B Chromosomes’ Sequences in Yellow-Necked Mice Apodemus flavicollis—Exploring the Transcription
Abstract
:1. Introduction
2. Materials and Methods
2.1. Samples
2.2. RNA and DNA Extraction
2.3. Primer Design
2.4. PCR
2.5. RT-PCR
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Makunin, A.I.; Dementyeva, P.V.; Graphodatsky, A.S.; Volobouev, V.T.; Kukekova, A.V.; Trifonov, V.A. Genes on B chromosomes of vertebrates. Mol. Cytogenet. 2014, 7, 99. [Google Scholar] [CrossRef] [Green Version]
- Jones, R.N.; Rees, H. B Chromosomes; Acad Press: London, UK, 1982. [Google Scholar]
- Camacho, J.P.M.; Sharbel, T.F.; Beukeboom, L.W. B-chromosome evolution. Philos. Trans. R. Soc. B Biol. Sci. 2000, 355, 163–178. [Google Scholar] [CrossRef] [PubMed]
- Jones, R.N.; Houben, A. B chromosomes in plants: Escapeees from the A chromosome genome? Trends Plant Sci. 2003, 8, 417–423. [Google Scholar] [CrossRef]
- Burt, A.; Trivers, R. Genes in Conflict: The Biology of Selfish Genetic Elements; Harvard University Press: Cambridge, MA, USA, 2006; pp. 325–380. [Google Scholar]
- Houben, A.; Banaei-Moghaddam, A.M.; Klemme, S.; Timmis, J.N. Evolution and biology of supernumerary B chromosomes. Cell Mol. Life Sci. 2014, 71, 467–478. [Google Scholar] [CrossRef]
- Bidau, C.J. A nucleolar-organizing B chromosome showing segregation–distortion in the grasshopper Dichroplus pratensis (Melanoplinae, Acrididae). Can. J. Genet. Cytol. 1986, 28, 138–148. [Google Scholar] [CrossRef]
- Brockhouse, C.; Bass, J.A.B.; Feraday, R.M.; Straus, N.A. Supernumerary chromosome evolution in the Simulium vernum group (Diptera: Simuliidae). Genome 1989, 32, 516–521. [Google Scholar] [CrossRef]
- Miao, V.P.; Covert, S.F.; VanEtten, H.D. A fungal gene for antibiotic resistance on a dispensable (“B”) chromosome. Science 1991, 254, 1773–1776. [Google Scholar] [CrossRef] [PubMed]
- Tanić, N.; Vujošević, M.; Dedović-Tanić, N.; Dimitrijević, B. Differential gene expression in yellow-necked mice Apodemus flavicollis (Rodentia, Mammalia) with and without B chromosomes. Chromosoma 2005, 113, 418–427. [Google Scholar] [CrossRef]
