Management of Lipedema with Ketogenic Diet: 22-Month Follow-Up
Abstract
:1. Introduction
2. Case Presentation
2.1. Data Collection and Questionnaires
2.2. Bioimpedance Analysis
2.3. Nutritional Plan
2.4. Ketosis Assessment
2.5. Side and Unwanted Effects of the KD
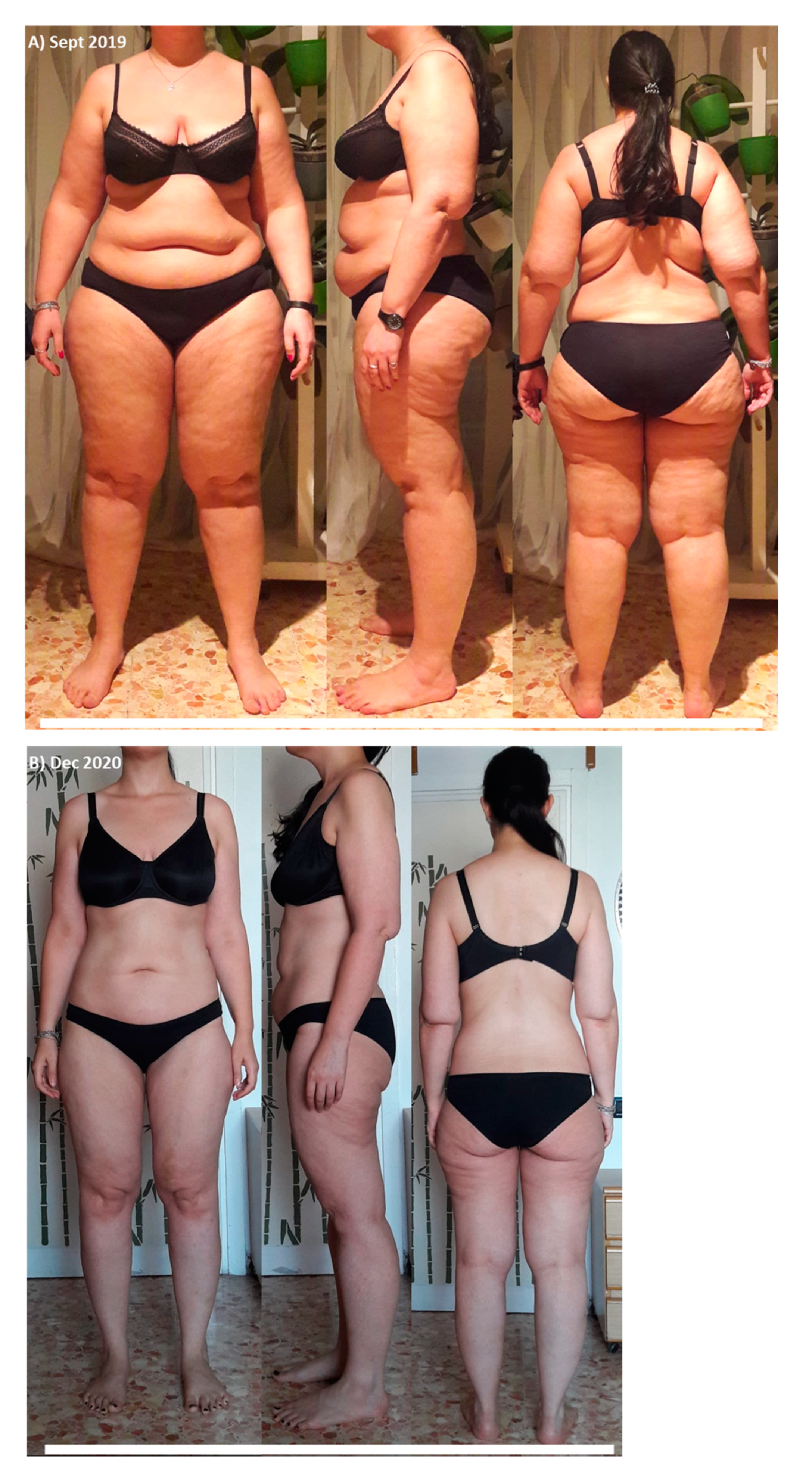
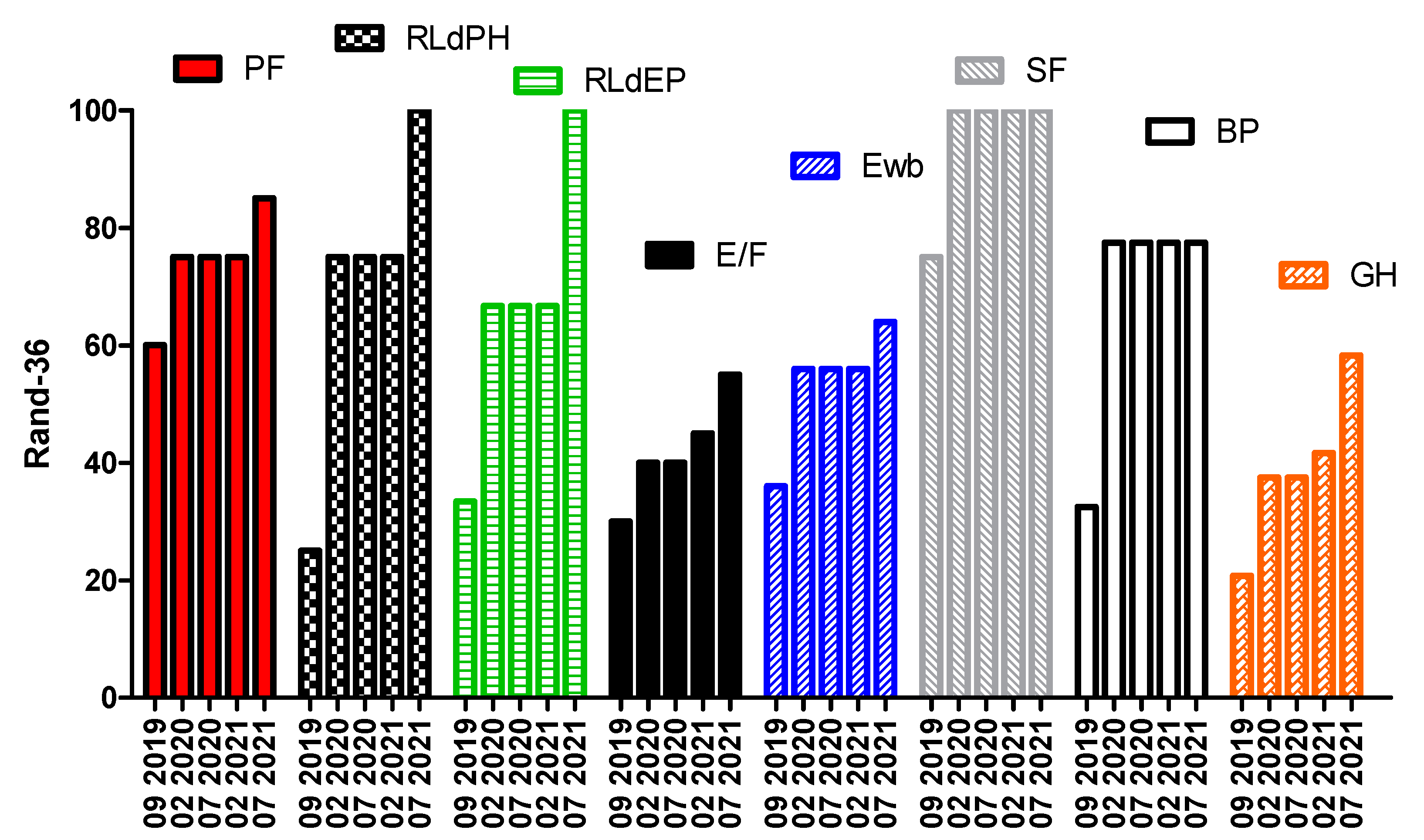
3. Results
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Acknowledgments
Conflicts of Interest
References
- Allen, E.V.; Hines, E.A.J. Lipedema of the legs: A syndrome characterised by fat legs and orthostatic edema. Proc. Staff Meet. Mayo Clin. 1940, 15, 184–187. [Google Scholar]
- Al-Ghadban, S.; Cromer, W.; Allen, M.; Ussery, C.; Badowski, M.; Harris, D.; Herbst, K.L. Dilated Blood and Lymphatic Microvessels, Angiogenesis, Increased Macrophages, and Adipocyte Hypertrophy in Lipedema Thigh Skin and Fat Tissue. J. Obes. 2019, 2019, 8747461. [Google Scholar] [CrossRef]
- Reich-Schupke, S.; Schmelle, W.; Brauer, W.J.; Cornely, M.E.; Faerber, G.; Ludwig, M.; Lulay, G.; Miller, A.; Rapprich, S.; Richter, D.F.; et al. S1 guidelines: Lipedema. J. Dtsch. Dermatol. Ges. 2017, 15, 758–767. [Google Scholar] [CrossRef]
- Herbst, K.L.; Kahn, L.A.; Iker, E.; Ehrlich, C.; Wright, T.; McHutchison, L.; Schwartz, J.; Sleigh, M.; Donahue, P.M.; Lisson, K.H.; et al. Standard of care for lipedema in the United States. Phlebology 2021, 28, 2683555211015887. [Google Scholar] [CrossRef]
- Herbst, K.L. Rare adipose disorders (RADs) masquerading as obesity. Acta Pharmacol. Sin. 2012, 33, 155–172. [Google Scholar] [CrossRef] [Green Version]
