Cryptic Diversity of Isaria-like Species in Guizhou, China
Abstract
:1. Introduction
2. Materials and Methods
2.1. Fungal Materials and Identification
2.2. DNA Extraction, Polymerase Chain Reaction Amplification and Nucleotide Sequencing
2.3. Sequence Alignment and Phylogenetic Analyses
3. Results
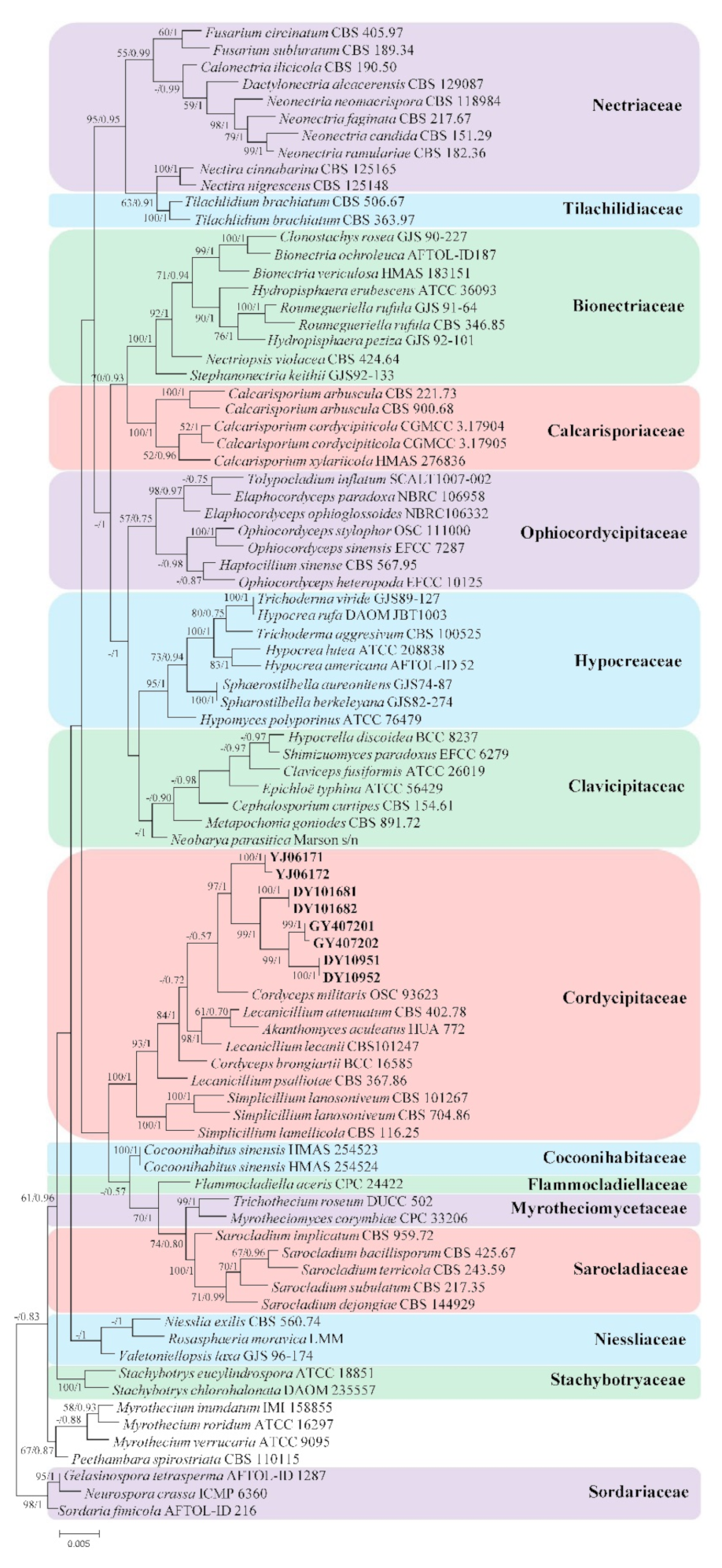
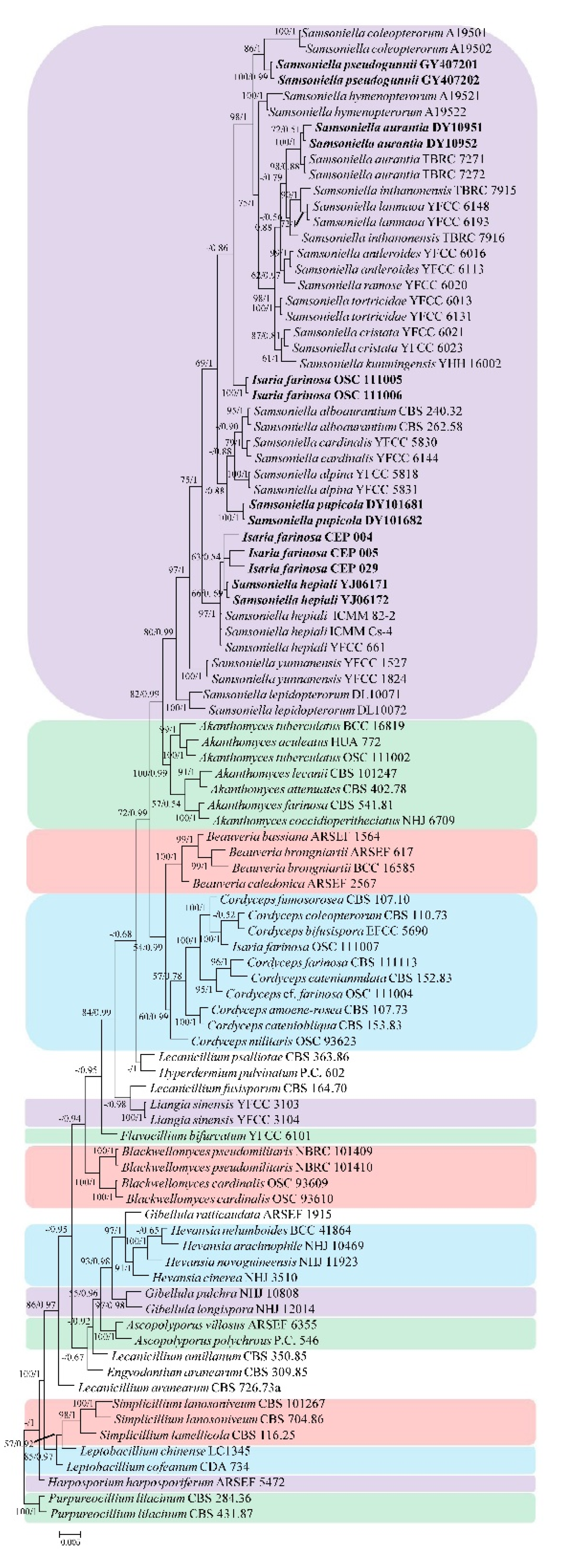
3.1. Phylogenetic Analyses
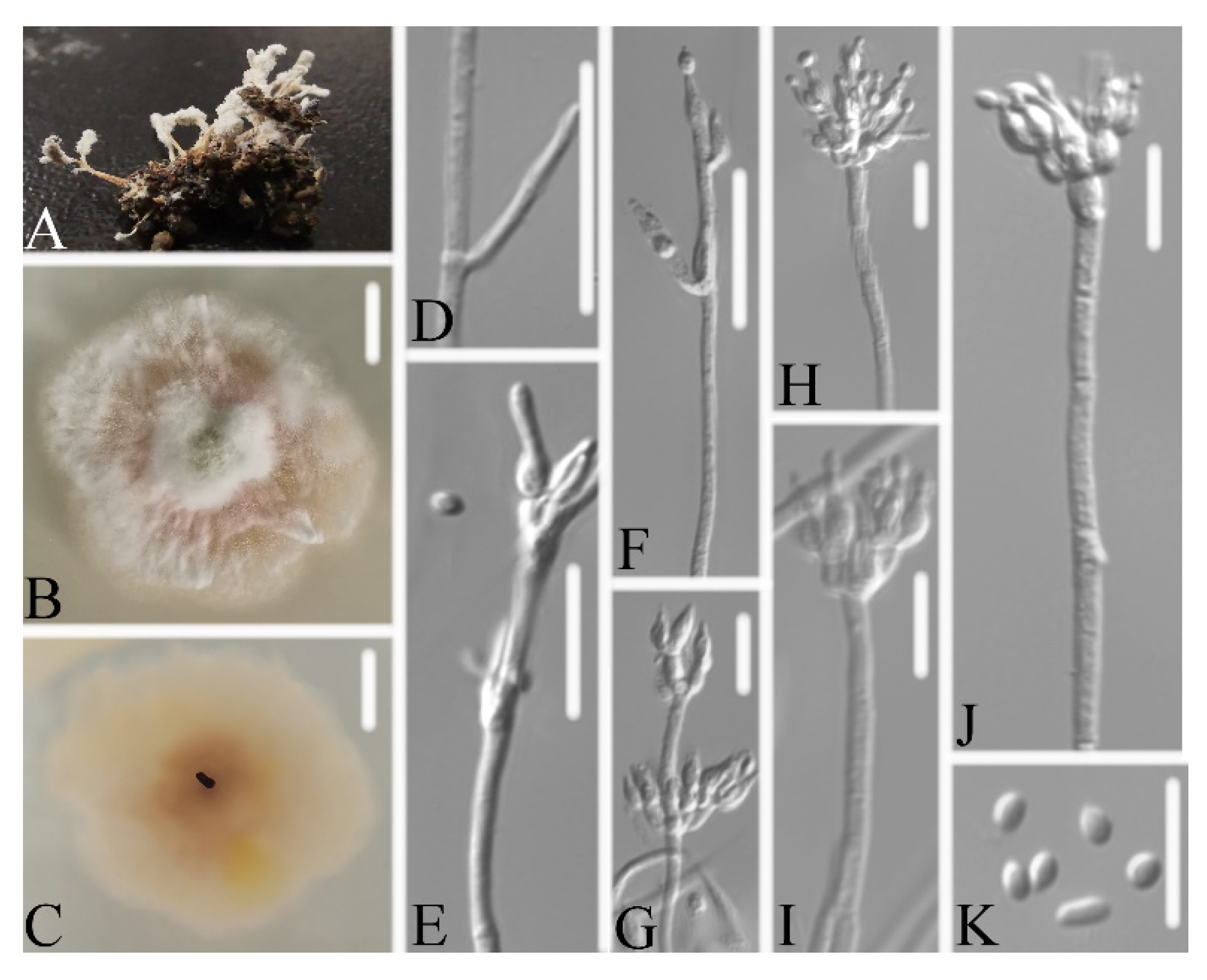
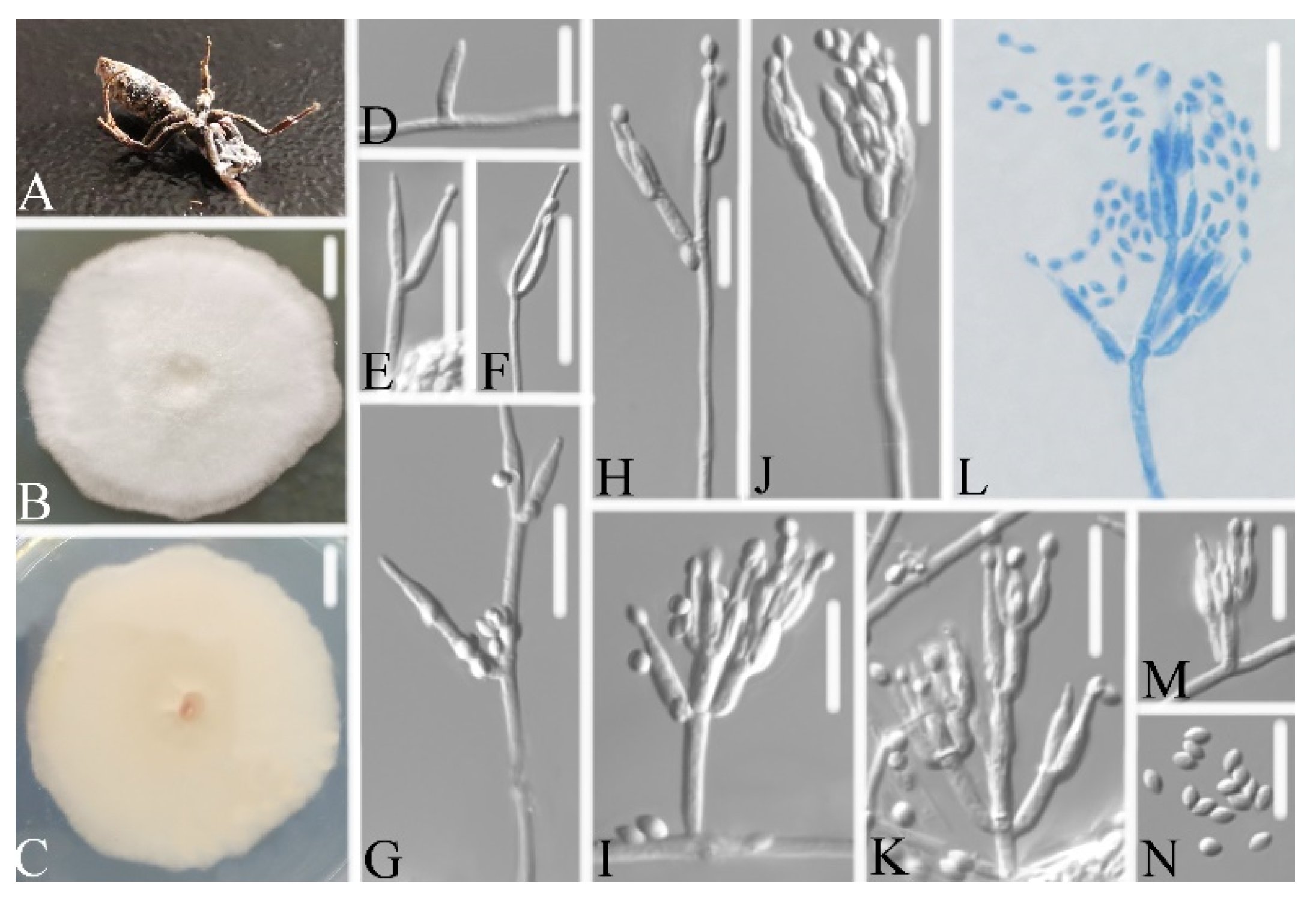
3.2. Taxonomy
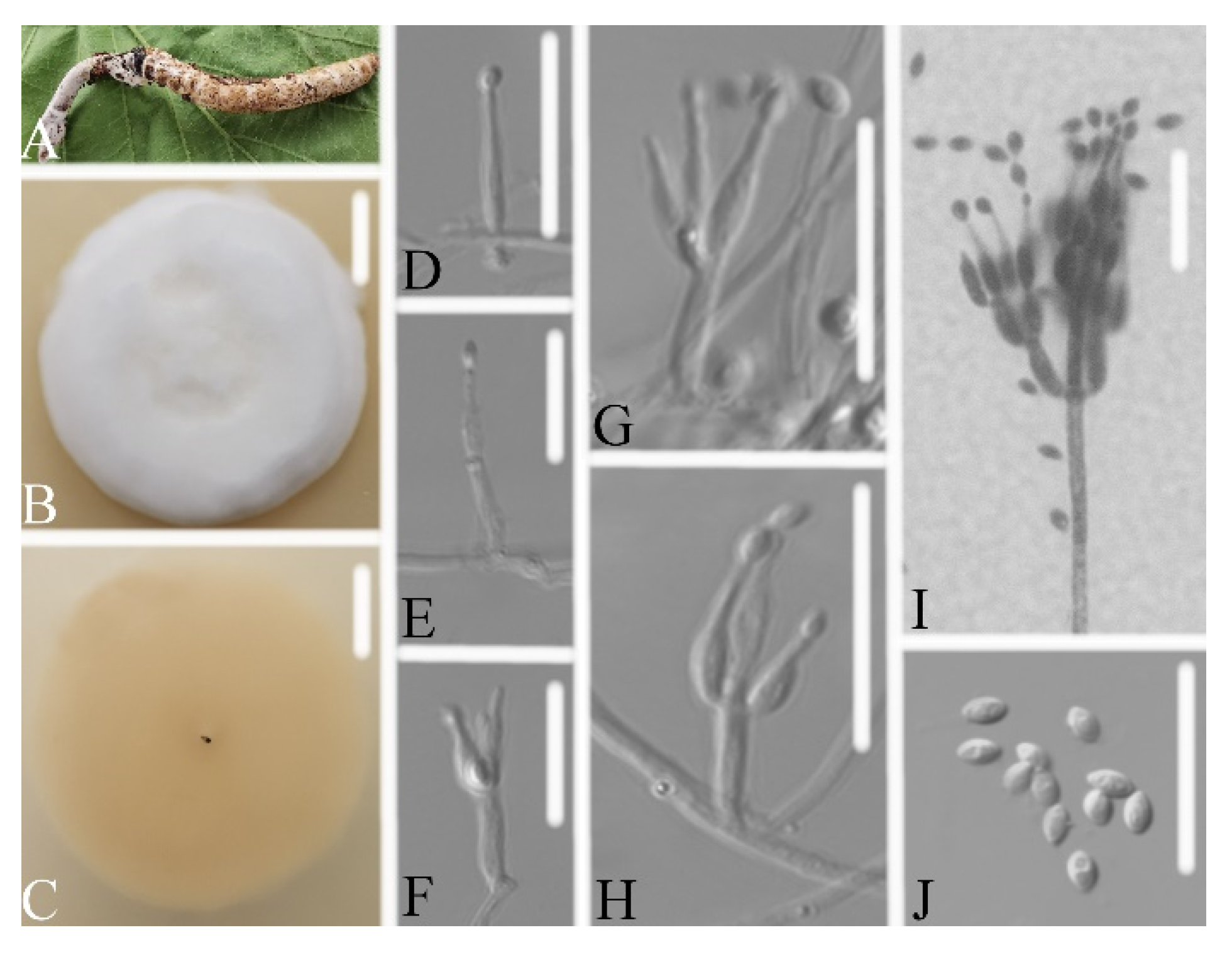
3.2.1. Samsoniella pseudogunnii W.H. Chen, Y.F. Han, J.D. Liang, and Z.Q. Liang, sp. nov.
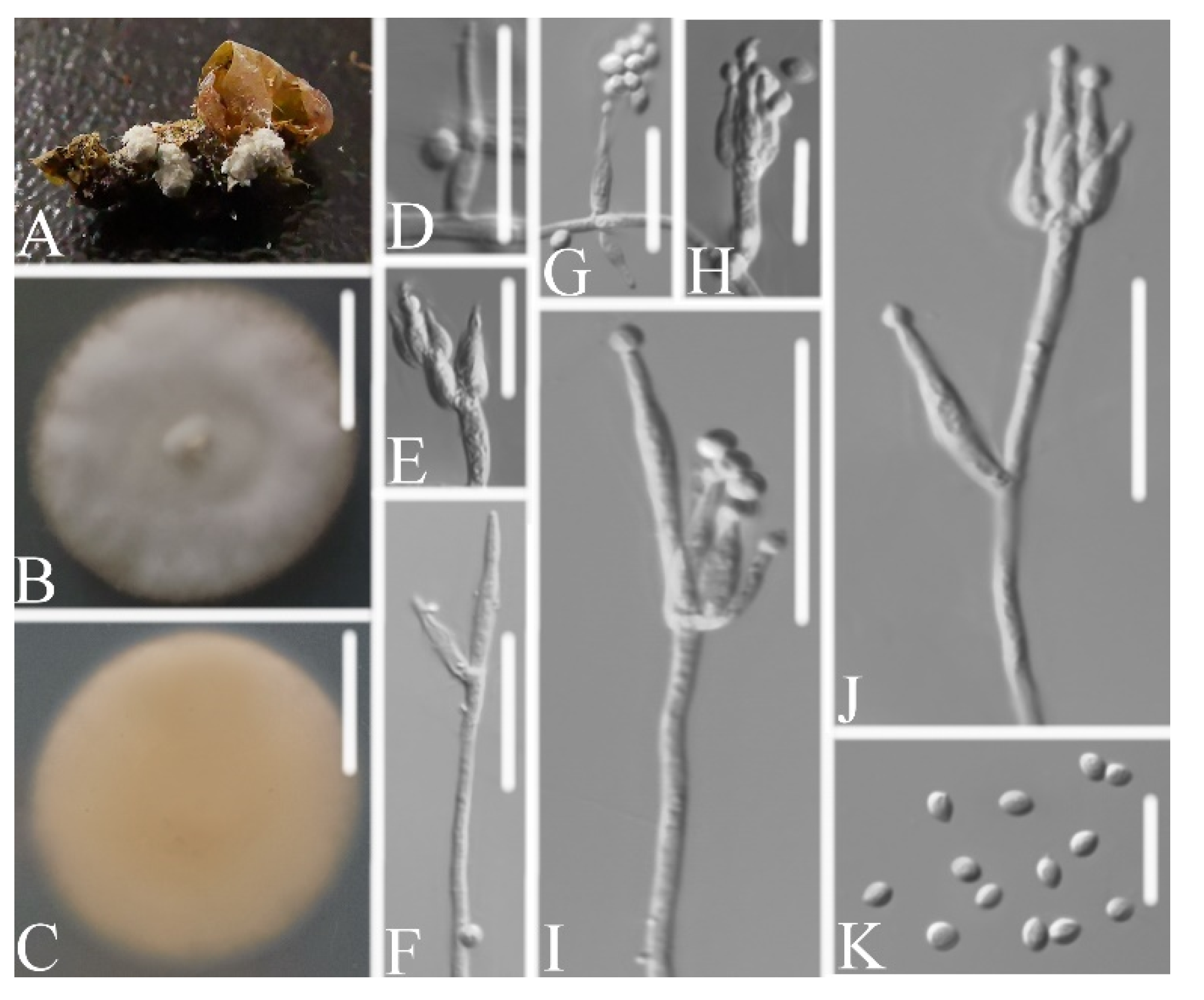
3.2.2. Samsoniella Pupicola W.H. Chen, Y.F. Han, J.D. Liang, and Z.Q. Liang, sp. nov.
3.2.3. Samsoniella aurantia Mongkols., Noisrip., Thanakitp., Spatafora, and Luangsa-ard, Mycologia 110(1): 249
3.2.4. Samsoniella hepiali (Q.T. Chen, and R.Q. Dai ex R.Q. Dai, X.M. Li, A.J. Shao, Shu F. Lin, J.L. Lan, Wei H. Chen, and C.Y. Shen) H. Yu, R.Q. Dai, Y.B. Wang, Y. Wang, and Zhu L. Yang, Fungal Diversity 103: 31
4. Discussion
Author Contributions
Funding
Institutional Review Board Statement
Informed Consent Statement
Data Availability Statement
Acknowledgments
Conflicts of Interest
References
- Fries, E.M. Systema Mycologicum; Ex Officina Berlingiana: Lund & Greifswald, Sweden, 1821; Volume 1, pp. 1–726. [Google Scholar]
- Brown, A.H.; Smith, G. The genus Paecilomyces Bainier and its perfect stage Byssochlamys Westling. Trans. Br. Mycol. Soc. 1957, 40, 17–89. [Google Scholar] [CrossRef]
- De Hoog, G.S. The genera Beauveria, Isaria, Tritirachium and Acrodontium Gen. Nov; Centraalbureau voor Schimmelcultures: Utrecht, The Netherlands, 1972; pp. 1–41. [Google Scholar]
- Samson, R.A. Paecilomyces and some allied Hyphomycetes; Centraalbureau voor Schimmelcultures: Utrecht, The Netherlands, 1974; Volume 6, pp. 1–119. [Google Scholar]
- Hodge, K.T.; Gams, W.; Samson, R.A.; Korf, R.P.; Seifert, K.A. Lectotypification and status of Isaria Pers.: Fr. Taxon 2005, 54, 485–489. [Google Scholar] [CrossRef]
- Luangsa-ard, J.J.; Hywel-Jones, N.L.; Samson, R.A. The order level polyphyletic nature of Paecilomyces sensu lato as revealed through 18S-generated rRNA phylogeny. Mycologia 2004, 96, 773–780. [Google Scholar] [CrossRef] [PubMed]
- Luangsa-Ard, J.J.; Hywel-Jones, N.L.; Manoch, L.; Samson, R.A. On the relationships of Paecilomyces sect. Isarioidea species. Mycol. Res. 2005, 109, 581–589. [Google Scholar] [CrossRef] [Green Version]
- Gams, W.; Hodge, K.T.; Samson, R.A.; Korf, R.P.; Seifert, K.A. (1684) Proposal to conserve the name Isaria (anamorphic fungi) with a conserved type. Taxon 2005, 54, 537. [Google Scholar] [CrossRef]
- Kepler, R.M.; Luangsa-ard, J.J.; Hywel-Jones, N.L.; Quandt, A.; Sung, G.-H.; Rehner, S.A.; Aime, M.C.; Henkel, T.W.; Sanjuan, T.; Zare, R.; et al. A phylogenetically-based nomenclature for Cordycipitaceae (Hypocreales). IMA Fungus 2017, 8, 335–353. [Google Scholar] [CrossRef]
- Mongkolsamrit, S.; Noisripoom, W.; Thanakitpipattana, D.; Wutikhun, T.; Spatafora, J.W.; Luangsa-ard, J. Disentangling cryptic species with isaria-like morphs in Cordycipitaceae. Mycologia 2018, 110, 230–257. [Google Scholar] [CrossRef]
