Systemic Inflammation Associated with Immune Reconstitution Inflammatory Syndrome in Persons Living with HIV
Abstract
1. Introduction
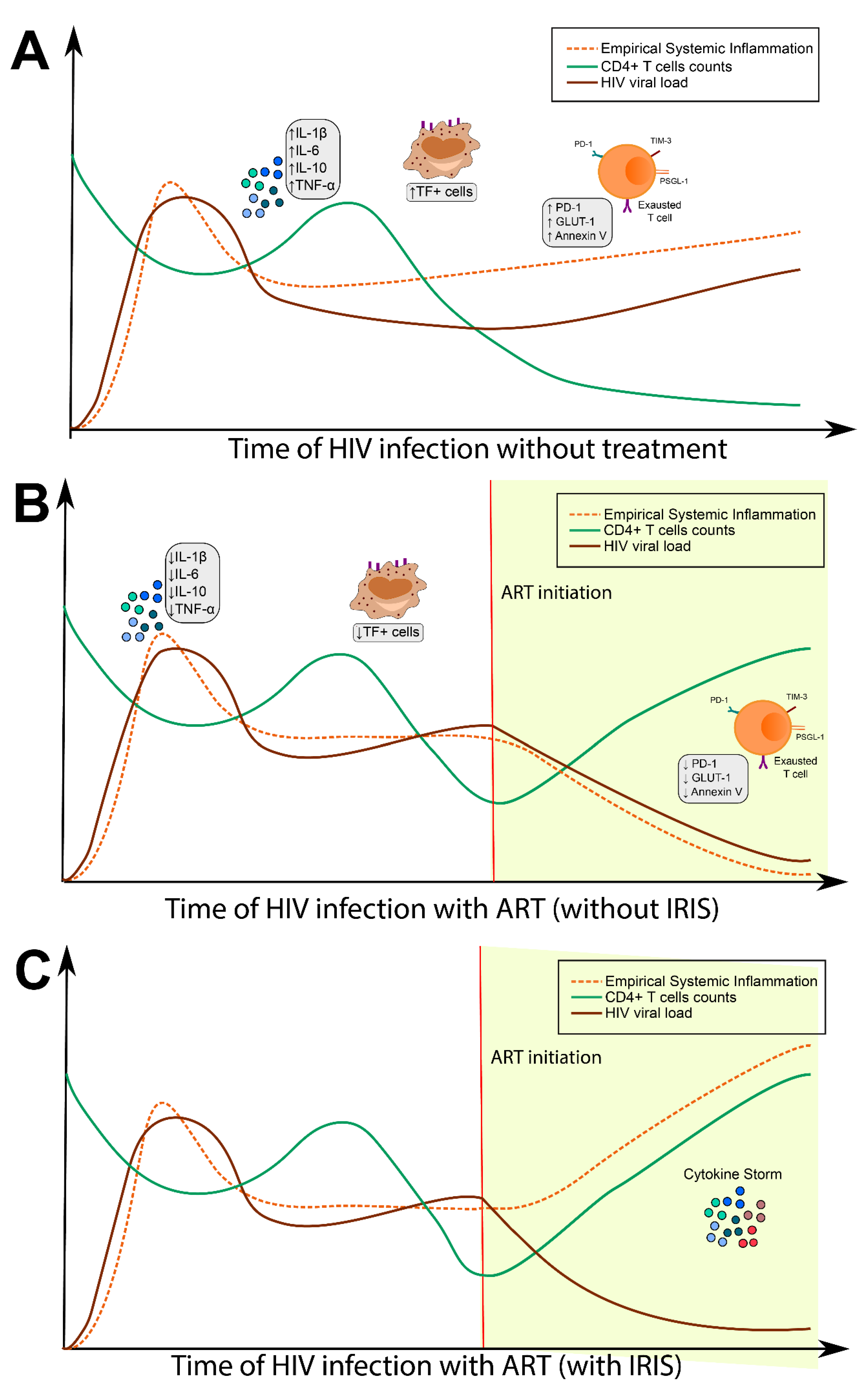
2. Inflammatory Activation in Persons Living with HIV and IRIS Development
2.1. Innate and Adaptive Immune Activation: Relationship with Progression of HIV
2.2. Exhaustion of T Cells and Contribution to Immune Dysregulation
2.3. Metabolic Consequences of Chronic Inflammation Associated with HIV Infection
2.4. Hypercoagulation as a Consequence of Persistent Immune Activation
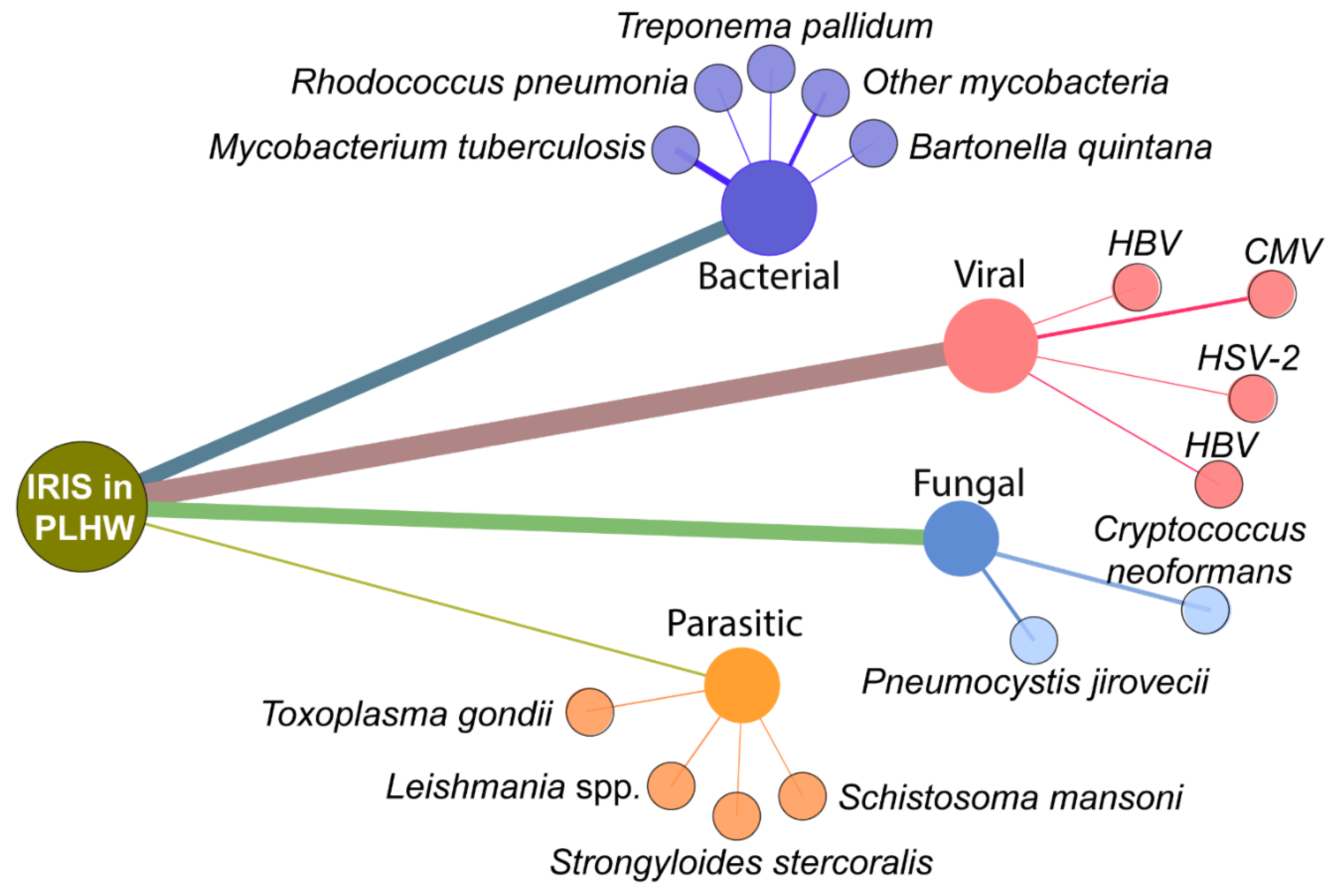
3. Opportunistic Infection in HIV and Its Impact on the Inflammatory Activation
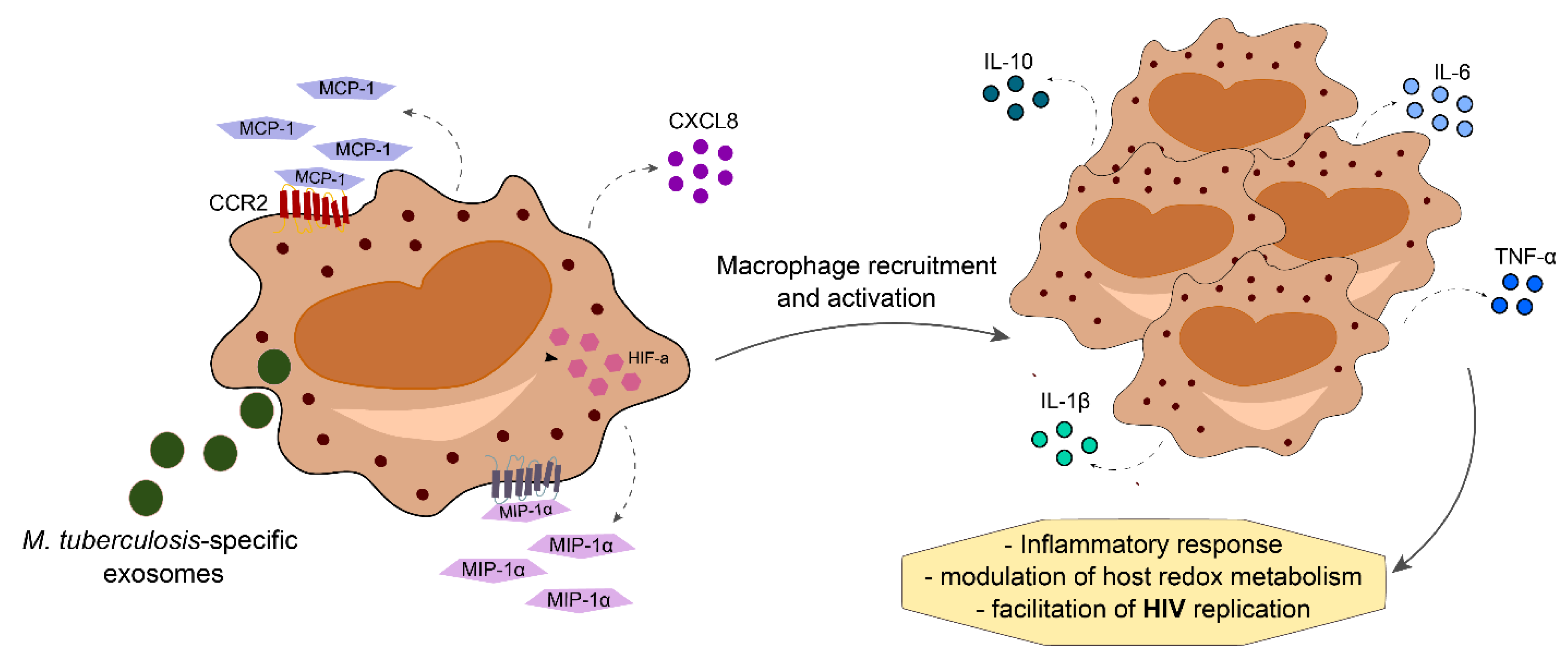
3.1. Mycobacterial Co-Infection
3.2. Viral Co-Infections
3.3. Fungal and Parasitic Co-Infections
4. Use of Host Markers to Predict IRIS
5. Host-Directed Therapies to Prevent and Treat IRIS in PLWH
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Abbreviations
| PLWH | People living with HIV |
| ART | Antiretroviral therapy |
| OI | Opportunistic infections |
| Mtb | Mycobacterium tuberculosis |
| MAC | Mycobacterium avium complex |
| CMV | Cytomegalovirus |
| KSHV | Kaposi sarcoma associated herpesvirus |
| HCV | Hepatitis C virus |
| HBV | Hepatitis B virus |
| IRIS | Immune Reconstitution Inflammatory Syndrome |
| SI | Systemic inflammation |
| NICMs | Noninfectious comorbidities |
| IL | Interleukin |
| TNF | Tissue necrosis factor |
| CRP | C-reactive protein |
| TLR | Toll-like receptors |
| AIDS | Acquired Immunodeficiency Syndrome |
| HLA | Human leukocyte antigen |
| CXCR | chemokine receptor |
| LBP | Lipopolysaccharide-binding protein |
| CXCL | C-X-C motif chemokine ligand |
| CCL | C–C motif chemokine ligand |
| PD | Programmed cell death receptor |
| CTLA | Cytotoxic T-lymphocyte-associated protein |
| LAG | Lymphocyte-activation gene |
| OXPHOS | Oxidative phosphorylation |
| ATP | Adenosine triphosphate |
| GLUT | Glucose transporter |
| HAND | HIV-associated neurocognitive disorder |
| CKD | Chronic kidney diseases |
| CVD | Cardiovascular disease |
| TF | Tissue factor |
| PAR | Protease activated receptor |
| WHO | World Health Organization |
| MIP | Macrophage inflammatory protein |
| HIF | Hypoxia-inducible factor |
| IFN | Interferon |
| MMPs | Matrix metalloproteinases |
| TIMP | tissue inhibitors of metalloproteinases |
| TB-IRIS | tuberculosis-associated immune reconstitution inflammatory syndrome |
| M-IRIS | Mycobacterial IRIS |
| C-IRIS | Cryptococcus IRIS |
| VZV | Varicella-zoster virus |
| EOD | End-organ diseases |
| HHV | Human herpesvirus |
| CSF | Cerebrospinal fluid |
| PJP | Pneumocystis pneumonia |
| LTA4H | leukotriene A4 hydroxylase |
| KIR | killer-cell immunoglobulin-like receptors |
| BMI | Body mass index |
| DTG | Dolutegravir |
| INSPIRING | International Study of Patients with HIV on Rifampicin ING |
References
- World Health Organization. HIV/AIDS. Available online: https://www.who.int/news-room/fact-sheets/detail/hiv-aids (accessed on 20 December 2020).
