, Lachnospiraceae, and Veillonella members of the Firmicutes phyla and Coriobacteriaceae of the Actinobacteria phyla increased over 24 h. The Lachnospiraceae levels were sustained for up to 48 h and Coriobacteriaceae and Bifidobacteraceae Actinobacteria increased [171]. The same holds for the constituent metabolic products derived from Ginkgo or other so-called superfoods and their whole food source.
10. Conclusions
New concepts and divergent views, differing from the dogmatic and classic understanding, represent a paradigm shift and potential for discovering novel treatments of diseases. Building on our previous work on the metabolic interaction between any host and any given organism at the small molecule level [99] and the involvement of the MGB axis when coupled with host co-metabolism, we have elucidated the common underpinnings of disease pathogenesis in AD and VaD and how natural compounds and the MGB hold promise to incentivize research and alternative treatments for this potentially treatable form of dementia. Moreover, a growing body of evidence suggests a common etiology for Alzheimer’s disease, cardiovascular disease, and VaD [17,142,173,174,175].
The use of Ginkgo biloba is not new in medicine as it has been used in China for centuries. For Ginkgo biloba to be fully accepted and integrated into clinical practice, its effectiveness must first be evidence-based. Further, we must also consider the second genome and vast metabolism of the microbiota–gut–brain axis in the metabolism of this compound to fully assess its effectiveness. Nevertheless, there remains a place for Ginkgo biloba in medicinal chemistry, especially when one considers bacterial co-metabolism. It only seems appropriate to offer GLE as an adjunct treatment in Western medicine as some clinical trials are demonstrating efficacy of Ginkgo biloba in the treatment of dementia symptoms. Unfortunately, biases remain that can hinder phytoceutical development and acceptance. Many pharmaceutical companies are funding clinical trials, which may compel researchers to anticipate obtaining positive data in support of funding for Ginkgo biloba. Patients educated about Ginkgo biloba can often demonstrate anecdotal faith in the drug, compelling them to believe in its success. This acceptance can hinder the evaluation of any improvement in function. Moreover, many pharmaceutical companies are funding these trials, which compels researchers to expect positive data in support of Ginkgo biloba. Whole Ginkgo biloba longa and its extracts as medicine and phytoceutical are gaining support and consideration with the power of clinically statistically significant results, low cost, low adverse reactions, and its prominent use in Eastern medicine for decades. Hopefully, the evidence and mechanistic understanding will garner GLE use in Western medicine. Moreover, we call for the concomitant exploration of co-metabolism for this drug with our gut microbiota as it relates to the microbiota–gut–brain axis [17]. Moving forward, there remains a place for Ginkgo biloba in medicinal chemistry and an opportunity that needs development for Ginkgo biloba to be accepted and integrated into clinical practice.
Even though there are a substantial number of clinical trials available, the efficacy of these Ginkgo-derived compounds or extracts in many clinical trials is limited in scope. For many trials, the statistical power has been low. It is not easy to gather patients for studies, as many of the participants are elderly and rely heavily on family support. Finding a family unit that has time to contribute to the study is an obstacle. However, regardless of the obstacles, further research for alternative treatments for dementia is essential. Lack of research in this area, either because of the large time commitment needed and the cost of conducting trials [9], hampers substantial research data. Therefore, the amount of data accumulated thus far is not sufficient to provide statistically sound evidence, even though clinical trials are proving the efficacy of Ginkgo biloba in treatment of dementia symptoms. So, interest and further research and trials in this direction are warranted to help develop more information and evidence regarding its potential use in the treatment of dementia.
What can be concluded from the clinical trials discussed in this review is that the pathophysiology of vascular-associated dementia is among the many etiologies of AD that provide numerous targets for treatment. With the pharmacodynamic range of EGb761 discussed, EGb761 is still a viable treatment option for both Alzheimer’s disease and vascular dementia [72]. Ginkgo biloba extract is an important and popular dietary supplement, which has a role in the prevention of neurological damage and improved memory enhancement [176] of patients suffering from AD or VaD. The possibility of improving cognition, stabilizing and decreasing the decline of mental function in patients with dementia using the Ginkgolides and the flavonoid glycosides exists. We suggest that GLE can help affected individuals and inconclusive studies should be revisited, taking the MGB axis and microbial metabolism into account when studying natural products. However, little exploration has been undertaken against the backdrop of gut microbiota, also called the second brain and the second genome, within us. How the microbiota modulates the effects of natural products and the downstream chemical profile of bacteria-derived constituents is an underappreciated and potentially transforming aspect of these brain-modulating nutraceuticals or phytoceuticals. Revisiting the clinical trials and considering the nutritional aspects and microbial constitution of the elderly should shed light on the varied results found with natural products and prosaic foods for AD and VaD.
Funding
This research received no external funding.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
Abbreviations
Alzheimer’s disease (AD), vascular dementia (VaD), microbiota–gut–brain (MGB) axis, Ginkgo biloba (G. biloba), Ginkgo biloba leaf extracts (GLE), Ginkgo biloba leaf extract (EGb761), blood–brain barrier (BBB), receptor for advanced glycation end-products (RAGE), mitogen-activated protein kinase (MAPK), acetylcholinesterase (AChE), interleukins (IL), cerebrospinal fluid (CSF), complementary alternative medicine (CAM), 1-methyl-4-phenyl-1,2,3,6-tetrahydropyridine (MPTP), monoamine oxidase (MAO), superoxide dismutase (SOD), myeloperoxidase (MPO), hyperhomocysteinemia (HHcy), tau kinase (GSK-β), protein phosphatase 2A (PP2A), G-protein receptor kinase 2 (GRK-2), short-chain fatty organic acids (SCFAs).
References
- Stelzmann, R.A.; Norman Schnitzlein, H.; Reed Murtagh, F. An English translation of Alzheimer’s 1907 paper, “Uber eine eigenartige Erkankung der Hirnrinde”. Clin. Anat. 1995, 8, 429–431. [Google Scholar] [CrossRef]
- Kalaria, R.N.; Maestre, G.E.; Arizaga, R.; Friedland, R.P.; Galasko, D.; Hall, K.; Luchsinger, J.A.; Ogunniyi, A.; Perry, E.K.; Potocnik, F.; et al. Alzheimer’s disease and vascular dementia in developing countries: Prevalence, management, and risk factors. Lancet Neurol. 2008, 7, 812–826. [Google Scholar] [CrossRef]
- Evans, D.A.; Funkenstein, H.H.; Albert, M.S.; Scherr, P.A.; Cook, N.R.; Chown, M.J.; Hebert, L.E.; Hennekens, C.H.; Taylor, J.O. Prevalence of Alzheimer’s Disease in a Community Population of Older Persons: Higher Than Previously Reported. JAMA J. Am. Med. Assoc. 1989, 262, 2551–2556. [Google Scholar] [CrossRef]
- Obrenovich, M.; Jaworski, H.; Tadimalla, T.; Mistry, A.; Sykes, L.; Perry, G.; Bonomo, R.A. The role of the microbiota–gut–brain axis and antibiotics in ALS and neurodegenerative diseases. Microorganisms 2020, 8, 784. [Google Scholar] [CrossRef] [PubMed]
- Wimo, A.; Prince, M. Alzheimer’s Disease International World Alzheimer Report 2010. Available online: https://scholar.google.com/scholar?hl=en&as_sdt=0%2C5&authuser=1&q=Wimo%2C+A.%2C+Prince%2C+M.+%282010%29+Alzheimer’s+Disease+International+World+Alzheimer+Report+2010.+The+Global+Economic+Impact+of+Dementia%2C+4&btnG= (accessed on 10 May 2021).
