Ammonium Sorption from Landfill Leachates Using Natural and Modified Zeolites: Pre-Tests for a Novel Application of the Ion Exchanger Loop Stripping Process
Abstract
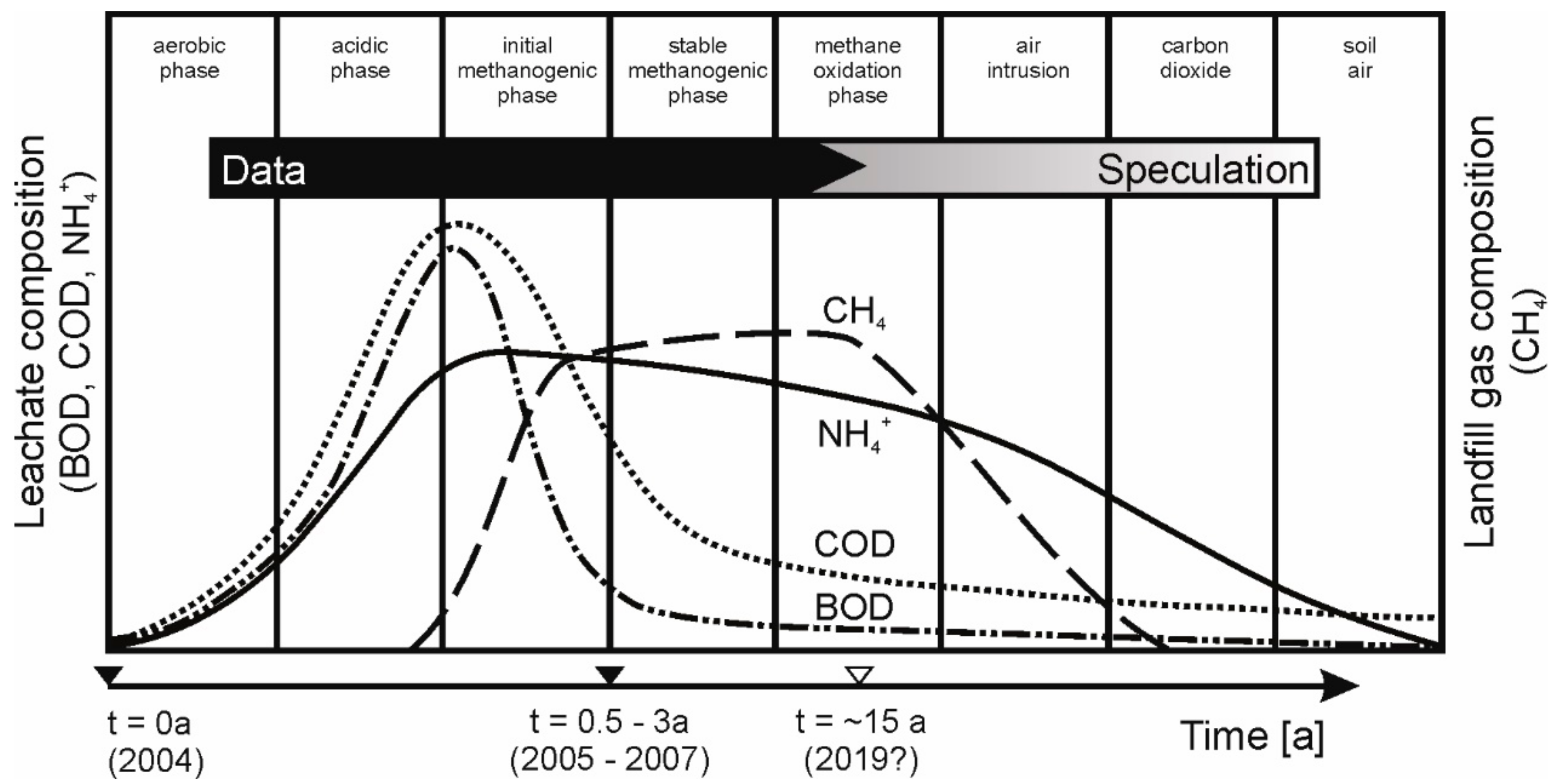
:1. Introduction
2. Materials and Methods
2.1. Materials
2.2. Methods
3. Results
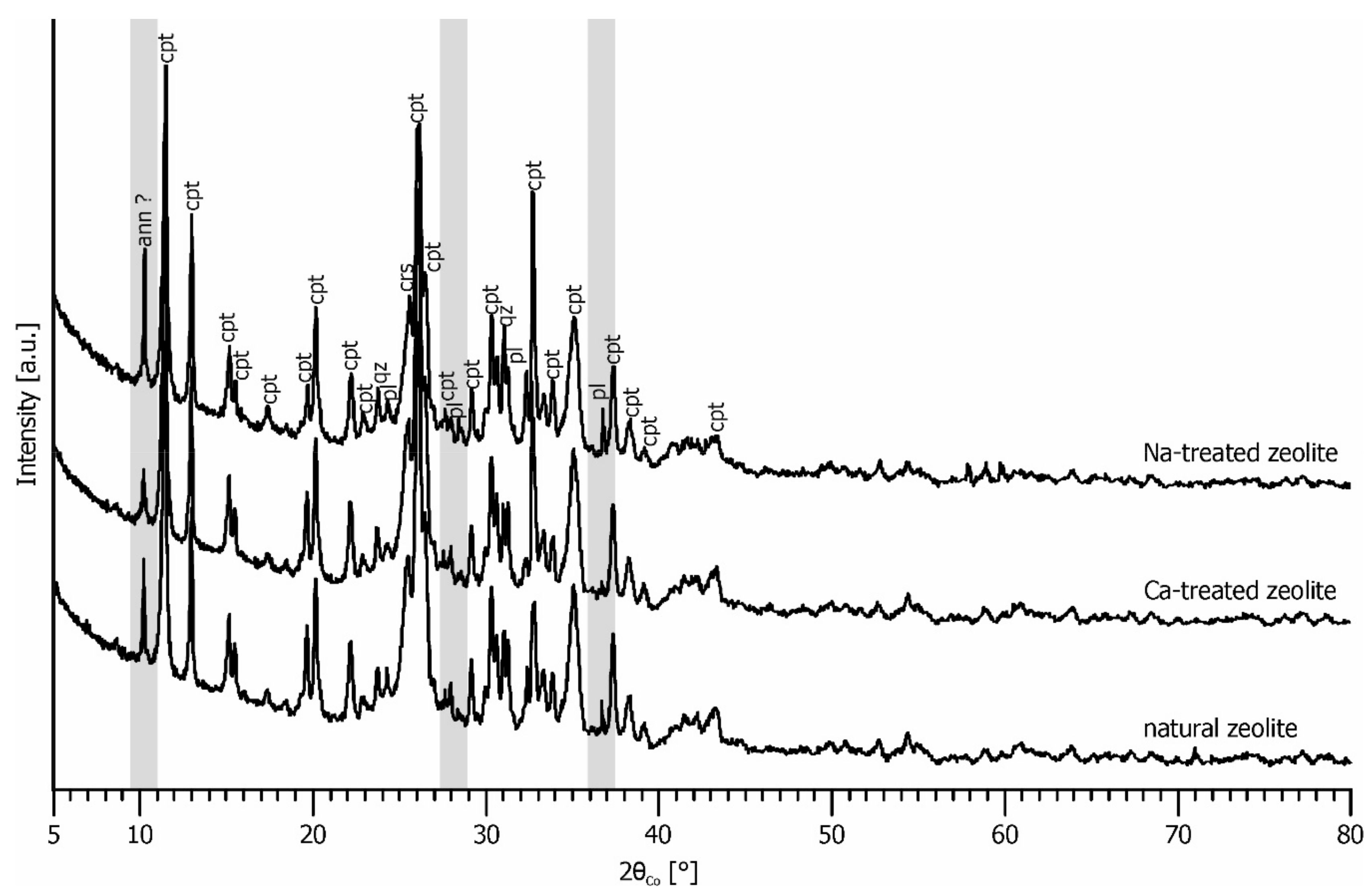
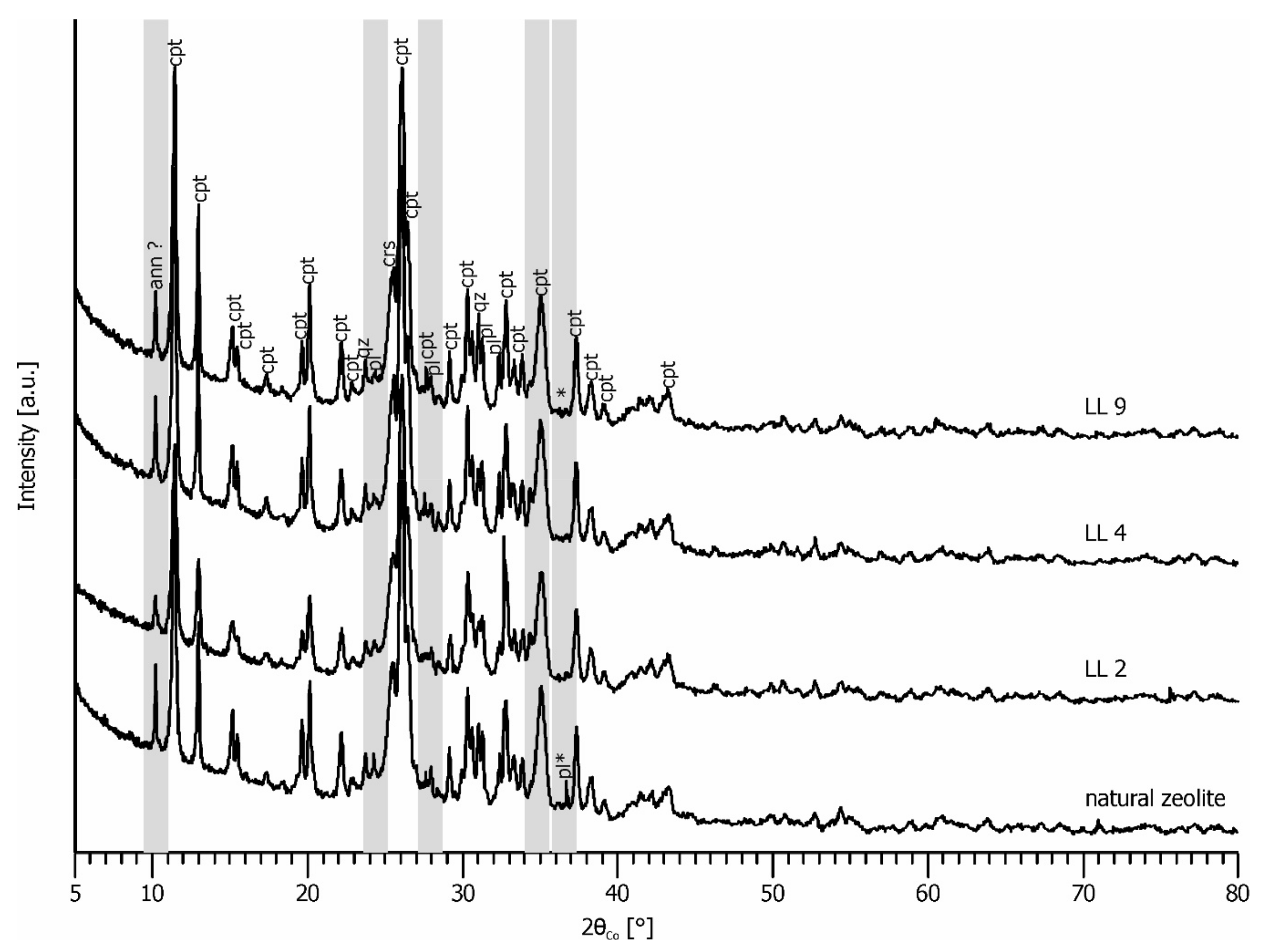
3.1. Zeolite Characterization
3.2. Leachate Characterization
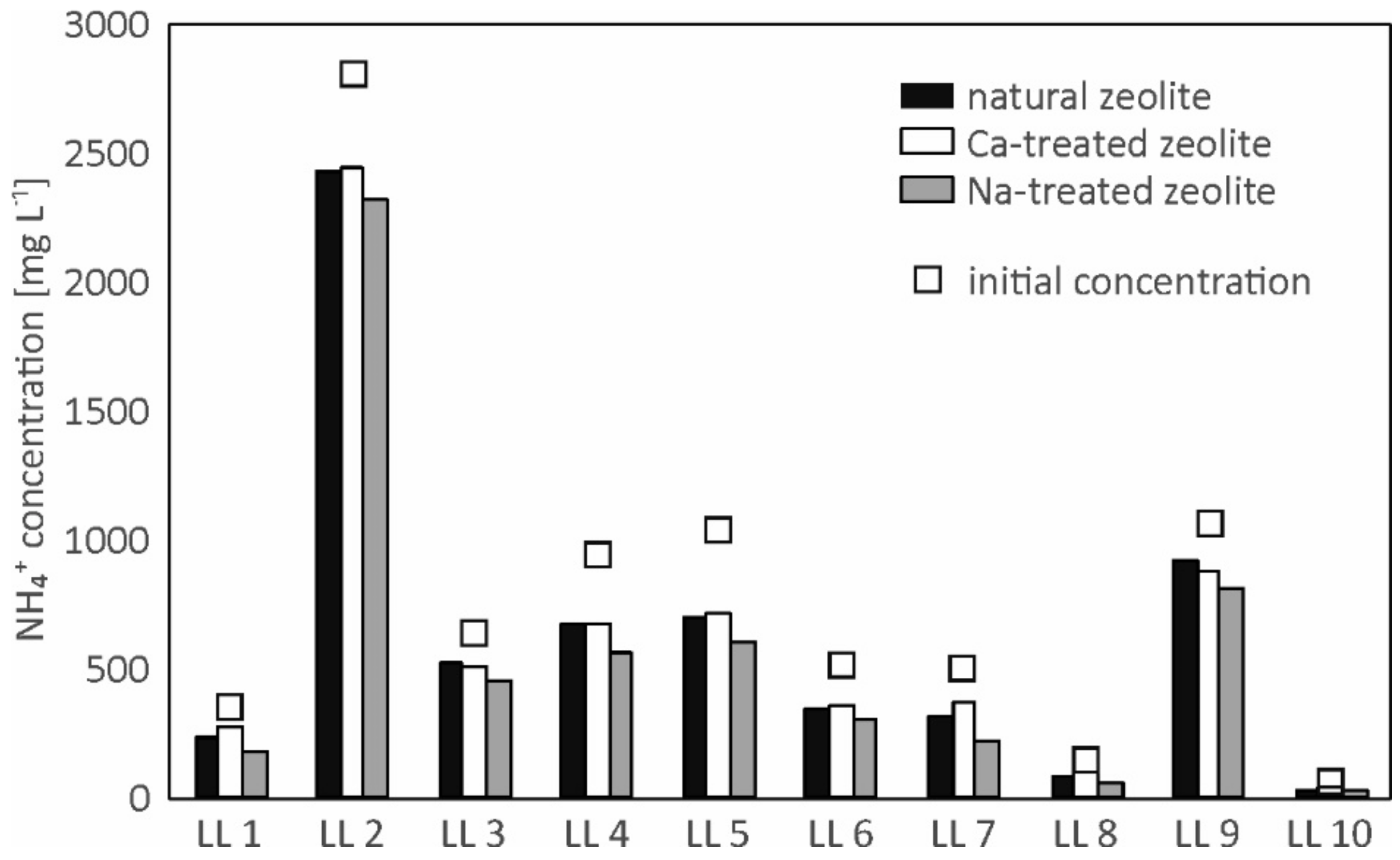
3.3. Sorption Efficiency
3.4. Zeolite Stability
4. Discussion
5. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Kjeldsen, P.; Barlaz, M.; Rooker, A.; Braun, A.; Ledin, A.; Christensen, T. Present and Long-Term Composition of MSW Landfill Leachate: A Review. Crit. Rev. Environ. Sci. Technol. 2002, 32, 297–336. [Google Scholar] [CrossRef]
- Barlaz, M.; Ham, D.; Schaefer, D.; Isaacson, R. Methane production from municipal refuse: A review of enhancement techniques and microbial dynamics. Crit. Rev. Environ. Control 1990, 19, 557–584. [Google Scholar] [CrossRef]
- Christensen, T.; Kjeldsen, P.; Bjerg, P.; Jensen, D.; Christensen, J.; Baun, A.; Albrechtsen, H.J.; Heron, G. Review: Biogeochemistry of landfill leachate plumes. Appl. Geochem. 2001, 16, 659–718. [Google Scholar] [CrossRef]
- Burton, S.; Watson-Craik, I. Ammonia and nitrogen fluxes in landfill sites: Applicability to sustainable landfilling. Waste Manag. Res. 1998, 16, 41–53. [Google Scholar] [CrossRef]