- Chen, W.-H.; Liu, C.; Han, Y.-F.; Liang, J.-D.; Liang, Z.-Q. Akanthomyces araneogenum, a new Isaria-like araneogenous species. Phytotaxa 2018, 379, 66–72. [Google Scholar] [CrossRef] [Green Version]
- Chen, W.H.; Han, Y.F.; Liang, J.D.; Tian, W.Y.; Liang, Z.Q. Morphological and phylogenetic characterizations reveal three new species of Samsoniella (Cordycipitaceae, Hypocreales) from Guizhou, China. MycoKeys 2020, 74, 1–15. [Google Scholar] [CrossRef] [PubMed]
- Chen, W.-H.; Liu, C.; Han, Y.-F.; Liang, J.-D.; Tian, W.-Y.; Liang, Z.-Q. Three novel insect-associated species of Simplicillium (Cordycipitaceae, Hypocreales) from Southwest China. MycoKeys 2019, 58, 83–102. [Google Scholar] [CrossRef] [PubMed]
- Zou, X.; Liu, A.; Liang, Z.; Han, Y.; Yang, M. Hirsutella liboensis, a new entomopathogenic species affecting Cossidae (Lepidoptera) in China. Mycotaxon 2010, 111, 39–44. [Google Scholar] [CrossRef]
- Liang, J.D.; Han, Y.F.; Zhang, J.W.; Du, W.; Liang, Z.Q.; Li, Z.Z. Optimal culture conditions for keratinase production by a novel thermophilic Myceliophthora thermophila strain GZUIFR-H49-1. J. Appl. Microbiol. 2011, 110, 871–880. [Google Scholar] [CrossRef]
- White, T.; Bruns, T.; Lee, S.; Taylor, J. Amplification and Direct Sequencing of Fungal Ribosomal RNA Genes for Phylogenetics. In PCR Protocols: A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar] [CrossRef]
- Rakotonirainy, M.; Cariou, M.; Brygoo, Y.; Riba, G. Phylogenetic relationships within the genus Metarhizium based on 28S rRNA sequences and isozyme comparison. Mycol. Res. 1994, 98, 225–230. [Google Scholar] [CrossRef]
- Castlebury, L.A.; Rossman, A.Y.; Sung, G.-H.; Hyten, A.S.; Spatafora, J.W. Multigene phylogeny reveals new lineage for Stachybotrys chartarum, the indoor air fungus. Mycol. Res. 2004, 108, 864–872. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Van den Brink, J.; Samson, R.A.; Hagen, F.; Boekhout, T.; de Vries, R.P. Phylogeny of the industrial relevant, thermophilic genera Myceliophthora and Corynascus. Fungal Divers. 2012, 52, 197–207. [Google Scholar] [CrossRef] [Green Version]
- Wang, Y.-B.; Wang, Y.; Fan, Q.; Duan, D.-E.; Zhang, G.-D.; Dai, R.-Q.; Dai, Y.-D.; Zeng, W.-B.; Chen, Z.-H.; Li, D.-D.; et al. Multigene phylogeny of the family Cordycipitaceae (Hypocreales): New taxa and the new systematic position of the Chinese cordycipitoid fungus Paecilomyces hepiali. Fungal Divers. 2020, 103, 1–46. [Google Scholar] [CrossRef]
- Katoh, K.; Standley, D.M. MAFFT multiple sequence alignment software version 7: Improvements in performance and usability. Mol. Biol. Evol. 2013, 30, 772–780. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tamura, K.; Stecher, G.; Peterson, D.; Filipski, A.; Kumar, S. MEGA6: Molecular evolutionary genetics analysis version 6.0. Mol. Biol. Evol. 2013, 30, 2725–2729. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Vaidya, G.; Lohman, D.J.; Meier, R. SequenceMatrix: Concatenation software for the fast assembly of multi-gene datasets with character set and codon information. Cladistics 2011, 27, 171–180. [Google Scholar] [CrossRef]
- Kalyaanamoorthy, S.; Minh, B.Q.; Wong, T.; Von Haeseler, A.; Jermiin, L.S. ModelFinder: Fast model selection for accurate phylogenetic estimates. Nat. Methods 2017, 14, 587–589. [Google Scholar] [CrossRef] [Green Version]
