Abstract
The associations of fetal fatty acids status to immune-related health parameters later in life are unclear. Our aim is to collect all available information on the relationship between fatty acid status at birth and allergy in childhood. Systematic literature search was performed on Ovid MEDLINE, Cochrane Library, and Embase. The search retrieved 897 articles without duplicates; 14 articles remained after excluding those that did not fit into our inclusion criteria. When the dichotomous parameter of suffering or not from allergic condition in childhood was analyzed, cord blood eicosapentaenoic acid (EPA) values proved to be significantly lower in allergic than non-allergic children in four comparisons from three studies. When the linear parameters of odds ratios and relative risks for allergy were taken into consideration, high cord blood EPA, but also high docosahexaenoic acid (DHA) and high total n-3 long-chain polyunsaturated fatty acid values were associated to clinically relevant reduction (at least 38%) in eight comparisons from five studies. Within the cord blood samples, higher EPA, docosapentaenoic acid, and DHA values were significantly and negatively associated in eight correlation analyses from three studies with laboratory parameters considered to reflect allergic trait. The data reported here may provide information for defining optimal fatty acid intakes for pregnant women.
1. Introduction
The most important long-chain polyunsaturated fatty acids (LCPUFAs), docosahexaenoic acid (C22:6n-3, DHA) and arachidonic acid (C20:4-6, AA), are bioconversion products from the essential fatty acids (EFAs), alpha-linolenic acid (C18:3n-3, ALA) and linoleic acid (C18:2n-6, LA), respectively. Although the efficiency of the conversion of EFAs to LCPUFAs is questioned throughout the human lifespan [1], it can be assumed with good reason that the perinatal period of rapid human growth and development represents the most critical stage. The need of LCPUFA supply to the human fetus was emphatically put forward as early as nearly half a century ago [2], and five decades of ongoing research yielded extensive data in this field.
Overall, 70 randomized controlled trials (RCTs) involving 19,927 pregnant women and investigating the addition of n-3 LCPUFAs either as supplements or as foods to the diet were summarized in a relatively recent Cochrane review [3]. Both preterm birth [< 37 weeks; relative risk (RR): 0.89; 95% confidence interval (95% CI): 0.81 to 0.97; 10,340 participants] and early preterm birth [<34 weeks; RR: 0.58; 95% CI: 0.44 to 0.77; 5204 participants] were significantly lower in women who received n-3 LCPUFA compared with no n-3 fatty acids. There was also a possibly reduced risk of perinatal death (RR: 0.75; 95% CI: 0.54 to 1.03; 7416 participants) and possibly fewer neonatal care admissions (RR: 0.92; 95% CI: 0.83 to 1.03; 6921 participants). In parallel with reduction in preterm birth and early preterm birth, prolonged gestation (>42 weeks) increased in women who received n-3 LCPUFA compared with no n-3 fatty acids (RR: 1.61; 95% CI: 1.11 to 2.33; 5141 participants). Important conclusions of this Cochrane review were that: (a) more studies comparing n-3 LCPUFA and placebo to establish causality in relation to preterm birth are not needed, and (b) further trials are needed to improve understanding of underlying mechanisms [3].
Development of the immune system is one of the major mechanisms influencing fetal wellbeing and pregnancy outcome, and patterns of LCPUFA exposure in pregnancy was reported to influence the fetal immune system. Decreased consumption of n-6 EFA and LCPUFAs in favor of more anti-inflammatory n-3 EFA and LCPUFAs in modern diets, has demonstrated the potential protective role of n-3 fatty acids in allergic and respiratory diseases [4,5,6]. Moreover, n-3 EFA and LCPUFAs may alter the T helper cell balance by inhibiting cytokine production and may further modify cellular membrane and induce eicosanoid metabolism and later gene expression [4,7,8].
Human studies showed that higher levels of n-3 LCPUFA were associated with reduction in neonatal oxidative stress, reduced production of inflammatory leukotriene B4, and altered T-cell function [9,10]. Inverse correlations between n-3 LCPUFA levels and neonatal T-cell cytokine production were also reported, consistent with data of adult studies showing reduction of T-cell cytokine production with fish oil supplementation [9,11]. Moreover, intrauterine LCPUFA supply may be associated with beneficial immunological consequences also after birth. Studies on supplemental n-3 LCPUFAs in pregnant women demonstrated reduced sensitization of infants to egg, reduced risk and severity of atopic dermatitis in the first year of life, and reduced persistent wheeze and asthma at ages of three to five years [12]. These observations indicate that LCPUFAs play a clinically significant role in immune development.
Despite encouraging data of the beneficial effects of LCPUFA supplementation to pregnant women on long-term immunity in the offspring, the exact associations of fetal fatty acids status to immune-related health parameters later in life are yet to be determined. Therefore, we decided to carry out a systematic review on objectively assessed neonatal fatty acids status at birth and occurrence of allergic diseases and clinical signs of atopy in early childhood.
2. Materials and Methods
This systematic review was registered prospectively in PROSPERO, under: CRD42021281397. The methodology and the results are reported according to the PRISMA (Preferred Reporting Items for Systematic Reviews and Meta-Analyses) guidelines for systematic reviews [13,14] (Table S1), as well as the guideline of the Cochrane Handbook of Systematic Reviews and Intervention [15].
2.1. Eligibility Criteria
We included studies on biological samples, allowing evaluation of LCPUFA status of healthy term newborns at birth (cord blood lipids, cord vessels wall lipids); whereas fatty acid studies on maternal biological samples obtained at delivery, on human milk (colostrum), or those on biological samples obtained from the offspring after birth, as well as data of not healthy infants (including preterm babies) were excluded. We included studies on fatty acid status but excluded studies on other blood lipids (e.g., HDL, LDL cholesterol, and triglyceride). Studies on incidence and prevalence of allergies, clinical signs of atopy, or other immune-related soluble factors in childhood were included, but inflammation in general or inflammatory factors were excluded. No restriction on clinical study type (observational or RCT) was applied; we excluded reviews, editorials, and comments not publishing original data. To sum up, we included studies reporting on fatty acid status in healthy newborns at birth in correlation with allergy-related data obtained in infancy or childhood.
2.2. Literature Search
Electronic literature search was performed on the following databases from the inception of each database up to September 2021: Embase, Cochrane Central Register of Controlled Trials (CENTRAL), and Ovid MEDLINE. No language restriction was applied. The search strategy was developed with terms related to newborns, allergy, and fatty acids. The search strategy on Ovid MEDLINE was as follows: (cord.mp OR newborn.mp OR infant.mp OR perinatal.mp OR postnatal.mp) AND (immune.mp OR immune*.mp OR allergy.mp OR allerg*.mp OR atopy.mp OR atopic.mp OR inflammation.mp OR infection.mp) AND (arachidonic.mp OR arachidonic acid.mp OR docosahexaenoic.mp OR docosahexenoic.mp OR docosahexaenoic acid.mp). The detailed search strategy for Ovid MEDLINE is available in List S1.
We manually searched the references of the included articles and related reviews for potentially relevant articles. We also searched grey literature for congress abstracts that might be relevant. All citations were then combined, and duplicates were excluded.
2.3. Study Selection, Risk of Bias Assessment
Pairs of review authors (B.B., E.M., É.S., T.M.) independently screened the abstract, title, or both of every record to determine potentially relevant articles. The abstract screening was performed on an online program (http://abstrackr.cebm.brown.edu; accessed on 15 November 2021) [16]. Then, the pairs of reviewers screened the full-text articles for inclusion and exclusion criteria independently on Rayyan.ai [17]. Disagreements between reviewers were resolved by further discussion until consensus. Risk of bias was also assessed independently by two authors (É.S., T.M.) according to the Cochrane Risk of Bias 2 (RoB2) tool for the RCTs [18], while the cohort studies and case-control studies were assessed using the ROBINS-I tool [19].
2.4. Data Extraction and Synthesis
One author (É.S) extracted data from the included articles, while the other author (T.M.) checked the data for accuracy and completeness. From full-text publications, the following data were extracted: first author, journal, year of publication, study type, place of study, type of sample, and reported outcomes. If the study published outcomes for multiple time points, we extracted data for each time point.