- Adnađević, T.; Jovanović, V.M.; Blagojević, J.; Budinski, I.; Čabrilo, B.; Bijelić-Čabrilo, O.; Vujošević, M. Possible influence of B chromosomes on genes included in immune response and parasite burden in Apodemus flavicollis. PLoS ONE 2014, 9, e112260. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vujošević, M.; Rajičić, M.; Blagojević, J. B Chromosomes in Populations of Mammals Revisited. Genes 2018, 9, 487. [Google Scholar] [CrossRef] [Green Version]
- Valente, G.T.; Conte, M.A.; Fantinatti, B.E.A.; Cabral-de-Mello, D.C.; Carvalho, R.F.; Vicari, M.R.; Kocher, T.D.; Martins, C. Origin and evolution of B chromosomes in the cichlid fish Astatotilapia latifasciata based on integrated genomic analyses. Mol. Biol. Evol. 2014, 31, 2061–2072. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clark, F.E.; Conte, M.A.; Kocher, T.D. Genomic characterization of a B chromosome in Lake Malawi cichlid fishes. Genes 2018, 9, 610. [Google Scholar] [CrossRef]
- Leach, C.R. Molecular evidence for transcription of genes on a B chromosome in Crepis capillaris. Genetics 2005, 171, 269–278. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banaei-Moghaddam, A.M.; Meier, K.; Karimi-Ashtiyani, R.; Houben, A. Formation and Expression of Pseudogenes on the B Chromosome of Rye. Plant Cell 2013, 25, 2536–2544. [Google Scholar] [CrossRef] [Green Version]
- Ma, W.; Gabriel, T.S.; Martis, M.M.; Gursinsky, T.; Schubert, V.; Vrána, J.; Doležel, J.; Grundlach, H.; Altschmied, L.; Scholz, U.; et al. Rye B chromosomes encode a functional Argonaute-like protein with in vitro slicer activities similar to its A chromosome paralog. New Phytol. 2017, 213, 916–928. [Google Scholar] [CrossRef] [PubMed]
- Huang, W.; Du, Y.; Zhao, X.; Jin, W. B chromosome contains active genes and impacts the transcription of A chromosomes in maize (Zea mays L.). BMC Plant Biol. 2016, 16, 88. [Google Scholar] [CrossRef] [Green Version]
- Van Vugt, J.J.F.A.; de Nooijer, S.; Stouthamer, R.; de Jong, H. NOR activity and repeat sequences of the paternal sex ratio chromosome of the parasitoid wasp Trichogramma kaykai. Chromosoma 2005, 114, 410–419. [Google Scholar] [CrossRef]
- Ruiz-Estévez, M.; López-León, M.D.; Cabrero, J.; Camacho, J.P.M. B-chromosome ribosomal DNA is functional in the grasshopper Eyprepocnemis plorans. PLoS ONE 2012, 7, e36600. [Google Scholar] [CrossRef]