- Paolacci, S.; Precone, V.; Acquaviva, F.; Chiurazzi, P.; Fulcheri, E.; Pinelli, M.; Buffelli, F.; Michelini, S.; Herbst, K.L.; Unfer, V.; et al. Genetics of lipedema: New perspectives on genetic research and molecular diagnoses. Eur. Rev. Med. Pharmacol. Sci. 2019, 23, 5581–5594. [Google Scholar]
- Kruppa, P.; Georgiou, I.; Biermann, N.; Prantl, L.; Klein-Weigel, P.; Ghods, M. Lipedema—Pathogenesis, Diagnosis, and Treatment Options. Dtsch. Aerzteblatt Int. 2020, 117, 396–403. [Google Scholar] [CrossRef]
- Hardy, D.; Williams, A. Best practice guidelines for the management of lipoedema. Br. J. Community Nurs. 2017, 22, S44–S48. [Google Scholar] [CrossRef] [Green Version]
- Bertsch, T.; Erbacher, G.; Elwell, R.; Partsch, H. Lipoedema: A paradigm shift and consensus. J. Wound Care 2020, 29, 1–51. [Google Scholar] [CrossRef]
- Bauer, A.T.; von Lukowicz, D.; Lossagk, K.; Aitzetmueller, M.; Moog, P.; Cerny, M.; Erne, H.; Schmauss, D.; Duscher, D.; Machens, H.G. New Insights on Lipedema: The Enigmatic Disease of the Peripheral Fat. Plast. Reconstr. Surg. 2019, 144, 1475–1484. [Google Scholar] [CrossRef] [PubMed]
- Wiedner, M.; Aghajanzadeh, D.; Richter, D.F. Differential diagnoses and treatment of lipedema. Plast. Aesthetic Res. 2020, 7, 10. [Google Scholar] [CrossRef] [Green Version]
- Ghods, M.; Georgiou, I.; Schmidt, J.; Kruppa, P. Disease progression and comorbidities in lipedema patients: A 10-year retrospective analysis. Dermatol. Ther. 2020, 33, e14534. [Google Scholar] [CrossRef]
- Cannataro, R.; Caroleo, M.C.; Fazio, A.; La Torre, C.; Plastina, P.; Gallelli, L.; Lauria, G.; Cione, E. Ketogenic Diet and microRNAs Linked to Antioxidant Biochemical Homeostasis. Antioxidants 2019, 8, 269. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Dupuis, N.; Curatolo, N.; Benoist, J.-F.; Auvin, S. Ketogenic diet exhibits anti-inflammatory properties. Epilepsia 2015, 56, e95–e98. [Google Scholar] [CrossRef]
