Whole-Genome Resequencing Points to Candidate DNA Loci Affecting Body Temperature under Cold Stress in Siberian Cattle Populations
Abstract
1. Introduction
2. Materials and Methods
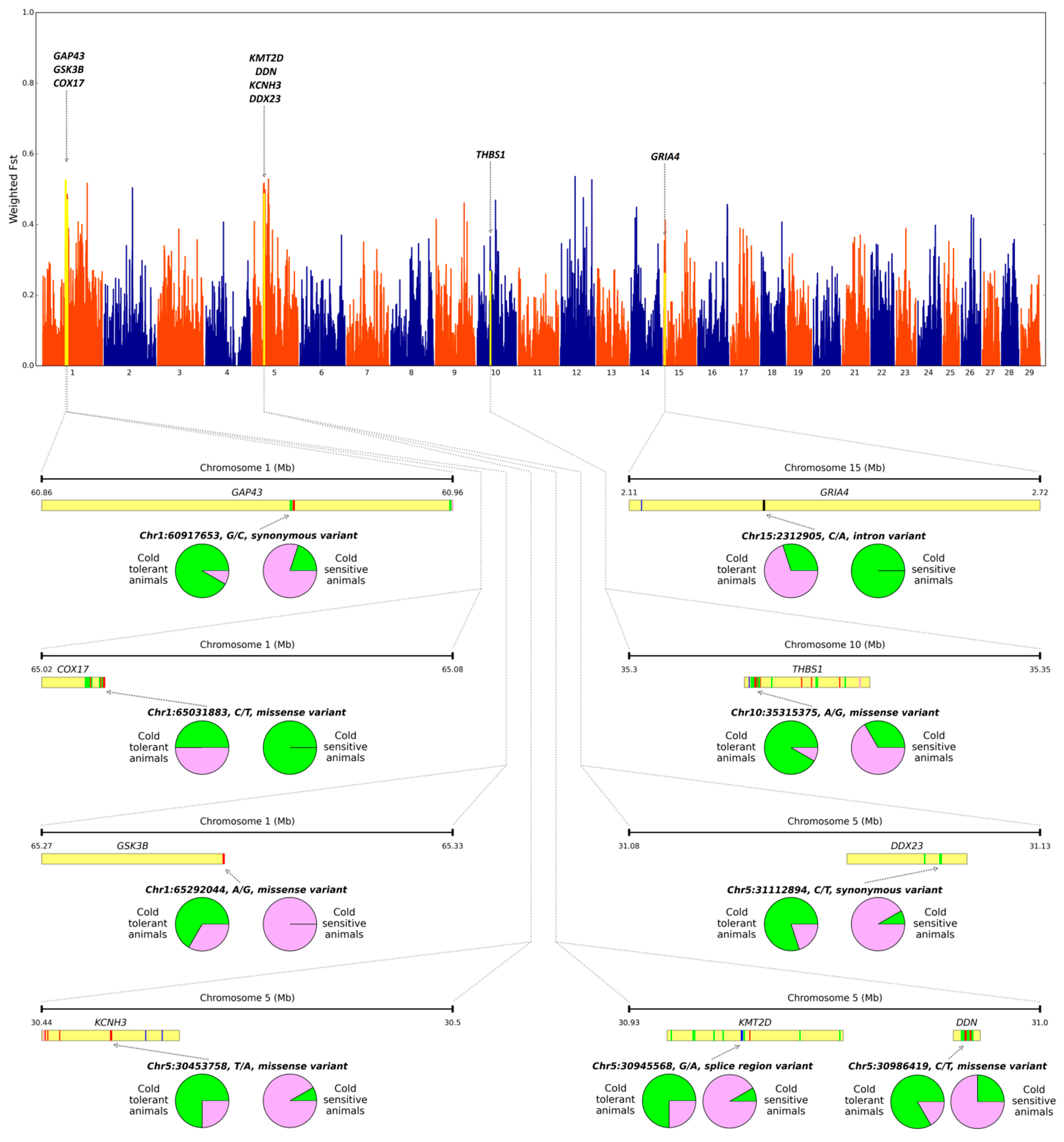
3. Results
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Young, B.A. Cold stress as it affects animal production. J. Anim. Sci. 1981, 52, 154–163. [Google Scholar] [CrossRef]
- Okumura, J.; Mori, N.; Muramatsu, T.; Tasaki, I.; Saito, F. Analysis of factors affecting year-round performance of single comb white leghorn laying hens reared under an open-sided housing system. Poult. Sci. 1988, 67, 1130–1138. [Google Scholar] [CrossRef] [PubMed]
- Piao, M.Y.; Baik, M. Seasonal variation in carcass characteristics of korean cattle steers. Asian-Australas. J. Anim. Sci. 2015, 28, 442–450. [Google Scholar] [CrossRef] [PubMed]
- Ghasemi, E.; Azad-Shahraki, M.; Khorvash, M. Effect of different fat supplements on performance of dairy calves during cold season. J. Dairy Sci. 2017, 100, 5319–5328. [Google Scholar] [CrossRef] [PubMed]
- Kang, H.J.; Piao, M.Y.; Park, S.J.; Na, S.W.; Kim, H.J.; Baik, M. Effects of ambient temperature and rumen-protected fat supplementation on growth performance, rumen fermentation and blood parameters during cold season in Korean cattle steers. Asian-Australas. J. Anim. Sci. 2019, 32, 657–664. [Google Scholar] [CrossRef]
- Weldenegodguad, M.; Popov, R.; Pokharel, K.; Ammosov, I.; Ming, Y.; Ivanova, Z.; Kantanen, J. Whole-genome sequencing of three native cattle breeds originating from the northernmost cattle farming regions. Front. Genet. 2018, 9, 728. [Google Scholar] [CrossRef] [PubMed]
- Banerjee, D.; Upadhyay, R.C.; Chaudhary, U.B.; Kumar, R.; Singh, S.; Ashutosh; Mohanarao, G.J.; Polley, S.; Mukherjee, A.; Das, T.K.; et al. Seasonal variation in expression pattern of genes under hsp70 : Seasonal variation in expression pattern of genes under HSP70 Family in heat and cold-adapted goats (Capra hircus). Cell Stress Chaperones 2014, 19, 401–408. [Google Scholar] [CrossRef]
- Gan, M.; Shen, L.; Fan, Y.; Guo, Z.; Liu, B.; Chen, L.; Tang, G.; Jiang, Y.; Li, X.; Zhang, S.; et al. High altitude adaptability and meat quality in Tibetan pigs: A reference for local pork processing and genetic improvement. Animals 2019, 9, 1080. [Google Scholar] [CrossRef]
- Librado, P.; Der Sarkissian, C.; Ermini, L.; Schubert, M.; Jónsson, H.; Albrechtsen, A.; Fumagalli, M.; Yang, M.A.; Gamba, C.; Seguin-Orlando, A.; et al. Tracking the origins of Yakutian horses and the genetic basis for their fast adaptation to subarctic environments. Proc. Natl. Acad. Sci. USA 2015, 112, E6889–E6897. [Google Scholar] [CrossRef]