- Bundesminster für Umwelt. Verordnung über die Ablagerung von Abfällen (Deponieverordnung); Bundesminister für Umwelt: Vienna, Austria, 1996. [Google Scholar]
- Schachermayer, E.; Lampert, C. Aufkommen und Behandlung von Deponiesickerwasser—Bestandsaufnahme an ausgewählten österreichischen Deponien; Umweltbundesamt: Vienna, Austria, 2010. [Google Scholar]
- Pesendorfer, S.; Ellersdorfer, M. Nutrient recovery and utilization potential from wastewater in Austria. Minisymposium Verfahrenstechnik 2019, 15, 115. [Google Scholar]
- Robinson, H.A. Review of the Composition of Leachates from Domestic Wastes in Landfill Sites. In The Technical Aspects of Controlled Waste Management; Report for the UK Department of the Environment, Waste Science and Research; Aspinwall & Company, Ltd.: London, UK, 1995. [Google Scholar]
- Ehrig, H.J. Leachate Quality. In Sanitary Landfilling Process Technology and Environmental Impact; Academic Press: London, UK, 1989; pp. 213–229. [Google Scholar]
- Andreottola, G.; Cannas, P.; Cossu, R. Overview on Landfill Leachate Quality; CISA Environmental Sanitary Engineering Centre: Caligary, Italy, 1990. [Google Scholar]
- Bundesminister für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft. Verordnung über die Begrenzung von Sickerwasseremissionen aus Abfalldeponien (AEV Deponiesickerwasser); Bundesminister für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Vienna, Austria, 2003. [Google Scholar]
