Short-Term Effect of Induced Alterations in Testosterone Levels on Fasting Plasma Amino Acid Levels in Healthy Young Men
Abstract
:1. Introduction
2. Materials and Methods
2.1. Subjects and Study Outline
2.2. Amino Acid Analysis
2.3. Statistical Analysis
2.3.1. Monitoring Free Amino Acids across Time Points with Testosterone Fluctuations
2.3.2. Modeling the Best Amino Acids in Response to Testosterone Fluctuations
3. Results
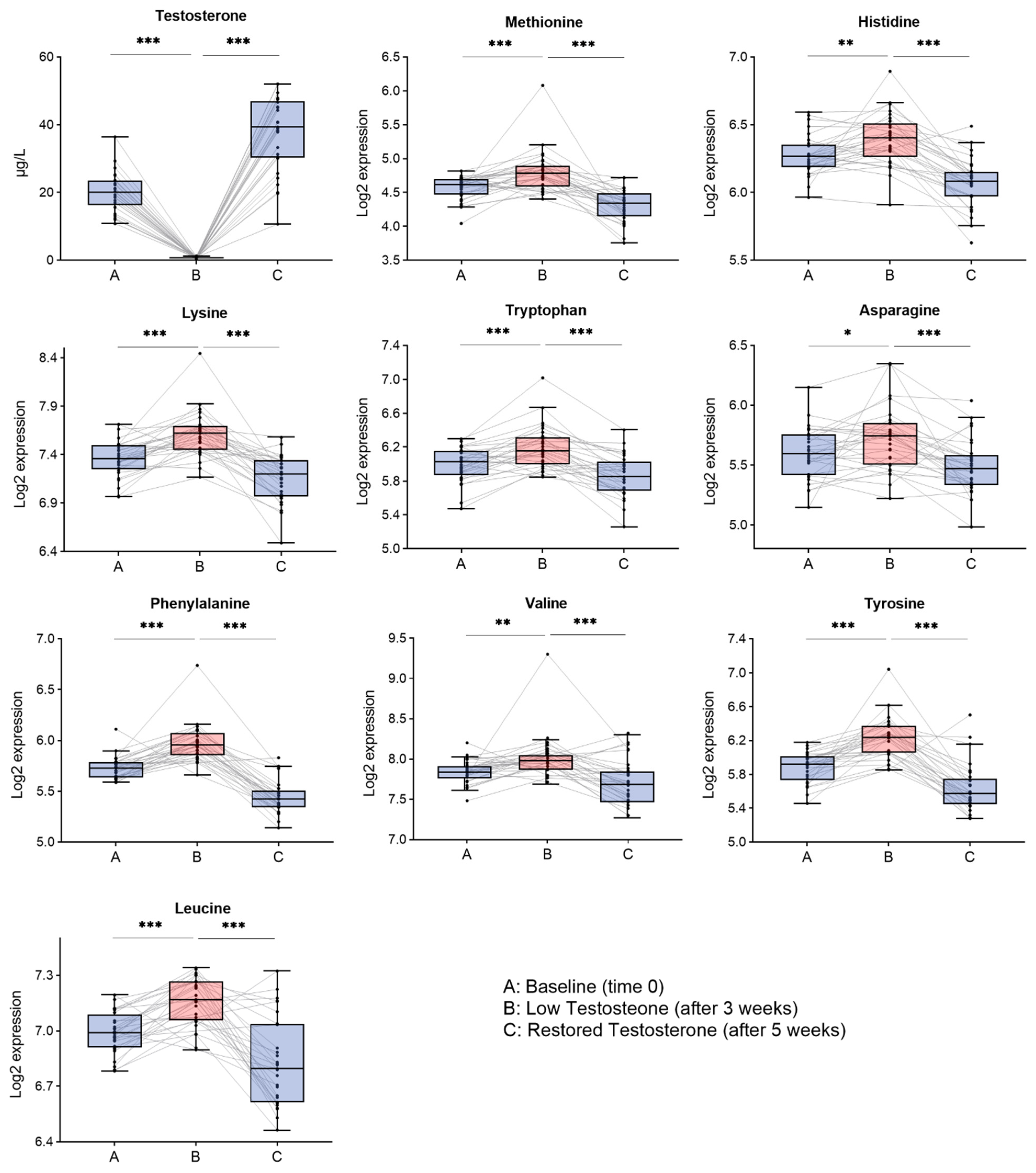
3.1. Amino Acids Significantly Changed by Testosterone
3.2. Branched Chain Amino Acids and Aromatic Amino Acids Significantly Changed by Testosterone
3.3. Selected Amino Acids That Best Reflect Changes in Testosterone
4. Discussion
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
References
- Rossetti, M.L.; Steiner, J.L.; Gordon, B.S. Androgen-mediated regulation of skeletal muscle protein balance. Mol. Cell. Endocrinol. 2017, 447, 35–44. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Kraemer, W.J.; Ratamess, N.A.; Nindl, B.C. Recovery responses of testosterone, growth hormone, and IGF-1 after resistance exercise. J. Appl. Physiol. 2017, 122, 549–558. [Google Scholar] [CrossRef] [PubMed]
- Protein and Amino Acids—Recommended Dietary Allowances—NCBI Bookshelf. Available online: https://www.ncbi.nlm.nih.gov/books/NBK234922/ (accessed on 24 September 2021).