- Zhang, D.; Gao, F.; Jakovlić, I.; Zou, H.; Zhang, J.; Li, W.X.; Wang, G.T. PhyloSuite: An integrated and scalable desktop platform for streamlined molecular sequence data management and evolutionary phylogenetics studies. Mol. Ecol. Resour. 2020, 20, 348–355. [Google Scholar] [CrossRef]
- Ronquist, F.; Teslenko, M.; Van Der Mark, P.; Ayres, D.L.; Darling, A.; Hoehna, S.; Larget, B.; Liu, L.; Suchard, M.A.; Huelsenbeck, J.P. MrBayes 3.2: Efficient Bayesian Phylogenetic Inference and Model Choice Across a Large Model Space. Syst. Biol. 2012, 61, 539–542. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Drummond, A.J.; Rambaut, A. BEAST: Bayesian evolutionary analysis by sampling trees. BMC Evol. Biol. 2007, 7, 214. [Google Scholar] [CrossRef] [Green Version]
- Silvestro, D.; Michalak, I. raxmlGUI: A graphical front-end for RAxML. Org. Divers. Evol. 2012, 12, 335–337. [Google Scholar] [CrossRef]
- Sung, G.-H.; Hywel-Jones, N.L.; Sung, J.-M.; Luangsa-ard, J.J.; Shrestha, B.; Spatafora, J.W. Phylogenetic classification of Cordyceps and the clavicipitaceous fungi. Stud. Mycol. 2007, 57, 5–59. [Google Scholar] [CrossRef] [Green Version]
- D’Alessandro, C.P.; Jones, L.R.; Humber, R.A.; López Lastra, C.C.; Sosa-Gomez, D.R. Characterization and phylogeny of Isaria spp. strains (Ascomycota: Hypocreales) using ITS 1-5.8 S-ITS 2 and elongation factor 1-alpha sequences. J. Basic Microbiol. 2014, 54, S21–S31. [Google Scholar] [CrossRef]
- Zimmermann, G. The entomopathogenic fungi Isaria farinosa (formerly Paecilomyces farinosus) and the Isaria fumosorosea species complex (formerly Paecilomyces fumosoroseus): Biology, ecology and use in biological control. Biocontrol Sci. Technol. 2008, 18, 865–901. [Google Scholar] [CrossRef]
- Lin, Y.; Liu, Y.J.; Wang, T.; Chen, M.J. Revision of taxonomic status of several Isaria-like strains. J. Microbiol. China 2021. (In Chinese) [Google Scholar] [CrossRef]
- Dai, R.-Q.; Shen, C.-Y.; Li, X.-M.; Lan, J.-L.; Lin, S.-F.; Shao, A.-J. Response to neotypification of Paecilomyces hepiali (Hypocreales) (Wang & al., 2015). Taxon 2018, 67, 784–786. [Google Scholar] [CrossRef] [Green Version]
| Name | Length | Direction | Sequence 5′-3′ | Optimised PCR Protocols | References | |
|---|---|---|---|---|---|---|
| ITS | ITS5 | 22 | forward | GGAAGTAAAAGTCGTAACAAGG | (95 °C: 30 s, 51 °C: 50 s, 72 °C: 45 s) × 33 cycles | [16] |
| ITS4 | 20 | reverse | TCCTCCGCTTATTGATATGC | |||
| LSU | LROR | 17 | forward | ACCCGCTGAACTTAAGC | (94 °C: 30 s, 51 °C: 1 min, 72 °C: 2 min) × 33 cycles | [17] |
| LR5 | 17 | reverse | TCCTGAGGGAAACTTCG | |||
| RPB1 | CRPB1 | 20 | forward | CAYCCWGGYTTYATCAAGAA | (94 °C: 30 s, 55 °C: 30 s, 72 °C: 1 min) × 33 cycles | [18] |
| RPB1Cr | 23 | reverse | CCNGCDATNTCRTTRTCCATRTA | |||
| RPB2 | RPB2-5F3 | 20 | forward | GACGACCGTGATCACTTTGG | (94 °C: 30 s, 54 °C: 40 s, 72 °C: 1 min 20 s) × 33 cycles | [19] |
| RPB2-7Cr2 | 20 | reverse | CCCATGGCCTGTTTGCCCAT | |||
| TEF | 983F | 23 | forward | GCYCCYGGHCAYCGTGAYTTYAT | (94 °C: 30 s, 58 °C: 1 min 20 s, 72 °C: 1 min) × 33 cycles | [19] |
| 2218R | 23 | reverse | ATGACACCRACRGCRACRGTYTG | |||
| Species | Strain No. | Host/ Substratum | GenBank Accession No. | ||||
|---|---|---|---|---|---|---|---|
| ITS | LSU | RPB1 | RPB2 | TEF | |||
| Akanthomyces aculeatus | HUA 772 | Lepidoptera; Sphingidae | - | KC519370 | - | - | KC519366 |