3. Results
After removing duplicates, the search resulted in 897 articles (Figure 1). After title/abstract screening, most articles were excluded; full text screening was evaluated on 70 articles. From these articles, eight were duplicates of another publication with the same population and same time point [20,21,22,23,24,25,26] and nine were excluded because they were reviews [27,28,29,30,31] or in vitro cell studies (cultured cells from cord blood) [32,33,34,35]. Furthermore, 27 articles were excluded because they investigated wrong populations; in many studies, infants were supplemented [36,37,38,39,40,41,42,43,44,45,46,47], while in others, blood was drawn later than at birth [48,49,50,51,52,53,54,55,56,57]; one study investigated placental lipid fatty acid composition [58], in two other studies no blood was drawn from the child [59,60], and in two further studies breast milk fatty acid composition was studied [61,62]. In nine studies, the outcome was not within our inclusion criteria (there was no direct link between fatty acids and immune-related factors [63,64,65,66,67,68,69,70], or inflammatory markers were only studied [71]). We also found two conference abstracts [72,73] and a trial protocol [74] where results have been not published in full article yet. Finally, 14 articles were included in the analysis (Table 1).
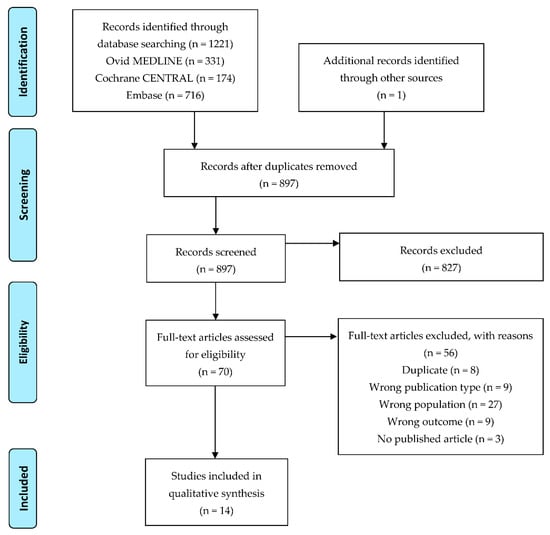
Figure 1.
Flow diagram of study selection.
3.1. Description of Included Studies
Most of the included studies were either RCTs (n = 6) [75,76,77,78,79,80] or birth cohort studies (n = 5) [81,82,83,84,85]. Three studies were published in two different articles [75,78,79,80,81,82], in that either the parameters analyzed in cord plasma [78,79] or the follow-up points [75,80,81,82] were different. For the purposes of the present review, we considered these related articles as independent publications.
Six studies reported cord blood fatty acid compositional data after maternal fish oil or n-3 LCPUFA supplementation during pregnancy, while four studies reported cord blood fatty acid data according to the appearance of allergy/atopy later on. Various cord blood constituents were used to characterize fatty acid status at birth: total plasma in four studies alone [78,79,84,86] and in one study in combination with umbilical artery and vein wall lipids [83], total serum in one study [87], plasma or serum phospholipids in five studies [76,77,81,82,88], and red blood cell (RBC) membrane lipids in three studies [75,80,85]. We provide detailed description of the type of lipid sample used, the method of fatty acid determination, and the list of fatty acids published in the given study in Table S2. Different surrogate parameters of allergy in the offspring were evaluated at birth in five studies [75,78,79,80]; we give description of the putative usefulness of these surrogate parameters in Table S3. Direct clinical signs and symptoms for allergy were estimated at no less than 11 different ages from 6 months [84] to 8 years [82].
When we compared the fatty acids identified and reported by the different studies (Table S2), we found that most of the articles presented fatty acid data as a percentage of total fatty acids by weight [76,79,80,81,82,83,84,85,87,88]. Only one study presented it as μg/106 cells [75], one as μmol/L [86], and one as mol% [77]. As for fatty acid determination, most research groups used the generally accepted capillary gas chromatographic (GC) determination [75,76,80,83,87], mostly with flame-ionization detector (FID) [77,81,84,85,86], and only a few determined fatty acids by mass spectrometry (MS) [79,82,88].
Three articles published no information about the family history of allergies [78,79,81], in two articles some newborns had at least one allergic parent [82,83], and in one article the number of newborns with a family history of maternal allergy was not described, but data were controlled for this confounding factor in the multivariate analysis [85]. Only two studies included healthy mothers with no allergic history [84,86], while in one article the included mothers were divided into two subgroups based on allergy [87]. The other articles investigated newborns of allergic mothers [75,80] or high-risk newborns with a family history of allergic disease [76,77,88]. In the included studies, twelve had moderate risk of bias, while serious risk of bias was identified in two studies (Tables S4 and S5).


Table 1.
Characteristics of the included studies.
Table 1.
Characteristics of the included studies.
| First Author, Year of Publication | Study Type | Place of Study | Subgroup of Infants | Type of Sample | Investigated Immune-Related Diseases/Factors |
|---|---|---|---|---|---|
| Barden AE, 2004 [75] | RCT | Subiaco, Australia | Maternal fish oil or olive oil supplementation during pregnancy | Cord blood RBC | Cord plasma F2-isoprostanes |
| Barman M, 2019 [81] | Birth cohort study | Sweden | Mothers living on a farm/not | Cord blood serum PL | Allergy at 18 and 36 months |
| Barman M, 2020 [82] | Birth cohort study | Sweden | Mothers living on a farm/not | Cord blood serum PL | Allergy at 1, 3, 5, and 8 years |
| Best KP, 2018 [76] | RCT | Australia | Maternal n-3 LCPUFA or placebo supplementation | Cord blood plasma PL | Allergic symptoms at 1, 3, and 6 years |
| Byberg K, 2008 [86] | Nested case-control study | Stavanger, Norway | Atopic/non-atopic | Cord blood plasma | Atopy, sCD23, and IgE at 3 years |
| Dirix CEH, 2009 [83] | Birth cohort study | Maastricht, The Netherlands | No | Cord blood plasma PL; umbilical artery and vein walls PL | Immune-related measurements at 7 years |
| Furuhjelm C, 2011 [77] | RCT | Sweden | Maternal n-3 LCPUFA or placebo supplementation | Cord blood plasma PL | Allergic symptoms up to 2 years |
| Galli E, 1994 [88] | Cohort study | Rome, Italy | Atopic/non-atopic | Cord blood serum PL | Atopy in the first 12 months |
| Montes R, 2013 [84] | Birth cohort study | Sabadell, Spain | Atopic/non-atopic | Cord blood plasma | Atopic eczema at 6 and 14 months |
| Mozurkewich EL, 2016 [78] | RCT | Michigan, USA | Maternal EPA/DHA/placebo supplementation | Cord blood plasma | Specialized pro-resolving mediators in cord plasma |
| Mozurkewich EL, 2018 [79] | RCT | Michigan, USA | Maternal EPA/DHA/placebo supplementation | Cord blood plasma | Cytokines in cord plasma |
| Newson RB, 2004 [85] | Birth cohort study | UK | No | Cord blood RBC PL | Wheezing and eczema at 18–30 and 30–42 months |
| See VHL, 2017 [80] | RCT | Subiaco, Australia | Maternal fish oil or olive oil supplementation during pregnancy | Cord blood RBC | Pro-resolving mediators at birth and 12 years |
| Yu G, 1996 [87] | Case-control study | Linköping, Sweden | Allergy: yes/no | Cord blood serum PL | Allergy during the first 6 years |
DHA: docosahexaenoic acid, EPA: eicosapentaenoic acid, LCPUFA: long-chain polyunsaturated fatty acid, PL: phospholipids, RBC: red blood cells, RCT: randomized controlled trial.
3.2. Fatty Acid Status at Birth and Development of Allergy in Childhood
Fatty acid status at birth was compared between children who developed and did not develop allergy in seven studies [77,81,82,84,86,87,88](Table 2). EPA values at birth were significantly lower in children who developed allergy in three studies [77,82,86] that addressed the ages of birth to 2 years [77], 3 years in two studies [82,86], and 8 years [82]. Significantly higher AA/EPA ratio was reported for the ages of birth to 2 years in children who developed allergy in one study [77]. In apparent contrast, significantly lower dihomo-gamma-linolenic acid (C20:3-n-6) and AA values were reported in one study [88] for children who were diagnosed with allergy at the age of 12 months; however, neither EPA nor DHA values were reported in this study.

Table 2.
