Bioactive Compounds in Garlic (Allium sativum) and Black Garlic as Antigout Agents, Using Computer Simulation
Abstract
:1. Introduction
2. Materials and Methods
2.1. Preparation and Sample Extraction
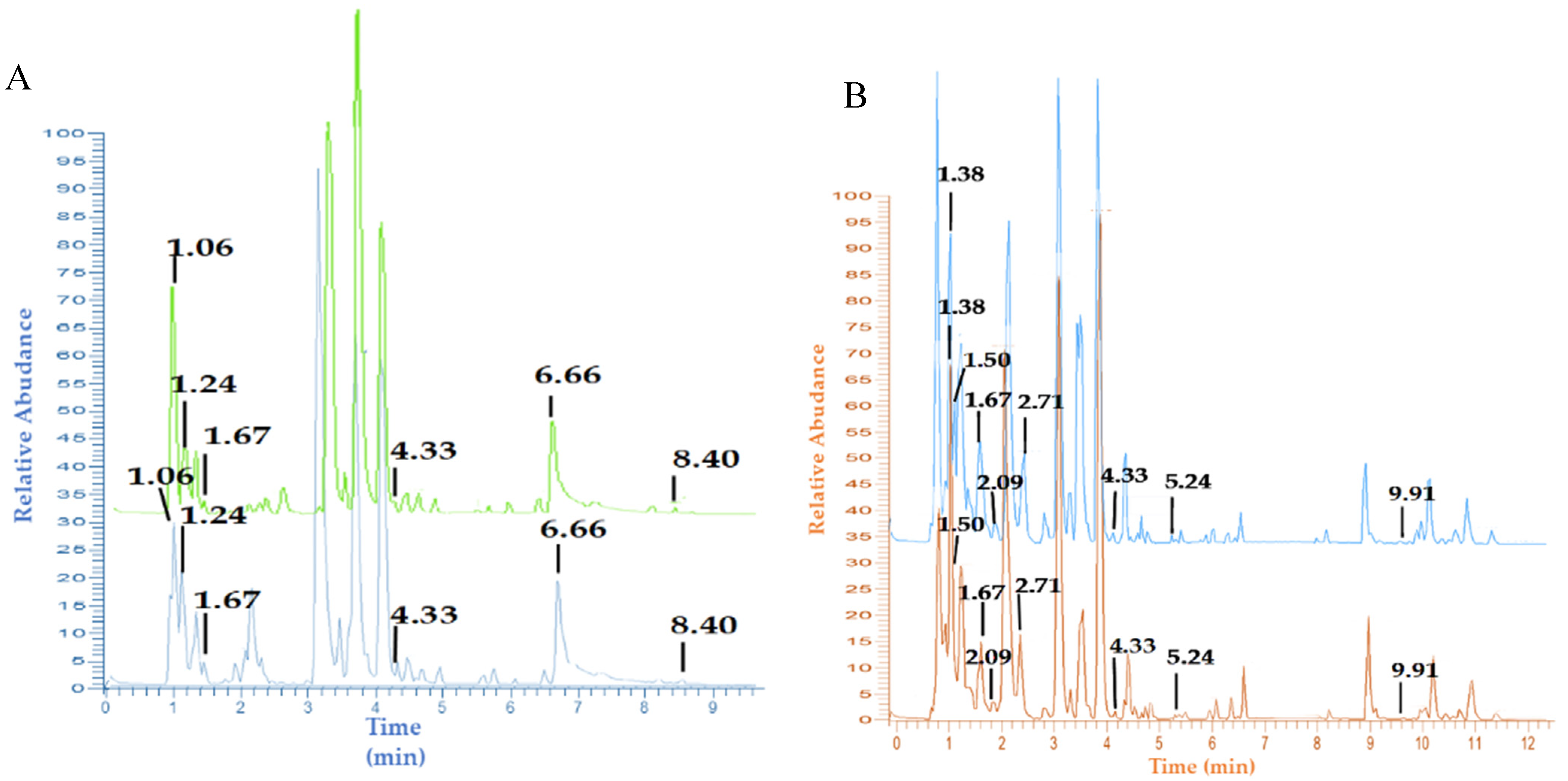
2.2. Identification of Metabolites by UHPLC-Q-Orbitrap-MS/MS
2.3. Statistical Analysis
2.4. Molecular Dynamics
2.4.1. Protein Preparation
2.4.2. Molecular Dynamics Simulation
2.5. Ensemble Docking
2.6. Screening of Bioactive Compounds as Drug Candidates
3. Results
3.1. Metabolite Profiles of Garlic and Black Garlic Extracts
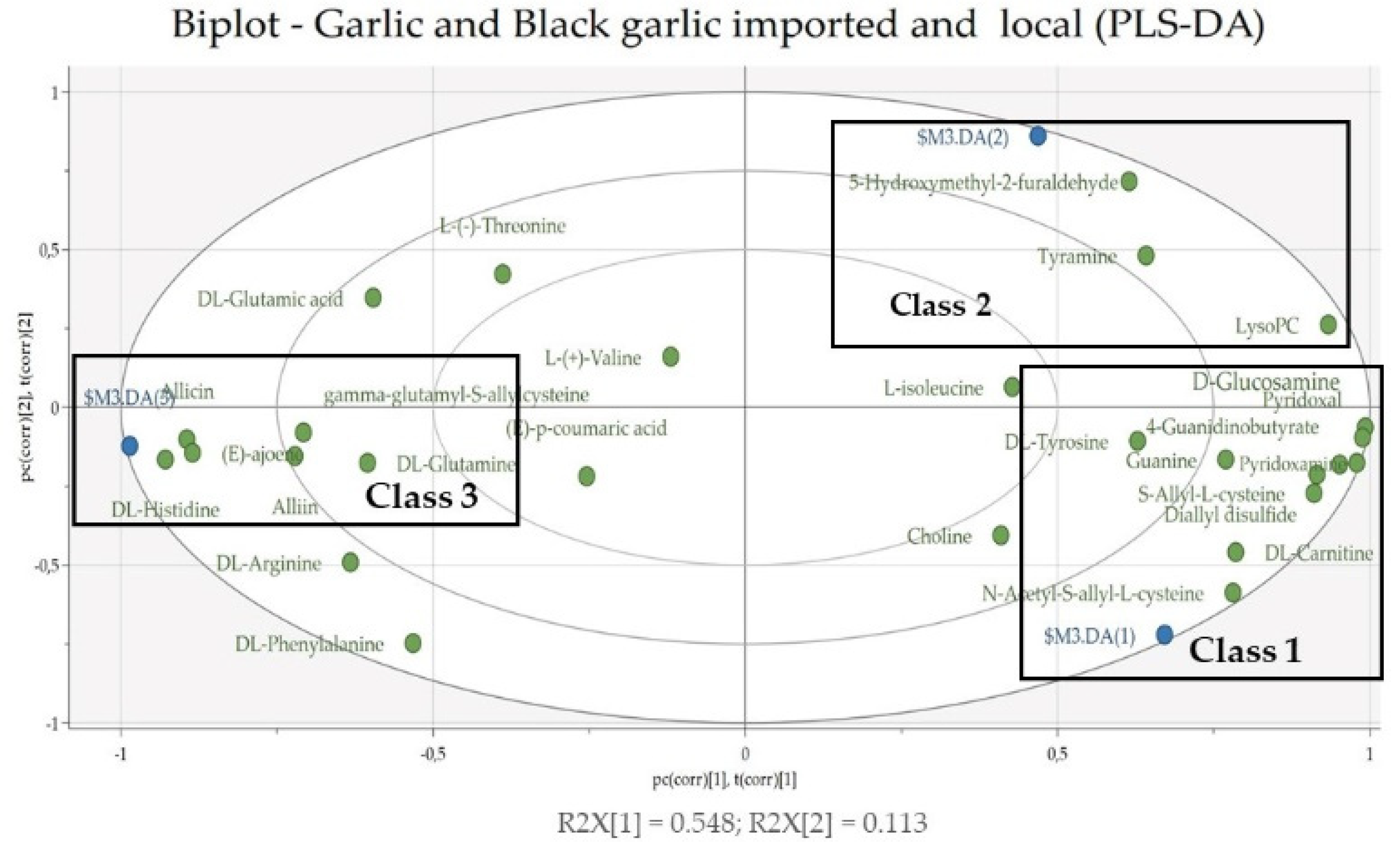
3.2. Multivariate Data Analysis
3.3. Molecular Dynamic Simulation (MD)
3.4. Ensemble Docking
3.5. Screening of Bioactive Compounds as Drug Candidates
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Dalbeth, N.; Merriman, T.R.; Stamp, L.K. Gout. Lancet 2016, 388, 2039–2052. [Google Scholar] [CrossRef]
- Kuo, C.-F.; Grainge, M.J.; Mallen, C.; Zhang, W.; Doherty, M. Comorbidities in patients with gout prior to and following diagnosis: Case-control study. Ann. Rheum. Dis. 2014, 75, 210–217. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Bardin, T.; Richette, P. Impact of comorbidities on gout and hyperuricaemia: An update on prevalence and treatment options. BMC Med. 2017, 15, 123. [Google Scholar] [CrossRef]
- Ragab, G.; Elshahaly, M.; Bardin, T. Gout: An old disease in new perspective—A review. J. Adv. Res. 2017, 8, 495–511. [Google Scholar] [CrossRef]
- Battelli, M.G.; Polito, L.; Bortolotti, M.; Bolognesi, A. Xanthine Oxidoreductase in Drug Metabolism: Beyond a Role as a De-toxifying Enzyme. Curr. Med. Chem. 2016, 23, 4027–4036. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Chung, H.Y.; Baek, B.S.; Song, S.H.; Kim, M.S.; Huh, J.I.; Shim, K.H.; Kim, K.W.; Lee, K.H. Xanthine Dehydrogenase/Xanthine Oxldase and Oxidative Stress. J. Am. Aging Assoc. 1997, 20, 127–140. [Google Scholar]
