Epithermal Bicolor Black and White Calcite Spheres from Herja Ore Deposit, Baia Mare Neogene Ore District, Romania-Genetic Considerations
Abstract
:1. Introduction
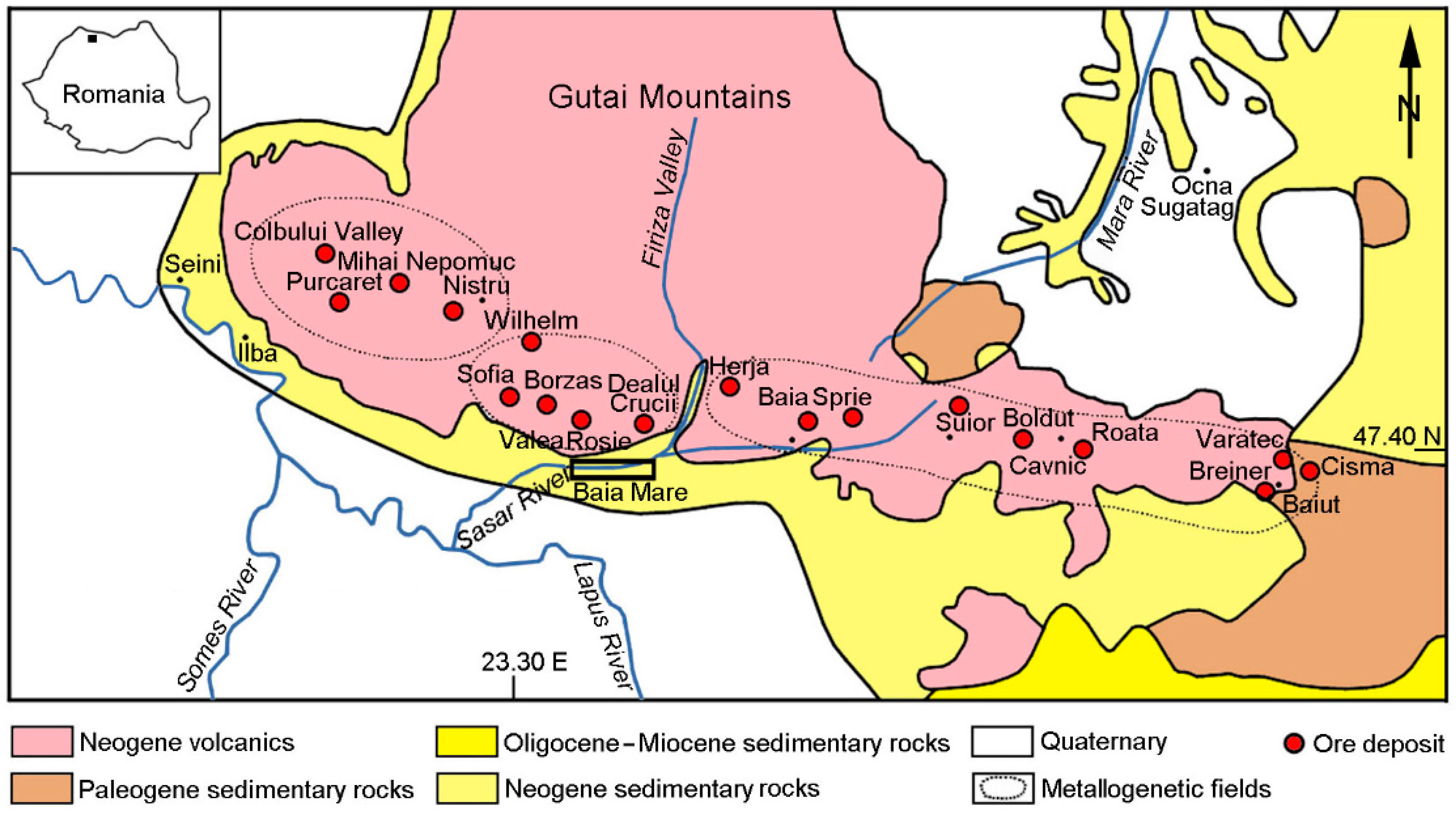
2. Geological Setting
3. Previous Data
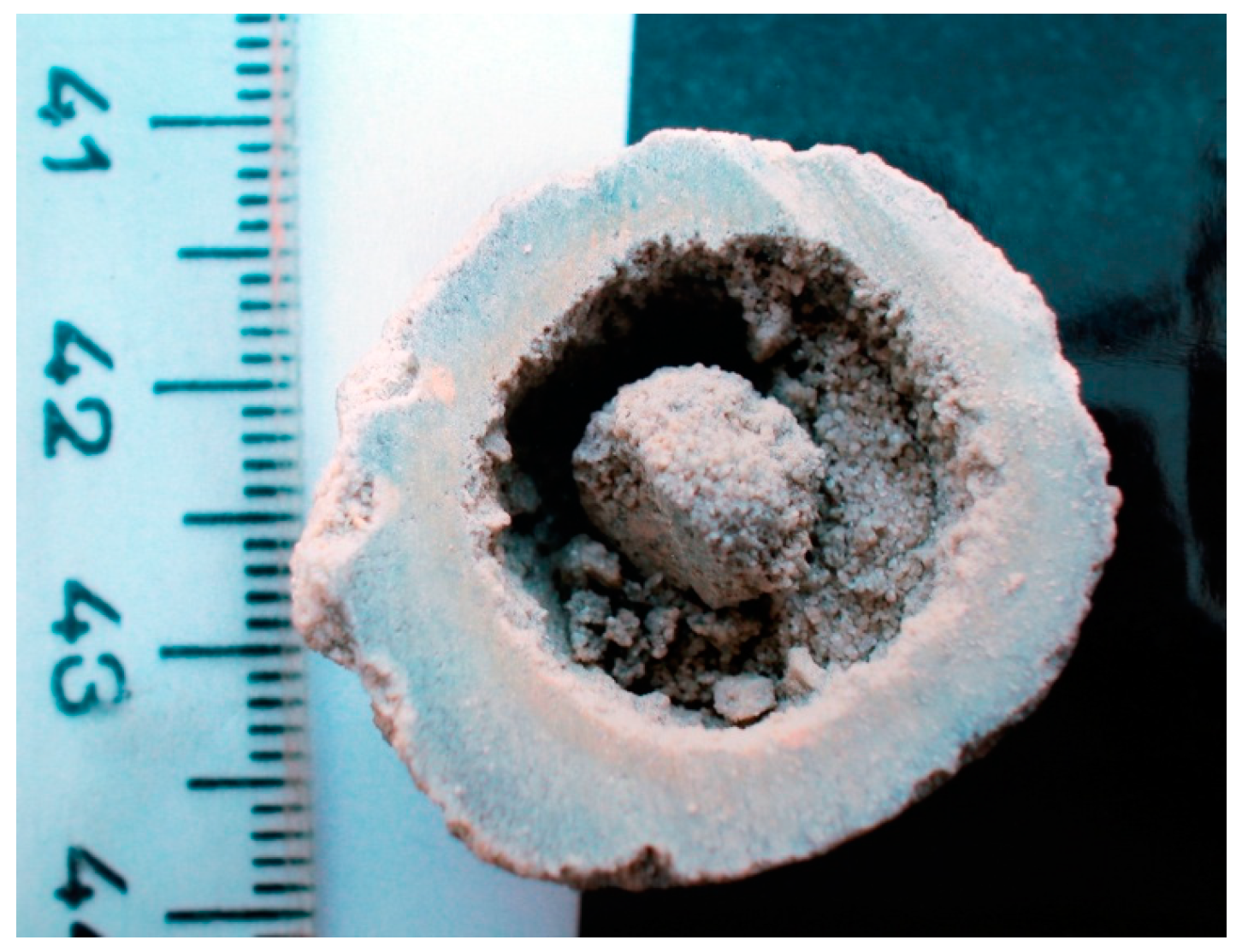
4. New Observations on Carbonate Spheres from the Herja Ore Deposit/Baia Mare Ore District
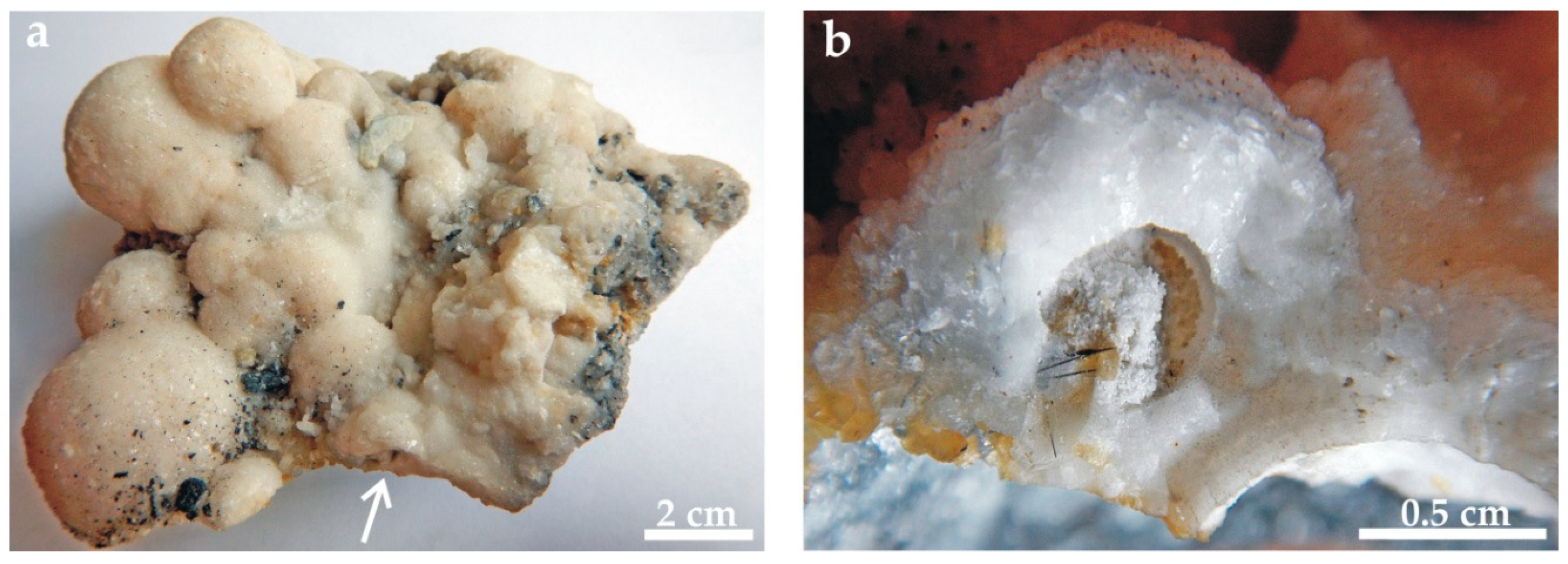
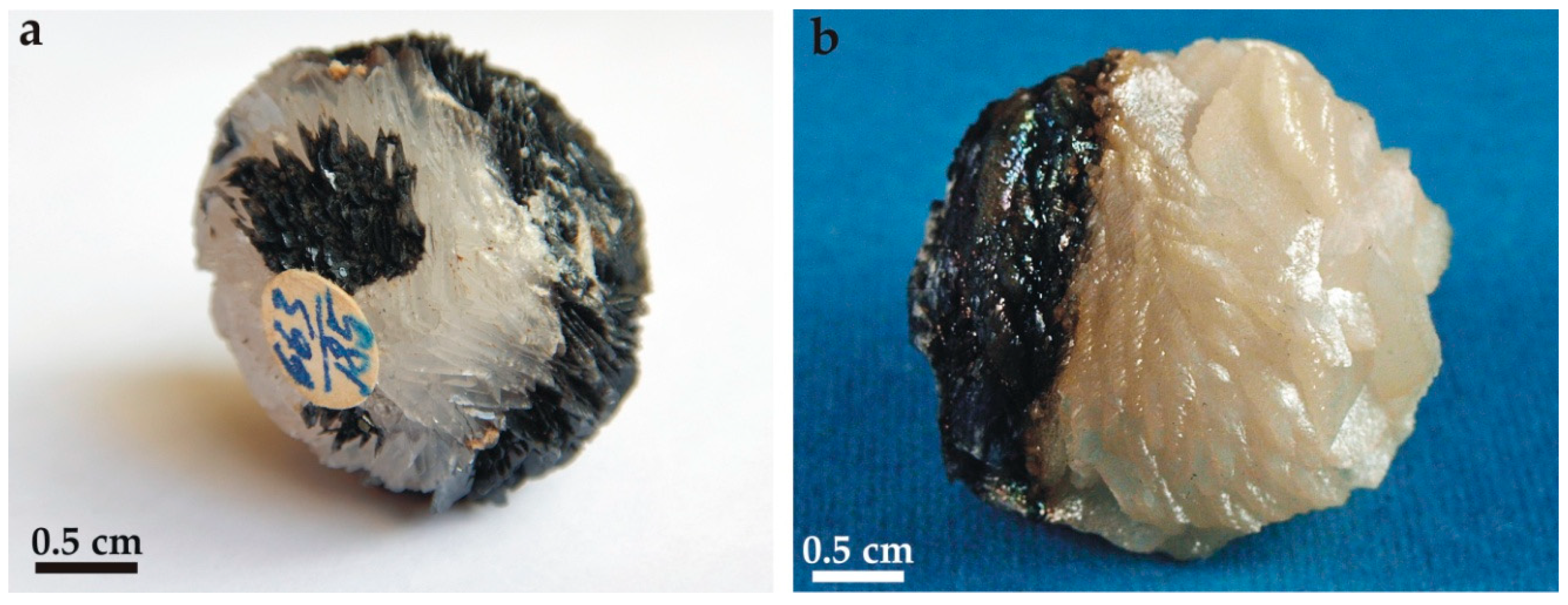
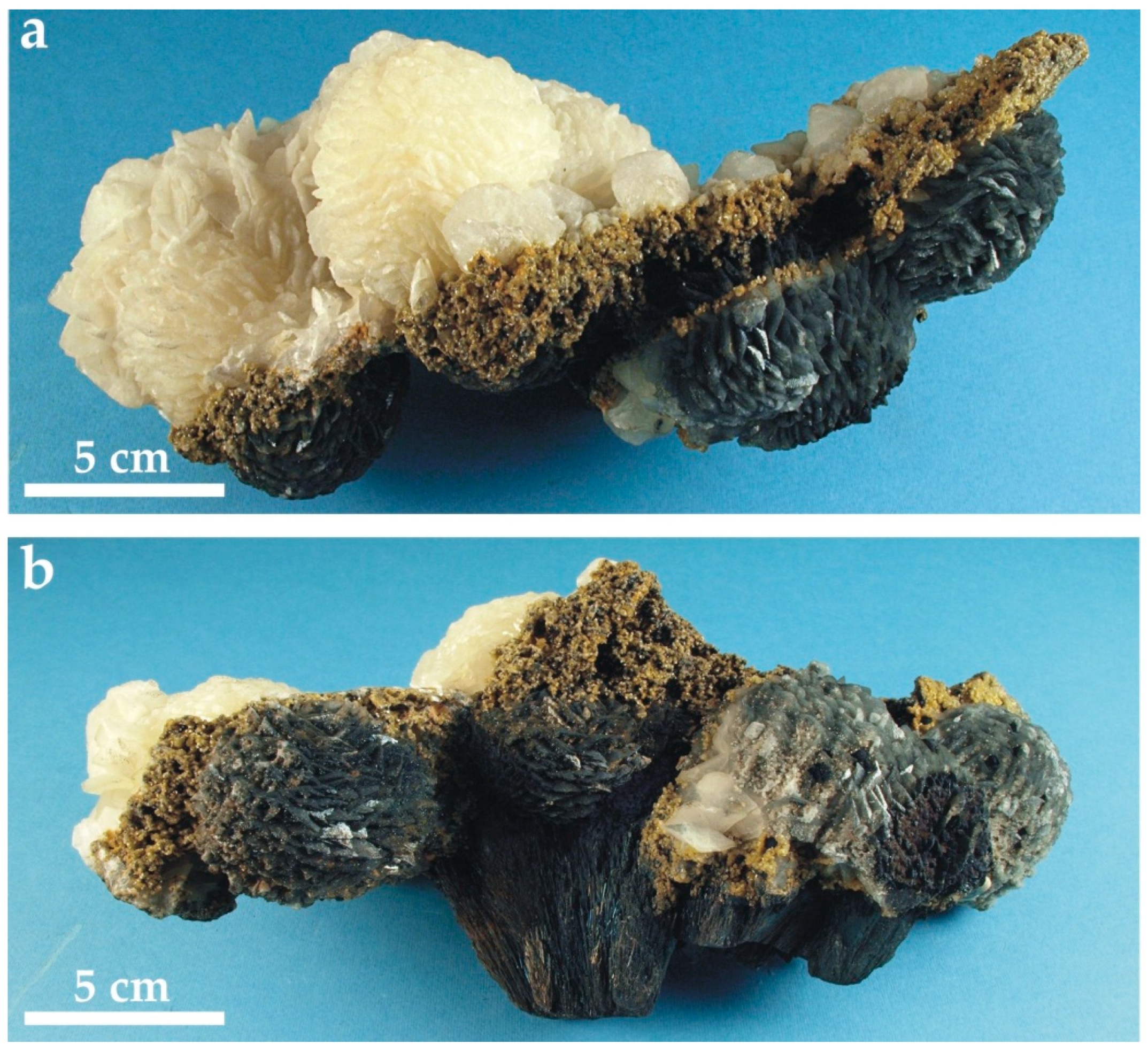
4.1. The Attached Carbonate Spheres
4.2. The Loose Carbonate Hydrothermal Spheres
4.3. Mineral Assemblage of Carbonate Spheres
5. Discussion
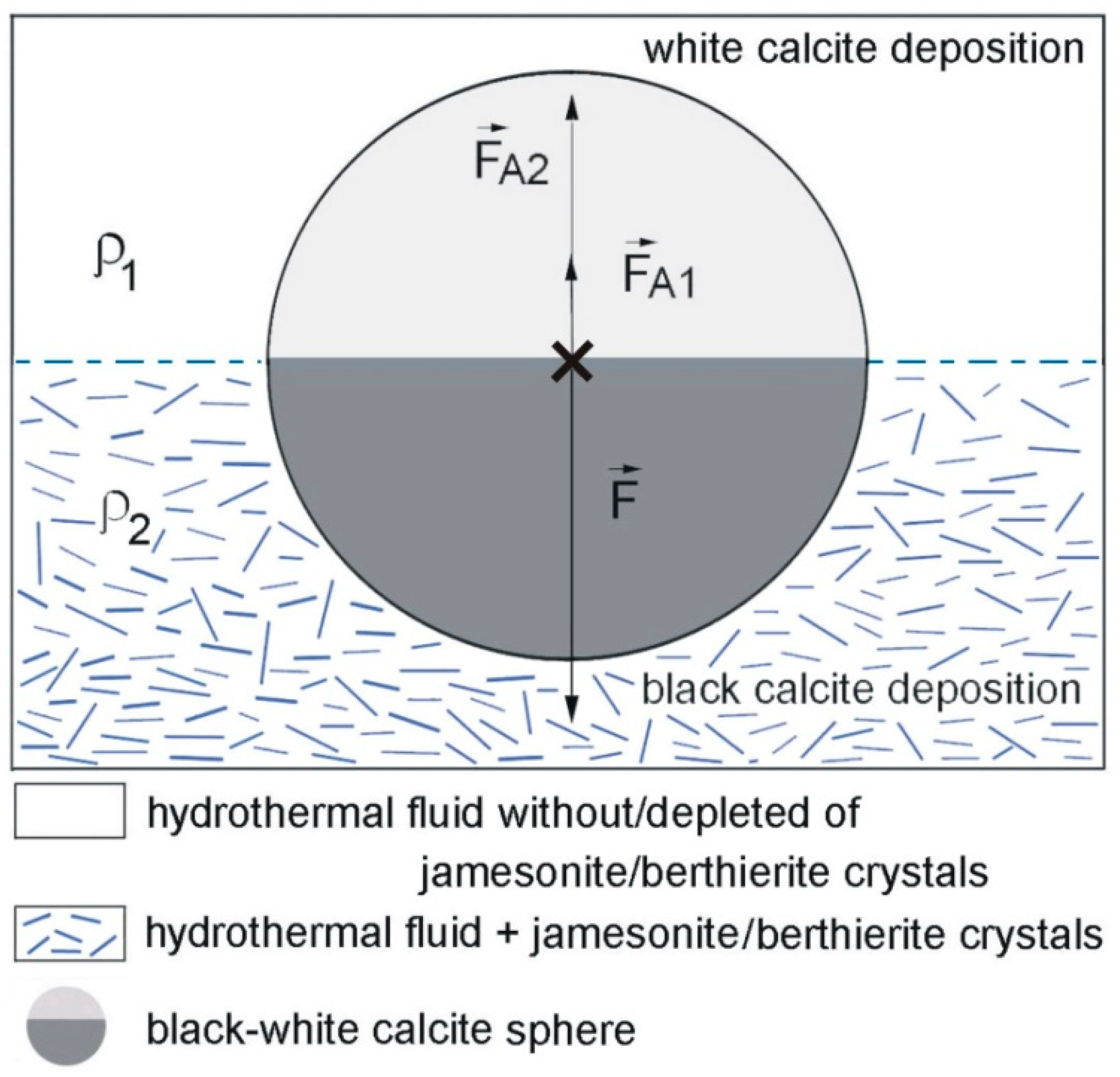
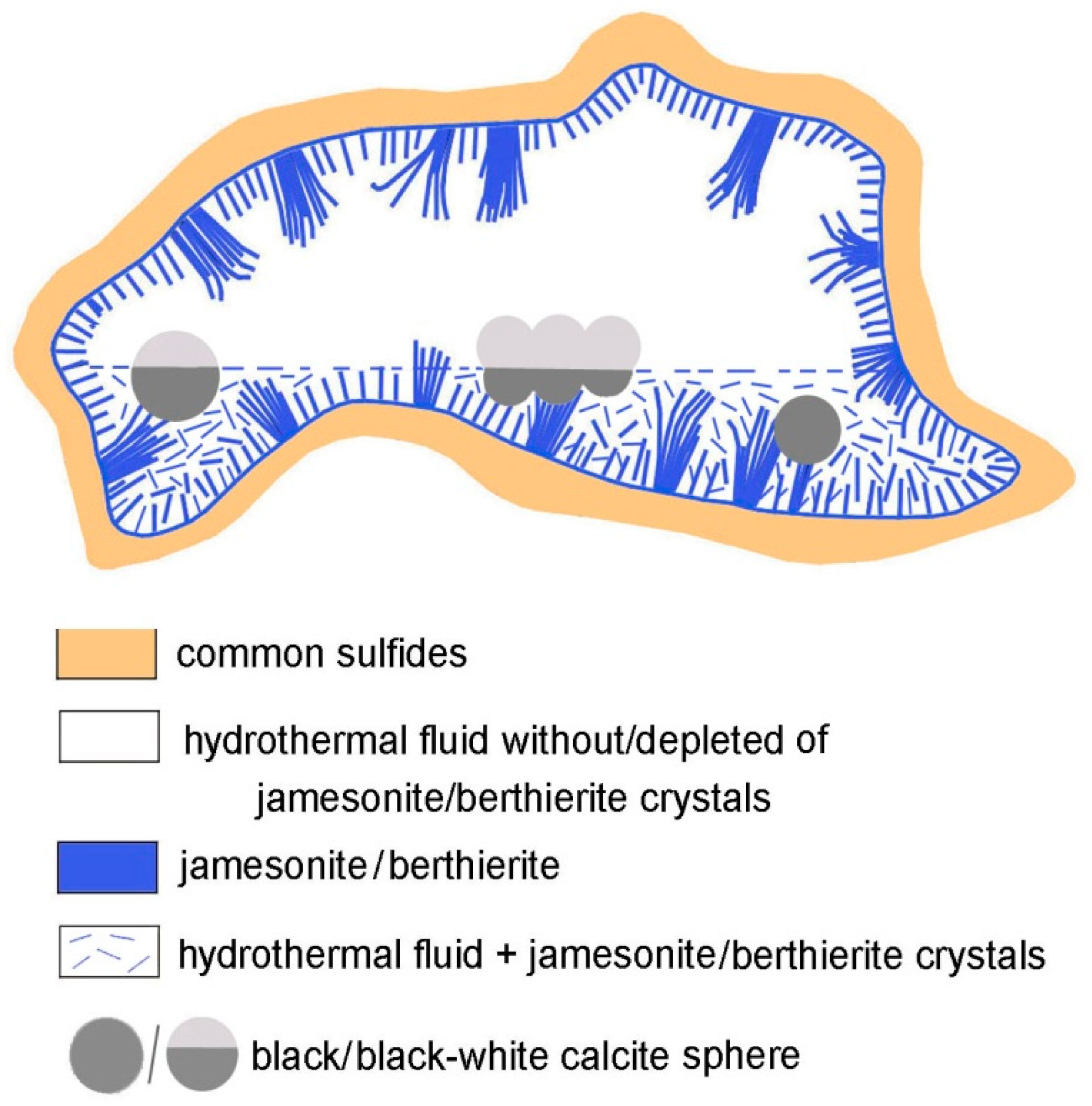
5.1. Environment of Formation
5.2. Genetic Mechanism of Bicolor Spheres Formation
6. Conclusions
Author Contributions
Acknowledgments
Conflicts of Interest
References
- Krenner, J.; Loczka, J. Fizélyit, egy új magyar ezüstérc. Matematika és Természettudományi Értesítő, A Magyar Tudományos Akadémia III. Osztályának Folyóirata 1923, 42, 18–19. [Google Scholar]
- Damian, G. Studiul mineralogic, geochimic și genetic al zăcământului polimetalic de la Herja. Ph.D. Thesis, Universitatea București, București, România, 1996. [Google Scholar]
- Bodiu, A.; Popescu, G.; Ilie, P.; Beregic, V. Regiunea minieră Baia Mare (monografie); Oficiul de Documentare și Publicații Tehnice al Ministerului Minelor, Petrolului și Geologiei: București, România, 1970; p. 130. [Google Scholar]
