pH-Dependent Adsorption of Peptides on Montmorillonite for Resisting UV Irradiation
Abstract
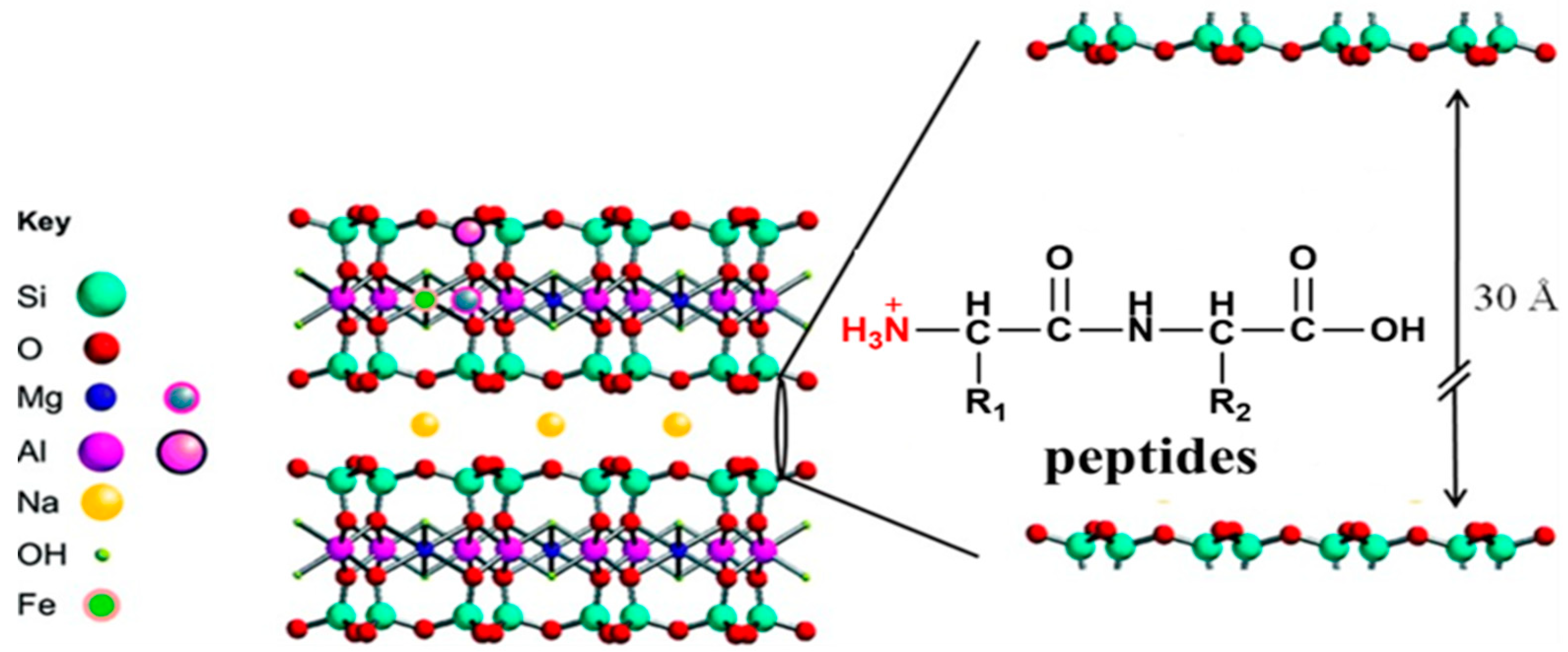
1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Sample Preparation
2.3. HPLC-MS Methods
2.4. X-Ray Diffraction (XRD) Analysis
3. Results
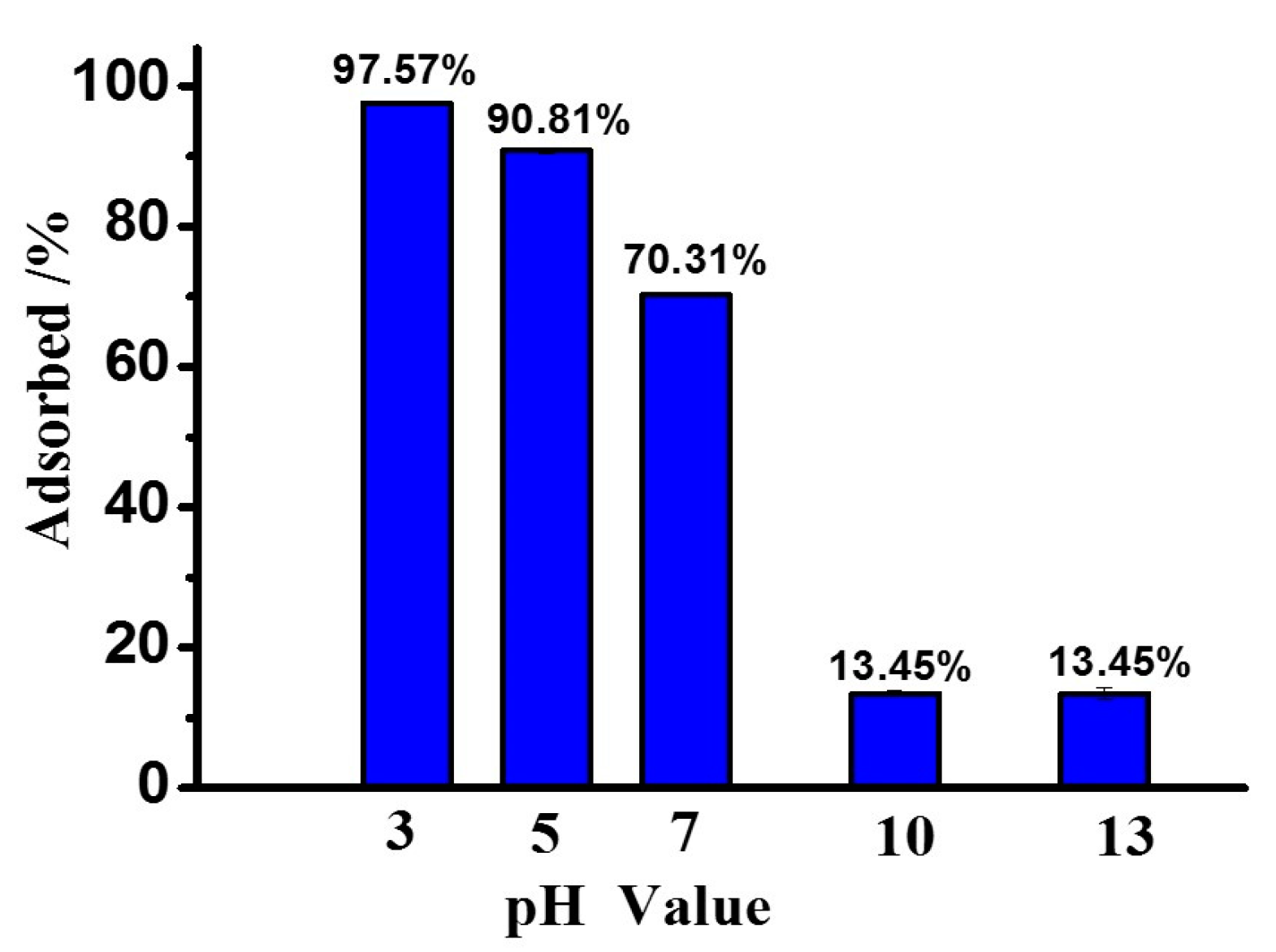
3.1. Adsorption of Peptides on MMT under Different pH Conditions
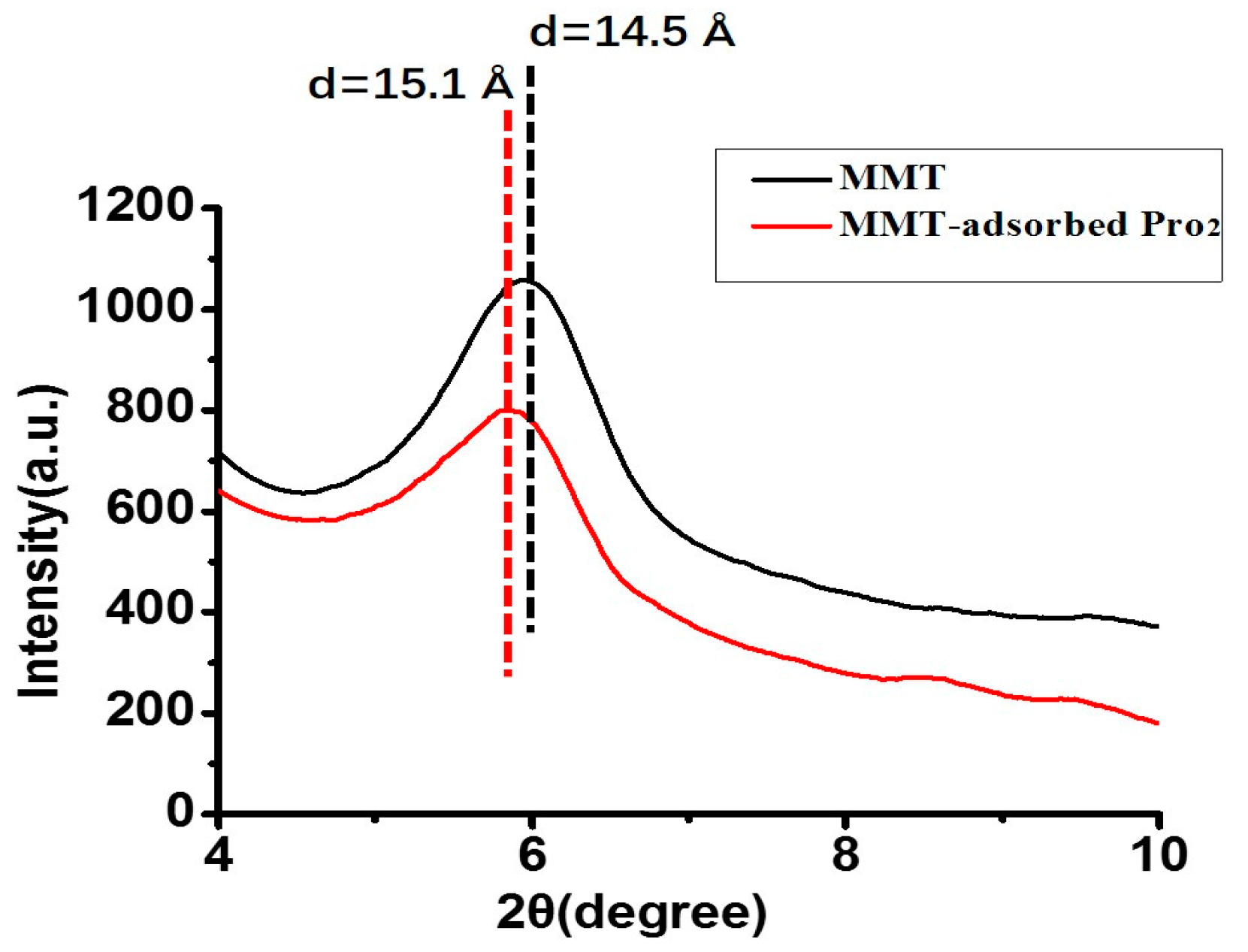
3.2. X-Ray Diffraction Analysis
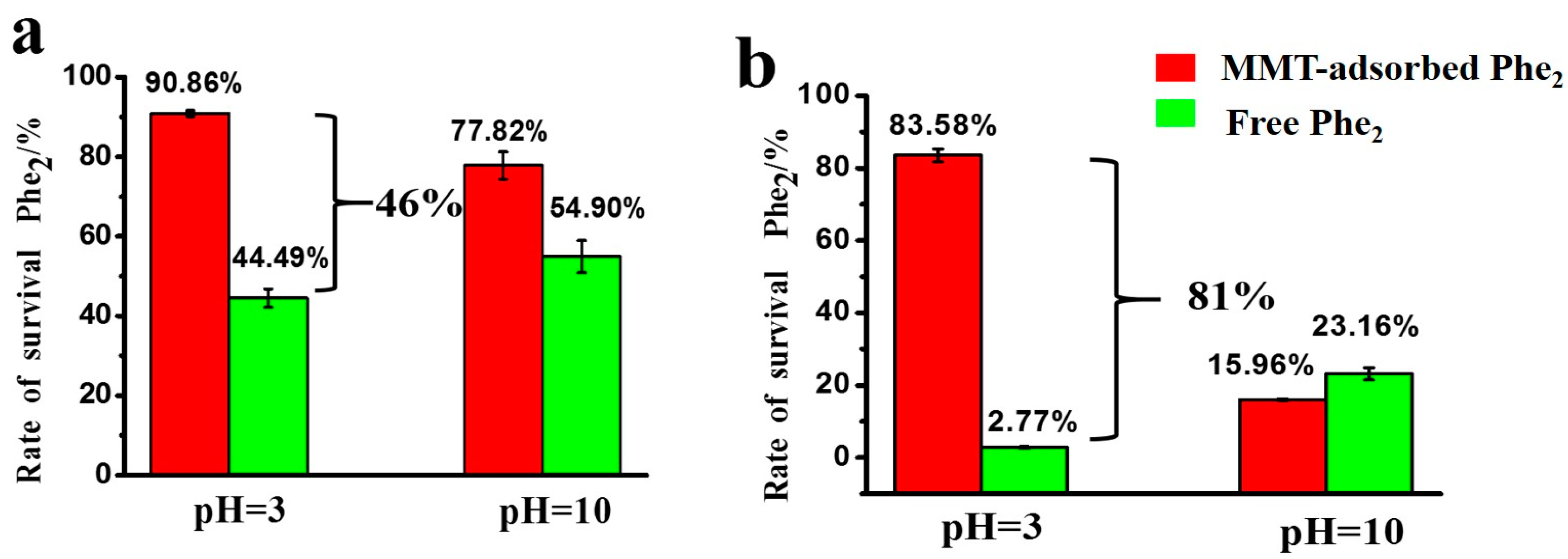
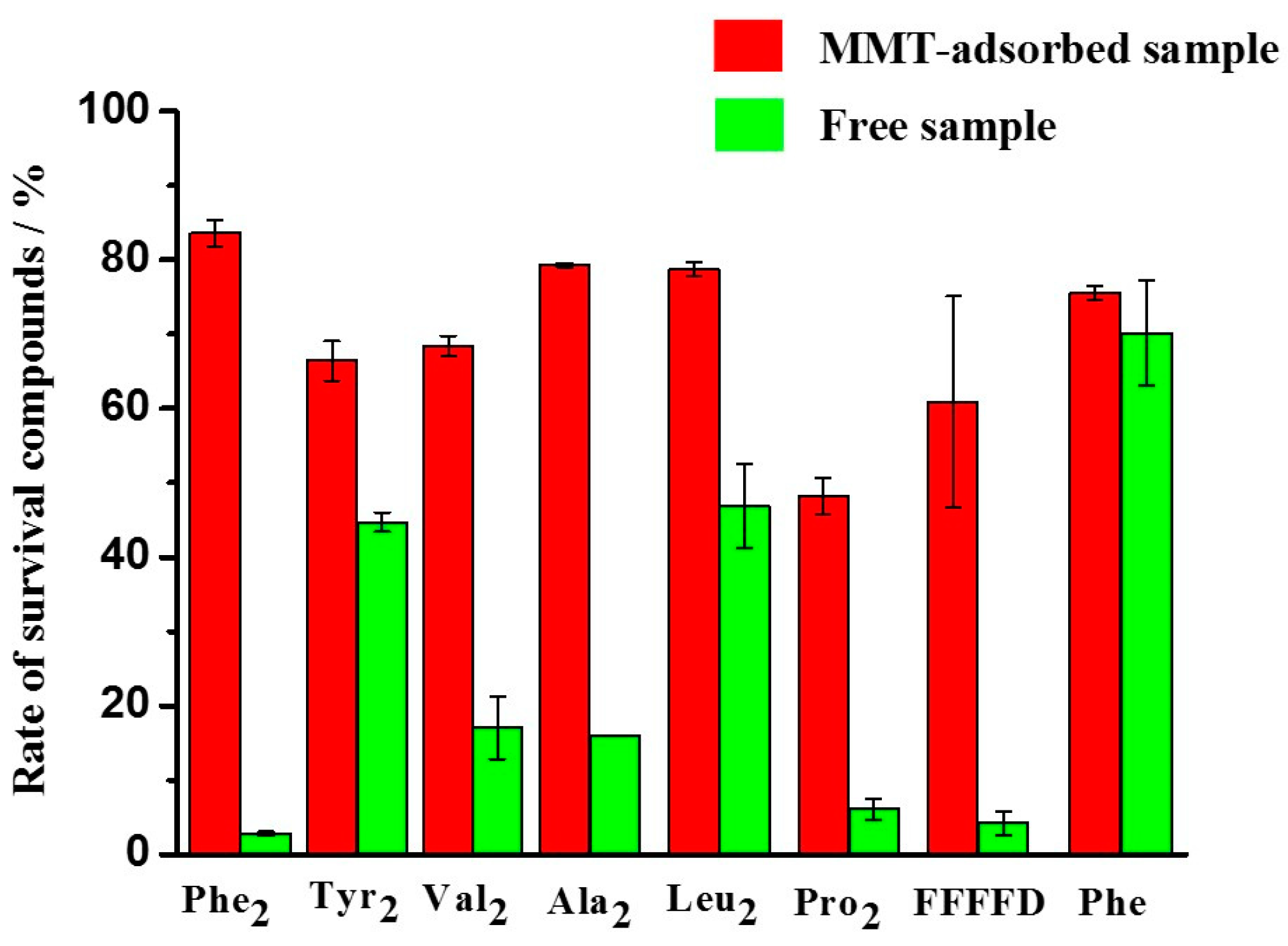
3.3. Protective Effect of MMT on Adsorbed Peptides against UV Irradiation
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Prins, L.J. Emergence of Complex Chemistry on an Organic Monolayer. Accounts Chem. Res. 2015, 48, 1920–1928. [Google Scholar] [CrossRef] [PubMed]
- Ruiz-Mirazo, K.; Briones, C.; De La Escosura, A. Prebiotic Systems Chemistry: New Perspectives for the Origins of Life. Chem. Rev. 2013, 114, 285–366. [Google Scholar] [CrossRef] [PubMed]
- Miller, S.L.; Urey, H.C. Organic compound synthesis on the primitive earth. Science 1959, 130, 245–251. [Google Scholar] [CrossRef] [PubMed]
- Kvenvolden, K.; Lawless, J.; Pering, K.; Peterson, E.; Flores, J.; Ponnamperuma, C.; Kaplan, I.R.; Moore, C. Evidence for extraterrestrial amino-acids and hydrocarbons in the Murchison meteorite. Nature 1970, 228, 923–926. [Google Scholar] [CrossRef]