Comparison of fatty acid status at birth between allergic and non-allergic children.
3.3. Relationship of Fatty Acid Status at Birth to Odds Ratios and Relative Risks of Allergy in Children
The wide variety of parameters characterizing fatty acid status and the widely different time points of evaluating allergy rendered it impossible to carry out formal meta-analysis within the present systematic review. However, odds ratios (ORs) or RRs of allergy reported in the studies reviewed allow some comparative mathematical representation of the results (Table 3). Altogether, 10 significantly different ORs or RRs for allergy were reported from five studies [76,82,84,85,86]. In four studies including altogether 1042 participants, n-3 LCPUFA supplementation during pregnancy [76] as well as high DHA or n-3 LCPUFA [84] or high eicosapentaenoic acid (EPA, C20:5n-3) in cord plasma [82,86] were associated with significantly reduced RRs or ORs of various clinical manifestations of allergy (Table 3). The extent of the reduction in ORs or RRs for allergy in favor of n-3 fatty acids was at least one third (highest OR or RR reported: 0.62). In a sub-study with 1191 participants of the Avon Longitudinal Study of Parents and Children [85], high RBC AA/EPA ratios were associated with enhanced OR of eczema at the age of 18 to 30 months, high AA/ALA ratios with enhanced OR of wheezing at the age of 30 to 42 months, whereas high ALA/n-3 LCPUFA ratios were marginally, but significantly associated with decreased OR of wheezing (Table 3).

Table 3.
Significantly different odds ratios/relative risks in the included studies.
3.4. Correlation between Fatty Acids and Allergy-Related Laboratory Parameters at Birth
Statistically significant correlations were reported between cord blood fatty acid values and some laboratory parameters considered by the authors of the given papers to be immunologically relevant (F2-isoprostanes, soluble CD23 receptors, interleukin (IL) 1β, 4-hydroxy-DHA, 14-hydroxy-DHA, 17-hydroxy-DHA, and 18-hydroxy-EPA) in five studies [75,78,79,80,86] (Table 4). In a study on 83 pregnant atopic women, cord red blood cell (RBC) EPA values were significantly and inversely associated to both cord blood plasma and urinary F2-isoprostanes, whereas cord blood RBC DHA values were significantly and inversely correlated with urinary F2-isoprostanes only [75]. Cord blood plasma EPA, docosapentaenoic acid (DPA), DHA, and total n-3 PUFA values were significantly and negatively correlated with soluble CD23 receptor levels in another study investigating 35 children who subsequently developed allergic sensitization and atopic dermatitis before the age of 3 years [86].

Table 4.
Significant correlations between fatty acids and allergy-related laboratory parameters at birth.
Cord blood plasma DHA values were significantly and inversely correlated to IL 1β concentrations in a study on 118 women participating in a n-3 LCPUFA supplementation trial [79]. In contrast, pooled maternal and cord plasma DHA values significantly and positively correlated with 4-hydroxy-DHA, 14-hydroxy-DHA, 17-hydroxy-DHA, and 18-hydroxy-EPA in a n-3 LCPUFA supplementation study in 60 pregnant women [78], whereas cord RBC EPA values significantly and positively correlated with 18-hydroxy-EPA values in another supplementation study including 83 participants [80] (Table 4).
4. Discussion
Recent developments in infant nutrition put further emphasis on the question of the role of LCPUFA supply to the fetus. Both a Cochrane Database Systematic Review on LCPUFA supplementation trials in infancy [89] and a systematic review and meta-analysis of fatty acid compositional data of human milk in various populations [90] substantially underpinned the importance of preformed DHA in infant nutrition. Today, DHA is a mandatory constituent of infant formula within the European Union [91], and mandatory inclusion of also AA is under vivid discussion in the medical literature [92,93]. If the diet of infants must contain preformed LCPUFA, it is logical to consider recommending for pregnant women the intake of certain amounts of certain type(s) of EFAs and/or LCPUFAs. Before embarking on recommendations, however, it is essential to better understand the potentially different effects of different LCPUFAs on various aspects of fetal development.
The major finding of the present review is the clear preventive relation between higher n-3 LCPUFA status at birth and the occurrence of allergy in childhood. When the dichotomous parameter of suffering or not from allergic condition in childhood was analyzed, EPA proved to be distinctive fatty acid in cord blood lipids. When the linear parameters of ORs and RRs were taken into consideration, besides EPA, DHA and total n-3 LCPUFA values were also significantly associated to clinically relevant reduction (more than 33%) in allergy. At birth, i.e., within the cord blood sample itself, higher EPA, docosapentaenoic acid, and DHA values were significantly and negatively associated with various laboratory parameters considered to reflect allergic trait.
Although it is generally accepted that DHA is the paramount LCPUFA in infant nutrition, based on the data obtained in the present systematic review it might be tempting to speculate that for the nutrition of the fetus, EPA may play an equally important role, at least as far as the intrauterine programming of the immune system is concerned. EPA will be converted by the cyclo-oxygenase and lipoxygenase enzymes into three and five series of prostaglandins and leukotrienes, which have less inflammatory effects than the two and four series of eicosanoids synthesized by the same enzymes from AA [94]. Furthermore, EPA serves as precursor for resolvins and maresins with anti-inflammatory effects [95]. However, data from animal studies indicate that tissue accretion of EPA is several folds lower than that of DHA or AA [96]. Therefore, isolated or unbalanced administration of EPA during pregnancy would not fit to the physiological fetal fatty acid accretion profiles. The data generated in the present systematic review cannot be directly translated into dietary advice but can serve as background for optimizing the design of further supplementation studies.
Among the seven studies comparing fatty acid status at birth between allergic and non-allergic children in our present systematic review, significantly lower contributions of EPA to cord blood lipids were reported in parallel with lack of difference in DHA values in three studies [77,82,86], whereas significant difference in DHA was not reported in any of the studies. However, the mostly observatory nature of the data reviewed here allows more to generate ideas than to support them.
It is to be noted that the appearance of allergic or atopic symptoms in the offspring may be differently related to LCPUFA status at birth in different groups of mothers. In the children of mothers with no allergy, LCPUFA supplementation in infancy significantly reduced the hazard ratios of any allergic diseases and skin allergic diseases, but failed to influence the hazard ratio of wheeze/asthma [41]. In the same study, in children of mothers with allergy, LCPUFA supplementation in infancy reduced the hazard ratio of wheeze/asthma but failed to influence the hazard for all allergic illnesses and skin allergic illness [41]. Moreover, population-specific associations were reported between blood parameters and asthma subtypes in a study involving altogether 3738 African American, Mexican American, and Puerto Rican children with median ages of 12 to 14 years [97].
Our present approach to try to differentiate between the effects of different n-3 LCPUFAs does not stand in the literature. Effects on n-3 LCPUFA supplementation either in the form of DHA or in the form of EPA were compared both in RCTs and in meta-analyses. In a randomized, crossover, head-to-head study in 154 healthy women and men with abdominal obesity and low-grade systematic inflammation, supplementation with DHA compared with supplementation with EPA resulted in significantly greater reduction in IL-18 values (DHA versus control: −18.15 ± 6.25 pmol/L, EPA versus control: −2.12 ± 6.29 pmol/L, mean ± SEM, DHA versus EPA: p = 0.01), whereas changes in C-reactive protein, IL-6, and tumor necrosis factor-α (TNF-α) were not significantly different [98]. The greater effect of DHA than that of EPA is in line with a meta-analysis showing that a major part of anti-inflammatory effects of marine-derived mixed n-3 LCPUFA can be attributed to DHA [99]. Gene expression analyses in 44 participants of the above-mentioned supplementation study showed no difference between EPA or DHA treatment on the expression of IL-10, IL-1β, and TNF-α genes [100]. Comparison in a 6-week trial of two different doses of EPA (600 mg/day or 1800 mg/day) with DHA (600 mg/day) in 121 healthy subjects showed no difference in IL-6, TNF-α, and vascular cell adhesion molecule 1 values among the three supplementation groups [101]. The anti-inflammatory effects of DHA and EPA were compared also in a recent pairwise and network meta-analyses of 5 and 20 RCTs including data from 411 and 1231 participants, respectively [102]. In both pairwise and network meta-analyses of these supplementation trials, EPA and DHA had similar effects on plasma C-reactive protein, IL-6, and TNF-α concentrations [102].