- Kovacs, M.; Fülöp, A. Baia Mare metallogenetic district. In: Ore deposits and other classic localities in the Eastern Carpathians: From metamorphics to volcanics (eds. G. Iancu and M. Kovacs). Acta Mineral.-Petrogr. Szeged, Field Guide Series 2010, 19, 5–13. [Google Scholar]
- Seghedi, I.; Balintoni, I.; Szakács, A. Interplay of tectonics and neogene post-collisional magmatism in the intracarpathian region. Lithos 1998, 45, 483–497. [Google Scholar] [CrossRef]
- Seghedi, I.; Downes, H. Geochemistry and tectonic development of Cenozoic magmatism in the Carpathian–Pannonian region. Gondwana Res. 2011, 20, 655–672. [Google Scholar] [CrossRef]
- Kovacs, M.; Seghedi, I.; Yamamoto, M.; Fülöp, A.; Pécskay, Z.; Jurje, M. Miocene volcanism in the Oaş–Gutâi Volcanic Zone, Eastern Carpathians, Romania: Relationship to geodynamic processes in the Transcarpathian Basin. Lithos 2017, 294–295, 304–318. [Google Scholar] [CrossRef]
- Bailly, L.; Milesi, J.-P.; Leroy, J.; Marcoux, E. Les minéralisations épithermales à Au-Cu-Zn-Sb du district de Baia Mare (Nord Roumanie): Nouvelles données minéralogiques et microthermométriques. C. R. Acad. Sci. Ser. IIa: Sci. Terre Planets 1998, 327, 385–390. [Google Scholar] [CrossRef]
- Neubauer, F.; Lips, A.; Kouzmanov, K.; Lexa, J.; Ivăşcanu, P. 1: Subduction, slab detachment and mineralization: The Neogene in the Apuseni Mountains and Carpathians. Ore Geol. Rev. 2005, 27, 13–44. [Google Scholar] [CrossRef]
