Does Physical Exercise Always Improve Bone Quality in Rats?
Abstract
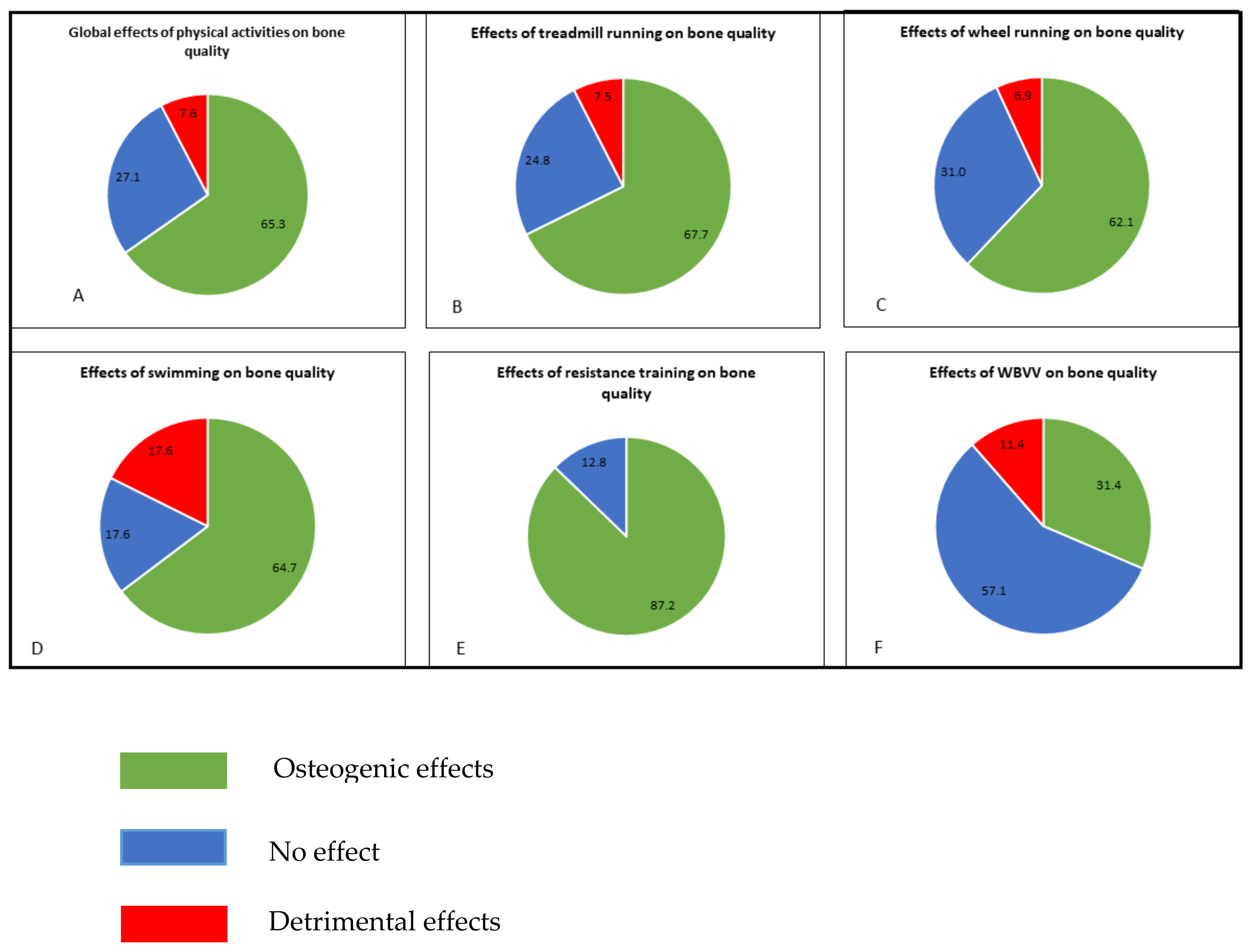
:1. Introduction
2. Population and Bones Studied
2.1. Population Studied
2.2. Bones Studied
3. Bone Parameters Studied
3.1. Bone Mineral Density
3.2. Bone Micro-Architecture
3.3. Bone Mechanical Properties
3.4. Osteocyte Lineage Biology
3.5. Osteoclast Biology
4. Effects of Physical Exercises on Bone Health Status
4.1. Effects of Treadmill Running Exercises
4.1.1. Treadmill Protocol Characteristics
4.1.2. Specific Changes in Bone Mass, Structure and Strength
Healthy Rats
Osteopenic Models
4.1.3. Specific Changes in Osteoblast/Osteocyte Lineage Biology
Healthy Rats
Osteopenic Models
4.1.4. Specific Changes in Osteoclast Biology
4.2. Voluntary Wheel Running Protocols
4.2.1. Protocol Characteristics
4.2.2. Specific Changes in Bone Mass, Structure and Strength
Healthy Rats
Osteopenic Models
4.2.3. Specific Changes in Osteoblast/Osteocyte Lineage Biology
Healthy Rats
Osteopenic Models
4.2.4. Specific Changes in Osteoclast Biology
Healthy Rats
4.3. Effects of Swimming Exercises
4.3.1. Swimming Protocol Characteristics
4.3.2. Specific Changes in Bone Mass, Structure and Strength
Healthy Rats
Osteopenic Models
4.3.3. Specific Changes in Osteoblast/Osteocyte Lineage Biology
Healthy Rats
4.3.4. Specific Changes in Osteoclast Biology
4.4. Effects of Resistance Exercises
4.4.1. Resistance Protocol Characteristics
4.4.2. Specific Changes in Bone Mass, Structure and Strength
Osteopenic Model
4.4.3. Specific Changes in Osteoblast/Osteocyte Lineage Biology
Healthy Rats
Osteopenic Models
4.4.4. Specific Changes in Osteoclast Biology
Healthy Rats
Osteopenic Models
4.5. Whole-Body Vibrations Exercise
4.5.1. Whole-Body Vibrations Protocol Characteristics
4.5.2. Specific Changes in Bone Mass, Structure and Strength
4.5.3. Specific Changes in Osteoblast/Osteocyte Lineage Biology
Healthy Rats
Osteopenic Models
4.5.4. Specific Changes in Osteoclast Biology
Osteopenic Models
5. Final General Discussion
Supplementary Materials
Author Contributions
Funding
Conflicts of Interest
Abbreviations
| ACTH | Adrenocorticotropic hormone |
| a.ES | active erosion surface |
| BMD | bone mineral density |
| BMPs | bone morphogenetic proteins |
| BS | bone surface (BS) |
| BV/TV | bone volume/tissue volume |
| CAS | chronic aversive stimuli |
| DXA | dual-energy X-ray Absorptiometry |
| ES | erosion surface |
| GRF | ground reaction forces |
| HCRs | high-capacity runners |
| HSPs | heat shock proteins |
| It | intermittent running |
| LCRs | low-capacity runners |
| LMHFV | low-magnitude, high-frequency vibration |
| MAR | metaphyseal mineral apposition rate |
| Oc.S | osteoclast surface |
| OPG | osteoprotegerin |
| OVX | ovariectomized |
| RANKL | receptor activator of nuclear factor kappa-B ligand |
| SRT | simulated resistance training |
| TRAP | tartrate-resistant acid phosphatase |
| CTx | terminal collagen cross-links |
| Tb.N | trabecular number |
| Ct.Th | cortical thickness |
| Tb.BMD | trabecular BMD |
| Tb.Sp | trabecular spacing |
| Tb.Th | trabecular thickness |
| WBVV | whole-body vertical vibration |
References
- Reginster, J.-Y.; Burlet, N. Osteoporosis: A still increasing prevalence. Bone 2006, 38, 4–9. [Google Scholar] [CrossRef]
- Sözen, T.; Özışık, L.; Başaran, N.Ç. An overview and management of osteoporosis. Eur. J. Rheumatol. 2017, 4, 46–56. [Google Scholar] [CrossRef]
- Wade, S.W.; Strader, C.; Fitzpatrick, L.A.; Anthony, M.S.; O’Malley, C.D. Estimating prevalence of osteoporosis: Examples from industrialized countries. Arch. Osteoporos. 2014, 9, 182. [Google Scholar] [CrossRef]
- Cooper, C. Epidemiology of osteoporosis. Osteoporos. Int. 1999, 9 (Suppl. 2), S2. [Google Scholar] [CrossRef]
- Lane, J.M.; Serota, A.C.; Raphael, B. Osteoporosis: Differences and similarities in male and female patients. Orthop. Clin. N. Am. 2006, 37, 601–609. [Google Scholar] [CrossRef]
- Wilk, R.; Skrzypek, M.; Kowalska, M.; Kusz, D.; Wielgórecki, A.; Horyniecki, M.; Sliwiak, J.; Piejczyk, S.; Pluskiewicz, W. Standardized incidence and trend of osteoporotic hip fracture in Polish women and men: A nine year observation. Maturitas 2014, 77, 59–63. [Google Scholar] [CrossRef] [PubMed]
- Antonelli, M.; Einstadter, D.; Magrey, M. Screening and treatment of osteoporosis after hip fracture: Comparison of sex and race. J. Clin. Densitom. 2014, 17, 479–483. [Google Scholar] [CrossRef] [PubMed]
- Johnell, O.; Odén, A.; De Laet, C.; Garnero, P.; Delmas, P.D.; Kanis, J.A. Biochemical indices of bone turnover and the assessment of fracture probability. Osteoporos. Int. 2002, 13, 523–526. [Google Scholar] [CrossRef] [PubMed]
- Garnero, P.; Delmas, P.D. Contribution of bone mineral density and bone turnover markers to the estimation of risk of osteoporotic fracture in postmenopausal women. J. Musculoskelet. Neuronal Interact. 2004, 4, 50–63. [Google Scholar] [PubMed]
- Kanis, J.A.; Johnell, O.; De Laet, C.; Johansson, H.; Oden, A.; Delmas, P.; Eisman, J.; Fujiwara, S.; Garnero, P.; Kroger, H.; et al. A meta-analysis of previous fracture and subsequent fracture risk. Bone 2004, 35, 375–382. [Google Scholar] [CrossRef]
- Kanis, J.A.; Johansson, H.; Oden, A.; Johnell, O.; De Laet, C.; Eisman, J.A.; McCloskey, E.V.; Mellstrom, D.; Melton, L.J.; Pols, H.A.P.; et al. A family history of fracture and fracture risk: A meta-analysis. Bone 2004, 35, 1029–1037. [Google Scholar] [CrossRef] [PubMed]
- Riggs, B.L.; Melton, L.J. The worldwide problem of osteoporosis: Insights afforded by epidemiology. Bone 1995, 17, S505–S511. [Google Scholar] [CrossRef]
- Bonaiuti, D.; Shea, B.; Iovine, R.; Negrini, S.; Robinson, V.; Kemper, H.C.; Wells, G.; Tugwell, P.; Cranney, A. Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst. Rev. 2002, CD000333. [Google Scholar] [CrossRef]
- Shea, B.; Bonaiuti, D.; Iovine, R.; Negrini, S.; Robinson, V.; Kemper, H.C.; Wells, G.; Tugwell, P.; Cranney, A. Cochrane Review on exercise for preventing and treating osteoporosis in postmenopausal women. Eur. Medicophys. 2004, 40, 199–209. [Google Scholar]
- Stránský, M.; Rysavá, L. Nutrition as prevention and treatment of osteoporosis. Physiol. Res. 2009, 58 (Suppl. 1), S7–S11. [Google Scholar]
- Howe, T.E.; Shea, B.; Dawson, L.J.; Downie, F.; Murray, A.; Ross, C.; Harbour, R.T.; Caldwell, L.M.; Creed, G. Exercise for preventing and treating osteoporosis in postmenopausal women. Cochrane Database Syst. Rev. 2011, CD000333. [Google Scholar] [CrossRef]
- Warburton, D.E.R.; Nicol, C.W.; Bredin, S.S.D. Health benefits of physical activity: The evidence. CMAJ 2006, 174, 801–809. [Google Scholar] [CrossRef] [Green Version]
- Warburton, D.E.; Charlesworth, S.; Ivey, A.; Nettlefold, L.; Bredin, S.S. A systematic review of the evidence for Canada’s Physical Activity Guidelines for Adults. Int. J. Behav. Nutr. Phys. Acta 2010, 7, 39. [Google Scholar] [CrossRef] [Green Version]
- Wolff, J. The Law of Bone Remodelling; Springer: Berlin/Heidelberg, Germany, 1986; ISBN 978-3-642-71033-9. [Google Scholar]
- Santos, L.; Elliott-Sale, K.J.; Sale, C. Exercise and bone health across the lifespan. Biogerontology 2017, 18, 931–946. [Google Scholar] [CrossRef] [Green Version]
- Rubin, C.T.; Lanyon, L.E. Regulation of bone formation by applied dynamic loads. J. Bone Joint Surg. Am. 1984, 66, 397–402. [Google Scholar] [CrossRef]