| A. attenuates | CBS 402.78 | Leaf litter (Acer saccharum) | - | AF339565 | EF468888 | EF468935 | EF468782 |
| A. coccidioperitheciatus | NHJ 6709 | Araneae (Spider) | - | EU369042 | EU369067 | - | EU369025 |
| A. farinosa | CBS 541.81 | - | AY624180 | MF416553 | MF416655 | MF416449 | JQ425686 |
| A. tuberculatus | BCC 16819 | Lepidoptera (Adult moth) | - | GQ249987 | - | - | GQ250037 |
| A. tuberculatus | OSC 111002 | Lepidoptera | - | DQ518767 | DQ522384 | - | DQ522338 |
| Ascopolyporus polychrous | P.C. 546 | Plant | - | DQ118737 | DQ127236 | - | DQ118745 |
| A. villosus | ARSEF 6355 | Plant | - | AY886544 | DQ127241 | - | DQ118750 |
| Beauveria bassiana | ARSEF 1564 | Lepidoptera; Arctiidae | - | - | HQ880833 | HQ880905 | HQ880974 |
| B. brongniartii | ARSEF 617 | Coleoptera; Scarabaeidae | - | AB027381 | HQ880854 | HQ880926 | HQ880991 |
| B. brongniartii | BCC 16585 | Coleoptera (Anomala cuprea) | JN049867 | JF415967 | JN049885 | JF415991 | JF416009 |
| B. caledonica | ARSEF 2567 | Soil | - | AF339520 | HQ880889 | HQ880961 | EF469057 |
| Bionectria ochroleuca | AFTOL-ID187 | - | - | DQ862027 | - | DQ862013 | DQ862029 |
| B. vericulosa | HMAS 183151 | Plant | HM050304 | HM050302 | - | - | - |
| Blackwellomyces cardinalis | OSC 93609 | Lepidoptera; Tineidae (Larva) | - | AY184962 | DQ522370 | - | DQ522325 |
| B. cardinalis | OSC 93610 | Lepidoptera; Tineidae (Larva) | - | AY184963 | EF469088 | - | EF469059 |
| B. pseudomilitaris | NBRC 101409 | Lepidoptera (Larva) | - | JN941393 | JN992482 | - | - |
| B. pseudomilitaris | NBRC 101410 | Lepidoptera (Larva) | - | JN941394 | JN992481 | - | - |
| Calcarisporium arbuscula | CBS 221.73 | - | AY271809 | - | - | - | - |
| C. arbuscula | CBS 900.68 | Hymenomycetes (Agarics sp.) | KT945003 | KX442598 | - | KX442597 | KX442596 |
| C. cordycipiticola | CGMCC 3.17904 | Cordycipitaceae (Cordyceps militaris) | KT945001 | KX442604 | - | KX442607 | KX442605 |
| C. cordycipiticola | CGMCC 3.17905 | Cordycipitaceae (Cordyceps militaris) | KT944999 | KX442599 | - | KX442594 | KX442593 |
| C. xylariicola | HMAS 276836 | Xylariaceae (Xylaria sp.) | KX442603 | KX442601 | - | KX442606 | KX442595 |
| Calonectria ilicicola | CBS 190.50 | Plant | GQ280605 | GQ280727 | - | KM232307 | AY725726 |
| Cephalosporium curtipes | CBS 154.61 | Uredinales (Hemileia vastatrix) | AJ292404 | AF339548 | - | EF468947 | EF468802 |
| Claviceps fusiformis | ATCC 26019 | Poaceae | JN049817 | - | - | - | DQ522320 |
| Clonostachys rosea | GJS 90-227 | Plant | - | AY489716 | - | - | AY489611 |
| Cocoonihabitus sinensis | HMAS 254523 | Saturniidae (Cocoon) | KY924870 | KY924869 | - | - | - |
| C. sinensis | HMAS 254524 | Saturniidae (Cocoon) | MF687395 | MF687396 | - | - | - |
| Cordyceps amoene-rosea | CBS 107.73 | Coleoptera (Pupa) | MH860646 | MH872342 | MF416651 | - | - |
| C. bifusispora | EFCC 5690 | Lepidoptera (Pupa) | - | EF468806 | EF468854 | EF468909 | EF468746 |
| C. cateniannulata | CBS 152.83 | Coleoptera (Adult) | NR_111169 | NG_067333 | - | - | - |
| C. cateniobliqua | CBS 153.83 | Lepidoptera (Adoxophyesprivatana) | NR_111170 | - | - | - | JQ425688 |
| C. cf. farinosa | OSC 111004 | Lepidoptera (Pupa) | - | EF468840 | EF468886 | - | EF468780 |