- Rui, L. Energy metabolism in the liver. Compr. Physiol. 2014, 4, 177–197. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nie, C.; He, T.; Zhang, W.; Zhang, G.; Ma, X. Branched Chain Amino Acids: Beyond Nutrition Metabolism. Int. J. Mol. Sci. 2018, 19, 954. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Lynch, C.J.; Adams, S.H. Branched-chain amino acids in metabolic signalling and insulin resistance. Nat. Rev. Endocrinol. 2014, 10, 723–736. [Google Scholar] [CrossRef] [Green Version]
- Neinast, M.; Murashige, D.; Arany, Z. Branched Chain Amino Acids. Annu. Rev. Physiol. 2019, 81, 139–164. [Google Scholar] [CrossRef]
- Henriksson, J. Effect of exercise on amino acid concentrations in skeletal muscle and plasma. J. Exp. Biol. 1991, 160, 149–165. [Google Scholar] [CrossRef]
- Fanelli, G.; Gevi, F.; Belardo, A.; Zolla, L. Metabolic patterns in insulin-sensitive male hypogonadism. Cell Death Dis. 2018, 9, 653. [Google Scholar] [CrossRef] [Green Version]
- Kelly, D.M.; Jones, T.H. Testosterone: A metabolic hormone in health and disease. J. Endocrinol. 2013, 217, R25–R45. [Google Scholar] [CrossRef] [Green Version]
- Sahlin, K.B.; Pla, I.; Sanchez, A.; Pawłowski, K.; Leijonhufvud, I.; Appelqvist, R.; Marko-Varga, G.; Giwercman, A.; Malm, J. Short-term effect of pharmacologically induced alterations in testosterone levels on common blood biomarkers in a controlled healthy human model. Scand. J. Clin. Lab. Investig. 2019, 80, 25–31. [Google Scholar] [CrossRef]
- Malm, J.; Végvári, Á.; Rezeli, M.; Upton, P.; Danmyr, P.; Nilsson, R.; Steinfelder, E.; Marko-Varga, G. Large scale biobanking of blood—The importance of high density sample processing procedures. J. Proteom. 2012, 76, 116–124. [Google Scholar] [CrossRef] [PubMed]
- Greenfield, M.L.; Kuhn, J.E.; Wojtys, E.M. A statistics primer. Confidence intervals. Am. J. Sports Med. 1998, 26, 145–149. [Google Scholar] [CrossRef]
- Matthews, D.E. An Overview of Phenylalanine and Tyrosine Kinetics in Humans. J. Nutr. 2007, 137, 1549S. [Google Scholar] [CrossRef]
