Notably, the composition of RM greatly varies because of the difference in bauxite composition and production process. For example, the iron content in RM varies in the range of 7–24%, SiO
2 in the range of 8–23%, Al
2O
3 in the range of 7–19%, and the variation range of trace elements is also large [
50]. It is impossible to have a general process to recover a certain valuable component from RM. Although the studies described in the following subsections focus mostly on a specific RM, their research guidance on RM with different properties is significant.
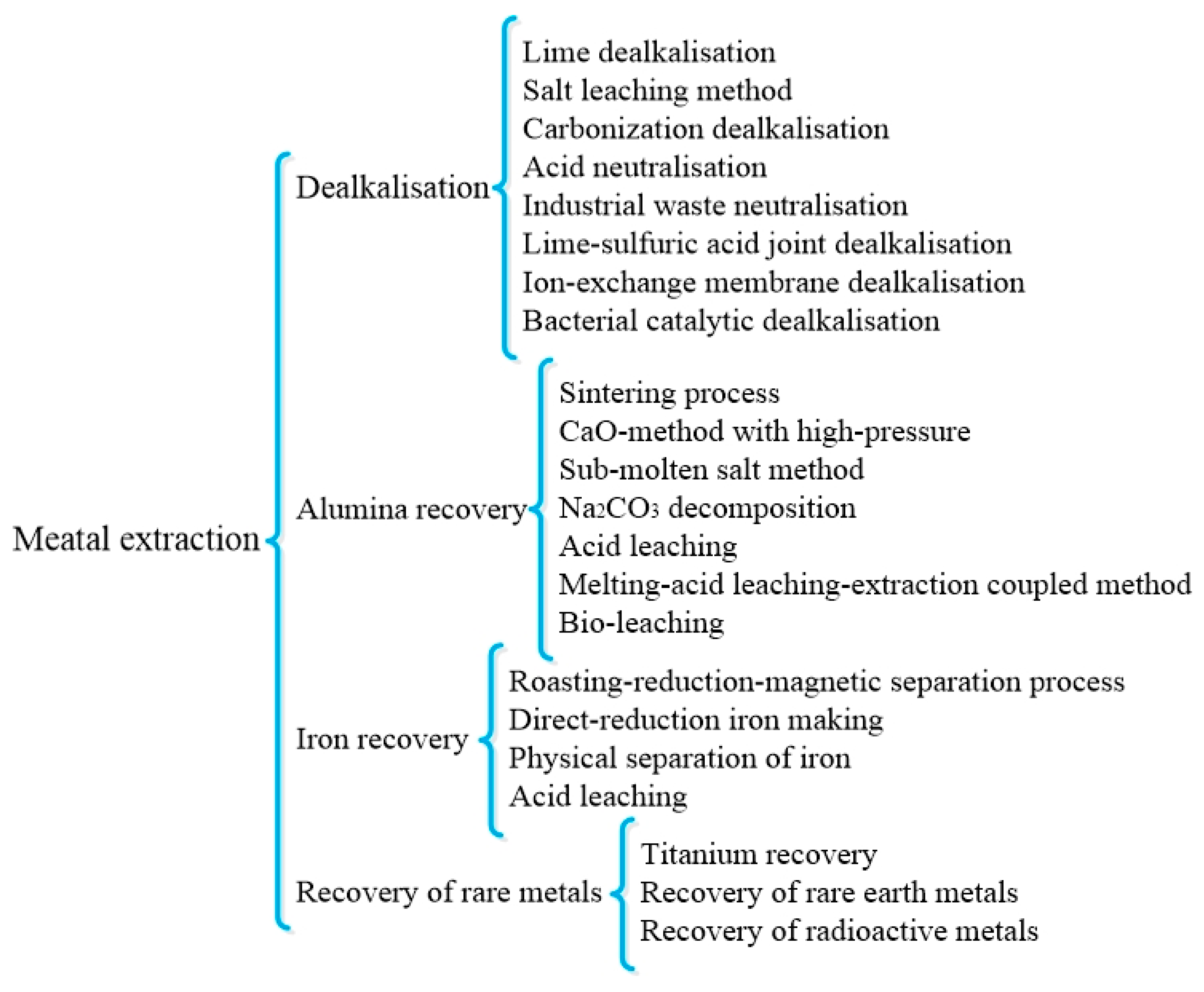
Figure 1.
