The Use of Response Surface Methodology as a Statistical Tool for the Optimisation of Waste and Pure Canola Oil Biodegradation by Antarctic Soil Bacteria
Abstract
1. Introduction
2. Materials and Methods
2.1. Sample Collection and Preparation
2.2. Screening of WCO and PCO Degradation by Bacterial Consortia
2.3. Optimisation of Bacterial Growth and Canola Oil Degradation through One-Factor-at-a-Time (OFAT)
2.4. Statistical Optimisation using Response Surface Methodology (RSM)
2.4.1. Screening for Significant Factors Using Plackett-Burman Design (PBD)
2.4.2. Optimisation of WCO and PCO Degradation Using Central Composite Design (CCD)
2.5. Statistical Analysis
3. Results
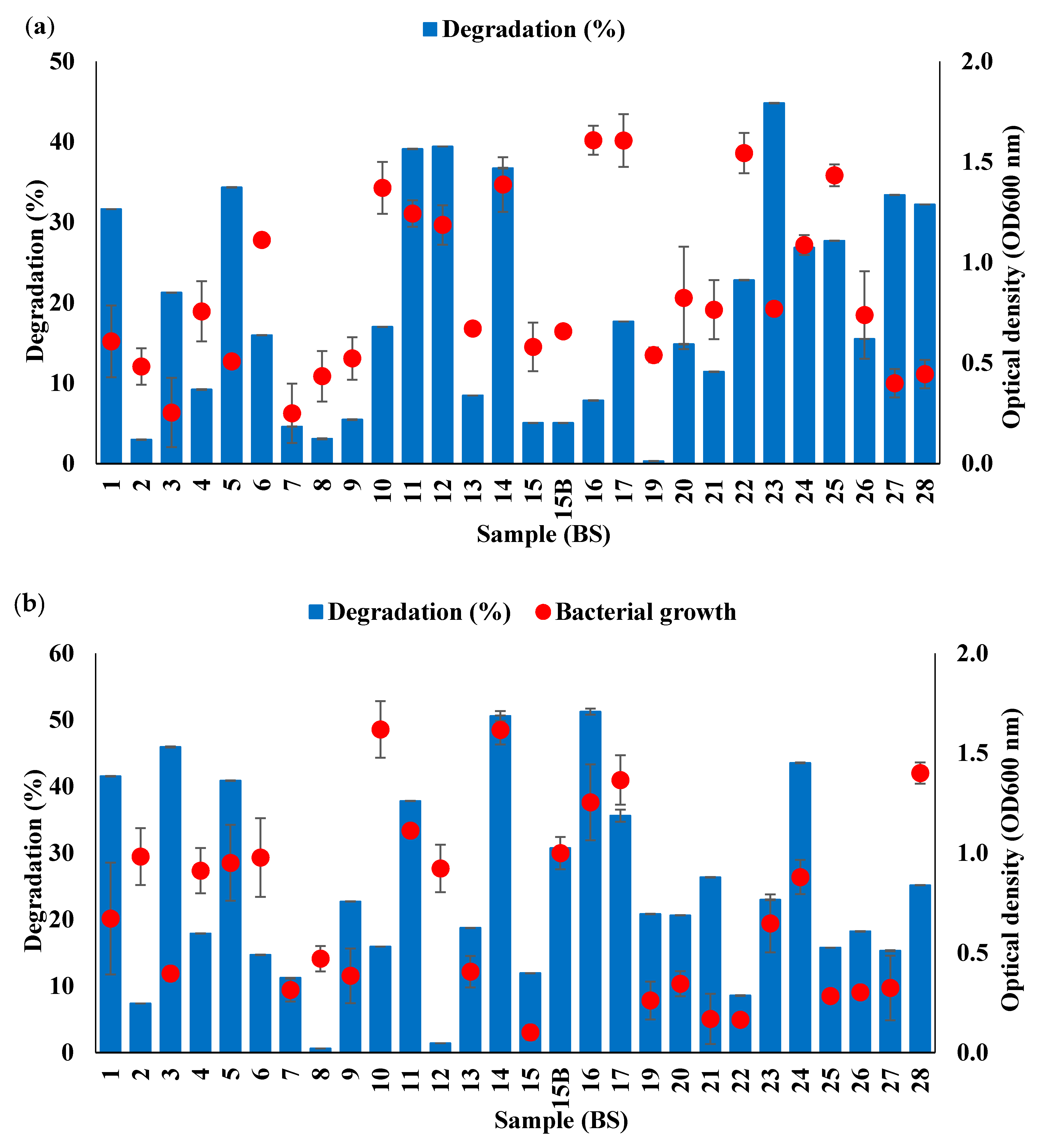
3.1. Screening of WCO and PCO-Degrading Antarctic Bacterial Consortia
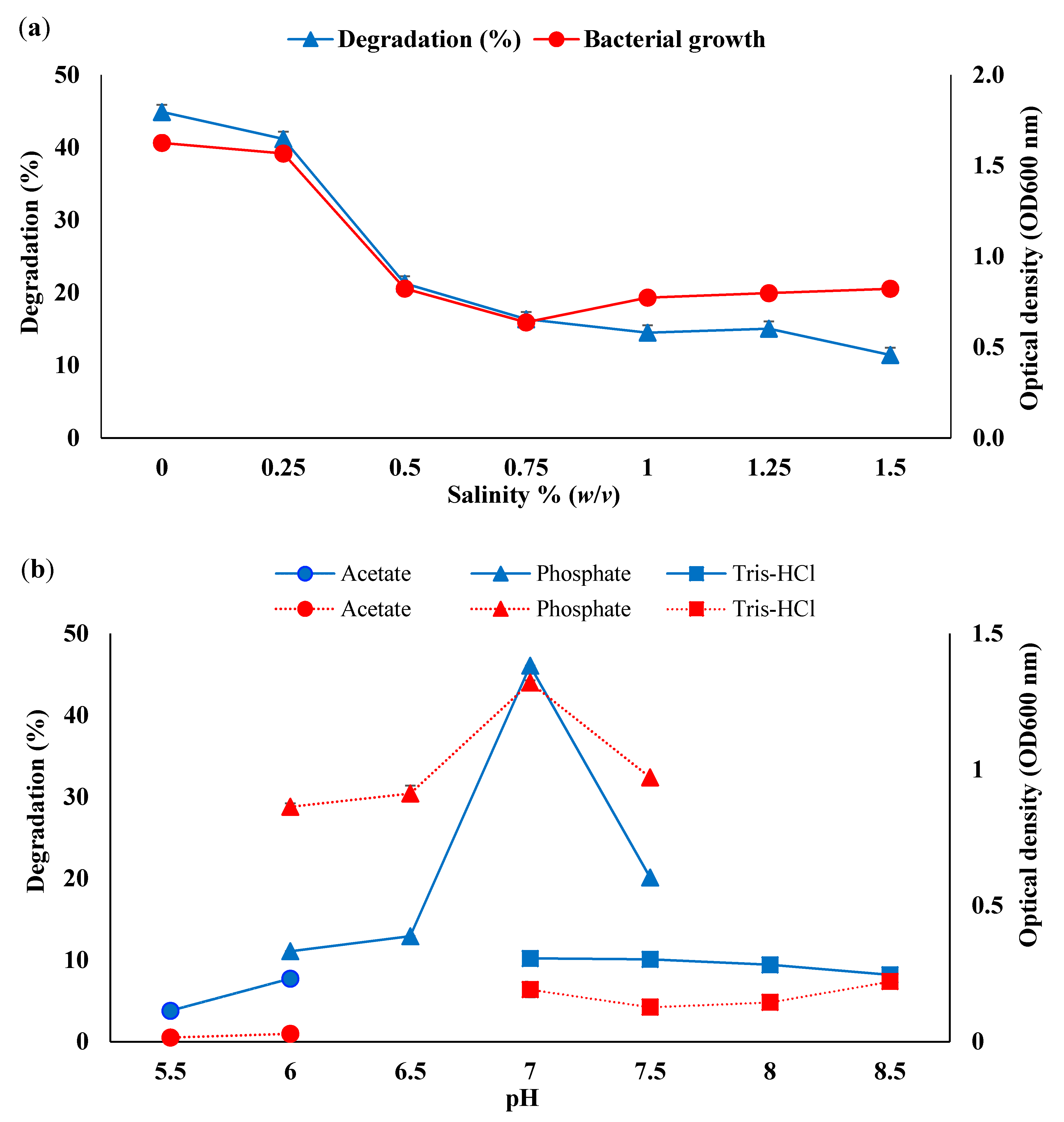
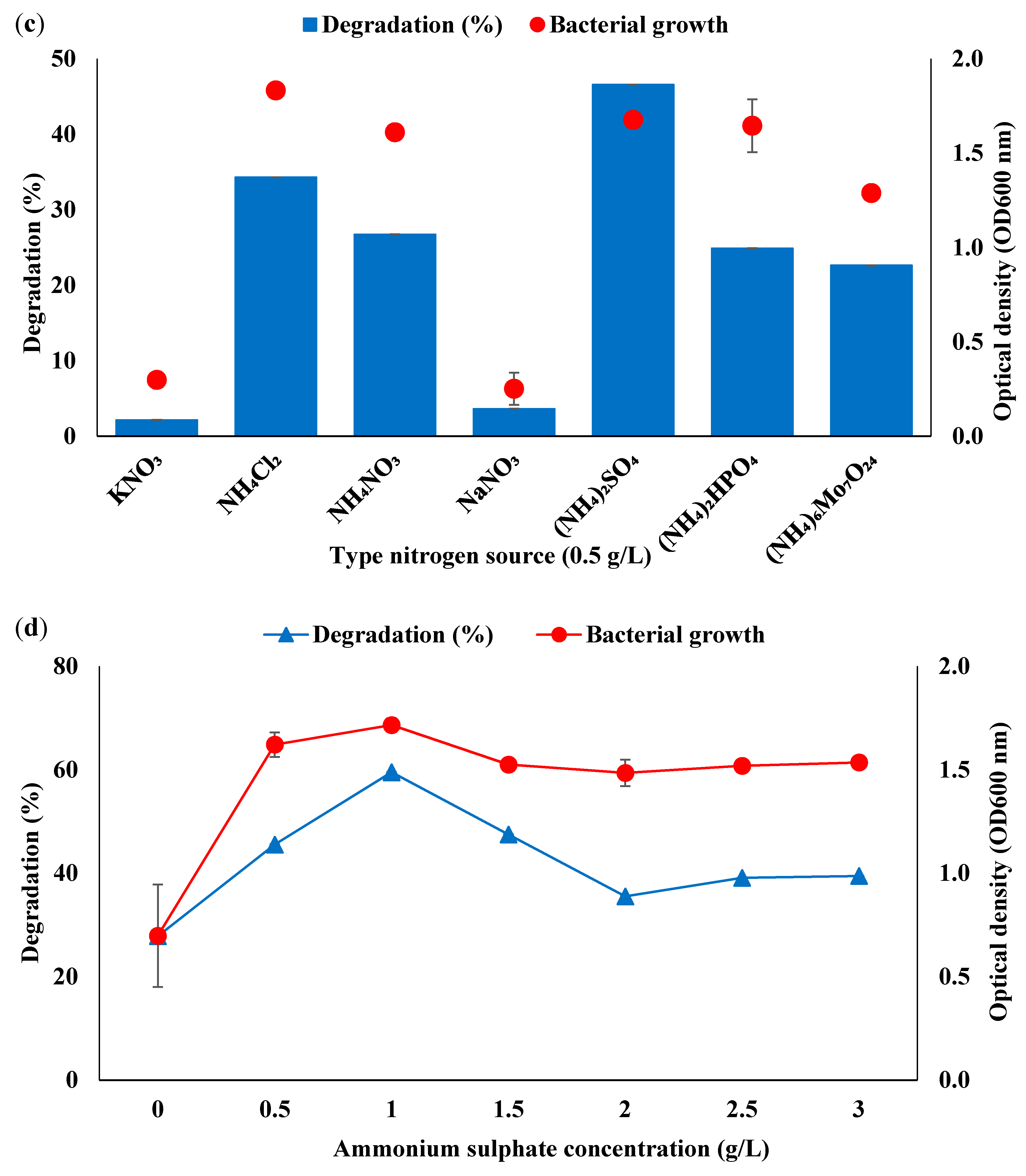
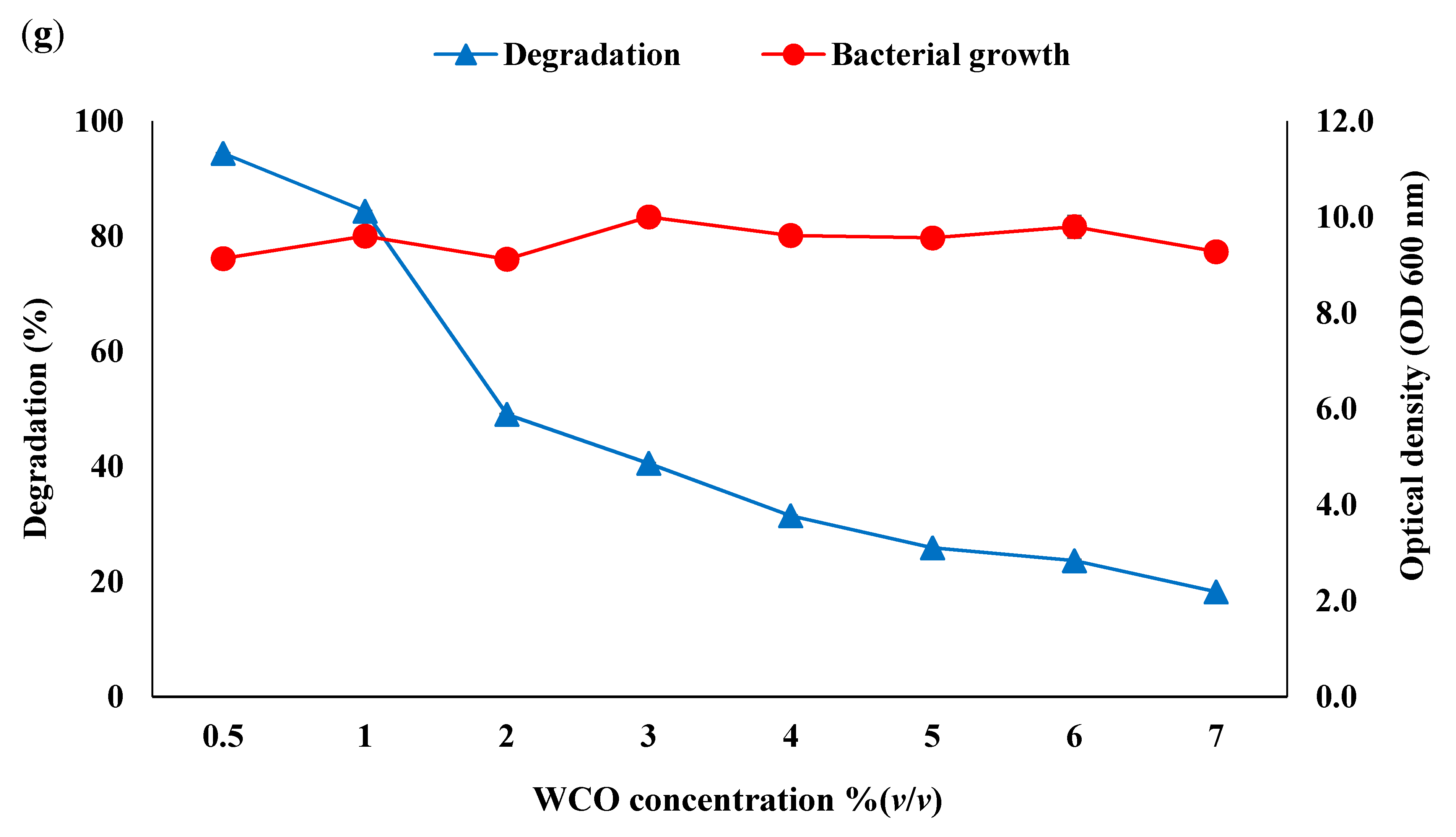
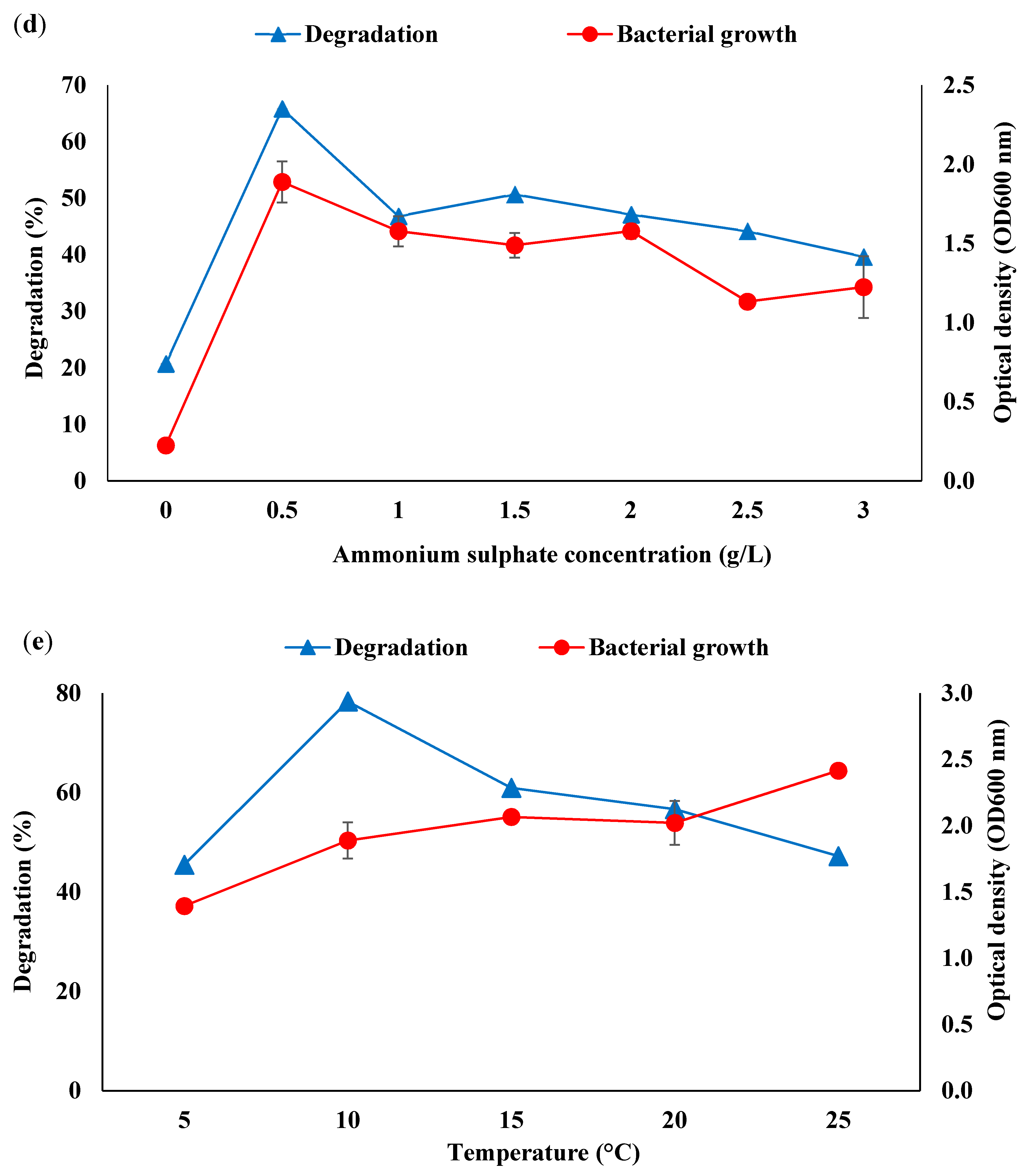
3.2. Optimisation of Consortium BS14 Growth and Canola Oil Degradation Using One-Factor-at-a-Time
3.3. Optimised Bacterial Growth and Degradation by Response Surface Method (RSM)
3.3.1. Plackett-Burman Design (PBD)
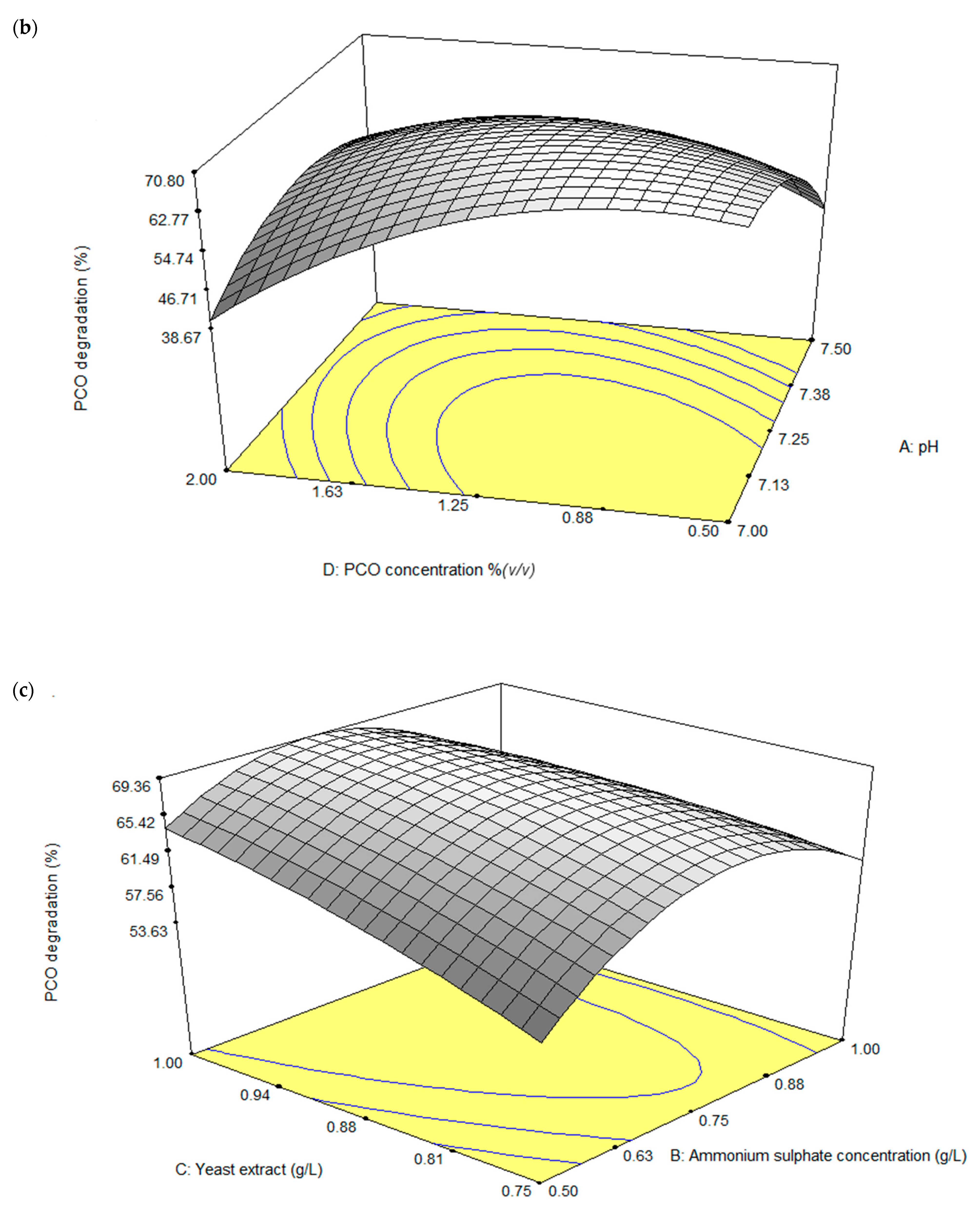
3.3.2. Central Composite Design (CCD)
4. Conclusions
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Shahbandeh, M. Vegetable Oils: Production Worldwide 2012/13–2019/20, by Type. 2020. Available online: https://www.statista.com/statistics/263933/production-of-vegetable-oils-worldwide-since-2000/#statisticContainer (accessed on 15 July 2020).
- Gunstone, F.D. Vegetable Oils in Foods Technology: Composition, Properties and Uses; Blackwell Publishing Ltd.: Hoboken, NJ, USA, 2002; pp. 107–133. [Google Scholar]
- Hageman, G.; Kikken, R.; Ten, H.F.; Kleinjans, J. Assessment of mutagenic activity of repeatedly used deep-frying fats. Mutat. Res. 1988, 204, 593–604. [Google Scholar] [CrossRef]
- Ananey-Obiri, D.; Matthews, L.; Azahrani, M.H.; Ibrahim, S.A.; Galanakis, C.M.; Tahergorabi, R. Application of protein-based edible coatings for fat uptake reduction in deep-fat fried foods with an emphasis on muscle food proteins. Trends Food Sci. Technol. 2018, 80, 167–174. [Google Scholar] [CrossRef]
- Australia Antarctic Division. Pollution and Waste. Department of the Environment and Energy. 2012. Available online: https://www.antarctica.gov.au (accessed on 12 December 2018).
- Hughes, K.A.; Constable, A.; Frenot, Y.; Lopez-Martinez, J.; Mclvor, E.; Njastad, B.; Terauds, A.; Liggett, D.; Roldan, G.; Wilmotte, A.; et al. Antarctic environmental protection: Strengthening the links between science and governance. Environ. Sci. Policy 2018, 83, 86–95. [Google Scholar] [CrossRef]