- Parker, E.T.; Zhou, M.; Burton, A.S.; Glavin, D.P.; Dworkin, J.P.; Krishnamurthy, R.; Fernández, F.M.; Bada, J.L. A Plausible Simultaneous Synthesis of Amino Acids and Simple Peptides on the Primordial Earth. Angew. Chem. Int. Ed. 2014, 53, 8132–8136. [Google Scholar] [CrossRef]
- Iqubal, M.A.; Sharma, R.; Jheeta, S. Kamaluddin Thermal Condensation of Glycine and Alanine on Metal Ferrite Surface: Primitive Peptide Bond Formation Scenario. Life 2017, 7, 15. [Google Scholar] [CrossRef]
- Martin, R.B. Free energies and equilibria of peptide bond hydrolysis and formation. Biopolymers 1998, 45, 351–353. [Google Scholar] [CrossRef]
- Lahav, N.; White, D.; Chang, S. Peptide formation in the prebiotic era: Thermal condensation of glycine in fluctuating clay environments. Science 1978, 201, 67–69. [Google Scholar] [CrossRef]
- Rode, B.M. Peptide and the origin of life. Peptides 1999, 20, 773–786. [Google Scholar] [CrossRef]
- Imai, E.; Honda, H.; Hatori, K.; Brack, A.; Matsuno, K. Elongation of oligopeptides in a simulated submarine hydrothermal system. Science 1999, 283, 831–833. [Google Scholar] [CrossRef]
- Schwendinger, M.G.; Rode, B.M. Investigations on the mechanism of the salt-induced peptide formation. Orig. life Evol. Biosphere 1992, 22, 349–359. [Google Scholar] [CrossRef]
- Canavelli, P.; Islam, S.; Powner, M.W. Peptide ligation by chemoselective aminonitrile coupling in water. Nature 2019, 571, 546–549. [Google Scholar] [CrossRef] [PubMed]
- Cockell, C.S.; Horneck, G. The history of the UV radiation climate of the earth—Theoretical and space-based observations. Photochem. Photobiol. 2001, 73, 447–451. [Google Scholar] [CrossRef]
