Transcriptional Analysis-Based Alterations Affecting Neuritogenesis of the Peripheral Nervous System in Psoriasis
Abstract
:1. Introduction
2. Materials and Methods
2.1. Criteria for Combining the Transcriptome Sequencing Data of from Three Published Psoriatic Datasets
2.2. RNA Sequencing Data Processing
2.3. Differential Expression Analysis
2.4. Functional Annotation, Enrichment Analysis, and Statistics
3. Results
3.1. Peripheral Nervous System-Associated Transcript Expression Alterations in Psoriasis
3.2. Differentially Expressed Transcripts Affecting Axon-Related Alterations in Non-Lesional and Lesional Psoriatic Skin
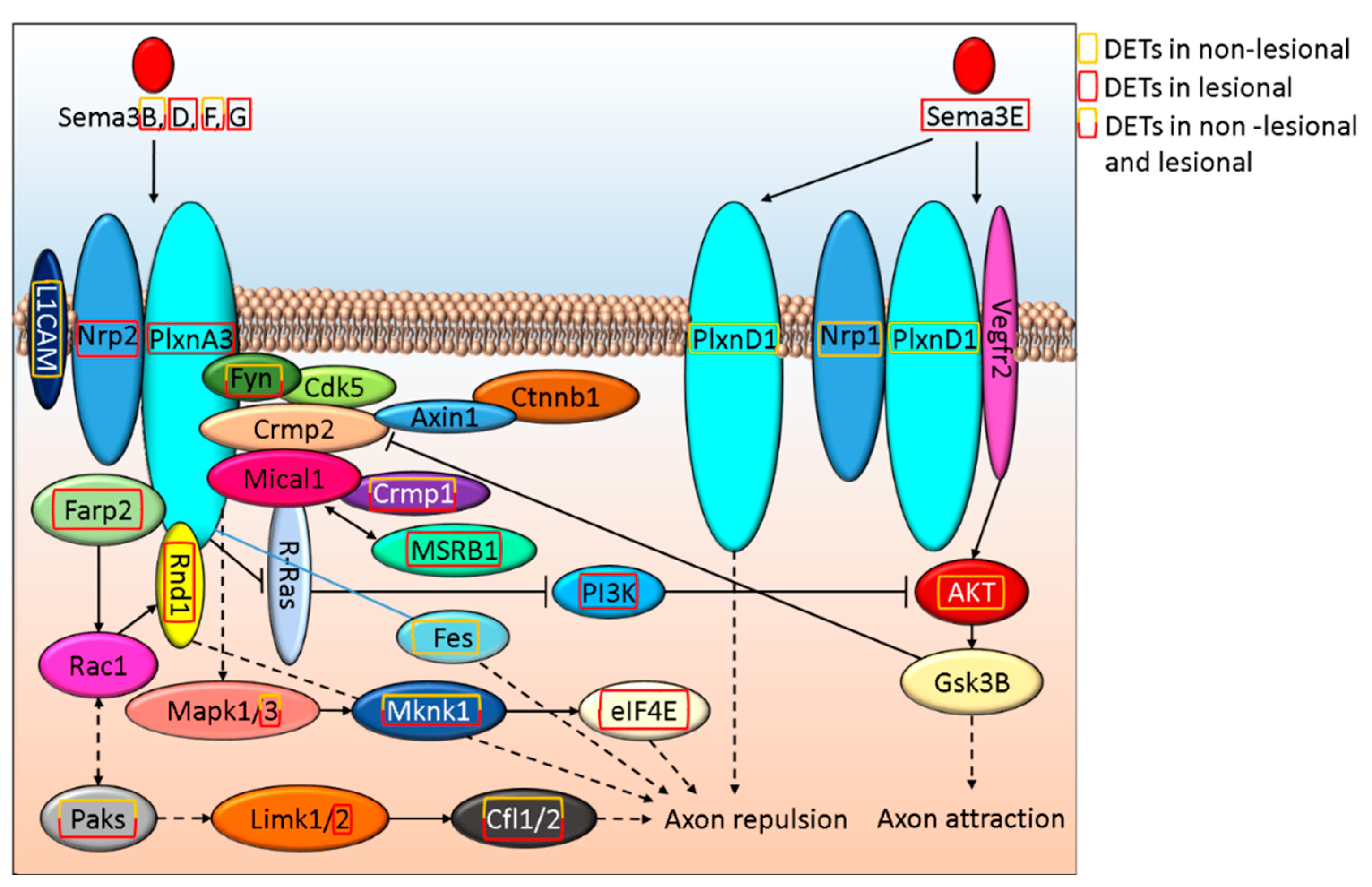
3.3. Semaphorin-Plexin Signaling, An Important Regulator of Axon Formation, Is Differentially Affected in Non-Lesional and Lesional Psoriatic Skin
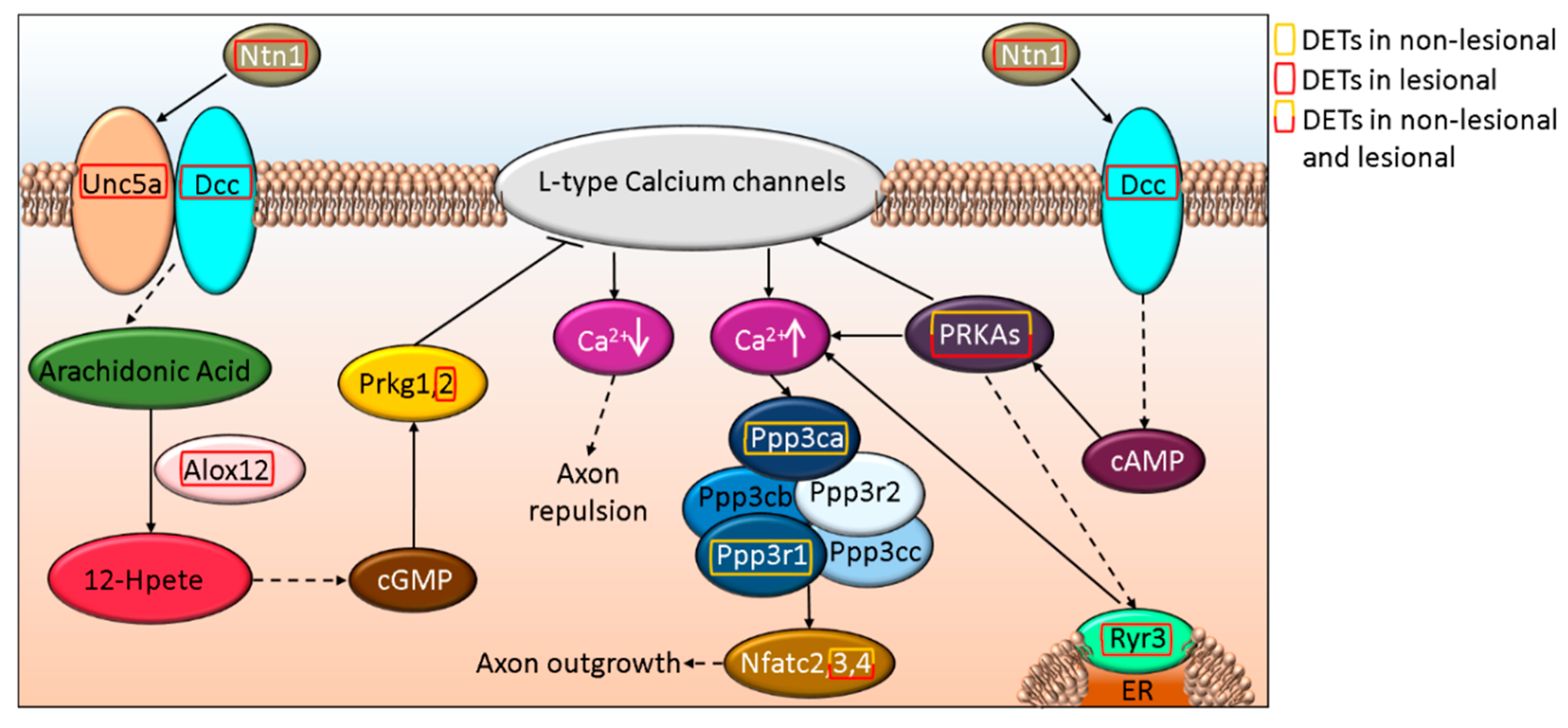
3.4. ROBO-DCC-UNC5 Signaling Regulates Axon Formation and Differentially Affected in Non-Lesional and Lesional Psoriatic Skin
3.5. Disturbed WNT5A Signaling Potentially Affect Cutaneous Axon Growth in Psoriasis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Chiricozzi, A.; Romanelli, P.; Volpe, E.; Borsellino, G.; Romanelli, M. Scanning the Immunopathogenesis of Psoriasis. Int. J. Mol. Sci. 2018, 19, 179. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Gubán, B.; Vas, K.; Balog, Z.; Manczinger, M.; Bebes, A.; Groma, G.; Szell, M.; Kemény, L.; Batacsorgő, Z. Abnormal regulation of fibronectin production by fibroblasts in psoriasis. Br. J. Dermatol. 2015, 174, 533–541. [Google Scholar] [CrossRef] [Green Version]
- Szlavicz, E.; Szabo, K.; Groma, G.; Bata-Csorgo, Z.; Pagani, F.; Kemény, L.; Szell, M. Splicing factors differentially expressed in psoriasis alter mRNA maturation of disease-associated EDA+ fibronectin. Mol. Cell. Biochem. 2017, 436, 189–199. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Eyre, R.W.; Krueger, G.G. Response to injury of skin involved and uninvolved with psoriasis, and its relation to disease activity: Koebner and’reverse’Koebner reactions. Br. J. Dermatol. 1982, 106, 153–159. [Google Scholar] [CrossRef]
- Gudjonsson, J.E.; Ding, J.; Li, X.; Nair, R.P.; Tejasvi, T.; Qin, Z.; Ghosh, D.; Aphale, A.; Gumucio, D.L.; Voorhees, J.J.; et al. Global Gene Expression Analysis Reveals Evidence for Decreased Lipid Biosynthesis and Increased Innate Immunity in Uninvolved Psoriatic Skin. J. Investig. Dermatol. 2009, 129, 2795–2804. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Szabó, K.; Bata-Csörgő, Z.; Dallos, A.; Bebes, A.; Francziszti, L.; Dobozy, A.; Kemény, L.; Széll, M. Regulatory Networks Contributing to Psoriasis Susceptibility. Acta Derm. Venereol. 2014, 94, 380–385. [Google Scholar] [CrossRef] [Green Version]
- Oh, S.; Chung, H.; Chang, S.; Lee, S.-H.; Seok, S.H.; Lee, H. Effect of Mechanical Stretch on the DNCB-induced Proinflammatory Cytokine Secretion in Human Keratinocytes. Sci. Rep. 2019, 9, 5156. [Google Scholar] [CrossRef]
- Baumbauer, K.M.; DeBerry, J.; Adelman, P.; Miller, R.H.; Hachisuka, J.; Lee, K.H.; Ross, S.E.; Koerber, H.R.; Davis, B.; Albers, K. Keratinocytes can modulate and directly initiate nociceptive responses. eLife 2015, 4, 09674. [Google Scholar] [CrossRef] [PubMed]
- Yosipovitch, G.; Chan, Y.; Tay, Y.; Goh, C. Thermosensory abnormalities and blood flow dysfunction in psoriatic skin. Br. J. Dermatol. 2003, 149, 492–497. [Google Scholar] [CrossRef]
- Sulzberger, M.B. BUGS. Arch. Dermatol. 1971, 104, 220. [Google Scholar] [CrossRef]
- Weiner, S.R.; Bassett, L.W.; Reichman, R.P. Protective effect of poliomyelitis on psoriatic arthritis. Arthritis Rheum. 1985, 28, 703–706. [Google Scholar] [CrossRef]