- Wimo, A.; Guerchet, M.; Ali, G.; Wu, Y.; Prina, A.W.; Winblad, B.; Jönsson, L.; Liu, Z.; Prince, M. The Worldwide Costs of Dementia 2015 and Comparisons with 2010; Elsevier: Amsterdam, The Netherland, 2017; Available online: https://www.sciencedirect.com/science/article/pii/S1552526016300437 (accessed on 13 July 2022).
- von Gunten, A.; Schlaefke, S.; Überla, K. Efficacy of Ginkgo biloba extract EGb 761® in dementia with behavioural and psychological symptoms: A systematic review. World J. Biol. Psychiatry 2016, 17, 622–633. [Google Scholar] [CrossRef]
- Dong, H.; Csernansky, C.A.; Martin, M.V.; Bertchume, A.; Vallera, D.; Csernansky, J.G. Acetylcholinesterase inhibitors ameliorate behavioral deficits in the Tg2576 mouse model of Alzheimer’s disease. Psychopharmacology 2005, 181, 145–152. [Google Scholar] [CrossRef]
- DeKosky, S.T.; Fitzpatrick, A.; Ives, D.G.; Saxton, J.; Williamson, J.; Lopez, O.L.; Burke, G.; Fried, L.; Kuller, L.H.; Robbins, J.; et al. The Ginkgo Evaluation of Memory (GEM) study: Design and baseline data of a randomized trial of Ginkgo biloba extract in prevention of dementia. Contemp. Clin. Trials 2006, 27, 238–253. [Google Scholar] [CrossRef] [PubMed]
- Snitz, B.E.; O’Meara, E.S.; Carlson, M.C.; Arnold, A.M.; Ives, D.G.; Rapp, S.R.; Saxton, J.; Lopez, O.L.; Dunn, L.O.; Sink, K.M.; et al. Ginkgo biloba for preventing cognitive decline in older adults a randomized trial. JAMA—J. Am. Med. Assoc. 2009, 302, 2663–2670. [Google Scholar] [CrossRef]
- Harman, D. Aging: A theory based on free radical and radiation chemistry. Sci. Aging Knowl. Environ. 2002, 37, cp14. Available online: https://scholar.google.com/scholar?hl=en&as_sdt=0%2C5&q=Harman+D.+Aging%3A+A+theory+based+on+free+radical+and+ra-+diation+chemistry.+J+Gerontol+1956%3B11%3A298–300&btnG= (accessed on 13 July 2022). [CrossRef]
- Aliev, G.; Gasimov, E.; Obrenovich, M.E.; Fischbach, K.; Shenk, J.C.; Smith, M.A.; Perry, G. Atherosclerotic lesions and mitochondria DNA deletions in brain microvessels: Implication in the pathogenesis of Alzheimer’s disease. Vasc. Health Risk Manag. 2008, 4, 721–730. [Google Scholar] [CrossRef]
- Globus, M.Y.; Alonso, O.; Dietrich, W.D.; Busto, R.; Ginsberg, M.D. Glutamate Release and Free Radical Production Following Brain Injury: Effects of Posttraumatic Hypothermia. J. Neurochem. 1995, 65, 1704–1711. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.A.; Perry, G.; Richey, P.L.; Sayre, L.M.; Anderson, V.E.; Beal, M.F.; Kowall, N. Oxidative damage in Alzheimer’s. Nature 1996, 382, 120–121. [Google Scholar] [CrossRef] [PubMed]
- Iadecola, C. The Pathobiology of Vascular Dementia. Neuron 2013, 80, 844–866. [Google Scholar] [CrossRef] [PubMed]
- Frerichs, K.U. Neuroprotective Strategies in Nature—Novel Clues for the Treatment of Stroke and Trauma. In Current Progress in the Understanding of Secondary Brain Damage from Trauma and Ischemia; Springer: Wien, Vienna, 1999; Volume 73, pp. 57–61. [Google Scholar] [CrossRef]
- Obrenovich, M.; Tabrez, S.; Siddiqui, B.; McCloskey, B.; Perry, G. The microbiota–gut–brain axis–heart shunt part II: Prosaic foods and the brain–heart connection in Alzheimer disease. Microorganisms 2020, 8, 493. [Google Scholar] [CrossRef] [PubMed]
- Müller, W.E.; Heiser, J.; Leuner, K. Effects of the standardized Ginkgo biloba extract EGb 761® on neuroplasticity. Int. Psychogeriatr. 2012, 24, S21–S24. [Google Scholar] [CrossRef]
- Brunt, V.E.; Casso, A.G.; Gioscia-Ryan, R.A.; Sapinsley, Z.J.; Ziemba, B.P.; Clayton, Z.S.; Bazzoni, A.E.; Vandongen, N.S.; Richey, J.J.; Hutton, D.A.; et al. Gut Microbiome-Derived Metabolite Trimethylamine N-Oxide Induces Aortic Stiffening and Increases Systolic Blood Pressure with Aging in Mice and Humans. Hypertension 2021, 78, 499–511. [Google Scholar] [CrossRef]
- Louca, P.; Menni, C.; Padmanabhan, S. Genomic Determinants of Hypertension With a Focus on Metabolomics and the Gut Microbiome. Am. J. Hypertens. 2020, 33, 473–481. [Google Scholar] [CrossRef]
- Tang, W.H.W.; Wang, Z.; Levison, B.S.; Koeth, R.A.; Britt, E.B.; Fu, X.; Wu, Y.; Hazen, S.L. Intestinal microbial metabolism of phosphatidylcholine and cardiovascular risk. N. Engl. J. Med. 2013, 368, 1575–1584. [Google Scholar] [CrossRef]
- Tang, W.H.W.; Wang, Z.; Kennedy, D.J.; Wu, Y.; Buffa, J.A.; Agatisa-Boyle, B.; Li, X.S.; Levison, B.S.; Hazen, S.L. Gut microbiota-dependent trimethylamine N-oxide (TMAO) pathway contributes to both development of renal insufficiency and mortality risk in chronic kidney disease. Circ. Res. 2015, 116, 448–455. [Google Scholar] [CrossRef]
- Xie, H.; Wang, J.R.; Yau, L.F.; Liu, Y.; Liu, L.; Han, Q.B.; Zhao, Z.; Jiang, Z.H. Quantitative analysis of the flavonoid glycosides and terpene trilactones in the extract of ginkgo biloba and evaluation of their inhibitory activity towards fibril formation of β-amyloid peptide. Molecules 2014, 19, 4466–4478. [Google Scholar] [CrossRef]
- Blacher, E.; Dadali, T.; Bespalko, A.; Haupenthal, V.J.; Grimm, M.O.W.; Hartmann, T.; Lund, F.E.; Stein, R.; Levy, A. Alzheimer’s disease pathology is attenuated in a CD38-deficient mouse model. Ann. Neurol. 2015, 78, 88–103. [Google Scholar] [CrossRef] [PubMed]
- Wan, W.B.; Cao, L.; Liu, L.M.; Kalionis, B.; Chen, C.; Tai, X.T.; Li, Y.M.; Xia, S. EGb761 provides a protective effect against Aβ1-42 oligomer-induced cell damage and blood-brain barrier disruption in an in vitro bEnd.3 endothelial model. PLoS ONE 2014, 9, e113126. [Google Scholar] [CrossRef] [PubMed]
- Obrenovich, M.E.; Smith, M.A.; Siedlak, S.L.; Chen, S.G.; Torre, J.C.; Perry, G.; Aliev, G. Overexpression of GRK2 in Alzheimer disease and in a chronic hypoperfusion rat model is an early marker of brain mitochondrial lesions. Neurotox. Res. 2006, 10, 43–56. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, J.; Yang, B.; Zheng, Y.; Yao, M.; Sun, M.; Xu, L.; Lin, C.; Chang, D.; Tian, F. Ginkgo biloba Extract Inhibits Astrocytic Lipocalin-2 Expression and Alleviates Neuroinflammatory Injury via the JAK2/STAT3 Pathway After Ischemic Brain Stroke. Front. Pharmacol. 2018, 9, 518. [Google Scholar] [CrossRef] [PubMed]
