Mitogenomics, Phylogeny and Morphology Reveal Ophiocordyceps pingbianensis Sp. Nov., an Entomopathogenic Fungus from China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Materials and Isolation
2.2. Morphological Observations
2.3. DNA Extraction, PCR and Sequencing of Nuclear Genes
2.4. Sequencing, Assembly and Annotation of Mitogenome
2.5. Sequence Analyses of Mitogenome
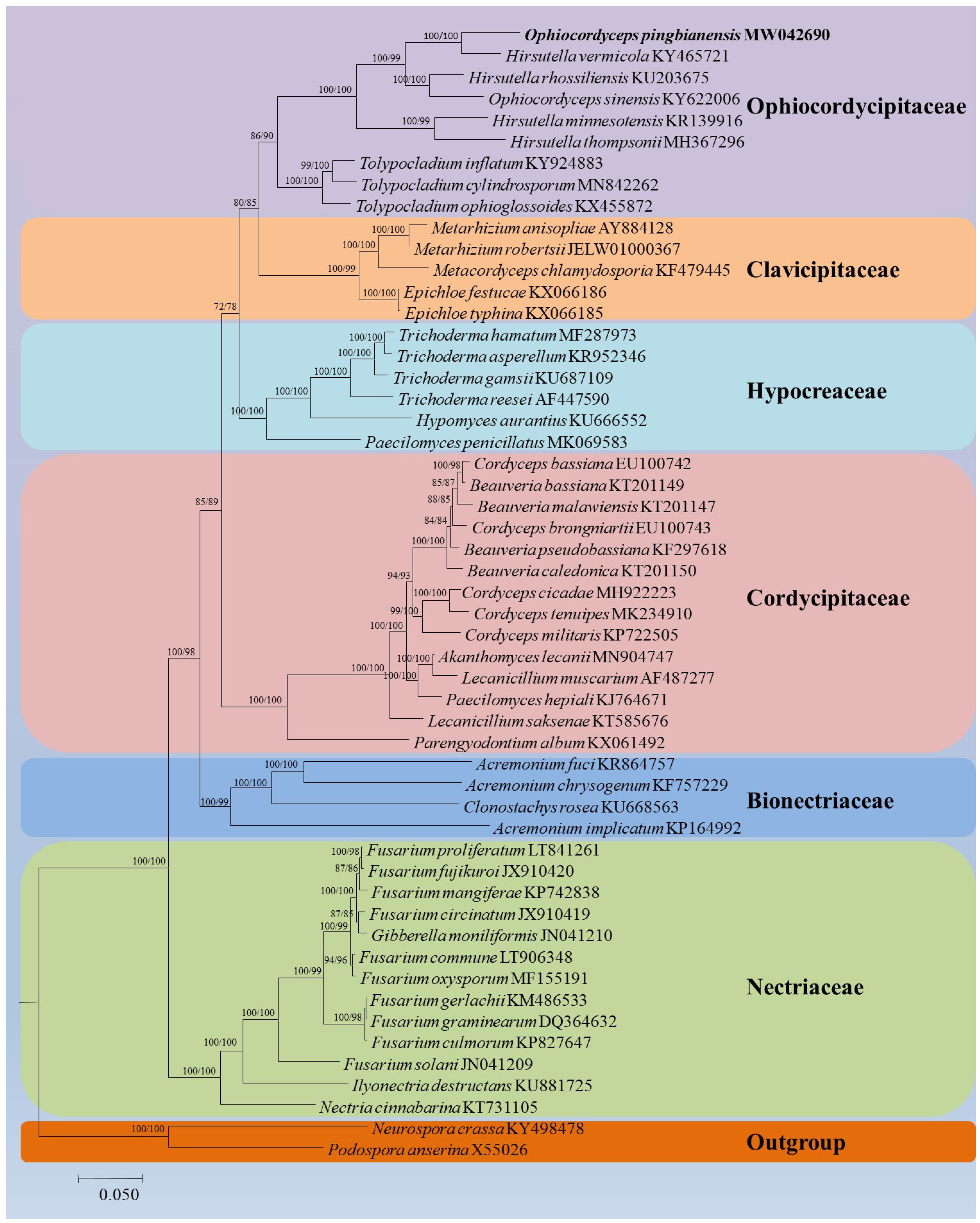
2.6. Phylogenetic Analyses
3. Results
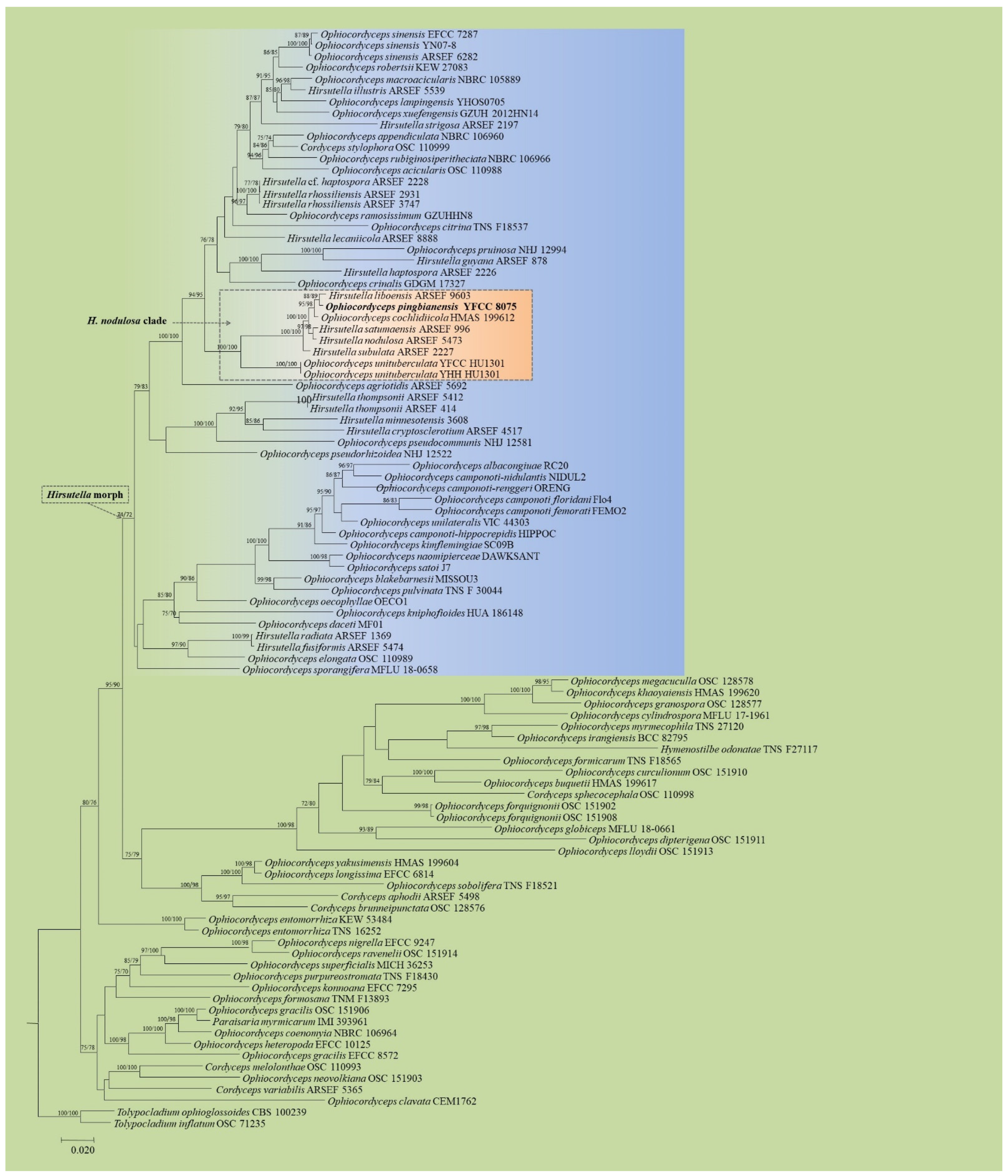
3.1. Phylogenetic Analyses Based on Nuclear Genes
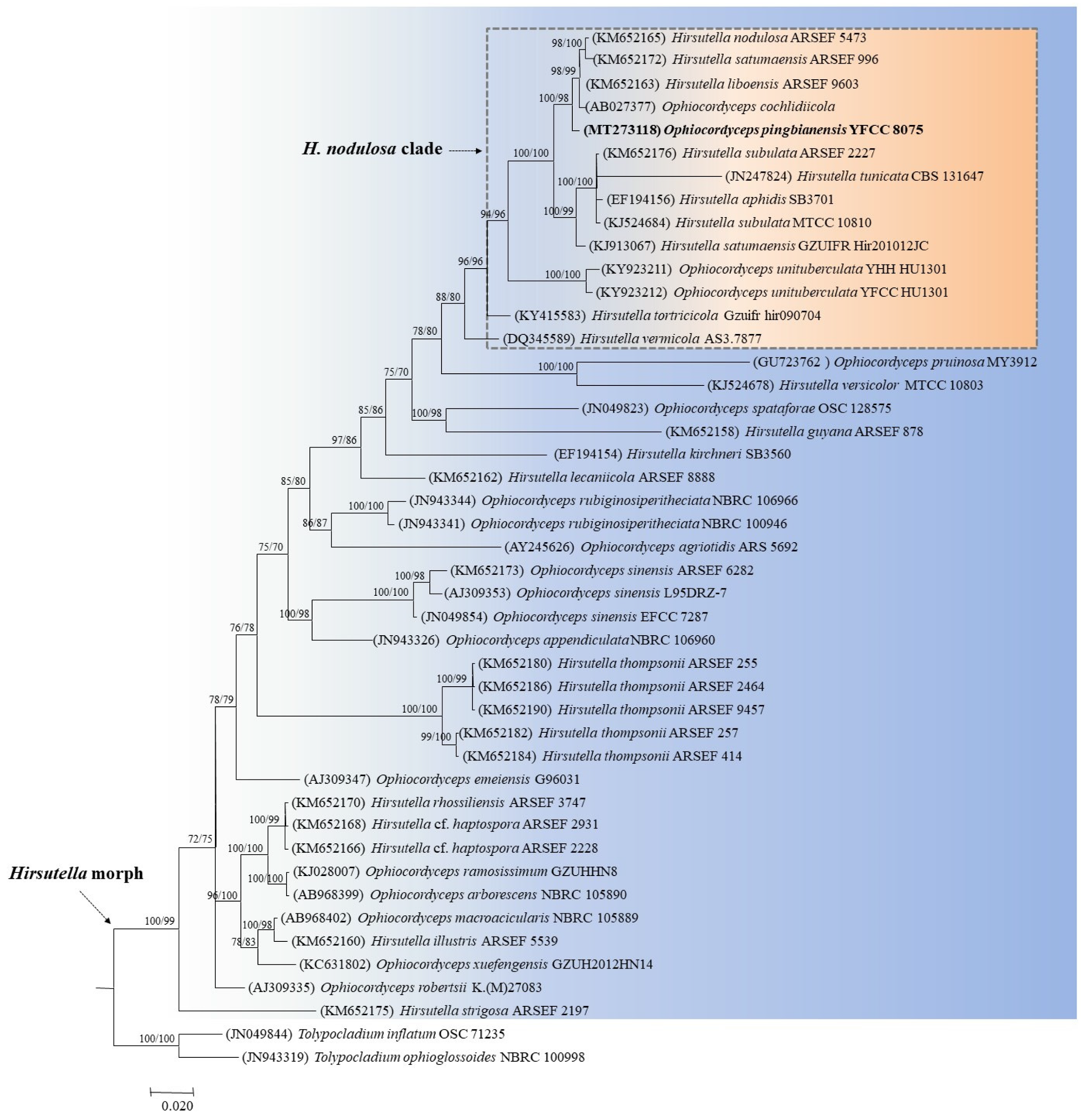
3.2. Phylogenetic Analysis Based on Mitochondrial Genes
3.3. Taxonomy
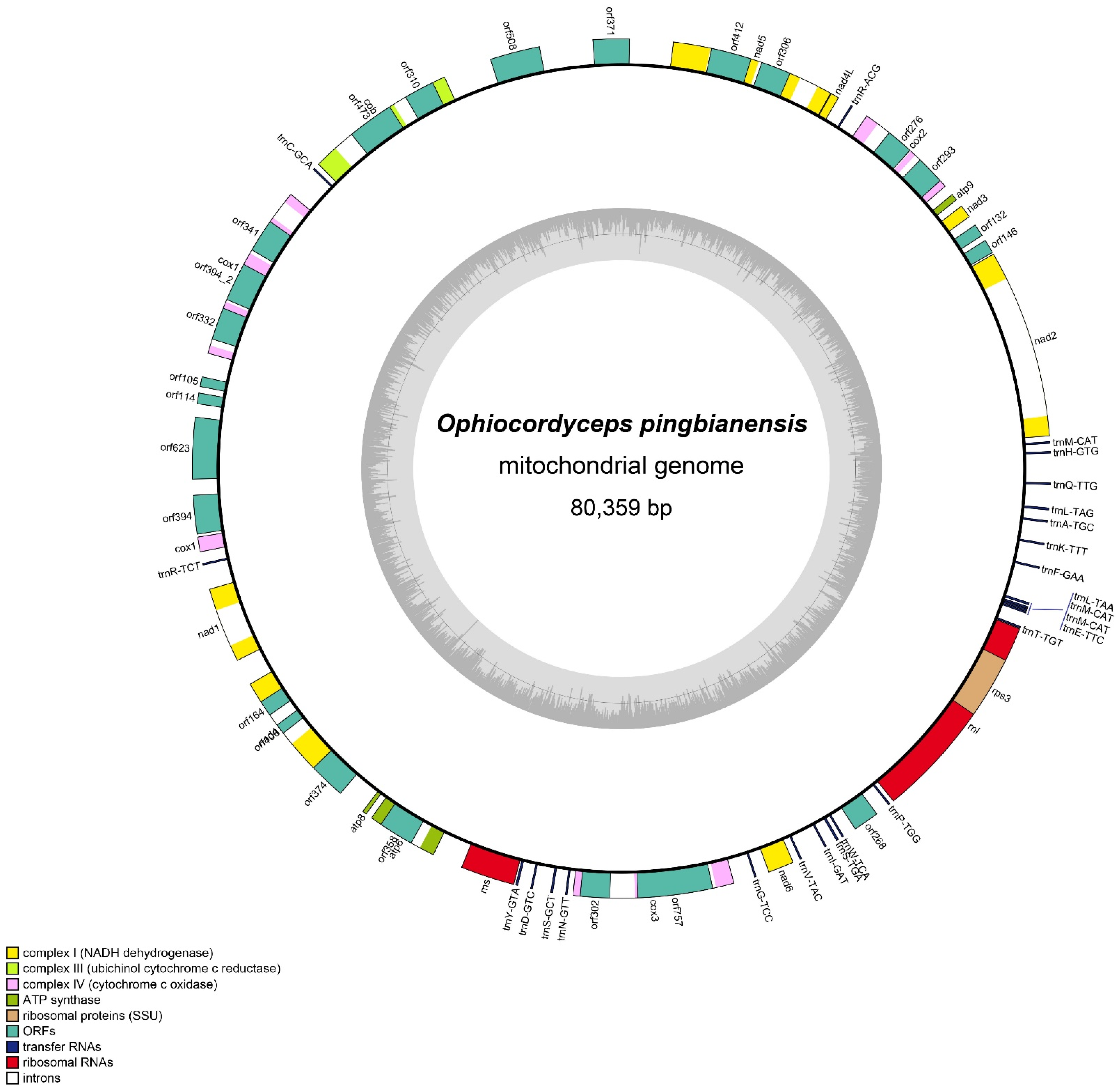
3.4. Mitogenomic Characteristics of Ophiocordyceps Pingbianensis
3.5. Codon Usage
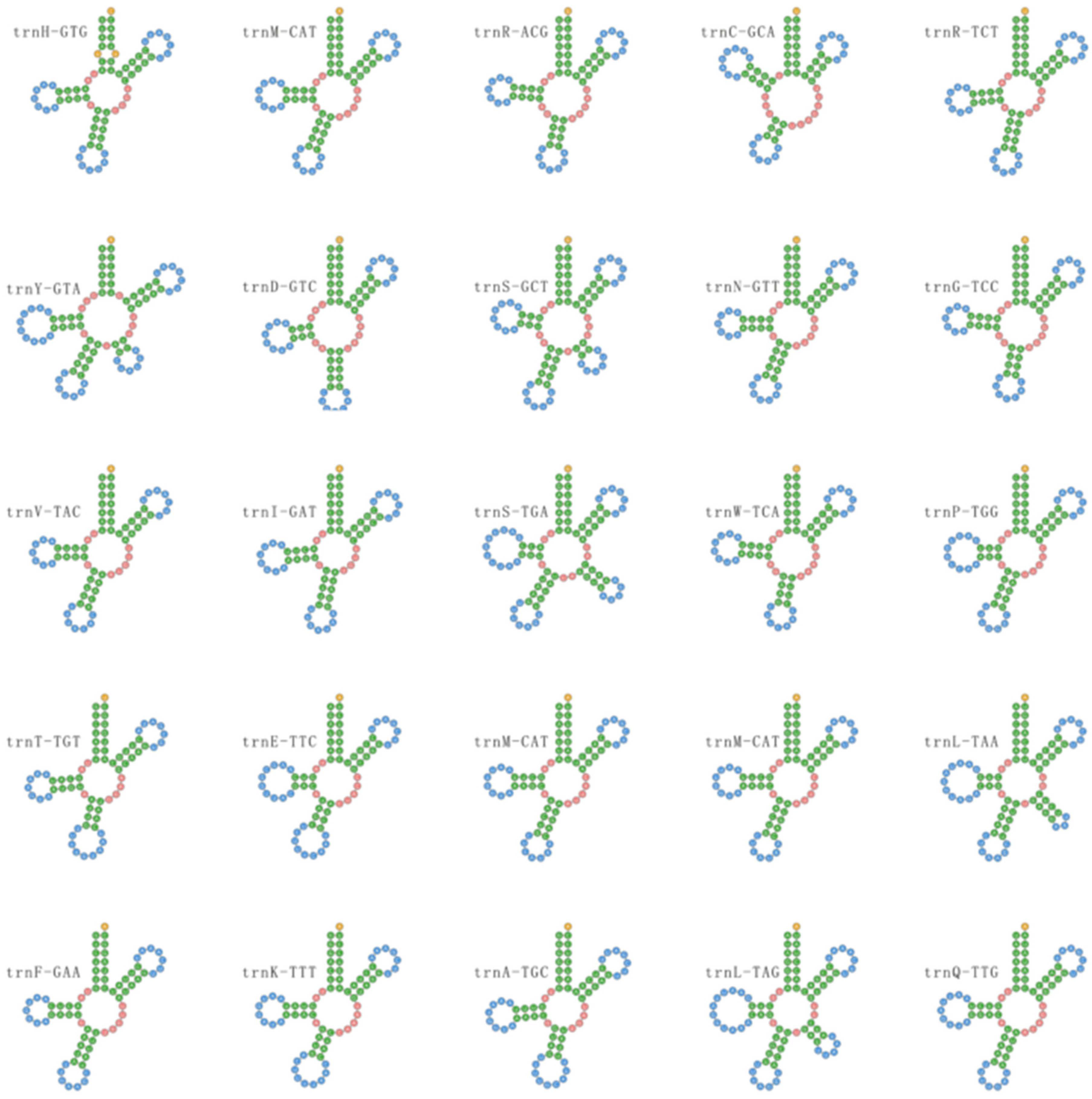
3.6. Transfer RNAs
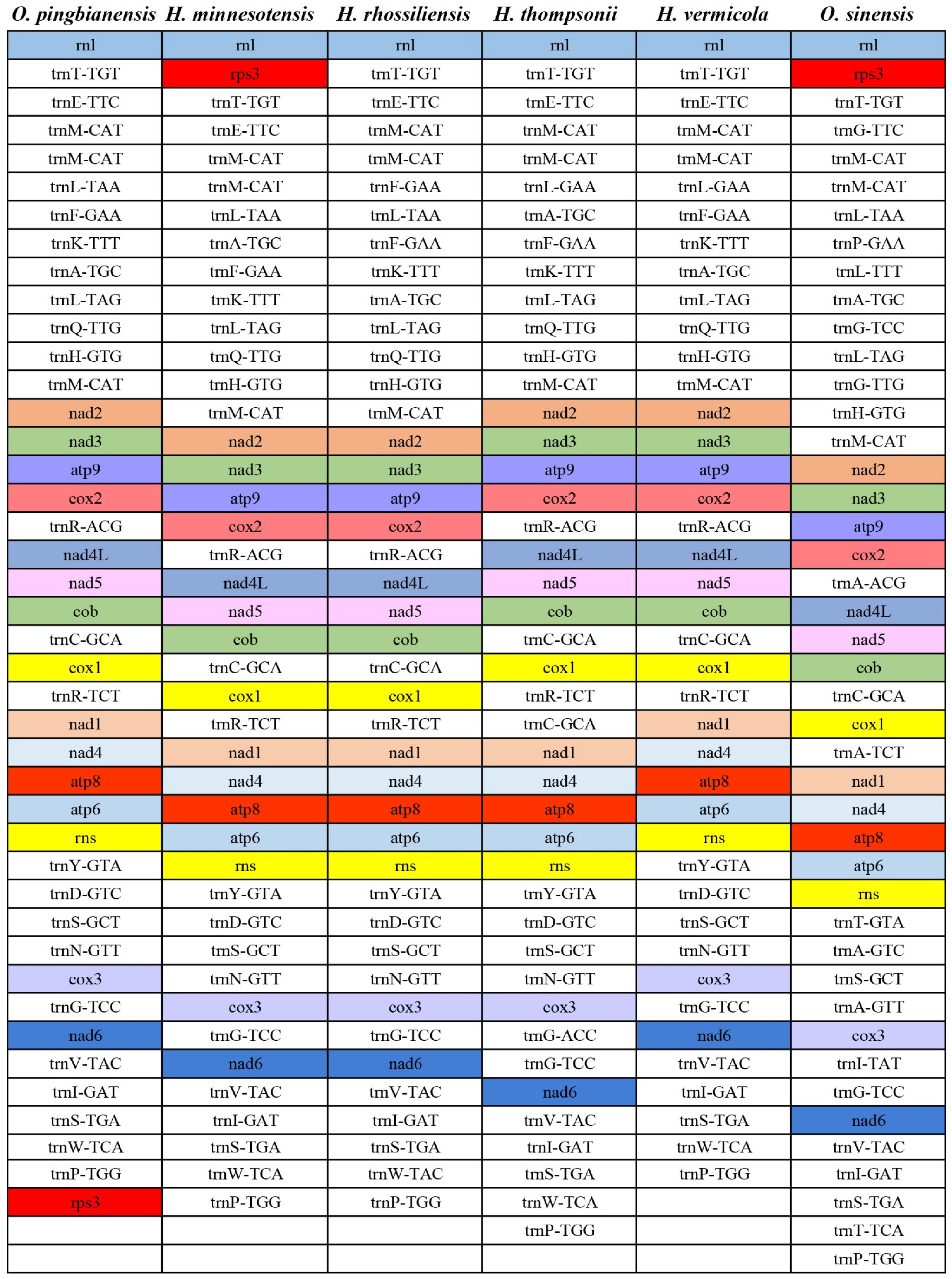
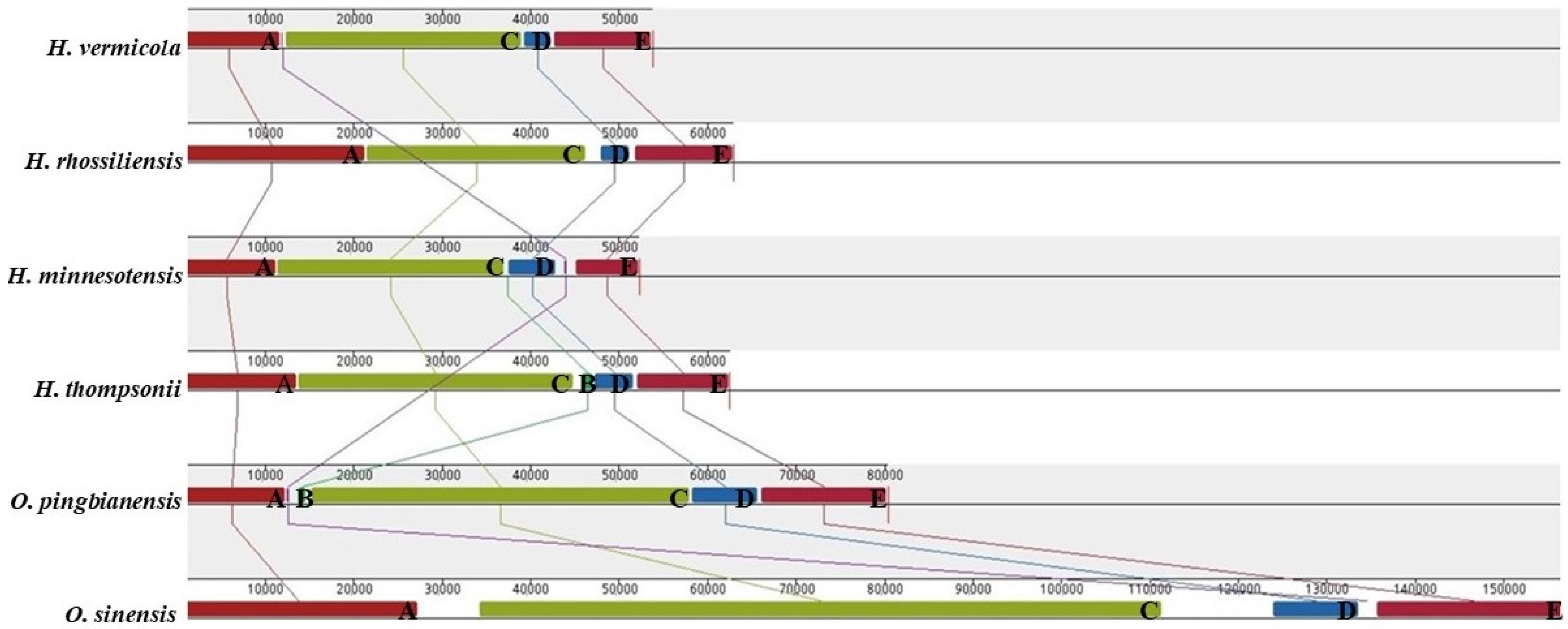
3.7. Gene Arrangement Analysis
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Sung, G.H.; Hywel-Jones, N.L.; Sung, J.M.; Luangsa-Ard, J.J.; Shrestha, B.; Spatafora, J.W. Phylogenetic classification of Cordyceps and the clavicipitaceous fungi. Stud. Mycol. 2007, 57, 5–59. [Google Scholar] [CrossRef] [Green Version]
- Petch, T. Notes on entomogenous fungi. Trans. Brit. Mycol. Soc. 1931, 16, 55–75. [Google Scholar] [CrossRef]
- Shrestha, B.; Zhang, W.M.; Zhang, Y.J.; Liu, X.Z. What is the Chinese caterpillar fungus Ophiocordyceps sinensis (Ophiocordycipitaceae)? Mycology 2010, 1, 228–236. [Google Scholar] [CrossRef] [Green Version]
- Kobayasi, Y. The genus Cordyceps and its allies. Science reports of the Tokyo Bunrika Daigaku. Sci. Rep. Tokyo Bun. Daigaku 1941, 5, 53–260. [Google Scholar]
- Kobayasi, Y. Keys to the taxa of the genera Cordyceps and Torrubiella. Trans. Mycol. Soc. Jpn. 1982, 23, 329–364. [Google Scholar]
- Mains, E.B. North American entomogenous species of Cordyceps. Mycologia 1958, 50, 169–222. [Google Scholar] [CrossRef]