Classification of main metal disposal methods from RM [
51]. Reproduced with permission from Wang et al., Journal of Cleaner Production; published by Elsevier, 2018 [
51].
4.1.1. Recovery of Iron from RM
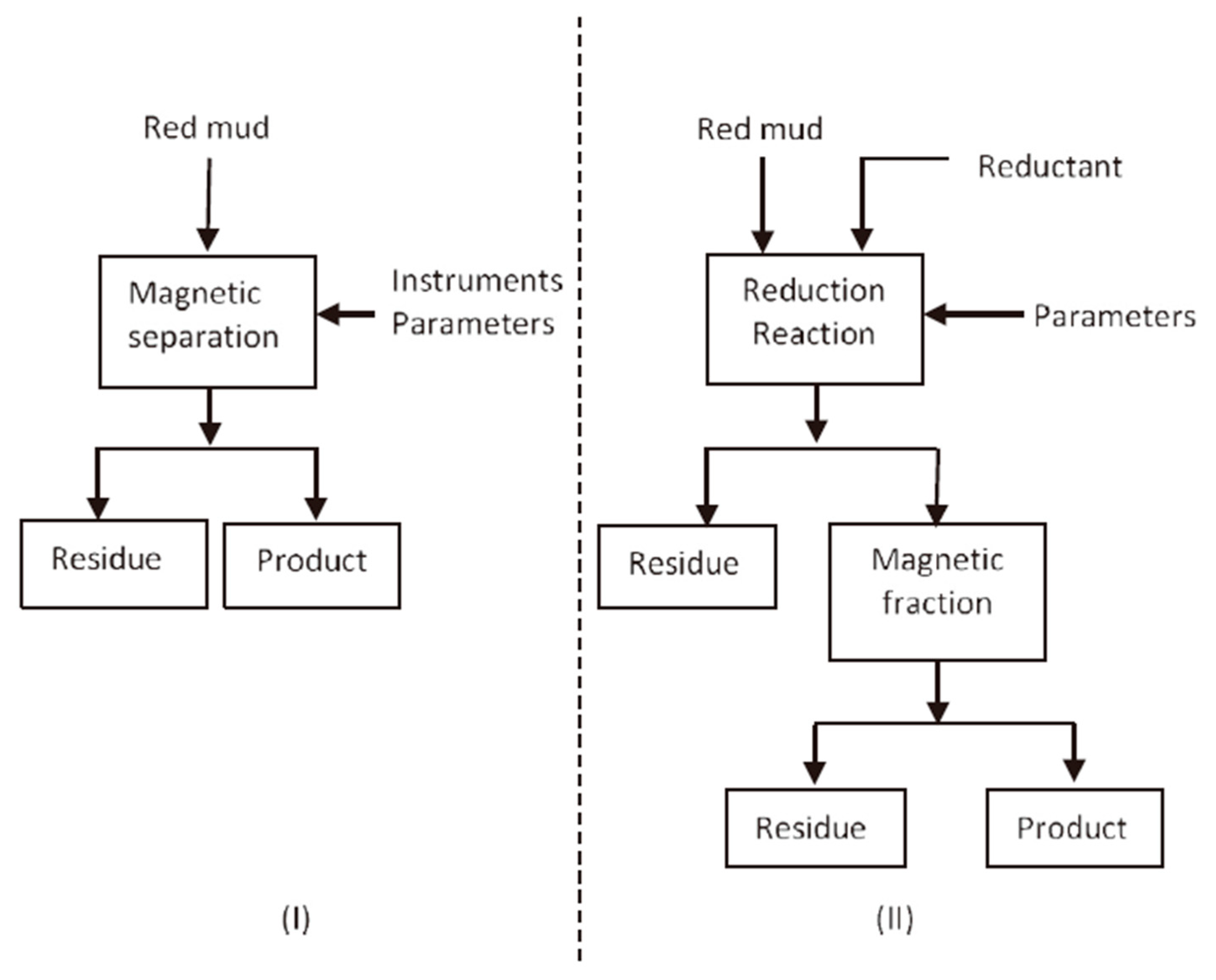
The methods of extracting iron from RM can be divided into pyrometallurgical recovery and direct magnetic separation.