| C. coleopterorum | CBS 110.73 | Coleoptera (Larva) | AY624177 | JF415988 | JN049903 | JF416006 | JF416028 |
| C. farinosa | CBS 111113 | - | - | MF416554 | MF416656 | MF416450 | MF416499 |
| C. fumosorosea | CBS 107.10 | - | - | MF416556 | MF416659 | MF416453 | MF416502 |
| C. militaris | OSC 93623 | Lepidoptera (Pupa) | - | AY184966 | DQ522377 | AY545732 | DQ522332 |
| Dactylonectria alcacerensis | CBS 129087 | Plant (Vitis vinifera) | JF735333 | KM231629 | - | - | JF735819 |
| Elaphocordyceps ophioglossoides | NBRC106332 | - | JN943322 | JN941409 | - | - | - |
| E. paradoxa | NBRC 106958 | - | JN943324 | JN941411 | - | - | - |
| Engyodontium aranearum | CBS 309.85 | Araneae (Spider) | - | AF339526 | DQ522387 | DQ522439 | DQ522341 |
| Epichloë typhina | ATCC 56429 | Poaceae (Festuca rubra) | JN049832 | U17396 | - | DQ522440 | AF543777 |
| Flammocladiella aceris | CPC 24422 | Plant (Acer platanoides) | KR611883 | KR611901 | - | - | - |
| Flavocillium bifurcatum | YFCC 6101 | Noctuidae (Larva) | - | MN576781 | MN576841 | MN576897 | MN576951 |
| Fusarium circinatum | CBS 405.97 | - | U61677 | - | - | JX171623 | KM231943 |
| F. subluratum | CBS 189.34 | Soil | HQ897830 | KM231680 | - | - | - |
| Gelasinospora tetrasperma | AFTOL-ID 1287 | - | - | DQ470980 | - | DQ470932 | DQ471103 |
| Gibellula longispora | NHJ 12014 | Araneae (Spider) | - | - | EU369055 | - | EU369017 |
| G. pulchra | NHJ 10808 | Araneae (Spider) | - | EU369035 | EU369056 | - | EU369018 |
| G. ratticaudata | ARSEF 1915 | Araneae (Spider) | - | DQ518777 | DQ522408 | - | DQ522360 |
| Haptocillium sinense | CBS 567.95 | Nematode | AJ292417 | AF339545 | - | - | - |
| Harposporium harposporiferum | ARSEF 5472 | - | - | NG_060621 | - | - | - |
| Hevansia arachnophile | NHJ 10469 | Araneae (Spider) | - | EU369031 | EU369047 | - | EU369008 |
| H. cinerea | NHJ 3510 | Araneae (Spider) | - | - | EU369048 | - | EU369009 |
| H. nelumboides | BCC 41864 | Araneae (Spider) | JN201871 | JN201873 | - | - | JN201867 |
| H. novoguineensis | NHJ 11923 | Araneae (Spider) | - | EU369032 | EU369052 | - | EU369013 |
| Hyperdermium pulvinatum | P.C. 602 | Hemiptera (Scale insect) | - | DQ118738 | DQ127237 | - | DQ118746 |
| Hydropisphaera erubescens | ATCC 36093 | - | - | AF193230 | - | AY545731 | DQ518174 |
| H. peziza | GJS 92-101 | Plant (Bark) | - | AY489730 | - | - | AY489625 |
| Hypocrea americana | AFTOL-ID 52 | - | DQ491488 | AY544649 | - | - | DQ471043 |
| H. lutea | ATCC 208838 | On decorticated conifer wood | - | AF543791 | - | DQ522446 | AF543781 |
| H. rufa | DAOM JBT1003 | - | JN942883 | JN938865 | |||
| H. discoidea | BCC 8237 | - | JN049840 | DQ384937 | - | DQ452461 | DQ384977 |
| Hypomyces polyporinus | ATCC 76479 | - | - | AF543793 | - | - | AF543784 |
| Isaria farinosa | CEP 004 | Soil | JN998783 | - | - | - | JN998763 |
| I. farinosa | CEP 005 | Soil | JN998784 | - | - | - | JN998764 |
| I. farinosa | CEP 029 | Trialeurodes vaporariorum | JN998785 | - | - | - | JN998765 |
| I. farinosa | OSC 111005 | Lepidoptera (Pupa) | - | DQ518772 | DQ522394 | - | DQ522348 |
| I. farinosa | OSC 111006 | Lepidoptera (Pupa) | - | EF469080 | EF469094 | - | EF469065 |
| I. farinosa | OSC 111007 | Lepidoptera (Pupa) | - | DQ518773 | DQ522395 | DQ522449 | DQ522349 |
| Lecanicillium antillanum | CBS 350.85 | Hymenomycetes (Agaric sp.) | - | AF339536 | DQ522396 | DQ522450 | DQ522350 |