- Navarro-Domínguez, B.; Ruiz-Ruano, F.J.; Cabrero, J.; Corral, J.M.; López-León, M.D.; Sharbel, T.F.; Camacho, J.P.M. Protein-coding genes in B chromosomes of the grasshopper Eyprepocnemis plorans. Sci. Rep. 2017, 7, 45200. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Trifonov, V.A.; Dementyeva, P.V.; Larkin, D.M.; O’Brien, P.C.M.; Perelman, P.L.; Yang, F.; Ferguson-Smith, M.A.; Grasphodatsky, A.S. Transcription of a protein-coding gene on B chromosomes of the Siberian roe deer (Capreolus pygargus). BMC Biol. 2013, 11, 90. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Valente, G.T.; Nakajima, R.T.; Fantinatti, B.E.A.; Marques, D.F.; Almeida, R.O.; Simões, R.P.; Martins, C. B chromosomes: From cytogenetics to systems biology. Chromosoma 2017, 126, 73–81. [Google Scholar] [CrossRef]
- Carchilan, M.; Kumke, K.; Mikolajewski, S.; Houben, A. Rye B chromosomes are weakly transcribed and might alter the transcriptional activity of A chromosome sequences. Chromosoma 2009, 118, 607–616. [Google Scholar] [CrossRef] [PubMed]
- Banaei-Moghaddam, A.M.; Schubert, V.; Kumke, K.; Weiβ, O.; Klemme, S.; Nagaki, K.; Macas, J.; González-Sánchez, M.; Heredia, V.; Gómez-Revilla, D.; et al. Nondisjunction in favor of a chromosome: The mechanism of rye B chromosome drive during pollen mitosis. Plant Cell 2012, 24, 4124–4134. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Banaei-Moghaddam, A.M.; Martis, M.M.; Macas, J.; Gundlach, H.; Himmelbach, A.; Altschmied, L.; Mayer, K.; Houben, A. Genes on B chromosomes: Old questions revisited with new tools. Biochim. Biophys. Acta (BBA)-Gene Regul. Mech. 2015, 1849, 64–70. [Google Scholar] [CrossRef]
- Akbari, O.S.; Antoshechkin, I.; Hay, B.A.; Ferree, P.M. Transcriptome profiling of Nasonia vitripennis testis reveals novel transcripts expressed from the selfish B chromosome, paternal sex ratio. G3 Genes Genomes Genet. 2013, 3, 1597–1605. [Google Scholar] [CrossRef] [Green Version]
- Ramos, É.; Cardoso, A.L.; Brown, J.; Marques, D.F.; Fantinatti, B.E.A.; Cabral-de-Mello, D.C.; Oliveira, R.A.; O’Neill, R.J.; Martins, C. The repetitive DNA element BncDNA, enriched in the B chromosome of the cichlid fish Astatotilapia latifasciata, transcribes a potentially noncoding RNA. Chromosoma 2017, 126, 313–323. [Google Scholar] [CrossRef] [Green Version]