- Cannataro, R.; Perri, M.; Caroleo, M.C.; Gallelli, L.; De Sarro, G.; Cione, E. Modulation of MicroRNAs Linked to Pain-migraine by Ketogenic Diet (P14-007-19). Curr. Dev. Nutr. 2019, 3, nzz052.P14–007–19. [Google Scholar] [CrossRef] [Green Version]
- Cannataro, R.; Cione, E. Lipedema and Nutrition: What’s the link? Acta Sci. Nutr. Health 2020, 4, 86–89. [Google Scholar]
- Keith, L.; Seo, C.; Rowsemitt, C.; Pfeffer, M.; Wahi, M.; Staggs, M.; Dudek, J.; Gower, B.; Carmody, M. Ketogenic diet as a potential intervention for lipedema. Med. Hypotheses 2021, 146, 110435. [Google Scholar] [CrossRef]
- Wheless, J.W. History of the ketogenic diet. Epilepsia 2008, 49, 3–5. [Google Scholar] [CrossRef]
- Oliveira, C.L.; Mattingly, S.; Schirrmacher, R.; Sawyer, M.B.; Fine, E.J.; Prado, C.M. A Nutritional Perspective of Ketogenic Diet in Cancer: A Narrative Review. J. Acad. Nutr. Diet. 2018, 118, 668–688. [Google Scholar] [CrossRef]
- Dowis, K.; Banga, S. The Potential Health Benefits of the Ketogenic Diet: A Narrative Review. Nutrients 2021, 13, 1654. [Google Scholar] [CrossRef]
- Barbanti, P.; Fofi, L.; Aurilia, C.; Egeo, G.; Caprio, M. Ketogenic diet in migraine: Rationale, findings and perspectives. Neurol. Sci. 2017, 38, 111–115. [Google Scholar] [CrossRef]
- Di Lorenzo, C.; Ballerini, G.; Barbanti, P.; Bernardini, A.; D’Arrigo, G.; Egeo, G.; Frediani, F.; Garbo, R.; Pierangeli, G.; Prudenzano, M.P.; et al. Applications of Ketogenic Diets in Patients with Headache: Clinical Recommendations. Nutrients 2021, 13, 2307. [Google Scholar] [CrossRef]