- Cevaal, P.M.; Bekker, L.-G.; Hermans, S. TB-IRIS pathogenesis and new strategies for intervention: Insights from related inflammatory disorders. Tuberculosis 2019, 118, 101863. [Google Scholar] [CrossRef] [PubMed]
- Sharma, S.K.; Soneja, M. HIV & immune reconstitution inflammatory syndrome (IRIS). Indian J. Med. Res. 2011, 134, 866–877. [Google Scholar] [CrossRef] [PubMed]
- Furman, D.; Campisi, J.; Verdin, E.; Carrera-Bastos, P.; Targ, S.; Franceschi, C.; Ferrucci, L.; Gilroy, D.W.; Fasano, A.; Miller, G.W.; et al. Chronic inflammation in the etiology of disease across the life span. Nat. Med. 2019, 25, 1822–1832. [Google Scholar] [CrossRef] [PubMed]
- Deeks, S.G.; Overbaugh, J.; Phillips, A.; Buchbinder, S. HIV infection. Nat. Rev. Dis. Prim. 2015, 1, 15035. [Google Scholar] [CrossRef]
- Deeks, S.G. HIV Infection, Inflammation, Immunosenescence, and Aging. Annu. Rev. Med. 2011, 62, 141–155. [Google Scholar] [CrossRef]
- Borges Álvaro, H.; O’Connor, J.L.; Phillips, A.N.; Rönsholt, F.F.; Pett, S.; Vjecha, M.J.; French, M.A.; Lundgren, J.D.; Insight, S.; Groups, E.S.; et al. Factors Associated with Plasma IL-6 Levels During HIV Infection. J. Infect. Dis. 2015, 212, 585–595. [Google Scholar] [CrossRef]
- Neuhaus, J.; Jacobs, D.R., Jr.; Baker, J.V.; Calmy, A.; Duprez, D.; La Rosa, A.; Kuller, L.H.; Pett, S.L.; Ristola, M.; Ross, M.J.; et al. Markers of Inflammation, Coagulation, and Renal Function Are Elevated in Adults with HIV Infection. J. Infect. Dis. 2010, 201, 1788–1795. [Google Scholar] [CrossRef]
- Hsu, D.C.; Breglio, K.F.; Pei, L.; Wong, C.-S.; Andrade, B.B.; Sheikh, V.; Smelkinson, M.; Petrovas, C.; Rupert, A.; Gil-Santana, L.; et al. Emergence of Polyfunctional Cytotoxic CD4+ T Cells in Mycobacterium avium Immune Reconstitution Inflammatory Syndrome in Human Immunodeficiency Virus-Infected Patients. Clin. Infect. Dis. 2018, 67, 437–446. [Google Scholar] [CrossRef]
- Paiardini, M.; Müller-Trutwin, M. HIV-associated chronic immune activation. Immunol. Rev. 2013, 254, 78–101. [Google Scholar] [CrossRef]
- Sereti, I.; Altfeld, M. Immune activation and HIV: An enduring relationship. Curr. Opin. HIV AIDS 2016, 11, 129–130. [Google Scholar] [CrossRef]
- Sokoya, T.; Steel, H.C.; Nieuwoudt, M.; Rossouw, T. HIV as a Cause of Immune Activation and Immunosenescence. Mediat. Inflamm. 2017, 2017, 1–16. [Google Scholar] [CrossRef] [PubMed]
- Iwasaki, A. Innate Immune Recognition of HIV-1. Immunity 2012, 37, 389–398. [Google Scholar] [CrossRef] [PubMed]
- Chang, J.J.; Lacas, A.; Lindsay, R.J.; Doyle, E.H.; Axten, K.L.; Pereyra, F.; Rosenberg, E.S.; Walker, B.D.; Allen, T.M.; Altfeld, M. Differential regulation of toll-like receptor pathways in acute and chronic HIV-1 infection. AIDS 2012, 26, 533–541. [Google Scholar] [CrossRef] [PubMed]
- Brown, J.N.; Kohler, J.J.; Coberley, C.R.; Sleasman, J.W.; Goodenow, M.M. HIV-1 Activates Macrophages Independent of Toll-Like Receptors. PLoS ONE 2008, 3, e3664. [Google Scholar] [CrossRef]
- Liu, Z.; Cumberland, W.G.; Hultin, L.E.; Prince, H.E.; Detels, R.; Giorgi, J.V. Elevated CD38 Antigen Expression on CD8+ T Cells Is a Stronger Marker for the Risk of Chronic HIV Disease Progression to AIDS and Death in the Multicenter AIDS Cohort Study Than CD4+ Cell Count, Soluble Immune Activation Markers, or Combinations of HLA-DR and CD38 Expression. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1997, 16, 83–92. [Google Scholar] [CrossRef]
- Hazenberg, M.D.; Stuart, J.W.; Otto, S.A.; Borleffs, J.C.; Boucher, C.A.; de Boer, R.J.; Miedema, F.; Hamann, D. T-cell division in human immunodeficiency virus (HIV)-1 infection is mainly due to immune activation: A longitudinal analysis in patients before and during highly active antiretroviral therapy (HAART). Blood 2000, 95, 249–255. [Google Scholar] [CrossRef]
- Connell, B.J.; Hermans, L.E.; Wensing, A.M.J.; Schellens, I.; Schipper, P.J.; Van Ham, P.M.; De Jong, D.T.C.M.; Otto, S.; Mathe, T.; Moraba, R.; et al. Immune activation correlates with and predicts CXCR4 co-receptor tropism switch in HIV-1 infection. Sci. Rep. 2020, 10, 1–10. [Google Scholar] [CrossRef]
- van den Dries, L.; Claassen, M.A.A.; Groothuismink, Z.M.A.; Van Gorp, E.; Boonstra, A. Immune activation in prolonged cART-suppressed HIV patients is comparable to that of healthy controls. Virology 2017, 509, 133–139. [Google Scholar] [CrossRef]
- Kaur, A.; Hale, C.L.; Ramanujan, S.; Jain, R.K.; Johnson, R.P. Differential Dynamics of CD4+ and CD8+ T-Lymphocyte Proliferation and Activation in Acute Simian Immunodeficiency Virus Infection. J. Virol. 2000, 74, 8413–8424. [Google Scholar] [CrossRef]
- Kroeze, S.; Wit, F.W.; Rossouw, T.M.; Steel, H.C.; Kityo, C.M.; Siwale, M.; Akanmu, S.; Mandaliya, K.; De Jager, M.; Ondoa, P.; et al. Plasma Biomarkers of Human Immunodeficiency Virus–Related Systemic Inflammation and Immune Activation in Sub-Saharan Africa Before and During Suppressive Antiretroviral Therapy. J. Infect. Dis. 2019, 220, 1029–1033. [Google Scholar] [CrossRef]
- Yi, J.S.; Cox, M.A.; Zajac, A.J. T-cell exhaustion: Characteristics, causes and conversion. Immunology 2010, 129, 474–481. [Google Scholar] [CrossRef] [PubMed]
- Wherry, E.J. T cell exhaustion. Nat. Immunol. 2011, 12, 492–499. [Google Scholar] [CrossRef] [PubMed]
- Blank, C.U.; Haining, W.N.; Held, W.; Hogan, P.G.; Kallies, A.; Lugli, E.; Lynn, R.C.; Philip, M.; Rao, A.; Restifo, N.P.; et al. Defining ‘T cell exhaustion’. Nat. Rev. Immunol. 2019, 19, 665–674. [Google Scholar] [CrossRef] [PubMed]
- Fenwick, C.; Joo, V.; Jacquier, P.; Noto, A.; Banga, R.; Perreau, M.; Pantaleo, G. T-cell exhaustion in HIV infection. Immunol. Rev. 2019, 292, 149–163. [Google Scholar] [CrossRef] [PubMed]
- Day, C.L.; Kaufmann, D.E.; Kiepiela, P.; Brown, J.A.; Moodley, E.S.; Reddy, S.; Mackey, E.W.; Miller, J.D.; Leslie, A.J.; DePierres, C.; et al. PD-1 expression on HIV-specific T cells is associated with T-cell exhaustion and disease progression. Nature 2006, 443, 350–354. [Google Scholar] [CrossRef]
- Macatangay, B.J.C.; Gandhi, R.T.; Jones, R.B.; McMahon, D.K.; Lalama, C.M.; Bosch, R.J.; Cyktor, J.C.; Thomas, A.S.; Borowski, L.; Riddler, S.A.; et al. T cells with high PD-1 expression are associated with lower HIV-specific immune responses despite long-term antiretroviral therapy. AIDS 2020, 34, 15–24. [Google Scholar] [CrossRef]
- Hoffmann, M.; Pantazis, N.; Martin, G.E.; Hickling, S.; Hurst, J.; Meyerowitz, J.; Willberg, C.B.; Robinson, N.; Brown, H.; Fisher, M.; et al. Exhaustion of Activated CD8 T Cells Predicts Disease Progression in Primary HIV-1 Infection. PLoS Pathog. 2016, 12, e1005661. [Google Scholar] [CrossRef]
- Rallón, N.; García, M.; García-Samaniego, J.; Cabello, A.; Álvarez, B.; Restrepo, C.; Nistal, S.; Górgolas, M.; Benito, J.M. Expression of PD-1 and Tim-3 markers of T-cell exhaustion is associated with CD4 dynamics during the course of untreated and treated HIV infection. PLoS ONE 2018, 13, e0193829. [Google Scholar] [CrossRef]
- Buck, M.D.; O’Sullivan, D.; Pearce, E.L. T cell metabolism drives immunity. J. Exp. Med. 2015, 212, 1345–1360. [Google Scholar] [CrossRef]
- Maciver, N.J.; Michalek, R.D.; Rathmell, J.C. Metabolic Regulation of T Lymphocytes. Annu. Rev. Immunol. 2013, 31, 259–283. [Google Scholar] [CrossRef]
- Pearce, E.L.; Poffenberger, M.C.; Chang, C.-H.; Jones, R.G. Fueling Immunity: Insights into Metabolism and Lymphocyte Function. Science 2013, 342, 1242454. [Google Scholar] [CrossRef] [PubMed]
- Gaber, T.; Strehl, C.; Buttgereit, T.G.C.S.F. Metabolic regulation of inflammation. Nat. Rev. Rheumatol. 2017, 13, 267–279. [Google Scholar] [CrossRef] [PubMed]
- Sáez-Cirión, A.; Sereti, I. Immunometabolism and HIV-1 pathogenesis: Food for thought. Nat. Rev. Immunol. 2020, 21, 5–19. [Google Scholar] [CrossRef] [PubMed]
