Tomentosin a Sesquiterpene Lactone Induces Antiproliferative and Proapoptotic Effects in Human Burkitt Lymphoma by Deregulation of Anti- and Pro-Apoptotic Genes
Abstract
:1. Introduction
2. Materials and Methods
2.1. Cell Culture
2.2. Cytotoxicity Assay
2.3. Apoptotic Assay
2.4. Cell Cycle Analysis
2.5. Gene Expression Analysis
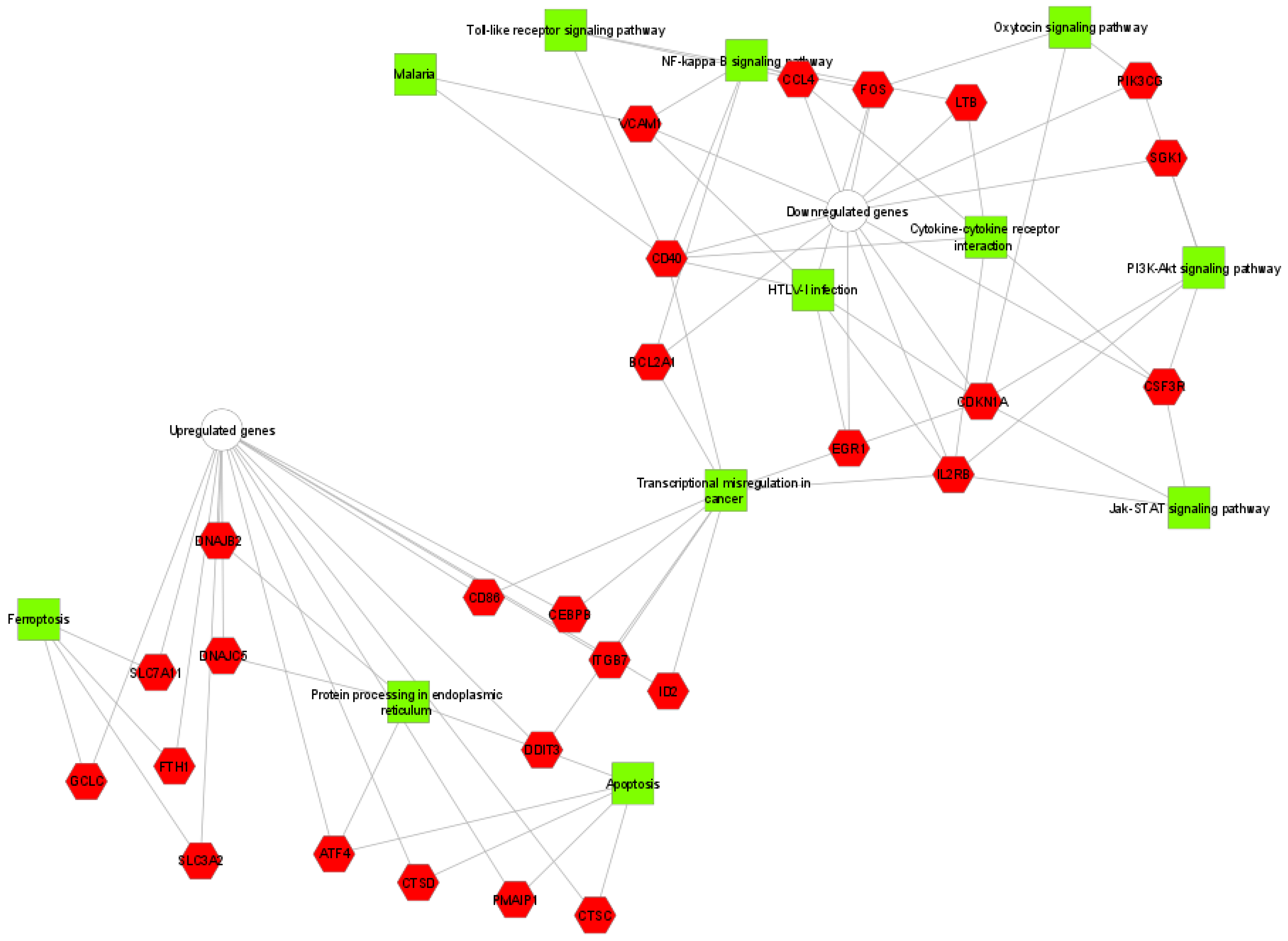
2.6. Functional Classification and Pathway Analysis of DEGs
2.7. Statistical Data Analysis
3. Results
3.1. Effects of Tomentosin on Cell Viability in Human Burkitt Lymphoma Cells
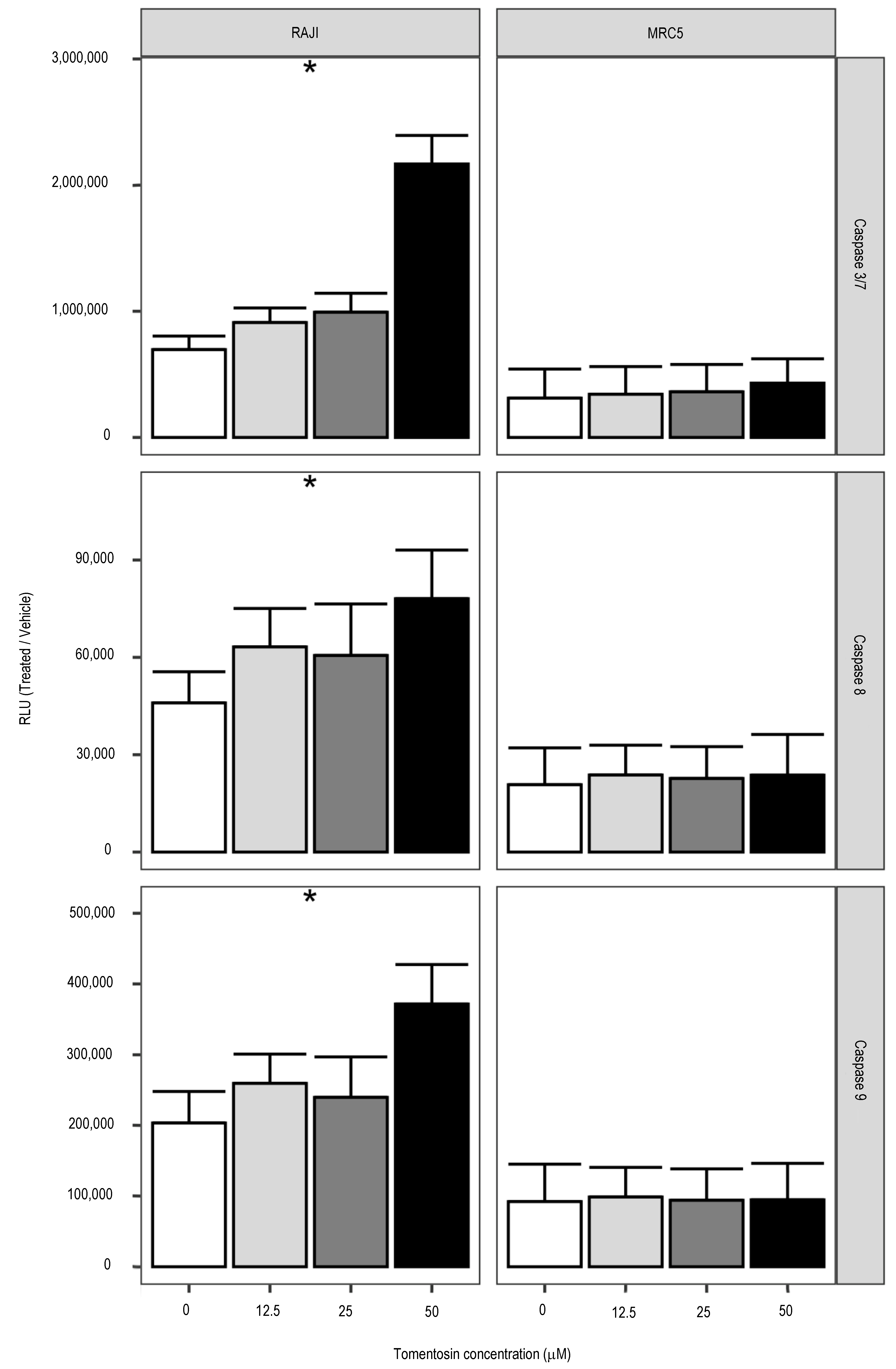
3.2. Effects of Tomentosin on Apoptotic Cellular Processes in Human Burkitt Lymphoma Cells
3.3. Effects of Tomentosin on Cell Cycle in Human Burkitt Lymphoma Cells
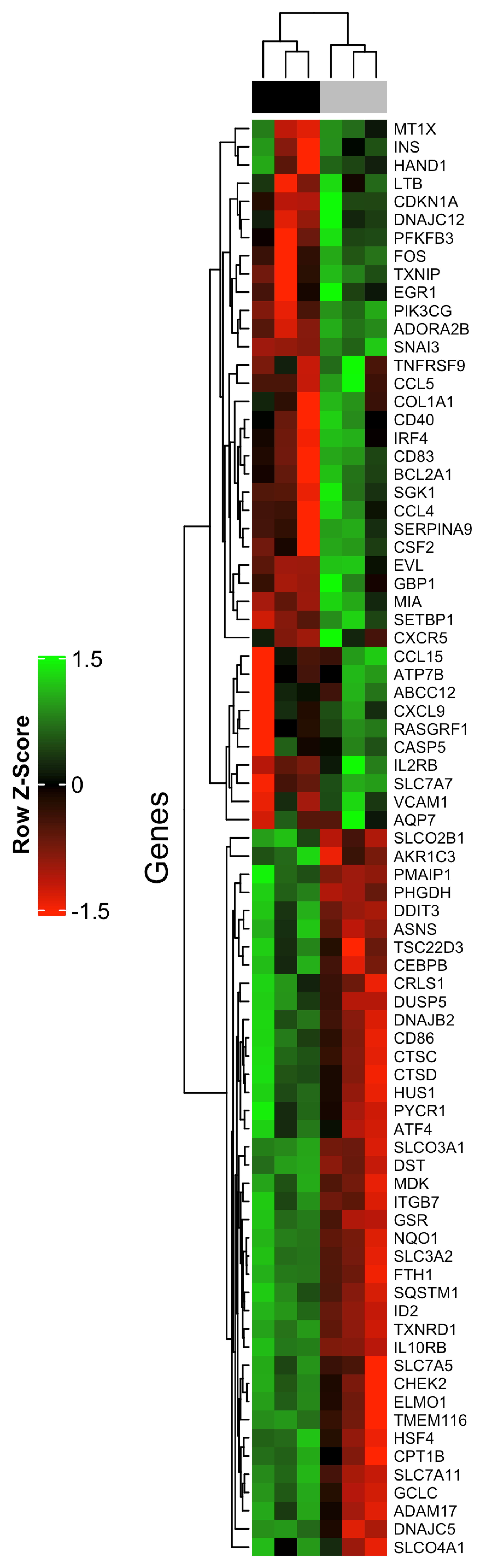
3.4. Effects of Tomentosin on Gene Expression Profiling in Human Burkitt Lymphoma Cells
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Conflicts of Interest
Abbreviations
References
- Casulo, C.; Friedberg, J.W. Burkitt lymphoma- a rare but challenging lymphoma. Best Pract. Res. Clin. Haematol. 2018, 31, 279–284. [Google Scholar] [CrossRef] [PubMed]