- Martins, C.; Jehangir, M. A genomic glimpse of B chromosomes in cichlids. Genes Genomics 2021, 3, 199–208. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, K.; Terai, Y.; Mizoiri, S.; Aibara, M.; Nishihara, H.; Watanabe, M.; Kuroiwa, A.; Hirai, H.; Hirai, Y.; Matsuda, Y.; et al. B chromosomes have a functional effect on female sex determination in Lake Victoria cichlid fishes. PLoS Genet. 2011, 7, e1002203. [Google Scholar] [CrossRef] [PubMed]
- Clark, F.E.; Conte, M.A.; Ferreira-Bravo, I.A.; Poletto, A.B.; Martins, C.; Kocher, T.D. Dynamic sequence evolution of a sex-associated B chromosome in Lake Malawi cichlid fish. J. Hered. 2017, 108, 53–62. [Google Scholar] [CrossRef] [Green Version]
- Vujošević, M.; Blagojević, J.; Radosavljević, J.; Bejaković, D. B chromosome polymorphism in populations of Apodemus flavicollis in Yugoslavia. Genetica 1991, 83, 167–170. [Google Scholar] [CrossRef]
- Vujošević, M.; Blagojević, J. Seasonal changes of B-chromosome frequencies within the population of Apodemus flavicollis (Rodentia) on Cer mountain in Yugoslavia. Acta Theriol 1995, 40, 131–137. [Google Scholar] [CrossRef] [Green Version]
- Vujošević, M.; Blagojević, J. Does environment affect polymorphism of B chromosomes in the yellow-necked mouse Apodemus flavicollis. Z Säugetierk 2000, 65, 313–317. [Google Scholar]
- Blagojević, J.; Vujošević, M. Supernumerary chromosomes of Apodemus flavicollis (Rodentia, Mammalia). The highest number of B-chromosomes. Arh. Biol. Nauka 1991, 43, 31–32. [Google Scholar]
- Vujošević, M. B-chromosome polymorphism in Apodemus flavicollis (Rodentia, Mammalia) during five years. Caryologia 1992, 45, 347–352. [Google Scholar] [CrossRef]
- Blagojević, J.; Vujošević, M. B chromosomes and developmental homeostasis in the yellow-necked mouse, Apodemus flavicollis (Rodentia, Mammalia): Effects on nonmetric traits. Heredity 2004, 93, 249–254. [Google Scholar] [CrossRef] [PubMed]
- Wójcik, J.M.; Wójcik, A.M.; Macholán, M.; Piálek, J.; Zima, J. The mammalian model for population studies of B chromosomes: The wood mouse (Apodemus). Cytogenet. Genome Res. 2004, 106, 264–270. [Google Scholar] [CrossRef]
- Blagojević, J.; Vujošević, M. The role of B chromosomes in the population dynamics of yellow-necked wood mice Apodemus flavicollis (Rodentia, Mammalia). Genome 1995, 38, 472–478. [Google Scholar] [CrossRef]