- Garcia-Pichel, F. Solar ultraviolet and the evolutionary history of cyanobacteria. Orig. Life Evol. Biosph. 1998, 28, 321–347. [Google Scholar] [CrossRef] [PubMed]
- Nuevo, M.; Auger, G.; Blanot, D.; d’Hendecourt, L. A detailed study of the amino acids produced from the vacuum UV irradiation of interstellar ice analogs. Orig. Life Evol. Biosph. 2008, 38, 37–56. [Google Scholar] [CrossRef] [PubMed]
- Sagan, C.; Khare, B.N. Long-wavelength ultraviolet photoproduction of amino acids on the primitive Earth. Science 1971, 173, 417–420. [Google Scholar] [CrossRef] [PubMed]
- Powner, M.W.; Anastasi, C.; Crowe, M.A.; Parkes, A.L.; Raftery, J.; Sutherland, J.D. On the prebiotic synthesis of ribonucleotides: Photoanomerisation of cytosine nucleosides and nucleotides revisited. ChemBioChem 2007, 8, 1170–1179. [Google Scholar] [CrossRef]
- Patel, B.H.; Percivalle, C.; Ritson, D.J.; Duffy, C.D.; Sutherland, J.D. Common origins of RNA, protein and lipid precursors in a cyanosulfidic protometabolism. Nat. Chem. 2015, 7, 301–307. [Google Scholar] [CrossRef]
- Janicki, M.J.; Roberts, S.J.; Šponer, J.; Powner, M.W.; Góra, R.W.; Szabla, R. Photostability of oxazoline RNA-precursors in UV-rich prebiotic environments. Chem. Commun. 2018, 54, 13407–13410. [Google Scholar] [CrossRef]
- Bonfio, C.; Valer, L.; Scintilla, S.; Shah, S.; Evans, D.J.; Jin, L.; Szostak, J.W.; Sasselov, D.D.; Sutherland, J.D.; Mansy, S.S. UV-light-driven prebiotic synthesis of iron-sulfur clusters. Nat. Chem. 2017, 9, 1229–1234. [Google Scholar] [CrossRef]