- Tămaș, C.G.; Mârza, I.; Har, N.; Denuț, I. The black calcite and its mineral assemblage in Herja ore deposit, Romania. Eur. J. Mineral. 2018, 30, 1141–1153. [Google Scholar] [CrossRef]
- Petrulian, N. Etude chalcographique du gisement de plomb et de zinc de Herja (Transylvanie, Roumanie). An. Inst. Géol. Roum. 1931, XVI, 539–574. [Google Scholar]
- Koch, S.; Grasselly, G.; Paděra, K. Contributions to the jamesonite problem. Acta Mineral.-Petrogr. 1960, 13, 17–32. [Google Scholar]
- Náray-Szabó, I. Abstract and discussion of paper by Koch et al. (1960). Zentralblatt für Mineralogie 1961, I (Kristallographie & Mineralogie), 273. [Google Scholar]
- Popescu, G.C. Observații asupra câtorva aspecte morfologice ale unor cristale din zăcământul Herja (Baia Mare). Anal. Univ. Buc. Seria Șt. Naturii (Geologie-Geografie) 1964, XIII, 93–105. [Google Scholar]
- Socolescu, M.; Rădulescu, D. Consideration sur la structure des complexes filonien hydrotermaux de la region de Baia mare. Acta Geol. Acad. Sci. Hung. 1971, 15, 41–48. [Google Scholar]
- Pomârleanu, V. Geotermometria și aplicarea ei la unele minerale din România; Editura Academiei Române: București, România, 1971; p. 158. [Google Scholar]
- Moțiu, A.; Ghiurcă, V.; Țîrlea, I. Aspecte paragenetice noi în zăcămintele de sulfuri din regiunea Baia Mare. Studia UBB Seria Geologia-Mineralogia 1972, 1, 11–19. [Google Scholar]