- Paoli, A.; Mancin, L.; Giacona, M.C.; Bianco, A.; Caprio, M. Effects of a ketogenic diet in overweight women with polycystic ovary syndrome. J. Transl. Med. 2020, 18, 104. [Google Scholar] [CrossRef]
- Paoli, A. Ketogenic Diet for Obesity: Friend or Foe? Int. J. Environ. Res. Public Health 2014, 11, 2092–2107. [Google Scholar] [CrossRef] [Green Version]
- Cannataro, R.; Perri, M.; Gallelli, L.; Caroleo, M.C.; De Sarro, G.; Cione, E. Ketogenic Diet Acts on Body Remodeling and MicroRNAs Expression Profile. MicroRNA 2019, 8, 116–126. [Google Scholar] [CrossRef]
- Møller, N. Ketone Body, 3-Hydroxybutyrate: Minor Metabolite—Major Medical Manifestations. J. Clin. Endocrinol. Metab. 2020, 105, dgaa370. [Google Scholar] [CrossRef]
- Ruan, H.-B.; Crawford, P. Ketone bodies as epigenetic modifiers. Curr. Opin. Clin. Nutr. Metab. Care 2018, 21, 260–266. [Google Scholar] [CrossRef]
- Boison, D. New insights into the mechanisms of the ketogenic diet. Curr. Opin. Neurol. 2017, 30, 187–192. [Google Scholar] [CrossRef] [Green Version]
- Di Renzo, L.; Cinelli, G.; Romano, L.; Zomparelli, S.; De Santis, G.L.; Nocerino, P.; Bigioni, G.; Arsini, L.; Cenname, G.; Pujia, A.; et al. Potential Effects of a Modified Mediterranean Diet on Body Composition in Lipoedema. Nutrients 2021, 13, 358. [Google Scholar] [CrossRef]
- Cannataro, R.; Fazio, A.; La Torre, C.; Caroleo, M.; Cione, E. Polyphenols in the Mediterranean Diet: From Dietary Sources to microRNA Modulation. Antioxidants 2021, 10, 328. [Google Scholar] [CrossRef]
- American College of Rheumatology. Western Ontario and McMaster Universities Osteoarthritis Index (WOMAC)—General Description. ACR. Retrieved 6 June 2012. Available online: https://www.rheumatology.org/I-Am-A/Rheumatologist/Research/Clinician-Researchers (accessed on 9 December 2021).