- Matheson, B.; Walker, K.Z.; Taylor, D.M.; Peterkin, R.; Lugg, D.; O’Dea, K. Effect on serum lipids of mono-saturated oil and margarine in the diet of an Antarctic expedition. Am. J. Clin. Nutr. 1996, 63, 933–938. [Google Scholar] [CrossRef] [PubMed]
- Antarctic Treaty, Waste Disposal and Waste Management. 2014. Available online: www.ats.aq (accessed on 30 March 2019).
- Dangi, A.K.; Sharman, B.; Hil, R.T.; Shukla, P. Bioremediation through microbes: Systems biology and metabolic engineering approach. Crit. Rev. Biotechnol. 2018, 39, 79–98. [Google Scholar] [CrossRef]
- Abatenh, E.; Gizaw, B.; Tsegaye, Z.; Misganaw, W. The role of microorganisms in bioremediation—A review. Environ. Biol. 2017, 2, 38–46. [Google Scholar] [CrossRef]
- Kumar, S.; Mathur, A.; Signh, V.; Nandy, S.; Khare, S.K.; Negi, S. Bioremediation of waste cooking oil using a novel lipase produced by Penicillium chrysogenum SNP5 grown in solid medium containing waste grease. Biosour. Technol. 2012, 120, 300–304. [Google Scholar] [CrossRef]
- Okino-Delgado, C.H.; Prado, D.Z.D.; Facanali, R.; Marques, M.M.O.; Nascimento, A.S.; Fernandes, C.J.D.C.; Zambuzzi, W.F.; Fleuri, L.F. Bioremediation of cooking oil waste using lipases from wastes. PLoS ONE 2017, 12, e0186246. [Google Scholar] [CrossRef]
- Massie, J.; Roberts, G.; White, P.J. Selective isolation of Bacillus sphaericus from soil by use of acetate as the only major source of carbon. Appl. Environ. Microbiol. 1985, 49, 1478–1481. [Google Scholar] [CrossRef]
- Deppe, U.; Richnow, H.; Michaelis, W.; Antranikian, G. Degradation of crude oil by an Arctic microbial consortium. Extremophiles 2005, 9, 461–470. [Google Scholar] [CrossRef]
- Jing, W.; Hongke, X.; Shaohui, G. Isolation and characteristics of a microbial consortium for effectively degrading phenanthrene. Pet. Sci. 2007, 4, 68–75. [Google Scholar] [CrossRef]
- Patowary, K.; Patowary, R.; Kalita, M.C.; Deka, S. Development of an efficient bacterial consortium for the potential remediation of hydrocarbons from contaminated sites. Front. Microbiol. 2016, 14, 1092. [Google Scholar] [CrossRef] [PubMed]
- Nzila, A.; Thukair, A.; Sankara, S.; Razzak, S.A. Characterisation of aerobic oil and grease-degrading bacteria in wastewater. Environ. Technol. 2016, 38, 1–10. [Google Scholar] [CrossRef]
- Piakong, M.T.; Zaida, N.Z. Effectiveness of single and microbial consortium of locally isolated beneficial microorganisms (LIBeM) in bioaugmentation of oil sludge contaminated soil at different concentration levels: A laboratory scale. J. Bioremed. Biodegrad. 2018, 9, 430. [Google Scholar] [CrossRef]
- Azubuike, C.C.; Chikere, C.B.; Olpokwasili, G.C. Bioremediation techniques-classification based on site of application: Principles, advantages, limitations and prospects. World Microbiol. Biotechnol. 2016, 32, 180. [Google Scholar] [CrossRef] [PubMed]