- Jojić, V.; Blagojević, J.; Ivanović, A.; Bugarski-Stanojević, V.; Vujošević, M. Morphological integration of the mandible in yellow-necked field mice: The effects of B chromosomes. J. Mammal. 2007, 88, 689–695. [Google Scholar] [CrossRef] [Green Version]
- Jojić, V.; Blagojević, J.; Vujošević, M. B chromosomes and cranial variability in yellow-necked field mice (Apodemus flavicollis). J. Mammal. 2011, 92, 396–406. [Google Scholar] [CrossRef] [Green Version]
- Vujošević, M.; Jojić, V.; Bugarski-Stanojević, V.; Blagojević, J. Habitat quality and B chromosomes in the yellow-necked mouse Apodemus flavicollis. Ital. J. Zool. 2007, 74, 313–316. [Google Scholar] [CrossRef]
- Zima, J.; Piálek, J.; Macholán, M. Possible heterotic effects of B chromosomes on body mass in a population of Apodemus flavicollis. Can. J. Zool. 2003, 81, 1312–1317. [Google Scholar] [CrossRef]
- Vujošević, M.; Blagojević, J.; Jojić-Šipetić, V.; Bugarski-Stanojević, V.; Adnađević, T.; Stmenković, G. Distribution of B chromosomes in age categories of the yellow-necked mouse Apodemus flavicollis (Mammalia, Rodentia). Arch. Biol. Sci. 2009, 61, 653–658. [Google Scholar] [CrossRef]
- Rajičić, M.; Romanenko, S.A.; Karamysheva, T.V.; Blagojević, J.; Adnađević, T.; Budinski, I.; Bogdanov, A.S.; Trifonov, V.A.; Rubtsov, N.B.; Vujošević, M. The origin of B chromosomes in yellow-necked mice (Apodemus flavicollis)—Break rules but keep playing the game. PLoS ONE 2017, 12, e0172704. [Google Scholar] [CrossRef]
- Karamysheva, T.; Romanenko, S.; Makunin, A.; Rajičić, M.; Bogdanov, A.; Trifonov, V.; Blagojević, J.; Vujošević, M.; Orishchenko, K.; Rubtsov, N. New Data on Organization and Spatial Localization of B-Chromosomes in Cell Nuclei of the Yellow-Necked Mouse Apodemus flavicollis. Cells 2021, 10, 1819. [Google Scholar] [CrossRef] [PubMed]
- Makunin, A.I.; Rajičić, M.; Karamysheva, T.V.; Romanenko, S.A.; Druzhkova, A.S.; Blagojević, J.; Vujošević, M.; Rubtsov, N.B.; Graphodatsky, A.S.; Trifonov, V.A. Low-pass single-chromosome sequencing of human small supernumerary marker chromosomes (sSMCs) and Apodemus B chromosomes. Chromosoma 2018, 127, 301–311. [Google Scholar] [CrossRef] [Green Version]
- Bugarski-Stanojević, V.; Stamenković, G.; Blagojević, J.; Liehr, T.; Kosyakova, N.; Rajičić, M.; Vujošević, M. Exploring supernumeraries—A new marker for screening of B-chromosomes presence in the yellow necked mouse Apodemus flavicollis. PLoS ONE 2016, 11, e0160946. [Google Scholar] [CrossRef]