- De la Torre, J.C. Alzheimer disease as a vascular disorder: Nosological evidence. Stroke 2002, 33, 1152–1162. [Google Scholar] [CrossRef] [PubMed]
- Querfurth, H.W.; LaFerla, F.M. Alzheimer’s disease. N. Engl. J. Med. 2010, 362, 329–344. [Google Scholar] [CrossRef] [PubMed]
- Humpel, C. Chronic mild cerebrovascular dysfunction as a cause for Alzheimer’s disease? Exp. Gerontol. 2011, 46, 225. [Google Scholar] [CrossRef]
- Obrenovich, M.E.; Palacios, H.H.; Gasimov, E.; Leszek, J.; Aliev, G. The GRK2 Overexpression Is a Primary Hallmark of Mitochondrial Lesions during Early Alzheimer Disease. Cardiovasc. Psychiatry Neurol. 2009, 14, 1–14. [Google Scholar] [CrossRef]
- EE, B. Neurovascular unit dysfunction: A vascular component of Alzheimer disease? Neurology 2007, 68, 1730–1732. [Google Scholar] [CrossRef]
- Serrano-pozo, A.; Frosch, M.P.; Masliah, E.; Hyman, B.T.; Holtzman, D.M.; Herz, J.; Bu, G.; Masters, C.L.; Selkoe, D.J.; Goedert, M.; et al. Neuropathological Alterations in Alzheimer Disease. Cold Spring Harb. Perspect. Med. 2011, 1, a006189. [Google Scholar] [CrossRef]
- Itagaki, S.; McGeer, P.L.; Akiyama, H.; Zhu, S.; Selkoe, D. Relationship of microglia and astrocytes to amyloid deposits of Alzheimer disease. J. Neuroimmunol. 1989, 24, 173–182. [Google Scholar] [CrossRef] [PubMed]
- El Khoury, J.; Toft, M.; Hickman, S.E.; Means, T.K.; Terada, K.; Geula, C.; Luster, A.D. Ccr2 deficiency impairs microglial accumulation and accelerates progression of Alzheimer-like disease. Nat. Med. 2007, 13. [Google Scholar] [CrossRef] [PubMed]
- Helmut, K.; Hanisch, U.K.; Noda, M.; Verkhratsky, A. Physiology of microglia. Physiol. Rev. 2011, 91, 432–438. [Google Scholar] [CrossRef]
- Jin, M.; Selkoe, D.J. Systematic analysis of time-dependent neural effects of soluble amyloid β oligomers in culture and in vivo: Prevention by scyllo-inositol. Neurobiol. Dis. 2015, 82, 152–163. [Google Scholar] [CrossRef] [PubMed]
- Colciaghi, F.; Borroni, B.; Zimmermann, M.; Bellone, C.; Longhi, A.; Padovani, A.; Cattabeni, F.; Christen, Y.; Di Luca, M. Amyloid precursor protein metabolism is regulated toward alpha-secretase pathway by Ginkgo biloba extracts. Neurobiol. Dis. 2004, 16, 454–460. [Google Scholar] [CrossRef] [PubMed]
- Demarin, V.; Kes, V.B.; Trkanjec, Z.; Budišić, M.; Pašić, M.B.; Črnac, P.; Budinčević, H. Efficacy and safety of Ginkgo biloba standardized extract in the treatment of vascular cognitive impairment: A randomized, double-blind, placebo-controlled clinical trial. Neuropsychiatr. Dis. Treat. 2017, 13, 483–490. [Google Scholar] [CrossRef]
- Nowak, A.; Kojder, K.; Zielonka-Brzezicka, J.; Wróbel, J.; Bosiacki, M.; Fabiańska, M.; Wróbel, M.; Sołek-Pastuszka, J.; Klimowicz, A. The Use of Ginkgo Biloba, L. as a Neuroprotective Agent in the Alzheimer’s Disease. Front. Pharmacol. 2021, 12, 3136. [Google Scholar] [CrossRef] [PubMed]
- Grundke-Iqbal, I.; Iqbal, K.; Tung, Y.C.; Quinlan, M.; Wisniewski, H.M.; Binder, L.I. Abnormal phosphorylation of the microtubule-associated protein tau (tau) in Alzheimer cytoskeletal pathology. Proc. Natl. Acad. Sci. USA 1986, 83, 4913–4917. [Google Scholar] [CrossRef]
- Brustolin, S.; Giugliani, R.; Félix, T.M. Genetics of homocysteine metabolism and associated disorders. Braz. J. Med. Biol. Res. 2010, 43, 1–7. [Google Scholar] [CrossRef]
- Tawfik, A.; Samra, Y.A.; Elsherbiny, N.M.; Al-Shabrawey, M. Implication of hyperhomocysteinemia in blood retinal barrier (BRB) dysfunction. Biomolecules 2020, 10, 1119. [Google Scholar] [CrossRef]
- Anderson, A.J.; Su, J.H.; Cotman, C.W. DNA damage and apoptosis in Alzheimer’s disease: Colocalization with c- Jun immunoreactivity, relationship to brain area, and effect of postmortem delay. J. Neurosci. 1996, 16, 1710–1719. [Google Scholar] [CrossRef] [PubMed]
- De La Monte, S.M.; Sohn, Y.K.; Wands, J.R. Correlates of p53- and Fas (CD95)-mediated apoptosis in Alzheimer’s disease. J. Neurol. Sci. 1997, 152, 73–83. [Google Scholar] [CrossRef] [PubMed]
- Kihiko, M.E.; Tucker, H.M.; Rydel, R.E.; Estus, S. c-Jun Contributes to Amyloid β-Induced Neuronal Apoptosis but Is Not Necessary for Amyloid β-Induced c-jun Induction. J. Neurochem. 1999, 73, 2609–2612. [Google Scholar] [CrossRef] [PubMed]
- Zeng, K.; Li, M.; Hu, J.; Mahaman, Y.A.R.; Bao, J.; Huang, F.; Xia, Y.; Liu, X.; Wang, Q.; Wang, J.-Z.; et al. Ginkgo biloba Extract EGb761 Attenuates Hyperhomocysteinemia-induced AD Like Tau Hyperphosphorylation and Cognitive Impairment in Rats. Curr. Alzheimer Res. 2017, 15, 89. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Yang, D.; Wyss-Coray, T.; Yan, J.; Gan, L.; Sun, Y.; Mucke, L. Wild-type but not Alzheimer-mutant amyloid precursor protein confers resistance against p53-mediated apoptosis. Proc. Natl. Acad. Sci. USA 1999, 96, 7547–7552. [Google Scholar] [CrossRef] [PubMed]
- Der Yang, S.; Yu, J.S.; Liu, W.K.; Yen, S.H. Synergistic control mechanism for abnormal site phosphorylation of Alzheimer’s diseased brain tau by kinase FA/GSK-3 alpha. Biochem. Biophys. Res. Commun. 1993, 197, 400–406. [Google Scholar] [CrossRef]
- Davies, P.; Maloney, A.J. Selective loss of central cholinergic neurons in Alzheimer’s disease. Lancet 1976, 2, 1403. Available online: http://www.ncbi.nlm.nih.gov/pubmed/63862 (accessed on 11 January 2015). [CrossRef]
- Bartus, R.T.; Dean, R.L.; Beer, B.; Lippa, A.S. The cholinergic hypothesis of geriatric memory dysfunction. Science 1982, 217, 408–414. Available online: http://www.ncbi.nlm.nih.gov/pubmed/7046051 (accessed on 11 January 2015). [CrossRef]
- Wang, J.; Mao, S.; Wang, J.; Jiao, Y.; Zhang, X.; Zhang, Y.; Wang, Z.; Zhang, Q.; Li, S. Effect of Ginkgo biloba extract on cognitive function and neurotransmitter levels in rats with vascular dementia. Indian J. Anim. Res. 2018, 52, 1141–1145. [Google Scholar] [CrossRef]