- Yi, H.; Shin, K.; Shin, C. Development of the Sleep Quality Scale. J. Sleep Res. 2006, 15, 309–316. [Google Scholar] [CrossRef]
- Hays, R.D.; Sherbourne, C.D.; Mazel, R.M. The rand 36-item health survey 1.0. Health Econ. 1993, 2, 217–227. [Google Scholar] [CrossRef]
- Bijur, P.E.; Silver, W.; Gallagher, E.J. Reliability of the Visual Analog Scale for Measurement of Acute Pain. Acad. Emerg. Med. 2001, 8, 1153–1157. [Google Scholar] [CrossRef]
- Lukaski, H.C.; Bolonchuk, W.W.; Hall, C.B.; Siders, W.A. Validation of tetrapolar bioelectrical impedance method to assess human body composition. J. Appl. Physiol. 1985, 60, 1327–1332. [Google Scholar] [CrossRef] [Green Version]
- Kyle, U.G.; Bosaeus, I.; De Lorenzo, A.D.; Deurenberg, P.; Elia, M.; Gomez, J.M.; Heitmann, B.L.; Kent-Smith, L.; Melchior, J.-C.; Pirlich, M.; et al. Bioelectrical impedance analysis—Part I: Review of principles and methods. Clin. Nutr. 2004, 23, 1226–1243. [Google Scholar] [CrossRef]
- Priglinger, E.; Strohmeier, K.; Weigl, M.; Lindner, C.; Auer, D.; Gimona, M.; Barsch, M.; Jacak, J.; Redl, H.; Grillari, J.; et al. SVF-derived extracellular vesicles carry characteristic miRNAs in lipedema. Sci. Rep. 2020, 10, 7211. [Google Scholar] [CrossRef]
- Kabisch, S.; Wenschuh, S.; Buccellato, P.; Spranger, J.; Pfeiffer, A.F. Affordability of Different Isocaloric Healthy Diets in Germany—An Assessment of Food Prices for Seven Distinct Food Patterns. Nutrients 2021, 13, 3037. [Google Scholar] [CrossRef]
- Faerber, G. Ernährungstherapie bei Lipödem und Adipositas—Ergebnisse Eines Leitliniengerechten Therapiekonzepts. Vasomed 2017, 29, 122–123. [Google Scholar]

| Nutritional Plan | |
|---|---|
| Breakfast | 30 g of rye bread |
| 40 g of ham | |
| 20 g of spreadable cheese | |
| 1 espresso coffee without sugar or other sweetener | |
| Snacks (during the day) | 50 g of parmesan cheese |
| 20 g of nuts (walnuts, hazelnuts, cashews, almond) | |
| 40 g of chicken sliced breast | |
| Vegetables at will (from a list that considered a limited intake of carbohydrates) | |
| Lunch | 200 g of salmon |
| A large bowl of mixed salad | |
| 2 tablespoons of extra virgin of olive oil | |
| Vinegar and spices at will | |
| Dinner | 1 whole egg and 100 mL of pasteurized egg white |
| A large bowl of grilled vegetable (zucchini, eggplant) | |
| 2 tablespoons of extra virgin of olive oil | |
| Vinegar and spices at will | |
| Drink at least 2 L of water, with carbonated sweetened beverages permitted | |
| Biochemical Parameters | Baseline | 7th Month | 14th Month | 21st Month | Normal Range |
|---|---|---|---|---|---|
| Glycemia mg/dL | 99 | 95 | 92 | 90 | 70–100 |
| Insulin mIU/L | 29.3 | 14 | 10 | 11 | 4–25 |
| HOMA-IR | 7.16 | 3.28 | 2.27 | 2.44 | <2.60 |
| HbA1C mmol/mol | 32 | 26 | 25 | 26 | 20–38 |
| Vitamin D ng/mL | 15 | 30 | 32 | 32 | 30–50 |
| Hb g/dL | 13.9 | 13.2 | 13.7 | 13.5 | 12.0–15.5 |
| Serum iron µg/dL | 102 | 89 | 98 | 95 | 60–170 |
| Serum calcium mmol/L | 2.4 | 2.2 | 2.29 | 2.3 | 2.2–2.7 |