- Xie, S.; Yang, X.; Gao, Y.; Jiao, W.; Li, X.; Li, Y.; Ning, Z. Performance differences of Rhode Island Red, Bashang Long-Tail chicken, and their reciprocal crossbreds under natural cold stress. Asian-Australas. J. Anim. Sci. 2017, 30, 1507–1514. [Google Scholar] [CrossRef]
- Kudinov, A.A.; Dementieva, N.V.; Mitrofanova, O.V.; Stanishevskaya, O.I.; Fedorova, E.S.; Larkina, T.A.; Mishina, A.I.; Plemyashov, K.V.; Griffin, D.K.; Romanov, M.N. Genome-wide association studies targeting the yield of extraembryonic fluid and production traits in Russian white chickens. BMC Genom. 2019, 20, 270. [Google Scholar] [CrossRef] [PubMed]
- Phillips, C.J.C. Principles of Cattle Production, 3rd ed.; CABI Wallingford: Oxfordshire, UK, 2018. [Google Scholar]
- Howard, J.T.; Kachman, S.D.; Snelling, W.M.; Pollak, E.J.; Ciobanu, D.C.; Kuehn, L.A.; Spangler, M.L. Beef cattle body temperature during climatic stress: A genome-wide association study. Int. J. Biometeorol. 2014, 58, 1665–1672. [Google Scholar] [CrossRef] [PubMed]
- Yurchenko, A.A.; Daetwyler, H.D.; Yudin, N.; Schnabel, R.D.; Vander Jagt, C.J.; Soloshenko, V.; Lhasaranov, B.; Popov, R.; Taylor, J.F.; Larkin, D.M. Scans for signatures of selection in Russian cattle breed genomes reveal new candidate genes for environmental adaptation and acclimation. Sci. Rep. 2018, 8, 12984. [Google Scholar] [CrossRef]
- Buggiotti, L.; Yurchenko, A.A.; Yudin, N.S.; Vander Jagt, C.J.; Vorobieva, N.V.; Kusliy, M.A.; Vasiliev, S.K.; Rodionov, A.N.; Boronetskaya, O.I.; Zinovieva, N.A.; et al. Demographic history, adaptation, and NRAP convergent evolution at amino acid residue 100 in the world northernmost cattle from Siberia. Mol. Biol. Evol. 2021. [Google Scholar] [CrossRef]
- Xu, Q.; Wang, Y.C.; Liu, R.; Brito, L.F.; Kang, L.; Yu, Y.; Wang, D.S.; Wu, H.J.; Liu, A. Differential gene expression in the peripheral blood of Chinese Sanhe cattle exposed to severe cold stress. Genet. Mol. Res. 2017, 16, gmr16029593. [Google Scholar] [CrossRef]
- Cao, K.X.; Hao, D.; Wang, J.; Peng, W.W.; Yan, Y.J.; Cao, H.X.; Sun, F.; Chen, H. Cold exposure induces the acquisition of brown adipocyte gene expression profiles in cattle inguinal fat normalized with a new set of reference genes for QRT-PCR. Res. Vet. Sci. 2017, 114, 1–5. [Google Scholar] [CrossRef] [PubMed]
- Pokharel, K.; Weldenegodguad, M.; Popov, R.; Honkatukia, M.; Huuki, H.; Lindeberg, H.; Peippo, J.; Reilas, T.; Zarovnyaev, S.; Kantanen, J. Whole blood transcriptome analysis reveals footprints of cattle adaptation to sub-arctic conditions. Anim. Genet. 2019, 50, 217–227. [Google Scholar] [CrossRef]