- Armstrong, M.D.; Stave, U. A study of plasma free amino acid levels. III. Variations during growth and aging. Metabolism 1973, 22, 571–578. [Google Scholar] [CrossRef]
- Pla, I.; Sahlin, K.B.; Pawłowski, K.; Appelqvist, R.; Marko-Varga, G.; Sanchez, A.; Malm, J. A pilot proteomic study reveals different protein profiles related to testosterone and gonadotropin changes in a short-term controlled healthy human cohort. J. Proteom. 2020, 220, 103768. [Google Scholar] [CrossRef]
- Sarabhai, T.; Roden, M. Hungry for your alanine: When liver depends on muscle proteolysis. J. Clin. Investig. 2019, 129, 4563–4566. [Google Scholar] [CrossRef]
- Ferrando, A.A.; Tipton, K.D.; Doyle, D.; Phillips, S.M.; Cortiella, J.; Wolfe, R.R. Testosterone injection stimulates net protein synthesis but not tissue amino acid transport. Am. J. Physiol. 1998, 275, E864–E871. [Google Scholar] [CrossRef] [PubMed]
- Wang, T.J.; Larson, M.G.; Vasan, R.S.; Cheng, S.; Rhee, E.P.; McCabe, E.; Lewis, G.D.; Fox, C.S.; Jacques, P.F.; Fernandez, C.; et al. Metabolite profiles and the risk of developing diabetes. Nat. Med. 2011, 17, 448–453. [Google Scholar] [CrossRef]
- Zhou, Y.; Qiu, L.; Xiao, Q.; Wang, Y.; Meng, X.; Xu, R.; Wang, S.; Na, R. Obesity and diabetes related plasma amino acid alterations. Clin. Biochem. 2013, 46, 1447–1452. [Google Scholar] [CrossRef]
- Forslund, A.H.; Hambræus, L.; van Beurden, H.; Holmbäck, U.; El-Khoury, A.E.; Hjorth, G.; Olsson, R.; Stridsberg, M.; Wide, L.; Åkerfeldt, T.; et al. Inverse relationship between protein intake and plasma free amino acids in healthy men at physical exercise. Am. J. Physiol. Endocrinol. Metab. 2000, 278, E857–E867. [Google Scholar] [CrossRef] [Green Version]
- Eriksson, L.S.; Broberg, S.; Björkman, O.; Wahren, J. Ammonia metabolism during exercise in man. Clin. Physiol. 1985, 5, 325–336. [Google Scholar] [CrossRef]
- Bergström, J.; Fürst, P.; Hultman, E. Free amino acids in muscle tissue and plasma during exercise in man. Clin. Physiol. 1985, 5, 155–160. [Google Scholar] [CrossRef] [PubMed]
- Sahlin, K.; Katz, A.; Broberg, S. Tricarboxylic acid cycle intermediates in human muscle during prolonged exercise. Am. J. Physiol. 1990, 259, C834–C841. [Google Scholar] [CrossRef] [PubMed]
