The Advantages of Inorganic Fertilization for the Mass Production of Copepods as Food for Fish Larvae in Aquaculture
Abstract
:1. Introduction
2. Materials and Methods
2.1. Experimental Design
2.2. Physicochemical Analyses
2.3. Biological Analyses
2.4. Statistical Analysis
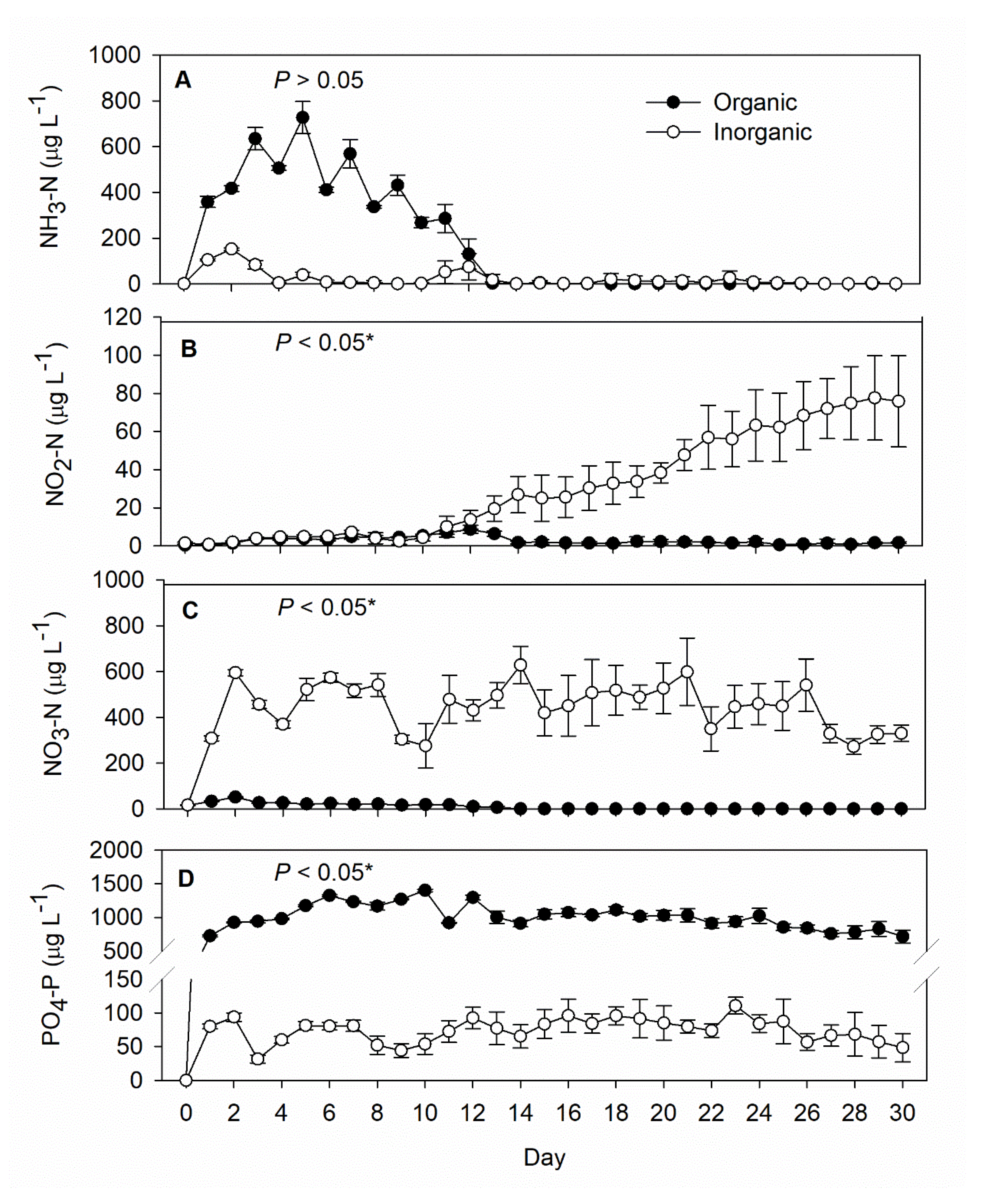
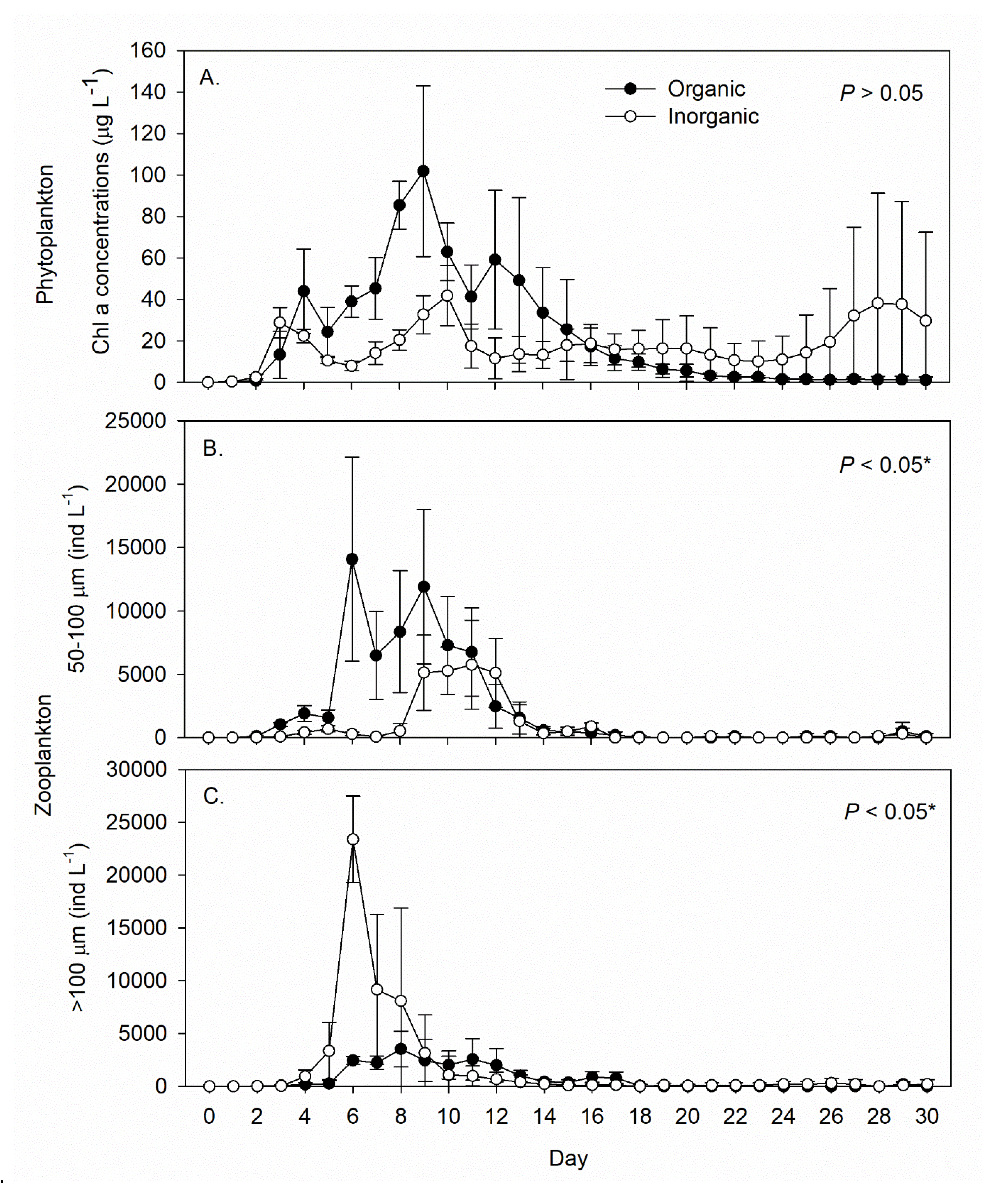
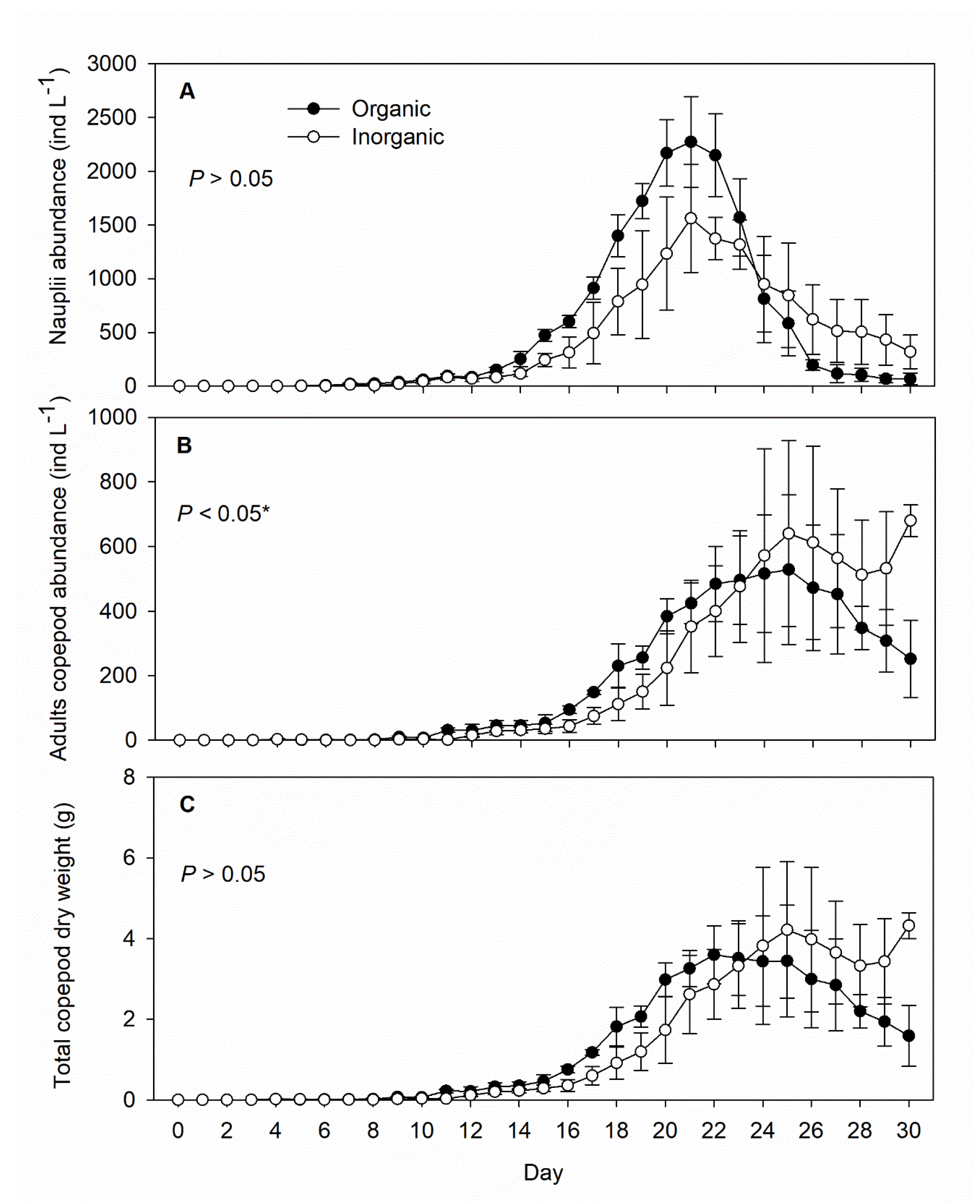
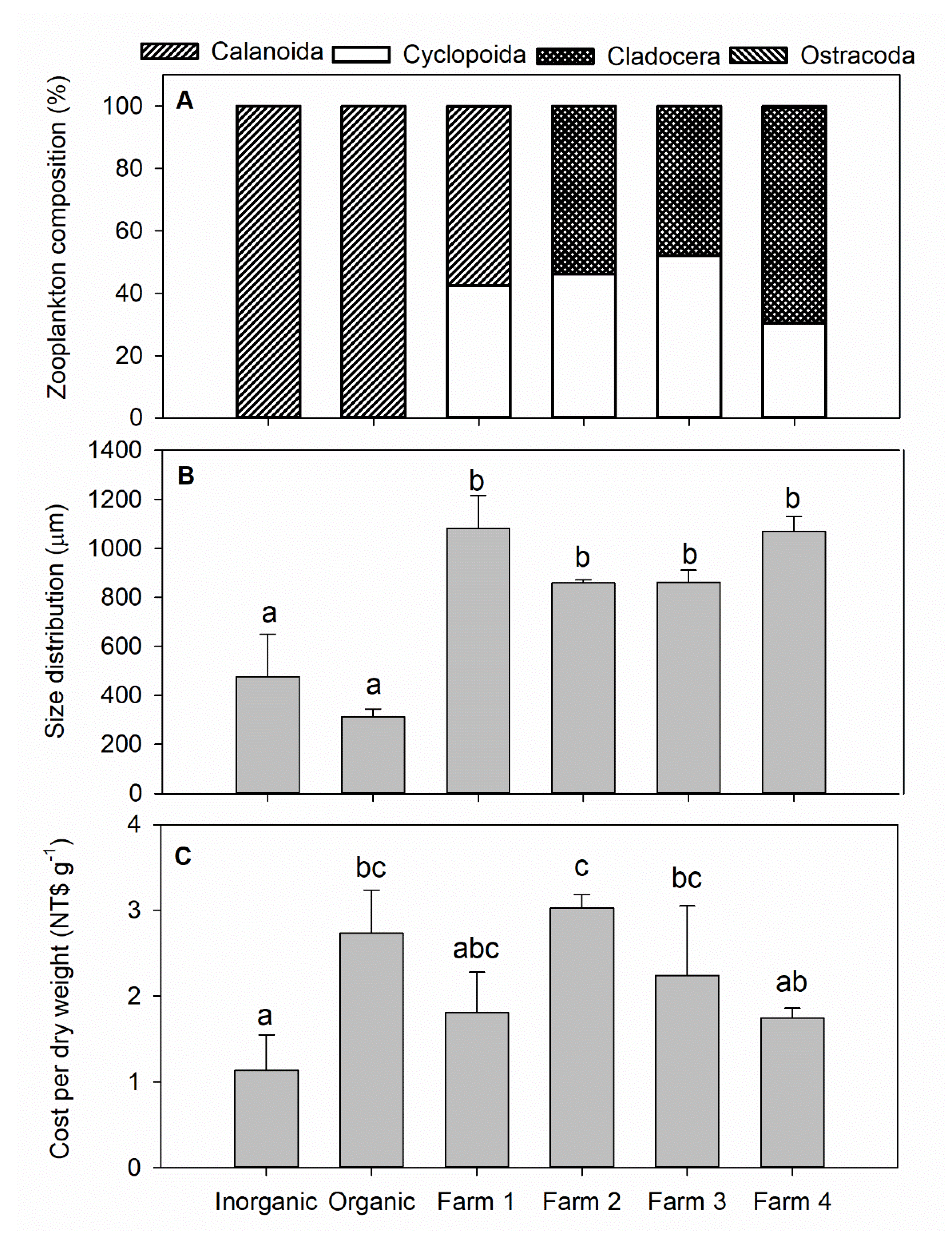
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- FAO. The State of World Fisheries and Aquaculture (SOFIA); FAO: Rome, Italy, 2020. [Google Scholar]
- Cahu, C.; Infante, J.Z.; Takeuchi, T. Nutritional components affecting skeletal development in fish larvae. Aquaculture 2003, 227, 245–258. [Google Scholar] [CrossRef] [Green Version]
- Tew, K.S.; Chang, Y.C.; Meng, P.J.; Leu, M.Y.; Glover, D.C. Towards sustainable exhibits–application of an inorganic fertilization method in coral reef fish larviculture in an aquarium. Aquac. Res. 2016, 47, 2748–2756. [Google Scholar] [CrossRef]
- Dhont, J.; Dierckens, K.; Støttrup, J.; Van Stappen, G.; Wille, M.; Sorgeloos, P. Rotifers, Artemia and copepods as live feeds for fish larvae in aquaculture. In Advances in Aquaculture Hatchery Technology; Allan, G., Burnell, G., Eds.; Woodhead Publishing: Cambridge, UK, 2013; pp. 157–202. [Google Scholar]
- Aragão, C.; Conceição, L.E.; Dinis, M.T.; Fyhn, H.J. Amino acid pools of rotifers and Artemia under different conditions: Nutritional implications for fish larvae. Aquaculture 2004, 234, 429–445. [Google Scholar] [CrossRef]
- Hagiwara, A.; Marcial, H.S. The use of non-Brachionus plicatilis species complex rotifer in larviculture. Hydrobiologia 2019, 844, 163–172. [Google Scholar] [CrossRef] [Green Version]