- Sanjuan, T.I.; Fanco-Molano, A.E.; Kepler, R.M.; Spatafora, J.W.; Tabima, J.; Vasco-palacios, A.M.; Restrepo, S. Five new species of entomopathogenic fungi from the Amazon and evolution of neotropical Ophiocordyceps. Fungal Biol. 2015, 119, 901–916. [Google Scholar] [CrossRef] [Green Version]
- Spatafora, J.W.; Quandt, C.A.; Kepler, R.M.; Sung, G.H.; Shrestha, B.; Hywel-Jones, N.L.; Luangsa-ard, J.J. New 1F1N species combinations in Ophiocordycipitaceae (Hypocreales). IMA Fungus 2015, 6, 357–362. [Google Scholar] [CrossRef]
- Araújo, J.P.M.; Evans, H.C.; Kepler, R.M.; Hughes, D.P. Zombie-ant fungi across continents: 15 new species and new combinations within Ophiocordyceps. I. Myrmecophilous hirsutelloid species. Stud. Mycol. 2018, 90, 119–160. [Google Scholar] [CrossRef]
- Khonsanit, A.; Luangsa-ard, J.J.; Thanakitpipattana, D.; Kobmoo, N.; Piasai1, O. Cryptic species within Ophiocordyceps myrmecophila complex on formicine ants from Thailand. Mycol. Prog. 2018, 18, 147–161. [Google Scholar] [CrossRef]
- Luangsa-ard, J.J.; Tasanathai, K.; Thanakitpipattana, D.; Khonsanit, A.; Stadler, M. Novel and interesting Ophiocordyceps spp. (Ophiocordycipitaceae, Hypocreales) with superficial perithecia from Thailand. Stud. Mycol. 2018, 89, 125–142. [Google Scholar] [CrossRef] [PubMed]
- Wijayawardene, N.N.; Hyde, K.D.; Lumbsch, H.T.; Liu, J.K.; Maharachchikumbura, S.S.; Ekanayaka, A.H.; Tian, Q.; Phookamsak, R. Outline of Ascomycota 2017. Fungal Divers. 2018, 88, 167–263. [Google Scholar] [CrossRef]
- Tasanathai, K.; Noisripoom, W.; Chaitika, T.; Khonsanit, A.; Hasin, S.; Luangsa-Ard, J.J. Phylogenetic and morphological classification of Ophiocordyceps species on termites from Thailand. MycoKeys 2019, 56, 101–129. [Google Scholar] [CrossRef] [PubMed]