- Pitkanen, H.; Mero, A.N.; Oja, S.S.; Komi, P.V.; Rusko, H.E.; Nummela, A.; Saransaari, P.I.; Takala, T. Effects of training on the exercise-induced changes in serum amino acids and hormones. J. Strength Cond. Res. 2002, 16, 390–398. [Google Scholar] [PubMed]
- Svanberg, E.; Moller-Loswick, A.C.; Matthews, D.E.; Korner, U.; Andersson, M.; Lundholm, K. Effects of amino acids on synthesis and degradation of skeletal muscle proteins in humans. Am. J. Physiol. 1996, 271, E718–E724. [Google Scholar] [CrossRef] [PubMed]
- Peake, J.M.; Tan, S.J.; Markworth, J.F.; Broadbent, J.A.; Skinner, T.L.; Cameron-Smith, D. Metabolic and hormonal responses to isoenergetic high-intensity interval exercise and continuous moderate-intensity exercise. Am. J. Physiol. Endocrinol. Metab. 2014, 307, E539–E552. [Google Scholar] [CrossRef] [PubMed] [Green Version]
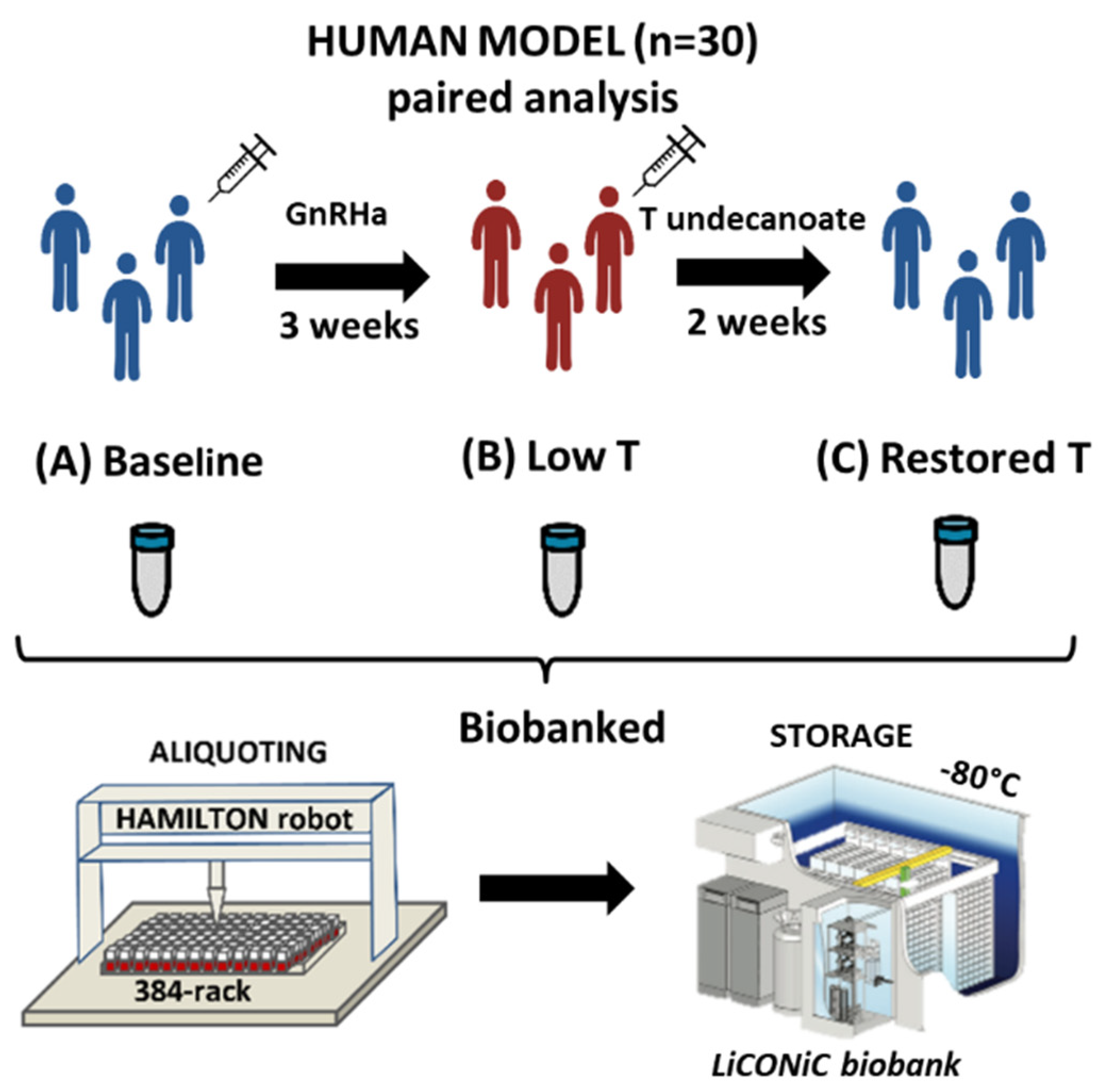
| Parameter | Baseline (A) | Low T (B) | Restored T (C) |
|---|---|---|---|
| T (nmol/L) | 19.97 ± 5.7 | 0.71 ± 0.3 | 37.39 ± 11.1 |
| FSH (IU/L) | 3.09 ± 1.7 | 0.15 ± 0.1 | 0.14 ± 0.1 |
| LH (IU/L | 4.99 ± 1.6 | 0.14 ± 0.1 | 0.14 ± 0.1 |
| AA | q-Value (FDR < 0.05) | CI (B-A) | p-Value (B-A) | CI (C-B) | p-Value (C-B) | CI (C-A) | p-Value (C-A) |
|---|---|---|---|---|---|---|---|
| Asn | <0.001 a | 0.117 | <0.05 | −0.229 | <0.0001 | −0.112 | <0.05 |
| (0.011; 0.222) | (−0.34; −0.119) | (−0.198; −0.027) | |||||
| Val e | <0.001 b | 0.159 | <0.01 | −0.323 | <0.001 | −0.146 | <0.05 |
| (0.073; 0.23) | (−0.47; −0.187) | (−0.278; −0.015) | |||||
| Met e | <0.0001 b | 0.215 | <0.001 | −0.448 | <0.001 | −0.253 | <0.01 |
| (0.123; 0.314) | (−0.589; −0.321) | (−0.379; −0.082) | |||||
| Leu e | <0.0001 b | 0.191 | <0.001 | −0.376 | <0.0001 | −0.191 | <0.01 |
| (0.136; 0.24) | (−0.508; −0.264) | (−0.286; −0.086) | |||||
| Tyr | <0.0001 b | 0.286 | <0.0001 | −0.615 | <0.0001 | −0.281 | <0.001 |
| (0.211; 0.413) | (−0.737; −0.492) | (−0.393; −0.18) | |||||
| Phe e | <0.0001 b | 0.249 | <0.0001 | −0.547 | <0.0001 | −0.291 | <0.0001 |
| (0.196; 0.303) | (−0.623; −0.473) | (−0.363; −0.223) | |||||