| L. attenuatum | CBS 402.78 | Leaf litter of Acer saccharum | - | AF339565 | EF468888 | EF468935 | EF468782 |
| L. aranearum | CBS 726.73a | Arachnida (Spider) | - | AF339537 | EF468887 | EF468934 | EF468781 |
| L. fusisporum | CBS 164.70 | Hymenomycetes (Coltricia perennis) | - | AF339549 | EF468889 | - | EF468783 |
| L. psalliotae | CBS 367.86 | Puccinia graminis | - | KM283800 | - | - | KM283823 |
| L. lecanii | CBS101247 | Hemiptera (Coccus viridis) | JN049836 | KM283794 | - | KM283859 | DQ522359 |
| Leptobacillium chinense | LC 1345 | submerged wood | - | JQ410322 | - | - | - |
| L. coffeanum | CDA 734 | Plant (Coffea arabica) | - | MF066032 | - | - | - |
| Liangia sinensis | YFCC 3103 | Fungi (Beauveria yunnanensis) | - | MN576782 | MN576842 | MN576898 | MN576952 |
| L. sinensis | YFCC 3104 | Fungi (Beauveria yunnanensis) | - | MN576783 | MN576843 | MN576899 | MN576953 |
| Metapochonia goniodes | CBS 891.72 | Fungi | AJ292409 | AF339550 | DQ522401 | DQ522458 | DQ522354 |
| Myrotheciomyces corymbiae | CPC 33206 | Plant (Corymbia variegata) | NR_160351 | NG_064542 | - | - | - |
| Myrothecium inundatum | IMI 158855 | Hymenomycetes (Russula nigricans) | - | AY489731 | - | - | AY489626 |
| M. roridum | ATCC 16297 | Soil | - | AY489708 | - | - | AY489603 |
| M. verrucaria | ATCC 9095 | Plant (Gossypium sp.) | - | AY489713 | - | - | AY489608 |
| Nectira cinnabarina | CBS 125165 | Plant (Aesculus sp.) | HM484548 | HM484562 | - | KM232402 | HM484527 |
| N. nigrescens | CBS 125148 | Plant (Dicotyledonous tree) | HM484707 | HM484720 | - | KM232403 | HM484672 |
| Nectriopsis violacea | CBS 424.64 | Fungi (Fuligo sp.) | - | AY489719 | - | - | - |
| Neobarya parasitica | Marson s/n | Fungi (Bertia moriformis) | KP899626 | KP899626 | - | - | - |
| Neonectria candida | CBS 151.29 | Plant (Malus sylvestris) | JF735313 | AY677333 | - | - | JF735791 |
| N. faginata | CBS 217.67 | - | HQ840385 | HQ840382 | - | DQ789797 | JF268746 |
| N. neomacrispora | CBS 118984 | - | HQ840388 | HQ840379 | - | DQ789810 | JF268754 |
| N. ramulariae | CBS 182.36 | - | HM054157 | HM042435 | - | DQ789793 | HM054092 |
| Neurospora crassa | ICMP 6360 | - | AY681193 | AY681158 | - | - | - |
| Niesslia exilis | CBS 560.74 | - | - | AY489720 | - | - | AY489614 |
| Ophiocordyceps heteropoda | EFCC 10125 | Cicadidae (Tibicen bihamatus) | JN049852 | EF468812 | - | EF468914 | EF468752 |
| O. sinensis | EFCC 7287 | Lepidoptera (Ghostmoth) | JN049854 | EF468827 | - | EF468924 | EF468767 |
| O. stylophor | OSC 111000 | Insect (Larvae) | JN049828 | DQ518766 | - | DQ522433 | DQ522337 |
| Peethambara spirostriata | CBS 110115 | Plant (Theobroma cacao) | - | AY489724 | - | EF692516 | AY489619 |
| Purpureocillium lilacinum | CBS 284.36 | Soil | - | AY624227 | EF468898 | EF468941 | EF468792 |
| P. lilacinum | CBS 431.87 | Nematoda (Meloidogyne sp.) | HQ842812 | EF468844 | EF468897 | EF468940 | EF468791 |
| Rosasphaeria moravica | LMM | - | JF440985 | - | - | JF440986 | JF440987 |
| Roumegueriella rufula | GJS 91-64 | - | - | EF469082 | - | EF469116 | EF469070 |
| R. rufula | CBS 346.85 | - | - | DQ518776 | - | DQ522461 | DQ522355 |
| Samsoniella alboaurantium | CBS 240.32 | Lepidoptera (Pupa) | - | JF415979 | JN049895 | JF415999 | JF416019 |