- Bundesminister für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft. Verordnung über Deponien (Deponieverordnung 2008–DVO 2008); Bundesminister für Land- und Forstwirtschaft, Umwelt und Wasserwirtschaft: Vienna, Austria, 2008. [Google Scholar]
- Laner, D.; Fellner, J.S. Ableitung standortspezifischer Nachsorgeentlassungskriterien: Fallstudien. Depo Tech 2012, 11, 539–544. [Google Scholar]
- Hollauf, B. Ammonsulfat zur selektiven nicht-katalytischen Stickoxidreduktion von Zementwerksabgasen. Ph.D. Thesis, Montanuniversität Leoben, Leoben, Austria, 2010. [Google Scholar]
- Wang, S.; Peng, Y. Review: Natural zeolites as effective adsorbents in water and wastewater treatment. Chem. Eng. J. 2010, 156, 11–24. [Google Scholar] [CrossRef]
- Höllen, D.; Klammer, D.; Letofsky-Papst, I.; Dietzel, M. Hydrothermal Alteration of Diatomite for Removal of Aqueous Cu2+, Pb2+ and Zn2+. Mater. Sci. Eng. B 2012, 10, 523–533. [Google Scholar]
- Höllen, D.; Klammer, D.; Letofsky-Papst, I.; Raab, G.; Dietzel, M. Synthesis of hierarchically structured materials: Microporous diatoms and nanoporous hydroxyaluminosilicate. Nanotechnol. Environ. Eng. 2016, 1, 1–9. [Google Scholar] [CrossRef]
- Stocker, K.; Ellersdorfer, M.; Lehner, M.; Raith, J.R. Characterization and Utilization of Natural Zeolites in Technical Applications. Berg- und Hüttenmännische Monatshefte 2017, 162, 142–147. [Google Scholar] [CrossRef]