- Quandt, C.A.; Kepler, R.M.; Gams, W.; Araújo, J.P.; Ban, S.; Evans, H.C.; Luangsa-Ard, J.J. Phylogenetic-based nomenclatural proposals for Ophiocordycipitaceae (Hypocreales) with new combinations in Tolypocladium. IMA Fungus 2014, 5, 121–134. [Google Scholar] [CrossRef]
- Ban, S.; Sakane, T.; Nakagiri, A. Three new species of Ophiocordyceps and overview of anamorph types in the genus and the family Ophiocordyceptaceae. Mycol. Prog. 2015, 14, 1017. [Google Scholar] [CrossRef]
- Hodge, K.T. Revisionary Studies in Hirsutella (Anamorphic Hyphomycetes: Clavicipitaceae). Ph.D. Thesis, Cornell University, New York, NY, USA, 1998. [Google Scholar]
- Simmons, D.R.; Kepler, R.M.; Rehner, S.A.; Groden, E. Phylogeny of Hirsutella species (Ophiocordycipitaceae) from the USA: Remedying the paucity of Hirsutella sequence data. IMA Fungus 2015, 6, 345–356. [Google Scholar] [CrossRef]
- Wang, Y.B.; Nguyen, T.T.; Dai, Y.D.; Yu, H.; Wu, C.K. Molecular phylogeny and morphology of Ophiocordyceps unituberculata sp. nov. (Ophiocordycipitaceae), a pathogen of caterpillars (Noctuidae, Lepidoptera) from Yunnan, China. Mycol. Prog. 2018, 17, 745–753. [Google Scholar] [CrossRef]
- Williams, S.T.; Foster, P.G.; Littlewood, D.T. The complete mitochondrial genome of a turbinid vetigastropod from MiSeq Illumina sequencing of genomic DNA and steps towards a resolved gastropod phylogeny. Gene 2014, 533, 38–47. [Google Scholar] [CrossRef]
- Botero-Castro, F.; Tilak, M.K.; Justy, F.; Catzeflis, F.; Delsuc, F.; Douzery, E.J. Next generation sequencing and phylogenetic signal of complete mitochondrial genomes for resolving the evolutionary history of leaf-nosed bats (Phyllostomidae). Mol. Phylogenet. Evol. 2013, 69, 728–739. [Google Scholar] [CrossRef]
- James, T.Y.; Pelin, A.; Bonen, L.; Ahrendt, S.; Sain, D.; Corradi, N.; Stajich, J.E. Shared signatures of parasitism and phylogenomics unite the Cryptomycota and Microsporidia. Curr. Biol. 2013, 23, 1548–1553. [Google Scholar] [CrossRef] [Green Version]