- Nagarajan, J.; Nawawi, N.M.; Ibrahim, A.L. Rhodococcus UKMP-5M, an endogenous lipase producing actinomycete from Peninsular Malaysia. Biologia 2014, 69, 123–132. [Google Scholar] [CrossRef]
- Zahri, K.N.M.; Zulkharnain, A.; Sabri, S.; Gomez-Fuentes, G.; Ahmad, S.A. Research trends of biodegradation of cooking oil in Antractica from 2001 to 2021: A bibliometric analysis based on the scopus database. Int. J. Environ. Res. Public Health 2021, 18, 2050. [Google Scholar] [CrossRef] [PubMed]
- Ibrahim, S.; Zahri, K.N.M.; Convey, P.; Khalil, K.A.; Gomez-Fuentes, C.; Zulkharnain, A.; Alias, S.A.; Gonzalez-Rocha, G.; Ahmad, S.A. Optimisation of biodegradation conditions for waste canola oil by cold-adapted Rhodococcus sp. AQ5-07 from Antarctica. Electron. J. Biotechnol. 2020, 48, 1–12. [Google Scholar] [CrossRef]
- Ibrahim, S.; Zulkharnain, A.; Zahri, K.N.M.; Lee, G.L.Y.; Convey, P.; Gomez-Fuentes, C.; Sabri, S.; Khalil, K.; Alias, S.A.; Gonzalez-Rocha, G.; et al. Effect of heavy metals and other xenobiotics on biodegradation of waste canola oil by cold-adapted Rhodococcus sp. strain AQ5-07. Rev. Mex. Ing. Quím. 2020, 19, 1041–1052. [Google Scholar] [CrossRef]
- Si-Zhong, Y.; Hui-Jun, J.; Zhi, W.; Rui-Xia, H.; Yan-Jun, J.; Xiu-Mei, L.; Shao-Peng, Y. Bioremediation of oil spills in cold environments: A review. Pedosphere 2009, 19, 371–381. [Google Scholar] [CrossRef]
- Hoover, R.B.; Pikuta, E.V. Psychrophilic and psychrotolerant microbial extremophiles in polar environments. In Polar Microbiology: The Ecology, Biodiversity and Bioremediation Potential of Microorganisms in Extremely Cold Environments; Bej, A.K., Aislabie, J., Atlas, R.M., Eds.; CRC Press: Boca Raton, FL, USA, 2019; pp. 115–156. [Google Scholar]
- Ruberto, L.; Vazquez, S.C.; MacCormack, W.P. Effectiveness of the natural bacterial flora, biostimulation and biodegradation on the bioremediation of a hydrocarbon contaminated Antarctic soil. Int. Biodeterior. Biodegrad. 2003, 52, 115–125. [Google Scholar] [CrossRef]
- Ruberto, L.A.M.; Vazquez, S.; Lobalbo, A.; MacCormack, W.P. Psychrotolerant hydrocarbon-degrading Rhodococcus strains isolated from polluted Antarctic soils. Ant. Sci. 2005, 17, 47–56. [Google Scholar] [CrossRef]
- Kumar, D.; Kim, J. New insights into bioremediation strategies for oil-contaminated soil in cold environments. Int. Biodeterior. Biodegrad. 2019, 142, 58–72. [Google Scholar] [CrossRef]
- Roslee, A.F.A.; Zakaria, N.N.; Convey, P.; Zulkharnain, A.; Lee, G.L.Y.; Gomez-Fuentes, C.; Ahmad, S.A. Statistical optimisation of growth conditions and diesel degradation by the Antarctic bacterium, Rhodococcus sp. Strain AQ5-07. Extremophiles 2019, 24, 277–291. [Google Scholar] [CrossRef]
- Abdulrasheed, M.; Zulkharnain, A.; Zakaria, N.N.; Roslee, A.F.A.; Khalil, K.A.; Napis, S.; Convey, P.; Gonzalez-Rocha, G.; Ahmad, S.A. Response surface methodology optimisation and kinetics if diesel degradation by a cold-adapted Antarctic bacterium, Arthrobacter sp. strain AQ5-05. Sustainability 2020, 12, 6966. [Google Scholar] [CrossRef]