Growing interest and accumulating evidence in the immunological role of intrauterine LCPUFA status gave rise to systematic overviews of the effect of n-3 LCPUFA supplementation on childhood allergy as well [103,104]. Systematic review of five RCTs with altogether 949 participants showed that n-3 LCPUFA supplementation during pregnancy reduced 12-month prevalence of positive egg skin prick test (OR: 0.33; 95% CI: 0.16 to 0.70) and childhood asthma (OR: 0.35; 95% CI: 0.15 to 0.79), and significantly reduced cord blood IL-13 levels [103]. However, in two of the five studies reviewed by Klemens et al. [103], intrauterine LCPUFA supplementation was followed by similar supplementation during lactation, thus the findings reported cannot be attributed solely to the intervention during pregnancy. Five years later, another systematic review identified five prenatal and one prenatal/postnatal RCTs (including four out of the five in the review of Klemens et al. [103]) addressing the question of LCPUFA supplementation in pregnant women on allergy outcomes in their children [104]. N-3 LCPUFA (EPA and DHA) supplementation showed a clear reduction in medically diagnosed IgE mediated allergy in children aged 12 to 36 months (RR: 0.66; 95% CI: 0.44 to 0.98; 2 RCTs; 823 participants), but not beyond 36 months, or if allergy diagnoses on the basis of parental reports were included [104]. Seven RCTs involving 2047 children were included into a more recent systematic review and meta-analysis on the effects of n-3 LCPUFA supplementation during pregnancy on asthma or wheeze of children [105]. N-3 LCPUFA supplementation reduced significantly the incidence of wheeze/asthma (RR: 0.81; 95% CI: 0.66 to 0.99), while the incidence of childhood asthma was not significantly reduced (RR: 0.89; 95% CI: 0.67 to 1.17) [105].The above-mentioned systematic reviews [103,104] were aimed to obtain data on the clinical efficacy of n-3 LCPUFA supplementation during pregnancy, thus they included RCTs only. In the present systematic review, we addressed the question of the relationship of cord blood fatty acid status at birth (as surrogate cumulative parameter of fetal fatty acid status) to the development of allergy in childhood, thus we included all types of studies. Furthermore, characterization of LCPUFA status at birth was among the eligibility criteria in the present study, whereas it was no prerequisite in the previous systematic reviews. Consequently, from the 14 studies included into the present review, only one [77] was included into the previous reviews.
The present systematic review has various limitations which usually accompany studies relating fatty acid status to clinical diagnoses and/or to surrogate biochemical parameters of clinical outcomes. First, chromatographic methods of fatty acid determination are far from being standardized; there are differences in the type of sample analyzed, in the palette of the fatty acids reported, and in the way the results are expressed. Second, identical diagnostic expressions, such as “allergy” or “atopy”, might cover slightly different meanings in different studies. Third, laboratory parameters considered immune-related or allergy-related by the authors of the studies reviewed here may characterize fairly different degrees as well as different aspects of the development of allergy. Although our supplementary tables showing detailed description of the methods of fatty acid analyses as well as the rationality behind using a given laboratory parameter as immune- or allergy-related may underpin the reliability of our conclusion, there remains some methodological uncertainties that should be considered at the evaluation of the data reported.
EFA and LCPUFA intake of pregnant women is a practical question. Existing evidence suggest that some enhancement of EFA and LCPUFA intake of pregnant women may beneficially influence some pregnancy outcomes. The findings of our present systematic review may provide useful information for defining optimal EFA and LCPUFA intakes for pregnant women.
Supplementary Materials
The following supporting information can be downloaded at: https://www.mdpi.com/article/10.3390/life12040526/s1, Table S1: Prisma 2020 Checklist. List S1: Search strategy for Ovid MEDLINE. Table S2: Determined fatty acids and methods of determination in the included studies. Table S3: Immune-related biochemical factors and their usefulness in allergic diseases. Table S4: Risk of bias assessment for included non-randomized (cohort and case-control). Table S5: Risk of bias assessment for included randomized controlled trials.
Author Contributions
Conceptualization, T.D.; methodology, É.S., T.M., B.B. and E.M.; software, É.S.; validation, T.M.; data curation, É.S. and T.M.; writing—original draft preparation, T.D. and É.S.; writing—review and editing, T.D. and É.S.; visualization, É.S.; supervision, T.D.; funding acquisition, T.D. and É.S. All authors have read and agreed to the published version of the manuscript.
Funding
This research was funded by the National Research, Development and Innovation Office (NKFIH) grant number K-120193 (T.D.), and the University of Pécs, Research Fund (KA-2021-07) grant number K-300954 (É.S.). The APC was funded by the University of Pécs.
Institutional Review Board Statement
Not applicable.
Informed Consent Statement
Not applicable.
Data Availability Statement
Not applicable.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Janssen, C.I.; Kiliaan, A.J. Long-Chain Polyunsaturated Fatty Acids (LCPUFA) from genesis to senescence: The influence of LCPUFA on neural development, aging, and neurodegeneration. Prog. Lipid Res. 2014, 53, 1–17. [Google Scholar] [CrossRef]
- Crawford, M.A.; Hassam, A.G.; Williams, G. Essential fatty acids and fetal brain growth. Lancet 1976, 1, 452–453. [Google Scholar] [CrossRef]
- Middleton, P.; Gomersall, J.C.; Gould, J.F.; Shepherd, E.; Olsen, S.F.; Makrides, M. Omega-3 fatty acid addition during pregnancy. Cochrane Database Syst. Rev. 2018, 11, CD003402. [Google Scholar] [CrossRef]
- Shek, L.P.; Chong, M.F.; Lim, J.Y.; Soh, S.E.; Chong, Y.S. Role of dietary long-chain polyunsaturated fatty acids in infant allergies and respiratory diseases. Clin. Dev. Immunol. 2012, 2012, 730568. [Google Scholar] [CrossRef]
- Hwang, I.; Cha, A.; Lee, H.; Yoon, H.; Yoon, T.; Cho, B.; Lee, S.; Park, Y. N-3 polyunsaturated fatty acids and atopy in Korean preschoolers. Lipids 2007, 42, 345–349. [Google Scholar] [CrossRef]
- Adams, S.; Lopata, A.L.; Smuts, C.M.; Baatjies, R.; Jeebhay, M.F. Relationship between serum omega-3 fatty acid and asthma endpoints. Int. J. Environ. Res. Public Health 2018, 16, 43. [Google Scholar] [CrossRef]
- Jaudszus, A.; Gruen, M.; Watzl, B.; Ness, C.; Roth, A.; Lochner, A.; Barz, D.; Gabriel, H.; Rothe, M.; Jahreis, G. Evaluation of suppressive and pro-resolving effects of EPA and DHA in human primary monocytes and T-helper cells. J. Lipid Res. 2013, 54, 923–935. [Google Scholar] [CrossRef]
- Lee, J.; Choi, Y.R.; Kim, M.; Park, J.M.; Kang, M.; Oh, J.; Lee, C.J.; Park, S.; Kang, S.M.; Manabe, I.; et al. Common and differential effects of docosahexaenoic acid and eicosapentaenoic acid on helper T-cell responses and associated pathways. BMB Rep. 2021, 54, 278–283. [Google Scholar] [CrossRef]
- Prescott, S.L.; Dunstan, J.A. Prenatal fatty acid status and immune development: The pathways and the evidence. Lipids 2007, 42, 801–810. [Google Scholar] [CrossRef]
- Bruschi, M.; Santucci, L.; Petretto, A.; Bartolocci, M.; Marchisio, M.; Ghiggeri, G.M.; Verrina, E.; Ramenghi, L.A.; Panfoli, I.; Candiano, G. Association between maternal omega-3 polyunsaturated fatty acids supplementation and preterm delivery: A proteomic study. FASEB J. 2020, 34, 6322–6334. [Google Scholar] [CrossRef]
- Trebble, T.M.; Wootton, S.A.; Miles, E.A.; Mullee, M.; Arden, N.K.; Ballinger, A.B.; Stroud, M.A.; Burdge, G.C.; Calder, P.C. Prostaglandin E2 production and T cell function after fish-oil supplementation: Response to antioxidant cosupplementation. Am. J. Clin. Nutr. 2003, 78, 376–382. [Google Scholar] [CrossRef] [PubMed]
- Miles, E.A.; Childs, C.E.; Calder, P.C. Long-Chain Polyunsaturated Fatty Acids (LCPUFAs) and the developing immune system: A narrative review. Nutrients 2021, 13, 247. [Google Scholar] [CrossRef] [PubMed]
- Moher, D.; Shamseer, L.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015 statement. Syst Rev. 2015, 4, 1. [Google Scholar] [CrossRef] [PubMed]
- Shamseer, L.; Moher, D.; Clarke, M.; Ghersi, D.; Liberati, A.; Petticrew, M.; Shekelle, P.; Stewart, L.A.; Group, P.-P. Preferred Reporting Items for Systematic Review and Meta-Analysis Protocols (PRISMA-P) 2015: Elaboration and explanation. BMJ 2015, 350, g7647. [Google Scholar] [CrossRef] [PubMed]
- Lefebvre, C.; Glanville, J.; Briscoe, S.; Littlewood, A.; Marshall, C.; Metzendorf, M.-I.; Noel-Storr, A.; Rader, T.; Shokraneh, F.; Thomas, J.; et al. Chapter 4: Searching for and selecting studies. In Cochrane Handbook for Systematic Reviews of Interventions Version 6.2 (Updated February 2021); Higgins, J., Thomas, J., Chandler, J., Cumpston, M., Li, T., Page, M., Welch, V., Eds.; Cochrane: London, UK, 2021; Available online: www.training.cochrane.org/handbook (accessed on 27 January 2022).