- Liu, W.; Cai, Y.L.; Zhang, Q.Q.; Chen, L.F.; Shu, F.; Ma, X.L.; Bian, Y.B. The mitochondrial genome of Morchella importuna (272.2 kb) is the largest among fungi and contains numerous introns, mitochondrial non-conserved open reading frames and repetitive sequences. Int. J. Biol. Macromol. 2019, 143, 373–381. [Google Scholar] [CrossRef]
- Kang, X.; Hu, L.; Shen, P.; Li, R.; Liu, D. SMRT sequencing revealed mitogenome characteristics and mitogenome-wide DNA modification pattern in Ophiocordyceps sinensis. Front. Microbiol. 2017, 8, 1422. [Google Scholar] [CrossRef]
- Zhang, Y.J.; Zhang, S.; Liu, X. The complete mitochondrial genome of the nematode endoparasitic fungus Hirsutella minnesotensis. Mitochondrial DNA Part A 2016, 27, 2693–2694. [Google Scholar] [CrossRef] [PubMed]
- Zhang, Y.J.; Wang, L.; Zhang, S.; Li, J.H. Mitochondrial genome, comparative analysis and evolutionary insights into the entomopathogenic fungus Hirsutella thompsonii. Environ. Microbiol. 2018, 20, 3393–3405. [Google Scholar] [CrossRef]
- Wang, Y.B.; Wang, Y.; Fan, Q.; Duan, D.E.; Zhang, G.D.; Dai, R.Q.; Dai, Y.D.; Zeng, W.B.; Chen, Z.H.; Li, D.D.; et al. Multigene phylogeny of the family Cordycipitaceae (Hypocreales): New taxa and the new systematic position of the Chinese cordycipitoid fungus Paecilomyces hepiali. Fungal Divers. 2020, 103, 1–46. [Google Scholar] [CrossRef]
- Liu, X.Z.; Chen, S.Y. Screening isolates of Hirsutella species for biocontrol of Heterodera glycines. Biocontrol. Sci. Tech. 2001, 11, 151–160. [Google Scholar] [CrossRef]
- Wang, Y.B.; Yu, H.; Dai, Y.D.; Wu, C.K.; Zeng, W.B.; Yuan, F.; Liang, Z.Q. Polycephalomyces agaricus, a new hyperparasite of Ophiocordyceps sp. infecting melolonthid larvae in southwestern China. Mycol. Prog. 2015, 14, 70. [Google Scholar] [CrossRef]
- Vilgalys, R.; Hester, M. Rapid genetic identification and mapping of enzymatically amplified ribosomal DNA from several Cryptococcus species. J. Bacteriol. 1990, 172, 4238–4246. [Google Scholar] [CrossRef] [Green Version]