- Wang, J.; Zhang, H.Y.; Tang, X.C. Cholinergic deficiency involved in vascular dementia: Possible mechanism and strategy of treatment. Acta Pharmacol. Sin. 2009, 30, 879–888. [Google Scholar] [CrossRef]
- Kumaran, D.; Udayabanu, M.; Kumar, M.; Aneja, R.; Katyal, A. Involvement of angiotensin converting enzyme in cerebral hypoperfusion induced anterograde memory impairment and cholinergic dysfunction in rats. Neuroscience 2008, 155, 626–639. [Google Scholar] [CrossRef] [PubMed]
- Kuang, X.; Du, J.R.; Liu, Y.X.; Zhang, G.Y.; Peng, H.Y. Postischemic administration of Z-Ligustilide ameliorates cognitive dysfunction and brain damage induced by permanent forebrain ischemia in rats. Pharmacol. Biochem. Behav. 2008, 88, 213–221. [Google Scholar] [CrossRef] [PubMed]
- Jeong, S.-J.; Sohn, E.; Kim, Y.J.; Lim, H.-S. Hwangryunhaedok-tang Exerts Neuropreventive Effect on Memory Impairment by reducing Cholinergic System Dysfunction and Inflammatory Response in Vascular Dementia Rat Model. Adv. Integr. Med. 2019, 6, S106. [Google Scholar] [CrossRef]
- Pohanka, M.; Dobes, P. Caffeine inhibits acetylcholinesterase, but not butyrylcholinesterase. Int. J. Mol. Sci. 2013, 14, 9873–9882. [Google Scholar] [CrossRef] [PubMed]
- Yancheva, S.; Ihl, R.; Schlaefke, S.; Hoerr, R. Ginkgo-biloba-Extract EGb 761®, donepezil or both combined in the treatment of Alzheimer’s disease with neuropsychiatric features: A randomised, double-blind trial. Z. Für Phyther. 2006, 27, V36. [Google Scholar] [CrossRef]
- Aksoy, P.; White, T.A.; Thompson, M.; Chini, E.N. Regulation of intracellular levels of NAD: A novel role for CD38. Biochem. Biophys. Res. Commun. 2006, 345, 1386–1392. [Google Scholar] [CrossRef]
- Young, G.S.; Choleris, E.; Lund, F.E.; Kirkland, J.B. Decreased cADPR and increased NAD+ in the Cd38-/- mouse. Biochem. Biophys. Res. Commun. 2006, 346, 188–192. [Google Scholar] [CrossRef]
- Kellenberger, E.; Kuhn, I.; Schuber, F.; Muller-Steffner, H. Flavonoids as inhibitors of human CD38. Bioorganic Med. Chem. Lett. 2011, 21, 3939–3942. [Google Scholar] [CrossRef]
- Al-Abady, Z.N.; Al-Shabany, A.J. Luteolin, a novel CD3 cycles inhibitor, is proposed regulator for modulation NAD-mediated glycol sis activity. Res. J. Pharm. Biol. Chem. Sci. 2018, 9, 486–495. [Google Scholar]
- Jiang, M.; Li, J.; Peng, Q.; Liu, Y.; Liu, W.; Luo, C.; Peng, J.; Li, J.; Yung, K.K.L.; Mo, Z. Neuroprotective effects of bilobalide on cerebral ischemia and reperfusion injury are associated with inhibition of pro-inflammatory mediator production and down-regulation of JNK1/2 and p38 MAPK activation. J. Neuroinflammation 2014, 11, 167. [Google Scholar] [CrossRef]
- Yuan, J.; Yankner, B.A. Apoptosis in the nervous system. Nature 2000, 407, 802–809. [Google Scholar] [CrossRef] [PubMed]
- Mahady, G.B. Ginkgo biloba: A Review of Quality, Safety, and Efficacy. Nutr. Clin. Care 2001, 4, 140–147. [Google Scholar] [CrossRef]
- Gold, P.E.; Cahill, L.; Wenk, G.L. The lowdown on Ginkgo biloba. Sci. Am. 2003, 288, 86–91. [Google Scholar] [CrossRef] [PubMed]
- Jeong, H.S.; Kim, K.H.; Lee, I.S.; Park, J.Y.; Kim, Y.; Kim, K.S.; Jang, H.J. Ginkgolide A ameliorates non-alcoholic fatty liver diseases on high fat diet mice. Biomed. Pharmacother. 2017, 88, 625–634. [Google Scholar] [CrossRef]
- Tao, Z.; Jin, W.; Ao, M.; Zhai, S.; Xu, H.; Yu, L. Evaluation of the anti-inflammatory properties of the active constituents in Ginkgo biloba for the treatment of pulmonary diseases. Food Funct. 2019, 10, 2209–2220. [Google Scholar] [CrossRef]
- Abebe, W. Herbal medication: Potential for adverse interactions with analgesic drugs. J. Clin. Pharm. Ther. 2002, 27, 391–401. [Google Scholar] [CrossRef]
- Lechler, E.; Penick, G.D. Blood clotting defect in hibernating ground squirrels (Citellus tridecemlineatus). Am. J. Physiol. 1963, 205, 985–988. [Google Scholar] [CrossRef]
- Nishino, A.; Suzuki, M.; Ohtani, H.; Motohashi, O.; Umezawa, K.; Nagura, H.; Yoshimoto, T. Thrombin May Contribute to the Pathophysiology of Central Nervous System Injury. J. Neurotrauma 2009, 10, 167–179. [Google Scholar] [CrossRef]
- Napryeyenko, O.; Sonnik, G.; Tartakovsky, I. Efficacy and tolerability of Ginkgo biloba extract EGb 761® by type of dementia: Analyses of a randomised controlled trial. J. Neurol. Sci. 2009, 283, 224–229. [Google Scholar] [CrossRef]
- Shi, J.; Perry, G.; Smith, M.A.; Friedland, R.P. Vascular abnormalities: The insidious pathogenesis of Alzheimer’s disease☆. Neurobiol. Aging 2000, 21, 357–361. [Google Scholar] [CrossRef]
- Nilsson, P.M. Early Vascular Aging in Hypertension. Front. Cardiovasc. Med. 2020, 7, 6. [Google Scholar] [CrossRef] [PubMed]
- Flint, H.J.; Duncan, S.H.; Scott, K.P.; Louis, P. Interactions and competition within the microbial community of the human colon: Links between diet and health: Minireview. Environ. Microbiol. 2007, 9, 1101–1111. [Google Scholar] [CrossRef] [PubMed]
- Biagi, E.; Nylund, L.; Candela, M.; Ostan, R.; Bucci, L.; Pini, E.; Nikkïla, J.; Monti, D.; Satokari, R.; Franceschi, C.; et al. Through ageing, and beyond: Gut microbiota and inflammatory status in seniors and centenarians. PLoS ONE 2010, 5, e10667. [Google Scholar] [CrossRef]
- Tan, J.K.; McKenzie, C.; Mariño, E.; Macia, L.; Mackay, C.R. Metabolite-Sensing G Protein–Coupled Receptors—Facilitators of Diet-Related Immune Regulation. Annu. Rev. Immunol. 2017, 35, 371–402. [Google Scholar] [CrossRef]
- Obrenovich, M. Leaky Gut, Leaky Brain? Microorganisms 2018, 6, 107. [Google Scholar] [CrossRef]
- Tran, L.; Greenwood-Van Meerveld, B. Age-associated remodeling of the intestinal epithelial barrier. J. Gerontol.-Ser. A Biol. Sci. Med. Sci. 2013, 68, 1045–1056. [Google Scholar] [CrossRef]