- Cockell, C.S. The ultraviolet history of the terrestrial planets—Implications for biological evolution. Planet. Space Sci. 2000, 48, 203–214. [Google Scholar] [CrossRef]
- Cleaves, H.J.; Miller, S.L. Oceanic protection of prebiotic organic compounds from UV radiation. Proc. Natl. Acad. Sci. USA 1998, 95, 7260–7263. [Google Scholar] [CrossRef] [PubMed]
- Scappini, F.; Casadei, F.; Zamboni, R.; Monti, S.; Giorgianni, P.; Capobianco, M.L. Laboratory simulation of UV irradiation from the Sun on amino acids. I: Irradiation of tyrosine. Int. J. Astrobiol. 2007, 6, 123–129. [Google Scholar] [CrossRef]
- Scappini, F.; Capobianco, M.L.; Casadei, F.; Zamboni, R.; Giorgianni, P. Laboratory simulation of UV irradiation from the Sun on amino acids. II. Irradiation of phenylalanine and tryptophan. Int. J. Astrobiol. 2007, 6, 281–289. [Google Scholar] [CrossRef]
- Scappini, F.; Casadei, F.; Zamboni, R.; Franchi, M.; Gallori, E.; Monti, S. Protective effect of clay minerals on adsorbed nucleic acid against UV radiation: Possible role in the origin of life. Int. J. Astrobiol. 2004, 3, 17–19. [Google Scholar] [CrossRef]
- Papke, K.G. Montmorillonite deposits in Nevada. Clays Clay Miner. 1969, 17, 211–222. [Google Scholar] [CrossRef]
- Zaia, D.A.M. A review of adsorption of amino acids on minerals: Was it important for origin of life? Amino Acids 2004, 27, 113–118. [Google Scholar] [CrossRef] [PubMed]