- Bonewald, L.F. Osteocytes as dynamic multifunctional cells. Ann. N. Y. Acad. Sci. 2007, 1116, 281–290. [Google Scholar] [CrossRef]
- Bonewald, L.F.; Johnson, M.L. Osteocytes, mechanosensing and Wnt signaling. Bone 2008, 42, 606–615. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Daly, R.M.; Duckham, R.L.; Gianoudis, J. Evidence for an interaction between exercise and nutrition for improving bone and muscle health. Curr. Osteoporos. Rep. 2014, 12, 219–226. [Google Scholar] [CrossRef] [PubMed]
- Marques, E.A.; Mota, J.; Carvalho, J. Exercise effects on bone mineral density in older adults: A meta-analysis of randomized controlled trials. Age (Dordr) 2012, 34, 1493–1515. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Musumeci, G.; Loreto, C.; Leonardi, R.; Castorina, S.; Giunta, S.; Carnazza, M.L.; Trovato, F.M.; Pichler, K.; Weinberg, A.M. The effects of physical activity on apoptosis and lubricin expression in articular cartilage in rats with glucocorticoid-induced osteoporosis. J. Bone Miner. Metab. 2013, 31, 274–284. [Google Scholar] [CrossRef]
- Lelovas, P.P.; Xanthos, T.T.; Thoma, S.E.; Lyritis, G.P.; Dontas, I.A. The laboratory rat as an animal model for osteoporosis research. Comp. Med. 2008, 58, 424–430. [Google Scholar]
- Meakin, L.B.; Price, J.S.; Lanyon, L.E. The Contribution of Experimental in vivo Models to Understanding the Mechanisms of Adaptation to Mechanical Loading in Bone. Front. Endocrinol. (Lausanne) 2014, 5, 154. [Google Scholar] [CrossRef] [Green Version]
- Iwamoto, J.; Takeda, T.; Sato, Y. Effect of treadmill exercise on bone mass in female rats. Exp. Anim. 2005, 54, 1–6. [Google Scholar] [CrossRef] [Green Version]
- Saino, H.; Luther, F.; Carter, D.H.; Natali, A.J.; Turner, D.L.; Shahtaheri, S.M.; Aaron, J.E. Evidence for an extensive collagen type III proximal domain in the rat femur. II. Expansion with exercise. Bone 2003, 32, 660–668. [Google Scholar] [CrossRef]
- Gao, X.; Ma, W.; Dong, H.; Yong, Z.; Su, R. Establishing a rapid animal model of osteoporosis with ovariectomy plus low calcium diet in rats. Int. J. Clin. Exp. Pathol. 2014, 7, 5123–5128. [Google Scholar]
- Morey, E.R. Spaceflight and Bone Turnover: Correlation with a New Rat Model of Weightlessness. Bioscience 1979, 29, 168–172. [Google Scholar] [CrossRef]
- Ju, Y.-I.; Sone, T.; Ohnaru, K.; Choi, H.-J.; Fukunaga, M. Differential effects of jump versus running exercise on trabecular architecture during remobilization after suspension-induced osteopenia in growing rats. J. Appl. Physiol. 2012, 112, 766–772. [Google Scholar] [CrossRef] [PubMed]
- Ju, Y.-I.; Sone, T.; Ohnaru, K.; Choi, H.-J.; Choi, K.-A.; Fukunaga, M. Jump exercise during hindlimb unloading protect against the deterioration of trabecular bone microarchitecture in growing young rats. Springerplus 2013, 2, 35. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ju, Y.-I.; Sone, T.; Okamoto, T.; Fukunaga, M. Jump exercise during remobilization restores integrity of the trabecular architecture after tail suspension in young rats. J. Appl. Physiol. 2008, 104, 1594–1600. [Google Scholar] [CrossRef] [Green Version]
- Falcai, M.J.; Louzada, M.J.Q.; de Paula, F.J.A.; Okubo, R.; Volpon, J.B. A modified technique of rat tail suspension for longer periods of observation. Aviat. Space Environ. Med. 2012, 83, 1176–1180. [Google Scholar] [CrossRef]
- Globus, R.K.; Morey-Holton, E. Hindlimb unloading: Rodent analog for microgravity. J. Appl. Physiol. 2016, 120, 1196–1206. [Google Scholar] [CrossRef]
- Chowdhury, P.; Long, A.; Harris, G.; Soulsby, M.E.; Dobretsov, M. Animal model of simulated microgravity: A comparative study of hindlimb unloading via tail versus pelvic suspension. Physiol. Rep. 2013, 1, e00012. [Google Scholar] [CrossRef]
- Arahão, G.S.; Shimano, A.C.; Picado, C.H.F. Effects of physical activity on the mechanical properties of osteopenic female rats’ femurs and tibiae. Acta Ortopédica Bras. 2006, 14, 242–245. [Google Scholar] [CrossRef]
- Kang, Y.-S.; Kim, J.-C.; Kim, J.-S.; Kim, S.H. Effects of Swimming Exercise on Serum Irisin and Bone FNDC5 in Rat Models of High-Fat Diet-Induced Osteoporosis. J. Sports Sci. Med. 2019, 18, 596–603. [Google Scholar]
- Markiewicz-Górka, I.; Kuropka, P.; Januszewska, L.; Jaremków, A.; Pawłowski, P.; Pawlas, N.; Prokopowicz, A.; Gonzalez, E.; Nikodem, A.; Pawlas, K. Influence of physical training on markers of bone turnover, mechanical properties, morphological alterations, density and mineral contents in the femur of rats exposed to cadmium and/or alcohol. Toxicol. Ind. Health 2019, 35, 277–293. [Google Scholar] [CrossRef]
- Rochester, E.; Wickman, B.E.; Bell, A.; Simecka, C.; Clayton, Z.S.; Hakkak, R.; Hooshmand, S. A diet containing high- versus low-daidzein does not affect bone density and osteogenic gene expression in the obese Zucker rat model. Food Funct. 2019, 10, 6851–6857. [Google Scholar] [CrossRef] [PubMed]
- Yuasa, Y.; Miyakoshi, N.; Kasukawa, Y.; Nagahata, I.; Akagawa, M.; Ono, Y.; Sato, C.; Tsuchie, H.; Nozaka, K.; Nagasawa, H.; et al. Effects of bazedoxifene and low-intensity aerobic exercise on bone and fat parameters in ovariectomized rats. J. Bone Miner. Metab. 2020, 38, 179–187. [Google Scholar] [CrossRef] [PubMed]
- Ocarino, N.M.; Boeloni, J.N.; Silva, J.F.; Goes, A.M.; Marubayashi, U.; Serakides, R. Different effect of the nitric oxide synthase inhibition between the cortical and trabecular bone of osteoporotic female rats submitted or no the physical activity. Sci. Sports 2010, 25, 260–266. [Google Scholar] [CrossRef]
- Fonseca, H.; Moreira-Gonçalves, D.; Coriolano, H.-J.A.; Duarte, J.A. Bone quality: The determinants of bone strength and fragility. Sports Med. 2014, 44, 37–53. [Google Scholar] [CrossRef] [PubMed]
- Robling, A.G.; Burr, D.B.; Turner, C.H. Recovery periods restore mechanosensitivity to dynamically loaded bone. J. Exp. Biol. 2001, 204, 3389–3399. [Google Scholar]
- Blake, G.M.; Fogelman, I. The role of DXA bone density scans in the diagnosis and treatment of osteoporosis. Postgrad Med. J. 2007, 83, 509–517. [Google Scholar] [CrossRef] [Green Version]
- Hamann, N.; Kohler, T.; Müller, R.; Brüggemann, G.-P.; Niehoff, A. The effect of level and downhill running on cortical and trabecular bone in growing rats. Calcif. Tissue Int. 2012, 90, 429–437. [Google Scholar] [CrossRef]
- Järvinen, T.L.N.; Kannus, P.; Pajamäki, I.; Vuohelainen, T.; Tuukkanen, J.; Järvinen, M.; Sievänen, H. Estrogen deposits extra mineral into bones of female rats in puberty, but simultaneously seems to suppress the responsiveness of female skeleton to mechanical loading. Bone 2003, 32, 642–651. [Google Scholar] [CrossRef]
- Welch, J.M.; Weaver, C.M.; Turner, C.H. Adaptations to free-fall impact are different in the shafts and bone ends of rat forelimbs. J. Appl. Physiol. 2004, 97, 1859–1865. [Google Scholar] [CrossRef] [Green Version]
- Warner, S.E.; Shea, J.E.; Miller, S.C.; Shaw, J.M. Adaptations in cortical and trabecular bone in response to mechanical loading with and without weight bearing. Calcif. Tissue Int. 2006, 79, 395–403. [Google Scholar] [CrossRef]
- Bodnar, M.; Skalicky, M.; Viidik, A.; Erben, R.G. Interaction between exercise, dietary restriction and age-related bone loss in a rodent model of male senile osteoporosis. Gerontology 2012, 58, 139–149. [Google Scholar] [CrossRef] [PubMed]
- Bouxsein, M.L.; Boyd, S.K.; Christiansen, B.A.; Guldberg, R.E.; Jepsen, K.J.; Müller, R. Guidelines for assessment of bone microstructure in rodents using micro-computed tomography. J. Bone Miner. Res. 2010, 25, 1468–1486. [Google Scholar] [CrossRef] [PubMed]