- Igoshin, A.V.; Yurchenko, A.A.; Belonogova, N.M.; Petrovsky, D.V.; Aitnazarov, R.B.; Soloshenko, V.A.; Yudin, N.S.; Larkin, D.M. Genome-wide association study and scan for signatures of selection point to candidate genes for body temperature maintenance under the cold stress in Siberian cattle populations. BMC Genet. 2019, 20, 5–14. [Google Scholar] [CrossRef]
- Cheruiyot, E.K.; Haile-Mariam, M.; Cocks, B.G.; MacLeod, I.M.; Xiang, R.; Pryce, J.E. New loci and neuronal pathways for resilience to heat stress in cattle. Sci. Rep. 2021, 11, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Hu, Z.-L.; Park, C.A.; Reecy, J.M. Building a livestock genetic and genomic information knowledgebase through integrative developments of animal QTLdb and CorrDB. Nucleic Acids Res. 2019, 47, D701–D710. [Google Scholar] [CrossRef]
- Xiang, R.; van den Berg, I.; MacLeod, I.M.; Hayes, B.J.; Prowse-Wilkins, C.P.; Wang, M.; Bolormaa, S.; Liu, Z.; Rochfort, S.J.; Reich, C.M.; et al. Quantifying the contribution of sequence variants with regulatory and evolutionary significance to 34 bovine complex traits. Proc. Natl. Acad. Sci. USA 2019, 116, 19398–19408. [Google Scholar] [CrossRef]
- Li, H.; Durbin, R. Fast and accurate short read alignment with Burrows-Wheeler transform. Bioinformatics 2009, 25, 1754–1760. [Google Scholar] [CrossRef] [PubMed]
- DePristo, M.A.; Banks, E.; Poplin, R.; Garimella, K.V.; Maguire, J.R.; Hartl, C.; Philippakis, A.A.; del Angel, G.; Rivas, M.A.; Hanna, M.; et al. A framework for variation discovery and genotyping using next-generation DNA sequencing data. Nat. Genet. 2011, 43, 491–498. [Google Scholar] [CrossRef]
- Weir, B.S.; Cockerham, C.C. Estimating F-statistics for the analysis of population structure. Evolution 1984, 38, 1358–1370. [Google Scholar] [CrossRef] [PubMed]
- Danecek, P.; Auton, A.; Abecasis, G.; Albers, C.A.; Banks, E.; DePristo, M.A.; Handsaker, R.E.; Lunter, G.; Marth, G.T.; Sherry, S.T.; et al. The variant call format and VCFtools. Bioinformatics 2011, 27, 2156–2158. [Google Scholar] [CrossRef]
- Grant, J.R.; Arantes, A.S.; Liao, X.; Stothard, P. In-depth annotation of SNPs arising from resequencing projects using NGS-SNP. Bioinformatics 2011, 27, 2300–2301. [Google Scholar] [CrossRef]
- Hinrichs, A.S.; Raney, B.J.; Speir, M.L.; Rhead, B.; Casper, J.; Karolchik, D.; Kuhn, R.M.; Rosenbloom, K.R.; Zweig, A.S.; Haussler, D.; et al. UCSC data integrator and variant annotation integrator. Bioinformatics 2016, 32, 1430–1432. [Google Scholar] [CrossRef]