- Akgül, M. Enhancement of the anionic dye adsorption capacity of clinoptiloliteby Fe3+ grafting. J. Hazard. Mater. 2014, 267, 1–8. [Google Scholar] [CrossRef] [PubMed]
- Karadag, D.; Tok, S.; Akgul, E.; Turan, M.; Ozturk, M.; Demir, A. Ammonium removal from sanitary landfill leachate using natural Gördes clinoptilolite. J. Hazard. Mater. 2008, 153, 60–66. [Google Scholar] [CrossRef] [PubMed]
- Vocciante, M.; De Folly D’Auris, A.; Finocchi, A.; Tagliabue, M.; Bellettato, M.; Ferrucci, A.; Reverberi, A.P.; Ferro, S. Adsorption of ammonium on clinoptilolite in presence of competing cations: Investigation on groundwater remediation. J. Clean. Prod. 2018, 198, 480–487. [Google Scholar] [CrossRef]
- Ellersdorfer, M. The ion-exchanger–loop-stripping process: Ammonium recovery from sludge liquor using NaCl-treated clinoptilolite and simultaneous air stripping. Water Sci. Technol. 2018, 77, 695–705. [Google Scholar] [CrossRef] [PubMed]
- Lubensky, J.; Ellersdorfer, M.; Stocker, K.; Lechleitner, A. Pilotversuche zum Recycling von Ammonium aus Trübwässern an einer Kläranlage. Recy DepoTech 2018, 14, 729–734. [Google Scholar]
- Stocker, K.; Ellersdorfer, M.; Lechleitner, A.; Lubensky, J.; Raith, J.R. Impact of concentrated acid, base and salt pretreatments on the characteristics of natural clinoptilolite and its ammonium uptake from model solution and real effluents. Microporous Mesoporous Mat. 2019, 288, 109553. [Google Scholar] [CrossRef]