Figure 2 shows some technical schemes.
Considering that the iron in RM is hematite and goethite with weak magnetic properties, direct magnetic process is applied as a basic technology for iron recovery from RM by using high-gradient magnetic separator. In comparison with pyrometallurgical recovery, recovering iron from RM by direct magnetic separation reduces energy costs, while maintaining Ti and other metals that are easily leachable [
38]. Hammond et al. used a high-intensity magnet to separate the iron from RM (in slurry form). The resulting non-magnetic parts can be used in building materials or added back to Bayer processes, while magnetic products can be used as raw materials for iron production or as pigments for pottery [
52]. The separation scheme is shown in
Figure 2I. However, owing to the dissemination of fine iron ore, direct magnetic separation is inefficient.
One of the most widely studied methods to recover iron from RM is pyrometallurgy. The conversion of weakly magnetic hematite to strongly magnetic magnetite or metallic iron can improve the recovery efficiency of iron [
53,
54,
55].
Figure 2II shows the whole scheme of the process. The process requires energy and a reductant in the solid phase or gaseous phase. Li et al. [
56] used the vertical resistance furnace to recover Fe from RM by reduction roasting followed by magnetic separation. Sodium salts played an important role in facilitating the growth of metallic iron particles during reduction roasting. The leaching ratios of Fe increased from 51.9% to 94.7% in the presence of sodium salts. After reductive roasting under a reduction temperature of 1050 °C, 60 min of reduction time, 6% quantity of Na
2SO
4 and 6% quantity of Na
2CO
3, and magnetic concentrate containing 90.2% iron was extracted from roasted materials by magnetic separation method, and the iron recovery was 95.0% [
56].
Liu et al. studied the recovery of iron from RM by magnetic separation after co-roasting with pyrite [
57]. Experimental results showed that by adding pyrite to RM for reducing roasting, its iron oxide was transformed into magnetite. Based on the thermogravimetric and differential thermal analysis of roasting pyrite, the pyrite under N
2 atmosphere had four weight-loss stages at 120 °C (dehydration stage) and in the range of 500–750 °C. Upon testing, they pointed out that the added pyrite produced thermo-decomposed products (element sulfur, iron monosulfide, and pyrrhotite) during roasting, which all had the reduction ability to react with hematite. Their research provided a new method to remove hematite from RM.
4.1.2. Technology of Removing Sodium and Extracting Aluminum from RM
Aluminum can be recovered from RM by alkali roasting with sodium carbonate and high-temperature hydrometallurgy [
53,
58]. In high-temperature hydrometallurgy, the aluminum in RM can be leached at high alkalinity (molar ratio of Na
2O to Al
2O
3 > 10) and high temperatures (>260 °C) in autoclaves. However, this process has not been industrialized because of low alumina concentration in the leach solution and equipment-corrosion problems at high temperatures.
Therefore, the most common process of extracting aluminum is alkali roasting. In this process, the alumina can be converted to sodium aluminate (soluble) by thoroughly mixing RM with sodium carbonate (Na
2CO
3) and calcining it from 800–1200 °C, followed by leaching with water. The main reactions occurring in the roasting system are as follows:
CaO/Ca(OH)
2 is used to replace Na
2O Equations (6) and (7) in sodalite and cancrinite. Then, CO
32− and Al(OH)
4− in RM are cemented out in the form of CaCO
3 and hydrocalumite. When the added CaO reaches the saturation state, the CaO–Na
2O–Al
2O
3–SiO
2–H
2O system becomes balanced. In addition to proper temperature, CaO/RM, and stirring strength, Ca
2+ in lime can partly replace Na
+ in sodalite and cancrinite. The NaOH produced in the replacement reaction enters the solution and increases the pH of the solution. Through washing and separating, the recovered NaOH returns to the alumina extraction process for reuse.