- Verma, P.; Sharma, A.; Sodhi, M.; Thakur, K.; Kataria, R.S.; Niranjan, S.K.; Bharti, V.K.; Kumar, P.; Giri, A.; Kalia, S.; et al. Transcriptome analysis of circulating PBMCs to understand mechanism of high altitude adaptation in native cattle of Ladakh region. Sci. Rep. 2018, 8, 7681. [Google Scholar] [CrossRef] [PubMed]
- Long, Y.; Li, X.; Li, F.; Ge, G.; Liu, R.; Song, G.; Li, Q.; Qiao, Z.; Cui, Z. Transcriptional programs underlying cold acclimation of common carp (Cyprinus carpio L.). Front. Genet. 2020, 11, 556418. [Google Scholar] [CrossRef]
- Pitt, D.; Bruford, M.W.; Barbato, M.; Orozco-terWengel, P.; Martínez, R.; Sevane, N. Demography and rapid local adaptation shape Creole cattle genome diversity in the tropics. Evol. Appl. 2019, 12, 105–122. [Google Scholar] [CrossRef]
- Coppe, A.; Agostini, C.; Marino, I.A.M.; Zane, L.; Bargelloni, L.; Bortoluzzi, S.; Patarnello, T. Genome evolution in the cold: Antarctic icefish muscle transcriptome reveals selective duplications increasing mitochondrial function. Genome Biol. Evol. 2013, 5, 45–60. [Google Scholar] [CrossRef]
- Bhardwaj, S.; Singh, S.; Ganguly, I.; Bhatia, A.K.; Bharti, V.K.; Dixit, S.P. Genome-wide diversity analysis for signatures of selection of Bos indicus adaptability under extreme agro-climatic conditions of temperate and tropical ecosystems. Anim. Gene 2021, 20, 200115. [Google Scholar] [CrossRef]
- Lin, J.; Cao, C.; Tao, C.; Ye, R.; Dong, M.; Zheng, Q.; Wang, C.; Jiang, X.; Qin, G.; Yan, C.; et al. Cold adaptation in pigs depends on UCP3 in beige adipocytes. J. Mol. Cell Biol. 2017, 9, 364–375. [Google Scholar] [CrossRef]
- Tavares, E.; Miñano, F.J. RANTES: A new prostaglandin dependent endogenous pyrogen in the rat. Neuropharmacology 2000, 39, 2505–2513. [Google Scholar] [CrossRef]
- Liu, P.; Guo, L.; Mao, H.; Gu, Z. Serum proteomics analysis reveals the thermal fitness of crossbred dairy buffalo to chronic heat stress. J. Therm. Biol. 2020, 89, 102547. [Google Scholar] [CrossRef]
- Freitas, P.H.F.; Wang, Y.; Yan, P.; Oliveira, H.R.; Schenkel, F.S.; Zhang, Y.; Xu, Q.; Brito, L.F. Genetic diversity and signatures of selection for thermal stress in cattle and other two Bos species adapted to divergent climatic conditions. Front. Genet. 2021, 12, 604823. [Google Scholar] [CrossRef]
- Shore, A.M.; Karamitri, A.; Kemp, P.; Speakman, J.R.; Graham, N.S.; Lomax, M.A. Cold-induced changes in gene expression in brown adipose tissue, white adipose tissue and liver. PLoS ONE 2013, 8, e68933. [Google Scholar] [CrossRef] [PubMed]
- Srikanth, K.; Kwon, A.; Lee, E.; Chung, H. Characterization of genes and pathways that respond to heat stress in Holstein calves through transcriptome analysis. Cell Stress Chaperones 2017, 22, 29–42. [Google Scholar] [CrossRef] [PubMed]