- Dang, C.V.; O’Donnell, K.A.; Juopperi, T. The great MYC escape in tumorigenesis. Cancer Cell 2005, 8, 177–178. [Google Scholar] [CrossRef] [Green Version]
- Dunleavy, K.; Little, R.F.; Wilson, W.H. Update on Burkitt Lymphoma. Hematol. Oncol. Clin. N. Am. 2016, 30, 1333–1343. [Google Scholar] [CrossRef]
- Bishop, P.C.; Rao, V.K.; Wilson, W.H. Burkitt’s lymphoma: Molecular pathogenesis and treatment. Cancer Investig. 2000, 18, 574–583. [Google Scholar] [CrossRef] [PubMed]
- Jaffe, E.S. The 2008 WHO classification of lymphomas: Implications for clinical practice and translational research. Hematol. Am. Soc. Hematol. Educ. Program 2009, 2009, 523–531. [Google Scholar] [CrossRef] [Green Version]
- Saleh, K.; Michot, J.M.; Camara-Clayette, V.; Vassetsky, Y.; Ribrag, V. Burkitt and Burkitt-Like Lymphomas: A Systematic Review. Curr. Oncol. Rep. 2020, 22, 1–9. [Google Scholar] [CrossRef] [PubMed]
- Minard-Colin, V.; Aupérin, A.; Pillon, M.; Burke, G.A.A.; Barkauskas, D.A.; Wheatley, K.; Delgado, R.F.; Alexander, S.; Uyttebroeck, A.; Bollard, C.M.; et al. Rituximab for High-Risk, Mature B-Cell Non-Hodgkin’s Lymphoma in Children. N. Engl. J. Med. 2020, 382, 2207. [Google Scholar] [CrossRef]