- Malhan, D.; Muelke, M.; Rosch, S.; Schaefer, A.B.; Merboth, F.; Weisweiler, D.; Heiss, C.; Arganda-Carreras, I.; El Khassawna, T. An Optimized Approach to Perform Bone Histomorphometry. Front. Endocrinol. (Lausanne) 2018, 9, 666. [Google Scholar] [CrossRef] [PubMed]
- Bourrin, S.; Genty, C.; Palle, S.; Gharib, C.; Alexandre, C. Adverse effects of strenuous exercise: A densitometric and histomorphometric study in the rat. J. Appl. Physiol. 1994, 76, 1999–2005. [Google Scholar] [CrossRef]
- Recker, R.R.; Barger-Lux, M.J. The elusive concept of bone quality. Curr. Osteoporos. Rep. 2004, 2, 97–100. [Google Scholar] [CrossRef]
- Cole, J.H.; van der Meulen, M.C.H. Whole bone mechanics and bone quality. Clin. Orthop. Relat. Res. 2011, 469, 2139–2149. [Google Scholar] [CrossRef] [Green Version]
- Kitagawa, J.; Nakahara, Y. Associations of daily walking steps with calcaneal ultrasound parameters and a bone resorption marker in elderly Japanese women. J. Physiol. Anthr. 2008, 27, 295–300. [Google Scholar] [CrossRef] [Green Version]
- Toumi, H.; Benaitreau, D.; Pallu, S.; Mazor, M.; Hambli, R.; Ominsky, M.; Lespessailles, E. Effects of anti-sclerostin antibody and running on bone remodeling and strength. Bone Rep. 2015, 2, 52–58. [Google Scholar] [CrossRef] [Green Version]
- Aguirre, J.I.; Plotkin, L.I.; Stewart, S.A.; Weinstein, R.S.; Parfitt, A.M.; Manolagas, S.C.; Bellido, T. Osteocyte apoptosis is induced by weightlessness in mice and precedes osteoclast recruitment and bone loss. J. Bone Miner. Res. 2006, 21, 605–615. [Google Scholar] [CrossRef]
- Boudenot, A.; Presle, N.; Uzbekov, R.; Toumi, H.; Pallu, S.; Lespessailles, E. Effect of interval-training exercise on subchondral bone in a chemically-induced osteoarthritis model. Osteoarthr. Cartil. 2014, 22, 1176–1185. [Google Scholar] [CrossRef]
- Iijima, H.; Aoyama, T.; Ito, A.; Yamaguchi, S.; Nagai, M.; Tajino, J.; Zhang, X.; Kuroki, H. Effects of short-term gentle treadmill walking on subchondral bone in a rat model of instability-induced osteoarthritis. Osteoarthr. Cartil. 2015, 23, 1563–1574. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Volpon, J.B.; Silva, A.V.; Falcai, M.J.; Louzada, M.J.Q.; Zamarioli, A.; Kotake, B.G.D.S.; Issa, J.P.M. The osteogenic effects of swimming on bone mass, strength, and microarchitecture in rats with unloading-induced bone loss. Microsc. Res. Tech. 2015, 78, 784–791. [Google Scholar] [CrossRef] [PubMed]
- Pichler, K.; Loreto, C.; Leonardi, R.; Reuber, T.; Weinberg, A.M.; Musumeci, G. RANKL is downregulated in bone cells by physical activity (treadmill and vibration stimulation training) in rat with glucocorticoid-induced osteoporosis. Histol. Histopathol. 2013, 28, 1185–1196. [Google Scholar] [CrossRef] [PubMed]
- Bonnet, N.; Beaupied, H.; Vico, L.; Dolleans, E.; Laroche, N.; Courteix, D.; Benhamou, C.-L. Combined effects of exercise and propranolol on bone tissue in ovariectomized rats. J. Bone Miner. Res. 2007, 22, 578–588. [Google Scholar] [CrossRef]
- Bonnet, N.; Laroche, N.; Beaupied, H.; Vico, L.; Dolleans, E.; Benhamou, C.L.; Courteix, D. Doping dose of salbutamol and exercise training: Impact on the skeleton of ovariectomized rats. J. Appl. Physiol. 2007, 103, 524–533. [Google Scholar] [CrossRef] [PubMed]
- Smith, M.Z.; Goettsch, B.M.; Van Ramshorst, R.D.; O’Brien, J.A.; Jaque, S.V.; Sumida, K.D. Resistance training and bone mineral density during growth. Int. J. Sports Med. 2008, 29, 316–321. [Google Scholar] [CrossRef] [Green Version]
- Bourrin, S.; Palle, S.; Pupier, R.; Vico, L.; Alexandre, C. Effect of physical training on bone adaptation in three zones of the rat tibia. J. Bone Miner. Res. 1995, 10, 1745–1752. [Google Scholar] [CrossRef]
- Notomi, T.; Karasaki, I.; Okazaki, Y.; Okimoto, N.; Kato, Y.; Ohura, K.; Noda, M.; Nakamura, T.; Suzuki, M. Insulinogenic sucrose+amino acid mixture ingestion immediately after resistance exercise has an anabolic effect on bone compared with non-insulinogenic fructose+amino acid mixture in growing rats. Bone 2014, 65, 42–48. [Google Scholar] [CrossRef]
- Boudenot, A.; Maurel, D.B.; Pallu, S.; Ingrand, I.; Boisseau, N.; Jaffré, C.; Portier, H. Quick benefits of interval training versus continuous training on bone: A dual-energy X-ray absorptiometry comparative study. Int. J. Exp. Pathol. 2015, 96, 370–377. [Google Scholar] [CrossRef]
- Nakajima, D.; Kim, C.S.; Oh, T.W.; Yang, C.Y.; Naka, T.; Igawa, S.; Ohta, F. Suppressive effects of genistein dosage and resistance exercise on bone loss in ovariectomized rats. J. Physiol. Anthr. Appl. Hum. Sci. 2001, 20, 285–291. [Google Scholar] [CrossRef] [Green Version]
- Gross, T.S.; Poliachik, S.L.; Ausk, B.J.; Sanford, D.A.; Becker, B.A.; Srinivasan, S. Why rest stimulates bone formation: A hypothesis based on complex adaptive phenomenon. Exerc. Sport Sci. Rev. 2004, 32, 9–13. [Google Scholar] [CrossRef] [PubMed]
- Robling, A.G.; Turner, C.H. Mechanical Signaling for Bone Modeling and Remodeling. Crit. Rev. Eukaryot. Gene Exp. 2009, 19, 319–338. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Rubin, C.T.; Lanyon, L.E. Regulation of bone mass by mechanical strain magnitude. Calcif. Tissue Int. 1985, 37, 411–417. [Google Scholar] [CrossRef] [PubMed]
- Forwood, M.R.; Parker, A.W. Repetitive loading, in vivo, of the tibia and femora of rats: Effects of a single bout of treadmill running. Calcif. Tissue Int. 1992, 50, 193–196. [Google Scholar] [CrossRef] [PubMed]
- Fuchs, R.K.; Shea, M.; Durski, S.L.; Winters-Stone, K.M.; Widrick, J.; Snow, C.M. Individual and combined effects of exercise and alendronate on bone mass and strength in ovariectomized rats. Bone 2007, 41, 290–296. [Google Scholar] [CrossRef]
- Kannus, P.; Sievänen, H.; Järvinen, T.L.; Järvinen, M.; Kvist, M.; Oja, P.; Vuori, I.; Jozsa, L. Effects of free mobilization and low- to high-intensity treadmill running on the immobilization-induced bone loss in rats. J. Bone Miner. Res. 1994, 9, 1613–1619. [Google Scholar] [CrossRef] [PubMed]
- Kannus, P.; Järvinen, T.L.; Sievänen, H.; Kvist, M.; Rauhaniemi, J.; Maunu, V.M.; Hurme, T.; Jozsa, L.; Järvinen, M. Effects of immobilization, three forms of remobilization, and subsequent deconditioning on bone mineral content and density in rat femora. J. Bone Miner. Res. 1996, 11, 1339–1346. [Google Scholar] [CrossRef]
- Leppänen, O.V.; Sievänen, H.; Jokihaara, J.; Pajamäki, I.; Kannus, P.; Järvinen, T.L.N. Pathogenesis of age-related osteoporosis: Impaired mechano-responsiveness of bone is not the culprit. PLoS ONE 2008, 3, e2540. [Google Scholar] [CrossRef]
- Rico, H.; Aznar, L.; Hernández, E.R.; Seco, C.; Sanchez-Atrio, A.; Villa, L.F.; Gervas, J.J. Effects of potassium bicarbonate supplementation on axial and peripheral bone mass in rats on strenuous treadmill training exercise. Calcif. Tissue Int. 1999, 65, 242–245. [Google Scholar] [CrossRef]
- Snyder, A.; Zierath, J.R.; Hawley, J.A.; Sleeper, M.D.; Craig, B.W. The effects of exercise mode, swimming vs. running, upon bone growth in the rapidly growing female rat. Mech. Ageing Dev. 1992, 66, 59–69. [Google Scholar] [CrossRef]
- Touron, J.; Perrault, H.; Julian, V.; Maisonnave, L.; Deat, P.; Auclair-Ronzaud, J.; Salles, J.; Walrand, S.; Hermet, J.; Rigaudiere, J.-P.; et al. Impact of Eccentric or Concentric Training on Body Composition and Energy Expenditure. Med. Sci. Sports Exerc. 2019, 51, 1944–1953. [Google Scholar] [CrossRef]
