Contributions to a Discussion of Spinosaurus aegyptiacus as a Capable Swimmer and Deep-Water Predator
Abstract
1. Introduction
2. Methods
3. Discussion
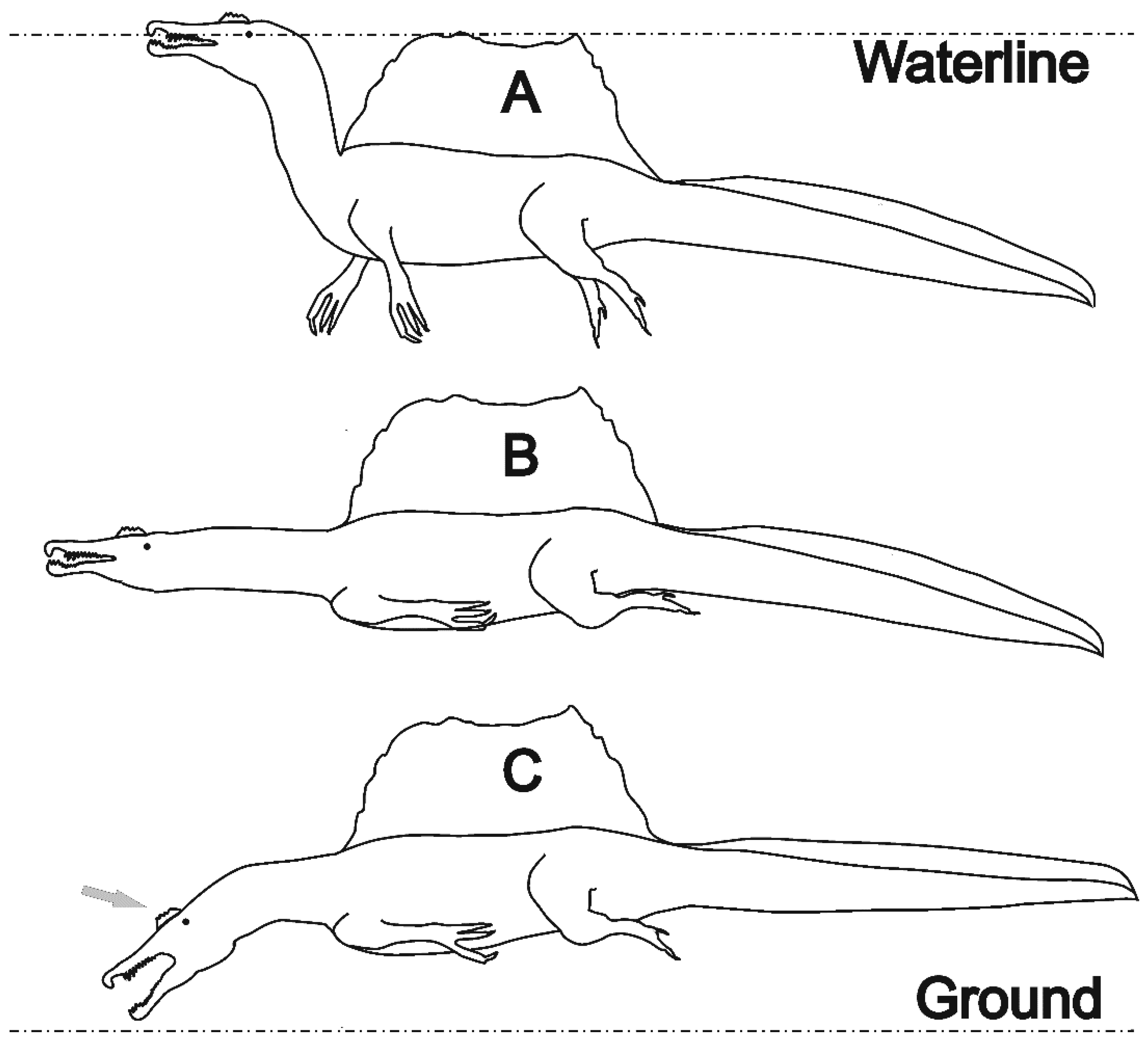
3.1. Buoyancy and Balance Aspects
3.2. Hydrodynamic Aspects
3.3. Is the Head Crest an Adaptation to ‘Pivot’ Feeding?
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Stromer, E. Ergebnisse der Forschungsreisen Prof. E. Stromers in den Wüsten Ägyptens. II. Wirbeltier-Reste der Baharîje-Stufe (unterstes Cenoman). 3. Das Original des Theropoden Spinosaurus aegyptiacus nov. gen., nov. spec. Abh. Der Königlichen Bayer. Akad. Der Wiss. Math. -Phys. Kl. 1915, 28, 1–33. [Google Scholar]
- Martill, D.M.; Ibrahim, N.; Brito, P.M.; Baider, L.; Zhouri, S.; Loveridge, R.; Naish, D.; Hing, R. A new plattenkalk Konservat Lagerstätte in the Upper Cretaceous of Gara Sbaa, south-eastern Morocco. Cretac. Res. 2011, 32, 433–446. [Google Scholar] [CrossRef]
- Sereno, P.C.; Dutheil, D.B.; Iarochene, M.; Larsson, H.C.; Lyon, G.H.; Magwene, P.M.; Sidor, C.A.; Varricchio, D.J.; Wilson, J.A. Predatory dinosaurs from the Sahara and Late Cretaceous faunal differentiation. Science 1996, 272, 986–991. [Google Scholar] [CrossRef] [PubMed]
- Taquet, P. A curious specialisation of the skull of some Cretaceous carnivorous dinosaurs: The long and narrow snout of spinosaurids. Comptes Rendus del’Académie Des Sci. 1984, 299, 217–222. [Google Scholar]
- Ibrahim, N.; Sereno, P.C.; Dal Sasso, C.; Maganuco, S.; Fabbri, M.; Martill, D.M.; Zouhri, S.; Myhrvold, N.; Iurino, D.A. Semiaquatic adaptations in a giant predatory dinosaur. Science 2014, 345, 1613–1616. [Google Scholar] [CrossRef]
- Amiot, R.; Buffetaut, E.; Lécuyer, C.; Wang, X.; Boudad, L.; Ding, Z.; Fourel, F.; Hutt, S.; Martineau, F.; Medeiros, M.A. Oxygen isotope evidence for semi-aquatic habits among spinosaurid theropods. Geology 2010, 38, 139–142. [Google Scholar] [CrossRef]
- Ibrahim, N.; Maganuco, S.; Dal Sasso, C.; Fabbri, M.; Auditore, M.; Bindellini, G.; Martill, D.M.; Zouhri, S.; Mattarelli, D.A.; Unwin, D.M. Tail-propelled aquatic locomotion in a theropod dinosaur. Nature 2020, 581, 67–70. [Google Scholar] [CrossRef]
- Gimsa, J.; Sleigh, R.; Gimsa, U. The riddle of Spinosaurus aegyptiacus’ dorsal sail. Geol. Mag. 2016, 153, 544–547. [Google Scholar] [CrossRef]
- Arden, T.M.; Klein, C.G.; Zouhri, S.; Longrich, N.R. Aquatic adaptation in the skull of carnivorous dinosaurs (Theropoda: Spinosauridae) and the evolution of aquatic habits in spinosaurids. Cretac. Res. 2019, 93, 275–284. [Google Scholar] [CrossRef]
- Vullo, R.; Allain, R.; Cavin, L. Convergent evolution of jaws between spinosaurid dinosaurs and pike conger eels. Acta Palaeontol. Pol. 2016, 61, 825–828. [Google Scholar] [CrossRef]