- Mahor, D.; Prasad, G.S. Biochemical Characterization of Kluyveromyces lactis Adenine Deaminase and Guanine Deaminase and Their Potential Application in Lowering Purine Content in Beer. Front. Bioeng. Biotechnol. 2018, 6, 180. [Google Scholar] [CrossRef]
- Mahor, D.; Priyanka, A.; Prasad, G.S.; Thakur, K.G. Functional and Structural Characterization of Purine Nucleoside Phos-phorylase from Kluyveromyces Lactis and Its Potential Applications in Reducing Purine Content in Food. PLoS ONE 2016, 11, e0164279. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, L.; Yang, D.; Wang, Y.; Yang, X.; Chen, K.; Luo, X.; Xu, J.; Liu, Y.; Tang, Z.; Zhang, Q.; et al. 5′-Nucleotidase Plays a Key Role in Uric Acid Metabolism of Bombyx mori. Cells 2021, 10, 2243. [Google Scholar] [CrossRef]
- Forkmann, A.T.M.C.M. As Compared to Allopurinol, Urate-Lowering Therapy with Febuxostat Has Superior Effects on Oxidative Stress and Pulse Wave Velocity in Patients with Severe Chronic Tophaceous Gout. Rheumatol. Int. 2014, 34, 101–109. [Google Scholar]
- Eddouks, M.; Ajebli, M.; Hebi, M. Ethnopharmacological survey of medicinal plants used in Daraa-Tafilalet region (Province of Errachidia), Morocco. J. Ethnopharmacol. 2017, 198, 516–530. [Google Scholar] [CrossRef]
- Jiang, T.A. Health Benefits of Culinary Herbs and Spices. J. AOAC Int. 2019, 102, 395–411. [Google Scholar] [CrossRef] [PubMed]
- Bayan, L.; Koulivand, P.H.; Gorji, A. Garlic: A review of potential therapeutic effects. Avicenna J. Phytomed. 2014, 4, 1–14. [Google Scholar] [CrossRef]
- Kimura, S.; Tung, Y.-C.; Pan, M.-H.; Su, N.-W.; Lai, Y.-J.; Cheng, K.-C. Black garlic: A critical review of its production, bioactivity, and application. J. Food Drug Anal. 2017, 25, 62–70. [Google Scholar] [CrossRef] [Green Version]