- Borcoș, M.; Lang, B.; Boștinescu, S.; Gheorghiță, I. Neogene hydrothermal ore deposits in the volcanic Gutâi Mountains. III. Dealul Crucii-Băiuț District. A. Herja, Baia Sprie and Șuior ore deposits. Rev. Roum. Géol.-Géophys.-Géogr. Ser. Geol. 1975, 19, 21–35. [Google Scholar]
- Ghiurcă, V. Aus der Welt der runden Jamesonitkristale von Baia Mare. Auffschluss 1985, 36, 145–150. [Google Scholar]
- Ghiurcă, V. Globular crystal aggregates in the jamesonite assemblage. Studii și Cercetări Geologie-Geografie, Complexul Muzeal Bistrița-Năsăud 2005, 10, 41–46. [Google Scholar]
- Ghiurcă, V.; Moțiu, A. Curved jamesonite crystals from Romania. Mineral. Rec. 1986, 17, 375–376. [Google Scholar]
- Popescu, G.C.; Petrescu, M. Contribuții la studiul optic cantitativ și cu microsonda electronică a unor sulfosăruri de la Herja (Munții Gutâi). Stud. și Cerc. de Geol.-Geof.-Gegr. Seria Geol. 1989, 34, 15–19. [Google Scholar]
- Edelstein, O.; Bernád, A.; Kovacs, M.; Crihan, M.; Pécskay, Z. Preliminary data regarding the K-Ar ages of some eruptive rocks from Baia Mare Neogene volcanic zone. Rév. Roum. Géol. 1992, 36, 45–60. [Google Scholar]
- Udubașa, S. Contribution to the study of “plumosite’’ of Herja. Rom. J. Mineral 1992, 75, 46–47. [Google Scholar]
- Udubașa, G.; Cristea, C.; Șerbănescu, A.; Ghiurcă, V.; Udubașa, S. Fibrous sulphosalts in some ores of north Romania. In Proceedings of the Third Geology Symposium, Baia Mare, Romania, 3–4 October 1993. [Google Scholar]
- Udubașa, S.; Udubașa, G.; Ghiurcă, V. Needle-like sulfosalts in hydrothermal ores in Romania. In Proceedings of the IMA 16th General Meeting, Pisa, Italy, 4–9 September 1994; pp. 415–416. [Google Scholar]