- Kim, J.-S.; Lee, Y.-H.; Yi, H.-K. Gradual downhill running improves age-related skeletal muscle and bone weakness: Implication of autophagy and bone morphogenetic proteins. Exp. Physiol. 2016, 101, 1528–1540. [Google Scholar] [CrossRef] [PubMed]
- Turner, R.T.; Lotinun, S.; Hefferan, T.E.; Morey-Holton, E. Disuse in adult male rats attenuates the bone anabolic response to a therapeutic dose of parathyroid hormone. J. Appl. Physiol. 2006, 101, 881–886. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tromp, A.M.; Bravenboer, N.; Tanck, E.; Oostlander, A.; Holzmann, P.J.; Kostense, P.J.; Roos, J.C.; Burger, E.H.; Huiskes, R.; Lips, P. Additional weight bearing during exercise and estrogen in the rat: The effect on bone mass, turnover, and structure. Calcif. Tissue Int. 2006, 79, 404–415. [Google Scholar] [CrossRef] [PubMed]
- Fang, J.; Gao, J.; Gong, H.; Zhang, T.; Zhang, R.; Zhan, B. Multiscale experimental study on the effects of different weight-bearing levels during moderate treadmill exercise on bone quality in growing female rats. Biomed. Eng. Online 2019, 18, 33. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Tang, L.; Gao, X.; Yang, X.; Zhang, D.; Zhang, X.; Du, H.; Han, Y.; Sun, L. Combination of Weight-Bearing Training and Anti-MSTN Polyclonal Antibody Improve Bone Quality In Rats. Int. J. Sport Nutr. Exerc. Metab. 2016, 26, 516–524. [Google Scholar] [CrossRef]
- van der Wiel, H.E.; Lips, P.; Graafmans, W.C.; Danielsen, C.C.; Nauta, J.; van Lingen, A.; Mosekilde, L. Additional weight-bearing during exercise is more important than duration of exercise for anabolic stimulus of bone: A study of running exercise in female rats. Bone 1995, 16, 73–80. [Google Scholar] [CrossRef]
- Omi, N.; Nakamura, T.; Ezawa, I. Modulation of bone mass and turnover in growing rats by voluntary weight-bearing exercise and glucose supplementation. J. Nutr. Sci. Vitaminol. 1998, 44, 409–421. [Google Scholar] [CrossRef] [Green Version]
- Huang, T.-H.; Chang, F.-L.; Lin, S.-C.; Liu, S.-H.; Hsieh, S.S.; Yang, R.-S. Endurance treadmill running training benefits the biomaterial quality of bone in growing male Wistar rats. J. Bone Miner. Metab. 2008, 26, 350–357. [Google Scholar] [CrossRef]
- Ríos-Kristjánsson, J.G.; Rizo-Roca, D.; Kristjánsdóttir, K.M.; Núñez-Espinosa, C.A.; Torrella, J.R.; Pagès, T.; Viscor, G. A three-criteria performance score for rats exercising on a running treadmill. PLoS ONE 2019, 14, e0219167. [Google Scholar] [CrossRef]
- Bu, S.; Chen, Y.; Wang, S.; Zhang, F.; Ji, G. Treadmill training regulates β-catenin signaling through phosphorylation of GSK-3β in lumbar vertebrae of ovariectomized rats. Eur. J. Appl. Physiol. 2012, 112, 3295–3304. [Google Scholar] [CrossRef] [PubMed]
- David, V.; Martin, A.; Lafage-Proust, M.-H.; Malaval, L.; Peyroche, S.; Jones, D.B.; Vico, L.; Guignandon, A. Mechanical loading down-regulates peroxisome proliferator-activated receptor gamma in bone marrow stromal cells and favors osteoblastogenesis at the expense of adipogenesis. Endocrinology 2007, 148, 2553–2562. [Google Scholar] [CrossRef] [PubMed]
- Snyder, K.L.; Kram, R.; Gottschall, J.S. The role of elastic energy storage and recovery in downhill and uphill running. J. Exp. Biol. 2012, 215, 2283–2287. [Google Scholar] [CrossRef] [Green Version]
- Davicco, M.J.; Horcajada-Molteni, M.N.; Gaumet-Meunier, N.; Lebecque, P.; Coxam, V.; Barlet, J.P. Endurance training and bone metabolism in middle-aged rats. Mech. Ageing Dev. 1999, 109, 83–96. [Google Scholar] [CrossRef]
- Huang, T.H.; Yang, R.S.; Hsieh, S.S.; Liu, S.H. Effects of caffeine and exercise on the development of bone: A densitometric and histomorphometric study in young Wistar rats. Bone 2002, 30, 293–299. [Google Scholar] [CrossRef]
- Joo, Y.-I.; Sone, T.; Fukunaga, M.; Lim, S.-G.; Onodera, S. Effects of endurance exercise on three-dimensional trabecular bone microarchitecture in young growing rats. Bone 2003, 33, 485–493. [Google Scholar] [CrossRef]
- Simões, P.A.; Zamarioli, A.; Blóes, P.; Mazzocato, F.C.; Pereira, L.H.A.; Volpon, J.B.; Shimano, A.C. Effect of treadmill exercise on lumbar vertebrae in ovariectomized rats: Anthropometrical and mechanical analyses. Acta Bioeng. Biomech. 2008, 10, 39–41. [Google Scholar]
- Takeda, S.; Kobayashi, Y.; Park, J.-H.; Ezawa, I.; Omi, N. Effect of different intake levels of dietary protein and physical exercise on bone mineral density and bone strength in growing male rats. J. Nutr. Sci. Vitaminol. 2012, 58, 240–246. [Google Scholar] [CrossRef] [Green Version]
- Hagihara, Y.; Fukuda, S.; Goto, S.; Iida, H.; Yamazaki, M.; Moriya, H. How many days per week should rats undergo running exercise to increase BMD? J. Bone Miner. Metab. 2005, 23, 289–294. [Google Scholar] [CrossRef]
- Chen, X.; Aoki, H.; Fukui, Y. Effect of exercise on the bone strength, bone mineral density, and metal content in rat femurs. Biomed. Mater. Eng. 2004, 14, 53–59. [Google Scholar]
- Hagihara, Y.; Nakajima, A.; Fukuda, S.; Goto, S.; Iida, H.; Yamazaki, M. Running exercise for short duration increases bone mineral density of loaded long bones in young growing rats. Tohoku J. Exp. Med. 2009, 219, 139–143. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Pajamäki, I.; Kannus, P.; Vuohelainen, T.; Sievänen, H.; Tuukkanen, J.; Järvinen, M.; Järvinen, T.L.N. The bone gain induced by exercise in puberty is not preserved through a virtually life-long deconditioning: A randomized controlled experimental study in male rats. J. Bone Miner. Res. 2003, 18, 544–552. [Google Scholar] [CrossRef] [PubMed]
- Horcajada, M.; Coxam, V.; Davicco, M.; Gaumet, N.; Pastoureau, P.; Leterrier, C.; Culioli, J.; Barlet, J. Influence of treadmill running on femoral bone in young orchidectomized rats. J. Appl. Physiol. 1997, 83, 129–133. [Google Scholar] [CrossRef]
- Yeh, J.K.; Aloia, J.F.; Tierney, J.M.; Sprintz, S. Effect of treadmill exercise on vertebral and tibial bone mineral content and bone mineral density in the aged adult rat: Determined by dual energy X-ray absorptiometry. Calcif. Tissue Int. 1993, 52, 234–238. [Google Scholar] [CrossRef] [PubMed]
- Yao, Z.; Lafage-Proust, M.-H.; Plouët, J.; Bloomfield, S.; Alexandre, C.; Vico, L. Increase of both angiogenesis and bone mass in response to exercise depends on VEGF. J. Bone Miner. Res. 2004, 19, 1471–1480. [Google Scholar] [CrossRef]
- Franz, J.R.; Lyddon, N.E.; Kram, R. Mechanical work performed by the individual legs during uphill and downhill walking. J. Biomech. 2012, 45, 257–262. [Google Scholar] [CrossRef] [Green Version]
- Gottschall, J.S.; Kram, R. Ground reaction forces during downhill and uphill running. J. Biomech. 2005, 38, 445–452. [Google Scholar] [CrossRef]
- Snyder, K.L.; Farley, C.T. Energetically optimal stride frequency in running: The effects of incline and decline. J. Exp. Biol. 2011, 214, 2089–2095. [Google Scholar] [CrossRef] [Green Version]
- Lee, D.V. Effects of grade and mass distribution on the mechanics of trotting in dogs. J. Exp. Biol. 2011, 214, 402–411. [Google Scholar] [CrossRef] [Green Version]
- Hawkins, S.A.; Schroeder, E.T.; Wiswell, R.A.; Jaque, S.V.; Marcell, T.J.; Costa, K. Eccentric muscle action increases site-specific osteogenic response. Med. Sci. Sports Exerc. 1999, 31, 1287–1292. [Google Scholar] [CrossRef]