- Qiu, Z.; Zheng, Z.; Zhang, B.; Sun-Waterhouse, D.; Qiao, X. Formation, nutritional value, and enhancement of characteristic components in black garlic: A review for maximizing the goodness to humans. Compr. Rev. Food Sci. Food Saf. 2020, 19, 801–834. [Google Scholar] [CrossRef] [Green Version]
- El-Saber Batiha, G.; Magdy Beshbishy, A.; Wasef, L.G.; Elewa, Y.H.; Al-Sagan, A.A.; El-Hack, A.; Taha, M.E.; Abd-Elhakim, Y.M.; Prasad Devkota, H. Chemical Constituents and Pharmacological Activities of Garlic (Allium sativum L.): A Review. Nutrients 2020, 12, 872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Shang, A.; Cao, S.Y.; Xu, X.Y.; Gan, R.Y.; Tang, G.Y.; Corke, H.; Mavumengwana, V.; Li, H. Bin Bioactive Compounds and Biological Functions of Garlic (Allium sativum L.). Foods 2019, 8, 246. [Google Scholar] [CrossRef] [Green Version]
- Johnson, P.; Loganathan, C.; Iruthayaraj, A.; Poomani, K.; Thayumanavan, P. S-allyl cysteine as potent anti-gout drug: Insight into the xanthine oxidase inhibition and anti-inflammatory activity. Biochimie 2018, 154, 1–9. [Google Scholar] [CrossRef]
- Campbell, A.J.; Lamb, M.L.; Joseph-McCarthy, D. Ensemble-Based Docking Using Biased Molecular Dynamics. J. Chem. Inf. Model. 2014, 54, 2127–2138. [Google Scholar] [CrossRef]
- Emir, A.; Emir, C.; Yıldırım, H. Characterization of Phenolic Profile by LC-ESI-MS/MS and Enzyme Inhibitory Activities of Two Wild Edible Garlic: Allium Nigrum L. and Allium subhirsutum L. J. Food Biochem. 2020, 44, e13165. [Google Scholar] [CrossRef] [PubMed]
- Emir, C.; Emir, A. Phytochemical Analyses with LC-MS/MS and in Vitro Enzyme Inhibitory Activities of an Endemic Species “Allium Stylosum O. Schwarz” (Amaryllidaceae). S. Afr. J. Bot. 2021, 136, 70–75. [Google Scholar] [CrossRef]