- Pécskay, Z.; Edelstein, O.; Kovacs, M.; Bernád, A.; Crihan, M. K-Ar age determination of Neogene volcanic rocks from te Gutâi Mts. (Eastern Carpathians, Romania). Geol. Carpath. 1994, 45, 357–363. [Google Scholar]
- Huber, P.; Mureșan, I. Die Erzlagerstätten Herja. Lapis 1996, 27–34. [Google Scholar]
- Cook, N.J.; Damian, G. New data on “plumosite” and other sulphosalt minerals from the Herja hydrothermal vein deposit, Baia Mare District, Rumania. Geol. Carpath. 1997, 48, 387–399. [Google Scholar]
- Kovacs, M.; Edelstein, O.; Gábor, M.; Bonhomme, M.; Pécskay, Z. Neogene magmatism and metallogeny in Oaș-Gutâi-Țibleș Mts.; a new approach based on radiometric datings. Rom. J. Miner. Depos. 1997, 78, 35–45. [Google Scholar]
- Kovacs, M. Petrogeneza rocilor magmatice de subducție din aria central-sud-estică a munților Gutâi; Editura Dacia: Cluj-Napoca, România, 2002; p. 201. [Google Scholar]
- Mârza, I.; Hallbauer, D.K.; Forray, F. Hollow, non-fixed hydrotermal concretions—A mineralogical curiosity from the Herja (Baia Mare) ore deposit. Rom. J. Miner. Depos. 2004, 81, 130–134. [Google Scholar]
- Ghițulescu, P.T. Distribution de la minéralisation dans les gisements d’ge tertiaire de Transylvanie. Bull. Soc. Roum. Geol. 1935, 2, 56–97. [Google Scholar]
- Almășan, B. Exploatarea zăcămintelor minerale din România; Editura Tehnică: București, Romania, 1984; Vol. 2, p. 204. [Google Scholar]