- Bailey, J.B. Neural spine elongation in dinosaurs: Sailbacks or buffalo-backs? J. Paleontol. 1997, 71, 1124–1146. [Google Scholar] [CrossRef]
- Benton, M.J. Ectothermy and the success of dinosaurs. Evolution 1979, 33, 983–997. [Google Scholar] [CrossRef] [PubMed]
- Wheeler, P. Elaborate CNS cooling structures in large dinosaurs. Nature 1978, 275, 441–443. [Google Scholar] [CrossRef] [PubMed]
- Hone, D.W.E.; Holtz Jr, T.R. A century of spinosaurs-a review and revision of the Spinosauridae with comments on their ecology. Acta Geol. Sin. -Engl. Ed. 2017, 91, 1120–1132. [Google Scholar] [CrossRef]
- Henderson, D.M. A buoyancy, balance and stability challenge to the hypothesis of a semi-aquatic Spinosaurus Stromer, 1915 (Dinosauria: Theropoda). PeerJ 2018, 6, e5409. [Google Scholar] [CrossRef] [PubMed]
- Allain, R.; Xaisanavong, T.; Richir, P.; Khentavong, B. The first definitive Asian spinosaurid (Dinosauria: Theropoda) from the early cretaceous of Laos. Naturwissenschaften 2012, 99, 369–377. [Google Scholar] [CrossRef]
- Sues, H.-D.; Frey, E.; Martill, D.M.; Scott, D.M. Irritator challengeri, a spinosaurid (Dinosauria: Theropoda) from the Lower Cretaceous of Brazil. J. Vertebr. Paleontol. 2002, 22, 535–547. [Google Scholar] [CrossRef]
- Schade, M.; Rauhut, O.W.; Evers, S.W. Neuroanatomy of the spinosaurid Irritator challengeri (Dinosauria: Theropoda) indicates potential adaptations for piscivory. Sci. Rep. 2020, 10, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Charig, A.J.; Milner, A.C. Baryonyx, a remarkable new theropod dinosaur. Nature 1986, 324, 359–361. [Google Scholar] [CrossRef]
- Charig, A.J.; Milner, A.C. Baryonyx walkeri, a fish-eating dinosaur from the Wealden of Surrey. Bull. -Nat. Hist. Mus. Geol. Ser. 1997, 53, 11–70. [Google Scholar]
- Malafaia, E.; Gasulla, J.; Escaso, F.; Narvaéz, I.; Ortega, F. An update of the spinosaurid (Dinosauria: Theropoda) fossil record from the Lower Cretaceous of the Iberian Peninsula: Distribution, diversity, and evolutionary history. J. Iber. Geol. 2020, 46, 431–444. [Google Scholar] [CrossRef]
- Foffa, D.; Cuff, A.R.; Sassoon, J.; Rayfield, E.J.; Mavrogordato, M.N.; Benton, M.J. Functional anatomy and feeding biomechanics of a giant Upper Jurassic pliosaur (Reptilia: Sauropterygia) from Weymouth Bay, Dorset, UK. J. Anat. 2014, 225, 209–219. [Google Scholar] [CrossRef]
- Smyth, R.S.; Ibrahim, N.; Martill, D.M. Sigilmassasaurus is Spinosaurus: A reappraisal of African spinosaurines. Cretac. Res. 2020, 114, 104520. [Google Scholar] [CrossRef]
- Gemmell, B.J.; Sheng, J.; Buskey, E.J. Morphology of seahorse head hydrodynamically aids in capture of evasive prey. Nat. Commun. 2013, 4, 1–8. [Google Scholar] [CrossRef]