- Castro De Souza, V.; Villarroel-Rocha, J.; Gomes De Araújo, M.J.; Sapag, K.; Pergher, S.B.C. Basic Treatment in Natural Clinoptilolite for Improvement of Physicochemical Properties. Minerals 2018, 8, 595. [Google Scholar] [CrossRef]
- Vollprecht, D.; Küppers, B.; Pomberger, R.; Machiels, L.; Bernardo, E.; Krook, J. Das EU Training Network for Resource Recovery Through Enhanced Landfill Mining (NEW-MINE). Recy DepoTech 2018, 14, 561–568. [Google Scholar]
- Kjeldahl, J. Neue Methode zur Bestimmung des Stickstoffs in organischen Körpern. Zeitschrift für Analytische Chemie 1883, 22, 366–382. [Google Scholar] [CrossRef]
- Kang, K.H.; Shin, H.; Park, H. Characterization of humic substances present in landfill leachates with different landfill ages and its implications. Water Res. 2002, 36, 4023–4032. [Google Scholar] [CrossRef]
- Kirdy, T.; Veselov, A. Phytoprotective Effect of Ammonium Humate at High Copper Concentrations in the Environment. Biol. Bull. 2017, 44, 1284–1288. [Google Scholar] [CrossRef]
- Wang, Y.F.; Lin, F.; Pang, W.Q. Ammonium exchange in aqueous solution using Chinese natural clinoptilolite and modified zeolite. J. Hazard. Mater. 2007, 142, 160–164. [Google Scholar] [CrossRef] [PubMed]

| Sample | Landfill Class | pH | Color/Transparency | Solid Particles | NH4+ Concentration (mg L−1) |
|---|---|---|---|---|---|
| LL 1 | MW | 7.7 | Dark brown to black | None | 350 |
| LL 2 | MW | 8.2 | Dark brown to black | None | 2805 |