- Byron, C.W.; Small, K.; Brodley, C.E.; Lau, J.; Trikalinos, T.A. Deploying an interactive machine learning system in an evidence-based practice center: Abstrackr. In Proceedings of the IHI ‘12: ACM International Health Informatics Symposium, Miami, FL, USA, 28–30 January 2012; pp. 819–824. [Google Scholar]
- Ouzzani, M.; Hammady, H.; Fedorowicz, Z.; Elmagarmid, A. Rayyan-a web and mobile app for systematic reviews. Syst. Rev. 2016, 5, 210. [Google Scholar] [CrossRef]
- Sterne, J.A.C.; Savovic, J.; Page, M.J.; Elbers, R.G.; Blencowe, N.S.; Boutron, I.; Cates, C.J.; Cheng, H.Y.; Corbett, M.S.; Eldridge, S.M.; et al. RoB 2: A revised tool for assessing risk of bias in randomised trials. BMJ 2019, 366, l4898. [Google Scholar] [CrossRef]
- Sterne, J.A.; Hernan, M.A.; Reeves, B.C.; Savovic, J.; Berkman, N.D.; Viswanathan, M.; Henry, D.; Altman, D.G.; Ansari, M.T.; Boutron, I.; et al. ROBINS-I: A tool for assessing risk of bias in non-randomised studies of interventions. BMJ 2016, 355, i4919. [Google Scholar] [CrossRef]
- Chase, P.; Boulware, D.; Krischer, J.; Rodriguez, H.; Donaldson, D.; Chritton, S.; Muller, S.; Clare-Salzler, M. Inflammatory cytokines in the pre-autoimmune phase of type 1 diabetes. Diabetes 2010, 59, A502. [Google Scholar]
- Scalabrin, D.; Mitmesser, S.; Birch, E.; Khoury, J.; Bean, J.; Harris, C.; Berseth, C. Lower incidence and less recurrence of allergic manifestations is observed in children who received docosahexaenoic acid/arachidonic acid in infancy via breast milk or supplemented formula. Allergy 2011, 66, 711. [Google Scholar] [CrossRef]
- Nct. Nutritional Intervention to Prevent Diabetes. 2006. Available online: https://clinicaltrials.gov/show/NCT00333554 (accessed on 27 January 2022).
- Mozurkewich, E.L.; Clinton, C.M.; Romero, V.C.; Berman, D.R.; Barks, J.; Djuric, Z.; Dalli, J.; Serhan, C.N. Omega-3 fatty acid-derived pro-resolving lipid mediators in the human fetus: An exploratory secondary analysis of the mothers, omega-3, & mental health study. Reprod. Sci. 2013, 20, 297A. [Google Scholar]
- Mozurkewich, E.L.; Clinton, C.M.; Romero, V.C.; Tyner, J.; Brown, S.; Williams, J.Z.; Schrader, R.; Djuric, Z. Prenatal omega-3 fatty acid supplementation reduces pro-inflammatory cytokine production in umbilical cord blood: A secondary analysis of the mothers, omega-3, & mental health study. Reprod. Sci. 2013, 20, 248A. [Google Scholar]
- Barman, M.; Nilsson, S.; Torinsson Naluai, A.; Sandin, A.; Wold, A.; Sandberg, A.S. Single nucleotide polymorphisms in fatty acid desaturases is associated with cord blood long chain PUFA proportions and development of allergy. Allergy 2015, 70, 402. [Google Scholar] [CrossRef]
- Chase, P.; Boulware, D.; Krischer, J.; Daniels, M.; Nathan, B.; Chritton, S.; Muller, S.; Clare-Salzler, M. Vitamin D and inflammatory cytokine levels in infants at high genetic risk for type 1 diabetes. Diabetes 2010, 59, A467. [Google Scholar]
- Calder, P.C. Early life programming of immune and lung function: Can we now exclude a role of arachidonic acid exposure? Br. J. Nutr. 2009, 102, 331–333. [Google Scholar] [CrossRef][Green Version]
- Ciaccio, C.E.; Girdhar, M. Effect of maternal ω3 fatty acid supplementation on infant allergy. Ann. Allergy Asthma Immunol. 2014, 112, 191–194. [Google Scholar] [CrossRef]
- Gomez, R.; Romero, R.; Edwin, S.S.; David, C. Pathogenesis of preterm labor and preterm premature rupture of membranes associated with intraamniotic infection. Infect. Dis. Clin. North. Am. 1997, 11, 135–176. [Google Scholar] [CrossRef]
- Calder, P.C. Polyunsaturated fatty acids and cytokine profiles: A clue to the changing prevalence of atopy? Clin. Exp. Allergy 2003, 33, 412–415. [Google Scholar] [CrossRef]
- Guesry, P. The role of nutrition in brain development. Prev. Med. 1998, 27, 189–194. [Google Scholar] [CrossRef][Green Version]
- Loppi, M.; Businco, L.; Arcese, G.; Ziruolo, G.; Nisini, R. Cord blood mononuclear leukocytes of neonates at risk of atopy have a deficiency of arachidonic acid. J. Investig. Allergol. Clin. Immunol. 1994, 4, 272–276. [Google Scholar]
- Gold, D.R.; Willwerth, B.M.; Tantisira, K.G.; Finn, P.W.; Schaub, B.; Perkins, D.L.; Tzianabos, A.; Ly, N.P.; Schroeter, C.; Gibbons, F.; et al. Associations of cord blood fatty acids with lymphocyte proliferation, IL-13, and IFN-gamma. J. Allergy Clin. Immunol. 2006, 117, 931–938. [Google Scholar] [CrossRef][Green Version]
- Dunstan, J.A.; Mori, T.A.; Barden, A.; Beilin, L.J.; Taylor, A.L.; Holt, P.G.; Prescott, S.L. Fish oil supplementation in pregnancy modifies neonatal allergen-specific immune responses and clinical outcomes in infants at high risk of atopy: A randomized, controlled trial. J. Allergy Clin. Immunol. 2003, 112, 1178–1184. [Google Scholar] [CrossRef] [PubMed]
- Espiritu, M.M.; Lin, H.; Foley, E.; Tsang, V.; Rhee, E.; Perlman, J.; Cunningham-Rundles, S. Omega-3 fatty acids modulate neonatal cytokine response to endotoxin. J. Perinat. Med. 2016, 44, 711–721. [Google Scholar] [CrossRef] [PubMed]
- See, V.H.L.; Mas, E.; Prescott, S.L.; Beilin, L.J.; Burrows, S.; Barden, A.E.; Huang, R.C.; Mori, T.A. Effects of postnatal omega-3 fatty acid supplementation on offspring pro-resolving mediators of inflammation at 6 months and 5 years of age: A double blind, randomized controlled clinical trial. Prostaglandins Leukot Essent Fat. Acids 2017, 126, 126–132. [Google Scholar] [CrossRef] [PubMed]
- Nct. Assessment of Arachidonic Acid Supplementation in Infant Formula on the Immune Response of Infants. 2014. Available online: https://clinicaltrials.gov/show/NCT02092857 (accessed on 27 January 2022).