- Lord, R.D.J. The lens as an indicator of age in cotonatial rabbits. J. Wildl. Manag. 1959, 23, 358–360. [Google Scholar] [CrossRef]
- Koressaar, T.; Remm, M. Enhancements and modifications of primer design program Primer3. Bioinform 2007, 23, 1289–1291. [Google Scholar] [CrossRef] [Green Version]
- Ahnert, S.E.; Fink, T.M.A.; Zinovyev, A. How much non-coding DNA do eukaryotes require? J. Theor. Biol. 2008, 252, 587–592. [Google Scholar] [CrossRef] [Green Version]
- Balakirev, E.S.; Ayala, F.J. Pseudogenes: Are they “junk” or functional DNA? Annu. Rev. Genet. 2003, 37, 123–151. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Sandelin, A.; Bailey, P.; Bruce, S.; Engström, P.G.; Klos, J.K.; Wasserman, W.W.; Ericson, J.; Lenhard, B. Arrays of ultraconserved non-coding regions span the loci of key developmental genes in vertebrate genomes. BMC Genom. 2004, 5, 99. [Google Scholar] [CrossRef] [Green Version]
- Pink, R.C.; Wicks, K.; Caley, D.P.; Punch, E.K.; Jacobs, L.; Carter, D.R.F. Pseudogenes: Pseudo-functional or key regulators in health and disease? RNA 2011, 17, 792–798. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pennisi, E. ENCODE Project writes eulogy for junk DNA. Science 2012, 337, 1159–1161. [Google Scholar] [CrossRef]
- Mattick, J.S. The central role of RNA in human development and cognition. FEBS Lett. 2011, 585, 1600–1616. [Google Scholar] [CrossRef]
- Li, Y.; Kang, J.; Horwitz, M.S. Interaction of an adenovirus 14.7-kilodalton protein inhibitor of tumor necrosis factor alpha cytolysis with a new member of the GTPase superfamily of signal transducers. J. Virol. 1997, 71, 1576–1582. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zheng, D.; Frankish, A.; Baertsch, R.; Kapranov, P.; Reymond, A.; Choo, S.W.; Lu, Y.; Denoeud, F.; Antonarakis, S.E.; Snyder, M.; et al. Pseudogenes in the ENCODE regions: Consensus annotation, analysis of transcription, and evolution. Genome Res. 2007, 17, 839–851. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Johnsson, P.; Morris, K.V.; Grandér, D. Pseudogenes: A novel source of trans-acting antisense RNAs. Pseudogenes Methods Mol. Biol. 2014, 1167, 213–226. [Google Scholar] [CrossRef] [PubMed]