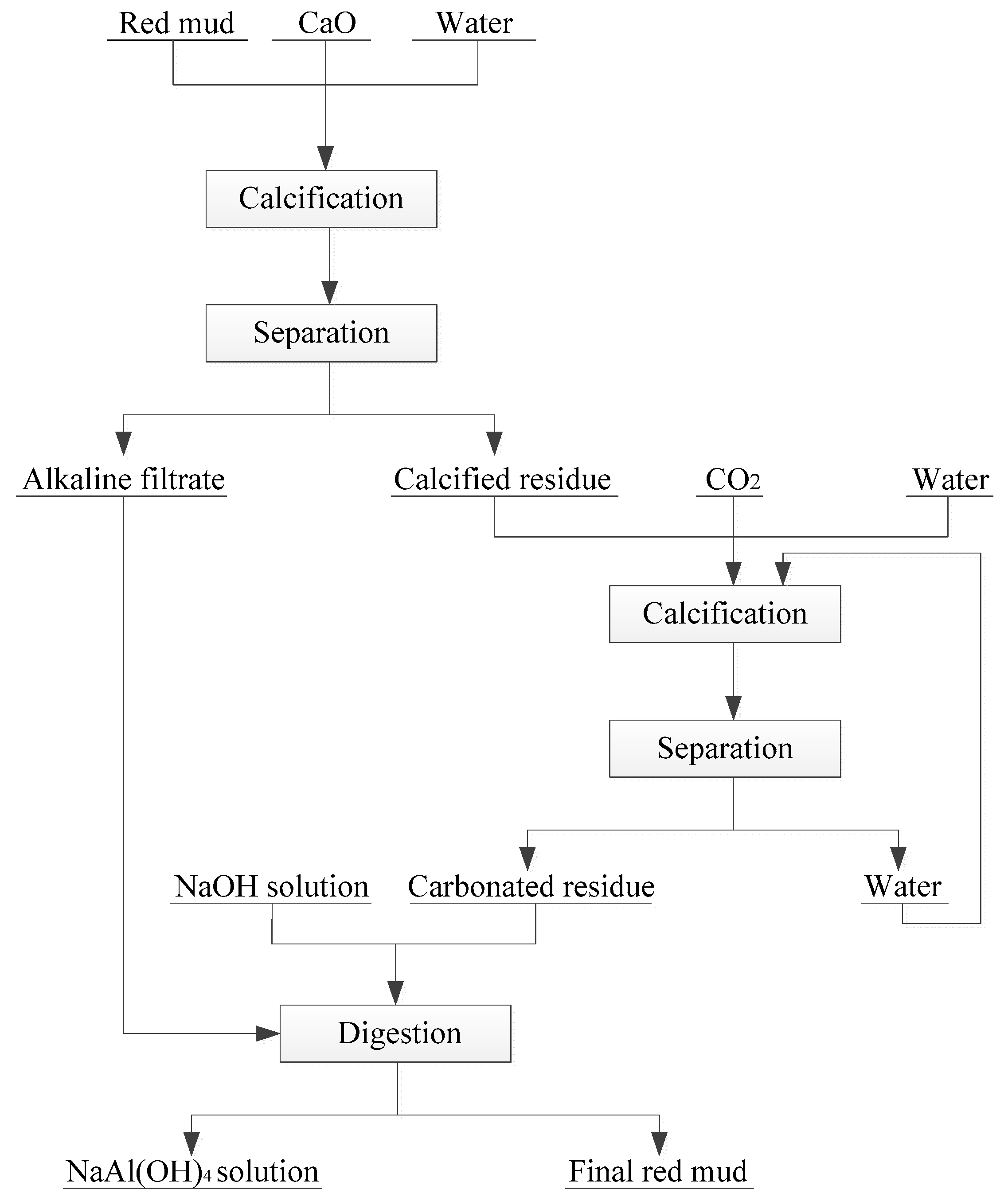
In recent years, some new technologies have been formed to treat RM, such as hydrothermal lime method, normal pressure lime method, lime–soda sintering method, and combined calcification–carbonization method [
59]. These methods have achieved good results in controlling the alkalinity of RM in sintering process and combination method. Among which, combined calcification–carbonization method can regulate the alkaline and recover Al
2O
3 in RM. Zhang et al. proposed a novel combined calcification–carbonization method to recover aluminum and sodium from Bayer RM [
51]. The novel process consists of two basic steps: calcification and carbonization (
Figure 3). First, lime is added into the RM used to transform the main mineral phase Na
2O∙Al
2O
3·xSiO
2·(6 − 2x)H
2O into hydrogarnet 3CaO·Al
2O
3·xSiO
2·(6 − 2x)H
2O and the Na
2O dissolved in the solution. Then, the above calcified solid product was carbonized by high-pressure CO
2, which decomposes it into calcium carbonate, calcium silicate, and aluminum hydroxide. Finally, under the low concentration of NaOH solution, aluminum hydroxide in the carbonized product can be easily extracted. The RM treated by this method can result in an alumina leaching rate of 46.5%, and the Na
2O content in the leaching residue drops to 0.3% or less. These two steps turn RM into a nonhazardous and near-neutral residue that can be used for building material production.
In addition to the lime method, acid leaching [
60], three-waste neutralization [