- Rasdi, N.W.; Qin, J.G. Copepod supplementation as a live food improved growth and survival of Asian seabass Lates calcarifer larvae. Aquac. Res. 2018, 49, 3606–3613. [Google Scholar] [CrossRef]
- Payne, M.F.; Rippingale, R.J. Intensive cultivation of the calanoid copepod Gladioferens imparipes. Aquaculture 2001, 201, 329–342. [Google Scholar] [CrossRef]
- Abatzopoulos, T.J.; Beardmore, J.; Clegg, J.; Sorgeloos, P. Artemia: Basic and Applied Biology; Springer Science & Business Media: Berlin/Heidelberg, Germany, 2013; Volume 1. [Google Scholar]
- Buskey, E.J. Behavioral characteristics of copepods that affect their suitability as food for larval fishes. In Copepods in Aquaculture; Lee, C.S., O’Bryen, P.J., Marcus, N.H., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2005; pp. 91–105. [Google Scholar]
- Bengtson, D.A. Status of marine aquaculture in relation to live prey: Past, present and future. In Live Feeds in Marine Aquaculture; Støttrup, J.G., McEvoy, L.A., Eds.; Wiley-Blackwell: Hoboken, NJ, USA, 2003; pp. 1–16. [Google Scholar]
- Drillet, G.; Lombard, F. A first step towards improving copepod cultivation using modelling: The effects of density, crowding, cannibalism, tank design and strain selection on copepod egg production yields. Aquac. Res. 2015, 46, 1638–1647. [Google Scholar] [CrossRef]
- Jepsen, P.M.; Thoisen, C.V.; Carron-Cabaret, T.; Pinyol-Gallemí, A.; Nielsen, S.L.; Hansen, B.W. Effects of salinity, commercial salts, and water type on cultivation of the cryptophyte microalgae Rhodomonas salina and the calanoid copepod Acartia tonsa. J. World Aquac. Soc. 2019, 50, 104–118. [Google Scholar] [CrossRef] [Green Version]