- Rafi, M.; Karomah, A.H.; Heryanto, R.; Septaningsih, D.A.; Kusuma, W.A.; Amran, M.B.; Rohman, A.; Prajogo, B. Metabolite profiling of Andrographis paniculata leaves and stem extract using UHPLC-Orbitrap-MS/MS. Nat. Prod. Res. 2020, 36, 625–629. [Google Scholar] [CrossRef] [PubMed]
- Anandakrishnan, R.; Aguilar, B.; Onufriev, A.V. H++ 3.0: Automating pK prediction and the preparation of biomolecular structures for atomistic molecular modeling and simulations. Nucleic Acids Res. 2012, 40, W537–W541. [Google Scholar] [CrossRef] [Green Version]
- Bitencourt-Ferreira, G.; Pintro, V.O.; de Azevedo, W.F. Docking with AutoDock4. Methods Mol. Biol. 2019, 2053, 125–148. [Google Scholar] [CrossRef] [PubMed]
- Zhang, J.; Chen, H.; Fan, C.; Gao, S.; Zhang, Z.; Bo, L. Classification of the botanical and geographical origins of Chinese honey based on 1H NMR profile with chemometrics. Food Res. Int. 2020, 137, 109714. [Google Scholar] [CrossRef]
- Eriksson, L.; Trygg, J.; Wold, S. CV-ANOVA for significance testing of PLS and OPLS® models. J. Chemom. 2008, 22, 594–600. [Google Scholar] [CrossRef]
- Martínez, L. Automatic Identification of Mobile and Rigid Substructures in Molecular Dynamics Simulations and Fractional Structural Fluctuation Analysis. PLoS ONE 2015, 10, e0119264. [Google Scholar] [CrossRef] [Green Version]
- Seth, R.; Asr, K.; Buchbinder, R.; Bombardier, C.; Cj, E. Allopurinol for Chronic Gout (Review); Cochrane Library: Hoboken, NJ, USA, 2014; pp. 1–82. [Google Scholar]
- Cividini, F.; Pesi, R.; Chaloin, L.; Allegrini, S.; Camici, M.; Cros-Perrial, E.; Dumontet, C.; Jordheim, L.; Tozzi, M. The purine analog fludarabine acts as a cytosolic 5′-nucleotidase II inhibitor. Biochem. Pharmacol. 2015, 94, 63–68. [Google Scholar] [CrossRef]
- Kutryb-Zajac, B.; Mierzejewska, P.; Slominska, E.M.; Smolenski, R.T. Therapeutic Perspectives of Adenosine Deaminase In-hibition in Cardiovascular Diseases. Molecules 2020, 25, 4652. [Google Scholar] [CrossRef]