- Farber, E.M.; Lanigan, S.W.; Boer, J. The Role of Cutaneous Sensory Nerves in the Maintenance of Psoriasis. Int. J. Dermatol. 1990, 29, 418–420. [Google Scholar] [CrossRef] [PubMed]
- Raychaudhuri, S.P.; Farber, E.M. Are sensory nerves essential for the development of psoriatic lesions? J. Am. Acad. Dermatol. 1993, 28, 488–489. [Google Scholar] [CrossRef]
- Joseph, T.; Kurian, J.; Warwick, D.; Friedmann, P. Unilateral remission of psoriasis following traumatic nerve palsy. Br. J. Dermatol. 2005, 152, 185–186. [Google Scholar] [PubMed]
- Kane, D.; Lockhart, J.C.; Balint, P.V.; Mann, C.; Ferrell, W.R.; McInnes, I. Protective effect of sensory denervation in inflammatory arthritis (evidence of regulatory neuroimmune pathways in the arthritic joint). Ann. Rheum. Dis. 2005, 64, 325–327. [Google Scholar] [CrossRef] [Green Version]
- Kecici, A.S.; Göktay, F.; Tutkavul, K.; Günes, P.; Yasar, S. Unilateral improvement of nail psoriasis with denervation injury. Clin. Exp. Dermatol. 2018, 43, 339–341. [Google Scholar] [CrossRef]
- Sowell, J.K.; Pippenger, M.A.; Crowe, M.J. Psoriasis Contralateral to Hemiparesis Following Cerebrovascular Accident. Int. J. Dermatol. 1993, 32, 598–599. [Google Scholar] [CrossRef]
- Veale, D.; Farrell, M.; FitzGerald, O. Mechanism of joint sparing in a patient with unilateral psoriatic arthritis and a longstanding hemiplegia. Rheumatology 1993, 32, 413–416. [Google Scholar] [CrossRef]
- Stratigos, A.J.; Katoulis, A.K.; Stavrianeas, N.G. Spontaneous clearing of psoriasis after stroke. J. Am. Acad. Dermatol. 1998, 38, 768–770. [Google Scholar] [CrossRef]
- Qin, B.; Sun, C.; Chen, L.; Wang, S.; Yang, J.; Xie, Z.; Shen, Z. The nerve injuries attenuate the persistence of psoriatic lesions. J. Dermatol. Sci. 2021, 102, 85–93. [Google Scholar] [CrossRef]
- Zhu, T.H.; Nakamura, M.; Farahnik, B.; Abrouk, M.; Lee, K.; Singh, R.; Gevorgyan, A.; Koo, J.; Bhutani, T. The Role of the Nervous System in the Pathophysiology of Psoriasis: A Review of Cases of Psoriasis Remission or Improvement Following Denervation Injury. Am. J. Clin. Dermatol. 2016, 17, 257–263. [Google Scholar] [CrossRef]
- González, C.; Franco, M.; Londoño, A.; Valenzuela, F. Breaking paradigms in the treatment of psoriasis: Use of botulinum toxin for the treatment of plaque psoriasis. Dermatol. Ther. 2020, 33, 14319. [Google Scholar] [CrossRef]
- Aschenbeck, K.A.; Hordinsky, M.K.; Kennedy, W.R.; Wendelschafer-Crabb, G.; Ericson, M.E.; Kavand, S.; Bertin, A.; Dykstra, D.D.; Panoutsopoulou, I.G. Neuromodulatory treatment of recalcitrant plaque psoriasis with onabotulinumtoxinA. J. Am. Acad. Dermatol. 2018, 79, 1156–1159. [Google Scholar] [CrossRef] [Green Version]
- Amalia, S.N.; Uchiyama, A.; Baral, H.; Inoue, Y.; Yamazaki, S.; Fujiwara, C.; Sekiguchi, A.; Yokoyama, Y.; Ogino, S.; Torii, R.; et al. Suppression of neuropeptide by botulinum toxin improves imiquimod-induced psoriasis-like dermatitis via the regulation of neuroimmune system. J. Dermatol. Sci. 2021, 101, 58–68. [Google Scholar] [CrossRef]
- Farber, E.M.; Nickoloff, B.J.; Recht, B.; Fraki, J.E. Stress, symmetry, and psoriasis: Possible role of neuropeptides. J. Am. Acad. Dermatol. 1986, 14, 305–311. [Google Scholar] [CrossRef]
- Saraceno, R.; Kleyn, C.E.; Terenghi, G.; Griffiths, C.E.M. The role of neuropeptides in psoriasis. Br. J. Dermatol. 2006, 155, 876–882. [Google Scholar] [CrossRef]