- Kalra, S.; Pant, C.K.; Pathak, H.D.; Mehata, M.S. Studies on the adsorption of peptides of glycine/alanine on montmorillonite clay with or without co-ordinated divalent cations. Colloid Surface A 2003, 212, 43–50. [Google Scholar] [CrossRef]
- Hashizume, H. Adsorption of nucleic acid bases, ribose, and phosphate by some clay minerals. Life 2015, 5, 637–650. [Google Scholar] [CrossRef]
- Ferris, J.P.; Hagan, W.J. The Adsorption and Reaction of Adenine-Nucleotides on Montmorillonite. Orig. Life Evol. Biosph. 1986, 17, 69–84. [Google Scholar] [CrossRef]
- Bernal, J.D. The physical basis of life. Proc. R. Soc. Lond. A 1951, 62, 537–558. [Google Scholar]
- Boillot, F.; Chabin, A.; Bure, C.; Venet, M.; Belsky, A.; Bertrand-Urbaniak, M.; Delmas, A.; Brack, A.; Barbier, B. The Perseus Exobiology mission on MIR: Behaviour of amino acids and peptides in earth orbit. Orig. Life Evol. Biosph. 2002, 32, 359–385. [Google Scholar] [CrossRef] [PubMed]
- Scappini, F.; Capobianco, M.L.; Casadei, F.; Zamboni, R. Laboratory simulation of ultraviolet irradiation from the Sun on amino acids. III. irradiation of glycine-tyrosine. Int. J. Astrobiol. 2009, 8, 63–68. [Google Scholar] [CrossRef]
- Ma, B.G.; Chen, L.; Ji, H.F.; Chen, Z.H.; Yang, F.R.; Wang, L.; Qu, G.; Jiang, Y.Y.; Ji, C.; Zhang, H.Y. Characters of very ancient proteins. Biochem. Biophys. Res. Commun. 2008, 366, 607–611. [Google Scholar] [CrossRef] [PubMed]
- Ranjan, S.; Sasselov, D.D. Constraints on the Early Terrestrial Surface UV Environment Relevant to Prebiotic Chemistry. Astrobiology 2017, 17, 169–204. [Google Scholar] [CrossRef]