- Rim, J.S.; Kozak, L.P. Regulatory motifs for CREB-binding protein and Nfe2l2 transcription factors in the upstream enhancer of the mitochondrial uncoupling protein 1 gene. J. Biol. Chem. 2002, 277, 34589–34600. [Google Scholar] [CrossRef] [PubMed]
- Eslamizad, M.; Albrecht, D.; Kuhla, B. The effect of chronic, mild heat stress on metabolic changes of nutrition and adaptations in rumen papillae of lactating dairy cows. J. Dairy Sci. 2020, 103, 8601–8614. [Google Scholar] [CrossRef] [PubMed]
- Mottillo, E.P.; Desjardins, E.M.; Crane, J.D.; Smith, B.K.; Green, A.E.; Ducommun, S.; Henriksen, T.I.; Rebalka, I.A.; Razi, A.; Sakamoto, K.; et al. Lack of adipocyte AMPK exacerbates insulin resistance and hepatic steatosis through brown and beige adipose tissue function. Cell Metab. 2016, 24, 118–129. [Google Scholar] [CrossRef] [PubMed]
- Singh, A.K.; Upadhyay, R.C.; Chandra, G.; Kumar, S.; Malakar, D.; Singh, S.V.; Singh, M.K. Genome-wide expression analysis of the heat stress response in dermal fibroblasts of Tharparkar (Zebu) and Karan-Fries (Zebu × Taurine) cattle. Cell Stress Chaperones 2020, 25, 327–344. [Google Scholar] [CrossRef] [PubMed]
- Khaibullina, A.; Kenyon, N.; Guptill, V.; Quezado, M.M.; Wang, L.; Koziol, D.; Wesley, R.; Moya, P.R.; Zhang, Z.; Saha, A.; et al. In a model of batten disease, palmitoyl protein thioesterase-1 deficiency is associated with brown adipose tissue and thermoregulation abnormalities. PLoS ONE 2012, 7, e48733. [Google Scholar] [CrossRef] [PubMed]
- Walsh, I.M.; Bowman, M.A.; Soto Santarriaga, I.F.; Rodriguez, A.; Clark, P.L. Synonymous codon substitutions perturb cotranslational protein folding in vivo and impair cell fitness. Proc. Natl. Acad. Sci. USA 2020, 117, 3528–3534. [Google Scholar] [CrossRef]
- Tang, X.; Miao, Y.; Luo, Y.; Sriram, K.; Qi, Z.; Lin, F.-M.; Gu, Y.; Lai, C.-H.; Hsu, C.-Y.; Peterson, K.L.; et al. Suppression of endothelial AGO1 promotes adipose tissue browning and improves metabolic dysfunction. Circulation 2020, 142, 365–379. [Google Scholar] [CrossRef]
- Wu, J.; Cohen, P.; Spiegelman, B.M. Adaptive thermogenesis in adipocytes: Is beige the new brown? Genes Dev. 2013, 27, 234–250. [Google Scholar] [CrossRef]
- Swain, L.L.; Mishra, C.; Sahoo, S.S.; Nayak, G.; Pradhan, S.K.; Mishra, S.R.; Dige, M. An in vivo and in silico analysis of novel variation in TMBIM6 gene affecting cardiopulmonary traits of Indian goats. J. Therm. Biol. 2020, 88, 102491. [Google Scholar] [CrossRef]
- Weldenegodguad, M.; Pokharel, K.; Niiranen, L.; Soppela, P.; Ammosov, I.; Honkatukia, M.; Lindeberg, H.; Peippo, J.; Reilas, T.; Mazzullo, N.; et al. Adipose gene expression profiles reveal novel insights into the adaptation of northern Eurasian semi-domestic reindeer (Rangifer tarandus). bioRxiv 2021. [Google Scholar] [CrossRef]
