Abstract
This study introduces a new zircon reference material, LKZ-1, for the in situ U–Pb dating and O–Hf isotopic and trace element analyses. The secondary ion mass spectrometric analyses for this gem-quality single-crystal zircon yielded a weighted mean 206Pb/238U age of 572.6 ± 2.0 Ma (2σ, n = 22, MSWD = 0.90), with moderately high U concentrations (619 ± 21 ppm, 1 SD), restricted Th/U ratios (0.146 ± 0.002, 1 SD), and negligible common Pb content (206Pbc < 0.2%). A comparable 206Pb/238U age (570.0 ± 2.5 Ma, 2σ) was produced by the isotope dilution-thermal ionization mass spectrometry. The secondary ion mass spectrometric and laser ablation-assisted multiple collector inductively coupled plasma mass spectrometer analyses respectively showed that LKZ-1 had little variation in O (δ18OV-SMOW = 10.65 ± 0.14‰; laser fluorination value = 10.72 ± 0.02‰; 1 SD) and Hf (176Hf/177Hf = 0.281794 ± 0.000016, 1 SD) isotopic compositions. LKZ-1 was also fairly homogeneous in its chemical composition (RSD of laser ablation ICPMS data ≤ 10%), displaying a relatively uniform chondrite-normalized rare earth element pattern ((Lu/Gd)N = 31 ± 3, Eu/Eu* = 0.43 ± 0.17, Ce/Ce* = 44 ± 32; 1 SD). These consistencies suggest that the LKZ-1 zircon is a suitable working standard for geochronological and geochemical analyses.
1. Introduction
Geochemical and geochronological data for rocks and minerals have long been obtained by wet chemical and isotopic analyses [1,2]. Despite the constraints of time-consuming and labor-intensive sample preparation, wet analysis is still widely used, particularly when a high level of precision is demanded. However, if the sample is composed of chemically or isotopically heterogeneous domains that have their own petrogenetic significance, microsampling strategies are inevitably required.
Technical advances during the past several decades have facilitated the routine analysis of tiny single crystals at the subgrain scale. The high spatial resolution of microbeam techniques, typically of the micrometer- or submicrometer-scale [3,4], provides an excellent opportunity to integrate the chemical and isotopic data from individual subgrain domains with textural observations (e.g., [5]). Zircon (ZrSiO4) has been a prime target of in situ analysis using secondary ion mass spectrometry (SIMS) [6,7] or laser ablation (LA)-assisted inductively coupled plasma mass spectrometry (ICPMS) [8,9], mainly for its ability to retain the original isotopic signature due to its strong resistance to physicochemical breakdown and slow intracrystalline diffusion of constituent ions [10]. Zircon can provide U–Pb age constraints, as well as trace element and O–Hf isotope data for given microdomains. The integration of such multifaceted zircon data has proven to be a powerful tool for addressing the diverse issues related to crustal evolution [11,12,13,14].
Microbeam analysis can be performed rapidly and requires minimal sample preparation; however, the measured data should be carefully corrected and calibrated for spectral and isobaric interference and instrumental fractionation. This process is generally achieved using matrix-matched standards, which are used as a reference material to check the data accuracy and, more importantly, as a primary standard to calculate the inter-elemental isotope ratios or instrumental mass fractionation (IMF) factors. In the latter case, the relative isotopic composition of the standard should be uniform and accurately known, because it directly affects the results calculated for unknown samples. Many zircon standards have been suggested for U–Th–Pb dating and chemical/isotopic analyses. Among these, 91500 [15], FC1 [16], Temora [17,18], and Plešovice [19] are widely used; however, their supply is quite limited. For example, the International Association of Geoanalysts provides only a limited amount of zircon 91500 per laboratory, with the recommendation that it be used initially to establish an in-house reference material. It is also noted that some zircon standards are chemically inhomogeneous [19,20]. Thus, there is still demand for high-quality zircon standards.