- Legat, F.J.; Griesbacher, T.; Schicho, R.; Althuber, P.; Schuligoi, R.; Kerl, H.; Wolf, P. Repeated subinflammatory ultraviolet B irradiation increases substance P and calcitonin gene-related peptide content and augments mustard oil-induced neurogenic inflammation in the skin of rats. Neurosci. Lett. 2002, 329, 309–313. [Google Scholar] [CrossRef]
- Dias, M.P.; Newton, D.J.; McLeod, G.A.; Belch, J.J.F.; Khan, F. Vasoactive properties of calcitonin gene-related peptide in Human Skin. Int. Angiol. 2011, 30, 424–428. [Google Scholar] [PubMed]
- Raychaudhuri, S.P.; Raychaudhuri, S.K. Role of NGF and neurogenic inflammation in the pathogenesis of psoriasis. Prog. Brain Res. 2004, 146, 433–437. [Google Scholar]
- Glinski, W.; Brodecka, H.; Glinska-Ferenz, M.; Kowalski, D. Neuropeptides in psoriasis: Possible role of beta-endorphin in the pathomechanism of the disease. Int. J. Dermatol. 1994, 33, 356–360. [Google Scholar] [CrossRef] [PubMed]
- Naukkarinen, A.; Nickoloff, B.J.; Farber, E.M. Quantification of Cutaneous Sensory Nerves and Their Substance P Content in Psoriasis. J. Investig. Dermatol. 1989, 92, 126–129. [Google Scholar] [CrossRef] [Green Version]
- Siiskonen, H.; Harvima, I. Mast Cells and Sensory Nerves Contribute to Neurogenic Inflammation and Pruritus in Chronic Skin Inflammation. Front. Cell. Neurosci. 2019, 13, 422. [Google Scholar] [CrossRef]
- Choi, J.E.; Di Nardo, A. Skin neurogenic inflammation. Semin. Immunopathol. 2018, 40, 249–259. [Google Scholar] [CrossRef]
- Szepietowski, J.C.; Reich, A. Itch in Psoriasis Management. Diagnosis and Therapy of Tattoo Complications. Itch Manag. Clin. Pract. 2016, 50, 102–110. [Google Scholar]
- Szepietowski, J.; Reich, A. Pruritus in psoriasis: An update. Eur. J. Pain 2015, 20, 41–46. [Google Scholar] [CrossRef] [PubMed]
- Tsianakas, A.; Mrowietz, U. Pruritus bei Psoriasis. Der Hautarzt 2016, 67, 601–605. [Google Scholar] [CrossRef]
- Arck, P.; Paus, R. From the Brain-Skin Connection: The Neuroendocrine-Immune Misalliance of Stress and Itch. Neuroimmunomodulation 2006, 13, 347–356. [Google Scholar] [CrossRef]
- Leon, A.; Rosen, J.D.; Hashimoto, T.; Fostini, A.C.; Paus, R.; Yosipovitch, G. Itching for an answer: A review of potential mechanisms of scalp itch in psoriasis. Exp. Dermatol. 2019, 28, 1397–1404. [Google Scholar] [CrossRef] [Green Version]
- Pithadia, D.J.; Reynolds, K.A.; Lee, E.B.; Wu, J.J. Psoriasis-associated cutaneous pain: Etiology, assessment, impact, and management. J. Dermatol. Treat. 2018, 30, 435–440. [Google Scholar] [CrossRef]
- Li, Y.; Lin, P.; Wang, S.; Li, S.; Wang, R.; Yang, L.; Wang, H. Quantitative analysis of differentially expressed proteins in psoriasis vulgaris using tandem mass tags and parallel reaction monitoring. Clin. Proteom. 2020, 17, 30. [Google Scholar] [CrossRef] [PubMed]
- Szél, E.; Bozó, R.; Hunyadi-Gulyás, É.; Manczinger, M.; Szabó, K.; Kemény, L.; Bata-Csörgő, Z.; Groma, G. Comprehensive Proteomic Analysis Reveals Intermediate Stage of Non-Lesional Psoriatic Skin and Points out the Importance of Proteins Outside this Trend. Sci. Rep. 2019, 9, 1382. [Google Scholar] [CrossRef]
- Manczinger, M.; Kemeny, L. Novel Factors in the Pathogenesis of Psoriasis and Potential Drug Candidates Are Found with Systems Biology Approach. PLoS ONE 2013, 8, e80751. [Google Scholar] [CrossRef] [Green Version]
- Li, B.; Tsoi, L.C.; Swindell, W.R.; Gudjonsson, J.E.; Tejasvi, T.; Johnston, A.; Ding, J.; Stuart, P.E.; Xing, X.; Kochkodan, J.J.; et al. Transcriptome Analysis of Psoriasis in a Large Case–Control Sample: RNA-Seq Provides Insights into Disease Mechanisms. J. Investig. Dermatol. 2014, 134, 1828–1838. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tsoi, L.C.; Iyer, M.K.; Stuart, P.E.; Swindell, W.R.; Gudjonsson, J.E.; Tejasvi, T.; Sarkar, M.K.; Li, B.; Ding, J.; Voorhees, J.J.; et al. Analysis of long non-coding RNAs highlights tissue-specific expression patterns and epigenetic profiles in normal and psoriatic skin. Genome Biol. 2015, 16, 24. [Google Scholar] [CrossRef] [Green Version]