- Welling, M.M.; Nabuurs, R.J.A.; Van Der Weerd, L. Potential role of antimicrobial peptides in the early onset of Alzheimer’s disease. Alzheimer’s Dement. 2015, 11, 51–57. [Google Scholar] [CrossRef]
- Rocher, M.N.; Carré, D.; Spinnewyn, B.; Schulz, J.; Delaflotte, S.; Pignol, B.; Chabrier, P.E.; Auguet, M. Long-term treatment with standardized Ginkgo biloba Extract (EGb 761) attenuates cognitive deficits and hippocampal neuron loss in a gerbil model of vascular dementia. Fitoterapia 2011, 82, 1075–1080. [Google Scholar] [CrossRef]
- Biron, K.E.; Dickstein, D.L.; Gopaul, R.; Jefferies, W.A. Amyloid triggers extensive cerebral angiogenesis causing blood brain barrier permeability and hypervascularity in alzheimer’s disease. PLoS ONE 2011, 6, e23789. [Google Scholar] [CrossRef]
- Agúndez, J.A.G.; Jiménez-Jiménez, F.J.; Alonso-Navarro, H.; García-Martín, E. Drug and xenobiotic biotransformation in the blood–brain barrier: A neglected issue. Front. Cell. Neurosci. 2014, 8, 335. [Google Scholar] [CrossRef]
- Jones, R.M.; Mercante, J.W.; Neish, A.S. Neish Reactive Oxygen Production Induced by the Gut Microbiota: Pharmacotherapeutic Implications. Curr. Med. Chem. 2012, 19, 1519–1529. [Google Scholar] [CrossRef] [PubMed]
- Aliev, G.; Smith, M.A.; Obrenovich, M.E.; De La Torre, J.C.; Perry, G. Role of vascular hypoperfusion-induced oxidative stress and mitochondria failure in the pathogenesis of Alzheimer disease. Neurotox. Res. 2003, 5, 491–504. [Google Scholar] [CrossRef] [PubMed]
- Aliev, G.; Palacios, H.H.; Lipsitt, A.E.; Fischbach, K.; Lamb, B.T.; Obrenovich, M.E.; Morales, L.; Gasimov, E.; Bragin, V. Nitric Oxide as an Initiator of Brain Lesions During the Development of Alzheimer Disease. Neurotox. Res. 2009, 16, 293–305. [Google Scholar] [CrossRef] [PubMed]
- Aliev, G.; Obrenovich, M.E.; Tabrez, S.; Jabir, N.R.; Reddy, V.P.; Li, Y.; Burnstock, G.; Cacabelos, R.; Kamal, M.A. Link between cancer and Alzheimer disease via oxidative stress induced by nitric oxide-dependent mitochondrial DNA overproliferation and deletion. Oxidative Med. Cell. Longev. 2013, 2013, 962984. [Google Scholar] [CrossRef] [PubMed]
- Aliev, G.; Liu, J.; Puchowicz, M.; Xu, K.; Siedlak, S.L.; Obrenovich, M.E.; Shenk, J.C.; Smith, M.A.; Gasimov, E.; LaManna, J.C.; et al. Does the oxidative stress and cerebral atherosclerosis initiate brain hypoperfusion and the development of alzheimer disease? Atheroscler. Suppl. 2008, 9, 154. [Google Scholar] [CrossRef]
- Aliev, G.; Shenk, J.C.; Liu, J.; Puchowicz, M.; Xu, K.; Smith, M.A.; Siedlak, S.L.; Obrenovich, M.E.; Ames, B.N.; de la Torre, J.C.; et al. Preventative effect of mitochondrial antioxidants in apoe deficiency mouse model of brain hypoperfusion. J. Neurol. Sci. 2009, 283, 272. [Google Scholar] [CrossRef]
- Bastianetto, S.; Ramassamy, C.; Doré, S.; Christen, Y.; Poirier, J.; Quirion, R. The ginkgo biloba extract (EGb 761) protects hippocampal neurons against cell death induced by β-amyloid. Eur. J. Neurosci. 2000, 12, 1882–1890. [Google Scholar] [CrossRef]
- Sayre, L.M.; Zelasko, D.A.; Harris, P.L.R.; Perry, G.; Salomon, R.G.; Smith, M.A. 4-Hydroxynonenal-Derived Advanced Lipid Peroxidation End Products Are Increased in Alzheimer’s Disease. J. Neurochem. 1997, 68, 2092–2097. [Google Scholar] [CrossRef]
- Kelso, R.J.; Buszczak, M.; Quiñones, A.T.; Castiblanco, C.; Mazzalupo, S.; Cooley, L. Flytrap, a database documenting a GFP protein-trap insertion screen in Drosophila melanogaster. Nucleic Acids Res. 2004, 32, D418–D420. [Google Scholar] [CrossRef]
- Bridi, R.; Crossetti, F.P.; Steffen, V.M.; Henriques, A.T. The antioxidant activity of standardized extract of Ginkgo biloba (EGb 761) in rats. Phyther. Res. 2001, 15, 449–451. [Google Scholar] [CrossRef]
- Singh, S.K.; Srivastav, S.; Castellani, R.J.; Plascencia-Villa, G.; Perry, G. Neuroprotective and Antioxidant Effect of Ginkgo biloba Extract Against AD and Other Neurological Disorders. Neurotherapeutics 2019, 16, 666–674. [Google Scholar] [CrossRef] [PubMed]
- Varatharaj, A.; Galea, I. The blood-brain barrier in systemic inflammation. Brain. Behav. Immun. 2017, 60, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Spadoni, I.; Zagato, E.; Bertocchi, A.; Paolinelli, R.; Hot, E.; Di Sabatino, A.; Caprioli, F.; Bottiglieri, L.; Oldani, A.; Viale, G.; et al. A gut-vascular barrier controls the systemic dissemination of bacteria. Science 2015, 350, 830–834. [Google Scholar] [CrossRef]
- Maes, M.; Kubera, M.; Leunis, J.C. The gut-brain barrier in major depression: Intestinal mucosal dysfunction with an increased translocation of LPS from gram negative enterobacteria (leaky gut) plays a role in the inflammatory pathophysiology of depression. Neuro Endocrinol. Lett. 2008, 29, 117–124. [Google Scholar] [PubMed]
- Kolappan, S.; Coureuil, M.; Yu, X.; Nassif, X.; Egelman, E.H.; Craig, L. Structure of the neisseria meningitidis type IV pilus. Nat. Commun. 2016, 7, 13015. [Google Scholar] [CrossRef] [PubMed]
- Obrenovich, M.; Sankar Chittoor Mana, T.; Rai, H.; Shola, D.; Sass, C.; McCloskey, B.; Levison, B.S. Recent findings within the microbiota–gut–brain–endocrine metabolic interactome. Pathol. Lab. Med. Int. 2017, 9, 21–30. [Google Scholar] [CrossRef]
- de Abreu Feijó de Mello, A.; De Mello, M.F.; Carpenter, L.L.; Price, L.H. Update on stress and depression: The role of the hypothalamic-pituitary-adrenal (HPA) axis. Rev. Bras. Psiquiatr. 2003, 25, 231–238. [Google Scholar] [CrossRef]
- Haddad-Tóvolli, R.; Dragano, N.R.V.; Ramalho, A.F.S.; Velloso, L.A. Development and function of the blood-brain barrier in the context of metabolic control. Front. Neurosci. 2017, 11, 224. [Google Scholar] [CrossRef]
- Raza, M.W.; Shad, A.; Pedler, S.J.; Karamat, K.A. Penetration and activity of antibiotics in brain abscess. J. Coll. Physicians Surg. Pak. 2005, 15, 165–167. [Google Scholar]