This study introduces a new zircon reference material LKZ-1. We measured its U–Pb age and Hf isotopic composition using a high-resolution (HR)-SIMS, isotope dilution thermal ionization mass spectrometry (ID-TIMS), and LA-multiple collector (MC)-ICPMS. The oxygen isotopic composition of LKZ-1 was measured using a laser fluorination system and HR-SIMS. The chemical composition of LKZ-1 was measured using a quadrupole ICPMS connected to a femtosecond LA system.
2. Materials and Methods
2.1. LKZ-1
LKZ-1 is a single transparent pale yellow megacryst of gem-quality zircon with no visible inclusions or cracks (Figure 1). The total mass was 0.83 g. The LKZ-1 was imported from Sri Lanka by an online gem dealer and purchased by the Korea Basic Science Institute (KBSI) in 2014. No information about the nature of its host rock is available.

Figure 1.
A photograph of the LKZ-1 zircon (inset) and representative backscattered electron (upper) and cathodoluminescence (lower) images. The grain and spot numbers are the same as in Table 2.
Our previous LA-MC-ICPMS analyses confirmed that this zircon is in a 238U–230Th radioactive equilibrium [21]. It is noted that the preliminary ion microprobe U–Pb data for LKZ-1, presented by Kim et al. (2015) [22], are revised in this study.
2.2. HR-SIMS U–Pb Analysis
Fragments of the LKZ-1 zircon were mounted in epoxy with reference zircons for in situ isotopic and chemical analyses. The U–Th–Pb isotopic analyses were conducted using a sensitive high-resolution ion microprobe (SHRIMP IIe/MC) at the KBSI Ochang Center. Before the SHRIMP analyses, cathodoluminescence (CL) and backscattered electron (BSE) images of the fragments were examined using a scanning electron microscope (SEM) (JEOL JSM-6610LV). The primary O2– beam was focused into a ~25-μm-diameter spot at an accelerating voltage of 10 kV. The collector slit width was fixed at 100 μm, achieving a mass resolution of about 5000 at 1% peak height. We used the 91500 (206Pb/238U age = 1062.4 Ma, [15]) and SL13 (U = 238 ppm) standard zircons for Pb/U calibration and to determine the U abundance, respectively. The Pb/U ratios were calibrated against the 91500 zircon according to the power law relationship between Pb+/U+ and UO+/U+. The Th/U ratios were estimated using a fractionation factor derived from the measured 232Th16O+/238U16O+ versus 208Pb/206Pb of the SL13 standard. The common Pb was corrected using the 207Pb method [23]. Data processing was conducted using the SQUID 2.50 and Isoplot 3.75 software [24,25]. The weighted mean ages were calculated after excluding the outliers using the Student’s t-test and reported at the 95% confidence level.
2.3. ID-TIMS U–Pb Analysis
ID-TIMS U–Pb isotopic analyses were performed at the Jack Satterly Geochronology Laboratory in the Department of Earth Sciences, University of Toronto (Toronto, ON, Canada). Four fragments were selected for analyses. The fragments were chemically abraded prior to dissolution [26]. This process involved thermal annealing in a muffle furnace at 900 °C for 48 h followed by a chemical etch for 9 h in ~0.10 mL concentrated hydrofluoric acid and ~10 µL 8N nitric acid in Teflon dissolution vessels at 200 °C. The fragments were rinsed with distilled water, and washed in 8N HNO3. A mixed 205Pb–235U spike was added to the Teflon dissolution capsules during sample loading. The zircon-spike mixture was dissolved using ~0.10 mL concentrated hydrofluoric acid and ~0.02 mL 8N nitric acid at 200 °C [27] for 5 days, dried to a precipitate, and re-dissolved in ~0.15 mL 3N hydrochloric acid. The U and Pb were isolated from the zircon solutions using anion exchange chromatography, dried in dilute phosphoric acid, and deposited onto outgassed rhenium filaments with silica gel [28]. The U