- Wang, R.; Green, D.R. The immune diet: Meeting the metabolic demands of lymphocyte activation. F1000 Biol. Rep. 2012, 4, 9. [Google Scholar] [CrossRef] [PubMed]
- Pålsson-McDermott, E.M.; O’Neill, L.A. The Warburg effect then and now: From cancer to inflammatory diseases. BioEssays 2013, 35, 965–973. [Google Scholar] [CrossRef]
- Marelli-Berg, F.M.; Fu, H.; Mauro, C. Molecular mechanisms of metabolic reprogramming in proliferating cells: Implications for T-cell-mediated immunity. Immunology 2012, 136, 363–369. [Google Scholar] [CrossRef]
- Palmer, C.S.; Ostrowski, M.; Gouillou, M.; Tsai, L.; Yu, D.; Zhou, J.; Henstridge, D.C.; Maisa, A.; Hearps, A.C.; Lewin, S.R.; et al. Increased glucose metabolic activity is associated with CD4+ T-cell activation and depletion during chronic HIV infection. AIDS 2014, 28, 297–309. [Google Scholar] [CrossRef]
- Palmer, C.S.; Cherry, C.L.; Sada-Ovalle, I.; Singh, A.; Crowe, S.M. Glucose Metabolism in T Cells and Monocytes: New Perspectives in HIV Pathogenesis. EBioMedicine 2016, 6, 31–41. [Google Scholar] [CrossRef]
- Butterfield, T.R.; Landay, A.L.; Anzinger, J.J. Dysfunctional Immunometabolism in HIV Infection: Contributing Factors and Implications for Age-Related Comorbid Diseases. Curr. HIV/AIDS Rep. 2020, 17, 125–137. [Google Scholar] [CrossRef]
- Peltenburg, N.C.; Schoeman, J.C.; Hou, J.; Mora, F.; Harms, A.C.; Lowe, S.H.; Bierau, J.; Bakker, J.A.; Verbon, A.; Hankemeier, T.; et al. Persistent metabolic changes in HIV-infected patients during the first year of combination antiretroviral therapy. Sci. Rep. 2018, 8, 16947. [Google Scholar] [CrossRef]
- Hsue, P.Y.; Waters, D.D. HIV infection and coronary heart disease: Mechanisms and management. Nat. Rev. Cardiol. 2019, 16, 745–759. [Google Scholar] [CrossRef] [PubMed]
- Bibas, M.; Biava, G.; Antinori, A. HIV-Associated venous thromboembolism. Mediterr. J. Hematol. Infect. Dis. 2011, 3, e2011030. [Google Scholar] [CrossRef] [PubMed]
- Shen, Y.-M.P.; Frenkel, E.P. Thrombosis and a Hypercoagulable State in HIV-Infected Patients. Clin. Appl. Thromb. 2004, 10, 277–280. [Google Scholar] [CrossRef] [PubMed]
- Baker, J. Chronic HIV disease and activation of the coagulation system. Thromb. Res. 2013, 132, 495–499. [Google Scholar] [CrossRef]
- Mackman, N. The Role of Tissue Factor and Factor VIIa in Hemostasis. Anesth. Analg. 2009, 108, 1447–1452. [Google Scholar] [CrossRef]
- Schechter, M.E.; Andrade, B.B.; He, T.; Richter, G.H.; Tosh, K.W.; Policicchio, B.B.; Singh, A.P.; Raehtz, K.D.; Sheikh, V.; Ma, D.; et al. Inflammatory monocytes expressing tissue factor drive SIV and HIV coagulopathy. Sci. Transl. Med. 2017, 9, eaam5441. [Google Scholar] [CrossRef]
- Boulougoura, A.; Sereti, I. HIV infection and immune activation: The role of coinfections. Curr. Opin. HIV AIDS 2016, 11, 191–200. [Google Scholar] [CrossRef]
- Gopalan, N.; Andrade, B.B.; Swaminathan, S. Tuberculosis-immune reconstitution inflammatory syndrome in HIV: From pathogenesis to prediction. Expert Rev. Clin. Immunol. 2014, 10, 631–645. [Google Scholar] [CrossRef]
- Barber, D.L.; Andrade, B.B.; Sereti, I.; Sher, A. Immune reconstitution inflammatory syndrome: The trouble with immunity when you had none. Nat. Rev. Microbiol. 2012, 10, 150–156. [Google Scholar] [CrossRef]
- Sereti, I.; Sheikh, V.; Shaffer, D.; Phanuphak, N.; Gabriel, E.E.; Wang, J.; Nason, M.C.; Roby, G.; Ngeno, H.; Kirui, F.; et al. Prospective International Study of Incidence and Predictors of Immune Reconstitution Inflammatory Syndrome and Death in People Living with Human Immunodeficiency Virus and Severe Lymphopenia. Clin. Infect. Dis. 2020, 71, 652–660. [Google Scholar] [CrossRef]
- World Health Organization. Global Tuberculosis Report 2020; WHO: Geneva, Switzerland, 2020. [Google Scholar]
- Nightingale, S.D.; Byrd, L.T.; Southern, P.M.; Jockusch, J.D.; Cal, S.X.; Wynne, B.A. Incidence of Mycobacterium avium-intracellulare Complex Bacteremia in Human Immunodeficiency Virus-Positive Patients. J. Infect. Dis. 1992, 165, 1082–1085. [Google Scholar] [CrossRef] [PubMed]
- Gilks, C.; Brindle, R.J.; Mwachari, C.; Batchelor, B.; Bwayo, J.; Kimari, J.; Arbeit, R.D.; Von Reyn, C.F. Disseminated Mycobacterium avium infection among HIV-infected patients in Kenya. J. Acquir. Immune Defic. Syndr. Hum. Retrovirol. 1995, 8, 195–198. [Google Scholar] [CrossRef] [PubMed]
- Breglio, K.F.; Vinhaes, C.L.; Arriaga, M.B.; Nason, M.; Roby, G.; Adelsberger, J.; Andrade, B.B.; Sheikh, V.; Sereti, I. Clinical and immunologic predictors of Mycobacterium avium complex immune reconstitution inflammatory syndrome in a contemporary cohort of patients with HIV. J. Infect. Dis. 2020. [Google Scholar] [CrossRef] [PubMed]
- Gopalan, N.; Oliveira-De-Souza, D.; Vinhaes, C.L.; Akrami, K.; Fukutani, K.F.; Banu, K.; Padmapriyadarsini, C.; Ravichandran, N.; Andrade, B.B.; Swaminathan, S.; et al. Multifocal tuberculosis-associated immune reconstitution inflammatory syndrome—A case report of a complicated scenario. BMC Infect. Dis. 2019, 19, 529. [Google Scholar] [CrossRef]
- Narendran, G.; Andrade, B.B.; Porter, B.O.; Chandrasekhar, C.; Venkatesan, P.; Menon, P.A.; Subramanian, S.; Anbalagan, S.; Bhavani, K.P.; Sekar, S.; et al. Paradoxical Tuberculosis Immune Reconstitution Inflammatory Syndrome (TB-IRIS) in HIV Patients with Culture Confirmed Pulmonary Tuberculosis in India and the Potential Role of IL-6 in Prediction. PLoS ONE 2013, 8, e63541. [Google Scholar] [CrossRef]
- Manabe, Y.C.; Campbell, J.D.; Sydnor, E.; Moore, R.D. Immune Reconstitution Inflammatory Syndrome: Risk factors and treatment implications. JAIDS J. Acquir. Immune Defic. Syndr. 2007, 46, 456–462. [Google Scholar] [CrossRef]
- Price, P.; Murdoch, D.M.; Agarwal, U.; Lewin, S.R.; Elliott, J.H.; French, M.A. Immune Restoration Diseases Reflect Diverse Immunopathological Mechanisms. Clin. Microbiol. Rev. 2009, 22, 651–663. [Google Scholar] [CrossRef]
- Letang, E.; Miró, J.M.; Nhampossa, T.; Ayala, E.; Gascón, J.; Menéndez, C.; Alonso, P.L.; Naniche, D. Incidence and Predictors of Immune Reconstitution Inflammatory Syndrome in a Rural Area of Mozambique. PLoS ONE 2011, 6, e16946. [Google Scholar] [CrossRef][Green Version]
- Vinhaes, C.L.; De Souza, D.O.; Silveira-Mattos, P.S.; Nogueira, B.; Shi, R.; Wei, W.; Yuan, X.; Zhang, G.; Cai, Y.; Barry, C.E., 3rd; et al. Changes in inflammatory protein and lipid mediator profiles persist after antitubercular treatment of pulmonary and extrapulmonary tuberculosis: A prospective cohort study. Cytokine 2019, 123, 154759. [Google Scholar] [CrossRef]
- Oliveira-De-Souza, D.; Vinhaes, C.L.; Arriaga, M.B.; Kumar, N.P.; Cubillos-Angulo, J.M.; Shi, R.; Wei, W.; Yuan, X.; Zhang, G.; Cai, Y.; et al. Molecular degree of perturbation of plasma inflammatory markers associated with tuberculosis reveals distinct disease profiles between Indian and Chinese populations. Sci. Rep. 2019, 9, 8002. [Google Scholar] [CrossRef]
- Tyagi, P.; Pal, V.K.; Agrawal, R.; Singh, S.; Srinivasan, S.; Singh, A. Mycobacterium tuberculosis Reactivates HIV-1 via Exosome-Mediated Resetting of Cellular Redox Potential and Bioenergetics. mBio 2020, 11, e03293-19. [Google Scholar] [CrossRef] [PubMed]
- Schutz, C.; Barr, D.; Andrade, B.B.; Shey, M.S.; Ward, A.; Janssen, S.; Burton, R.; Wilkinson, R.; Sossen, B.; Fukutani, K.F.; et al. Clinical, microbiologic, and immunologic determinants of mortality in hospitalized patients with HIV-associated tuberculosis: A prospective cohort study. PLoS Med. 2019, 16, e1002840. [Google Scholar] [CrossRef] [PubMed]
- Weiss, G.; Goodnough, L.T. Anemia of Chronic Disease. N. Engl. J. Med. 2005, 352, 1011–1023. [Google Scholar] [CrossRef] [PubMed]