- Tsoi, L.C.; Stuart, P.E.; Tian, C.; Gudjonsson, J.E.; Das, S.; Zawistowski, M.; Ellinghaus, E.; Barker, J.N.; Chandran, V.; Dand, N.; et al. Large scale meta-analysis characterizes genetic architecture for common psoriasis associated variants. Nat. Commun. 2017, 8, 15382. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, F.; Wang, W.; Shen, C.; Li, H.; Zuo, X.; Zheng, X.; Yue, M.; Zhang, C.; Yu, L.; Chen, M.; et al. Epigenome-Wide Association Analysis Identified Nine Skin DNA Methylation Loci for Psoriasis. J. Investig. Dermatol. 2016, 136, 779–787. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Calautti, E.; Avalle, L.; Poli, V. Psoriasis: A STAT3-Centric View. Int. J. Mol. Sci. 2018, 19, 171. [Google Scholar] [CrossRef] [Green Version]
- Liang, Y.; Tsoi, L.C.; Xing, X.; Beamer, M.A.; Swindell, W.R.; Sarkar, M.; Berthier, C.C.; Stuart, P.E.; Harms, P.W.; Nair, R.P.; et al. A gene network regulated by the transcription factor VGLL3 as a promoter of sex-biased autoimmune diseases. Nat. Immunol. 2017, 18, 152–160. [Google Scholar] [CrossRef] [Green Version]
- Sahoo, P.; Smith, D.S.; Perrone-Bizzozero, N.; Twiss, J.L. Axonal mRNA transport and translation at a glance. J. Cell Sci. 2018, 131, jcs196808. [Google Scholar] [CrossRef] [Green Version]
- Costa, I.D.; Buchanan, C.N.; Zdradzinski, M.D.; Sahoo, P.K.; Smith, T.P.; Thames, E.; Kar, A.N.; Twiss, J.L. The functional organization of axonal mRNA transport and translation. Nat. Rev. Neurosci. 2021, 22, 77–91. [Google Scholar] [CrossRef]
- Bray, N.L.; Pimentel, H.; Melsted, P.; Pachter, H.P.L. Near-optimal probabilistic RNA-seq quantification. Nat. Biotechnol. 2016, 34, 525–527. [Google Scholar] [CrossRef]
- Harrow, J.; Frankish, A.; Gonzalez, J.M.; Tapanari, E.; Diekhans, M.; Kokocinski, F.; Aken, B.L.; Barrell, D.; Zadissa, A.; Searle, S.; et al. Gencode: The reference human genome annotation for The ENCODE Project. Genome Res. 2012, 22, 1760–1774. [Google Scholar] [CrossRef] [Green Version]
- Soneson, C.; Love, M.I.; Robinson, M.D. Differential analyses for RNA-seq: Transcript-level estimates improve gene-level inferences. F1000Research 2016, 4, 1521. [Google Scholar] [CrossRef]
- McCarthy, D.J.; Chen, Y.; Smyth, G.K. Differential expression analysis of multifactor RNA-Seq experiments with respect to biological variation. Nucleic Acids Res. 2012, 40, 4288–4297. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Schwämmle, V.; León, I.R.; Jensen, O.N. Assessment and Improvement of Statistical Tools for Comparative Proteomics Analysis of Sparse Data Sets with Few Experimental Replicates. J. Proteome Res. 2013, 12, 3874–3883. [Google Scholar] [CrossRef]
- Ritchie, M.; Phipson, B.; Wu, D.; Hu, Y.; Law, C.W.; Shi, W.; Smyth, G.K. limma powers differential expression analyses for RNA-sequencing and microarray studies. Nucleic Acids Res. 2015, 43, e47. [Google Scholar] [CrossRef] [PubMed]
- Liu, R.; Holik, A.Z.; Su, S.; Jansz, N.; Chen, K.; Leong, H.S.; Blewitt, M.; Asselin-Labat, M.-L.; Smyth, G.; Ritchie, M.E. Why weight? Modelling sample and observational level variability improves power in RNA-seq analyses. Nucleic Acids Res. 2015, 43, e97. [Google Scholar] [CrossRef]
- Berchtold, W. Comparison of the Kastenbaum-Bowman test and Fisher’s exact test. Archiv fur Genetik 1975, 48, 151–157. [Google Scholar]