- Ranjan, S.; Sasselov, D.D. Influence of the UV Environment on the Synthesis of Prebiotic Molecules. Astrobiology 2016, 16, 68–88. [Google Scholar] [CrossRef]
- Dashman, T.; Stotzky, G. Physical-properties of homoionic montmorillonite and kaolinite complexed with amino-acids and peptides. Soil Biol. Biochem. 1985, 17, 189–195. [Google Scholar] [CrossRef]
- Rimola, A.; Sodupe, M.; Ugliengo, P. Role of mineral surfaces in prebiotic chemical evolution. in silico quantum mechanical studies. Life 2019, 9, 10. [Google Scholar] [CrossRef]
- Holm, N.G.; Dumont, M.; Ivarsson, M.; Konn, C. Alkaline fluid circulation in ultramafic rocks and formationof nucleotide constituents: A hypothesis. Geochem. Trans. 2006, 7, 1–7. [Google Scholar] [CrossRef]
- Russell, M.J. The importance of being alkaline. Science 2003, 302, 580–581. [Google Scholar] [CrossRef]
- Sakata, K.; Kitadai, N.; Yokoyama, T. Effects of pH and temperature on dimerization rate of glycine: Evaluation of favorable environmental conditions for chemical evolution of life. Geochim. Cosmochim. Acta 2010, 74, 6841–6851. [Google Scholar] [CrossRef]
- Abou Mrad, N.; Ajram, G.; Rossi, J.C.; Boiteau, L.; Duvernay, F.; Pascal, R.; Danger, G. The Prebiotic C-Terminal Elongation of Peptides Can Be Initiated by N-Carbamoyl Amino Acids. Chem. A Eur. J. 2017, 23, 7418–7421. [Google Scholar] [CrossRef] [PubMed]
- Ramos, M.E.; Huertas, F.J. Adsorption of glycine on montmorillonite in aqueous solutions. Appl. Clay Sci. 2013, 80–81, 10–17. [Google Scholar] [CrossRef]