| Serum potassium mmol/L | 3.74 | 3.8 | 4.39 | 4.01 | 3.6–5.2 |
| AST U/L | 21 | 19 | 19 | 9 | 8–33 |
| ALT U/L | 21 | 16 | 13 | 12 | 4–36 |
| GGT U/L | 16 | 12 | 13 | 12 | 0–30 |
| Creatinine mg/dL | 0.93 | 0.87 | 0.85 | 0.72 | 0.6–1.2 |
| CRP mg/dL | 0.6 | 0.2 | 0.1 | 0.1 | 0.3–1.0 |
| Uric acid mg/dL | 2.5 | 3.5 | 3 | 3 | 2.7–7.3 |
| Circumference | 09 2019 | 10 2019 | 11 2019 | 12 2019 | 02 2020 | 04 2020 | 06 2020 | 08 2020 | 10 2020 | 12 2020 | D% |
|---|---|---|---|---|---|---|---|---|---|---|---|
| Arm sx | 43 | 42 | 40.5 | 39.5 | 39 | 38.5 | 36 | 36 | 35 | 32.5 | −24.42 |
| Arm dx | 44 | 42 | 40.5 | 40 | 40 | 38 | 37 | 36.5 | 35 | 32.5 | −26.14 |
| Forearm sx | 32 | 31 | 30 | 29 | 28 | 28 | 27 | 27 | 27 | 25.5 | −20.31 |
| Forearm dx | 32 | 30 | 30 | 29 | 29 | 29 | 26 | 27 | 27 | 25.5 | −20.31 |
| Waist | 109 | 99.5 | 96.5 | 96 | 96 | 94 | 90 | 88.5 | 86 | 83 | −23.85 |
| Hip | 134 | 123.5 | 116 | 105.5 | 103.5 | 102 | 104 | 102 | 101 | 96.5 | −27.98 |
| Coulotte | 133 | 132 | 127.5 | 124 | 120 | 118.5 | 115.5 | 115 | 111 | 107 | −19.55 |
| Thigh sx | 74 | 71.5 | 69.5 | 66 | 62 | 59.5 | 56.5 | 56.5 | 56 | 54 | −27.03 |
| Thigh dx | 74 | 71.5 | 69.5 | 67 | 64.5 | 61 | 58 | 57.5 | 56.5 | 54 | −27.03 |
| Knee sx | 47.5 | 47 | 46 | 44.5 | 43 | 41 | 40 | 42 | 42 | 39 | −17.9 |
| Knee dx | 48 | 47 | 46.5 | 44.5 | 44 | 42 | 42 | 42 | 42 | 39.5 | −17.71 |
| Calf sx | 50.5 | 48 | 47 | 46.5 | 46 | 45.5 | 44 | 44 | 43 | 41.5 | −17.82 |
| Calf dx | 50 | 48.5 | 48 | 46 | 46 | 44.5 | 44 | 44 | 43 | 41.5 | −17 |
| Ankle sx | 26 | 25 | 25 | 24,5 | 24 | 24 | 23.5 | 24 | 23.5 | 23.5 | −9.62 |
| Ankle dx | 26.5 | 25 | 24.5 | 24.5 | 24 | 24 | 23.5 | 24 | 23.5 | 23.5 | −11.32 |
| Time | WOMAC | SQS | VAS |
|---|---|---|---|
| 09 2019 | 45 | 37 | 9.2 |
| 02 2020 | 38 | 26 | 7.1 |
| 07 2020 | 37 | 24 | 6.7 |
| 02 2021 | 35 | 22 | 3.5 |
| 07 2021 | 21 | 19 | 3 |
| D% | −53.33 | −48.65 | −67.39 |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cannataro, R.; Michelini, S.; Ricolfi, L.; Caroleo, M.C.; Gallelli, L.; De Sarro, G.; Onorato, A.; Cione, E. Management of Lipedema with Ketogenic Diet: 22-Month Follow-Up. Life 2021, 11, 1402. https://doi.org/10.3390/life11121402
Cannataro R, Michelini S, Ricolfi L, Caroleo MC, Gallelli L, De Sarro G, Onorato A, Cione E. Management of Lipedema with Ketogenic Diet: 22-Month Follow-Up. Life. 2021; 11(12):1402. https://doi.org/10.3390/life11121402
Chicago/Turabian StyleCannataro, Roberto, Sandro Michelini, Lorenzo Ricolfi, Maria Cristina Caroleo, Luca Gallelli, Giovambattista De Sarro, Alberto Onorato, and Erika Cione. 2021. "Management of Lipedema with Ketogenic Diet: 22-Month Follow-Up" Life 11, no. 12: 1402. https://doi.org/10.3390/life11121402
APA StyleCannataro, R., Michelini, S., Ricolfi, L., Caroleo, M. C., Gallelli, L., De Sarro, G., Onorato, A., & Cione, E. (2021). Management of Lipedema with Ketogenic Diet: 22-Month Follow-Up. Life, 11(12), 1402. https://doi.org/10.3390/life11121402