- Eden, E.; Navon, R.; Steinfeld, I.; Lipson, D.; Yakhini, Z. GOrilla: A tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC Bioinform. 2009, 10, 48. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ferreira, J.A. The Benjamini-Hochberg method in the case of discrete test statistics. Int. J. Biostat. 2007, 3, 11. [Google Scholar] [CrossRef] [Green Version]
- Zhang, H.-L.; Wang, J.; Tang, L. Sema4D Knockdown in Oligodendrocytes Promotes Functional Recovery After Spinal Cord Injury. Cell Biophys. 2013, 68, 489–496. [Google Scholar] [CrossRef]
- Furue, K.; Ito, T.; Tsuji, G.; Kadono, T.; Furue, M. Psoriasis and the TNF/IL23/IL17 axis. Ital. J. Dermatol. Venereol. 2019, 154, 418–424. [Google Scholar] [CrossRef]
- Riol-Blanco, L.; Ordovas-Montanes, J.; Perro, M.; Naval, E.; Thiriot, A.; Alvarez, D.; Paust, S.; Wood, J.N.; Von Andrian, U.H. Nociceptive sensory neurons drive interleukin-23-mediated psoriasiform skin inflammation. Nat. Cell Biol. 2014, 510, 157–161. [Google Scholar] [CrossRef] [PubMed]
- Dattola, A.; Silvestri, M.; Tamburi, F.; Amoruso, G.F.; Bennardo, L.; Nisticò, S.P. Emerging role of anti-IL23 in the treatment of psoriasis: When humanized is very promising. Dermatol. Ther. 2020, 33, e14504. [Google Scholar] [CrossRef]
- Amoruso, G.; Nisticò, S.; Iannone, L.; Russo, E.; Rago, G.; Patruno, C.; Bennardo, L. Ixekizumab May Improve Renal Function in Psoriasis. Health 2021, 9, 543. [Google Scholar] [CrossRef] [PubMed]
- Worzfeld, T.; Offermanns, S. Semaphorins and plexins as therapeutic targets. Nat. Rev. Drug Discov. 2014, 13, 603–621. [Google Scholar] [CrossRef] [PubMed]
- Adams, R.H.; Eichmann, A. Axon Guidance Molecules in Vascular Patterning. Cold Spring Harb. Perspect. Biol. 2010, 2, a001875. [Google Scholar] [CrossRef] [Green Version]
- Suzuki, K.; Kumanogoh, A.; Kikutani, H. Semaphorins and their receptors in immune cell interactions. Nat. Immunol. 2007, 9, 17–23. [Google Scholar] [CrossRef] [PubMed]
- Kumanogoh, A.; Kikutani, H. Immunological functions of the neuropilins and plexins as receptors for semaphorins. Nat. Rev. Immunol. 2013, 13, 802–814. [Google Scholar] [CrossRef] [PubMed]
- Yoshida, Y. Semaphorin Signaling in Vertebrate Neural Circuit Assembly. Front. Mol. Neurosci. 2012, 5, 71. [Google Scholar] [CrossRef] [Green Version]
- Messina, A.; Ferraris, N.; Wray, S.; Cagnoni, G.; Donohue, D.E.; Casoni, F.; Kramer, P.R.; Derijck, A.A.; Adolfs, Y.; Fasolo, A.; et al. Dysregulation of Semaphorin7A/β1-integrin signaling leads to defective GnRH-1 cell migration, abnormal gonadal development and altered fertility. Hum. Mol. Genet. 2011, 20, 4759–4774. [Google Scholar] [CrossRef] [Green Version]
- Giacobini, P.; Prevot, V. Semaphorins in the development, homeostasis and disease of hormone systems. Semin. Cell Dev. Biol. 2013, 24, 190–198. [Google Scholar] [CrossRef] [PubMed]
- Kanth, S.M.; Gairhe, S.; Torabi-Parizi, P. The Role of Semaphorins and Their Receptors in Innate Immune Responses and Clinical Diseases of Acute Inflammation. Front. Immunol. 2021, 12, 1610. [Google Scholar] [CrossRef] [PubMed]
- Sabag, A.D.; Dias-Polak, D.; Bejar, J.; Sheffer, H.; Bergman, R.; Vadasz, Z. Altered expression of regulatory molecules in the skin of psoriasis. Immunol. Res. 2018, 66, 649–654. [Google Scholar] [CrossRef] [PubMed]
- Zhang, C.; Xiao, C.; Dang, E.; Cao, J.; Zhu, Z.; Fu, M.; Yao, X.; Liu, Y.; Jin, B.; Wang, G.; et al. CD100–Plexin-B2 Promotes the Inflammation in Psoriasis by Activating NF-κB and the Inflammasome in Keratinocytes. J. Investig. Dermatol. 2018, 138, 375–383. [Google Scholar] [CrossRef] [Green Version]