and Pb were analyzed using a VG M354 mass spectrometer with multiple Faraday collectors in static mode for Pb and a single Daly pulse-counting system in dynamic mode for U measurements. The dead time of the Daly measuring system for U was 14.5 ns. The mass discrimination correction for the Daly detector was constant at 0.03%/atomic mass unit. The amplifier gains and Daly characteristics were monitored using the SRM 982 Pb standard. A thermal mass fractionation correction of 0.1(±0.05)% per atomic mass unit for Pb and U was applied. The total common Pb in each zircon analysis was assumed to have the isotopic composition of laboratory blank (206Pb/204Pb = 18.49 ± 0.4%; 207Pb/204Pb = 15.59 ± 0.4%; 208Pb/204Pb = 39.36 ± 0.4%). The total amount of common Pb in the present analyses ranged from 0.5 to 4.7 picograms. The Pb/U and Pb/Pb isotopic ratios were corrected for IMF, common Pb in the spike, and blank. The Th/U ratios were calculated from the radiogenic 208Pb/206Pb ratio and 207Pb/206Pb age assuming concordance. Corrections for 230Th disequilibrium in 206Pb/238U and 207Pb/206Pb were made assuming Th/U of 4.2 in the magma. We used the decay constants of Jaffey et al. (1971) [29] (238U and 235U are 1.55125 × 10−10 and 9.8485 × 10−10 per year, respectively). The 238U/235U ratio of 137.88 was used for the 207Pb/206Pb model age calculations. All the age errors quoted in the text and table, and error ellipses in the concordia diagram are given at 2σ. Plotting and age calculations were obtained using the Isoplot/Ex 3.00 software [30].
2.4. Oxygen Isotope Analysis
Zircon oxygen isotopes were measured using a laser fluorination system at the Korea Polar Research Institute (KOPRI), and the Cameca IMS 1280 ion probe at the SIMS laboratory of the Institute of Geology and Geophysics, Chinese Academy of Sciences (CAS) in Beijing, China.
The laser fluorination system at the KOPRI for oxygen isotopic analysis is well described elsewhere [31]. About 3 mg of the LKZ-1 zircon was loaded in the nickel sample holder, and then the sample holder was placed in the reaction chamber. The chamber that was assembled with a purification line was evacuated to 10−3 mbar or better, and heated for over 10 h by an external heater to eliminate the absorbed moisture inside the chamber. Before the sample was analysed, the chamber was pre-fluorinated by a small amount of BrF5 for 1 h at room temperature to completely remove the moisture absorbed on the sample surfaces and inside the chamber. Any remaining gases in the reaction chamber were thoroughly evacuated by a diffusion pump (<10−4 mbar), and then sufficient BrF5 was introduced into the chamber for laser fluorination of the sample. The sample was gradually heated using a defocused CO2 infrared laser beam by automatically increasing the lasing power up to 60%. All the gaseous species derived from the samples were introduced into the purification line consisting of two cryogenic traps (liquid nitrogen, −196 °C) and a heated KBr getter. A molecular sieve (MS13X) pellet on the final cryogenic trap collected the pure O2 for 10 min at liquid nitrogen temperature. The oxygen yield of the sample was calculated by the pressure value of the recovered O2 gas from MS13X. The oxygen isotopic composition of the sample gas was analyzed using a dual-inlet isotope ratio mass spectrometer (MAT 253 Plus, Thermo Fisher Scientific, Waltham, MA, USA) connected on-line to the laser fluorination system. The measured 18O/16O ratios were normalized to the Vienna standard mean oceanic water (V-SMOW) (18O/16O = 0.0020052, [32]) and presented as δ18O notation. The long-term reproducibility of the laser fluorination system based on the repeated analyses (n = 28) of in-house obsidian standard is ±0.08‰ (1σ) for δ18O [31].