- Abate, T.G.; Nielsen, R.; Nielsen, M.; Drillet, G.; Jepsen, P.M.; Hansen, B.W. Economic feasibility of copepod production for commercial use: Result from a prototype production facility. Aquaculture 2015, 436, 72–79. [Google Scholar] [CrossRef]
- Drillet, G.; Frouël, S.; Sichlau, M.H.; Jepsen, P.M.; Højgaard, J.K.; Joarder, A.K.; Hansen, B.W. Status and recommendations on marine copepod cultivation for use as live feed. Aquaculture 2011, 315, 155–166. [Google Scholar] [CrossRef]
- Blanda, E.; Drillet, G.; Huang, C.C.; Hwang, J.S.; Højgaard, J.K.; Jakobsen, H.H.; Rayner, T.A.; Su, H.M.; Hansen, B.W. An analysis of how to improve production of copepods as live feed from tropical Taiwanese outdoor aquaculture ponds. Aquaculture 2017, 479, 432–441. [Google Scholar] [CrossRef]
- Luo, X.; Li, C.; Huang, X. Effect of diet on the development, survival, and reproduction of the calanoid copepod Pseudodiaptomus dubia. J. Oceanol. Limnol. 2019, 37, 1756–1767. [Google Scholar] [CrossRef]
- Li, C.; Luo, X.; Huang, X.; Gu, B. Influences of temperature on development and survival, reproduction and growth of a calanoid copepod (Pseudodiaptomus dubia). Sci. World J. 2009, 9, 866–879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kang’ombe, J.; Brown, J.A.; Halfyard, L.C. Effect of using different types of organic animal manure on plankton abundance, and on growth and survival of Tilapia rendalli (Boulenger) in ponds. Aquac. Res. 2006, 37, 1360–1371. [Google Scholar] [CrossRef]
- Adewumi, A. The growth and gonadal maturation of the African catfish, Clarias gariepinus (Burchell) broodstock fed differently heated soybean-based diets. Aquac. Nutr. 2006, 12, 267–274. [Google Scholar] [CrossRef]