- Brotto, M.; Johnson, M.L. Endocrine Crosstalk Between Muscle and Bone. Curr. Osteoporos. Rep. 2014, 12, 135–141. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Maurel, D.B.; Boisseau, N.; Pallu, S.; Rochefort, G.Y.; Benhamou, C.-L.; Jaffre, C. Regular exercise limits alcohol effects on trabecular, cortical thickness and porosity, and osteocyte apoptosis in the rat. Jt. Bone Spine 2013, 80, 492–498. [Google Scholar] [CrossRef] [PubMed]
- Robling, A.G.; Burr, D.B.; Turner, C.H. Partitioning a daily mechanical stimulus into discrete loading bouts improves the osteogenic response to loading. J. Bone Miner. Res. 2000, 15, 1596–1602. [Google Scholar] [CrossRef] [PubMed]
- Holy, X.; Zérath, E. Bone mass increases in less than 4 wk of voluntary exercising in growing rats. Med. Sci. Sports Exerc. 2000, 32, 1562–1569. [Google Scholar] [CrossRef]
- Chang, T.-K.; Huang, C.-H.; Huang, C.-H.; Chen, H.-C.; Cheng, C.-K. The influence of long-term treadmill exercise on bone mass and articular cartilage in ovariectomized rats. BMC Musculoskelet. Disord. 2010, 11, 185. [Google Scholar] [CrossRef] [Green Version]
- Horcajada-Molteni, M.N.; Davicco, M.J.; Collignon, H.; Lebecque, P.; Coxam, V.; Barlet, J.P. Does endurance running before orchidectomy prevent osteopenia in rats? Eur. J. Appl. Physiol. Occup. Physiol. 1999, 80, 344–352. [Google Scholar] [CrossRef]
- Horcajada-Molteni, M.N.; Davicco, M.J.; Coxam, V.; Lebecque, P.; Dominguez, B.; Ritz, P.; Culioli, J.; Barlet, J.P. Treadmill running starting 3 months after orchidectomy restores femoral bone mass in rats. Eur. J. Appl. Physiol. Occup. Physiol. 1999, 79, 251–259. [Google Scholar] [CrossRef]
- Thomsen, J.S.; Skalicky, M.; Viidik, A. Influence of physical exercise and food restriction on the biomechanical properties of the femur of ageing male rats. Gerontology 2008, 54, 32–39. [Google Scholar] [CrossRef]
- Notomi, T.; Okazaki, Y.; Okimoto, N.; Saitoh, S.; Nakamura, T.; Suzuki, M. A comparison of resistance and aerobic training for mass, strength and turnover of bone in growing rats. Eur. J. Appl. Physiol. 2000, 83, 469–474. [Google Scholar] [CrossRef]
- Iwamoto, J.; Shimamura, C.; Takeda, T.; Abe, H.; Ichimura, S.; Sato, Y.; Toyama, Y. Effects of treadmill exercise on bone mass, bone metabolism, and calciotropic hormones in young growing rats. J. Bone Miner. Metab. 2004, 22, 26–31. [Google Scholar] [CrossRef]
- Mathey, J.; Horcajada-Molteni, M.-N.; Chanteranne, B.; Picherit, C.; Puel, C.; Lebecque, P.; Cubizoles, C.; Davicco, M.-J.; Coxam, V.; Barlet, J.-P. Bone mass in obese diabetic Zucker rats: Influence of treadmill running. Calcif. Tissue Int. 2002, 70, 305–311. [Google Scholar] [CrossRef] [PubMed]
- Sipos, W.; Rauner, M.; Skalicky, M.; Viidik, A.; Hofbauer, G.; Schett, G.; Redlich, K.; Lang, S.; Pietschmann, P. Running has a negative effect on bone metabolism and proinflammatory status in male aged rats. Exp. Gerontol. 2008, 43, 578–583. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yingling, V.R.; Davies, S.; Silva, M.J. The effects of repetitive physiologic loading on bone turnover and mechanical properties in adult female and male rats. Calcif. Tissue Int. 2001, 68, 235–239. [Google Scholar] [CrossRef] [PubMed]
- Swift, S.N.; Baek, K.; Swift, J.M.; Bloomfield, S.A. Restriction of dietary energy intake has a greater impact on bone integrity than does restriction of calcium in exercising female rats. J. Nutr. 2012, 142, 1038–1045. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Seco, C.; Revilla, M.; Hernández, E.R.; Gervás, J.; González-Riola, J.; Villa, L.F.; Rico, H. Effects of zinc supplementation on vertebral and femoral bone mass in rats on strenuous treadmill training exercise. J. Bone Miner. Res. 1998, 13, 508–512. [Google Scholar] [CrossRef]
- Rico, H.; Gervas, J.J.; Hernández, E.R.; Seco, C.; Villa, L.F.; Revilla, M.; Sanchez-Atrio, A. Effects of alprazolam supplementation on vertebral and femoral bone mass in rats on strenuous treadmill training exercise. Calcif. Tissue Int. 1999, 65, 139–142. [Google Scholar] [CrossRef] [PubMed]
- Yeh, J.K.; Aloia, J.F.; Chen, M.M.; Tierney, J.M.; Sprintz, S. Influence of exercise on cancellous bone of the aged female rat. J. Bone Miner. Res. 1993, 8, 1117–1125. [Google Scholar] [CrossRef] [PubMed]
- Chen, Y.; Wang, S.; Bu, S.; Wang, Y.; Duan, Y.; Yang, S. Treadmill training prevents bone loss by inhibition of PPARγ expression but not promoting of Runx2 expression in ovariectomized rats. Eur. J. Appl. Physiol. 2011, 111, 1759–1767. [Google Scholar] [CrossRef] [PubMed]
- Iwamoto, J.; Yeh, J.K.; Aloia, J.F. Differential effect of treadmill exercise on three cancellous bone sites in the young growing rat. Bone 1999, 24, 163–169. [Google Scholar] [CrossRef]
- Maurel, D.B.; Boisseau, N.; Ingrand, I.; Dolleans, E.; Benhamou, C.-L.; Jaffre, C. Combined effects of chronic alcohol consumption and physical activity on bone health: Study in a rat model. Eur. J. Appl. Physiol. 2011, 111, 2931–2940. [Google Scholar] [CrossRef]
- Reed, A.H.; McCarty, H.L.; Evans, G.L.; Turner, R.T.; Westerlind, K.C. The effects of chronic alcohol consumption and exercise on the skeleton of adult male rats. Alcohol. Clin. Exp. Res. 2002, 26, 1269–1274. [Google Scholar] [CrossRef] [PubMed]
- Peng, Z.Q.; Väänänen, H.K.; Tuukkanen, J. Ovariectomy-induced bone loss can be affected by different intensities of treadmill running exercise in rats. Calcif. Tissue Int. 1997, 60, 441–448. [Google Scholar] [CrossRef] [PubMed]
- Karatosun, H.; Erdogan, A.; Yildiz, M.; Akgun, C.; Cetin, C. Effects of swimming training and free mobilization on bone mineral densities of rats with the immobilization-induced osteopenia. Saudi Med. J. 2006, 27, 312–316. [Google Scholar] [PubMed]
- Newhall, K.M.; Rodnick, K.J.; van der Meulen, M.C.; Carter, D.R.; Marcus, R. Effects of voluntary exercise on bone mineral content in rats. J. Bone Miner. Res. 1991, 6, 289–296. [Google Scholar] [CrossRef]
- Iwamoto, J.; Takeda, T.; Ichimura, S. Effects of exercise on bone mineral density in mature osteopenic rats. J. Bone Miner. Res. 1998, 13, 1308–1317. [Google Scholar] [CrossRef]
- Nordsletten, L.; Kaastad, T.S.; Madsen, J.E.; Reikerås, O.; Ovstebø, R.; Strømme, J.H.; Falch, J. The development of femoral osteopenia in ovariectomized rats is not reduced by high intensity treadmill training: A mechanical and densitometric study. Calcif. Tissue Int. 1994, 55, 436–442. [Google Scholar] [CrossRef]
- Peng, Z.; Tuukkanen, J.; Väänänen, H.K. Exercise can provide protection against bone loss and prevent the decrease in mechanical strength of femoral neck in ovariectomized rats. J. Bone Miner. Res. 1994, 9, 1559–1564. [Google Scholar] [CrossRef]
- Ocarino, N.M.; Marubayashi, U.; Cardoso, T.G.; Guimaraes, C.V.; Silva, A.E.; Torres, R.C.; Serakides, R. Physical activity in osteopenia treatment improved the mass of bones directly and indirectly submitted to mechanical impact. J. Musculoskelet. Neuronal Interact. 2007, 7, 84–93. [Google Scholar]
- Tamaki, H.; Akamine, T.; Goshi, N.; Kurata, H.; Sakou, T. Effects of exercise training and etidronate treatment on bone mineral density and trabecular bone in ovariectomized rats. Bone 1998, 23, 147–153. [Google Scholar] [CrossRef]
- Shimano, M.M.; Volpon, J.B. Biomechanics and structural adaptations of the rat femur after hindlimb suspension and treadmill running. Braz. J. Med. Biol. Res. 2009, 42, 330–338. [Google Scholar] [CrossRef] [Green Version]