- Brazaitis, P.; Watanabe, M.E. Crocodilian behaviour: A window to dinosaur behaviour? Hist. Biol. 2011, 23, 73–90. [Google Scholar] [CrossRef]
- Soares, D. An ancient sensory organ in crocodilians. Nature 2002, 417, 241–242. [Google Scholar] [CrossRef] [PubMed]
- Leitch, D.B.; Catania, K.C. Structure, innervation and response properties of integumentary sensory organs in crocodilians. J. Exp. Biol. 2012, 215, 4217–4230. [Google Scholar] [CrossRef] [PubMed]
- Butler, P.J.; Jones, D.R. Physiology of diving of birds and mammals. Physiol. Rev. 1997, 77, 837–899. [Google Scholar] [CrossRef] [PubMed]
- Ponganis, P.; Welch, T.; Welch, L.; Stockard, T. Myoglobin production in emperor penguins. J. Exp. Biol. 2010, 213, 1901–1906. [Google Scholar] [CrossRef]
- Helbo, S.; Fago, A. Functional properties of myoglobins from five whale species with different diving capacities. J. Exp. Biol. 2012, 215, 3403–3410. [Google Scholar] [CrossRef]
- Ferreras, J.M.; Ragucci, S.; Citores, L.; Iglesias, R.; Pedone, P.V.; Di Maro, A. Insight into the phylogenetic relationship and structural features of vertebrate myoglobin family. Int. J. Biol. Macromol. 2016, 93, 1041–1050. [Google Scholar] [CrossRef] [PubMed]
- Taylor, M.A. Stomach stones for feeding or buoyancy? The occurrence and function of gastroliths in marine tetrapods. Philos. Trans. R. Soc. London. Ser. B Biol. Sci. 1993, 341, 163–175. [Google Scholar]
- Henderson, D.M. Floating point: A computational study of buoyancy, equilibrium, and gastroliths in plesiosaurs. Lethaia 2006, 39, 227–244. [Google Scholar] [CrossRef]
- Wings, O. A review of gastrolith function with implications for fossil vertebrates and a revised classification. Acta Palaeontol. Pol. 2007, 52, 1–16. [Google Scholar]
- Lucas, K.N.; Thornycroft, P.J.; Gemmell, B.J.; Colin, S.P.; Costello, J.H.; Lauder, G.V. Effects of non-uniform stiffness on the swimming performance of a passively-flexing, fish-like foil model. Bioinspiration Biomim. 2015, 10, 056019. [Google Scholar] [CrossRef] [PubMed]
- Lauder, G.V.; Lim, J.; Shelton, R.; Witt, C.; Anderson, E.; Tangorra, J.L. Robotic models for studying undulatory locomotion in fishes. Mar. Technol. Soc. J. 2011, 45, 41–55. [Google Scholar] [CrossRef]
- Gvozdik, L.; Damme, R.V.; Damme, R.V. Triturus newts defy the running—swimming dilemma. Evolution 2006, 60, 2110–2121. [Google Scholar] [CrossRef]
- Godoy-Diana, R.; Thiria, B. On the diverse roles of fluid dynamic drag in animal swimming and flying. J. R. Soc. Interface 2018, 15, 20170715. [Google Scholar] [CrossRef]
- Glaser, R. Biophysics: An Introduction; Springer Science & Business Media: Berlin, Heidelberg, 2012. [Google Scholar]
- Hone, D.W.; Holtz Jr, T.R. Evaluating the ecology of Spinosaurus: Shoreline generalist or aquatic pursuit specialist? Palaeontol. Electron. 2021, 24, a03. [Google Scholar]