- Kovács, R.; Tămaș, C.G. Summary of previously identified minerals in Herja ore deposit, Herja-Băiuț metallogenetic field, Baia Mare metallogenetic district (Eastern Carpathians, Romania). In Proceedings of the Sesiunea Științifică Anuală “Ion Popescu Voitești”, Department of Geology, Babeș-Bolyai University, Cluj-Napoca, Romania, 15 December 2017; Filipescu, S., Bindiu-Haitonic, R., Eds.; Presa Universitară Clujeană: Cluj-Napoca, Romania, 2018; pp. 16–17. [Google Scholar]
- Borcoș, M.; Kräutner, H.G.; Udubașa, G.; Săndulescu, M.; Năstăseanu, S.; Bițoianu, C. Herja. In Atlas Geologic 1:100000, Harta Substanțelor Minerale Utile, Ediția a II-a; Ministerul Geologiei, Institutul de Geologie și Geofizică: București, Romania, 1984; Vol. 8, pp. 37–38. [Google Scholar]
- Udubașa, G.; Ilinca, G.; Marincea, Ș.; Săbău, G.; Rădan, S. Minerals in Romania: The state of te art 1991. Rom. J. Mineral. 1992, 75, 1–51. [Google Scholar]
- Pastero, L.; Costa, E.; Alessandria, B.; Rubbo, M.; Aquilano, D. The competition between {1014} cleavage and {0112} steep rhombohedra in gel grown calcite crystals. J. Cryst. Growth 2003, 247, 472–482. [Google Scholar] [CrossRef]
- Seilacher, A. Concretion morphologies reflecting diagenetic and epigenetic pathways. Sediment. Geol. 2001, 143, 41–57. [Google Scholar] [CrossRef]
- Ortoleva, P.; Merino, E.; Moore, C.; Chadam, J. Geochemical self-organization I: Reaction-transport feedbacks and modeling approach. Am. J. Sci. 1987, 287, 979–1007. [Google Scholar] [CrossRef]