For the SIMS analyses at the CAS, the Gaussian focused Cs+ primary ion beam was accelerated at 10 kV, with an intensity of ~1.6 nA. The spot size was approximately 20 μm in diameter (10 μm beam + 10 μm raster). A normal incidence electron flood gun was used to compensate for sample charging. The magnetic field was stabilized using a nuclear magnetic resonance controller. Negative secondary ions were extracted with a potential of −10 kV. The field aperture was 6000 × 6000 μm2. A ~120 μm entrance slit, 40 eV energy slit, ~133 transfer magnification, and 500 μm exit slit provided a mass resolution of ~2500 at 1% peak height. Under these conditions, the count rate of 16O– was typically ~1 × 109 cps/nA. The 16O and 18O ions were detected simultaneously using two Faraday cups with 1010 and 1011 Ω resistors, respectively. The in-run precision was typically better than 0.2 ‰ (2 standard errors). The IMF was corrected based on the 91500 zircon value suggested by Valley (2003) [7] (δ18OV-SMOW = 10.07 ± 0.03‰).
2.5. LA-MC-ICPMS U–Pb and Lu–Yb–Hf Isotope Analysis
Zircon U–Pb and Lu–Yb–Hf isotopes were measured using a Plasma II MC-ICPMS (Nu Instruments) equipped with an NWR193-nm ArF Excimer laser ablation system at the KBSI Ochang Center. The instrumental parameters for the U–Pb and Lu–Yb–Hf isotopic analyses are summarized in Table 1. The raw data were processed using Iolite 2.5 within the Igor Pro 6.3.5.5 software [33], and corrected for the background. The instrumental mass discrimination and laser-induced elemental fractionation during the U–Pb analysis were corrected by calibration against the 91500 zircon. No common Pb correction was performed. The ages were calculated using the Isoplot 3.75 software [25]. All the ratios were calculated with 2σ errors.

Table 1.
LA-MC-ICPMS instrument and operational parameters for U–Pb dating and Lu–Yb–Hf isotope analysis.
The 176Lu and 176Yb interferences on the 176Hf signal were corrected using the isotopic values suggested by Chu et al. (2002) [34] and Vervoort et al. (2004) [35], respectively, on the basis of previous MC-ICPMS results for mixed standard solutions [36]. The mass bias of the measured Hf isotopic ratios was corrected to 179Hf/177Hf = 0.7325 using an exponential correction law. The 176Lu/177Hf and 176Yb/177Hf ratios were calculated following the method of Iizuka and Hirata (2005) [37]. The εHf values were calculated using a 176Lu decay constant of 1.865 × 10−11 per year [38] and the chondritic values suggested by Blichert-Toft and Albarède (1997) [39].
2.6. LA-Quadrupole ICPMS Analysis
The minor and trace elements were analyzed using a 343 nm femtosecond laser ablation microprobe (J200 LA model, Applied Spectra Inc., Fremont, CA, USA) coupled to an iCapQ model (Thermo Fisher Scientific, Bremen, Germany) quadrupole ICPMS at the Core Research Facilities, Pusan National University, Korea. The zircon surfaces were ablated at a pulse repetition rate of 10 Hz and a pulse energy of 250 μJ, producing a ~30-μm-diameter crater pit. Helium (700 mL/min) was flushed into the sample cell to increase the sample transport efficiency and reduce aerosol deposition around the ablation pit [40]. The instrumental parameters of ICPMS were optimized to provide the highest sensitivity, whilst maintaining the ratio of ThO+/Th+ below 0.005. The zircon data were externally calibrated using NIST 610 standard glass, and internally normalized using 29Si and 91Zr. The reference values for the NIST 610 were taken from Jochum et al. (2011) [41], and the stoichiometric values of zircon (Si = 15.3%, Zr = 47.6%) were used for normalization.