- Rehner, S.A.; Samuels, G.J. Taxonomy and phylogeny of Gliocladium analysed from nuclear large subunit ribosomal DNA sequences. Mycol. Res. 1994, 98, 625–634. [Google Scholar] [CrossRef]
- Bischoff, J.F.; Rehner, S.A.; Humber, R.A. Metarhizium frigidum sp. nov.: A cryptic species of M. anisopliae and a member of the M. flavoviride Complex. Mycologia 2006, 98, 737–745. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.D.; Lee, S.B.; Taylor, J.W. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In Pcr Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Wang, Y.B.; Yu, H.; Dai, Y.D.; Chen, Z.H.; Zeng, W.B.; Yuan, F.; Liang, Z.Q. Polycephalomyces yunnanensis (Hypocreales), a new species of Polycephalomyces parasitizing Ophiocordyceps nutans and stink bugs (hemipteran adults). Phytotaxa 2015, 208, 34–44. [Google Scholar] [CrossRef]
- Bankevich, A.; Nurk, S.; Antipov, D.; Gurevich, A.A.; Dvorkin, M.; Kulikov, A.S.; Lesin, V.M.; Nikolenko, S.L.; Pham, S.; Prjibelski, A.D.; et al. SPAdes: A new genome assembly algorithm and its applications to single-cell sequencing. J. Comput. Biol. 2012, 19, 455–477. [Google Scholar] [CrossRef] [Green Version]
- Valach, M.; Burger, G.; Gray, M.W.; Lang, B.F. Widespread occurrence of organelle genome-encoded 5S rRNAs including permuted molecules. Nucleic Acids Res. 2014, 42, 13764–13777. [Google Scholar] [CrossRef] [Green Version]
- Lohse, M.; Drechsel, O.; Bock, R. Organellargenomedraw (ogdraw): A tool for the easy generation of high-quality custom graphical maps of plastid and mitochondrial genomes. Curr. Genet. 2007, 52, 267–274. [Google Scholar] [CrossRef]
- Larkin, M.A.; Blackshields, G.; Brown, N.P.; Chenna, R.; McGettigan, P.A.; McWilliam, H.; Valentin, F.; Wallace, I.M.; Wilm, A.; Lopez, R.; et al. Clustal W and Clustal X version 2.0. Bioinformatics 2007, 23, 2947–2948. [Google Scholar] [CrossRef] [Green Version]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular Evolutionary Genetics Analysis Version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [Green Version]