- Müller, S.C.; Ross, J. Spatial Structure Formation in Precipitation Reactions. J. Phys. Chem. A 2003, 107, 7997–8008. [Google Scholar] [CrossRef]
- Lu, T.; Sunagawa, I. Texture formation of agate in geode. Mineralogical Journal 1994, 17, 53–76. [Google Scholar]
- Petránek, J. Gravitationally banded (“Uruguay-type”) agates in basaltic rocks—Where and when? Bull. Geosci. 2004, 79, 195–204. [Google Scholar]
- Hickey, K.A.; Barker, S.L.L.; Dipple, G.M.; Arehart, G.B.; Donelick, R.A. The Brevity of Hydrothermal Fluid Flow Revealed by Thermal Halos around Giant Gold Deposits: Implications for Carlin-Type Gold Systems. Econ. Geol. 2014, 109, 1461–1487. [Google Scholar] [CrossRef]
- Aquilina, L.; de Dreuzy, J.-R.; Bour, O.; Davy, P. Porosity and fluid velocities in the upper continental crust (2 to 4 km) inferred from injection tests at the Soultz-sous-Forêts geothermal site. Geochim. Cosmochim Acta 2004, 68, 2405–2415. [Google Scholar] [CrossRef]
- Ge, S. Estimation of groundwater velocity in localized fracture zones from well temperature profiles. J. Volcanol. Geoth. Res. 1998, 84, 93–101. [Google Scholar] [CrossRef]
- Rodriguez-Blanco, J.D.; Shaw, S.; Benning, L.G. The kinetics and mechanisms of amorphous calcium carbonate (ACC) crystallization to calcite, viavaterite. Nanoscale 2011, 3, 265–271. [Google Scholar] [CrossRef]
- Goodwin, A.L.; Michel, F.M.; Phillips, B.L.; Keen, D.A.; Dove, M.T.; Reeder, R.J. Nanoporous Structure and Medium-Range Order in Synthetic Amorphous Calcium Carbonate. Chem. Mater. 2010, 22, 3197–3205. [Google Scholar] [CrossRef]