- Kwak, D.W.; Hwang, H.S.; Kwon, J.Y.; Park, Y.W.; Kim, Y.H. Co-infection with vaginal Ureaplasma urealyticum and Mycoplasma hominis increases adverse pregnancy outcomes in patients with preterm labor or preterm premature rupture of membranes. J. Matern. Neonatal Med. 2014, 27, 333–337. [Google Scholar] [CrossRef]
- Obrenovich, M.; Rai, H.; Mana, T.S.; Shola, D.; McCloskey, B.; Sass, C.; Levison, B. BAOJ Microbiology Dietary Co-Metabolism within the Microbiota-Gut-Brain-Endocrine Metabolic Interactome. BAO Microbiol. 2017, 2, 10–2174. [Google Scholar]
- Erickson, M.A.; Banks, W.A. Blood-brain barrier dysfunction as a cause and consequence of Alzheimer’s disease. J. Cereb. Blood Flow Metab. 2013, 33, 1500–1513. [Google Scholar] [CrossRef]
- Coureuil, M.; Lécuyer, H.; Bourdoulous, S.; Nassif, X. A journey into the brain: Insight into how bacterial pathogens cross blood-brain barriers. Nat. Rev. Microbiol. 2017, 15, 149–159. [Google Scholar] [CrossRef] [PubMed]
- Poole, S.; Singhrao, S.K.; Chukkapalli, S.; Rivera, M.; Velsko, I.; Kesavalu, L.; Crean, S. Active invasion of Porphyromonas gingivalis and infection-induced complement activation in ApoE-/- mice brains. J. Alzheimer’s Dis. 2014, 43, 67–80. [Google Scholar] [CrossRef] [PubMed]
- Braniste, V.; Al-Asmakh, M.; Kowal, C.; Anuar, F.; Abbaspour, A.; Tóth, M.; Korecka, A.; Bakocevic, N.; Guan, N.L.; Kundu, P.; et al. The gut microbiota influences blood-brain barrier permeability in mice. Sci. Transl. Med. 2014, 6, 263ra158. [Google Scholar] [CrossRef]
- Prakash Reddy, V.; Obrenovich, M.E.; Atwood, C.S.; Perry, G.; Smith, M.A. Involvement of Maillard reactions in Alzheimer disease. Neurotox. Res. 2002, 4, 191–209. [Google Scholar] [CrossRef]
- Yan, F.L.; Zheng, Y.; Zhao, F.D. Effects of ginkgo biloba extract EGb761 on expression of RAGE and LRP-1 in cerebral microvascular endothelial cells under chronic hypoxia and hypoglycemia. Acta Neuropathol. 2008, 116, 529–535. [Google Scholar] [CrossRef]
- Monnier, V.M.; Obrenovich, M.E. Wake Up and Smell the Maillard Reaction. Sci. Aging Knowl. Environ. 2002, 2002, pe21. [Google Scholar] [CrossRef]
- Shenk, J.C.; Liu, J.; Fischbach, K.; Xu, K.; Puchowicz, M.; Obrenovich, M.E.; Gasimov, E.; Alvarez, L.M.; Ames, B.N.; LaManna, J.C.; et al. The effect of acetyl-L-carnitine and R-α-lipoic acid treatment in ApoE4 mouse as a model of human Alzheimer’s disease. J. Neurol. Sci. 2009, 283, 199–206. [Google Scholar] [CrossRef]
- Abdel-Kader, R.; Hauptmann, S.; Keil, U.; Scherping, I.; Leuner, K.; Eckert, A.; Müller, W.E. Stabilization of mitochondrial function by Ginkgo biloba extract (EGb 761). Pharmacol. Res. 2007, 56, 493–502. [Google Scholar] [CrossRef]
- Vojdani, A.; Lerner, A.; Vojdani, E. Cross-Reactivity and Sequence Homology Between Alpha-Synuclein and Food Products: A Step Further for Parkinson’s Disease Synucleinopathy. Cells 2021, 10, 1111. [Google Scholar] [CrossRef] [PubMed]
- Kirschner, D.A.; Abraham, C.; Selkoe, D.J. X-ray diffraction from intraneuronal paired helical filaments and extraneuronal amyloid fibers in Alzheimer disease indicates cross-β conformation. Proc. Natl. Acad. Sci. USA 1986, 83, 503–507. [Google Scholar] [CrossRef] [PubMed]
- Wiśniewski, H.M.; Narang, H.K.; Terry, R.D. Neurofibrillary tangles of paired helical filaments. J. Neurol. Sci. 1976, 27, 173–181. [Google Scholar] [CrossRef] [PubMed]
- Obrenovich, M.E.; Monnier, V.M. Glycation stimulates amyloid formation. Sci. Aging Knowl. Environ. 2004, 2004, 3. [Google Scholar] [CrossRef] [PubMed]
- Younis, A.M.; Ibrahim, A.R.S.; Ibrahim, S.M.; Aboulsoud, K.A.; Kabbash, A.M. Microbial transformation of curcumin and evaluation of the biological activities of the isolated metabolites. J. Pharm. Sci. Res. 2016, 8, 1169. [Google Scholar]
- Bedir, E.; Tatli, I.I.; Khan, R.A.; Zhao, J.; Takamatsu, S.; Walker, L.A.; Goldman, P.; Khan, I.A. Biologically active secondary metabolites from Ginkgo biloba. J. Agric. Food Chem. 2002, 50, 3150–3155. [Google Scholar] [CrossRef]
- Chen, F.; Dong, R.R.; Zhong, K.L.; Ghosh, A.; Tang, S.S.; Long, Y.; Hu, M.; Miao, M.X.; Liao, J.M.; Sun, H.B.; et al. Antidiabetic drugs restore abnormal transport of amyloid-β across the blood-brain barrier and memory impairment in db/db mice. Neuropharmacology 2016, 101, 123–136. [Google Scholar] [CrossRef]
- Da Zhang, L.; Ma, L.; Zhang, L.; Dai, J.G.; Chang, L.G.; Huang, P.L.; Tian, X.Q. Hyperbaric oxygen and ginkgo biloba extract ameliorate cognitive and memory impairment via nuclear factor Kappa-B pathway in rat model of alzheimer’s disease. Chin. Med. J. 2015, 128, 3088–3093. [Google Scholar] [CrossRef]
- Perez-Muñoz, M.E.; Arrieta, M.C.; Ramer-Tait, A.E.; Walter, J. A critical assessment of the “sterile womb” and “in utero colonization” hypotheses: Implications for research on the pioneer infant microbiome. Microbiome 2017, 5, 48. [Google Scholar] [CrossRef]
- Kim, K.S. Mechanisms of microbial traversal of the blood-brain barrier. Nat. Rev. Microbiol. 2008, 6, 625–634. [Google Scholar] [CrossRef]
- Zipser, B.D.; Johanson, C.E.; Gonzalez, L.; Berzin, T.M.; Tavares, R.; Hulette, C.M.; Vitek, M.P.; Hovanesian, V.; Stopa, E.G. Microvascular injury and blood-brain barrier leakage in Alzheimer’s disease. Neurobiol. Aging 2007, 28, 977–986. [Google Scholar] [CrossRef] [PubMed]
- Banack, S.A.; Caller, T.A.; Stommel, E.W. The cyanobacteria derived toxin beta-N-methylamino-L-alanine and amyotrophic lateral sclerosis. Toxins 2010, 2, 2837–2850. [Google Scholar] [CrossRef] [PubMed]
- Roychowdhury, S.; Cadnum, J.; Glueck, B.; Obrenovich, M.; Donskey, C.; Cresci, G.A.M. Faecalibacterium prausnitzii and a Prebiotic Protect Intestinal Health in a Mouse Model of Antibiotic and Clostridium difficile Exposure. J. Parenter. Enter. Nutr. 2018, 42, 1156–1167. [Google Scholar] [CrossRef] [PubMed]
- Ait-Belgnaoui, A.; Durand, H.; Cartier, C.; Chaumaz, G.; Eutamene, H.; Ferrier, L.; Houdeau, E.; Fioramonti, J.; Bueno, L.; Theodorou, V. Prevention of gut leakiness by a probiotic treatment leads to attenuated HPA response to an acute psychological stress in rats. Psychoneuroendocrinology 2012, 37, 1885–1895. [Google Scholar] [CrossRef]