| S. alboaurantium | CBS 262.58 | Soil | - | AB080087 | MF416654 | MF416448 | MF416497 |
| S. alpina | YFCC 5818 | Hepialidae (Hepialus baimaensis) | - | MN576809 | MN576869 | MN576923 | MN576979 |
| S. alpina | YFCC 5831 | Hepialidae (Hepialus baimaensis) | - | MN576810 | MN576870 | MN576924 | MN576980 |
| S. antleroides | YFCC 6016 | Noctuidae (Larvae) | - | MN576803 | MN576863 | MN576917 | MN576973 |
| S. antleroides | YFCC 6113 | Noctuidae (Larvae) | - | MN576804 | MN576864 | MN576918 | MN576974 |
| S. aurantia | TBRC 7271 | Lepidoptera | - | MF140728 | MF140791 | MF140818 | MF140846 |
| S. aurantia | TBRC 7272 | Lepidoptera | - | MF140727 | - | MF140817 | MF140845 |
| S. aurantia | DY10951 | Lepidoptera (Pupa) | MZ827667 | MZ827827 | - | - | MZ855229 |
| S. aurantia | DY10952 | Lepidoptera (Pupa) | MZ827666 | MZ827084 | - | - | MZ855230 |
| S. cardinalis | YFCC 5830 | Limacodidae (Pupa) | - | MN576788 | MN576848 | MN576902 | MN576958 |
| S. cardinalis | YFCC 6144 | Limacodidae (Pupa) | - | MN576786 | MN576846 | MN576900 | MN576956 |
| S. coleopterorum | A19501 | Curculionidae (Snout beetle) | MT626376 | - | MT642600 | MN101585 | MN101586 |
| S. coleopterorum | A19502 | Curculionidae (Snout beetle) | MT626625 | - | MT642603 | MN101587 | MT642602 |
| S. cristata | YFCC 6021 | Saturniidae (Pupa) | - | MN576791 | MN576851 | MN576905 | MN576961 |
| S. cristata | YFCC 6023 | Saturniidae (Pupa) | - | MN576792 | MN576852 | MN576906 | MN576962 |
| S. hepiali | ICMM 82-2 | Fungi (Ophiocordyceps sinensis) | - | MN576794 | MN576854 | MN576908 | MN576964 |
| S. hepiali | ICMM Cs-4 | Fungi (Ophiocordycepssinensis) | - | MN576799 | MN576859 | MN576913 | MN576969 |
| S. hepiali | YFCC 661 | Fungi (Ophiocordycepssinensis) | - | MN576795 | MN576855 | MN576909 | MN576965 |
| S. hepiali | YJ06171 | Formicidae | MZ831866 | MZ831868 | MZ855241 | - | MZ855235 |
| S. hepiali | YJ06172 | Formicidae | MZ831867 | MZ831873 | - | - | MZ855236 |
| S. hymenopterorum | A19521 | Vespidae (Bee) | MN128224 | - | MT642601 | MT642604 | MN101588 |
| S. hymenopterorum | A19522 | Vespidae (Bee) | MN128081 | - | MN101589 | MN101590 | MN101591 |
| S. inthanonensis | TBRC 7915 | Lepidoptera (Pupa) | MF140761 | - | MF140790 | MF140815 | MF140849 |
| S. inthanonensis | TBRC 7916 | Lepidoptera (Pupa) | MF140760 | - | MF140789 | MF140814 | MF140848 |
| S. kunmingensis | YHH 16002 | Lepidoptera (Pupa) | - | MN576802 | MN576862 | MN576916 | MN576972 |
| S. lanmaoa | YFCC 6148 | Lepidoptera (Pupa) | - | MN576789 | MN576849 | MN576903 | MN576959 |
| S. lanmaoa | YFCC 6193 | Lepidoptera (Pupa) | - | MN576790 | MN576850 | MN576904 | MN576960 |
| S. lepidopterorum | DL10071 | Lepidoptera (Pupa) | MN128076 | - | MN101592 | MN101593 | MN101594 |
| S. lepidopterorum | DL10072 | Lepidoptera (Pupa) | MN128084 | - | - | MT642605 | MT642606 |
| S. pseudogunii | GY407201 | Lepidoptera (Larvae) | MZ827470 | MZ827010 | - | MZ855239 | MZ855233 |
| S. pseudogunii | GY407202 | Lepidoptera (Larvae) | MZ831863 | MZ831865 | - | MZ855240 | MZ855234 |
| S. pupicola | DY101681 | Lepidoptera (Pupa) | MZ827085 | MZ827009 | - | MZ855237 | MZ855231 |
| S. pupicola | DY101682 | Lepidoptera (Pupa) | MZ827008 | MZ827635 | - | MZ855238 | MZ855232 |
| S. ramose | YFCC 6020 | Limacodidae (Pupa) | - | MN576805 | MN576865 | MN576919 | MN576975 |