- Pathania, S.; Singh, P.K. Analyzing FDA-approved drugs for compliance of pharmacokinetic principles: Should there be a critical screening parameter in drug designing protocols? Expert Opin. Drug Metab. Toxicol. 2021, 17, 351–354. [Google Scholar] [CrossRef]
- Bin Choy, Y.; Prausnitz, M.R. The Rule of Five for Non-Oral Routes of Drug Delivery: Ophthalmic, Inhalation and Transdermal. Pharm. Res. 2011, 28, 943–948. [Google Scholar] [CrossRef] [Green Version]
- Cheng, F.; Li, W.; Zhou, Y.; Shen, J.; Wu, Z.; Liu, G.; Lee, P.W.; Tang, Y. admetSAR: A Comprehensive Source and Free Tool for Assessment of Chemical ADMET Properties. J. Chem. Inf. Model. 2012, 52, 3099–3105. [Google Scholar] [CrossRef] [PubMed]
- Daoudi, N.E.; Bouhrim, M.; Ouassou, H.; Bnouham, M. Medicinal Plants as a Drug Alternative Source for the Antigout Therapy in Morocco. Scientifica 2020, 2020, 1–10. [Google Scholar] [CrossRef] [PubMed]
- Yuan, H.; Sun, L.; Chen, M.; Wang, J. The Comparison of the Contents of Sugar, Amadori, and Heyns Compounds in Fresh and Black Garlic. J. Food Sci. 2016, 81, C1662–C1668. [Google Scholar] [CrossRef]
- Choi, I.S.; Cha, H.S.; Lee, Y.S. Physicochemical and Antioxidant Properties of Black Garlic. Molecules 2014, 19, 16811–16823. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Evangelista, W.; Weir, R.L.; Ellingson, S.R.; Harris, J.B.; Kapoor, K.; Smith, J.C.; Baudry, J. Ensemble-based docking: From hit discovery to metabolism and toxicity predictions. Bioorganic Med. Chem. 2016, 24, 4928–4935. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Setiawan, A.A.; Kumala, S.; Dian Ratih, L.; Yuliana, N.D. In Silico Study on S-Allyl Cysteine and Quercetin from Garlic (Allium Sativum Linn) as Xanthine Oxidase Inhibitor. In Proceedings of the International Seminar on Pharmaceutical Sciences and Technology, Philadelphia, PA, USA, 18–20 October 2018; pp. 1–7. [Google Scholar]