- Chen, F.; Li, L.; Xu, F.; Sun, Y.; Du, F.; Ma, X.; Zhong, C.; Li, X.; Wang, F.; Zhang, N.; et al. Systemic and cerebral exposure to and pharmacokinetics of flavonols and terpene lactones after dosing standardized Ginkgo biloba leaf extracts to rats via different routes of administration. Br. J. Pharmacol. 2013, 170, 440–457. [Google Scholar] [CrossRef]
- Aa, L.; Fei, F.; Tan, Z.; Aa, J.; Wang, G.; Liu, C. The pharmacokinetics study of ginkgolide A, B and the effect of food on bioavailability after oral administration of ginkgolide extracts in beagle dogs. Biomed. Chromatogr. 2018, 32, e4212. [Google Scholar] [CrossRef]
- Yan-Yan, Z.; Li-Li, G.; Guo-Ming, S.; Rong, R.; Jing-Zhen, T. Determination of ginkgolides A, B, C, J and bilobalide in plasma by LC-ESI (-)/MS/MS (QQQ) and its application to the pharmacokinetic study of ginkgo biloba extract in rats. Drug Res. 2016, 66, 520–526. [Google Scholar] [CrossRef]
- Murota, K.; Nakamura, Y.; Uehara, M. Flavonoid metabolism: The interaction of metabolites and gut microbiota. Biosci. Biotechnol. Biochem. 2018, 82, 600–610. [Google Scholar] [CrossRef]
- Kawabata, K.; Yoshioka, Y.; Terao, J. Role of intestinal microbiota in the bioavailability and physiological functions of dietary polyphenols. Molecules 2019, 24, 370. [Google Scholar] [CrossRef]
- Lin, W.; Wang, W.; Yang, H.; Wang, D.; Ling, W. Influence of Intestinal Microbiota on the Catabolism of Flavonoids in Mice. J. Food Sci. 2016, 81, H3026–H3034. [Google Scholar] [CrossRef]
- Bombardelli: Cosmetical Uses of Ginkgo Extracts and Constituents. Available online: https://scholar.google.com/scholar_lookup?title=CosmeticalusesofGinkgoextractsandconstituents&author=E.Bombardelli&author=A.Cristoni&author=&author=P.Morazzoni&publication_year=2000 (accessed on 10 May 2021).
- Choi, M.S.; Kim, J.K.; Kim, D.H.; Yoo, H.H. Effects of gut microbiota on the bioavailability of bioactive compounds from Ginkgo leaf extracts. Metabolites 2019, 9, 132. [Google Scholar] [CrossRef] [PubMed]
- Dethlefsen, L.; Huse, S.; Sogin, M.L.; Relman, D.A. The pervasive effects of an antibiotic on the human gut microbiota, as revealed by deep 16s rRNA sequencing. PLoS Biol. 2008, 6, e280. [Google Scholar] [CrossRef] [PubMed]
- Jin, M.J.; Kim, U.; Kim, I.S.; Kim, Y.; Kim, D.H.; Han, S.B.; Kwon, O.S.; Yoo, H.H. Effects of gut microflora on pharmacokinetics of hesperidin: A study on non-antibiotic and pseudo-germ-free rats. J. Toxicol. Environ. Health-Part A Curr. Issues 2010, 73, 1441–1450. [Google Scholar] [CrossRef] [PubMed]
- Kim, I.S.; Yoo, D.H.; Jung, I.H.; Lim, S.; Jeong, J.J.; Kim, K.A.; Bae, O.N.; Yoo, H.H.; Kim, D.H. Reduced metabolic activity of gut microbiota by antibiotics can potentiate the antithrombotic effect of aspirin. Biochem. Pharmacol. 2016, 122, 72–79. [Google Scholar] [CrossRef]
- Wang, L.; Ravichandran, V.; Yin, Y.; Yin, J.; Zhang, Y. Natural Products from Mammalian Gut Microbiota. Trends Biotechnol. 2019, 37, 492–504. [Google Scholar] [CrossRef]
- Le, T.; Bhushan, V.; Sochat, M. First Aid for the USMLE Step 1 2021; McGraw-Hill Education: New York, NY, USA, 2021. [Google Scholar]
- Román, G.C. Cholinergic dysfunction in vascular dementia. Curr. Psychiatry Rep. 2005, 7, 18–26. [Google Scholar] [CrossRef]
- Obrenovich, M.; Siddiqui, B.; McCloskey, B.; Reddy, V.P. The microbiota–gut–brain axis heart shunt part i: The french paradox, heart disease and the microbiota. Microorganisms 2020, 8, 490. [Google Scholar] [CrossRef]
- Román, G.C.; Kalaria, R.N. Vascular determinants of cholinergic deficits in Alzheimer disease and vascular dementia. Neurobiol. Aging 2006, 27, 1769–1785. [Google Scholar] [CrossRef]
- LaVoie, M.J.; Ostaszewski, B.L.; Weihofen, A.; Schlossmacher, M.G.; Selkoe, D.J. Dopamine covalently modifies and functionally inactivates parkin. Nat. Med. 2005, 11, 1214–1221. [Google Scholar] [CrossRef]
- Goyal, N.; Siddiqui, S.; Chatterjee, U.; Kumar, D.; Siddiqui, A. Neuropsychology of prefrontal cortex. Indian J. Psychiatry 2008, 50, 202–208. [Google Scholar] [CrossRef]
- Mazza, M.; Capuano, A.; Bria, P.; Mazza, S. Ginkgo biloba and donepezil: A comparison in the treatment of Alzheimer’s dementia in a randomized placebo-controlled double-blind study. Eur. J. Neurol. 2006, 13, 981–985. [Google Scholar] [CrossRef] [PubMed]
- Herrschaft, H.; Nacu, A.; Likhachev, S.; Sholomov, I.; Hoerr, R.; Schlaefke, S. Ginkgo biloba extract EGb 761® in dementia with neuropsychiatric features: A randomised, placebo-controlled trial to confirm the efficacy and safety of a daily dose of 240 mg. J. Psychiatr. Res. 2012, 46, 716–723. [Google Scholar] [CrossRef] [PubMed]
- Ahlemeyer, B.; Krieglstein, J. Pharmacological studies supporting the therapeutic use of Ginkgo biloba extract for Alzheimer’s disease. Pharmacopsychiatry 2003, 36, 8–14. [Google Scholar] [CrossRef]
- Kleijnen, J.; Knipschild, P. Ginkgo biloba for cerebral insufficiency. Br. J. Clin. Pharmacol. 1992, 34, 352–358. [Google Scholar] [CrossRef] [PubMed]
- Bastianetto, S.; Quirion, R. EGb 761 is a neuroprotective agent against beta-amyloid toxicity. Cell. Mol. Biol. 2002, 48, 693–697. [Google Scholar]
- Christen, Y. Oxidative stress and Alzheimer disease. Am. J. Clin. Nutr. 2000, 71, 621S–629S. [Google Scholar] [CrossRef]
- Boles Ponto, L.L.; Schultz, S.K. Ginkgo biloba Extract: Review of CNS Effects. Ann. Clin. Psychiatry 2003, 15, 109–119. [Google Scholar] [CrossRef]
- Yang, Y.; Liu, P.; Chen, L.; Liu, Z.; Zhang, H.; Wang, J.; Sun, X.; Zhong, W.; Wang, N.; Tian, K.; et al. Therapeutic effect of Ginkgo biloba polysaccharide in rats with focal cerebral ischemia/reperfusion (I/R) injury. Carbohydr. Polym. 2013, 98, 1383–1388. [Google Scholar] [CrossRef]