- Ryu, S.; Broussard, L.; Youn, C.; Song, B.; Norris, D.; Armstrong, C.A.; Kim, B.; Song, P.I. Therapeutic Effects of Synthetic Antimicrobial Peptides, TRAIL and NRP1 Blocking Peptides in Psoriatic Keratinocytes. Chonnam. Med. J. 2019, 55, 75–85. [Google Scholar] [CrossRef] [PubMed]
- Casper, M. 3-Dimensional Imaging of Cutaneous Nerve Endings. J. Investig. Dermatol. 2019, 139, 999–1001. [Google Scholar] [CrossRef] [Green Version]
- Julien, F.; Bechara, A.; Fiore, R.; Nawabi, H.; Zhou, H.; Hoyo-Becerra, C.; Bozon, M.; Rougon, G.; Grumet, M.; Püschel, A.W.; et al. Dual Functional Activity of Semaphorin 3B Is Required for Positioning the Anterior Commissure. Neuron 2005, 48, 63–75. [Google Scholar] [CrossRef]
- Chauvet, S.; Cohen, S.; Yoshida, Y.; Fekrane, L.; Livet, J.; Gayet, O.; Segu, L.; Buhot, M.-C.; Jessell, T.M.; Henderson, C.; et al. Gating of Sema3E/PlexinD1 Signaling by Neuropilin-1 Switches Axonal Repulsion to Attraction during Brain Development. Neuron 2007, 56, 807–822. [Google Scholar] [CrossRef] [Green Version]
- Bellon, A.; Luchino, J.; Haigh, K.; Rougon, G.; Haigh, J.; Chauvet, S.; Mann, F. VEGFR2 (KDR/Flk1) Signaling Mediates Axon Growth in Response to Semaphorin 3E in the Developing Brain. Neuron 2010, 66, 205–219. [Google Scholar] [CrossRef] [Green Version]
- Liu, Y.; Halloran, M.C. Central and Peripheral Axon Branches from One Neuron Are Guided Differentially by Semaphorin3D and Transient Axonal Glycoprotein-1. J. Neurosci. 2005, 25, 10556–10563. [Google Scholar] [CrossRef] [Green Version]
- Simona, M.; Antonio, P.; Massimo, B.; Alfonso, C. Neuronal Semaphorins Regulate a Primary Immune Response. Curr. Neurovasc. Res. 2006, 3, 295–305. [Google Scholar]
- Siems, S.B.; Jahn, O.; Eichel, M.; Kannaiyan, N.; Wu, L.M.N.; Sherman, D.L.; Kusch, K.; Hesse, D.; Jung, R.B.; Fledrich, R.; et al. Proteome profile of peripheral myelin in healthy mice and in a neuropathy model. eLife 2020, 9, 51406. [Google Scholar] [CrossRef]
- Kalpachidou, T.; Spiecker, L.; Kress, M.; Quarta, S. Rho GTPases in the Physiology and Pathophysiology of Peripheral Sensory Neurons. Cells 2019, 8, 591. [Google Scholar] [CrossRef] [Green Version]
- Carr, L.; Parkinson, D.B.; Dun, X.-P. Expression patterns of Slit and Robo family members in adult mouse spinal cord and peripheral nervous system. PLoS ONE 2017, 12, e0172736. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dun, X.-P.; Parkinson, D.B. Role of Netrin-1 Signaling in Nerve Regeneration. Int. J. Mol. Sci. 2017, 18, 491. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Boneschansker, L.; Nakayama, H.; Eisenga, M.; Wedel, J.; Klagsbrun, M.; Irimia, D.; Briscoe, D.M. Netrin-1 Augments Chemokinesis in CD4+ T Cells In Vitro and Elicits a Proinflammatory Response In Vivo. J. Immunol. 2016, 197, 1389–1398. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nakatsuji, Y.; Okuno, T.; Moriya, M.; Sugimoto, T.; Kinoshita, M.; Takamatsu, H.; Nojima, S.; Kimura, T.; Kang, S.; Ito, D.; et al. Elevation of Sema4A Implicates Th Cell Skewing and the Efficacy of IFN-β Therapy in Multiple Sclerosis. J. Immunol. 2012, 188, 4858–4865. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Koda, T.; Namba, A.; Kinoshita, M.; Nakatsuji, Y.; Sugimoto, T.; Sakakibara, K.; Tada, S.; Shimizu, M.; Yamashita, K.; Takata, K.; et al. Sema4A is implicated in the acceleration of Th17 cell-mediated neuroinflammation in the effector phase. J. Neuroinflamm. 2020, 17, 82–89. [Google Scholar] [CrossRef] [PubMed]