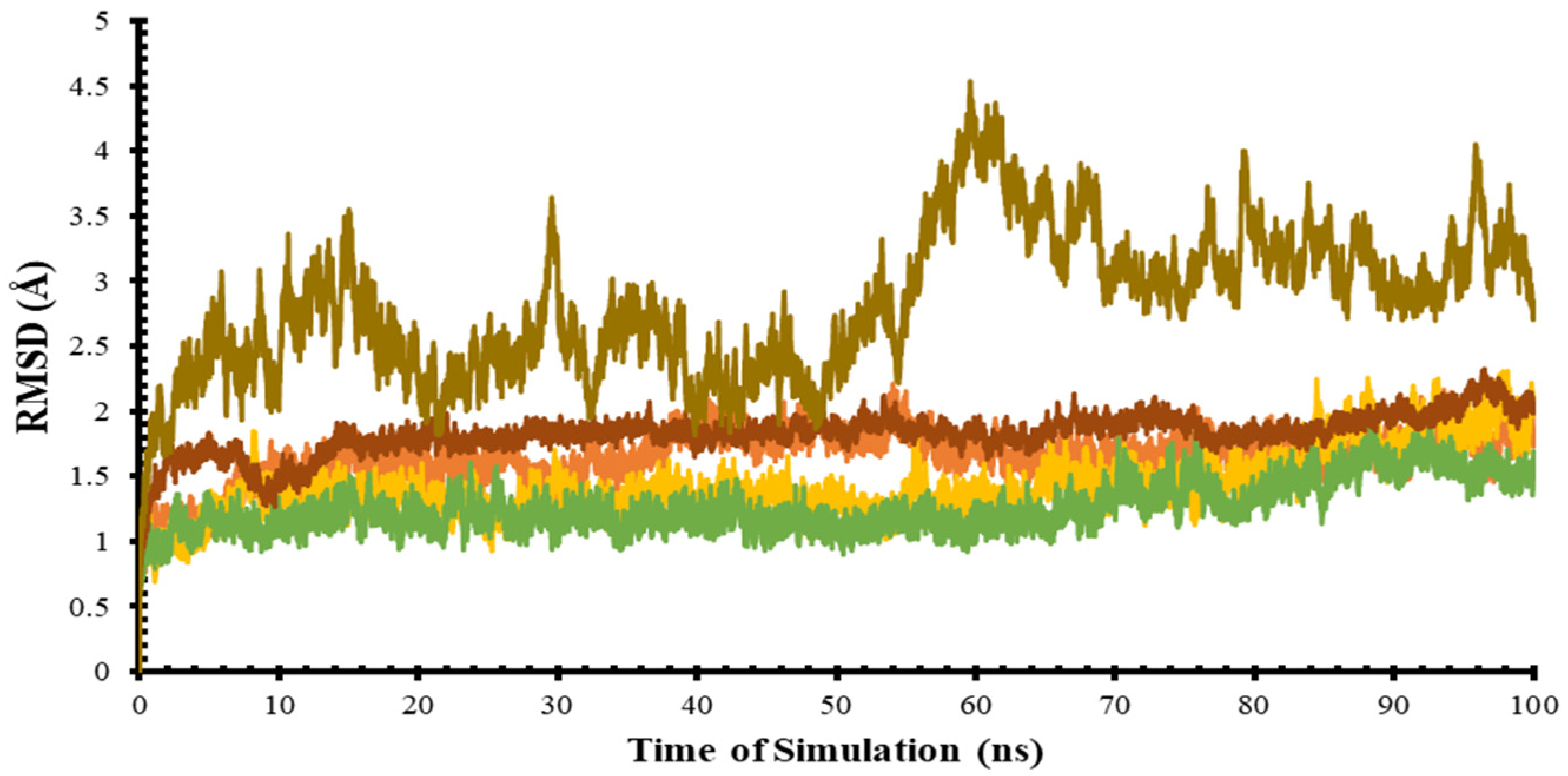
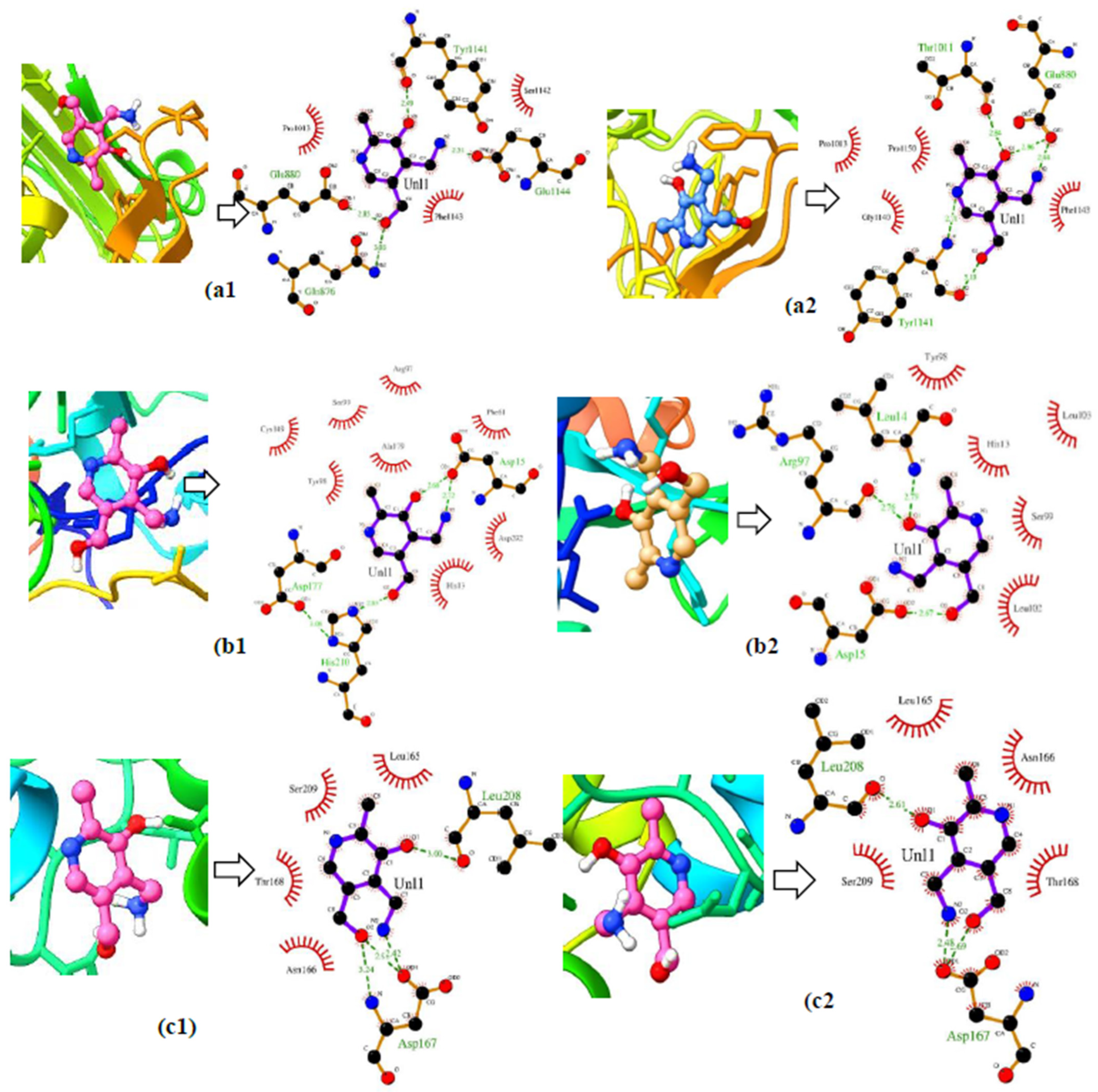
| Protein | Minimum and Maximum (Å) | Difference in Distance (Å) |
|---|---|---|
| Xanthine oxidase (XO) | 0.48–2.30 | 1.82 |
| Adenine deaminase (ADA) | 0.45–2.00 | 1.55 |
| Guanine deaminase (GDA) | 0.48–2.35 | 1.87 |
| Purine nucleoside phosphorylase (PNP) | 0.48–2.25 | 1.77 |
| 5-Nucleotidase II (NT5C2) | 0.70–4.50 | 3.80 |
| Name | Binding Energy (kcal/mol) | |||||
|---|---|---|---|---|---|---|
| XO | ADA | GDA | PNP | NT5C2-1497 | NT5C2-1498 | |
| Diallyl thiosulfinate (Allicin) | −4.74 ± 0.52 | −4.43 ± 0.38 | −4.46 ± 0.15 | −4.28 ± 0.23 | −3.94 ± 0.24 | −3.66 ± 0.18 |
| Alliin | −4.15 ± 0.38 | −4.72 ± 0.47 | −4.43 ± 0.24 | −4.81 ± 0.26 | −3.61 ± 0.36 | −3.60 ± 0.47 |
| S-allyl-L-cysteine | −4.04 ± 0.39 | −4.51 ± 0.29 | −4.12 ± 0.16 | −4.54 ± 0.20 | −3.72 ± 0.20 | −3.54 ± 0.27 |
| Diallyl disulfide | −2.80 ± 0.11 | −3.19 ± 0.33 | −3.39 ± 0.13 | −2.94 ± 0.22 | −3.27 ± 0.20 | −3.05 ± 0.06 |
| -Glutamyl-S-allyl-cysteine | −2.99 ± 0.42 | −4.10 ± 0. 48 | −2.94 ± 0.62 | −2.30 ± 0.57 | −3.47 ± 0.27 | −3.76 ± 0.72 |
| N-acetyl-S-allyl-L-cysteine | −3.85 ± 0.23 | −3.56 ± 0. 34 | −3.80 ± 0.41 | −4.36 ± 0.33 | −4.36 ± 0.28 | −4.25 ± 0.31 |
| (E)-Ajoene | −4.91 ± 0.12 | −5.32 ± 0. 24 | −4.94 ± 0.22 | −5.16 ± 0.28 | −4.58 ± 0.26 | −4.48 ± 0.28 |
| Pyridoxal | −5.01 ± 0.53 | −5.12 ± 0. 51 | −4.90 ± 0.27 | −4.71 ± 0.15 | −4.36 ± 0.26 | −4.64 ± 0.16 |
| Pyridoxamine | −5.26 ± 0.24 | −6.78 ± 0. 29 | −5.53 ± 0.29 | −6.06 ± 0.15 | −4.66 ± 0.30 | −4.68 ± 0.41 |
| DL-Carnitine | −2.39 ± 0.23 | −3.17 ± 0.30 | −2.52 ± 0.33 | −2.65 ± 0.30 | −3.44 ± 0.21 | −3.48 ± 0.44 |
| 4-Guanidinobutyric acid | −4.65 ± 0.43 | −3.60 ± 0.52 | −3.66 ± 0.21 | −4.58 ± 0.36 | −4.79 ± 0.47 | −4.17 ± 0.49 |