- Lecoq, B.; Potrel-Burgot, C.; Granier, P.; Sabatier, J.-P.; Marcelli, C. Comparison of bone loss induced in female rats by hindlimb unloading, ovariectomy, or both. Joint Bone Spine 2006, 73, 189–195. [Google Scholar] [CrossRef] [PubMed]
- Vicente, W.S.; dos Reis, L.M.; Graciolli, R.G.; Graciolli, F.G.; Dominguez, W.V.; Wang, C.C.; Fonseca, T.L.; Velosa, A.P.; Roschel, H.; Teodoro, W.R.; et al. Bone Plasticity in Response to Exercise Is Sex-Dependent in Rats. PLoS ONE 2013, 8, e64725. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Li, K.C.; Zernicke, R.F.; Barnard, R.J.; Li, A.F. Differential response of rat limb bones to strenuous exercise. J. Appl. Physiol. 1991, 70, 554–560. [Google Scholar] [CrossRef] [PubMed]
- Barengolts, E.I.; Lathon, P.V.; Curry, D.J.; Kukreja, S.C. Effects of endurance exercise on bone histomorphometric parameters in intact and ovariectomized rats. Bone Miner. 1994, 26, 133–140. [Google Scholar] [CrossRef]
- Nam, J.; Perera, P.; Rath, B.; Agarwal, S. Dynamic regulation of bone morphogenetic proteins in engineered osteochondral constructs by biomechanical stimulation. Tissue Eng. Part A 2013, 19, 783–792. [Google Scholar] [CrossRef] [Green Version]
- Oxlund, H.; Andersen, N.B.; Ortoft, G.; Orskov, H.; Andreassen, T.T. Growth hormone and mild exercise in combination markedly enhance cortical bone formation and strength in old rats. Endocrinology 1998, 139, 1899–1904. [Google Scholar] [CrossRef]
- Yeh, J.K.; Liu, C.C.; Aloia, J.F. Additive effect of treadmill exercise and 17 beta-estradiol replacement on prevention of tibial bone loss in adult ovariectomized rat. J. Bone Miner. Res. 1993, 8, 677–683. [Google Scholar] [CrossRef]
- Liu, M.; Zhong, C.; He, R.-X.; Chen, L.-F. Icariin associated with exercise therapy is an effective treatment for postmenopausal osteoporosis. Chin. Med. J. 2012, 125, 1784–1789. [Google Scholar]
- Bourrin, S.; Palle, S.; Genty, C.; Alexandre, C. Physical exercise during remobilization restores a normal bone trabecular network after tail suspension-induced osteopenia in young rats. J. Bone Miner. Res. 1995, 10, 820–828. [Google Scholar] [CrossRef]
- Bégot, L.; Collombet, J.; Renault, S.; Butigieg, X.; Andre, C.; Zerath, E.; Holy, X. Effects of high-phosphorus and/or low-calcium diets on bone tissue in trained male rats. Med. Sci. Sports Exerc. 2011, 43, 54–63. [Google Scholar] [CrossRef]
- Dimarco, N.M.; Dart, L.; Sanborn, C.B. Modified activity-stress paradigm in an animal model of the female athlete triad. J. Appl. Physiol. 2007, 103, 1469–1478. [Google Scholar] [CrossRef] [Green Version]
- Hydock, D.S.; Iwaniec, U.T.; Turner, R.T.; Lien, C.-Y.; Jensen, B.T.; Parry, T.L.; Schneider, C.M.; Hayward, R. Effects of voluntary wheel running on goserelin acetate-induced bone degeneration. Pathophysiology 2008, 15, 253–259. [Google Scholar] [CrossRef] [PubMed]
- Hertrampf, T.; Degen, G.H.; Kaid, A.A.; Laudenbach-Leschowsky, U.; Seibel, J.; Di Virgilio, A.L.; Diel, P. Combined effects of physical activity, dietary isoflavones and 17beta-estradiol on movement drive, body weight and bone mineral density in ovariectomized female rats. Planta Med. 2006, 72, 484–487. [Google Scholar] [CrossRef] [PubMed]
- Shiga, K.; Hara, H.; Okano, G.; Ito, M.; Minami, A.; Tomita, F. Ingestion of difructose anhydride III and voluntary running exercise independently increase femoral and tibial bone mineral density and bone strength with increasing calcium absorption in rats. J. Nutr. 2003, 133, 4207–4211. [Google Scholar] [CrossRef]
- Omi, N.; Morikawa, N.; Ezawa, I. The effect of voluntary exercise on bone mineral density and skeletal muscles in the rat model at ovariectomized and sham stages. Bone Min. 1994, 24, 211–222. [Google Scholar] [CrossRef]
- Banu, M.J.; Orhii, P.B.; Mejia, W.; McCarter, R.J.; Mosekilde, L.; Thomsen, J.S.; Kalu, D.N. Analysis of the effects of growth hormone, voluntary exercise, and food restriction on diaphyseal bone in female F344 rats. Bone 1999, 25, 469–480. [Google Scholar] [CrossRef]
- Fonseca, H.; Moreira-Gonçalves, D.; Esteves, J.L.S.; Viriato, N.; Vaz, M.; Mota, M.P.; Duarte, J.A. Voluntary exercise has long-term in vivo protective effects on osteocyte viability and bone strength following ovariectomy. Calcif. Tissue Int. 2011, 88, 443–454. [Google Scholar] [CrossRef] [PubMed]
- Lertsinthai, P.; Charoenphandhu, J.; Suntornsaratoon, P.; Krishnamra, N.; Charoenphandhu, N. Voluntary wheel running mitigates the stress-induced bone loss in ovariectomized rats. J. Bone Min. Metab. 2015, 33, 261–269. [Google Scholar] [CrossRef] [PubMed]
- Goulet, G.C.; Halonen, N.R.; Koch, L.G.; Britton, S.L.; Zernicke, R.F.; Kozloff, K.M. Osteoblast response to ovariectomy is enhanced in intrinsically high aerobic-capacity rats. Calcif. Tissue Int. 2011, 88, 325–335. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Guadalupe-Grau, A.; Fuentes, T.; Guerra, B.; Calbet, J.A.L. Exercise and bone mass in adults. Sports Med. 2009, 39, 439–468. [Google Scholar] [CrossRef] [PubMed]
- Courteix, D.; Lespessailles, E.; Peres, S.L.; Obert, P.; Germain, P.; Benhamou, C.L. Effect of physical training on bone mineral density in prepubertal girls: A comparative study between impact-loading and non-impact-loading sports. Osteoporos. Int. 1998, 8, 152–158. [Google Scholar] [CrossRef]
- Simkin, A.; Leichter, I.; Swissa, A.; Samueloff, S. The effect of swimming activity on bone architecture in growing rats. J. Biomech. 1989, 22, 845–851. [Google Scholar] [CrossRef]
- Swissa-Sivan, A.; Azoury, R.; Statter, M.; Leichter, I.; Nyska, A.; Nyska, M.; Menczel, J.; Samueloff, S. The effect of swimming on bone modeling and composition in young adult rats. Calcif. Tissue Int. 1990, 47, 173–177. [Google Scholar] [CrossRef] [PubMed]
- Huang, T.H.; Lin, S.C.; Chang, F.L.; Hsieh, S.S.; Liu, S.H.; Yang, R.S. Effects of different exercise modes on mineralization, structure, and biomechanical properties of growing bone. J. Appl. Physiol. 2003, 95, 300–307. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Huang, T.-H.; Hsieh, S.S.; Liu, S.-H.; Chang, F.-L.; Lin, S.-C.; Yang, R.-S. Swimming training increases the post-yield energy of bone in young male rats. Calcif. Tissue Int. 2010, 86, 142–153. [Google Scholar] [CrossRef] [PubMed]
- Gomes, R.J.; de Mello, M.A.R.; Caetano, F.H.; Sibuya, C.Y.; Anaruma, C.A.; Rogatto, G.P.; Pauli, J.R.; Luciano, E. Effects of swimming training on bone mass and the GH/IGF-1 axis in diabetic rats. Growth Horm. IGF Res. 2006, 16, 326–331. [Google Scholar] [CrossRef] [PubMed]
- Bourrin, S.; Ghaemmaghami, F.; Vico, L.; Chappard, D.; Gharib, C.; Alexandre, C. Effect of a five-week swimming program on rat bone: A histomorphometric study. Calcif. Tissue Int. 1992, 51, 137–142. [Google Scholar] [CrossRef]
- Swissa-Sivan, A.; Statter, M.; Brooks, G.A.; Azevedo, J.; Viguie, C.; Azoury, R.; Greenfield, C.; Oman, S.; Leichter, I.; Zinker, B.A. Effect of swimming on prednisolone-induced osteoporosis in elderly rats. J. Bone Miner. Res. 1992, 7, 161–169. [Google Scholar] [CrossRef]
- Rochdi, A.; Foucat, L.; Renou, J.P. Effect of thermal denaturation on water-collagen interactions: NMR relaxation and differential scanning calorimetry analysis. Biopolymers 1999, 50, 690–696. [Google Scholar] [CrossRef]
- Pelker, R.R.; Friedlaender, G.E.; Markham, T.C.; Panjabi, M.M.; Moen, C.J. Effects of freezing and freeze-drying on the biomechanical properties of rat bone. J. Orthop. Res. 1984, 1, 405–411. [Google Scholar] [CrossRef]