- Kaatz, S.E.; Morris, J.E.; Rudacille, J.B.; Johnson, J.A.; Clayton, R.D. Role of organic fertilizers in walleye (Sander vitreus) production in plastic-lined culture ponds. Aquac. Res. 2011, 42, 490–498. [Google Scholar] [CrossRef]
- Mischke, C.C.; Zimba, P.V. Plankton community responses in earthen channel catfish nursery ponds under various fertilization regimes. Aquaculture 2004, 233, 219–235. [Google Scholar] [CrossRef]
- Zeng, C.; Romano, N. Nitrogenous wastes: Often overlooked pollutants in aquatic environments. J. Mar. Sci. Res. Dev. 2013, 3, 1000e118. [Google Scholar] [CrossRef] [Green Version]
- Hargreaves, J.A. Nitrogen biogeochemistry of aquaculture ponds. Aquaculture 1998, 166, 181–212. [Google Scholar] [CrossRef]
- Culver, D.A. Effects of the N: P ratio in fertilizer for fish hatchery ponds. Int. Ver. Für Theor. Angew. Limnol. Verh. 1991, 24, 1503–1507. [Google Scholar] [CrossRef]
- Culver, D.A.; Madon, S.P.; Qin, J. Percid pond production techniques: Timing, enrichment, and stocking density manipulation. J. Appl. Aquac. 1994, 2, 9–32. [Google Scholar] [CrossRef]
- Qin, J.; Culver, D.A. The survival and growth of larval walleye, Stizostedion vitreum, and trophic dynamics in fertilized ponds. Aquaculture 1992, 108, 257–276. [Google Scholar] [CrossRef]
- Tew, K.S.; Conroy, J.D.; Culver, D.A. Effects of lowered inorganic phosphorus fertilization rates on pond production of percid fingerlings. Aquaculture 2006, 255, 436–446. [Google Scholar] [CrossRef]