- Lauder, G.V.; Madden, P.G. Fish locomotion: Kinematics and hydrodynamics of flexible foil-like fins. Exp. Fluids 2007, 43, 641–653. [Google Scholar] [CrossRef]
- Carpenter, K. The Carnivorous Dinosaurs; Indiana University Press: Bloomington, IN, USA, 2005. [Google Scholar]
- Sereno, P.C.; Beck, A.L.; Dutheil, D.B.; Gado, B.; Larsson, H.C.; Lyon, G.H.; Marcot, J.D.; Rauhut, O.W.; Sadleir, R.W.; Sidor, C.A. A long-snouted predatory dinosaur from Africa and the evolution of spinosaurids. Science 1998, 282, 1298–1302. [Google Scholar] [CrossRef] [PubMed]
- Aureliano, T.; Ghilardi, A.M.; Buck, P.V.; Fabbri, M.; Samathi, A.; Delcourt, R.; Fernandes, M.A.; Sander, M. Semi-aquatic adaptations in a spinosaur from the Lower Cretaceous of Brazil. Cretac. Res. 2018, 90, 283–295. [Google Scholar] [CrossRef]
- Busbey, A.B.; Thomason, J. The structural consequences of skull flattening in crocodilians. In Functional morphology in vertebrate paleontology; Thomason, J.J., Ed.; Cambridge University Press: Cambridge, UK, 1995; pp. 173–192. [Google Scholar]
- Thomason, J.; Russell, A. Mechanical factors in the evolution of the mammalian secondary palate: A theoretical analysis. J. Morphol. 1986, 189, 199–213. [Google Scholar] [CrossRef]
- Rayfield, E.J.; Milner, A.C.; Xuan, V.B.; Young, P.G. Functional morphology of spinosaur ‘crocodile-mimic’dinosaurs. J. Vertebr. Paleontol. 2007, 27, 892–901. [Google Scholar] [CrossRef]
- Cuff, A.R.; Rayfield, E.J. Feeding mechanics in spinosaurid theropods and extant crocodilians. PLoS ONE 2013, 8, e65295. [Google Scholar] [CrossRef] [PubMed]
- Palmer, C.; Young, M.T. Surface drag reduction and flow separation control in pelagic vertebrates, with implications for interpreting scale morphologies in fossil taxa. R. Soc. Open Sci. 2015, 2, 140163. [Google Scholar] [CrossRef][Green Version]
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gimsa, J.; Gimsa, U. Contributions to a Discussion of Spinosaurus aegyptiacus as a Capable Swimmer and Deep-Water Predator. Life 2021, 11, 889. https://doi.org/10.3390/life11090889
Gimsa J, Gimsa U. Contributions to a Discussion of Spinosaurus aegyptiacus as a Capable Swimmer and Deep-Water Predator. Life. 2021; 11(9):889. https://doi.org/10.3390/life11090889
Chicago/Turabian StyleGimsa, Jan, and Ulrike Gimsa. 2021. "Contributions to a Discussion of Spinosaurus aegyptiacus as a Capable Swimmer and Deep-Water Predator" Life 11, no. 9: 889. https://doi.org/10.3390/life11090889
APA StyleGimsa, J., & Gimsa, U. (2021). Contributions to a Discussion of Spinosaurus aegyptiacus as a Capable Swimmer and Deep-Water Predator. Life, 11(9), 889. https://doi.org/10.3390/life11090889