| D-Glucosamine | −5.23 ± 0.34 | −6.03 ± 0.39 | −5.27 ± 0.27 | −5.83 ± 0.23 | −4.03 ± 0.53 | −4.67 ± 0.35 |
| 5-hydroxymethyl-2-furaldehyde | −3.56 ± 0.29 | −3.70 ± 0.26 | −3.83 ± 0.20 | −3.92 ± 0.19 | −3.72 ± 0.23 | −3.34 ± 0.21 |
| Allopurinol (commercial) Xanthine (substrate) Hipoxanthine (substrate) | −4.82 ± 0.25 | |||||
| −4.92 ± 0.36 | ||||||
| −4.72 ± 0.38 | ||||||
| Erythro-9-(2-hydroxy-3-nonyl)adenine [EHNA] (commercial) Adenosine (substrate) | −4.73 ± 0.42 | |||||
| −4.52 ± 0.58 | ||||||
| Azepinomycin (commercial) Guanine (substrate) | −4.86 ± 0.28 | |||||
| −5.27 ± 0.16 | ||||||
| Ulodesine (commercial) | −6.03 ± 0.68 | |||||
| Guanosine (substrate) | −5.78 ± 0.67 | |||||
| Inosine (substrate) | −4.84 ± 0.40 | |||||
| Xanthosine (substrate) | −5.63 ± 0.55 | |||||
| Fludarabine (commercial) Adenosine monophosphate (substrate) Guanosine monophosphate (substrate) Inosine monophosphate (substrate) Xanthosine monophosphate (substrate) | −4.20 ± 0.52 | −4.04 ± 0.49 | ||||
| −4.18 ± 0.37 | −4.67 ± 0.33 | |||||
| −3.44 ± 0.36 | −4.67 ± 1,13 | |||||
| −4.73 ± 0.47 | −4.73 ± 0.80 | |||||
| −4.66 ± 0.51 | −4.63 ± 0.45 | |||||
| Ligand | Physicochemical Parameters | ADMET Parameters | |||||||
|---|---|---|---|---|---|---|---|---|---|
| MW a (<500 g mol−1) | LogP b (<5) | HBD c (<5) | HBA d (<10) | Bioavaibility (Score) | Human Intestinal Absorption (HIA) | AMES Mutagenesis | Carciogenicity | LD50 (mg/kg) (Predicted Toxicity Class) | |
| Allopurinol | 136.11 | −0.19 | 2 | 4 | GB (0.55) | HIA (+) | AMES (−) | NC | 1000 (IV) |
| Ulodesine | 264.28 | −0.80 | 4 | 6 | GB (0.55) | HIA (+) | AMES (−) | NC | 1000 (IV) |
| Erythro-9-(2-Hydroxy-3-nonyl)adenin [EHNA] | 277.37 | 2.30 | 3 | 5 | GB (0.55) | HIA (+) | AMES (−) | NC | 450 (IV) |
| Azepinomycin | 168.16 | −1.12 | 4 | 5 | GB (0.55) | HIA (+) | AMES (−) | NC | 2032 (V) |