- Molyneux, E.M.; Rochford, R.; Griffin, B.; Newton, R.; Jackson, G.; Menon, G.; Harrison, C.J.; Israels, T.; Bailey, S. Burkitt’s lymphoma. Lancet 2012, 379, 1234–1244. [Google Scholar] [CrossRef] [Green Version]
- Virdis, P.; Migheli, R.; Galleri, G.; Fancello, S.; Cadoni, M.P.L.; Pintore, G.; Petretto, G.L.; Marchesi, I.; Fiorentino, F.P.; di Francesco, A.; et al. Antiproliferative and proapoptotic effects of Inula viscosa extract on Burkitt lymphoma cell line. Tumor Biol. 2020, 42. [Google Scholar] [CrossRef] [Green Version]
- Rozenblat, S.; Grossman, S.; Bergman, M.; Gottlieb, H.; Cohen, Y.; Dovrat, S. Induction of G2/M arrest and apoptosis by sesquiterpene lactones in human melanoma cell lines. Biochem. Pharmacol. 2008, 75, 369–382. [Google Scholar] [CrossRef] [PubMed]
- Merghoub, N.; El Btaouri, H.; Benbacer, L.; Gmouh, S.; Trentesaux, C.; Brassart, B.; Attaleb, M.; Madoulet, C.; Wenner, T.; Amzazi, S.; et al. Tomentosin Induces Telomere Shortening and Caspase-Dependant Apoptosis in Cervical Cancer Cells. J. Cell. Biochem. 2017, 118, 1689–1698. [Google Scholar] [CrossRef]
- Lee, C.M.; Lee, J.; Nam, M.J.; Choi, Y.S.; Park, S.H. Tomentosin displays anti-carcinogenic effect in human osteosarcoma MG-63 cells via the induction of intracellular reactive oxygen species. Int. J. Mol. Sci. 2019, 20, 1508. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yang, H.; Zhao, H.; Dong, X.; Yang, Z.; Chang, W. Tomentosin induces apoptotic pathway by blocking inflammatory mediators via modulation of cell proteins in AGS gastric cancer cell line. J. Biochem. Mol. Toxicol. 2020, 34, e22501. [Google Scholar] [CrossRef] [PubMed]
- Xiang, P.; Guo, X.; Han, Y.-Y.; Gao, J.-M.; Tang, J.-J. Cytotoxic and Pro-apoptotic Activities of Sesquiterpene Lactones from Inula Britannica. Nat. Prod. Commun. 2016, 11, 7–10. [Google Scholar] [CrossRef] [Green Version]
- Yang, L.; Xie, J.; Almoallim, H.S.; Alharbi, S.A.; Chen, Y. Tomentosin inhibits cell proliferation and induces apoptosis in MOLT-4 leukemia cancer cells through the inhibition of mTOR/PI3K/Akt signaling pathway. J. Biochem. Mol. Toxicol. 2021, 35. [Google Scholar] [CrossRef]
- Ihaka, R.; Gentleman, R. R: A Language for Data Analysis and Graphics. J. Comput. Graph. Stat. 1996, 5, 299. [Google Scholar] [CrossRef]
- Ellis, B.; Gentleman, R.; Hahne, F.; Le Meur, N.; Sarkar, D.; Jiang, M. flowViz: Visualization for Flow Cytometry. R Package Version 1.56.0. 2021. Available online: https://bioconductor.org/packages/release/bioc/html/flowViz.html (accessed on 23 October 2021).