- Malla, M.A.; Dubey, A.; Yadav, S.; Kumar, A.; Hashem, A.; Abd_Allah, E.F. Understanding and designing the strategies for the microbe-mediated remediation of environmental contaminants using omics approaches. Front. Microbiol. 2018, 9, 1132. [Google Scholar] [CrossRef]
- Shahaby, A.F.; Alharti, A.A.; El Tarras, A.E. Bioremediation of petroleum oil by potential biosurfactant-producing bacteria using gravimetric assay. Int. J. Curr. Microbiol. Appl. Sci. 2015, 4, 390–403. [Google Scholar]
- Brooksbank, A.M.; Latchford, J.W.; Mudge, S.M. Degradation and modification of fats, oils and grease by commercial microbial supplements. World J. Microbiol. Biotechnol. 2014, 23, 977–985. [Google Scholar] [CrossRef]
- Thenmozhi, R.; Nagasathya, A.; Thajuddin, N. Studies on biodegradation of used engine oil by consortium cultures. Adv. Environ. Biol. 2011, 5, 1051–1057. [Google Scholar]
- Ginesy, M.; Rusanova-Naydenova, D.; Rova, U. Tuning of the carbon-to-nitrogen ratio for the production of L-Arginine by Escherichia coli. Fermentation 2017, 3, 60. [Google Scholar] [CrossRef]
- El-Sharkawi, H.M. Effect of nitrogen sources on microbial biomass nitrogen under different soil types. Int. Sch. Res. Not. 2012, 310727. [Google Scholar] [CrossRef]
- Chaudhary, D.K.; Bajagain, R.; Jeong, S.; Kim, J. Development of a bacterial consortium comprising oil-degraders and diazotropic bacteria for elimination of exogenous nitrogen requirement in bioremediation of diesel-contaminated soil. World J. Microbiol. Biotechnol. 2019, 35, 99. [Google Scholar] [CrossRef] [PubMed]
- Gupta, K.; Jain, N.K. Near-Net Shape Manufacturing of Miniature Spur Gears by Wire Spark Erosion Machining; Springer Nature: Singapore, 2017; pp. 1–117. [Google Scholar]
- Bradley, N. The Response Surface Methodology. Master’s Thesis, Indiana University of South Bend, South Bend, IN, USA, 2007; pp. 1–10. [Google Scholar]
- El-Sheekh, M.M.; Khairy, H.M.; Ghedaa, S.F.; El-Shenody, R.A. Application of Placket-Burman design for the high production of some valuable metabolites in marine alga Nannochloropsis oculata. Egypt J. Aquat. Res. 2016, 42, 57–64. [Google Scholar] [CrossRef]
- Aydar, A.Y. Utilisation of response surface methodology in optimisation of extraction of plant materials. In Statistical Approaches with Emphasis on Design of Experiments Applied to Chemical Processes; Valter, S., Ed.; IntechOpen: London, UK, 2018. [Google Scholar] [CrossRef]
- Lim, J.; Beh, H.; Ching, D.L.C.; Ho, Y.; Baloo, L.; Bashir, M.J.K.; Wee, S. Central composite design (CCD) applied for statistical optimisation of glucose and sucrose binary carbon mixture in enhancing the denitrification process. Appl. Water Sci. 2017, 7, 3719–3727. [Google Scholar] [CrossRef]