- Worthmann, A.; John, C.; Rühlemann, M.C.; Baguhl, M.; Heinsen, F.-A.; Schaltenberg, N.; Heine, M.; Schlein, C.; Evangelakos, I.; Mineo, C.; et al. Cold-induced conversion of cholesterol to bile acids in mice shapes the gut microbiome and promotes adaptive thermogenesis. Nat. Med. 2017, 23, 839–849. [Google Scholar] [CrossRef]
- Pereira-da-Silva, M.; Torsoni, M.A.; Nourani, H.V.; Augusto, V.D.; Souza, C.T.; Gasparetti, A.L.; Carvalheira, J.B.; Ventrucci, G.; Marcondes, M.C.C.G.; Cruz-Neto, A.P.; et al. Hypothalamic melanin-concentrating hormone is induced by cold exposure and participates in the control of energy expenditure in rats. Endocrinology 2003, 144, 4831–4840. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Sambeat, A.; Gulyaeva, O.; Dempersmier, J.; Sul, H.S. Epigenetic regulation of the thermogenic adipose program. Trends Endocrinol. Metab. 2017, 28, 19–31. [Google Scholar] [CrossRef]
- Gracey, A.Y.; Fraser, E.J.; Li, W.; Fang, Y.; Taylor, R.R.; Rogers, J.; Brass, A.; Cossins, A.R. Coping with cold: An integrative, multitissue analysis of the transcriptome of a poikilothermic vertebrate. Proc. Natl. Acad. Sci. USA 2004, 101, 16970–16975. [Google Scholar] [CrossRef]
- Yudin, N.S.; Larkin, D.M.; Ignatieva, E.V. A compendium and functional characterization of mammalian genes involved in adaptation to Arctic or Antarctic environments. BMC Genet. 2017, 18, 111. [Google Scholar] [CrossRef]
- Hancock, A.M.; Witonsky, D.B.; Gordon, A.S.; Eshel, G.; Pritchard, J.K.; Coop, G.; Di Rienzo, A. Adaptations to climate in candidate genes for common metabolic disorders. PLoS Genet. 2008, 4, e32. [Google Scholar] [CrossRef]
| Gene | SNP Position (UMD3.1), (RefSNP) | Reference/Alternate Allele | Sum of Ranks | Reference Allele Frequency | Functional Class | Literature Evidence | |
|---|---|---|---|---|---|---|---|
| Cold-Sensitive Group | Cold-Tolerant Group | ||||||
| DDX23 | Chr5:31,112,894 (rs108955444) | C/T | 0.002 | 0.08 | 0.80 | synonymous variant | Climate adaptation in cattle [29], cold adaptation in common carp [30] |
| MAATS1 | Chr1:65,062,344 (rs43234266) | T/C | 0.01 | 1.00 | 0.13 | synonymous variant | Adaptation of cattle to tropical climates [31] |
| GRIA4 | Chr15:2,312,905 (rs207668622) | C/A | 0.01 | 1.00 | 0.30 | intron variant | Cold [19] and heat [20] adaptations in cattle |
| COX17 | Chr1:65,031,883 (rs208045948) | C/T | 0.02 | 1.00 | 0.50 | missense variant | Adaptation of cattle to tropical climates [31], cold adaptation in Antarctic icefish [32] |