- Kim, M.S.; Lee, J.I.; Lee, W.Y.; Kim, S.E. Neuroprotective effect of Ginkgo biloba L. extract in a rat model of parkinson’s disease. Phytother. Res. 2004, 18, 663–666. [Google Scholar] [CrossRef]
- Wu, W.R.; Zhu, X.Z. Involvement of monoamine oxidase inhibition in neuroprotective and neurorestorative effects of Ginkgo biloba extract against MPTP-induced nigrostriatal dopaminergic toxicity in C57 mice. Life Sci. 1999, 65, 157–164. [Google Scholar] [CrossRef]
- Le Bars, P.L.; Katz, M.M.; Berman, N.; Itil, T.M.; Freedman, A.M.; Schatzberg, A.F. A placebo-controlled, double-blind, randomized trial of an extract of Ginkgo biloba for dementia. JAMA 1997, 278, 1327–1332. [Google Scholar] [CrossRef] [PubMed]
- Yancheva, S.; Ihl, R.; Nikolova, G.; Panayotov, P.; Schlaefke, S.; Hoerr, R. Ginkgo biloba extract EGb 761, donepezil or both combined in the treatment of Alzheimer’s disease with neuropsychiatric features: A randomised, double-blind, exploratory trial. Aging Ment Health 2009, 13, 183–190. [Google Scholar] [CrossRef] [PubMed]
- Mix, J.A.; Crews, W.D. A double-blind, placebo-controlled, randomized trial of Ginkgo biloba extract EGb 761® in a sample of cognitively intact older adults: Neuropsychological findings. Hum. Psychopharmacol. 2002, 17, 267–277. [Google Scholar] [CrossRef] [PubMed]
- DeKosky, S.T.; Williamson, J.D.; Fitzpatrick, A.L.; Kronmal, R.A.; Ives, D.G.; Saxton, J.A. Ginkgo biloba for prevention of dementia: A randomized controlled trial. JAMA—J. Am. Med. Assoc. 2008, 300, 2253–2262. [Google Scholar] [CrossRef]
- McCarney, R.; Fisher, P.; Iliffe, S.; van Haselen, R.; Griffin, M.; Van Der Meulen, J.; Warner, J. Ginkgo biloba for mild to moderate dementia in a community setting: A pragmatic, randomised, parallel-group, double-blind, placebo-controlled trial. Int. J. Geriatr. Psychiatry 2008, 23, 1222–1230. [Google Scholar] [CrossRef]
- Diamond, B.J.; Shiflett, S.C.; Feiwel, N.; Matheis, R.J.; Noskin, O.; Richards, J.A.; Schoenberger, N.E. Ginkgo biloba extract: Mechanisms and clinical indications. Arch. Phys. Med. Rehabil. 2000, 81, 668–678. [Google Scholar] [CrossRef]
- Yuan, Q.; Wang, C.W.; Shi, J.; Lin, Z.X. Effects of Ginkgo biloba on dementia: An overview of systematic reviews. J. Ethnopharmacol. 2017, 195, 1–9. [Google Scholar] [CrossRef]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Zock, P.L.; Kromhout, D.; Hollman, P.C.H. Effects of the pure flavonoids epicatechin and quercetin on vascular function and cardiometabolic health: A randomized, double-blind, placebo-controlled, crossover trial. Am. J. Clin. Nutr. 2015, 101, 914–921. [Google Scholar] [CrossRef]
- Dower, J.I.; Geleijnse, J.M.; Gijsbers, L.; Schalkwijk, C.; Kromhout, D.; Hollman, P.C. Supplementation of the pure flavonoids epicatechin and quercetin affects some biomarkers of endothelial dysfunction and inflammation in (pre)hypertensive adults: A randomized double-blind, placebo-controlled, crossover trial. J. Nutr. 2015, 145, 1459–1463. [Google Scholar] [CrossRef]
- Song, M.C.; Kim, E.J.; Kim, E.; Rathwell, K.; Nam, S.J.; Yoon, Y.J. Microbial biosynthesis of medicinally important plant secondary metabolites. Nat. Prod. Rep. 2014, 31, 1497–1509. [Google Scholar] [CrossRef]
- Schmitz, A.; Felder, S.; Höver, T.; Kehraus, S.; Neu, E.; Lohr, F.; König, G.M.; Schäberle, T.F. Antibiotics from gliding bacteria. Phytochem. Rev. 2013, 12, 507–516. [Google Scholar] [CrossRef]
- Pham, J.V.; Yilma, M.A.; Feliz, A.; Majid, M.T.; Maffetone, N.; Walker, J.R.; Kim, E.; Cho, H.J.; Reynolds, J.M.; Song, M.C.; et al. A review of the microbial production of bioactive natural products and biologics. Front. Microbiol. 2019, 10, 1404. [Google Scholar] [CrossRef] [PubMed]
- Pimentel, M.R.; Molina, G.; Dionísio, A.P.; Maróstica Junior, M.R.; Pastore, G.M. The Use of Endophytes to Obtain Bioactive Compounds and Their Application in Biotransformation Process. Biotechnol. Res. Int. 2011, 2011, 576286. [Google Scholar] [CrossRef] [PubMed]
- Jones, L.; Kumar, J.; Mistry, A.; Mana, T.S.C.; Perry, G.; Reddy, V.P.; Obrenovich, M. The transformative possibilities of the microbiota and mycobiota for health, disease, aging, and technological innovation. Biomedicines 2019, 7, 24. [Google Scholar] [CrossRef] [PubMed]
- Louis, P.; Flint, H.J. Formation of propionate and butyrate by the human colonic microbiota. Environ. Microbiol. 2017, 19, 29–41. [Google Scholar] [CrossRef]
- Ansell, J.; Parkar, S.; Paturi, G.; Rosendale, D.; Blatchford, P. Modification of the Colonic Microbiota; Elsevier: Amsterdam, The Netherland, 2013. [Google Scholar] [CrossRef]
- Bavington, C.; Page, C. Stopping Bacterial Adhesion: A Novel Approach to Treating Infections. Respiration 2005, 72, 335–344. [Google Scholar] [CrossRef]
- De La Torre, J.C. Critically attained threshold of cerebral hypoperfusion: The CATCH hypothesis of Alzheimer’s pathogenesis. Neurobiol. Aging 2000, 21, 331–342. [Google Scholar] [CrossRef]
- Etiene, D.; Kraft, J.; Ganju, N.; Gomez-Isla, T.; Gemelli, B.; Hyman, B.T.; Hedley-Whyte, E.T.; Wands, J.R.; De la Monte, S.M. Cerebrovascular pathology contributes to the heterogeneity of Alzheimer’s disease. J. Alzheimer’s Dis. 1998, 1, 119–134. [Google Scholar] [CrossRef]
- Kalaria, R.N.; Ballard, C. Overlap between pathology of Alzheimer disease and vascular dementia. Alzheimer Dis. Assoc. Disord. 1999, 13, S115–S123. [Google Scholar] [CrossRef]
- Ahlemeyer, B.; Krieglstein, J. Neuroprotective effects of Ginkgo biloba extract. Cell. Mol. Life Sci. 2003, 60, 1779–1792. [Google Scholar] [CrossRef]
Disclaimer/Publisher’s Note: The statements, opinions and data contained in all publications are solely those of the individual author(s) and contributor(s) and not of MDPI and/or the editor(s). MDPI and/or the editor(s) disclaim responsibility for any injury to people or property resulting from any ideas, methods, instructions or products referred to in the content. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).