- Shivakoti, R.; Yang, W.-T.; Gupte, N.; Berendes, S.; La Rosa, A.; Cardoso, S.W.; Mwelase, N.; Kanyama, C.; Pillay, S.; Samaneka, W.; et al. Concurrent Anemia and Elevated C-Reactive Protein Predicts HIV Clinical Treatment Failure, Including Tuberculosis, After Antiretroviral Therapy Initiation. Clin. Infect. Dis. 2015, 61, 102–110. [Google Scholar] [CrossRef]
- Chu, K.-A.; Hsu, C.-H.; Lin, M.-C.; Chu, Y.-H.; Hung, Y.-M.; Wei, J.C.-C. Association of iron deficiency anemia with tuberculosis in Taiwan: A nationwide population-based study. PLoS ONE 2019, 14, e0221908. [Google Scholar] [CrossRef]
- Belperio, P.S.; Rhew, D.C. Prevalence and outcomes of anemia in individuals with human immunodeficiency virus: A systematic review of the literature. Am. J. Med. 2004, 116 (Suppl. 7A), 27S–43S. [Google Scholar] [CrossRef]
- Pai, M.; Behr, M.A.; Dowdy, D.; Dheda, K.; Divangahi, M.; Boehme, C.C.; Ginsberg, A.; Swaminathan, S.; Spigelman, M.; Getahun, H.; et al. Tuberculosis. Nat. Rev. Dis. Prim. 2016, 2, 16076. [Google Scholar] [CrossRef]
- Barber, D.L.; Mayer-Barber, K.D.; Feng, C.G.; Sharpe, A.H.; Sher, A. CD4 T Cells Promote Rather than Control Tuberculosis in the Absence of PD-1–Mediated Inhibition. J. Immunol. 2011, 186, 1598–1607. [Google Scholar] [CrossRef]
- Tan, H.Y.; Yong, Y.K.; Shankar, E.M.; Paukovics, G.; Ellegård, R.; Larsson, M.; Kamarulzaman, A.; French, M.A.; Crowe, S.M. Aberrant Inflammasome Activation Characterizes Tuberculosis-Associated Immune Reconstitution Inflammatory Syndrome. J. Immunol. 2016, 196, 4052–4063. [Google Scholar] [CrossRef]
- Mahnke, Y.D.; Greenwald, J.H.; DerSimonian, R.; Roby, G.; Antonelli, L.R.V.; Sher, A.; Roederer, M.; Sereti, I. Selective expansion of polyfunctional pathogen-specific CD4+ T cells in HIV-1–infected patients with immune reconstitution inflammatory syndrome. Blood 2012, 119, 3105–3112. [Google Scholar] [CrossRef]
- Antonelli, L.R.V.; Mahnke, Y.; Hodge, J.N.; Porter, B.O.; Barber, D.L.; DerSimonian, R.; Greenwald, J.H.; Roby, G.; Mican, J.; Sher, A.; et al. Elevated frequencies of highly activated CD4+ T cells in HIV+ patients developing immune reconstitution inflammatory syndrome. Blood 2010, 116, 3818–3827. [Google Scholar] [CrossRef] [PubMed]
- Bourgarit, A.; Carcelain, G.; Martinez, V.; Lascoux, C.; Delcey, V.; Gicquel, B.; Vicaut, E.; Lagrange, P.H.; Sereni, D.; Autran, B. Explosion of tuberculin-specific Th1-responses induces immune restoration syndrome in tuberculosis and HIV co-infected patients. AIDS 2006, 20, F1–F7. [Google Scholar] [CrossRef] [PubMed]
- Tadokera, R.; Meintjes, G.; Skolimowska, K.H.; Wilkinson, K.A.; Matthews, K.; Seldon, R.; Chegou, N.N.; Maartens, G.; Rangaka, M.X.; Rebe, K.; et al. Hypercytokinaemia accompanies HIV-tuberculosis immune reconstitution inflammatory syndrome. Eur. Respir. J. 2011, 37, 1248–1259. [Google Scholar] [CrossRef] [PubMed]
- Andrade, B.B.; Singh, A.; Narendran, G.; Schechter, M.E.; Nayak, K.; Subramanian, S.; Anbalagan, S.; Jensen, S.M.R.; Porter, B.O.; Antonelli, L.R.D.V.; et al. Mycobacterial Antigen Driven Activation of CD14++CD16− Monocytes Is a Predictor of Tuberculosis-Associated Immune Reconstitution Inflammatory Syndrome. PLoS Pathog. 2014, 10, e1004433. [Google Scholar] [CrossRef] [PubMed]
- Tadokera, R.; Meintjes, G.A.; Wilkinson, K.A.; Skolimowska, K.H.; Walker, N.; Friedland, J.S.; Maartens, G.; Elkington, P.T.G.; Wilkinson, R.J. Matrix metalloproteinases and tissue damage in HIV -tuberculosis immune reconstitution inflammatory syndrome. Eur. J. Immunol. 2014, 44, 127–136. [Google Scholar] [CrossRef]
- Amaral, E.P.; Vinhaes, C.L.; Oliveira-De-Souza, D.; Nogueira, B.; Akrami, K.M.; Andrade, B.B. The Interplay Between Systemic Inflammation, Oxidative Stress, and Tissue Remodeling in Tuberculosis. Antioxid. Redox Signal. 2020. [Google Scholar] [CrossRef]
- Kübler, A.; Luna, B.; Larsson, C.; Ammerman, N.C.; Andrade, B.B.; Orandle, M.; Bock, K.W.; Xu, Z.; Bagci, U.; Mollura, D.J.; et al. Mycobacterium tuberculosisdysregulates MMP/TIMP balance to drive rapid cavitation and unrestrained bacterial proliferation. J. Pathol. 2015, 235, 431–444. [Google Scholar] [CrossRef]
- Vignesh, R.; Kumarasamy, N.; Lim, A.; Solomon, S.; Murugavel, K.G.; Balakrishnan, P.; Solomon, S.S.; Mayer, K.H.; Swathirajan, C.R.; Chandrasekaran, E.; et al. TB-IRIS After Initiation of Antiretroviral Therapy Is Associated With Expansion of Preexistent Th1 Responses Against Mycobacterium tuberculosis Antigens. JAIDS J. Acquir. Immune Defic. Syndr. 2013, 64, 241–248. [Google Scholar] [CrossRef]
- Haddow, L.J.; Dibben, O.; Moosa, M.-Y.S.; Borrow, P.; Easterbrook, P. Circulating inflammatory biomarkers can predict and characterize tuberculosis-associated immune reconstitution inflammatory syndrome. AIDS 2011, 25, 1163–1174. [Google Scholar] [CrossRef]
- Lawn, S.D.; Myer, L.; Edwards, D.; Bekker, L.-G.; Wood, R. Short-term and long-term risk of tuberculosis associated with CD4 cell recovery during antiretroviral therapy in South Africa. AIDS 2009, 23, 1717–1725. [Google Scholar] [CrossRef]
- Lapadula, G.; Soria, A.; Bandera, A.; Squillace, N.; Sabbatini, F.; Franzetti, F.; Migliori, G.B.; Gori, A. Unmasking tuberculosis in the era of antiretroviral treatment. Eur. Respir. J. 2012, 39, 1064–1075. [Google Scholar] [CrossRef] [PubMed]
- Gopal, R.; Rapaka, R.R.; Kolls, J.K. Immune reconstitution inflammatory syndrome associated with pulmonary pathogens. Eur. Respir. Rev. 2017, 26, 160042. [Google Scholar] [CrossRef] [PubMed]
- Balkhair, A.; Ahamed, S.; Sankhla, D. Unmasking Immune Reconstitution Inflammatory Syndrome (IRIS): A report of five cases and review of the literature. Sultan. Qaboos Univ. Med. J. 2011, 11, 95–103. [Google Scholar]
- Zolopa, A.R.; Andersen, J.; Komarow, L.; Sanne, I.; Sanchez, A.; Hogg, E.; Suckow, C.; Powderly, W.; ACTG A5164 Study Team. Early Antiretroviral Therapy Reduces AIDS Progression/Death in Individuals with Acute Opportunistic Infections: A Multicenter Randomized Strategy Trial. PLoS ONE 2009, 4, e5575. [Google Scholar] [CrossRef] [PubMed]
- Breton, G.; Duval, X.; Estellat, C.; Poaletti, X.; Bonnet, D.; Mvondo, D.M.; Longuet, P.; Leport, C.; Vildé, J. Determinants of Immune Reconstitution Inflammatory Syndrome in HIV Type 1–Infected Patients with Tuberculosis after Initiation of Antiretroviral Therapy. Clin. Infect. Dis. 2004, 39, 1709–1712. [Google Scholar] [CrossRef]
- Narendran, G.; Jyotheeswaran, K.; Senguttuvan, T.; Vinhaes, C.L.; Santhanakrishnan, R.K.; Manoharan, T.; Selvaraj, A.; Chandrasekaran, P.; Menon, P.A.; Bhavani, K.P.; et al. Characteristics of paradoxical tuberculosis-associated immune reconstitution inflammatory syndrome and its influence on tuberculosis treatment outcomes in persons living with HIV. Int. J. Infect. Dis. 2020, 98, 261–267. [Google Scholar] [CrossRef]
- Namale, P.E.; Abdullahi, L.H.; Fine, S.; Kamkuemah, M.; Wilkinson, R.J.; Meintjes, G. Paradoxical TB-IRIS in HIV-infected adults: A systematic review and meta-analysis. Future Microbiol. 2015, 10, 1077–1099. [Google Scholar] [CrossRef]
- Vinhaes, C.L.; Sheikh, V.; de-Souza, D.O.; Wang, J.; Rupert, A.; Roby, G.; Arriaga, M.B.; Fukutani, K.F.; Sawe, F.; Shaffer, D.; et al. An inflammatory composite score predicts mycobacterial IRIS in people with HIV and severe lymphopenia: A prospective international cohort study. J. Infect. Dis. 2020. [Google Scholar] [CrossRef]
- Lichtner, M.; Cicconi, P.; Vita, S.; Cozzi-Lepri, A.; Galli, M.; Caputo, S.L.; Saracino, A.; De Luca, A.; Moioli, M.; Maggiolo, F.; et al. Cytomegalovirus Coinfection Is Associated With an Increased Risk of Severe Non–AIDS-Defining Events in a Large Cohort of HIV-Infected Patients. J. Infect. Dis. 2015, 211, 178–186. [Google Scholar] [CrossRef]