- Burkhardt, C.; Müller, M.; Badde, A.; Garner, C.C.; Gundelfinger, E.D.; Püschel, A.W. Semaphorin 4B interacts with the post-synaptic density protein PSD-95/SAP90 and is recruited to synapses through a C-terminal PDZ-binding motif. FEBS Lett. 2005, 579, 3821–3828. [Google Scholar] [CrossRef] [Green Version]
- Nakagawa, Y.; Takamatsu, H.; Okuno, T.; Kang, S.; Nojima, S.; Kimura, T.; Kataoka, T.R.; Ikawa, M.; Toyofuku, T.; Katayama, I.; et al. Identification of Semaphorin 4B as a Negative Regulator of Basophil-Mediated Immune Responses. J. Immunol. 2011, 186, 2881–2888. [Google Scholar] [CrossRef] [Green Version]
- Zhu, K.; Ye, J.; Wu, M.; Cheng, H. Expression of Th1 and Th2 cytokine-associated transcription factors, T-bet and GATA-3, in peripheral blood mononuclear cells and skin lesions of patients with psoriasis vulgaris. Arch. Dermatol. Res. 2010, 302, 517–523. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Wang, B.; Zou, M.; Li, J.; Lü, G.; Zhang, Q.; Liu, F.; Lu, C. CircSEMA4B targets miR-431 modulating IL-1β-induced degradative changes in nucleus pulposus cells in intervertebral disc degeneration via Wnt pathway. Biochim. Biophys. Acta BBA Mol. Basis Dis. 2018, 1864, 3754–3768. [Google Scholar] [CrossRef] [PubMed]
- Romanowska, M.; Evans, A.; Kellock, D.; Bray, S.E.; McLean, K.; Donandt, S.; Foerster, J. Wnt5a Exhibits Layer-Specific Expression in Adult Skin, Is Upregulated in Psoriasis, and Synergizes with Type 1 Interferon. PLoS ONE 2009, 4, e5354. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Clark, C.E.J.; Liu, Y.; Cooper, H.M. The Yin and Yang of Wnt/Ryk axon guidance in development and regeneration. Sci. China Life Sci. 2014, 57, 366–371. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ghosh, M.C.; Collins, G.D.; Vandanmagsar, B.; Patel, K.; Brill, M.; Carter, A.; Lustig, A.; Becker, K.G.; Wood, W.W.; Emeche, C.D.; et al. Activation of Wnt5A signaling is required for CXC chemokine ligand 12–mediated T-cell migration. Blood 2009, 114, 1366–1373. [Google Scholar] [CrossRef]
- Zgraggen, S.; Huggenberger, R.; Kerl, K.; Detmar, M. An Important Role of the SDF-1/CXCR4 Axis in Chronic Skin In-flammation. PLoS ONE 2014, 9, e93665. [Google Scholar]
- Winge, M.C.; Ohyama, B.; Dey, C.N.; Boxer, L.; Li, W.; Ehsani-Chimeh, N.; Truong, A.K.; Wu, D.; Armstrong, A.W.; Makino, T.; et al. RAC1 activation drives pathologic interactions between the epidermis and immune cells. J. Clin. Investig. 2016, 126, 2661–2677. [Google Scholar] [CrossRef]



| Categories | Functions | Comparison | p-Value | Number of Molecules |
|---|---|---|---|---|
| Nervous System Development and Function | Morphology of nervous system | NL vs. H | 4.11E-17 | 236 |
| L vs. H | 5.28E-32 | 637 | ||
| Nervous System Development and Function, Neurological Disease | Abnormal morphology of nervous system | NL vs. H | 4.95E-13 | 188 |
| L vs. H | 2.80E-20 | 495 | ||
| Nervous System Development and Function, Tissue Morphology | Morphology of nervous tissue | NL vs. H | 1.11E-12 | 165 |
| L vs. H | 5.25E-22 | 439 | ||
| Nervous System Development and Function, Organismal Development, Tissue Development | Morphogenesis of nervous tissue | NL vs. H | 4.70E-10 | 144 |
| L vs. H | 4.46E-22 | 405 | ||
| Cell Morphology, Cellular Assembly and Organization, Cellular Development, Cellular Function and Maintenance, Cellular Growth and Proliferation, Nervous System Development and Function, Organismal Development, Tissue Development | Neuritogenesis | NL vs. H | 5.26E-10 | 142 |
| L vs. H | 6.62E-22 | 399 | ||
| Cell Morphology, Cellular Development, Cellular Growth and Proliferation, Nervous System Development and Function, Organismal Development, Tissue Development | Morphogenesis of neurons | NL vs. H | 6.60E-10 | 143 |