- D’Vaz, N.; Amarasekera, M.; Dunstan, J.; Meldrum, S.; Lee-Pullen, T.; Metcalfe, J.; Holt, B.; Serralha, M.; Tulic, M.; Mori, T.; et al. Basic and clinical immunology-3020. Fish oil supplementation in early infancy modulates developing infant immune responses but not clinical allergy. World Allergy Organ. J. 2013, 6, P196. [Google Scholar] [CrossRef][Green Version]
- Lucas, A.; Stafford, M.; Morley, R.; Abbott, R.; Stephenson, T.; MacFadyen, U.; Elias-Jones, A.; Clements, H. Efficacy and safety of long-chain polyunsaturated fatty acid supplementation of infant-formula milk: A randomised trial. Lancet 1999, 354, 1948–1954. [Google Scholar] [CrossRef]
- D’Vaz, N.; Meldrum, S.J.; Dunstan, J.A.; Lee-Pullen, T.F.; Metcalfe, J.; Holt, B.J.; Serralha, M.; Tulic, M.K.; Mori, T.A.; Prescott, S.L. Fish oil supplementation in early infancy modulates developing infant immune responses. Clin. Exp. Allergy 2012, 42, 1206–1216. [Google Scholar] [CrossRef]
- Foiles, A.M.; Kerling, E.H.; Wick, J.A.; Scalabrin, D.M.F.; Colombo, J.; Carlson, S.E. Formula with long-chain polyunsaturated fatty acids reduces incidence of allergy in early childhood. Pediatr. Allergy Immunol. 2016, 27, 156–161. [Google Scholar] [CrossRef]
- Scalabrin, D.M.; Birch, E.E.; Khoury, J.C.; Bean, J.; Harris, C.L.; Mitmesser, S.H.; Berseth, C.L. Impact of early nutrition on incidence of allergic manifestations in the first 3 years of life. J. Allergy Clin. Immunol. 2011, 127, AB33. [Google Scholar] [CrossRef]
- Lapillonne, A.; Pastor, N.; Zhuang, W.; Scalabrin, D.M.F. Infants fed formula with added long chain polyunsaturated fatty acids have reduced incidence of respiratory illnesses and diarrhea during the first year of life. BMC Pediatrics 2014, 14, 168. [Google Scholar] [CrossRef]
- Miklavcic, J.J.; Larsen, B.M.K.; Mazurak, V.C.; Scalabrin, D.M.F.; MacDonald, I.M.; Shoemaker, G.K.; Casey, L.; Van Aerde, J.E.; Clandinin, M.T. Reduction of arachidonate is associated with increase in B-cell activation marker in infants: A randomized trial. J. Pediatr. Gastroenterol. Nutr. 2017, 64, 446–453. [Google Scholar] [CrossRef]
- Gibson, R.A.; Barclay, D.; Marshall, H.; Moulin, J.; Maire, J.C.; Makrides, M. Safety of supplementing infant formula with long-chain polyunsaturated fatty acids and Bifidobacterium lactis in term infants: A randomised controlled trial. Br. J. Nutr. 2009, 101, 1706–1713. [Google Scholar] [CrossRef] [PubMed]
- Meldrum, S.J.; D’Vaz, N.; Dunstan, J.; Mori, T.A.; Prescott, S.L. The Infant Fish Oil Supplementation Study (IFOS): Design and research protocol of a double-blind, randomised controlled n-3 LCPUFA intervention trial in term infants. Contemp. Clin. Trials 2011, 32, 771–778. [Google Scholar] [CrossRef] [PubMed]
- Field, C.J.; Van Aerde, J.E.; Goruk, S.; Clandinin, M.T. Effect of feeding a formula supplemented with long-chain polyunsaturated fatty acids for 14 weeks improves the ex vivo response to a mitogen and reduces the response to a soy protein in infants at low risk for allergy. J. Pediatr. Gastroenterol. Nutr. 2010, 50, 661–669. [Google Scholar] [CrossRef] [PubMed]
- Laitinen, K.; Hoppu, U.; Hämäläinen, M.; Linderborg, K.; Moilanen, E.; Isolauri, E. Breast milk fatty acids may link innate and adaptive immune regulation: Analysis of soluble CD14, prostaglandin E2, and fatty acids. Pediatr. Res. 2006, 59, 723–727. [Google Scholar] [CrossRef][Green Version]
- Granot, E.; Golan, D.; Berry, E.M. Breast-fed and formula-fed infants do not differ in immunocompetent cell cytokine production despite differences in cell membrane fatty acid composition. Am. J. Clin. Nutr. 2000, 72, 1202–1205. [Google Scholar] [CrossRef]
- Lumia, M.; Luukkainen, P.; Takkinen, H.M.; Kaila, M.; Nwaru, B.I.; Nevalainen, J.; Salminen, I.; Uusitalo, L.; Niinistö, S.; Tuokkola, J.; et al. Cow’s milk allergy and the association between fatty acids and childhood asthma risk. J. Allergy Clin. Immunol. 2014, 134, 488–490. [Google Scholar] [CrossRef]
- Van Gool, C.J.A.W.; Thijs, C.; Dagnelie, P.C.; Henquet, C.J.M.; Van Houwelingen, A.C.; Schrander, J.; Menheere, P.P.C.A.; Van Den Brandt, P.A. Determinants of neonatal IgE level: Parity, maternal age, birth season and perinatal essential fatty acid status in infants of atopic mothers. Allergy 2004, 59, 961–968. [Google Scholar] [CrossRef]
- Granot, E.; Jakobovich, E.; Rabinowitz, R.; Levy, P.; Schlesinger, M. DHA supplementation during pregnancy and lactation affects infants’ cellular but not humoral immune response. Mediat. Inflamm. 2011, 2011, 493925. [Google Scholar] [CrossRef]
- Chase, H.P.; Boulware, D.; Rodriguez, H.; Donaldson, D.; Chritton, S.; Rafkin-Mervis, L.; Krischer, J.; Skyler, J.S.; Clare-Salzler, M.; Lescheck, E.; et al. Effect of docosahexaenoic acid supplementation on inflammatory cytokine levels in infants at high genetic risk for type 1 diabetes. Pediatr. Diabetes 2015, 16, 271–279. [Google Scholar] [CrossRef]
- Argaw, A.; Wondafrash, M.; Bouckaert, K.P.; Kolsteren, P.; Lachat, C.; Belachew, T.; De Meulenaer, B.; Huybregts, L. Effects of n-3 long-chain PUFA supplementation to lactating mothers and their breastfed children on child growth and morbidity: A 2 × 2 factorial randomized controlled trial in rural Ethiopia. Am. J. Clin. Nutr. 2018, 107, 454–464. [Google Scholar] [CrossRef]
- Niinistö, S.; Takkinen, H.M.; Erlund, I.; Ahonen, S.; Toppari, J.; Ilonen, J.; Veijola, R.; Knip, M.; Vaarala, O.; Virtanen, S.M. Fatty acid status in infancy is associated with the risk of type 1 diabetes-associated autoimmunity. Diabetologia 2017, 60, 1223–1233. [Google Scholar] [CrossRef] [PubMed]
- Ellul, S.; Marx, W.; Collier, F.; Saffery, R.; Tang, M.; Burgner, D.; Carlin, J.; Vuillermin, P.; Ponsonby, A.L. Plasma metabolomic profiles associated with infant food allergy with further consideration of other early life factors. Prostaglandins Leukot. Essent. Fat. Acids 2020, 159, 102099. [Google Scholar] [CrossRef] [PubMed]
- Jonsson, K.; Barman, M.; Moberg, S.; Sjöberg, A.; Brekke, H.K.; Hesselmar, B.; Sandberg, A.S.; Wold, A.E. Serum fatty acids in infants, reflecting family fish consumption, were inversely associated with allergy development but not related to farm residence. Acta Paediatr. 2016, 105, 1462–1471. [Google Scholar] [CrossRef] [PubMed]
- Keelan, J.A.; Mas, E.; D’Vaz, N.; Dunstan, J.A.; Li, S.; Barden, A.E.; Mark, P.J.; Waddell, B.J.; Prescott, S.L.; Mori, T.A. Effects of maternal n-3 fatty acid supplementation on placental cytokines, pro-resolving lipid mediators and their precursors. Reproduction 2015, 149, 171–178. [Google Scholar] [CrossRef]
- Furuhjelm, C.; Warstedt, K.; Larsson, J.; Fredriksson, M.; Böttcher, M.F.; Fälth-Magnusson, K.; Duchén, K. Fish oil supplementation in pregnancy and lactation may decrease the risk of infant allergy. Acta Paediatr. 2009, 98, 1461–1467. [Google Scholar] [CrossRef]
- Palmer, D.J.; Sullivan, T.; Gold, M.S.; Prescott, S.L.; Heddle, R.; Gibson, R.A.; Makrides, M. Randomized controlled trial of fish oil supplementation in pregnancy on childhood allergies. Allergy 2013, 68, 1370–1376. [Google Scholar] [CrossRef]
- Nct. DHA-1 for Lactating Mothers. 2012. Available online: https://clinicaltrials.gov/show/NCT01732874 (accessed on 28 January 2022).