- Rakic, T.; Kasagic-Vujanovic, I.; Jonanovic, M.; Jancic-Stojanovic, B.; Ivanovic, D. Comparison of full factorial design, central composite design and box-behnken design in chromatographic method development for the determination of fluconazole and its impurities. Anal. Lett. 2014, 47, 1334–1347. [Google Scholar] [CrossRef]
- Affandi, I.E.; Suratman, N.H.; Abdullah, S.; Ahmad, W.A.; Zakaria, Z.A. Degradation of oil and grease from high-strength industrial effluents using locally isolated aerobic biosurfactant-producing bacteria. Int. Biodeterior. Biodegrad. 2014, 95, 33–40. [Google Scholar] [CrossRef]
- Kis, A.; Laczi, K.; Zsiros, S.; Rakhely, G.; Perei, K. Biodegradation of animal fats and vegetable oils by Rhodococcus erythropolis PR4. Int. Biodeterior. Biodegrad. 2015, 105, 114–119. [Google Scholar] [CrossRef]
- Bedade, D.K.; Singhal, R.S. Isolation and characterisation and acrylamidase from Arthrobacter sp. DBV1 and its ability to biodegrade acrylamide. Appl. Biochem. Biotechnol. 2016, 182, 570–585. [Google Scholar] [CrossRef]
- Tian, X.; Wang, X.; Peng, S.; Wang, Z.; Tian, H. Isolation, screening, and crude oil degradation characteristics of hydrocarbons-degrading bacteria for treatment of oily wastewater. Water Sci. Technol. 2018, 78, 2626–2638. [Google Scholar] [CrossRef]
- Stallwood, B.; Shears, J.; Williams, P.A.; Hughes, K.A. Low temperature bioremediation of oil-contaminated soil using biostimulation and bioaugmentation with a Pseudomonas sp. from maritime Antarctica. J. Appl. Microbiol. 2005, 99, 794–802. [Google Scholar] [CrossRef]
- Aislabie, J.; McLeod, M.; Fraser, R. Potential for biodegradation of hydrocarbons in soil from the Ross Dependency, Antarctica. Appl. Microbiol. Biotechnol. 1998, 49, 210–214. [Google Scholar] [CrossRef]
- Ma, Y.; Wang, L.; Shao, Z. Pseudomonas, the dominant polycyclic aromatic hydrocarbon-degrading bacteria isolated from Antarctic soil and the role of large plasmids in horizontal gene transfer. Environ. Microbiol. 2006, 8, 455–465. [Google Scholar] [CrossRef] [PubMed]
- Goordial, J.; Davila, A.; Lacelle, D.; Pollard, W.; Marinova, M.M.; Greer, C.W.; Diruggiero, J.; Mckay, C.P.; Whyte, L.G. Nearing the cold-arid limits of microbial life in permafrost of an upper dry valley, Antarctica. ISME J. 2015, 10, 1613–1624. [Google Scholar] [CrossRef] [PubMed]
- Malavenda, R.; Rizzo, C.; Michaud, L.; Gerce, B.; Bruni, V.; Syidatk, C.; Hausmann, R.; Lo Giudice, A. Biosurfactant production by Arctic and Antarctic bacteria growing on hydrocarbons. Polar Biol. 2015, 38, 1565–1574. [Google Scholar] [CrossRef]
- Pearce, D.A.; Newsham, K.K.; Thome, M.A.S.; Calvo-Bado, L.; Krsek, M.; Laskaris, P.; Hodson, A.; Wellington, E.M. Metagenomic analysis of a southern maritime Antarctic soil. Front. Microbiol. 2012, 3, 403. [Google Scholar] [CrossRef]