- Love, M.I.; Huber, W.; Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 2014, 15, 1–21. [Google Scholar] [CrossRef] [Green Version]
- Wang, G.W.; Qin, J.J.; Cheng, X.R.; Shen, Y.H.; Shan, L.; Jin, H.Z.; Zhang, W.D. Inula sesquiterpenoids: Struc-tural diversity, cytotoxicity and anti-tumor activity. Expert Opin. Investig. Drugs 2014, 23, 317–345. [Google Scholar] [CrossRef] [PubMed]
- Quintana, J.; Estévez, F. Recent Advances on Cytotoxic Sesquiterpene Lactones. Curr. Pharm. Des. 2019, 24, 4355–4361. [Google Scholar] [CrossRef]
- Yee, J.; White, R.E.; Anderton, E.; Allday, M.J. Latent Epstein-Barr virus can inhibit apoptosis in B cells by blocking the induction of NOXA expression. PLoS ONE 2011, 6, e28506. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Crawley, J.B.; Williams, L.M.; Mander, T.; Brennan, F.M.; Foxwell, B.M.J. Interleukin-10 stimulation of phosphatidylinositol 3-kinase and p70 S6 kinase is required for the proliferative but not the antiinflammatory effects of the cytokine. J. Biol. Chem. 1996, 271, 16357–16362. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Zhou, J.-H.; Broussard, S.R.; Strle, K.; Freund, G.G.; Johnson, R.W.; Dantzer, R.; Kelley, K.W. IL-10 Inhibits Apoptosis of Promyeloid Cells by Activating Insulin Receptor Substrate-2 and Phosphatidylinositol 3′-Kinase. J. Immunol. 2001, 167, 4436–4442. [Google Scholar] [CrossRef] [PubMed]
- Tsao, C.C.; Geisen, C.; Abraham, R.T. Interaction between human MCM7 and Rad17 proteins is required for replication checkpoint signaling. EMBO J. 2004, 23, 4660–4669. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagy, B.; Lundán, T.; Larramendy, M.L.; Aalto, Y.; Zhu, Y.; Niini, T.; Edgren, H.; Ferrer, A.; Vilpo, J.; Elonen, E.; et al. Abnormal expression of apoptosis-related genes in haematological malignancies: Overexpression of MYC is poor prognostic sign in mantle cell lymphoma. Br. J. Haematol. 2003, 120, 434–441. [Google Scholar] [CrossRef] [Green Version]
- Feuerhake, F.; Kutok, J.L.; Monti, S.; Chen, W.; LaCasce, A.S.; Cattoretti, G.; Kurtin, P.; Pinkus, G.S.; De Levai, L.; Harris, N.L.; et al. NFκB activity, function, and target-gene signatures in primary mediastinal large B-cell lymphoma and diffuse large B-cell lymphoma subtypes. Blood 2005, 106, 1392–1399. [Google Scholar] [CrossRef]
- Mahadevan, D.; Spier, C.; Della Croce, K.; Miller, S.; George, B.; Riley, C.; Warner, S.; Grogan, T.M.; Miller, T.P. Transcript profiling in peripheral T-cell lymphoma, not otherwise specified, and diffuse large B-cell lymphoma identifies distinct tumor profile signatures. Mol. Cancer Ther. 2005, 4, 1867–1879. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Piva, R.; Pellegrino, E.; Mattioli, M.; Agnelli, L.; Lombardi, L.; Boccalatte, F.; Costa, G.; Ruggeri, B.A.; Cheng, M.; Chiarle, R.; et al. Functional validation of the anaplastic lymphoma kinase signature identifies CEBPB and BCl2A1 as critical target genes. J. Clin. Investig. 2006, 116, 3171–3182. [Google Scholar] [CrossRef]
- Monti, S.; Savage, K.J.; Kutok, J.L.; Feuerhake, F.; Kurtin, P.; Mihm, M.; Wu, B.; Pasqualucci, L.; Neuberg, D.; Aguiar, R.C.T.; et al. Molecular profiling of diffuse large B-cell lymphoma identifies robust subtypes including one characterized by host inflammatory response. Blood 2005, 105, 1851–1861. [Google Scholar] [CrossRef] [Green Version]
- Park, I.; Lee, S.; Whang, D.; Hong, W.; Choi, S.; Shin, H.; Choe, T.; Hong, S. Expression of a novel Bcl-2 related gene, Bfl-1, in various human cancers and cancer cell lines. Anticancer. Res. 1997, 17, 4619–4622. [Google Scholar]
- Kathpalia, V.P.; Mussak, E.N.; Chow, S.S.; Lam, P.H.; Skelley, N.; Time, M.; Markelewicz, R.J.; Kanduc, D.; Lomas, L.; Xiang, Z.; et al. Genome-wide transcriptional profiling in human squamous cell carcinoma of the skin identifies unique tumor-associated signatures. J. Dermatol. 2006, 33, 309–318. [Google Scholar] [CrossRef]