- Lee, S.A.; Sinclair, E.; Hatano, H.; Hsue, P.Y.; Epling, L.; Hecht, F.M.; Bangsberg, D.R.; Martin, J.N.; McCune, J.M.; Deeks, S.G.; et al. Impact of HIV on CD8+ T Cell CD57 Expression Is Distinct from That of CMV and Aging. PLoS ONE 2014, 9, e89444. [Google Scholar] [CrossRef]
- Freeman, M.L.; Mudd, J.C.; Shive, C.L.; Younes, S.-A.; Panigrahi, S.; Sieg, S.F.; Lee, S.A.; Hunt, P.W.; Calabrese, L.H.; Gianella, S.; et al. CD8 T-Cell Expansion and Inflammation Linked to CMV Coinfection in ART-treated HIV Infection. Clin. Infect. Dis. 2016, 62, 392–396. [Google Scholar] [CrossRef] [PubMed]
- Gianella, S.; Strain, M.C.; Rought, S.E.; Vargas, M.V.; Little, S.J.; Richman, D.D.; Spina, C.A.; Smith, D.M. Associations between Virologic and Immunologic Dynamics in Blood and in the Male Genital Tract. J. Virol. 2012, 86, 1307–1315. [Google Scholar] [CrossRef]
- Stein, D.S. Cytomegalovirus retinitis after initiation of highly active antiretroviral therapy. Lancet 1997, 350, 589–590. [Google Scholar] [CrossRef]
- Crum, N.F.; Blade, K.A. Cytomegalovirus retinitis after immune reconstitution. AIDS Read. 2005, 15, 186–188. [Google Scholar] [PubMed]
- Ruiz-Cruz, M.; La Barrera, C.A.-D.; Ablanedo-Terrazas, Y.; Reyes-Teran, G. Proposed Clinical Case Definition for Cytomegalovirus-Immune Recovery Retinitis. Clin. Infect. Dis. 2014, 59, 298–303. [Google Scholar] [CrossRef] [PubMed]
- Río, S.P.-D.; Rodriguez-Hernandez, A.; Anguiano-Landa, L.; Aguilar-Portillo, G.; Zavala-Trujillo, I.; Nava-Zavala, A.H.; Zavala-Cerna, M.G. Immune Reconstitution Inflammatory Syndrome and Cytomegalovirus Pneumonia Case Report: Highlights and Missing Links in Classification Criteria and Standardized Treatment. Case Rep. Infect. Dis. 2017, 2017, 9314580. [Google Scholar] [CrossRef]
- Panel on Opportunistic Infections in Adults and Adolescents with HIV. Guidelines for the Prevention and Treatment of Opportunistic Infections in Adults and Adolescents with HIV; Centers for Disease Control and Prevention tNIoH and the HIV Medicine Association of the Infectious Diseases Society of America: New York, NY, USA, 2020.
- Masiá, M.; Robledano, C.; De La Tabla, V.O.; Antequera, P.; Lumbreras, B.; Hernández, I.; Gutiérrez, F. Coinfection with Human Herpesvirus 8 Is Associated with Persistent Inflammation and Immune Activation in Virologically Suppressed HIV-Infected Patients. PLoS ONE 2014, 9, e105442. [Google Scholar] [CrossRef]
- WHO. Hepatitis B. Available online: https://www.who.int/news-room/fact-sheets/detail/hepatitis-b (accessed on 21 December 2020).
- Wilkinson, R.N.; Noeypatimanondh, S.; Gould, D.J. Infectivity of falciparum malaria patients for anopheline mosquitoes before and after chloroquine treatment. Trans. R. Soc. Trop. Med. Hyg. 1976, 70, 306–307. [Google Scholar] [CrossRef]
- Vinhaes, C.L.; Cruz, L.A.B.; Menezes, R.C.; Carmo, T.A.; Arriaga, M.B.; Queiroz, A.T.L.; Barral-Netto, M.; Andrade, B.B. Chronic Hepatitis B Infection Is Associated with Increased Molecular Degree of Inflammatory Perturbation in Peripheral Blood. Viruses 2020, 12, 864. [Google Scholar] [CrossRef]
- Andrade, B.B.; Hullsiek, K.H.; Boulware, D.R.; Rupert, A.; French, M.A.; Ruxrungtham, K.; Montes, M.L.; Price, H.; Barreiro, P.; Audsley, J.; et al. Biomarkers of Inflammation and Coagulation Are Associated with Mortality and Hepatitis Flares in Persons Coinfected With HIV and Hepatitis Viruses. J. Infect. Dis. 2013, 207, 1379–1388. [Google Scholar] [CrossRef]
- Crane, M.; Oliver, B.; Matthews, G.; Avihingsanon, A.; Ubolyam, S.; Markovska, V.; Chang, J.J.; Dore, G.J.; Price, P.; Visvanathan, K.; et al. Immunopathogenesis of Hepatic Flare in HIV/Hepatitis B Virus (HBV)–Coinfected Individuals after the Initiation of HBV-Active Antiretroviral Therapy. J. Infect. Dis. 2009, 199, 974–981. [Google Scholar] [CrossRef] [PubMed]
- Guidotti, L.G.; Ishikawa, T.; Hobbs, M.V.; Matzke, B.; Schreiber, R.; Chisari, F.V. Intracellular Inactivation of the Hepatitis B Virus by Cytotoxic T Lymphocytes. Immunity 1996, 4, 25–36. [Google Scholar] [CrossRef]
- Sherman, K.E.; Rouster, S.D.; Chung, R.T.; Rajicic, N. Hepatitis C Virus Prevalence among Patients Infected with Human Immunodeficiency Virus: A Cross-Sectional Analysis of the US Adult AIDS Clinical Trials Group. Clin. Infect. Dis. 2002, 34, 831–837. [Google Scholar] [CrossRef] [PubMed]
- Staples, C.T., Jr.; Rimland, D.; Dudas, D. Hepatitis C in the HIV (Human Immunodeficiency Virus) Atlanta V.A. (Veterans Affairs Medical Center) Cohort Study (HAVACS): The Effect of Coinfection on Survival. Clin. Infect. Dis. 1999, 29, 150–154. [Google Scholar] [CrossRef] [PubMed]
- Chen, J.Y.; Feeney, E.R.; Chung, R.T. HCV and HIV co-infection: Mechanisms and management. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 362–371. [Google Scholar] [CrossRef] [PubMed]
- Benhamou, Y.; Bochet, M.; Di Martino, V.; Charlotte, F.; Azria, F.; Coutellier, A.; Vidaud, M.; Opolon, P.; Katlama, C.; Poynard, T.; et al. Liver fibrosis progression in human immunodeficiency virus and hepatitis C virus coinfected patients. Hepatology 1999, 30, 1054–1058. [Google Scholar] [CrossRef] [PubMed]
- Di Martino, V.; Rufat, P.; Boyer, N.; Renard, P.; Degos, F.; Martinot-Peignoux, M.; Matheron, S.; Le Moing, V.; Vachon, F.; Degott, C.; et al. The influence of human immunodeficiency virus coinfection on chronic hepatitis C in injection drug users: A long-term retrospective cohort study. Hepatology 2001, 34, 1193–1199. [Google Scholar] [CrossRef]
- Thein, H.H.; Yi, Q.; Dore, G.J.; Krahn, M.D. Natural history of hepatitis C virus infection in HIV-infected individuals and the impact of HIV in the era of highly active antiretroviral therapy: A meta-analysis. AIDS 2008, 22, 1979–1991. [Google Scholar] [CrossRef]
- Soriano, V.; Berenguer, J. Extrahepatic comorbidities associated with hepatitis C virus in HIV-infected patients. Curr. Opin. HIV AIDS 2015, 10, 309–315. [Google Scholar] [CrossRef]
- Fernández-Montero, J.V.; Barreiro, P.; De Mendoza, C.; Labarga, P.; Soriano, V. Hepatitis C virus coinfection independently increases the risk of cardiovascular disease in HIV-positive patients. J. Viral Hepat. 2015, 23, 47–52. [Google Scholar] [CrossRef]
- Hodowanec, A.C.; Lee, R.D.; Brady, K.E.; Gao, W.; Kincaid, S.; Plants, J.; Bahk, M.; Mackman, N.; Landay, A.L.; Huhn, G.D. A matched cross-sectional study of the association between circulating tissue factor activity, immune activation and advanced liver fibrosis in hepatitis C infection. BMC Infect. Dis. 2015, 15, 190. [Google Scholar] [CrossRef] [PubMed]
- Márquez, M.; Romero-Cores, P.; Montes-Oca, M.; Martín-Aspas, A.; Soto-Cardenas, M.-J.; Guerrero, F.; Fernández-Gutierrez, C.; Girón-González, J.A. Immune Activation Response in Chronic HIV-Infected Patients: Influence of Hepatitis C Virus Coinfection. PLoS ONE 2015, 10, e0119568. [Google Scholar] [CrossRef] [PubMed]
- Kushner, L.E.; Wendelboe, A.M.; Lazzeroni, L.C.; Chary, A.; Winters, M.A.; Osinusi, A.; Kottilil, S.; Polis, M.A.; Holodniy, M. Immune Biomarker Differences and Changes Comparing HCV Mono-Infected, HIV/HCV Co-Infected, and HCV Spontaneously Cleared Patients. PLoS ONE 2013, 8, e60387. [Google Scholar] [CrossRef] [PubMed]
- Kalinoski, T.; Malenfant, J.; Yim, C.; Jeng, A. Case Report: A Case of Severe Cryptococcal Immune Reconstitution Inflammatory Syndrome Presenting with Brain and Intradural Abscesses in an HIV Patient. Am. J. Trop. Med. Hyg. 2020, 103, 713–718. [Google Scholar] [CrossRef]
- Boulware, D.R.; Meya, D.B.; Bergemann, T.L.; Wiesner, D.L.; Rhein, J.; Musubire, A.; Lee, S.J.; Kambugu, A.; Janoff, E.N.; Bohjanen, P.R. Clinical Features and Serum Biomarkers in HIV Immune Reconstitution Inflammatory Syndrome after Cryptococcal Meningitis: A Prospective Cohort Study. PLoS Med. 2010, 7, e1000384. [Google Scholar] [CrossRef]