- Muro, E.M.; Mah, N.; Andrade-Navarro, M.A. Functional evidence of post-transcriptional regulation by pseudogenes. Biochimie 2011, 93, 1916–1921. [Google Scholar] [CrossRef]
- Carninci, P. RNA dust: Where are the genes? DNA Res. 2010, 17, 51–59. [Google Scholar] [CrossRef] [Green Version]
- Lawo, S.; Bashkurov, M.; Mullin, M.; Ferreria, M.G.; Kittler, R.; Habermann, B.; Tagliaferro, A.; Poser, I.; Hutchins, J.R.A.; Hegemann, B.; et al. HAUS, the 8-Subunit human augmin complex, regulates centrosome and spindle integrity. Curr. Biol. 2009, 19, 816–826. [Google Scholar] [CrossRef] [Green Version]
- Mirzaa, G.M.; Vitre, B.; Carpenter, G.; Abramowicz, I.; Gleeson, J.G.; Paciorkowski, A.R.; Cleveland, D.W.; Dobyns, W.B.; O’Driscoll, M. Mutations in CENPE define a novel kinetochore-centromeric mechanism for microcephalic primordial dwarfism. Hum. Genet. 2014, 133, 1023–1039. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Carmello, B.O.; Coan, R.L.B.; Cardoso, A.L.; Ramos, E.; Fantinatti, B.E.A.; Marques, D.F.; Oliveira, R.A.; Valente, G.T.; Martins, C. The hnRNP Q-like gene is retroinserted into the B chromosomes of the cichlid fish Astatotilapia latifasciata. Chromosome Res. 2017, 25, 277–290. [Google Scholar] [CrossRef] [PubMed]
- Camacho, J.P.; Schmid, M.; Cabrero, J. B chromosomes and sex in animals. Sex Dev. 2011, 5, 155–166. [Google Scholar] [CrossRef] [PubMed]
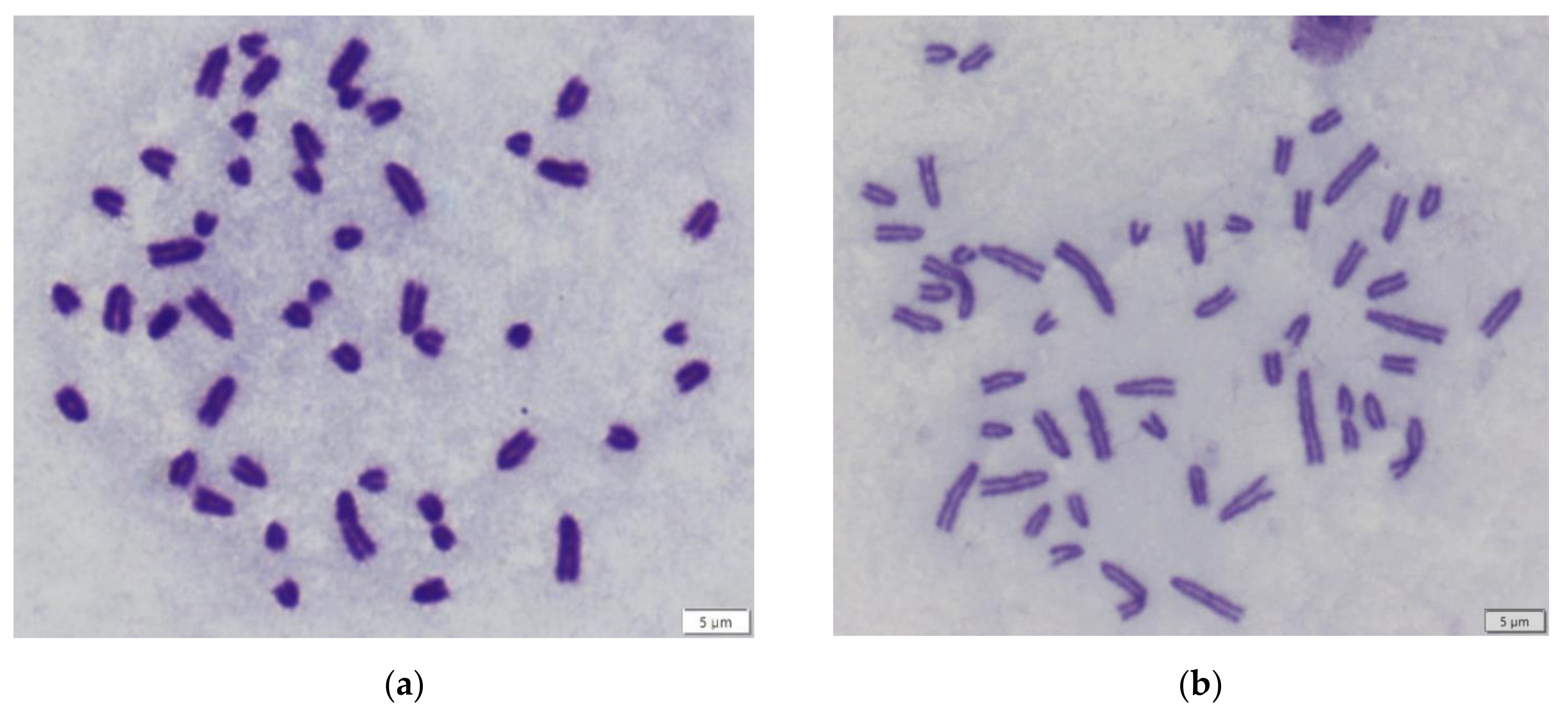



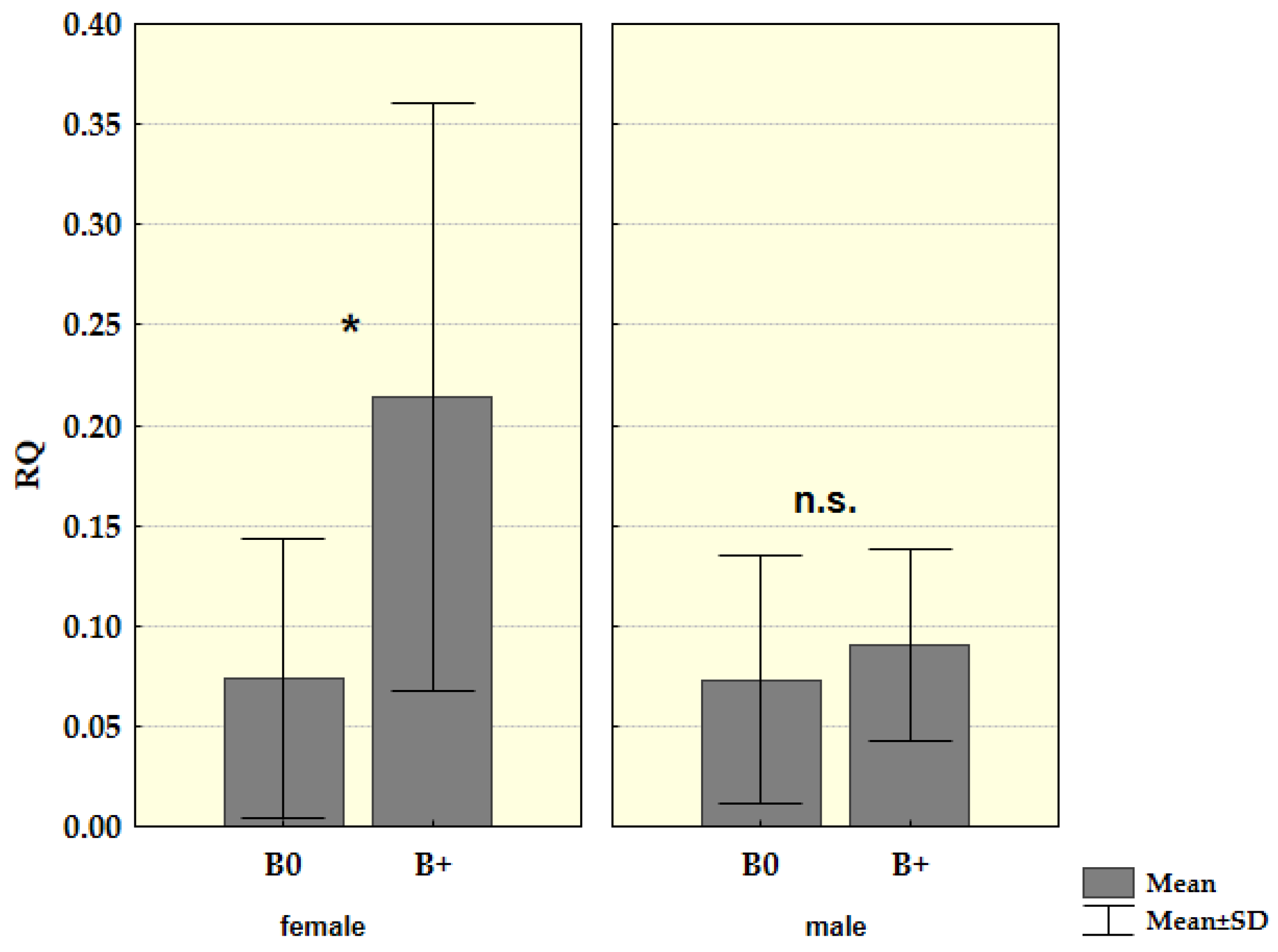
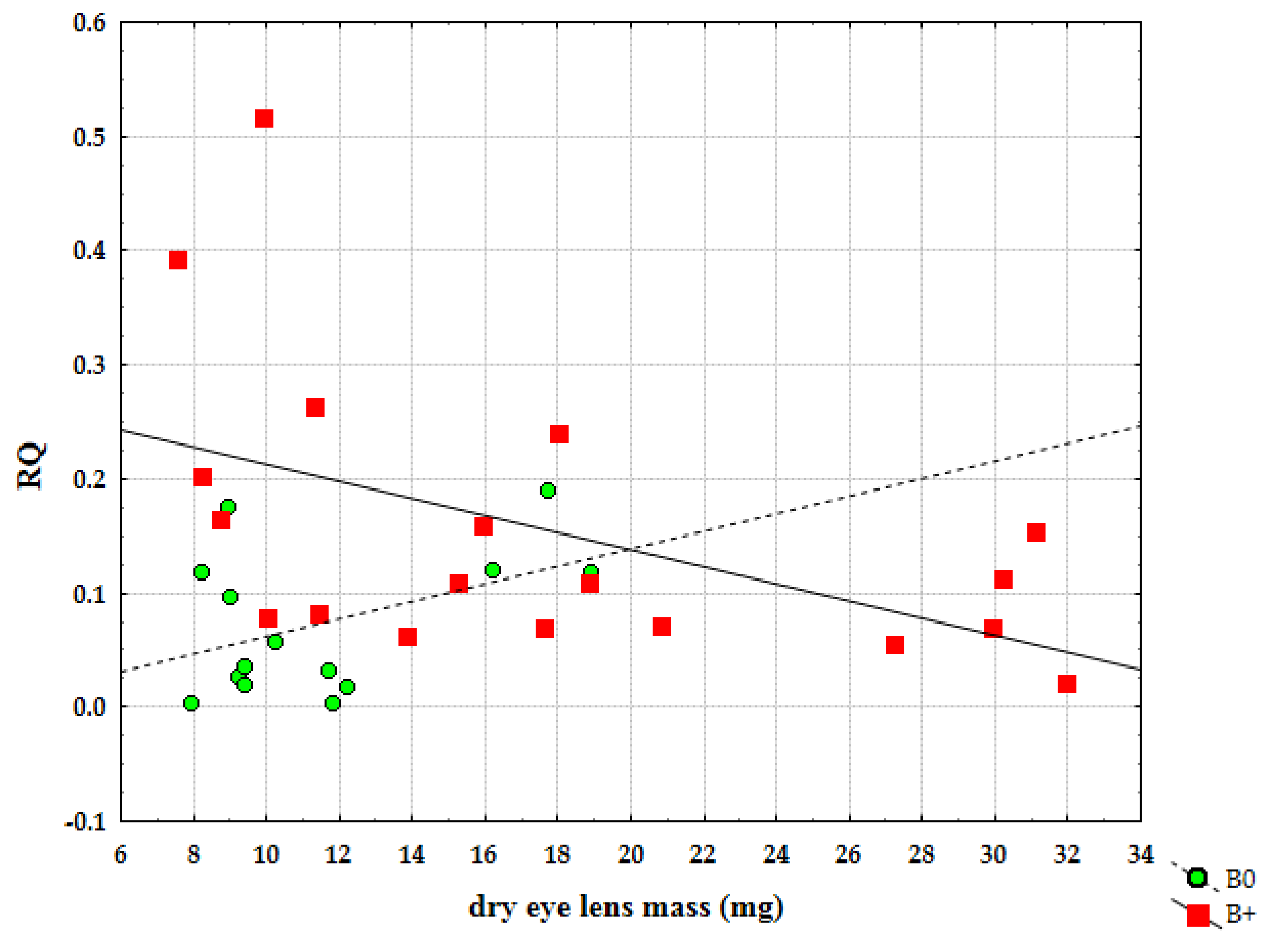
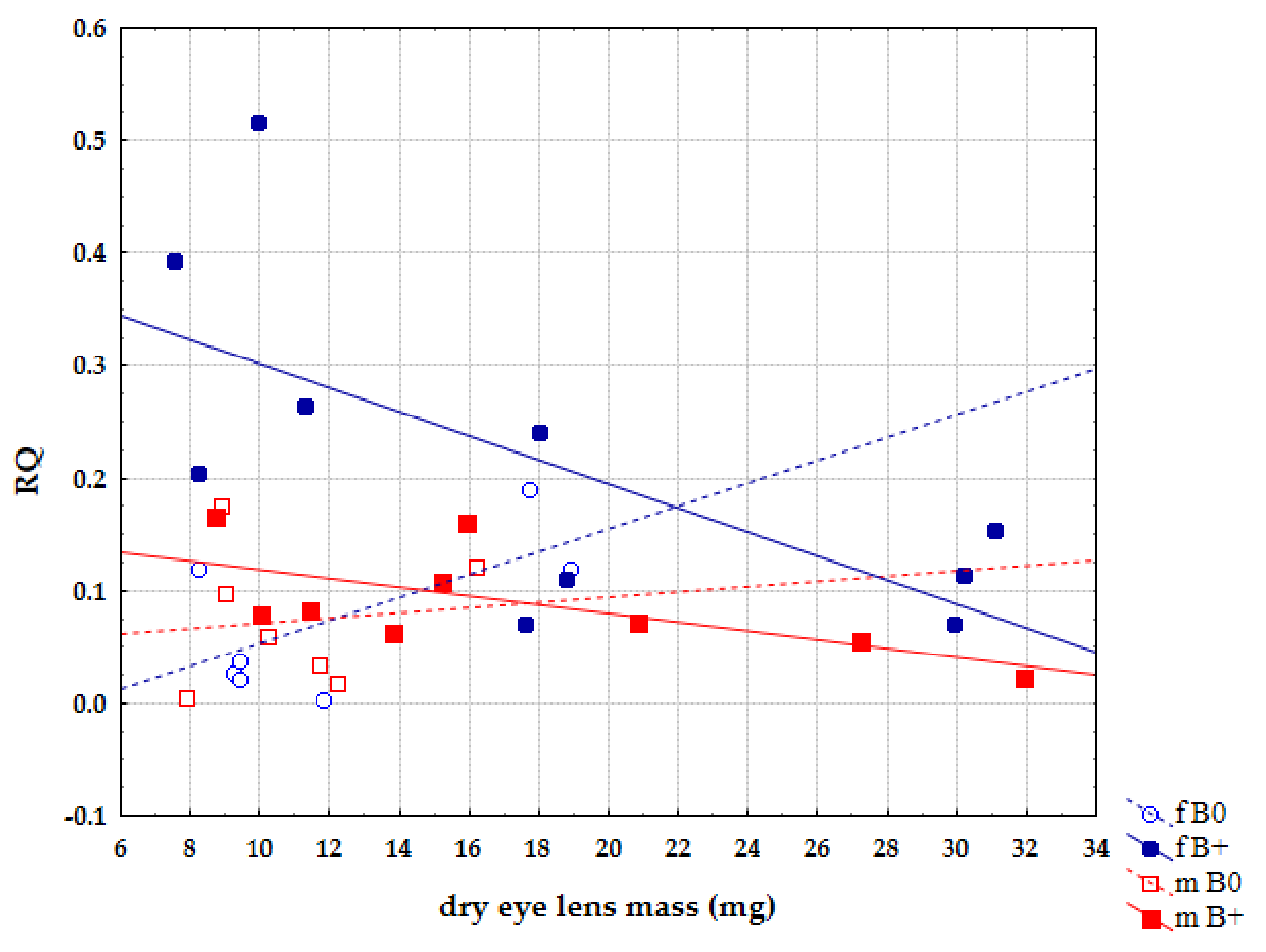
| Sample | Karyotype | Boody Mass (g) | Eyes Lens Mass (mg) | Locality |
|---|---|---|---|---|
| 3694 | 48,XX | 8.4 | 12.0 | IBISS 1 |
| 3695 | 48,XX,+1B | 8.1 | 11.3 | IBISS 1 |
| 3696 | 48,XY,+1B | 9.1 | 11.4 | IBISS 1 |
| 3698 | 48,XY | 9.9 | 11.7 | IBISS 1 |
| 3699 | 48,XY | 9.8 | 12.2 | IBISS 1 |
| 3796 | 48,XY,+1B | 19.8 | 13.8 | Bosilegrad |
| 3860 | 48,XX,+1B | 21.5 | 17.6 | Misača |
| 3907 | 48,XY,+1B | 21.1 | 15.2 | Ruski Krstur |
| 3913 | 48,XY,+1B | 21.4 | 15.9 | Ruski Krstur |
| 4056 | 48,XX,+1B | 18.2 | 9.9 | IBISS 1 |
| 4060 | 48,XX | 10.0 | 9.4 | IBISS 1 |
| 4061 | 48,XY | 9.1 | 8.9 | IBISS 1 |
| 4062 | 48,XY | 10.2 | 9.0 | IBISS 1 |
| 4063 | 48,XX | 10.3 | 9.2 | IBISS 1 |
| 4095 | 48,XX | 9.7 | 8.2 | Petnica |
| 4099 | 48,XX,+1B | 10.6 | 8.2 | IBISS 1 |
| 4112 | 48,XY,+1B | 11.8 | 8.7 | Vlasina |