- Jacob, A.P.; Culver, D.A. Experimental evaluation of the impacts of reduced inorganic phosphorus fertilization rates on juvenile saugeye production. Aquaculture 2010, 304, 22–33. [Google Scholar] [CrossRef]
- Kuo, J.; Chen, C.Y.; Han, C.C.; Ju, Y.M.; Tew, K.S. Analyses of diet preference of larval orange-spotted grouper (Epinephelus coioides) grown under inorganic fertilization method using next-generation sequencing. Aquaculture 2021, 531, 735916. [Google Scholar] [CrossRef]
- Tew, K.S.; Meng, P.J.; Lin, H.S.; Chen, J.H.; Leu, M.Y. Experimental evaluation of inorganic fertilization in larval giant grouper (Epinephelus lanceolatus Bloch) production. Aquac. Res. 2013, 44, 439–450. [Google Scholar] [CrossRef]
- Pai, S.C.; Riley, J. Determination of nitrate in the presence of nitrite in natural waters by flow injection analysis with a non-quantitative on-line cadmium reductor. Int. J. Environ. Anal. Chem. 1994, 57, 263–277. [Google Scholar] [CrossRef]
- Parsons, T.; Maita, Y.; Lalli, C.M. Amanual of Chemical and Biological Methods for Seawater Analysis; Elsevier: Amsterdam, The Netherlands, 1984. [Google Scholar]
- Chen, J.Y.; Zeng, C.; Jerry, D.R.; Cobcroft, J.M. Recent advances of marine ornamental fish larviculture: Broodstock reproduction, live prey and feeding regimes, and comparison between demersal and pelagic spawners. Rev. Aquac. 2020, 12, 1518–1541. [Google Scholar] [CrossRef]
- Divya, K.R.; AkbarAli, I.; Schmidt, B.V.; John, E.M.; Sureshkumar, S.; Thazhakot Vasunambesan, S. Improvement of nutritional quality of live feed for aquaculture: An overview. Aquac. Res. 2020, 51, 1–17. [Google Scholar]
- McKinnon, A.; Duggan, S.; Nichols, P.; Rimmer, M.; Semmens, G.; Robino, B. The potential of tropical paracalanid copepods as live feeds in aquaculture. Aquaculture 2003, 223, 89–106. [Google Scholar] [CrossRef]