- Kankaanpaa, P.; Nurmela, K.; Erkkila, A.; Kalliomaki, M.; Holmberg-Marttila, D.; Salminen, S.; Isolauri, E. Polyunsaturated fatty acids in maternal diet, breast milk, and serum lipid fatty acids of infants in relation to atopy. Allergy 2001, 56, 633–638. [Google Scholar] [CrossRef]
- Beck, M.; Zelczak, G.; Lentze, M.J. Abnormal fatty acid composition in umbilical cord blood of infants at high risk of atopic disease. Acta Paediatr. 2000, 89, 279–284. [Google Scholar] [CrossRef]
- Moodley, T.; Vella, C.; Djahanbakhch, O.; Branford-White, C.J.; Crawford, M.A. Arachidonic and docosahexaenoic acid deficits in Preterm neonatal mononuclear cell membranes. Implications for the immune response at birth. Nutr. Health 2009, 20, 167–185. [Google Scholar] [CrossRef]
- Yu, G.; Björkstén, B. Serum levels of phospholipid fatty acids in mothers and their babies in relation to allergic disease. Eur. J. Pediatr. 1998, 157, 298–303. [Google Scholar] [CrossRef]
- Barman, M.; Nilsson, S.; Naluai, Å.T.; Sandin, A.; Wold, A.E.; Sandberg, A.S. Single nucleotide polymorphisms in the FADS gene cluster but not the ELOVL2 gene are associated with serum polyunsaturated fatty acid composition and development of allergy (in a Swedish birth cohort). Nutrients 2015, 7, 10100–10115. [Google Scholar] [CrossRef] [PubMed]
- Furuhjelm, C.; Jenmalm, M.C.; Fälth-Magnusson, K.; DuchIn, K. Th1 and Th2 chemokines, vaccine-induced immunity, and allergic disease in infants after maternal ω-3 fatty acid supplementation during pregnancy and lactation. Pediatr. Res. 2011, 69, 259–264. [Google Scholar] [CrossRef] [PubMed]
- Krauss-Etschmann, S.; Hartl, D.; Rzehak, P.; Heinrich, J.; Shadid, R.; del Carmen Ramírez-Tortosa, M.; Campoy, C.; Pardillo, S.; Schendel, D.J.; Decsi, T.; et al. Decreased cord blood IL-4, IL-13, and CCR4 and increased TGF-β levels after fish oil supplementation of pregnant women. J. Allergy Clin. Immunol. 2008, 121, 464–470. [Google Scholar] [CrossRef] [PubMed]
- Romero, V.C.; Somers, E.C.; Stolberg, V.; Clinton, C.; Chensue, S.; Djuric, Z.; Berman, D.R.; Treadwell, M.C.; Vahratian, A.M.; Mozurkewich, E. Developmental programming for allergy: A secondary analysis of the mothers, Omega-3, and mental health study. Am. J. Obstet. Gynecol. 2013, 208, e311–e316. [Google Scholar] [CrossRef] [PubMed]
- Palmer, D.J.; Sullivan, T.; Gold, M.S.; Prescott, S.L.; Heddle, R.; Gibson, R.A.; Makrides, M. Effect of n-3 long chain polyunsaturated fatty acid supplementation in pregnancy on infants’ allergies in first year of life: Randomised controlled trial. BMJ 2012, 344, e184. [Google Scholar] [CrossRef]
- Harris, C.; Demmelmair, H.; Von Berg, A.; Lehmann, I.; Flexeder, C.; Koletzko, B.; Heinrich, J.; Standl, M. Associations between fatty acids and low-grade inflammation in children from the LISAplus birth cohort study. Eur. J. Clin. Nutr. 2017, 71, 1303–1311. [Google Scholar] [CrossRef]
- Chuang, S.L.; Wang, Y.; Demertzi, E.; Willetts, S.; Talbot, I.; Johnson, M.; Pantelidis, P.; Crawford, M.; Ogundipe, E. Fatty acid profiles in pregnancy and cord blood related to meconium microbiota. J. Matern. Fetal Neonatal. Med. 2014, 27, 331. [Google Scholar] [CrossRef]
- Balogun, K.; Balmert, L.; Jao, J.; Sun, S.; Bazinet, R.; Serghides, L. Maternal and cord plasma bioactive eicosanoid profiles differ in HIV+ and HIV−women. Top Antivir. Med. 2020, 28, 295–296. [Google Scholar]
- Nct. Docosahexaenoic Acid (DHA) Supplementation in High Risk Pregnancies. 2019. Available online: https://clinicaltrials.gov/show/NCT04069195 (accessed on 28 January 2022).
- Barden, A.E.; Mori, T.A.; Dunstan, J.A.; Taylor, A.L.; Thornton, A.; Croft, K.D.; Beilin, L.J.; Prescott, S.L. Fish oil supplementation in pregnancy lowers F2-isoprostanes in neonates at high risk of atopy. Free Radic. Res. 2004, 38, 233–239. [Google Scholar] [CrossRef]
- Best, K.P.; Sullivan, T.R.; Palmer, D.J.; Gold, M.; Martin, J.; Kennedy, D.; Makrides, M. Prenatal omega-3 LCPUFA and symptoms of allergic disease and sensitization throughout early childhood—A longitudinal analysis of long-term follow-up of a randomized controlled trial. World Allergy Organ J. 2018, 11, 10. [Google Scholar] [CrossRef]
- Furuhjelm, C.; Warstedt, K.; Fagerås, M.; Fälth-Magnusson, K.; Larsson, J.; Fredriksson, M.; Duchén, K. Allergic disease in infants up to 2 years of age in relation to plasma omega-3 fatty acids and maternal fish oil supplementation in pregnancy and lactation. Pediatr. Allergy Immunol. 2011, 22, 505–514. [Google Scholar] [CrossRef] [PubMed]
- Mozurkewich, E.L.; Greenwood, M.; Clinton, C.; Berman, D.; Romero, V.; Djuric, Z.; Qualls, C.; Gronert, K. Pathway markers for pro-resolving lipid mediators in maternal and umbilical cord blood: A secondary analysis of the mothers, Omega-3, and mental health study. Front. Pharmacol. 2016, 7, 274. [Google Scholar] [CrossRef] [PubMed]
- Mozurkewich, E.L.; Berman, D.R.; Vahratian, A.; Clinton, C.M.; Romero, V.C.; Chilimigras, J.L.; Vazquez, D.; Qualls, C.; Djuric, Z. Effect of prenatal EPA and DHA on maternal and umbilical cord blood cytokines. BMC Pregnancy Childbirth 2018, 18, 261. [Google Scholar] [CrossRef] [PubMed]
- See, V.H.L.; Mas, E.; Prescott, S.L.; Beilin, L.J.; Burrows, S.; Barden, A.E.; Huang, R.C.; Mori, T.A. Effects of prenatal n-3 fatty acid supplementation on offspring resolvins at birth and 12 years of age: A double-blind, randomised controlled clinical trial. Br. J. Nutr. 2017, 118, 971–980. [Google Scholar] [CrossRef]
- Barman, M.; Jonsson, K.; Wold, A.E.; Sandberg, A.S. Exposure to a farm environment during pregnancy increases the proportion of arachidonic acid in the cord sera of offspring. Nutrients 2019, 11, 238. [Google Scholar] [CrossRef]
- Barman, M.; Rabe, H.; Hesselmar, B.; Johansen, S.; Sandberg, A.S.; Wold, A.E. Cord blood levels of epa, a marker of fish intake, correlate with infants’ T-and B-lymphocyte phenotypes and risk for allergic disease. Nutrients 2020, 12, 3000. [Google Scholar] [CrossRef]
- Dirix, C.E.H.; Hogervorst, J.G.F.; Rump, P.; Hendriks, J.J.E.; Bruins, M.; Hornstra, G. Prenatal arachidonic acid exposure and selected immune-related variables in childhood. Br. J. Nutr. 2009, 102, 387–397. [Google Scholar] [CrossRef]