| LL 3 | MW | 8.7 | Dark brown to black | None | 645 |
| LL 4 | MW | 8.5 | Brown | None | 945 |
| LL 5 1 | RW | 8.3 | Grey to yellowish/cloudy | Transparent floating particles, black particles (0.063–0.2 mm) | 1037 |
| LL 6 | MW | 8.3 | Orange to brown | Black particles (0.063–2.0 mm) | 523 |
| LL 7 | MW | 8.6 | Orange to dark brown | Transparent floating particles, black particles (0.063–2.0 mm) | 503 |
| LL 8 | MW | 7.7 | (Dark) brown | Black flaky particles (2.0–6.3 mm) | 153 |
| LL 9 | MW/RW | 8.3 | Dark brown to black | Black flaky particles (2.0–6.3 mm) | 1073 |
| LL 10 | MW | 8.2 | Light brown/transparent | None | 75 |
| Sample | Initial NH4+ Concentration (mg L−1)/Cation Loading | Equilibrium NH4+ Concentration (mg L−1) | Equilibrium NH4+ Loading (mg g−1) | Removal Rate (%) | ||||||
|---|---|---|---|---|---|---|---|---|---|---|
| Natural | Ca | Na | Natural | Ca | Na | Natural | Ca | Na | ||
| LL 1 | 350 | 244 | 283 | 180 | 2.65 | 1.67 | 4.24 | 30.4 | 19.1 | 48.5 |
| LL 2 | 2805 | 2440 | 2444 | 2323 | 9.13 | 9.02 | 12.06 | 13.0 | 12.9 | 17.2 |
| LL 3 | 645 | 528 | 514 | 460 | 2.93 | 3.27 | 4.62 | 18.2 | 20.3 | 28.7 |
| LL 4 | 945 | 681 | 677 | 573 | 6.59 | 6.71 | 9.30 | 27.9 | 28.4 | 39.4 |
| LL 5 | 1037 | 713 | 717 | 604 | 8.12 | 8.00 | 10.82 | 31.3 | 30.9 | 41.7 |
| LL 6 | 523 | 347 | 368 | 311 | 4.40 | 3.89 | 5.30 | 33.6 | 29.7 | 40.5 |
| LL 7 | 503 | 320 | 374 | 232 | 4.58 | 3.22 | 6.77 | 36.4 | 25.6 | 53.9 |