- Edgar, R.C. MUSCLE: Multiple sequence alignment with high accuracy and high throughput. Nucleic Acids Res. 2004, 32, 1792–1797. [Google Scholar] [CrossRef] [Green Version]
- Stamatakis, A. RAxML-VI-HPC: Maximum likelihood-based phylogenetic analyses with thousands of taxa and mixed models. Bioinformatics 2006, 22, 2688–2690. [Google Scholar] [CrossRef] [PubMed]
- Ronquist, F.; Huelsenbeck, J.P. MrBayes 3: Bayesian phylogenetic inference under mixed models. Bioinformatics. 2003, 19, 1572–1574. [Google Scholar] [CrossRef] [Green Version]
- Brady, S. Notes on Hirsutella aphidis. Trans. Br. Mycol. Soc. 1985, 85, 752–756. [Google Scholar] [CrossRef]
- Zou, X.; Liu, A.Y.; Liang, Z.Q.; Han, Y.N.; Yang, M.F. Hirsutella liboensis, a new entomopathogenic species affecting Cossidae (Lepidoptera) in China. Mycotaxon 2010, 111, 34–44. [Google Scholar] [CrossRef]
- Minter, D.W.; Brady, B.L. Mononematous species of Hirsytella. Trans. Br. Mycol. Soc. 1980, 74, 271–282. [Google Scholar] [CrossRef]
- Aoki, K.; Nakasato, Y.; Fujimoto, I.; Suzuki, K. Studies on the new fungous parasites of silkworms, Bombyx Mori L. IV. Hirsutella satumaensis Aoki sp. nov. Bull. Seric. Exp. Stn. 1957, 14, 471–486. [Google Scholar]
- Zou, X.; Zhou, Y.M.; Liang, Z.Q.; Xu, F.L. A new species of the genus Hirsutella with helical twist neck of phialides parasitized on Tortricidae. Mycosystema 2016, 53, 807–813. [Google Scholar]
- Xiang, M.C.; Yang, E.C.; Xiao, Q.M.; Liu, X.Z.; Chen, S.Y. Hirsutella vermicola sp. nov., a new species parasitizing bacteria-feeding nematodes. Fungal Divers. 2006, 19, 217–234. [Google Scholar] [CrossRef]
- Wang, N.N.; Zhang, Y.J.; Hussain, M.; Li, K.; Xiang, M.H.; Liu, X.Z. The mitochondrial genome of the nematode endoparasitic fungus Hirsutella rhossiliensis. Mitochondrial DNA Part B 2016, 27, 2693–2694. [Google Scholar] [CrossRef] [Green Version]