| Fludarabine | 285.24 | −2.84 | 4 | 9 | GB (0.55) | HIA (+) | AMES (−) | NC | 13 (II) |
| Alliin | 177.22 | −0.67 | 2 | 3 | GB (0.55) | HIA (+) | AMES (−) | NC | 8000 (VI) |
| N-Acetyl-S-Allyl-L-Cysteine | 203.26 | 0.49 | 2 | 3 | GB (0.56) | HIA (+) | AMES (−) | NC | 4000 (V) |
| (E)-Ajoene | 234.41 | 3.00 | 0 | 3 | GB (0.55) | HIA (+) | AMES (−) | NC | 1600 (IV) |
| Pyridoxal | 167.16 | 0.40 | 2 | 13 | GB (0.55) | HIA (+) | AMES (−) | NC | 1120 (IV) |
| Pyridoxamine | 168.20 | 0.05 | 3 | 4 | GB (0.55) | HIA (+) | AMES (−) | NC | 5100 (VI) |
| 4-Guanidinobutyric acid | 145.16 | −0.88 | 3 | 2 | GB (0.55) | HIA (+) | AMES (−) | NC | 12,680 (VI) |
| D-Glucosamine | 179.17 | −2.55 | 5 | 19 | GB (0.55) | HIA (+) | AMES (−) | NC | 1000 (IV) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lestari, A.R.; Batubara, I.; Wahyudi, S.T.; Ilmiawati, A.; Achmadi, S.S. Bioactive Compounds in Garlic (Allium sativum) and Black Garlic as Antigout Agents, Using Computer Simulation. Life 2022, 12, 1131. https://doi.org/10.3390/life12081131
Lestari AR, Batubara I, Wahyudi ST, Ilmiawati A, Achmadi SS. Bioactive Compounds in Garlic (Allium sativum) and Black Garlic as Antigout Agents, Using Computer Simulation. Life. 2022; 12(8):1131. https://doi.org/10.3390/life12081131
Chicago/Turabian StyleLestari, Ayu Rahmania, Irmanida Batubara, Setyanto Tri Wahyudi, Auliya Ilmiawati, and Suminar Setiati Achmadi. 2022. "Bioactive Compounds in Garlic (Allium sativum) and Black Garlic as Antigout Agents, Using Computer Simulation" Life 12, no. 8: 1131. https://doi.org/10.3390/life12081131
APA StyleLestari, A. R., Batubara, I., Wahyudi, S. T., Ilmiawati, A., & Achmadi, S. S. (2022). Bioactive Compounds in Garlic (Allium sativum) and Black Garlic as Antigout Agents, Using Computer Simulation. Life, 12(8), 1131. https://doi.org/10.3390/life12081131