| Lys e | <0.0001 b | 0.245 | <0.0001 | −0.431 | <0.0001 | −0.214 | <0.01 |
| (0.171; 0.319) | (−0.567; −0.335) | (−0.31; −0.097) | |||||
| His e | <0.0001 a | 0.117 | <0.01 | −0.324 | <0.0001 | −0.207 | <0.0001 |
| (0.038; 0.195) | (−0.405; −0.243) | (−0.277; −0.138) | |||||
| Trp e | <0.0001 b | 0.205 | <0.001 | −0.338 | <0.0001 | −0.129 | <0.05 |
| (0.117; 0.302) | (−0.423; −0.226) | (−0.238; −0.017) | |||||
| T | <0.0001 a | −19.262 | <0.0001 | 36.683 | <0.0001 | 17.421 | <0.0001 |
| (−21.45; −17.075) | (32.451; 40.914) | (13.004; 21.837) |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Sahlin, K.B.; Pla, I.; de Siqueira Guedes, J.; Pawłowski, K.; Appelqvist, R.; Marko-Varga, G.; Domont, G.B.; César Sousa Nogueira, F.; Giwercman, A.; Sanchez, A.; et al. Short-Term Effect of Induced Alterations in Testosterone Levels on Fasting Plasma Amino Acid Levels in Healthy Young Men. Life 2021, 11, 1276. https://doi.org/10.3390/life11111276
Sahlin KB, Pla I, de Siqueira Guedes J, Pawłowski K, Appelqvist R, Marko-Varga G, Domont GB, César Sousa Nogueira F, Giwercman A, Sanchez A, et al. Short-Term Effect of Induced Alterations in Testosterone Levels on Fasting Plasma Amino Acid Levels in Healthy Young Men. Life. 2021; 11(11):1276. https://doi.org/10.3390/life11111276
Chicago/Turabian StyleSahlin, K. Barbara, Indira Pla, Jéssica de Siqueira Guedes, Krzysztof Pawłowski, Roger Appelqvist, György Marko-Varga, Gilberto Barbosa Domont, Fábio César Sousa Nogueira, Aleksander Giwercman, Aniel Sanchez, and et al. 2021. "Short-Term Effect of Induced Alterations in Testosterone Levels on Fasting Plasma Amino Acid Levels in Healthy Young Men" Life 11, no. 11: 1276. https://doi.org/10.3390/life11111276
APA StyleSahlin, K. B., Pla, I., de Siqueira Guedes, J., Pawłowski, K., Appelqvist, R., Marko-Varga, G., Domont, G. B., César Sousa Nogueira, F., Giwercman, A., Sanchez, A., & Malm, J. (2021). Short-Term Effect of Induced Alterations in Testosterone Levels on Fasting Plasma Amino Acid Levels in Healthy Young Men. Life, 11(11), 1276. https://doi.org/10.3390/life11111276