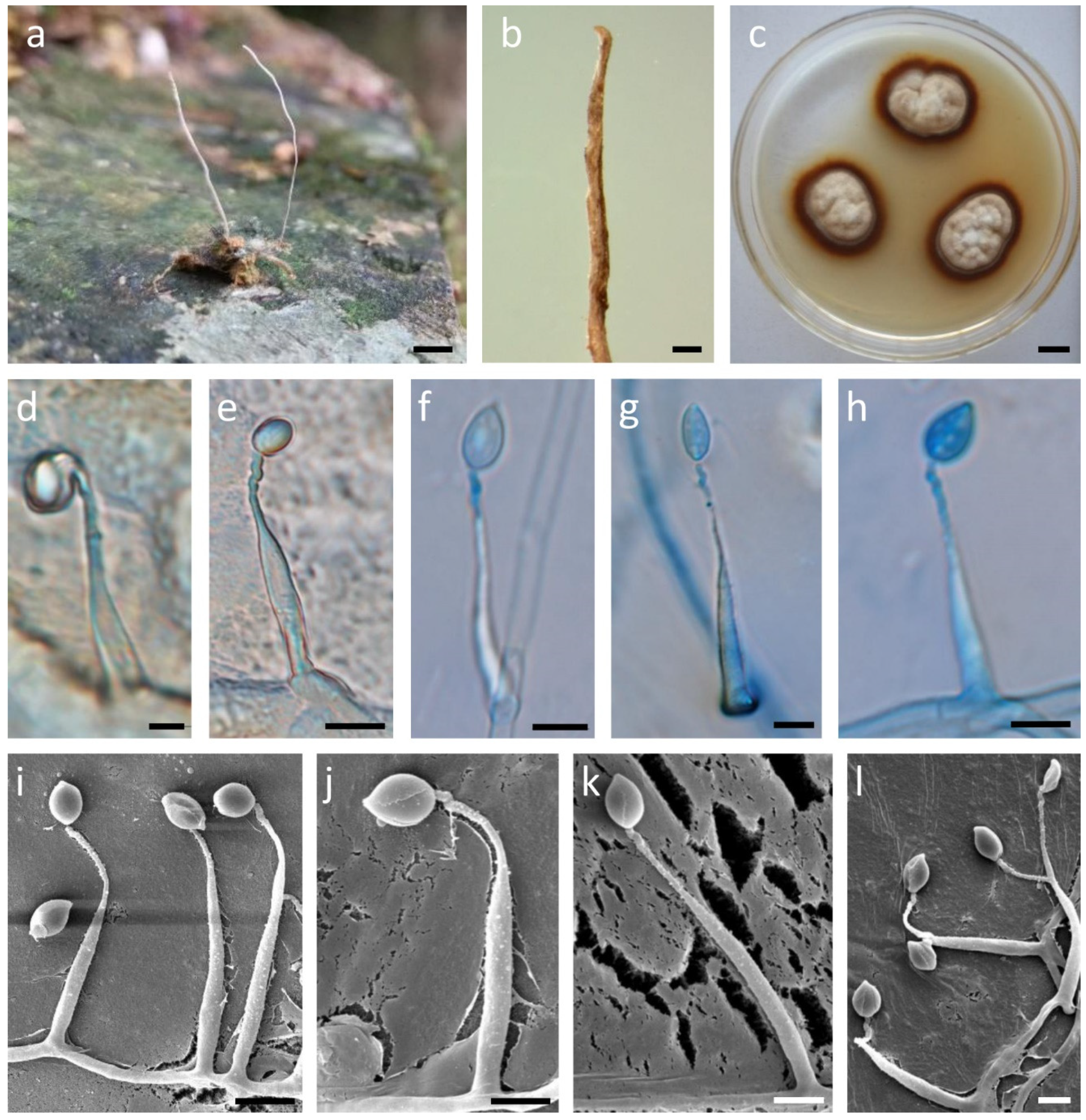
| Species | Host | Habitat | Synnemata/Stromata | Conidiogenous Cells | Conidia | References |
|---|---|---|---|---|---|---|
| H. aphidis | Aphids | On plant | Small or as short cushions | Narrowly conical, length (17–90 µm), neck width (0.6–1.0 µm), base width (2.6–4.0 µm) | Slightly crescent-shaped or cymbiform, usually in pairs, 7.0–18.3 × 2.8–4.6 µm, with mucous sheath | [42] |
| H. liboensis | Larvae of Cossidae (Lepidoptera) | In tree hole | Clustered | Base significantly swollen, length (28–30 µm), neck width (1–2 µm), twisting in 2–3 helices at the apex, base width (3–4.5 µm) | Fusiform or orange-segmented, in pairs or single, 6–8 × 3–5 µm, with mucous sheath | [43] |
| H. nodulosa | Lepidoptera, Acari | On plant | None | Base swollen, length (20–35 µm), base width (4 µm), neck often twists in a helix at the apex, neck width (1 µm), with tiny warts | Ellipsoid or like orange segments, 5–6 × 3 µm, with mucous sheath | [44] |
| O. pingbianensis | Larvae of tiger beetle (Coleoptera) | Clinging to fallen leaves | Geminate, 17–21 × 0.16–0.21 | Base obviously swollen, length (20.4–31.6 μm), base width (3.2–5.2 μm), neck width (1–1.5 µm), twisty and warty at the apex | Solitary, fusiform or oval, 5.3–7.5 µm long, 3.14.6 µm wide, with mucous sheath | This study |
| H. satumaensis | Larvae of Bombyx mori (Lepidoptera) | On plant | Clustered,3.0–6.5 × 0.5 mm. | With conoid to cylindrical base, size (5–17 × 3–4.5 μm), neck length (7 µm), twisting in a helix at the apex; base with warts | Fusiform or like orange segments, 5–7.5 × 3–5μm, with mucous sheath | [14,45] |
| H. subuluta | Larvae of Lepidoptera | On plant | Solitary, 15–50 × 0.1–0.3 mm | Phialidic, neck 6–12 µm long, base size (4–8 × 3–5 µm) | Narrowly ellipsoid, in pairs or single, 4–8 × 1.5–2.5 μm, with mucous sheath. | [16] |