- Oh, H.N.; Park, D.; Seong, H.J.; Kim, D.; Sul, W.J. Antarctic tundra soil metagenome as useful natural resource of cold-active lignocellulolytic enzymes. J. Microbiol. 2019, 57, 865–873. [Google Scholar] [CrossRef]
- Waschulin, V.; Borsetto, C.; James, R.; Newsham, K.K.; Donadio, S.; Corre, C.; Wellington, E. Metabolic potential of uncultured Antarctic soil bacteria revealed through long read metagenomic sequencing. BioRxiv 2020, 416412. [Google Scholar] [CrossRef]
- Wong, R.R.; Lim, Z.S.; Shaharuddin, N.A.; Zulkharnain, A.; Gomez-Fuentes, C.; Ahmad, S.A. Diesel in Antarctica and bibliometric study on its indigenous microorganisms as remediation agent. Int. J. Environ. Res. Public Health 2021, 18, 1512. [Google Scholar] [CrossRef] [PubMed]
- Knothe, G.; Steidly, K.R. A comparison of used cooking oils: A very heterogenous feedstock for biodiesel. Biores Technol. 2009, 100, 5796–5801. [Google Scholar] [CrossRef]
- Mannu, A.; Vlahopoulou, G.; Urgeghe, P.; Ferrom, M.; Del Caro, A.; Taras, A.; Garroni, S.; Rourke, J.P.; Cabizza, R.; Petretto, G.L. Variation of the chemical composition of waste cooking oils upon bentonite filtration. Resources 2019, 8, 108. [Google Scholar] [CrossRef]
- Zakaria, N.N.; Ahmad, S.A.; Lee, G.L.Y.; Yasid, N.A.; Manogaran, M.; Subramaniam, K.; Mazuki, T.A.T.; Nawawi, N.M.; Shukor, M.Y. Biodegradation of phenol by Antarctic bacterium Rhodococcus baikonurensis strain AQ5-001 in the presence of heavy metals. Malays. J. Biochem. Mol. Biol. 2019, 21, 29–36. [Google Scholar]
- Abdulrasheed, M.; Zakaria, N.N.; Roslee, A.F.A.; Shukor, M.Y.; Zulkharnain, A.; Napis, S.; Convey, P.; Alias, S.A.; Gonzalez-Rocha, G.; Ahmad, S.A. Biodegradation of diesel oil by cold-adapted bacterial strains of Arthrobacter spp. from Antarctica. Ant. Sci. 2020, 32, 341–353. [Google Scholar] [CrossRef]
- Food and Agriculture Organization (FAO). Chapter 7—Salty Soils. 2019. Available online: http://www.fao.org/3/r4082e/r4082e08.htm (accessed on 25 July 2020).
- Yan, N.; Marschner, P.; Cao, W.; Zuo, C.; Qin, W. Influence of salinity and water content on soil microorganism. Int. Soil Water Conserv. Res. 2015, 3, 316–323. [Google Scholar] [CrossRef]
- Wen-Wen, Z.; Chong, W.; Rui, X.; Li-Jie, W. Effects of salinity on the soil microbial community and soil fertility. J. Integr. Agric. 2019, 18, 1360–1368. [Google Scholar] [CrossRef]
- Wu, Y.; Zeng, J.; Zhu, Q.; Zhang, Z.; Lin, X. pH is the primary determination of the bacterial community structure in agriculture soils impacted by polycyclic aromatic hydrocarbon pollution. Sci. Rep. 2017, 7, 40093. [Google Scholar] [CrossRef] [PubMed]
- Todar, K.; Nutrition and Growth of Bacteria. Todar’s Online Textbook of Bacteriology. 2012. Available online: http://www.textbookofbacteriology.net (accessed on 26 March 2020).