| L vs. H | 6.85E-22 | 403 | ||
| Cellular Development, Cellular Growth and Proliferation, Nervous System Development and Function, Tissue Development | Development of neurons | NL vs. H | 1.12E-09 | 177 |
| L vs. H | 2.14E-24 | 517 |
| GO Term | Description | Comparison | p-Value | FDR q-Value | Enrichment (N, B, n, b) |
|---|---|---|---|---|---|
| GO:0010975 | Regulation of neuron projection development | NL vs. H | 1.98E-4 | 2.74E-2 | 1.72 (1442, 229, 139, 38) |
| L vs. H | 6.66E-10 | 2.79E-7 | 1.64 (1594, 260, 389, 104) | ||
| GO:0045664 | Regulation of neuron differentiation | NL vs. H | 2.68E-4 | 3.35E-2 | 1.67 (1442, 249, 139, 40) |
| L vs. H | 6.63E-11 | 4.07E-8 | 1.64 (1594, 285, 389, 114) | ||
| GO:0071526 | Semaphorin-plexin signaling pathway | NL vs. H | 4.09E-4 | 4.07E-2 | 5.19 (1442, 12, 139, 6) |
| L vs. H | 2.3E-5 | 1.37E-3 | 2.96 (1594, 18, 389, 13) |
| GO Term | Description | Comparison | p-Value | FDR q-Value | Enrichment (N, B, n, b) |
|---|---|---|---|---|---|
| GO:0048812 | Neuron projection morphogenesis | L vs. H | 2.94E-10 | 1.43E-7 | 1.96 (1594, 138, 389, 66) |
| GO:0097485 | Neuron projection guidance | 8.37E-7 | 9.17E-5 | 1.78 (1594, 124, 389, 54) | |
| GO:0031175 | Neuron projection development | 9.31E-7 | 9.74E-5 | 1.68 (1594, 159, 389, 65) | |
| GO:0010976 | Positive regulation of neuron projection development | 3.15E-6 | 2.54E-4 | 1.69 (1594, 141, 389, 58) | |
| GO:0010977 | Negative regulation of neuron projection development | 9.99E-5 | 4.76E-3 | 1.74 (1594, 87, 389, 37) |
| GO Term | Description | Comparison | p-Value | FDR q-Value | Enrichment (N, B, n, b) |
|---|---|---|---|---|---|
| GO:0050770 | Regulation of axonogenesis | L vs. H | 6.36E-7 | 7.32E-5 | 1.89 (1594, 102, 389, 47) |
| GO:0007411 | Axon guidance | 8.37E-7 | 9.06E-5 | 1.78 (1594, 124, 389, 54) | |
| GO:1902668 | Negative regulation of axon guidance | 8.36E-5 | 4.2E-3 | 3.00 (1594, 15, 389, 11) | |
| GO:0048843 | Negative regulation of axon extension involved in axon guidance | 9.41E-5 | 4.56E-3 | 3.15 (1594, 13, 389, 10) | |
| GO:0050771 | Negative regulation of axonogenesis | 2.61E-4 | 1.07E-2 | 2.00 (1594, 45, 389, 22) | |
| GO:0008045 | Motor neuron axon guidance | 6.07E-4 | 2.21E-2 | 2.73 (1594, 15, 389, 10) | |
| GO:0048841 | Regulation of axon extension involved in axon guidance | 6.07E-4 | 2.2E-2 | 2.73 (1594, 15, 389, 10) | |
| GO:1902667 | Regulation of axon guidance | 9.35E-4 | 3.19E-2 | 2.50 (1594, 18, 389, 11) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2022 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Romhányi, D.; Szabó, K.; Kemény, L.; Sebestyén, E.; Groma, G. Transcriptional Analysis-Based Alterations Affecting Neuritogenesis of the Peripheral Nervous System in Psoriasis. Life 2022, 12, 111. https://doi.org/10.3390/life12010111
Romhányi D, Szabó K, Kemény L, Sebestyén E, Groma G. Transcriptional Analysis-Based Alterations Affecting Neuritogenesis of the Peripheral Nervous System in Psoriasis. Life. 2022; 12(1):111. https://doi.org/10.3390/life12010111
Chicago/Turabian StyleRomhányi, Dóra, Kornélia Szabó, Lajos Kemény, Endre Sebestyén, and Gergely Groma. 2022. "Transcriptional Analysis-Based Alterations Affecting Neuritogenesis of the Peripheral Nervous System in Psoriasis" Life 12, no. 1: 111. https://doi.org/10.3390/life12010111
APA StyleRomhányi, D., Szabó, K., Kemény, L., Sebestyén, E., & Groma, G. (2022). Transcriptional Analysis-Based Alterations Affecting Neuritogenesis of the Peripheral Nervous System in Psoriasis. Life, 12(1), 111. https://doi.org/10.3390/life12010111