- Yeni, Y.N.; Brown, C.U.; Norman, T.L. Influence of bone composition and apparent density on fracture toughness of the human femur and tibia. Bone 1998, 22, 79–84. [Google Scholar] [CrossRef]
- Hart, K.J.; Shaw, J.M.; Vajda, E.; Hegsted, M.; Miller, S.C. Swim-trained rats have greater bone mass, density, strength, and dynamics. J. Appl. Physiol. 2001, 91, 1663–1668. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Falcai, M.J.; Zamarioli, A.; Okubo, R.; de Paula, F.J.A.; Volpon, J.B. The osteogenic effects of swimming, jumping, and vibration on the protection of bone quality from disuse bone loss. Scand. J. Med. Sci Sports 2015, 25, 390–397. [Google Scholar] [CrossRef] [PubMed]
- Tezval, M.; Biblis, M.; Sehmisch, S.; Schmelz, U.; Kolios, L.; Rack, T.; Stuermer, K.M.; Stuermer, E.K. Improvement of femoral bone quality after low-magnitude, high-frequency mechanical stimulation in the ovariectomized rat as an osteopenia model. Calcif. Tissue Int. 2011, 88, 33–40. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Ubago-Guisado, E.; Mata, E.; Sánchez-Sánchez, J.; Plaza-Carmona, M.; Martín-García, M.; Gallardo, L. Influence of different sports on fat mass and lean mass in growing girls. J. Sport Health Sci. 2017, 6, 213–218. [Google Scholar] [CrossRef] [Green Version]
- Aikawa, Y.; Wakasugi, Y.; Narukawa, T.; Yamashita, T.; Sasai, N.; Umemura, Y.; Omi, N.; Ohtsuki, M. Jump Exercise and Food Restriction on Bone Parameters in Young Female Rats. Calcif. Tissue Int. 2019, 105, 557–566. [Google Scholar] [CrossRef]
- Okubo, R.; Sanada, L.S.; Castania, V.A.; Louzada, M.J.Q.; de Paula, F.J.A.; Maffulli, N.; Shimano, A.C. Jumping exercise preserves bone mineral density and mechanical properties in osteopenic ovariectomized rats even following established osteopenia. Osteoporos. Int. 2017, 28, 1461–1471. [Google Scholar] [CrossRef]
- Cavalié, H.; Horcajada-Molteni, M.N.; Lebecque, P.; Davicco, M.J.; Coxam, V.; Lac, G.; Barlet, J.P. Progressive isometric force training and bone mass in rats. J. Musculoskelet. Neuronal Interact. 2003, 3, 47–52. [Google Scholar]
- Notomi, T.; Okimoto, N.; Okazaki, Y.; Tanaka, Y.; Nakamura, T.; Suzuki, M. Effects of tower climbing exercise on bone mass, strength, and turnover in growing rats. J. Bone Miner. Res. 2001, 16, 166–174. [Google Scholar] [CrossRef]
- Umemura, Y.; Ishiko, T.; Yamauchi, T.; Kurono, M.; Mashiko, S. Five jumps per day increase bone mass and breaking force in rats. J. Bone Miner. Res. 1997, 12, 1480–1485. [Google Scholar] [CrossRef]
- Umemura, Y.; Sogo, N.; Honda, A. Effects of intervals between jumps or bouts on osteogenic response to loading. J. Appl. Physiol. 2002, 93, 1345–1348. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Swift, J.M.; Gasier, H.G.; Swift, S.N.; Wiggs, M.P.; Hogan, H.A.; Fluckey, J.D.; Bloomfield, S.A. Increased training loads do not magnify cancellous bone gains with rodent jump resistance exercise. J. Appl. Physiol. 2010, 109, 1600–1607. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Nagasawa, S.; Honda, A.; Sogo, N.; Umemura, Y. Effects of low-repetition jump exercise on osteogenic response in rats. J. Bone Miner. Metab. 2008, 26, 226–230. [Google Scholar] [CrossRef]
- Umemura, Y.; Ishiko, T.; Tsujimoto, H.; Miura, H.; Mokushi, N.; Suzuki, H. Effects of jump training on bone hypertrophy in young and old rats. Int. J. Sports Med. 1995, 16, 364–367. [Google Scholar] [CrossRef]
- Umemura, Y.; Nagasawa, S.; Honda, A.; Singh, R. High-impact exercise frequency per week or day for osteogenic response in rats. J. Bone Miner. Metab. 2008, 26, 456–460. [Google Scholar] [CrossRef] [PubMed]
- Welch, J.M.; Turner, C.H.; Devareddy, L.; Arjmandi, B.H.; Weaver, C.M. High impact exercise is more beneficial than dietary calcium for building bone strength in the growing rat skeleton. Bone 2008, 42, 660–668. [Google Scholar] [CrossRef]
- Lin, H.-S.; Huang, T.-H.; Wang, H.-S.; Mao, S.-W.; Tai, Y.-S.; Chiu, H.-T.; Cheng, K.-Y.B.; Yang, R.-S. Short-term free-fall landing causes reduced bone size and bending energy in femora of growing rats. J. Sports Sci. Med. 2013, 12, 1–9. [Google Scholar] [PubMed]
- Notomi, T.; Okazaki, Y.; Okimoto, N.; Tanaka, Y.; Nakamura, T.; Suzuki, M. Effects of tower climbing exercise on bone mass, strength, and turnover in orchidectomized growing rats. J. Appl. Physiol. 2002, 93, 1152–1158. [Google Scholar] [CrossRef] [Green Version]
- Ju, Y.-I.; Sone, T.; Ohnaru, K.; Tanaka, K.; Yamaguchi, H.; Fukunaga, M. Effects of different types of jump impact on trabecular bone mass and microarchitecture in growing rats. PLoS ONE 2014, 9, e107953. [Google Scholar] [CrossRef] [Green Version]
- Honda, A.; Sogo, N.; Nagasawa, S.; Kato, T.; Umemura, Y. Bones benefits gained by jump training are preserved after detraining in young and adult rats. J. Appl. Physiol. 2008, 105, 849–853. [Google Scholar] [CrossRef] [Green Version]
- Rosa, B.V.; Firth, E.C.; Blair, H.T.; Vickers, M.H.; Morel, P.C.H.; Cockrem, J.F. Short-term voluntary exercise in the rat causes bone modeling without initiating a physiological stress response. Am. J. Physiol. Regul. Integr. Comp. Physiol. 2010, 299, R1037–R1043. [Google Scholar] [CrossRef] [PubMed]
- Umemura, Y.; Nagasawa, S.; Sogo, N.; Honda, A. Effects of jump training on bone are preserved after detraining, regardless of estrogen secretion state in rats. J. Appl. Physiol. 2008, 104, 1116–1120. [Google Scholar] [CrossRef] [PubMed]
- Honda, A.; Umemura, Y.; Nagasawa, S. Effect of high-impact and low-repetition training on bones in ovariectomized rats. J. Bone Miner. Res. 2001, 16, 1688–1693. [Google Scholar] [CrossRef] [PubMed]
- Honda, A.; Sogo, N.; Nagasawa, S.; Shimizu, T.; Umemura, Y. High-impact exercise strengthens bone in osteopenic ovariectomized rats with the same outcome as Sham rats. J. Appl. Physiol. 2003, 95, 1032–1037. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Yanagihara, G.R.; Paiva, A.G.; Gasparini, G.A.; Macedo, A.P.; Frighetto, P.D.; Volpon, J.B.; Shimano, A.C. High-impact exercise in rats prior to and during suspension can prevent bone loss. Braz. J. Med. Biol. Res. 2016, 49. [Google Scholar] [CrossRef] [Green Version]
- Notomi, T.; Lee, S.J.; Okimoto, N.; Okazaki, Y.; Takamoto, T.; Nakamura, T.; Suzuki, M. Effects of resistance exercise training on mass, strength, and turnover of bone in growing rats. Eur. J. Appl. Physiol. 2000, 82, 268–274. [Google Scholar] [CrossRef]
- Macias, B.R.; Swift, J.M.; Nilsson, M.I.; Hogan, H.A.; Bouse, S.D.; Bloomfield, S.A. Simulated resistance training, but not alendronate, increases cortical bone formation and suppresses sclerostin during disuse. J. Appl. Physiol. 2011, 112, 918–925. [Google Scholar] [CrossRef] [Green Version]
- Swift, J.M.; Swift, S.N.; Nilsson, M.I.; Hogan, H.A.; Bouse, S.D.; Bloomfield, S.A. Cancellous bone formation response to simulated resistance training during disuse is blunted by concurrent alendronate treatment. J. Bone Min. Res. 2011, 26, 2140–2150. [Google Scholar] [CrossRef]
- Notomi, T.; Okimoto, N.; Okazaki, Y.; Nakamura, T.; Suzuki, M. Tower climbing exercise started 3 months after ovariectomy recovers bone strength of the femur and lumbar vertebrae in aged osteopenic rats. J. Bone Miner. Res. 2003, 18, 140–149. [Google Scholar] [CrossRef]