- Joshi, P.C.; Aldersley, M.F.; Delano, J.W.; Ferris, J.P. Mechanism of montmorillonite catalysis in the formation of RNA oligomers. J. Am. Chem. Soc. 2009, 131, 13369–13374. [Google Scholar] [CrossRef] [PubMed]
- Todd, Z.R.; Szabla, R.; Szostak, J.W.; Sasselov, D.D. UV photostability of three 2-aminoazoles with key roles in prebiotic chemistry on the early earth. Chem. Commun. 2019, 55, 10388–10391. [Google Scholar] [CrossRef]
- Todd, Z.R.; Fahrenbach, A.C.; Magnani, C.J.; Ranjan, S.; Bjorkbom, A.; Szostak, J.W.; Sasselov, D.D. Solvated-electron production using cyanocuprates is compatible with the UV-environment on a Hadean-Archaean Earth. Chem. Commun. 2018, 54, 1121–1124. [Google Scholar] [CrossRef]


| Sample Name | Adsorbed/% | #Desorbed/% | |
|---|---|---|---|
| pH 10 | pH 3 | pH 10 | |
| Phe2 | 13.45% | 97.57% | 88.41% |
| Tyr2 | 2.09% | 80.25% | 97.25% |
| Val2 | 5.14% | 62.36% | 96.02% |
| Ala2 | 3.99% | 77.04% | 96.39% |
| Leu2 | 2.51% | 62.14% | 98.23% |
| Pro2 | 30.53% | 90.09% | 63.55% |
| FFFFD | 1.81% | 99.38% | 87.79% |
| Phe | 33.36% | 47.46% | 65.79% |
| Items | MMT-Adsorbed Sample | Free Sample | ||||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Survival Rate/% | pH = 10 | pH = 3 | pH = 10 | pH = 3 | ||||||
| Sample | 1 day | 5 days | 1 day | 5 days | 1 day | 5 days | 1 day | 5 days | ||
| Phe2 | 77.82% | 15.96% | 90.86% | 83.58% | 54.90% | 23.16% | 44.49% | 2.77% | ||
| Tyr2 | 50.72% | 33.29% | 79.39% | 66.42% | 61.55% | 41.98% | 64.77% | 44.69% | ||