| THBS1 | Chr10:35,315,375 (rs43707861) | A/G | 0.02 | 0.33 | 0.92 | missense variant | Cold [18] and heat [33] adaptations in cattle, cold adaptation in pigs [34] |
| Chr10:35,320,988 (rs17870352) | A/G | 0.02 | 0.33 | 0.90 | missense variant | ||
| CCL5 | Chr19:14,825,116 (rs208398974) | C/T | 0.02 | 0.25 | 1.00 | synonymous variant | Cold adaptation in cattle [18], thermoregulation in rats [35] |
| UPK1B | Chr1:64,592,185 (rs43652277) | A/G | 0.02 | 0.10 | 0.63 | missense variant | Adaptation of cattle to tropical climates [31] |
| PLA1A | Chr1:64,966,636 (rs43233262) | C/A | 0.03 | 0.00 | 0.83 | intron variant | Adaptation of buffaloes to heat stress [36] |
| NR1I2 | Chr1:65,236,459 (rs43235975) | T/C | 0.04 | 0.00 | 0.42 | synonymous variant | Adaptation of cattle to heat stress [31,37], cold stress response in mice [38] |
| ATF1 | Chr5:29,271,337 (rs210280224) | A/G | 0.06 | 0.00 | 0.63 | downstream gene variant | Adaptation of cattle to heat stress [39], regulation of brown adipose tissue thermogenesis in mammals [40] |
| PRKAG1 | Chr5:30,981,551 (rs29002398) | T/C | 0.06 | 0.08 | 0.83 | 3′-UTR variant | Adaptation of cattle to heat stress [41], regulation of brown adipose tissue thermogenesis in mammals [42] |
| IFNGR1 | Chr9:76,093,074 (rs41569368) | T/G | 0.06 | 0.83 | 0.33 | synonymous variant | Cold adaptation in cattle [18] |
| PPT1 | Chr3:106,629,521 (rs42791314) | T/C | 0.07 | 0.30 | 0.88 | missense variant | Heat adaptation in cattle [43], thermoregulation in mice [44] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Igoshin, A.; Yudin, N.; Aitnazarov, R.; Yurchenko, A.A.; Larkin, D.M. Whole-Genome Resequencing Points to Candidate DNA Loci Affecting Body Temperature under Cold Stress in Siberian Cattle Populations. Life 2021, 11, 959. https://doi.org/10.3390/life11090959
Igoshin A, Yudin N, Aitnazarov R, Yurchenko AA, Larkin DM. Whole-Genome Resequencing Points to Candidate DNA Loci Affecting Body Temperature under Cold Stress in Siberian Cattle Populations. Life. 2021; 11(9):959. https://doi.org/10.3390/life11090959
Chicago/Turabian StyleIgoshin, Alexander, Nikolay Yudin, Ruslan Aitnazarov, Andrey A. Yurchenko, and Denis M. Larkin. 2021. "Whole-Genome Resequencing Points to Candidate DNA Loci Affecting Body Temperature under Cold Stress in Siberian Cattle Populations" Life 11, no. 9: 959. https://doi.org/10.3390/life11090959
APA StyleIgoshin, A., Yudin, N., Aitnazarov, R., Yurchenko, A. A., & Larkin, D. M. (2021). Whole-Genome Resequencing Points to Candidate DNA Loci Affecting Body Temperature under Cold Stress in Siberian Cattle Populations. Life, 11(9), 959. https://doi.org/10.3390/life11090959