- Cardinale, M.; Bosco, C. The use of vibration as an exercise intervention. Exerc. Sport Sci. Rev. 2003, 31, 3–7. [Google Scholar] [CrossRef]
- Judex, S.; Rubin, C.T. Is bone formation induced by high-frequency mechanical signals modulated by muscle activity? J. Musculoskelet. Neuronal Interact. 2010, 10, 3–11. [Google Scholar] [PubMed]
- Ogawa, T.; Vandamme, K.; Zhang, X.; Naert, I.; Possemiers, T.; Chaudhari, A.; Sasaki, K.; Duyck, J. Stimulation of titanium implant osseointegration through high-frequency vibration loading is enhanced when applied at high acceleration. Calcif. Tissue Int. 2014, 95, 467–475. [Google Scholar] [CrossRef] [PubMed] [Green Version]
- Liang, Y.-Q.; Qi, M.-C.; Xu, J.; Xu, J.; Liu, H.-W.; Dong, W.; Li, J.-Y.; Hu, M. Low-magnitude high-frequency loading, by whole-body vibration, accelerates early implant osseointegration in ovariectomized rats. Mol. Med. Rep. 2014, 10, 2835–2842. [Google Scholar] [CrossRef] [Green Version]
- Chatterjee, M.; Hatori, K.; Duyck, J.; Sasaki, K.; Naert, I.; Vandamme, K. High-frequency loading positively impacts titanium implant osseointegration in impaired bone. Osteoporos. Int. 2015, 26, 281–290. [Google Scholar] [CrossRef]
- Hatori, K.; Camargos, G.V.; Chatterjee, M.; Faot, F.; Sasaki, K.; Duyck, J.; Vandamme, K. Single and combined effect of high-frequency loading and bisphosphonate treatment on the bone micro-architecture of ovariectomized rats. Osteoporos. Int. 2015, 26, 303–313. [Google Scholar] [CrossRef] [PubMed]
- Nowak, A.; Łochyński, D.; Pawlak, M.; Romanowski, W.; Krutki, P. High-magnitude whole-body vibration effects on bone resorption in adult rats. Aviat. Space Environ. Med. 2014, 85, 518–521. [Google Scholar] [CrossRef] [PubMed]
- Camargos, G.V.; Bhattacharya, P.; van Lenthe, G.H.; Del Bel Cury, A.A.; Naert, I.; Duyck, J.; Vandamme, K. Mechanical competence of ovariectomy-induced compromised bone after single or combined treatment with high-frequency loading and bisphosphonates. Sci. Rep. 2015, 5, 10795. [Google Scholar] [CrossRef] [Green Version]
- Hoffmann, D.B.; Sehmisch, S.; Hofmann, A.M.; Eimer, C.; Komrakova, M.; Saul, D.; Wassmann, M.; Stürmer, K.M.; Tezval, M. Comparison of parathyroid hormone and strontium ranelate in combination with whole-body vibration in a rat model of osteoporosis. J. Bone Miner. Metab. 2017, 35, 31–39. [Google Scholar] [CrossRef]
- Hoffmann, D.B.; Griesel, M.H.; Brockhusen, B.; Tezval, M.; Komrakova, M.; Menger, B.; Wassmann, M.; Stuermer, K.M.; Sehmisch, S. Effects of 8-Prenylnaringenin and Whole-Body Vibration Therapy on a Rat Model of Osteopenia. J. Nutr. Metab. 2016, 2016, 6893137. [Google Scholar] [CrossRef] [Green Version]
- Komrakova, M.; Hoffmann, D.B.; Nuehnen, V.; Stueber, H.; Wassmann, M.; Wicke, M.; Tezval, M.; Stuermer, K.M.; Sehmisch, S. The Effect of Vibration Treatments Combined with Teriparatide or Strontium Ranelate on Bone Healing and Muscle in Ovariectomized Rats. Calcif. Tissue Int. 2016, 99, 408–422. [Google Scholar] [CrossRef]
- Pasqualini, M.; Lavet, C.; Elbadaoui, M.; Vanden-Bossche, A.; Laroche, N.; Gnyubkin, V.; Vico, L. Skeletal site-specific effects of whole body vibration in mature rats: From deleterious to beneficial frequency-dependent effects. Bone 2013, 55, 69–77. [Google Scholar] [CrossRef] [PubMed]
- Maddalozzo, G.F.; Iwaniec, U.T.; Turner, R.T.; Rosen, C.J.; Widrick, J.J. Whole-body vibration slows the acquisition of fat in mature female rats. Int. J. Obes. (Lond.) 2008, 32, 1348–1354. [Google Scholar] [CrossRef] [Green Version]
- Xie, P.; Tang, Z.; Qing, F.; Chen, X.; Zhu, X.; Fan, Y.; Yang, X.; Zhang, X. Bone mineral density, microarchitectural and mechanical alterations of osteoporotic rat bone under long-term whole-body vibration therapy. J. Mech. Behav. Biomed. Mater. 2016, 53, 341–349. [Google Scholar] [CrossRef] [PubMed]
- Yang, P.; Jia, B.; Ding, C.; Wang, Z.; Qian, A.; Shang, P. Whole-body vibration effects on bone before and after hind-limb unloading in rats. Aviat. Space Environ. Med. 2009, 80, 88–93. [Google Scholar] [CrossRef] [PubMed]
- Gong, H.; Zhang, R.; Gao, J.; Zhang, M.; Liu, B.; Zhang, M.; Zhu, D. Whole body vibration with rest days could improve bone quality of distal femoral metaphysis by regulating trabecular arrangement. Sci. China Life Sci. 2019, 62, 95–103. [Google Scholar] [CrossRef]
- Tsigos, C.; Kyrou, I.; Kassi, E.; Chrousos, G.P. Stress, Endocrine Physiology and Pathophysiology; MDText.com, Inc.: Dartmouth, MA, USA, 2016. [Google Scholar]
- Droste, S.K.; Gesing, A.; Ulbricht, S.; Müller, M.B.; Linthorst, A.C.E.; Reul, J.M.H.M. Effects of long-term voluntary exercise on the mouse hypothalamic-pituitary-adrenocortical axis. Endocrinology 2003, 144, 3012–3023. [Google Scholar] [CrossRef] [PubMed]
- Droste, S.K.; Schweizer, M.C.; Ulbricht, S.; Reul, J.M.H.M. Long-term voluntary exercise and the mouse hypothalamic-pituitary-adrenocortical axis: Impact of concurrent treatment with the antidepressant drug tianeptine. J. Neuroendocr. 2006, 18, 915–925. [Google Scholar] [CrossRef]
- Isales, C.M.; Zaidi, M.; Blair, H.C. ACTH is a novel regulator of bone mass: ACTH and bone mass. Ann. N. Y. Acad. Sci. 2010, 1192, 110–116. [Google Scholar] [CrossRef] [PubMed]
- Elefteriou, F. Impact of the Autonomic Nervous System on the Skeleton. Physiol. Rev. 2018, 98, 1083. [Google Scholar] [CrossRef] [Green Version]
- de Souza, R.L.; Pitsillides, A.A.; Lanyon, L.E.; Skerry, T.M.; Chenu, C. Sympathetic nervous system does not mediate the load-induced cortical new bone formation. J. Bone Miner. Res. 2005, 20, 2159–2168. [Google Scholar] [CrossRef]
- Bassey, E.J.; Ramsdale, S.J. Increase in femoral bone density in young women following high-impact exercise. Osteoporos. Int. 1994, 4, 72–75. [Google Scholar] [CrossRef] [PubMed]
- Gomarasca, M.; Banfi, G.; Lombardi, G. Myokines: The endocrine coupling of skeletal muscle and bone. Adv. Clin. Chem. 2020, 94, 155–218. [Google Scholar] [CrossRef] [PubMed]
- Pagnotti, G.M.; Styner, M.; Uzer, G.; Patel, V.S.; Wright, L.E.; Ness, K.K.; Guise, T.A.; Rubin, J.; Rubin, C.T. Combating osteoporosis and obesity with exercise: Leveraging cell mechanosensitivity. Nat. Rev. Endocrinol. 2019, 15, 339–355. [Google Scholar] [CrossRef] [PubMed]
- Sherk, V.D.; Rosen, C.J. Senescent and apoptotic osteocytes and aging: Exercise to the rescue? Bone 2019, 121, 255–258. [Google Scholar] [CrossRef] [PubMed]
- Wang, Z.; Guo, J. Mechanical induction of BMP-7 in osteocyte blocks glucocorticoid-induced apoptosis through PI3K/AKT/GSK3β pathway. Cell Biochem. Biophys. 2013, 67, 567–574. [Google Scholar] [CrossRef]
- Massimini, M.; Palmieri, C.; De Maria, R.; Romanucci, M.; Malatesta, D.; De Martinis, M.; Maniscalco, L.; Ciccarelli, A.; Ginaldi, L.; Buracco, P.; et al. 17-AAG and Apoptosis, Autophagy, and Mitophagy in Canine Osteosarcoma Cell Lines. Vet. Pathol. 2017, 54, 405–412. [Google Scholar] [CrossRef]

© 2020 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Portier, H.; Benaitreau, D.; Pallu, S. Does Physical Exercise Always Improve Bone Quality in Rats? Life 2020, 10, 217. https://doi.org/10.3390/life10100217
Portier H, Benaitreau D, Pallu S. Does Physical Exercise Always Improve Bone Quality in Rats? Life. 2020; 10(10):217. https://doi.org/10.3390/life10100217
Chicago/Turabian StylePortier, Hugues, Delphine Benaitreau, and Stéphane Pallu. 2020. "Does Physical Exercise Always Improve Bone Quality in Rats?" Life 10, no. 10: 217. https://doi.org/10.3390/life10100217