| S. tortricidae | YFCC 6013 | Limacodidae (Pupa) | - | MN576807 | MN576867 | MN576921 | MN576977 |
| S. tortricidae | YFCC 6131 | Limacodidae (Pupa) | - | MN576806 | MN576866 | MN576920 | MN576976 |
| S. yunnanensis | YFCC 1527 | Limacodidae (Pupa) | - | MN576812 | MN576872 | MN576926 | MN576982 |
| S. yunnanensis | YFCC 1824 | Limacodidae (Pupa) | - | MN576813 | MN576873 | MN576927 | MN576983 |
| Sarocladium bacillisporum | CBS 425.67 | Soil | NR_145039 | MH870718 | - | - | - |
| S. dejongiae | CBS 144929 | Soil | NR_161153 | NG_067854 | - | - | - |
| S. implicatum | CBS 959.72 | Soil | HG965023 | MH878470 | - | - | - |
| S. subulatum | CBS 217.35 | Soil | MH855652 | NG_070566 | - | - | - |
| S. terricola | CBS 243.59 | Soil | MH857853 | MH869389 | - | - | - |
| Shimizuomyces paradoxus | EFCC 6279 | Smilacaceae (Smilax sieboldii) | JN049847 | EF469084 | - | EF469117 | EF469071 |
| Simplicillium lamellicola | CBS 116.25 | Hymenomycetes (Agaricus bisporus) | AJ292393 | MH866307 | - | DQ522462 | DQ522356 |
| S. lanosoniveum | CBS 101267 | Uredinales (Hemileia vastatrix) | - | AJ292395 | - | DQ522463 | DQ522357 |
| S. lanosoniveum | CBS 704.86 | Uredinales (Hemileia vastatrix) | AJ292396 | AF339553 | - | DQ522464 | DQ522358 |
| Sordaria fimicola | AFTOL-ID 216 | - | DQ518178 | - | - | - | DQ518175 |
| Sphaerostilbella aureonitens | GJS74-87 | - | FJ442633 | HM466683 | - | FJ442763 | - |
| S. berkeleyana | GJS82-274 | - | - | U00756 | - | - | AF543783 |
| Stachybotrys chlorohalonata | DAOM 235557 | - | JN942888 | JN938870 | - | - | - |
| S. eucylindrospora | ATCC 18851 | - | JN942887 | JN938869 | - | - | - |
| Stephanonectria keithii | GJS92-133 | Plant (Bark) | - | AY489727 | - | - | AY489622 |
| Tilachlidium brachiatum | CBS 363.97 | Hymenomycetes (Agaricus sp.) | KM231838 | KM231719 | - | KM232414 | KM231975 |
| T. brachiatum | CBS 506.67 | Hymenomycetes (Hypholoma fasciculare) | KM231839 | HQ232177 | - | KM232415 | KM231976 |
| Tolypocladium inflatum | SCALT1007-002 | Sclerotium | KC963032 | - | - | - | - |
| Trichoderma aggresivum | CBS 100525 | - | - | JN939837 | - | JQ014130 | - |
| T. viride | GJS89-127 | Plant (Bark) | - | AY489726 | - | - | AY489621 |
| Trichothecium roseum | DUCC 502 | Plant (Solanum lycopersicum) | JN937590 | JX458860 | - | - | - |
| Valetoniellopsis laxa | GJS 96-174 | - | - | AY015635 | - | AY015638 | - |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (https://creativecommons.org/licenses/by/4.0/).
Share and Cite
Chen, W.; Liang, J.; Ren, X.; Zhao, J.; Han, Y.; Liang, Z. Cryptic Diversity of Isaria-like Species in Guizhou, China. Life 2021, 11, 1093. https://doi.org/10.3390/life11101093
Chen W, Liang J, Ren X, Zhao J, Han Y, Liang Z. Cryptic Diversity of Isaria-like Species in Guizhou, China. Life. 2021; 11(10):1093. https://doi.org/10.3390/life11101093
Chicago/Turabian StyleChen, Wanhao, Jiandong Liang, Xiuxiu Ren, Jiehong Zhao, Yanfeng Han, and Zongqi Liang. 2021. "Cryptic Diversity of Isaria-like Species in Guizhou, China" Life 11, no. 10: 1093. https://doi.org/10.3390/life11101093
APA StyleChen, W., Liang, J., Ren, X., Zhao, J., Han, Y., & Liang, Z. (2021). Cryptic Diversity of Isaria-like Species in Guizhou, China. Life, 11(10), 1093. https://doi.org/10.3390/life11101093