- Piasecki, W.; Goodwin, A.E.; Eiras, J.C.; Nowak, B.F. Importance of copepoda in freshwater aquaculture. Zool. Stud. 2004, 43, 193–205. [Google Scholar]
- Støttrup, J. The elusive copepods: Their production and suitability in marine aquaculture. Aquac. Res. 2000, 31, 703–711. [Google Scholar] [CrossRef]
- Hansen, B.W.; Hansen, P.J.; Nielsen, T.G.; Jepsen, P.M. Effects of elevated pH on marine copepods in mass cultivation systems: Practical implications. J. Plankton Res. 2017, 39, 984–993. [Google Scholar] [CrossRef]
- Pedersen, M.F.; Hansen, P.J. Effects of high pH on a natural marine planktonic community. Mar. Ecol. Prog. Ser. 2003, 260, 19–31. [Google Scholar] [CrossRef] [Green Version]
- Buttino, I. The effect of low concentrations of phenol and ammonia on egg production rates, fecal pellet production and egg viability of the calanoid copepod Acartia clausi. Mar. Biol. 1994, 119, 629–634. [Google Scholar] [CrossRef]
- Jepsen, P.M.; Andersen, C.V.; Schjelde, J.; Hansen, B.W. Tolerance of un-ionized ammonia in live feed cultures of the calanoid copepod Acartia tonsa Dana. Aquac. Res. 2015, 46, 420–431. [Google Scholar] [CrossRef]
- Davis, A.K.; Anderson, R.S.; Spierling, R.; Leader, S.; Lesne, C.; Mahan, K.; Lundquist, T.; Benemann, J.R.; Lane, T.; Polle, J.E.W. Characterization of a novel strain of Tribonema minus demonstrating high biomass productivity in outdoor raceway ponds. Bioresour. Technol. 2021, 331, 125007. [Google Scholar] [CrossRef]