- Ganapathy, B.; Yahya, A.; Ibrahim, N. Bioremediation of palm oil mill effluent (POME) using indigenous Meyerozyma guilliermondii. Environ. Sci. Pollut. Res. Int. 2019, 26, 11113–11125. [Google Scholar] [CrossRef] [PubMed]
- Lee, G.L.Y.; Ahmad, S.A.; Yasid, N.A.; Zulkharnain, A.; Convey, P.; Johari, W.L.W.; Alias, S.A.; Gonzalez-Rocha, G.; Shukor, M.Y. Biodegradation of phenol by cold-adapted bacteria from Antarctic soils. Polar Biol. 2018, 41, 553–562. [Google Scholar] [CrossRef]
- Subramaniam, K.; Mazuki, T.A.T.; Shukor, M.Y.; Ahmad, S.A. Isolation and optimisation of phenol degradation by Antarctic isolate using one factor at a time. Malays. J. Biochem. Mol. Biol. 2019, 21, 79–86. [Google Scholar]
- Soletto, D.; Binaghi, L.; Lodi, A.; Carvalho, J.C.M.; Converti, A. Batch and fed-batch cultivations of Spirullina platensis using ammonium sulphate and urea as nitrogen sources. Aquaculture 2005, 243, 217–224. [Google Scholar] [CrossRef]
- Chien, S.H.; Gearhart, M.M.; Villagarcia, S. Comparison of ammonium sulfate with other nitrogen and sulfur fertilisers in increasing crop production and minimizing environmental impact: A review. Soil Sci. 2011, 176, 327–335. [Google Scholar] [CrossRef]
- Legrand, M.; Ducroz, F.; Wagenbach, D.; Mulvaney, R.; Hall, J. Ammonium in coastal Antarctic aerosol and snow: Role of polar ocean and penguin emissions. J. Geophys. Res. 1998, 103, 11043–11056. [Google Scholar] [CrossRef]
- Bokhorst, S.; van Logtestijn, R.; Convey, P.; Aerts, R. Nitrogen isotope fractionation explains the 15N enrichment of Antarctic cryptogams by volatilized ammonia from penguin and seal colonies. Polar Res. 2019, 38, 3355. [Google Scholar] [CrossRef]
- Bokhorst, S.; Convey, P.; Aerts, R. Nitrogen inputs by marine vertebrates drive abundance and richness in Antarctic terrestrial ecosystems. Curr. Biol. 2019, 29, 1721–1727. [Google Scholar] [CrossRef]
- Swiecilo, A.; Zych-Wezyk, I. Bacterial stress response as an adaptation to life in a soil environment. Pol. J. Environ. Stud. 2013, 22, 1577–1587. [Google Scholar]
- Wang, J.; Wang, J.; Zhang, Z.; Li, Y.; Zhang, B.; Zhang, Z.; Zhang, G. Cold-adapted bacteria for bioremediation of crude oil-contaminated soil. J. Chem. Technol. Biotechnol. 2015, 91, 2286–2297. [Google Scholar] [CrossRef]
- Jesus, H.E.D.; Peixoto, R.S.; Rasado, A.S. Bioremediation in Antarctic soils. J. Pet. Environ. Biotechnol. 2015, 6, 1–11. [Google Scholar] [CrossRef]
- World Meteorological Organization. New Record for Antarctic Contonet Reported. 2020. Available online: https://public.wmo.int/en/media/news/new-record-antarctic-continent-reported (accessed on 12 May 2021).
- Maliji, D.; Olama, Z.; Holail, H. Environmental studies on the microbial degradation of oil hydrocarbons and its application in Lebanese oil polluted coastal and marine ecosystem. Int. J. Curr. Microbiol. Appl. Sci. 2013, 2, 1–18. [Google Scholar]
- Hamzah, A.; Azmy, A.R.R.F.H.R.; Yussoff, N.A. Isolation and characterisation of bacteria degrading Sumandak and South Angsi oils. Sains Malays. 2010, 39, 161–168. [Google Scholar]
- Huang, L.; Xie, J.; Lv, B.; Shi, X.; Li, G.; Liang, F.; Lian, J. Optimisation of nutrient component for diesel oil degradation by Acinetobacter beijerinckii ZRS. Mar. Pollutt. Bull. 2013, 76, 325–332. [Google Scholar] [CrossRef]
- Arulazhagan, P.; Vasudevan, N.; Yeom, I.T. Biodegradation of polycyclic aromatic hydrocarbon by a halotolerant bacterial consortium isolated from marine environment. Int. J. Environ. Sci. Technol. 2010, 7, 639–652. [Google Scholar] [CrossRef]
- Farag, S.; Soliman, N.A.; Abdel-Fattah, Y.R. Statistical optimisation of crude oil biodegradation by a local marine bacterium isolate Pseudomonas sp. Sp48. Genet. Eng. Biotechnol. J. 2018, 16, 409–420. [Google Scholar] [CrossRef]
- Ng, C.; Leong, X.; Masbah, N.; Adam, S.K.; Kamisah, Y.; Jaarin, K. Heated vegetable oils and cardiovascular disease risk factors. Vascul. Pharmacol. 2014, 62, 38–46. [Google Scholar] [CrossRef] [PubMed]
- Henning, R.J.; Johnson, G.T.; Coyle, J.P.; Harbison, R.D. Acrolein can cause cardiovascular disease: A review. Cardiovasc. Toxicol. 2017, 17, 227–236. [Google Scholar] [CrossRef] [PubMed]
- Aslanzadeh, S.; Ishola, M.M.; Richards, T.; Taherzadeh, M.J. An overview of existing individual unit operations. In Biorefineries Integrated Biochemical Processes for Liquid Biofuels; Qureshi, N., Hodge, D.B., Vertès, A.A., Eds.; Elsevier: Amsterdam, The Netherlands, 2014; pp. 3–36. [Google Scholar] [CrossRef]
- Das, N.; Chandran, P. Microbial degradation of petroleum hydrocarbon contaminants: An overview. Biotechol. Res. Int. 2011, 2011, 941810. [Google Scholar] [CrossRef]
- Xu, X.; Liu, W.; Tian, S.; Wang, W.; Qi, Q.; Jiang, P.; Gao, X.; Li, F.; Li, H.; Yu, H. Petroleum hydrocarbon-degrading bacteria for the remediation of oil pollution under aerobic conditions: A perspective analysis. Front. Microbiol. 2018, 9, 2885. [Google Scholar] [CrossRef] [PubMed]
- Bahera, S.K.; Meena, H.; Chakraborty, S.; Meikap, B.C. Application of response surface method (RSM) for optimization of leaching parameters for ash reduction from low-grade coal. Int. J. Min. Sci. Technol. 2018, 18, 621–629. [Google Scholar] [CrossRef]






| Factors | Name | Unit | Experimental Value | ||
|---|---|---|---|---|---|