| Val2 | 84.16% | 56.23% | 83.74% | 68.41% | 85.25% | 60.47% | 76.14% | 14.03% | ||
| Ala2 | 63.29% | 50.76% | 87.43% | 79.31% | 82.76% | 19.16% | 79.63% | 15.92% | ||
| Leu2 | 84.55% | 27.17% | 92.30% | 78.61% | 87.31% | 44.56% | 86.72% | 42.75% | ||
| Pro2 | 18.78% | 17.36% | 46.57% | 45.41% | 21.12% | trace | 35.72% | 6.49% | ||
| FFFFD | 73.38% | 15.45% | 80.36% | 53.30% | 17.87% | 3.55% | 31.49% | 4.27% | ||
| Phe | 79.87% | 64.56% | 88.41% | 75.52% | 7.50% | 1.49% | 86.83% | 70.63% | ||
| Entry | Phe | Phe2 | FFFFD | Tyr2 | Val2 | Ala2 | Leu2 | Pro2 |
|---|---|---|---|---|---|---|---|---|
| pKa1 | 2.207 | 2.746 | 3.122 | 2.562 | 3.093 | 3.026 | 2.961 | 3.162 |
© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Lin, R.; Wang, Y.; Li, X.; Liu, Y.; Zhao, Y. pH-Dependent Adsorption of Peptides on Montmorillonite for Resisting UV Irradiation. Life 2020, 10, 45. https://doi.org/10.3390/life10040045
Lin R, Wang Y, Li X, Liu Y, Zhao Y. pH-Dependent Adsorption of Peptides on Montmorillonite for Resisting UV Irradiation. Life. 2020; 10(4):45. https://doi.org/10.3390/life10040045
Chicago/Turabian StyleLin, Rongcan, Yueqiao Wang, Xin Li, Yan Liu, and Yufen Zhao. 2020. "pH-Dependent Adsorption of Peptides on Montmorillonite for Resisting UV Irradiation" Life 10, no. 4: 45. https://doi.org/10.3390/life10040045
APA StyleLin, R., Wang, Y., Li, X., Liu, Y., & Zhao, Y. (2020). pH-Dependent Adsorption of Peptides on Montmorillonite for Resisting UV Irradiation. Life, 10(4), 45. https://doi.org/10.3390/life10040045