- Boulware, D.R.; Bonham, S.C.; Meya, D.B.; Wiesner, D.L.; Park, G.S.; Kambugu, A.; Janoff, E.N.; Bohjanen, P.R. Paucity of Initial Cerebrospinal Fluid Inflammation in Cryptococcal Meningitis Is Associated with Subsequent Immune Reconstitution Inflammatory Syndrome. J. Infect. Dis. 2010, 202, 962–970. [Google Scholar] [CrossRef]
- Jarvis, J.N.; Meintjes, G.; Bicanic, T.; Buffa, V.; Hogan, L.E.; Mo, S.; Tomlinson, G.S.; Kropf, P.; Noursadeghi, M.; Harrison, T.S. Cerebrospinal Fluid Cytokine Profiles Predict Risk of Early Mortality and Immune Reconstitution Inflammatory Syndrome in HIV-Associated Cryptococcal Meningitis. PLoS Pathog. 2015, 11, e1004754. [Google Scholar] [CrossRef]
- Khaw, Y.M.; Aggarwal, N.; Barclay, W.E.; Kang, E.; Inoue, M.; Shinohara, M.L. Th1-Dependent Cryptococcus-Associated Immune Reconstitution Inflammatory Syndrome Model with Brain Damage. Front. Immunol. 2020, 11, 529219. [Google Scholar] [CrossRef]
- Meya, D.B.; Okurut, S.; Zziwa, G.; Cose, S.; Boulware, D.R.; Janoff, E. HIV-Associated Cryptococcal Immune Reconstitution Inflammatory Syndrome Is Associated with Aberrant T Cell Function and Increased Cytokine Responses. J. Fungi 2019, 5, 42. [Google Scholar] [CrossRef]
- Boulware, D.R.; Meya, D.B.; Muzoora, C.; Rolfes, M.A.; Hullsiek, K.H.; Musubire, A.; Taseera, K.; Nabeta, H.W.; Schutz, C.; Williams, D.A.; et al. Timing of Antiretroviral Therapy after Diagnosis of Cryptococcal Meningitis. N. Engl. J. Med. 2014, 370, 2487–2498. [Google Scholar] [CrossRef]
- Chang, C.C.; Omarjee, S.; Lim, A.; Spelman, T.; Gosnell, B.I.; Carr, W.H.; Elliott, J.H.; Moosa, M.-Y.S.; Ndung’U, T.; A French, M.; et al. Chemokine Levels and Chemokine Receptor Expression in the Blood and the Cerebrospinal Fluid of HIV-Infected Patients with Cryptococcal Meningitis and Cryptococcosis-Associated Immune Reconstitution Inflammatory Syndrome. J. Infect. Dis. 2013, 208, 1604–1612. [Google Scholar] [CrossRef]
- Akilimali, N.A.; Chang, C.C.; Muema, D.M.; Reddy, T.; Moosa, M.-Y.S.; Lewin, S.R.; A French, M.; Ndung’U, T. Plasma But Not Cerebrospinal Fluid Interleukin 7 and Interleukin 5 Levels Pre-Antiretroviral Therapy Commencement Predict Cryptococcosis-Associated Immune Reconstitution Inflammatory Syndrome. Clin. Infect. Dis. 2017, 65, 1551–1559. [Google Scholar] [CrossRef]
- Phair, J.P.; Muñoz, A.; Detels, R.; A Kaslow, R.; Rinaldo, C.R.; Saah, A.J. The risk of Pneumocystis carinii pneumonia among men infected with human immunodeficiency virus type 1. Multicenter AIDS Cohort Study Group. N. Engl. J. Med. 1990, 322, 161–165. [Google Scholar] [CrossRef]
- Tato, L.R.; Cibrian, J.B.; Fábregas, A.C.; Mercadé, J.N.; Willekens, R.; Martin-Gomez, M.T.; Pascuet, E.R.; Ferrer, V.F. Immune reconstitution inflammatory syndrome in HIV-infected patients with Pneumocystis jirovecii pneumonia. Enferm. Infecc. Microbiol. Clín. 2018, 36, 621–626. [Google Scholar] [CrossRef] [PubMed]
- Israelski, D.M.; Chmiel, J.S.; Poggensee, L.; Phair, J.P.; Remington, J.S. Prevalence of Toxoplasma infection in a cohort of homosexual men at risk of AIDS and toxoplasmic encephalitis. JAIDS J. Acquir. Immune Defic. Syndr. 1993, 6, 414–418. [Google Scholar]
- Mathews, W.C.; Fullerton, S.C. Use of a clinical laboratory database to estimate Toxoplasma seroprevalence among human immunodeficiency virus-infected patients. Overcoming bias in secondary analysis of clinical records. Arch. Pathol. Lab. Med. 1994, 118, 807–810. [Google Scholar] [PubMed]
- Jones, J.L.; Wilson, M.; Kruszon-Moran, D.; Sanders-Lewis, K. Toxoplasma gondii Infection in the United States, 1999–2004, Decline from the Prior Decade. Am. J. Trop. Med. Hyg. 2007, 77, 405–410. [Google Scholar] [CrossRef] [PubMed]
- Luft, B.B.; Conley, F.F.; Remington, J.J.; Laverdiere, M.M.; Levine, J.J.; Strandberg, D.D.; Wagner, K.K.; Craven, P.P.; File, T.T.; Rice, N.N.; et al. Outbreak of Central-Nervous-System Toxoplasmosis in Western Europe and North America. Lancet 1983, 321, 781–784. [Google Scholar] [CrossRef]
- Wong, B.; Gold, J.W.M.; Brown, A.E.; Lange, M.; Fried, R.; Grieco, M.; Mildvan, N.; Giron, J.; Tapper, M.L.; Lerner, C.W.; et al. Central-Nervous-System Toxoplasmosis in Homosexual Men and Parenteral Drug Abusers. Ann. Intern. Med. 1984, 100, 36–42. [Google Scholar] [CrossRef] [PubMed]
- Leport, C.; Ch⩽ne, G.; Morlat, P.; Luft, B.J.; Rousseau, F.; Pueyo, S.; Hafner, R.; Miro, J.M.; Aubertin, J.; Salamon, R.; et al. Pyrimethamine for Primary Prophylaxis of Toxoplasmic Encephalitis in Patients with Human Immunodeficiency Virus Infection: A Double-Blind, Randomized Trial. ANRS 005-ACTG 154 Group MembersJ. Agence Nationale de Recherche sur le SIDA. AIDS Clinical Trial Group. J. Infect. Dis. 1996, 173, 91–97. [Google Scholar] [CrossRef]
- Luft, B.J.; Brooks, R.G.; Conley, F.K.; McCabe, R.E.; Remington, J.S. Toxoplasmic Encephalitis in Patients with Acquired Immune Deficiency Syndrome. JAMA 1984, 252, 913–917. [Google Scholar] [CrossRef] [PubMed]
- Balanza, N.; Erice, C.; Ngai, M.; Varo, R.; Kain, K.C.; Bassat, Q. Host-Based Prognostic Biomarkers to Improve Risk Stratification and Outcome of Febrile Children in Low- and Middle-Income Countries. Front. Pediatr. 2020, 8, 552083. [Google Scholar] [CrossRef] [PubMed]
- Lin, Q.; Wang, Y.; Luo, Y.; Tang, G.; Li, S.; Zhang, Y.; Mao, L.; Liu, W.; Wang, F.; Sun, Z. The Effect of Host Immunity on Predicting the Mortality of Carbapenem-Resistant Organism Infection. Front. Cell. Infect. Microbiol. 2020, 10, 480. [Google Scholar] [CrossRef] [PubMed]
- Silveira-Mattos, P.S.; Barreto-Duarte, B.; Vasconcelos, B.; Fukutani, K.F.; Vinhaes, C.L.; Oliveira-De-Souza, D.; Ibegbu, C.C.; Figueiredo, M.C.; Sterling, T.R.; Rengarajan, J.; et al. Differential Expression of Activation Markers by Mycobacterium tuberculosis-specific CD4+ T Cell Distinguishes Extrapulmonary From Pulmonary Tuberculosis and Latent Infection. Clin. Infect. Dis. 2019, 71, 1905–1911. [Google Scholar] [CrossRef]
- Fernandes, C.D.; Arriaga, M.B.; Costa, M.C.M.; Costa, M.C.M.; Costa, M.H.M.; Vinhaes, C.L.; Silveira-Mattos, P.S.; Fukutani, K.F.; Andrade, B.B. Host Inflammatory Biomarkers of Disease Severity in Pediatric Community-Acquired Pneumonia: A Systematic Review and Meta-analysis. Open Forum Infect. Dis. 2019, 6, ofz520. [Google Scholar] [CrossRef]
- Manabe, Y.C.; Andrade, B.B.; Gupte, N.; Leong, S.; Kintali, M.; Matoga, M.; Riviere, C.; Sameneka, W.; Lama, J.R.; Naidoo, K.; et al. A Parsimonious Host Inflammatory Biomarker Signature Predicts Incident TB and Mortality in Advanced HIV. Clin. Infect. Dis. 2019. [Google Scholar] [CrossRef]
- Narendran, G.; Kavitha, D.; Karunaianantham, R.; Gil-Santana, L.; Almeida-Junior, J.L.; Reddy, S.D.; Kumar, M.M.; Hemalatha, H.; Jayanthi, N.N.; Ravichandran, N.; et al. Role of LTA4H Polymorphism in Tuberculosis-Associated Immune Reconstitution Inflammatory Syndrome Occurrence and Clinical Severity in Patients Infected with HIV. PLoS ONE 2016, 11, e0163298. [Google Scholar] [CrossRef]
- De Sá, N.B.R.; Ribeiro-Alves, M.; Da Silva, T.P.; Pilotto, J.H.; Rolla, V.C.; Giacoia-Gripp, C.B.W.; Scott-Algara, D.; Morgado, M.G.; Teixeira, S.L.M. Clinical and genetic markers associated with tuberculosis, HIV-1 infection, and TB/HIV-immune reconstitution inflammatory syndrome outcomes. BMC Infect. Dis. 2020, 20, 591. [Google Scholar] [CrossRef]
- Price, P.; Morahan, G.; Huang, D.; Stone, E.; Cheong, K.Y.M.; Castley, A.; Rodgers, M.; McIntyre, M.Q.; Abraham, L.J.; French, M.A. Polymorphisms in cytokine genes define subpopulations of HIV-1 patients who experienced immune restoration diseases. AIDS 2002, 16, 2043–2047. [Google Scholar] [CrossRef]