| H. tortricicola | Larvae of Tortricidae (Lepidoptera) | In wilted leaf | Solitary | Base obviously swollen, length (18–22 µm), base width (3.5–4 µm), neck width (1–1.5 µm), neck often twists in 1–2 helices at the apex | Ellipsoid or like orange segments, in pairs or single, 2.7–3.6 × 1.4–1.8 μm, with mucous sheath | [46] |
| O. unituberculata | Larvae of Noctuidae (Lepidoptera) | In soil or cling to the fallen leaves | Clustered, 5–76 × 0.4–0.7 mm | With an inflated awl-shaped base, length (31.9–128.3 µm), base width (1.8–5.0 µm), neck width (0.5–1.2 µm). With a large periclinal protuberance near the apex | Solitary, lanceolate to fusiform, 6.3–10.6 × 1.9–3.7 µm, with mucous sheath | [18] |
| H. vermicola | Bacteria-feeding nematodes | In soil | None | Base obviously swollen, length (14–26 µm), base width (3.0–5.0 µm), neck width (1–2 µm), neck often twists in a helix towards the apex | More or less ellipsoid, single or in groups of 2–8 × 3–5 µm, with mucous sheath | [47] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, S.; Wang, Y.; Zhu, K.; Yu, H. Mitogenomics, Phylogeny and Morphology Reveal Ophiocordyceps pingbianensis Sp. Nov., an Entomopathogenic Fungus from China. Life 2021, 11, 686. https://doi.org/10.3390/life11070686
Chen S, Wang Y, Zhu K, Yu H. Mitogenomics, Phylogeny and Morphology Reveal Ophiocordyceps pingbianensis Sp. Nov., an Entomopathogenic Fungus from China. Life. 2021; 11(7):686. https://doi.org/10.3390/life11070686
Chicago/Turabian StyleChen, Siqi, Yuanbing Wang, Kongfu Zhu, and Hong Yu. 2021. "Mitogenomics, Phylogeny and Morphology Reveal Ophiocordyceps pingbianensis Sp. Nov., an Entomopathogenic Fungus from China" Life 11, no. 7: 686. https://doi.org/10.3390/life11070686
APA StyleChen, S., Wang, Y., Zhu, K., & Yu, H. (2021). Mitogenomics, Phylogeny and Morphology Reveal Ophiocordyceps pingbianensis Sp. Nov., an Entomopathogenic Fungus from China. Life, 11(7), 686. https://doi.org/10.3390/life11070686