- Montes, R.; Chisaguano, A.M.; Castellote, A.I.; Morales, E.; Sunyer, J.; López-Sabater, M.C. Fatty-acid composition of maternal and umbilical cord plasma and early childhood atopic eczema in a Spanish cohort. Eur. J. Clin. Nutr. 2013, 67, 658–663. [Google Scholar] [CrossRef]
- Newson, R.B.; Shaheen, S.O.; Henderson, A.J.; Emmett, P.M.; Sherriff, A.; Calder, P.C. Umbilical cord and maternal blood red cell fatty acids and early childhood wheezing and eczema. J. Allergy Clin. Immunol. 2004, 114, 531–537. [Google Scholar] [CrossRef]
- Byberg, K.; Øymar, K.; Aksnes, L. Fatty acids in cord blood plasma, the relation to soluble CD23 and subsequent atopy. Prostaglandins Leukot Essent Fat. Acids 2008, 78, 61–65. [Google Scholar] [CrossRef]
- Yu, G.; Kjellman, N.I.M.; Björkstén, B. Phospholipid fatty acids in cord blood: Family history and development of allergy. Acta Paediatr. 1996, 85, 679–683. [Google Scholar] [CrossRef] [PubMed]
- Galli, E.; Picardo, M.; Chini, L.; Passi, S.; Moschese, V.; Terminali, O.; Paone, F.; Fraioli, G.; Rossi, P. Analysis of polyunsaturated fatty acids in newborn sera: A screening tool for atopic disease? Br. J. Dermatol. 1994, 130, 752–756. [Google Scholar] [CrossRef] [PubMed]
- Jasani, B.; Simmer, K.; Patole, S.K.; Rao, S.C. Long chain polyunsaturated fatty acid supplementation in infants born at term. Cochrane Database Syst. Rev. 2017, 3, CD000376. [Google Scholar] [CrossRef] [PubMed]
- Bahreynian, M.; Feizi, A.; Kelishadi, R. Is fatty acid composition of breast milk different in various populations? A systematic review and meta-analysis. Int. J. Food Sci. Nutr. 2020, 71, 909–920. [Google Scholar] [CrossRef] [PubMed]
- Commission Delegated Regulation (EU) 2016/127 of 25 September 2015 Supplementing Regulation (EU) No 609/2013 of the European Parliament and of the Council as Regards the Specific Compositional and Information Requirements for Infant Formula and Follow-on Formula and as Regards Requirements on Information Relating to Infant and Young Child Feeding. Available online: http://data.europa.eu/eli/reg_del/2016/127/2021-07-15 (accessed on 1 February 2022).
- Koletzko, B.; Bergmann, K.; Brenna, J.T.; Calder, P.C.; Campoy, C.; Clandinin, M.T.; Colombo, J.; Daly, M.; Decsi, T.; Demmelmair, H.; et al. Should formula for infants provide arachidonic acid along with DHA? A position paper of the European Academy of Paediatrics and the Child Health Foundation. Am. J. Clin. Nutr. 2020, 111, 10–16. [Google Scholar] [CrossRef] [PubMed]
- Tounian, P.; Bellaiche, M.; Legrand, P. ARA or no ARA in infant formulae, that is the question. Arch. Pediatr. 2021, 28, 69–74. [Google Scholar] [CrossRef]
- Barden, A.E.; Mas, E.; Mori, T.A. n-3 Fatty acid supplementation and proresolving mediators of inflammation. Curr. Opin. Lipidol. 2016, 27, 26–32. [Google Scholar] [CrossRef]
- Hogenkamp, A.; Ehlers, A.; Garssen, J.; Willemsen, L.E.M. Allergy modulation by N-3 long chain polyunsaturated fatty acids and fat soluble nutrients of the Mediterranean diet. Front. Pharmacol. 2020, 11, 1244. [Google Scholar] [CrossRef]
- Ranade, P.S.; Rao, S.S. Maternal long-chain PUFA supplementation during protein deficiency improves brain fatty acid accretion in rat pups by altering the milk fatty acid composition of the dam. J. Nutr. Sci. 2013, 2, e5. [Google Scholar] [CrossRef]
- Wohlford, E.M.; Huang, P.F.; Elhawary, J.R.; Millette, L.A.; Contreras, M.G.; Witonsky, J.; Holweg, C.T.J.; Oh, S.S.; Lee, C.; Merenda, C.; et al. Racial/ethnic differences in eligibility for asthma biologics among pediatric populations. J. Allergy Clin. Immunol. 2021, 148, 1324–1331. [Google Scholar] [CrossRef]
- Allaire, J.; Couture, P.; Leclerc, M.; Charest, A.; Marin, J.; Lepine, M.C.; Talbot, D.; Tchernof, A.; Lamarche, B. A randomized, crossover, head-to-head comparison of eicosapentaenoic acid and docosahexaenoic acid supplementation to reduce inflammation markers in men and women: The Comparing EPA to DHA (ComparED) Study. Am. J. Clin. Nutr. 2016, 104, 280–287. [Google Scholar] [CrossRef] [PubMed]
- Li, K.; Huang, T.; Zheng, J.; Wu, K.; Li, D. Effect of marine-derived n-3 polyunsaturated fatty acids on C-reactive protein, interleukin 6 and tumor necrosis factor alpha: A meta-analysis. PLoS ONE 2014, 9, e88103. [Google Scholar] [CrossRef]
- Vors, C.; Allaire, J.; Marin, J.; Lepine, M.C.; Charest, A.; Tchernof, A.; Couture, P.; Lamarche, B. Inflammatory gene expression in whole blood cells after EPA vs. DHA supplementation: Results from the ComparED study. Atherosclerosis 2017, 257, 116–122. [Google Scholar] [CrossRef] [PubMed]
- Asztalos, I.B.; Gleason, J.A.; Sever, S.; Gedik, R.; Asztalos, B.F.; Horvath, K.V.; Dansinger, M.L.; Lamon-Fava, S.; Schaefer, E.J. Effects of eicosapentaenoic acid and docosahexaenoic acid on cardiovascular disease risk factors: A randomized clinical trial. Metabolism 2016, 65, 1636–1645. [Google Scholar] [CrossRef]
- Vors, C.; Allaire, J.; Mejia, S.B.; Khan, T.A.; Sievenpiper, J.L.; Lamarche, B. Comparing the effects of docosahexaenoic and eicosapentaenoic acids on inflammation markers using pairwise and network meta-analyses of randomized controlled trials. Adv. Nutr. 2021, 12, 128–140. [Google Scholar] [CrossRef]
- Klemens, C.M.; Berman, D.R.; Mozurkewich, E.L. The effect of perinatal omega-3 fatty acid supplementation on inflammatory markers and allergic diseases: A systematic review. BJOG 2011, 118, 916–925. [Google Scholar] [CrossRef]
- Gunaratne, A.W.; Makrides, M.; Collins, C.T. Maternal prenatal and/or postnatal n-3 long chain polyunsaturated fatty acids (LCPUFA) supplementation for preventing allergies in early childhood. Cochrane Database Syst. Rev. 2015, 7, CD010085. [Google Scholar] [CrossRef]
- Lin, J.; Zhang, Y.; Zhu, X.; Wang, D.; Dai, J. Effects of supplementation with omega-3 fatty acids during pregnancy on asthma or wheeze of children: A systematic review and meta-analysis. J. Matern. Fetal Neonatal Med. 2020, 33, 1792–1801. [Google Scholar] [CrossRef]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).