61], membrane desodium [
62], suspension carbonization [
63], selective flocculation [
64], and other domestic processes, salt leaching [
65], bacterial leaching [
66], and foreign processes are available for alkali removal in RM.
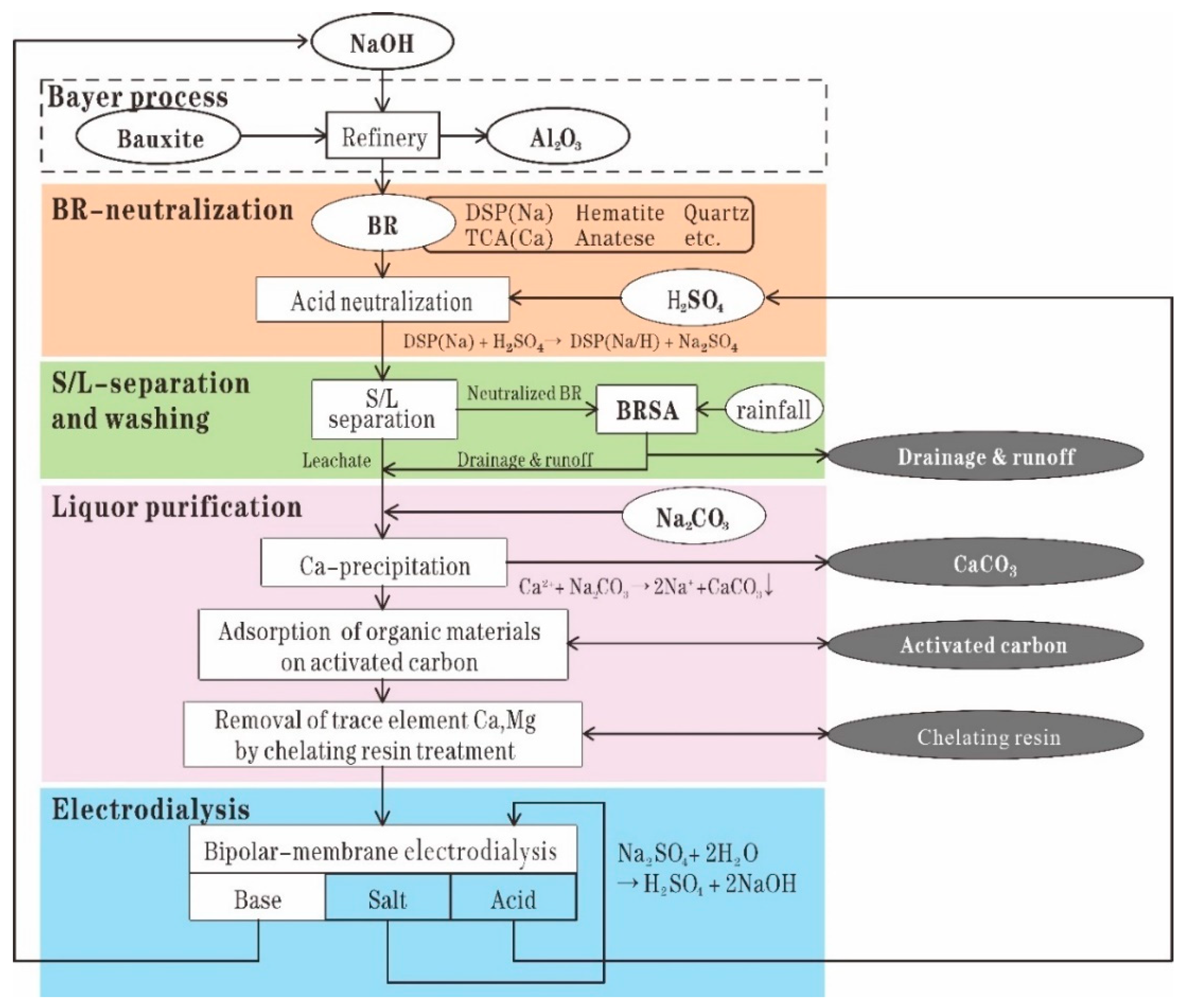
Sulfuric acid is the most commonly used regent used in RM neutralization owing to its low cost [
67]. Recently, CSIRO developed a process to recover sodium from RM, which consists of the following steps: leaching RM by sulfuric acid to extract sodium as sodium sulfate; splitting the sodium sulfate through bipolar-membrane electrodialysis into sulfuric acid and sodium hydroxide, which can be returned to the alumina refinery; and neutralizing RM by using sulfuric acid [
67]. The leached residues after sodium recovery show a low and stable pH and low soda. This property can provide great benefit to long-term residue management [
68].
Figure 4 shows the process of in situ remediation of RM after treatment by CSIRO’s soda recovery process.
4.1.3. Recovery of Titanium, Vanadium, and Rare-Earth Elements (REEs) from RM
Titanium is a rare metal in nature, because its distribution is too dispersed, and its extraction is difficult. However, many titanium resources are found in RM, especially TiO
2 in Indian RM, which accounts for more than 20% [
53]. RM is also rich in vanadium, which is typically mobile in RM leachate, and the concentrations of V ranges from 1.2–15.6 mg L
−1 [
45]. Vanadium exists in pentavalent form in RM leachate, is toxic, and is a possible human carcinogen [