- Vlasova-St Louis, I.; Chang, C.C.; Shahid, S.; A French, M.; Bohjanen, P.R. Transcriptomic Predictors of Paradoxical Cryptococcosis-Associated Immune Reconstitution Inflammatory Syndrome. Open Forum Infect. Dis. 2018, 5, 157. [Google Scholar] [CrossRef]
- Silva, C.A.; Graham, B.; Webb, K.; Ashton, L.V.; Harton, M.; Luetkemeyer, A.F.; Bokatzian, S.; Almubarak, R.; Mahapatra, S.; Hovind, L.; et al. A pilot metabolomics study of tuberculosis immune reconstitution inflammatory syndrome. Int. J. Infect. Dis. 2019, 84, 30–38. [Google Scholar] [CrossRef] [PubMed]
- Oliver, B.G.; Elliott, J.H.; Price, P.; Phillips, M.; Saphonn, V.; Vun, M.C.; Kaldor, J.M.; Cooper, D.A.; French, M.A. Mediators of Innate and Adaptive Immune Responses Differentially Affect Immune Restoration Disease Associated withMycobacterium tuberculosisin HIV Patients Beginning Antiretroviral Therapy. J. Infect. Dis. 2010, 202, 1728–1737. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Tan, H.Y.; Yong, Y.K.; Andrade, B.B.; Shankar, E.M.; Ponnampalavanar, S.; Omar, S.F.; Narendran, G.; Kamarulzaman, A.; Swaminathan, S.; Sereti, I.; et al. Plasma interleukin-18 levels are a biomarker of innate immune responses that predict and characterize tuberculosis-associated immune reconstitution inflammatory syndrome. AIDS 2015, 29, 421–431. [Google Scholar] [CrossRef] [PubMed]
- Musselwhite, L.W.; Andrade, B.B.; Ellenberg, S.S.; Tierney, A.; Belaunzaran-Zamudio, P.F.; Rupert, A.; Lederman, M.M.; Sanne, I.; Madero, J.G.S.; Sereti, I. Vitamin D, d -dimer, Interferon γ, and sCD14 Levels are Independently Associated with Immune Reconstitution Inflammatory Syndrome: A Prospective, International Study. EBioMedicine 2016, 4, 115–123. [Google Scholar] [CrossRef] [PubMed]
- Mizukawa, Y.; Kimishima, M.; Aoyama, Y.; Shiohara, T. Predictive biomarkers for cytomegalovirus reactivation before and after immunosuppressive therapy: A single-institution retrospective long-term analysis of patients with drug-induced hypersensitivity syndrome (DiHS)/drug reaction with eosinophilia and systemic syndrome (DRESS). Int. J. Infect. Dis. 2020, 100, 239–246. [Google Scholar] [CrossRef]
- Murthy, A.R.; Marulappa, R.; Hegde, U.; Kappadi, D.; Ambikathanaya, U.K.; Nair, P. Treatment Guidelines and Prognosis of Immune Reconstitution Inflammatory Syndrome Patients: A Review. J. Int. Oral Health 2015, 7, 92–95. [Google Scholar] [PubMed]
- Meintjes, G.; Wilkinson, R.J.; Morroni, C.; Pepper, D.J.; Rebe, K.; Rangaka, M.X.; Oni, T.; Maartens, G. Randomized placebo-controlled trial of prednisone for paradoxical tuberculosis-associated immune reconstitution inflammatory syndrome. AIDS 2010, 24, 2381–2390. [Google Scholar] [CrossRef]
- Stek, C.; Allwood, B.; Du Bruyn, E.; Buyze, J.; Schutz, C.; Thienemann, F.; Lombard, A.; Wilkinson, R.J.; Meintjes, G.; Lynen, L. The effect of HIV-associated tuberculosis, tuberculosis-IRIS and prednisone on lung function. Eur. Respir. J. 2020, 55, 1901692. [Google Scholar] [CrossRef]
- Beardsley, J.; Wolbers, M.; Kibengo, F.M.; Ggayi, A.-B.M.; Kamali, A.; Cuc, N.T.K.; Binh, T.Q.; Chau, N.V.V.; Farrar, J.; Merson, L.; et al. Adjunctive Dexamethasone in HIV-Associated Cryptococcal Meningitis. N. Engl. J. Med. 2016, 374, 542–554. [Google Scholar] [CrossRef]
- Musubire, A.K.; Meya, B.D.; Mayanja-Kizza, H.; Lukande, R.; Wiesner, L.D.; Bohjanen, P.R.; Boulware, D.R. Challenges in diagnosis and management of Cryptococcal immune reconstitution inflammatory syndrome (IRIS) in resource limited settings. Afr. Health Sci. 2012, 12, 226–230. [Google Scholar] [CrossRef]
- Dooley, K.E.; Kaplan, R.; Mwelase, N.; Grinsztejn, B.; Ticona, E.; Lacerda, M.; Sued, O.; Belonosova, E.; Ait-Khaled, M.; Angelis, K.; et al. Dolutegravir-based Antiretroviral Therapy for Patients Coinfected With Tuberculosis and Human Immunodeficiency Virus: A Multicenter, Noncomparative, Open-label, Randomized Trial. Clin. Infect. Dis. 2020, 70, 549–556. [Google Scholar] [CrossRef] [PubMed]
- Sierra-Madero, J.G.; Ellenberg, S.S.; Rassool, M.S.; Tierney, A.; Belaunzarán-Zamudio, P.F.; López-Martínez, A.; Piñeirúa-Menéndez, A.; Montaner, L.J.; Azzoni, L.; Benítez, C.R.; et al. A Randomized, Double-Blind, Placebo-Controlled Clinical Trial of a Chemokine Receptor 5 (CCR5) Antagonist to Decrease the Occurrence of Immune Reconstitution Inflammatory Syndrome in HIV-Infection: The CADIRIS Study. Lancet HIV 2014, 1, e60–e67. [Google Scholar] [CrossRef]
- Hachisu, Y.; Koga, Y.; Kasama, S.; Kaira, K.; Yatomi, M.; Aoki-Saito, H.; Tsurumaki, H.; Kamide, Y.; Sunaga, N.; Maeno, T.; et al. Treatment with Tumor Necrosis Factor-α Inhibitors, History of Allergy, and Hypercalcemia Are Risk Factors of Immune Reconstitution Inflammatory Syndrome in HIV-Negative Pulmonary Tuberculosis Patients. J. Clin. Med. 2019, 9, 96. [Google Scholar] [CrossRef] [PubMed]
- Keeley, A.J.; Parkash, V.; Tunbridge, A.; Greig, J.; Collini, P.J.; McKane, W.; Tattersall, R.S. Anakinra in the treatment of protracted paradoxical inflammatory reactions in HIV-associated tuberculosis in the United Kingdom: A report of two cases. Int. J. STD AIDS 2020, 31, 808–812. [Google Scholar] [CrossRef] [PubMed]
- Jain, S.; Bajgai, P.; Tigari, B.; Sharma, K.; Sharma, A.; Gupta, V.; Singh, R. Bevacizumab for paradoxical worsening treatment adjunct in HIV patient with choroidal tuberculoma. J. Ophthalmic Inflamm. Infect. 2016, 6, 421. [Google Scholar] [CrossRef]
- Lwin, N.; Boyle, M.; Davis, J.S. Adalimumab for Corticosteroid and Infliximab-Resistant Immune Reconstitution Inflammatory Syndrome in the Setting of TB/HIV Coinfection. Open Forum Infect. Dis. 2018, 5, ofy027. [Google Scholar] [CrossRef]
- Hsu, D.C.; Faldetta, K.F.; Pei, L.; Sheikh, V.; Utay, N.S.; Roby, G.; Rupert, A.; Fauci, A.S.; Sereti, I. A Paradoxical Treatment for a Paradoxical Condition: Infliximab Use in Three Cases of Mycobacterial IRIS. Clin. Infect. Dis. 2016, 62, 258–261. [Google Scholar] [CrossRef]
- Brunel, A.-S.; Reynes, J.; Tuaillon, E.; Rubbo, P.-A.; Lortholary, O.; Montes, B.; Le Moing, V.; Makinson, A. Thalidomide for steroid-dependent immune reconstitution inflammatory syndromes during AIDS. AIDS 2012, 26, 2110–2112. [Google Scholar] [CrossRef]
- Lortholary, O.; Fontanet, A.; Mémain, N.; Martin, A.; Sitbon, K.; Dromer, F. rench Cryptococcosis Study Group incidence and risk factors of immune reconstitution inflammatory syndrome complicating HIV-associated cryptococcosis in France. AIDS 2005, 19, 1043–1049. [Google Scholar] [CrossRef]
- Rowley, M.W.; Patel, A.; Zhou, W.; Wong, M.; Seetharam, A.B. Immune Reconstitution Syndrome with Initiation of Treatment of HBV/HIV Co-infection: Activity Flare associated with E antigen Seroconversion. Ann. Hepatol. 2019, 18, 220–224. [Google Scholar] [CrossRef]
- Mitsumoto, F.; Murata, M.; Kato, Y.; Ura, K.; Takayama, K.; Hiramine, S.; Ikezaki, H.; Shimizu, M.; Toyoda, K.; Ogawa, E.; et al. Hepatitis B Virus-related Immune Reconstitution Inflammatory Syndrome in Two Patients Coinfected with Human Immunodeficiency Virus Diagnosed with a Liver Biopsy. Intern. Med. 2014, 53, 2165–2170. [Google Scholar] [CrossRef] [PubMed][Green Version]
- Hill, A.M.; Mitchell, N.; Hughes, S.; Pozniak, A.L. Risks of cardiovascular or central nervous system adverse events and immune reconstitution inflammatory syndrome, for dolutegravir versus other antiretrovirals: Meta-analysis of randomized trials. Curr. Opin. HIV AIDS 2018, 13, 102–111. [Google Scholar] [CrossRef] [PubMed]
- European Medicine Agency. Tivicay, Summary of Product Characteristics. Available online: https://www.ema.europa.eu/en/documents/product-information/tivicay-epar-product-information_en.pdf (accessed on 15 January 2021).