| Low (−1) | Centre (0) | High (+1) | |||
| A | Salinity | % (w/v) | 0.00 | 0.13 | 0.25 |
| B | pH | - | 7.00 | 7.25 | 7.50 |
| C | (NH4)2SO4 concentration | g/L | 0.50 | 0.75 | 1.00 |
| D | Temperature | °C | 10.0 | 12.5 | 15.0 |
| E | Yeast extract | g/L | 0.75 | 1.00 | 1.25 |
| F | Initial oil concentration | % (v/v) | 0.50 | 1.25 | 2.00 |
| Source | Sum of Squares | DF | F Value | Prob > F |
|---|---|---|---|---|
| Model | 8404.79 | 6 | 2190.80 | 0.0005 *** |
| A | 33.73 | 1 | 79.13 | 0.0124 * |
| B | 17.16 | 1 | 40.26 | 0.0239 * |
| C | 32.69 | 1 | 76.70 | 0.0128 * |
| D | 3048.79 | 1 | 7152.30 | 0.0001 *** |
| E | 63.29 | 1 | 63.29 | 0.0067 ** |
| F | 2349.43 | 1 | 2349.43 | 0.0002 *** |
| Residual | 149.29 | 5 | ||
| Cor Total | 8405.65 | 11 | ||
| R-squared | 0.9999 | Pred R-squared | N/A | |
| Adj R-squared | 0.9994 | Adeq Precision | 128.8396 |
| Source | Sum of Squares | DF | F value | Prob > F |
|---|---|---|---|---|
| Model | 13,637.68 | 6 | 591.70 | 0.0001 *** |
| A | 10.24 | 1 | 3.56 | 0.1558 |
| B | 85.28 | 1 | 29.60 | 0.0122 * |
| C | 51.68 | 1 | 17.94 | 0.0241 * |
| D | 26.10 | 1 | 9.06 | 0.0572 |
| E | 274.27 | 1 | 95.20 | 0.0023 ** |
| F | 4402.62 | 1 | 1528.15 | <0.0001 *** |
| Residual | 298.30 | 5 | ||
| Cor. Total | 13,646.32 | 11 | ||
| R-squared | 0.9994 | Pred R-squared | 0.9827 | |
| Adj R-squared | 0.9977 | Adeq Precision | 55.992 |
| Source | Sum of Squares | DF | F value | Prob > F |
|---|---|---|---|---|
| Model | 33,527.03 | 27 | 11.32 | <0.0001 *** |
| A | 4.45 | 1 | 0.041 | 0.8412 |
| B | 24.02 | 1 | 0.22 | 0.6418 |
| C | 321.12 | 1 | 2.93 | 0.0930 |
| D | 36.49 | 1 | 0.33 | 0.5666 |
| E | 459.53 | 1 | 6.15 | 0.0153 * |
| F | 35,966.50 | 1 | 481.58 | <0.0001 *** |
| A2 | 0.066 | 1 | 6.001 × 10−4 | 0.9805 |
| B2 | 54.77 | 1 | 0.50 | 0.4829 |
| C2 | 12.37 | 1 | 0.11 | 0.7383 |
| D2 | 87.52 | 1 | 0.80 | 0.3758 |
| E2 | 579.62 | 1 | 7.76 | 0.0067 ** |
| F2 | 211.53 | 1 | 1.93 | 0.1708 |
| AB | 0.32 | 1 | 2.947 × 10−3 | 0.9569 |
| AC | 0.67 | 1 | 6.137 × 10−3 | 0.9379 |
| AD | 0.023 | 1 | 2.108 × 10−4 | 0.9885 |
| AE | 13.32 | 1 | 0.12 | 0.7289 |
| AF | 26.56 | 1 | 0.24 | 0.6247 |
| BC | 65.03 | 1 | 0.59 | 0.4448 |
| BD | 142.47 | 1 | 1.30 | 0.2596 |
| BE | 42.90 | 1 | 0.39 | 0.5344 |
| BF | 5.75 | 1 | 0.052 | 0.8197 |
| CD | 9.02 | 1 | 0.082 | 0.7754 |
| CE | 15.89 | 1 | 0.14 | 0.7050 |
| CF | 1.38 | 1 | 0.013 | 0.9111 |
| DE | 86.64 | 1 | 0.79 | 0.3782 |
| DF | 565.20 | 1 | 7.57 | 0.0074 ** |
| EF | 2.30 | 1 | 0.021 | 0.8853 |
| Residual | 4071.35 | 53 | ||
| Lack of Fit | 4071.45 | 49 | 4.94 | 0.0642 |
| Pure Error | 0.00 | 4 | 0.00 | |
| Cor Total | 43,246.92 | 80 | ||
| R-squared | 0.8688 | Pred R-squared | 0.8416 | |
| Adj R-squared | 0.8618 | Adeq Precision | 55.8624 |
| Source | Sum of Squares | DF | F value | Prob > F |
|---|---|---|---|---|
| Model | 8831.46 | 14 | 8.28 | 0.0001 *** |
| A | 555.71 | 1 | 7.16 | 0.0145 * |
| B | 0.30 | 1 | 3.927 × 10−3 | 0.9509 |
| C | 335.79 | 1 | 4.41 | 0.0531 |
| D | 434.13 | 1 | 5.59 | 0.0283 * |
| A2 | 1622.05 | 1 | 20.89 | 0.0002 *** |
| B2 | 1466.52 | 1 | 18.89 | 0.0003 *** |
| C2 | 301.62 | 1 | 3.88 | 0.0627 * |
| D2 | 3105.00 | 1 | 39.99 | <0.0001 *** |
| AB | 0.018 | 1 | 2.413 × 10−4 | 0.9878 |
| AC | 56.01 | 1 | 0.74 | 0.4047 |
| AD | 1363.48 | 1 | 17.56 | 0.0005 *** |
| BC | 603.67 | 1 | 7.77 | 0.0113 * |
| BD | 477.31 | 1 | 6.15 | 0.0222 * |
| CD | 17.89 | 1 | 0.23 | 0.6350 |
| Residual | 1142.88 | 15 | ||
| Lack of Fit | 821.09 | 10 | 1.28 | 0.4161 |
| Pure Error | 321.79 | 5 | ||
| Cor Total | 9974.34 | 29 | 8.28 | |
| R-squared | 0.8443 | Pred R-squared | 0.6001 | |
| Adj R-squared | 0.7743 | Adeq Precision | 10.4597 |
| Type of Oil | Degradation (%) | p Value | Efficiency (%) | |
|---|---|---|---|---|
| Expected Value | Actual Value | |||
| WCO | 44.84 | 44.96 | 0.985 | 99.73 |
| PCO | 46.90 | 50.39 | 0.801 | 93.07 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Zahri, K.N.M.; Zulkharnain, A.; Gomez-Fuentes, C.; Sabri, S.; Abdul Khalil, K.; Convey, P.; Ahmad, S.A. The Use of Response Surface Methodology as a Statistical Tool for the Optimisation of Waste and Pure Canola Oil Biodegradation by Antarctic Soil Bacteria. Life 2021, 11, 456. https://doi.org/10.3390/life11050456
Zahri KNM, Zulkharnain A, Gomez-Fuentes C, Sabri S, Abdul Khalil K, Convey P, Ahmad SA. The Use of Response Surface Methodology as a Statistical Tool for the Optimisation of Waste and Pure Canola Oil Biodegradation by Antarctic Soil Bacteria. Life. 2021; 11(5):456. https://doi.org/10.3390/life11050456
Chicago/Turabian StyleZahri, Khadijah Nabilah Mohd, Azham Zulkharnain, Claudio Gomez-Fuentes, Suriana Sabri, Khalilah Abdul Khalil, Peter Convey, and Siti Aqlima Ahmad. 2021. "The Use of Response Surface Methodology as a Statistical Tool for the Optimisation of Waste and Pure Canola Oil Biodegradation by Antarctic Soil Bacteria" Life 11, no. 5: 456. https://doi.org/10.3390/life11050456
APA StyleZahri, K. N. M., Zulkharnain, A., Gomez-Fuentes, C., Sabri, S., Abdul Khalil, K., Convey, P., & Ahmad, S. A. (2021). The Use of Response Surface Methodology as a Statistical Tool for the Optimisation of Waste and Pure Canola Oil Biodegradation by Antarctic Soil Bacteria. Life, 11(5), 456. https://doi.org/10.3390/life11050456