| 4115 | 48,XY,+2B | 11.4 | 10.0 | Vlasina |
| 4133 | 48,XX,+1B | 12.4 | 7.5 | Babin zub |
| 4134 | 48,XY | 15.6 | 10.2 | Babin zub |
| 4148 | 48,XX,+1B | 32.0 | 29.9 | Petnica |
| 4150 | 48,XY,+1B | 40.6 | 31.9 | Petnica |
| 4157 | 48,XX,+2B | 28.0 | 30.2 | Petnica |
| 4172 | 48,XX | 21.2 | 17.7 | IBISS 1 |
| 4174 | 48,XX,+1B | 20.6 | 18.0 | IBISS 1 |
| 4183 | 48,XX,+2B | 31.5 | 31.1 | Maljen |
| 4195 | 48,XY | 17.9 | 16.2 | Goč |
| 4209 | 48,XY | 9.0 | 7.9 | Vlasina |
| 4216 | 48,XX | 19.6 | 18.9 | Vlasina |
| 4218 | 48,XX | 14.5 | 11.8 | Vlasina |
| 4224 | 48,XX,+2B | 17.9 | 20.8 | Vlasina |
| 4227 | 48,XX,+3B | 17.0 | 18.8 | Vlasina |
| 4229 | 48,XY,+2B | 23.7 | 20.8 | Vlasina |
| 4230 | 48,XY,+1B | 18.6 | 27.2 | Vlasina |
| Gene | Sequencies | |
|---|---|---|
| Rraga | F | 5′GCGGGACAACATCTTCTGTA3′ |
| R | 5′ATCTTTTTCCAGTTCGCGG3′ | |
| Cenpe | F | 5′GAGCCAAGGACTGGCATTAGA3′ |
| R | 5′TGCAGCTTCGATTTGCCTTG3′ | |
| Haus6 | F | 5′TTGGCTACAGGGCTCAGTTC3′ |
| R | 5′CTTCCAGAAGAAGTTGGCGA3′ | |
| Calnexin | F | 5′ATGGAAGGGAAGTGGTTACTGT3′ |
| R | 5′GCTTTGTAGGTGACCTTTGGAG3′ | |
| Pgk | F | 5′ATGTCGCTTTCCAACAAGCTG3′ |
| R | 5′GCTCCATTGTCCAAGCAGAAT3′ | |
| β-aktin | F | 5′TGGACATCCGCAAAGACCTGTAC3′ |
| R | 5′TCAGGAGGAGCAATGATCTTGA3′ |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Rajičić, M.; Makunin, A.; Adnađević, T.; Trifonov, V.; Vujošević, M.; Blagojević, J. B Chromosomes’ Sequences in Yellow-Necked Mice Apodemus flavicollis—Exploring the Transcription. Life 2022, 12, 50. https://doi.org/10.3390/life12010050
Rajičić M, Makunin A, Adnađević T, Trifonov V, Vujošević M, Blagojević J. B Chromosomes’ Sequences in Yellow-Necked Mice Apodemus flavicollis—Exploring the Transcription. Life. 2022; 12(1):50. https://doi.org/10.3390/life12010050
Chicago/Turabian StyleRajičić, Marija, Alexey Makunin, Tanja Adnađević, Vladimir Trifonov, Mladen Vujošević, and Jelena Blagojević. 2022. "B Chromosomes’ Sequences in Yellow-Necked Mice Apodemus flavicollis—Exploring the Transcription" Life 12, no. 1: 50. https://doi.org/10.3390/life12010050
APA StyleRajičić, M., Makunin, A., Adnađević, T., Trifonov, V., Vujošević, M., & Blagojević, J. (2022). B Chromosomes’ Sequences in Yellow-Necked Mice Apodemus flavicollis—Exploring the Transcription. Life, 12(1), 50. https://doi.org/10.3390/life12010050