| LL 8 | 153 | 95 | 103 | 60 | 1.47 | 1.26 | 2.34 | 38.2 | 32.9 | 61.2 |
| LL 9 | 1073 | 929 | 888 | 816 | 3.61 | 4.62 | 6.42 | 13.4 | 17.2 | 24.0 |
| LL 10 | 75 | 36 | 51 | 32 | 0.97 | 0.59 | 1.08 | 51.8 | 31.3 | 57.8 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Vollprecht, D.; Frühauf, S.; Stocker, K.; Ellersdorfer, M. Ammonium Sorption from Landfill Leachates Using Natural and Modified Zeolites: Pre-Tests for a Novel Application of the Ion Exchanger Loop Stripping Process. Minerals 2019, 9, 471. https://doi.org/10.3390/min9080471
Vollprecht D, Frühauf S, Stocker K, Ellersdorfer M. Ammonium Sorption from Landfill Leachates Using Natural and Modified Zeolites: Pre-Tests for a Novel Application of the Ion Exchanger Loop Stripping Process. Minerals. 2019; 9(8):471. https://doi.org/10.3390/min9080471
Chicago/Turabian StyleVollprecht, Daniel, Sabrina Frühauf, Kristina Stocker, and Markus Ellersdorfer. 2019. "Ammonium Sorption from Landfill Leachates Using Natural and Modified Zeolites: Pre-Tests for a Novel Application of the Ion Exchanger Loop Stripping Process" Minerals 9, no. 8: 471. https://doi.org/10.3390/min9080471
APA StyleVollprecht, D., Frühauf, S., Stocker, K., & Ellersdorfer, M. (2019). Ammonium Sorption from Landfill Leachates Using Natural and Modified Zeolites: Pre-Tests for a Novel Application of the Ion Exchanger Loop Stripping Process. Minerals, 9(8), 471. https://doi.org/10.3390/min9080471