- Cheng, P.; Cheng, J.J.; Cobb, K.; Zhou, C.; Zhou, N.; Addy, M.; Chen, P.; Yan, X.; Ruan, R. Tribonema sp. and Chlorella zofingiensis co-culture to treat swine wastewater diluted with fishery wastewater to facilitate harvest. Bioresour. Technol. 2020, 297, 122516. [Google Scholar] [CrossRef]
- Malej, A.; Harris, R.P. Inhibition of copepod grazing by diatom exudates: A factor in the development of mucus aggregates? Mar. Ecol. Prog. Ser. 1993, 33–42. [Google Scholar] [CrossRef]
- Ianora, A.; Miralto, A.; Poulet, S.A.; Carotenuto, Y.; Buttino, I.; Romano, G.; Casotti, R.; Pohnert, G.; Wichard, T.; Colucci-D’Amato, L. Aldehyde suppression of copepod recruitment in blooms of a ubiquitous planktonic diatom. Nature 2004, 429, 403–407. [Google Scholar] [CrossRef]
- Miralto, A.; Barone, G.; Romano, G.; Poulet, S.; Ianora, A.; Russo, G.; Buttino, I.; Mazzarella, G.; Laabir, M.; Cabrini, M. The insidious effect of diatoms on copepod reproduction. Nature 1999, 402, 173–176. [Google Scholar] [CrossRef]
- Berk, S.; Brownlee, D.; Heinle, D.; Kling, H.; Colwell, R. Ciliates as a food source for marine planktonic copepods. Microb. Ecol. 1977, 4, 27–40. [Google Scholar] [CrossRef] [PubMed]
- Calbet, A.; Saiz, E. The ciliate-copepod link in marine ecosystems. Aquat. Microb. Ecol. 2005, 38, 157–167. [Google Scholar] [CrossRef]
- Wu, F.; Dai, M.; Huang, H.; Qi, Z. Plankton ciliate community responses to different aquatic environments in Nan’ao Island, a representative mariculture base in the South China Sea. Mar. Freshw. Res. 2018, 70, 426–436. [Google Scholar] [CrossRef]
- Lin, H.Y.; Yeh, W.Y.; Tsai, S.F.; Chiang, K.P.; Lin, J.H.Y.; Tsao, C.C.; Lin, H.J. Biological protective effects against Vibrio infections in grouper larvae using the Strombidium sp. NTOU1, a marine ciliate amenable for scaled-up culture and with an excellent bacteriovorous ability. Front. Mar. Sci. 2020, 7, 373. [Google Scholar] [CrossRef]