69,
70,
71]. Among the REEs in RM, scandium attracts the attention of many researchers because scandium accounts for more than 90% of REEs with economic value present in RM [
72,
73]. According to the properties and processing technology of bauxite, the content of Sc
2O
3 in RM ranges from 0.004–0.01 wt % [
74].
For recent years, direct acid leaching has been the main technology used to dissolve titanium and scandium from RM in acid medium, followed by precipitation, adsorption, ion exchange, and solvent extraction [
26,
75,
76,
77]. In leaching technology, sulfuric acid, nitric acid, and hydrochloric acid are often used in leaching scandium from RM. Sulfuric acid (H
2SO
4) is the best choice to improve the extraction efficiency of titanium in inorganic acids [
78]. Alkan et al. suggested a leaching process for recovering titanium from RM with sulfuric acid. They reported that the TiO
2 recovery reached approximately 67.3% [
79].
Helena et al. has tested the efficiency of ion exchange resins in recovering vanadium (V) from RM leachates at an alkaline pH of 11.5 and 13 [
80]. Results showed that anion exchange resin can be used to recover vanadium from RM leachate with high alkaline pH value (up to 13). In the subsequent treatment, the vanadium in the resin is easily eluted out by NaOH solution. Results showed that it can use suitable NaOH solution to extract V with the recoveries of 42% and 76% from resins treated with RM leachate at pH 13 and 11.5, respectively.