- Andreassen, J.-P.; Flaten, E.M.; Beck, R.; Lewis, A.E. Investigations of spherulitic growth in industrial crystallization. Chem. Eng. Res. Des. 2010, 88, 1163–1168. [Google Scholar] [CrossRef]








© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Mârza, I.; Tămaș, C.G.; Tetean, R.; Andreica, A.; Denuț, I.; Kovács, R. Epithermal Bicolor Black and White Calcite Spheres from Herja Ore Deposit, Baia Mare Neogene Ore District, Romania-Genetic Considerations. Minerals 2019, 9, 352. https://doi.org/10.3390/min9060352
Mârza I, Tămaș CG, Tetean R, Andreica A, Denuț I, Kovács R. Epithermal Bicolor Black and White Calcite Spheres from Herja Ore Deposit, Baia Mare Neogene Ore District, Romania-Genetic Considerations. Minerals. 2019; 9(6):352. https://doi.org/10.3390/min9060352
Chicago/Turabian StyleMârza, Ioan, Călin Gabriel Tămaș, Romulus Tetean, Alina Andreica, Ioan Denuț, and Réka Kovács. 2019. "Epithermal Bicolor Black and White Calcite Spheres from Herja Ore Deposit, Baia Mare Neogene Ore District, Romania-Genetic Considerations" Minerals 9, no. 6: 352. https://doi.org/10.3390/min9060352
APA StyleMârza, I., Tămaș, C. G., Tetean, R., Andreica, A., Denuț, I., & Kovács, R. (2019). Epithermal Bicolor Black and White Calcite Spheres from Herja Ore Deposit, Baia Mare Neogene Ore District, Romania-Genetic Considerations. Minerals, 9(6), 352. https://doi.org/10.3390/min9060352