- Dorr, P.; Westby, M.; Dobbs, S.; Griffin, P.; Irvine, B.; Macartney, M.; Mori, J.; Rickett, G.; Smith-Burchnell, C.; Napier, C.; et al. Maraviroc (UK-427,857), a Potent, Orally Bioavailable, and Selective Small-Molecule Inhibitor of Chemokine Receptor CCR5 with Broad-Spectrum Anti-Human Immunodeficiency Virus Type 1 Activity. Antimicrob. Agents Chemother. 2005, 49, 4721–4732. [Google Scholar] [CrossRef] [PubMed]
- Mertens, M.; Singh, J.A. Anakinra for Rheumatoid Arthritis: A Systematic Review. J. Rheumatol. 2009, 36, 1118–1125. [Google Scholar] [CrossRef] [PubMed]
- Branisteanu, D.C.; Branisteanu, D.E.; Feraru, C.I.; Branisteanu, C.I.; Moraru, A.; Zemba, M.; Balta, F. Influence of unilateral intravitreal bevacizumab injection on the incidence of symptomatic choroidal neovascularization in the fellow eye in patients with neovascular age-related macular degeneration (Review). Exp. Ther. Med. 2020, 20, 182. [Google Scholar] [CrossRef] [PubMed]
- Papamichael, K.; Chachu, K.A.; Vajravelu, R.K.; Vaughn, B.P.; Ni, J.; Osterman, M.T.; Cheifetz, A.S. Improved Long-term Outcomes of Patients With Inflammatory Bowel Disease Receiving Proactive Compared With Reactive Monitoring of Serum Concentrations of Infliximab. Clin. Gastroenterol. Hepatol. 2017, 15, 1580–1588.e3. [Google Scholar] [CrossRef] [PubMed]
- Franks, M.E.; MacPherson, G.R.; Figg, W.D. Thalidomide. Lancet 2004, 363, 1802–1811. [Google Scholar] [CrossRef]
- Somerville, L.K.; Henderson, A.P.; Chen, S.C.; Kok, J. Successful treatment of Cryptococcus neoformans immune reconstitution inflammatory syndrome in an immunocompetent host using thalidomide. Med. Mycol. Case Rep. 2015, 7, 12–14. [Google Scholar] [CrossRef]
| Author | Year of Publication | Title of the Study | Type of IRIS/Site | Described Biomarkers | Ref |
|---|---|---|---|---|---|
| Breglio et al. | 2020 | Clinical and immunologic predictors of Mycobacterium avium complex immune reconstitution inflammatory syndrome in a contemporary cohort of patients with HIV | MAC-IRIS/Plasma | Increased expression of CD38 in CD8 + T cells High levels of alkaline phosphatase and D-dimer | [55] |
| Haddow et al. | 2011 | Circulating inflammatory biomarkers can predict and characterize tuberculosis-associated immune reconstitution inflammatory syndrome | TB-IRIS/Plasma | Reduced IL-10 and CCL2 levels Increased CRP and IFN-γ levels | [81] |
| Vinhaes et al. | 2020 | An inflammatory composite score predicts mycobacterial IRIS in people with HIV and severe lymphopenia: A prospective international cohort study. | TB-IRIS and Viral-IRIS/Plasma | A composite score including augmented levels of IL-6, IL-10, IL-27, sCD14, IFN-γ, TNF-α, Hyaluronic acid and D-dimer, and lower levels of hemoglobin | [90] |
| Andrade et al. | 2013 | Biomarkers of inflammation and coagulation are associated with mortality and hepatitis flares in persons coinfected with HIV and hepatitis viruses | HBV and HCV associated-IRIS/Plasma | D-dimer, IL-6, IL-8 and sCD14 | [104] |
| Jarvis et al. | 2015 | Cerebrospinal fluid cytokine profiles predict risk of early mortality and immune reconstitution inflammatory syndrome in HIV-associated cryptococcal meningitis. | C-IRIS/Cerebrospinal Fluid | IL-6, IFN-g, IL-4, IL-10 and IL-27 Associated with protection | [121] |
| Boulware et al. | 2014 | Timing of antiretroviral therapy after diagnosis of cryptococcal meningitis | C-IRIS/Cerebrospinal Fluid | Decreased CSF leukocyte counts reduced levels of IFN-g, IL-6, IL-8 and TNF-a | [124] |
| Akilimali et al. | 2017 | Plasma, but Not Cerebrospinal Fluid Interleukin 7 and Interleukin 5 Levels Pre-Antiretroviral Therapy Commencement Predict Cryptococcosis-Associated Immune Reconstitution Inflammatory Syndrome. | C-IRIS/Plasma and Cerebrospinal Fluid | IL-7 and IL-5 | [126] |
| Narendran et al. | 2016 | Role of LTA4H Polymorphism in Tuberculosis-Associated Immune Reconstitution Inflammatory Syndrome Occurrence and Clinical Severity in Patients Infected with HIV | TB-IRIS/DNA sample extracted from blood | LTA4H polymorphism | [141] |
| de Sá et al. | 2020 | Clinical and genetic markers associated with tuberculosis, HIV-1 infection, and TB/HIV-immune reconstitution inflammatory syndrome outcomes | TB-IRIS/DNA sample extracted from blood | Polymorphisms in HLA-B, HLA-C, and KIR2 DL3 genes | [142] |
| Price et al. | 2002 | Polymorphisms in cytokine genes define subpopulations of HIV-1 patients who experienced immune restoration diseases | Mycobacteria/Viral IRIS/DNA sample extracted from blood | TNFA-308*2 allele | [143] |
| Vlasova-St Louis et al. | 2018 | Transcriptomic Predictors of Paradoxical Cryptococcosis-Associated Immune Reconstitution Inflammatory Syndrome | C-IRIS/Plasma | Elevated expression of AIM2, BEX1 and C1QB | [144] |
| Silva et al. | 2019 | A pilot metabolomics study of tuberculosis immune reconstitution inflammatory syndrome | TB-IRIS/Plasma | Increased levels of epoxyeicosatrienoic acid, 15-deoxy-Δ-12,14-PGJ2, hydroperoxylinoleic acid and phosphatidylethanolamine. Reduced levels of phosphatidylcholine | [145] |
| Oliver et al. | 2010 | Mediators of innate and adaptive immune responses differentially affect immune restoration disease associated with Mycobacterium tuberculosis in HIV patients beginning antiretroviral therapy | TB-IRIS/Plasma | Diminished CCL2 levels | [146] |
| Tan et al. | 2015 | Plasma interleukin-18 levels are a biomarker of innate immune responses that predict and characterize tuberculosis-associated immune reconstitution inflammatory syndrome | TB-IRIS/Plasma | High IL-18 levels prior to ART | [147] |
| Musselwhite et al. | 2016 | Vitamin D, D-dimer, Interferon gamma, and sCD14 Levels are Independently Associated with Immune Reconstitution Inflammatory Syndrome: A Prospective, International Study | TB-IRIS and other-IRIS/Plasma | A composite score including CRP, sCD14, IFN-γ and hemoglobin | [148] |
| Mizukawa et al. | 2020 | Predictive biomarkers for cytomegalovirus reactivation before and after immunosuppressive therapy: A single-institution retrospective long-term analysis of patients with drug-induced hypersensitivity syndrome (DiHS)/drug reaction with eosinophilia and systemic syndrome (DRESS) | CMV- IRIS/Plasma | High baseline levels of IL-8, IL-10, IL-12p70, IL-15, G-CSF and CCL2 | [149] |
| Author | Year | Types of Study Designs | Type of IRIS | Country | Drug | Evidence | Ref |
|---|---|---|---|---|---|---|---|
| Meintjes et al. | 2010 | Randomized-controlled trial | TB-IRIS | Cape Town, South Africa | Prednisone | Prophylactic prednisone during the first 4 weeks after the initiation of ART in adult patients at high-risk for tuberculosis-associated IRIS resulted in a 30% lower incidence of tuberculosis-associated IRIS than placebo. | [151] |
| Stek et al. | 2020 | Randomized-controlled trial | TB-IRIS | Cape Town, South Africa | In severe TB-IRIS, the effect was ameliorated by treatment with prednisone, improved lung function at week 4, possibly by reducing TB-IRIS; however, the 28-day course of prednisone did not improve lung function from week 12. | [152] | |
| Dooley et al. | 2020 | Randomized-controlled trial | TB-IRIS | There were 37 sites in 7 countries (Argentina, Brazil, Mexico, Peru, Russia, South Africa, and Thailand) | Dolutegravir | Tuberculosis-associated IRIS incidence was low in the Dolutegravir arm. | [155] |
| Sierra-Madero, et al. | 2015 | Randomized controlled trial | TB-IRIS | One site in South Africa and five in Mexico | Maraviroc | Maraviroc in an initial treatment regimen does not confer meaningful protection from the occurrence of IRIS. | [156] |
| Hachisu et al. | 2019 | Cohort study | TB-IRIS | Gunma, Japan | TNF-α inhibitor | The usage of TNF-α inhibitor was significantly associated with TB-IRIS development in non-HIV patients. | [157] |
| Keeley, et al. | 2020 | Case report | TB-IRIS | Patients from Ethiopian and Zimbabwe | Anakinra | Anakinra was used to achieve control of inflammation and to reduce and stop steroids in patients at risk of death or serious morbidity (in part due to high steroid requirements) with protracted paradoxical reactions to TB. | [158] |
| Jain, et al. | 2016 | Case report | TB-IRIS | India | Bevacizumab | The patient developed IRIS in the form of increased serous fluid, and they document its resolution with intravitreal bevacizumab. | [159] |
| Lwin, et al. | 2018 | Case report | TB-IRIS | Australia | Adalimumab | The case highlights the therapeutic effect of Adalimumab on IRIS without a negative impact on immunological and virological control of HIV infection in short-term follow-up. | [160] |
| Hsu, et al. | 2016 | Case report | TB-IRIS | Cameroon, Honduras and African American | Infliximab | In 3 patients with steroid-unresponsive mycobacterial IRIS, clinical improvement was temporally associated with the administration of infliximab without obvious adverse impact on immune recovery and HIV virologic control. | [161] |
| Brunel, et al. | 2012 | Case report | Cryptococcal meningitis-IRIS | France | Thalidomide | Two HIV-infected patients with CM showed rapid clinical remission and were able to stop corticosteroid treatment following treatment with thalidomide, allowing successful IRIS resolution | [162] |
| Lortholaryet al. | 2005 | Retrospective multicenter cohort | Cryptococcal meningitis-IRIS | France | Two patients received specific therapy for IRIS: thalidomide for 4 months, with a favorable progressive return to normal lymph-node size, with dramatic improvement within 10 days, and then lower doses for a total of 8 months. | [163] | |
| . Rowley, et al. | 2019 | Case report | Hepatitis B virus–IRIS | Nigeria | HAART (emtricitabine, tenofovir and raltegravir) | Patient with hepatitis B virus-related IRIS, HAART was continued, and he was discharged on hospital day 12 after symptoms improved significantly. | [164] |
| Mitsumoto, et al. | 2014 | Case report | Hepatitis B virus–IRIS | Japan | ART: raltegravir, tenofovir disoproxil fumarate, emtricitabine | Two patients with hepatitis B virus/HIV IRIS continue ART. The alanine aminotransferase levels of both patients gradually decreased, and his HBV DNA and HIV RNA levels reduce. | [165] |
Publisher’s Note: MDPI stays neutral with regard to jurisdictional claims in published maps and institutional affiliations. |
© 2021 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vinhaes, C.L.; Araujo-Pereira, M.; Tibúrcio, R.; Cubillos-Angulo, J.M.; Demitto, F.O.; Akrami, K.M.; Andrade, B.B. Systemic Inflammation Associated with Immune Reconstitution Inflammatory Syndrome in Persons Living with HIV. Life 2021, 11, 65. https://doi.org/10.3390/life11010065
Vinhaes CL, Araujo-Pereira M, Tibúrcio R, Cubillos-Angulo JM, Demitto FO, Akrami KM, Andrade BB. Systemic Inflammation Associated with Immune Reconstitution Inflammatory Syndrome in Persons Living with HIV. Life. 2021; 11(1):65. https://doi.org/10.3390/life11010065
Chicago/Turabian StyleVinhaes, Caian L., Mariana Araujo-Pereira, Rafael Tibúrcio, Juan M. Cubillos-Angulo, Fernanda O. Demitto, Kevan M. Akrami, and Bruno B. Andrade. 2021. "Systemic Inflammation Associated with Immune Reconstitution Inflammatory Syndrome in Persons Living with HIV" Life 11, no. 1: 65. https://doi.org/10.3390/life11010065
APA StyleVinhaes, C. L., Araujo-Pereira, M., Tibúrcio, R., Cubillos-Angulo, J. M., Demitto, F. O., Akrami, K. M., & Andrade, B. B. (2021). Systemic Inflammation Associated with Immune Reconstitution Inflammatory Syndrome in Persons Living with HIV. Life, 11(1), 65. https://doi.org/10.3390/life11010065