The direct acid leaching process of RM avoids the high energy consumption of roasting pretreatment and simplifies the technological process. However, direct leaching process shows low selectivity towards other valuable components, such as Al, Fe, and Sc. Therefore, these components commonly enter the leaching solution during acid leaching, which makes the following separation process more complex and consumes a large amount of chemicals during subsequent processes such as solvent extraction [
81]. To reduce the effect of impurity elements, many researchers have focused on developing novel acid leaching systems and organic extraction systems to improve the selectivity of scandium leaching.
Wang et al. first by used dilute sulfuric acid to leach scandium from an Australian RM [
5]. For the following extraction process, a comparative experiment was carried out by using three organic acid extractants, and the result showed that di (2-ethylhexyl) phosphoric acid (D2EHP) exhibited the highest performance. The recovery rate of scandium reached 99%, and no iron and aluminum impurities were formed with the synthetic organic extractant consisting of 0.05 M D2EHPA and 0.05 M tri-butyl-phosphate in Shellsol D70 (100% aliphatic diluent) under the organic solution, and the synthetic leaching solution was mixed in a 5:1 ratio at pH 0.25 and 40 °C.
For reducing the influence of impurity elements on the leaching process, smelting is often carried out before leaching step to remove iron and aluminum from RM. Borra et al. removed iron from RM by smelting reduction, followed by recovering REEs from the slag [
82]. By using 20 wt % wollastonite as flux and 5 wt % graphite as reducing agent, roasting experiments were carried out at 1500–1600 °C. More than 95% of the iron can be extracted from the RM. Under the leaching temperature of 90 °C and by using HCl and HNO
3, more than 70% of titanium, 95% of scandium, and approximately >70% of REEs can be leached.
Another challenge for extract REEs from RM using acid leaching method is the formation of silica gel, because it decreases the filtration efficiency of RM dramatically [
83]. Additionally, the silica gel may hinder the further dissolution of scandium and remarkably reduce the leaching kinetics. Recently, Rivera et al. extracted scandium from RM by dry digestion method with H
2SO
4 or HCl, followed by water leaching [
84]. During the novel process, the water consumption is reduced by up to 60% and silicon dissolution can be limited to less than 5 wt %. Most importantly, silica gel does not form in the process, which significantly improves the filterability of the leach liquor.
Alkan et al. reported a novel scandium leaching process from RM based on oxidative leaching conditions. In this study, the combination of hydrogen peroxide (H
2O
2) and sulfuric acid (H
2SO
4) was utilized as the leaching solution. Results showed that the addition of 2.5 M H
2SO
4 and 2.5 M H
2O
2 at 90 °C and leaching time of 30 min are the best leaching conditions from the inhibition of silica gel formation; the extraction efficiencies of Ti and Sc was the highest, which were 91% and 68%, respectively [
85]. Studies showed that the use of H
2SO
4 alone not only can form silicic acid and have the low selectivity to Ti, Sc and REEs, but also cause the co-precipitation of Sc and the rhomboclase phase, which greatly reduces the leaching rate of Sc. However, when H
2SO
4 and H
2O
2 are added together, it is observed that H
2O
2 provides an oxidizing atmosphere to precipitate the dissolved Si out in quartz phase, thereby preventing the formation of Si gel, and also forms titanium–peroxo–sulfate complex, which provides Ti with a high leaching efficiency. The medium formed in the leaching process also reduces the formation of rhomboclase phase, which entraps Sc during precipitation and is conducive to the formation of hematite [
85]. Consequently, the combined use of hydrogen peroxide in the leaching process can improve the leaching efficiencies of Ti, Sc, and REEs. Furthermore, the liquid–solid ratio, temperature, and other factors must be well controlled in the treatment process because of the acid concentration. The cost must also be considered in the process of recovering Ti, Sc, and REEs from RM.
In the following process, much effort has been made to extract REEs from base elements. Among them, supported ionic liquid phase is proven effective in up-taking REEs from the acid solution because of its high adsorption kinetics and high selectivity for REEs. This novel chromatographic method also exhibits high reusability and stability without decreasing the recovery efficiencies [
86].