- Saleh, A.; Zain, R.B.; Hussaini, H.; Ng, F.; Tanavde, V.; Hamid, S.; Chow, A.T.; Lim, G.S.; Abraham, M.T.; Teo, S.H.; et al. Transcriptional profiling of oral squamous cell carcinoma using formalin-fixed paraffin-embedded samples. Oral Oncol. 2010, 46, 379–386. [Google Scholar] [CrossRef] [Green Version]
- Yoon, H.S.; Hong, S.H.; Kang, H.J.; Ko, B.K.; Ahn, S.H.; Huh, J.R. Bfl-1 Gene Expression in Breast Cancer: Its Relationship with other Prognostic Factors. J. Korean Med. Sci. 2003, 18, 225–230. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Riker, A.I.; Enkemann, S.A.; Fodstad, O.; Liu, S.; Ren, S.; Morris, C.; Xi, Y.; Howell, P.; Metge, B.; Samant, R.S.; et al. The gene expression profiles of primary and metastatic melanoma yields a transition point of tumor progression and metastasis. BMC Med. Genomics 2008, 1, 1–16. [Google Scholar] [CrossRef] [Green Version]
- Lee, C.F.; Ling, Z.Q.; Zhao, T.; Fang, S.H.; Chang, W.C.; Lee, S.C.; Lee, K.R. Genomic-wide analysis of lymphatic metastasis-associated genes in human hepatocellular carcinoma. World J. Gastroenterol. 2009, 15, 356–365. [Google Scholar] [CrossRef] [PubMed]
- Dotto, G.P. p21(WAF1/Cip1): More than a break to the cell cycle? Biochim. Biophys. Acta Rev. Cancer 2000, 1471, M43–M56. [Google Scholar] [CrossRef]
- Abbas, T.; Dutta, A. p21 in cancer: Intricate networks and multiple activities. Nat. Rev. Cancer 2009, 9, 400–414. [Google Scholar] [CrossRef] [PubMed]
- De La Cueva, E.; García-Cao, I.; Herranz, M.; López, P.; García-Palencia, P.; Flores, J.M.; Serrano, M.; Fernández-Piqueras, J.; Martín-Caballero, J. Tumorigenic activity of p21Waf1/Cip1 in thymic lymphoma. Oncogene 2006, 25, 4128–4132. [Google Scholar] [CrossRef] [Green Version]
- Martín-Caballero, J.; Flores, J.M.; García-Palencia, P.; Serrano, M. Tumor Susceptibility of p21 Waf1/Cip1-deficient Mice 1. Cancer Res. 2001, 61, 6234–6238. [Google Scholar]
- Roninson, I.B. Oncogenic functions of tumour suppressor p21Waf1/Cip1/Sdi1: Association with cell senescence and tumour-promoting activities of stromal fibroblasts. Cancer Lett. 2002, 179, 1–14. [Google Scholar] [CrossRef]


| Cell Line | Treatment | IC50 | Confidence Interval 95% |
|---|---|---|---|
| Raji | Tomentosin | 42.62 µM | 39.53–46.47 µM |
| MRC5 | Tomentosin | >50.00 µM | — |
| Raji | Cispaltin | 9.43 µM | 8.23–11.21 µM |
| MRC5 | Cispaltin | 14.77 µM | 13.1–16.48 µM |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Virdis, P.; Marchesi, I.; Fiorentino, F.P.; Migheli, R.; Sanna, L.; Bordoni, V.; Pintore, G.; Galleri, G.; Muroni, M.R.; Bagella, L.; et al. Tomentosin a Sesquiterpene Lactone Induces Antiproliferative and Proapoptotic Effects in Human Burkitt Lymphoma by Deregulation of Anti- and Pro-Apoptotic Genes. Life 2021, 11, 1128. https://doi.org/10.3390/life11111128
Virdis P, Marchesi I, Fiorentino FP, Migheli R, Sanna L, Bordoni V, Pintore G, Galleri G, Muroni MR, Bagella L, et al. Tomentosin a Sesquiterpene Lactone Induces Antiproliferative and Proapoptotic Effects in Human Burkitt Lymphoma by Deregulation of Anti- and Pro-Apoptotic Genes. Life. 2021; 11(11):1128. https://doi.org/10.3390/life11111128
Chicago/Turabian StyleVirdis, Patrizia, Irene Marchesi, Francesco Paolo Fiorentino, Rossana Migheli, Luca Sanna, Valentina Bordoni, Giorgio Pintore, Grazia Galleri, Maria Rosaria Muroni, Luigi Bagella, and et al. 2021. "Tomentosin a Sesquiterpene Lactone Induces Antiproliferative and Proapoptotic Effects in Human Burkitt Lymphoma by Deregulation of Anti- and Pro-Apoptotic Genes" Life 11, no. 11: 1128. https://doi.org/10.3390/life11111128