- Chaijarasphong, T.; Munkongwongsiri, N.; Stentiford, G.D.; Aldama-Cano, D.J.; Thansa, K.; Flegel, T.W.; Sritunyalucksana, K.; Itsathitphaisarn, O. The shrimp microsporidian Enterocytozoon hepatopenaei (EHP): Biology, pathology, diagnostics and control. J. Invertebr. Pathol. 2021, 186, 107458. [Google Scholar] [CrossRef]
- Siddique, A.B.; Moniruzzaman, M.; Ali, S.; Dewan, M.; Islam, M.R.; Islam, M.; Amin, M.B.; Mondal, D.; Parvez, A.K.; Mahmud, Z.H. Characterization of pathogenic Vibrio parahaemolyticus isolated from fish aquaculture of the Southwest coastal area of Bangladesh. Front. Microbiol. 2021, 12, 266. [Google Scholar] [CrossRef]

Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Hong, G.-K.; Tew, K.S. The Advantages of Inorganic Fertilization for the Mass Production of Copepods as Food for Fish Larvae in Aquaculture. Life 2022, 12, 441. https://doi.org/10.3390/life12030441
Hong G-K, Tew KS. The Advantages of Inorganic Fertilization for the Mass Production of Copepods as Food for Fish Larvae in Aquaculture. Life. 2022; 12(3):441. https://doi.org/10.3390/life12030441
Chicago/Turabian StyleHong, Guo-Kai, and Kwee Siong Tew. 2022. "The Advantages of Inorganic Fertilization for the Mass Production of Copepods as Food for Fish Larvae in Aquaculture" Life 12, no. 3: 441. https://doi.org/10.3390/life12030441
APA StyleHong, G.-K., & Tew, K. S. (2022). The Advantages of Inorganic Fertilization for the Mass Production of Copepods as Food for Fish Larvae in Aquaculture. Life, 12(3), 441. https://doi.org/10.3390/life12030441