3. Results and Discussion
3.1. U–Pb Age
As shown in Figure 1, the LKZ-1 fragments were relatively dark under CL. The CL and BSE images showed no clear zonation patterns. The HR-SIMS (SHRIMP) U–Th–Pb isotope data are presented in Table 2. The common Pb proportion in 206Pb, calculated using the 204Pb counts and an assumed Pb isotopic composition after the model of Stacey and Kramers (1975) [42], was mostly less than 0.1%. The SHRIMP analyses produced consistently concordant data (Figure 2). The 207Pb-corrected data generated a weighted mean 206Pb/238U age of 572.6 ± 2.0 Ma (n = 22, MSWD = 0.90), which was consistent with the 204Pb-corrected 207Pb/206Pb age (weighted mean = 573 ± 12 Ma, n = 21, MSWD = 1.4). The relatively uniform concentrations of U (619 ± 21 ppm, 1 standard deviation, same hereafter unless otherwise stated) and Th (90.2 ± 4.2 ppm) yielded highly consistent Th/U ratios of 0.146 ± 0.002. The Plešovice zircon (ID-TIMS 206Pb/238U age = 337.13 ± 0.37 Ma; [19]), analyzed together with LKZ-1, yielded a weighted mean 206Pb/238U age of 338.7 ± 1.4 Ma (n = 15, MSWD = 0.81) (Table S1).

Table 2.
SHRIMP U–Th–Pb results for the LKZ-1 zircon.
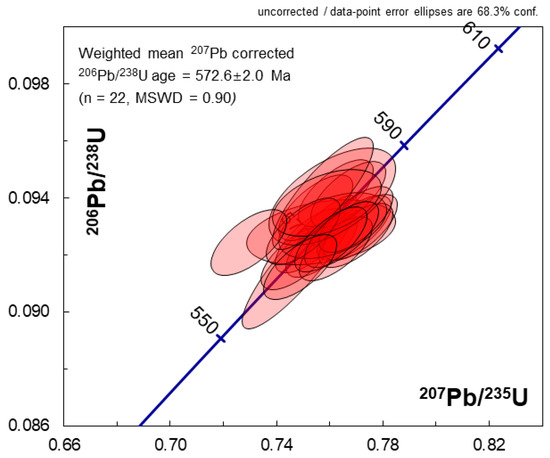
Figure 2.
206Pb/238U-207Pb/235U concordia diagram showing the SHRIMP results for the LKZ-1 zircon with ±1σ error ellipses. The plotted data are uncorrected for common Pb.
The LA-MC-ICPMS U–Pb results for the LKZ-1 zircon are listed in Table 3, and shown graphically in Figure 3. The LA-MC-ICPMS analyses produced concordant age data, with a weighted mean 206Pb/238U age of 574.8 ± 1.3 Ma (n = 28, MSWD = 1.13).

Table 3.
LA-MC-ICPMS U–Pb results for the LKZ-1 zircon.
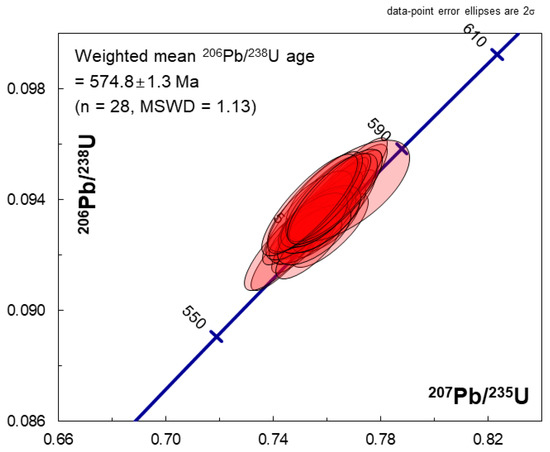
Figure 3.
206Pb/238U–207Pb/235U concordia diagram showing LA-MC-ICPMS results for the LKZ-1 zircon with ±2σ error ellipses.
Four U-Pb ID-TIMS zircon analyses produced two overlapping, concordant results and two that were slightly discordant (1.2%), but within error of the concordant results (Table 4, Figure 4). Four U-Pb data together yielded a weighted mean 207Pb/206Pb age of 575.22 ± 0.98 Ma (MSWD = 0.16, probability = 0.92). The weighted mean 206Pb/238U age of the four data was 570.0 ± 2.5 Ma (MSWD = 5.4, probability = 0.001), but if only the two concordant results are considered, a more precise 206Pb-238U age of 571.7 ± 1.1 Ma is obtained. The Th/U ratios (0.148 ± 0.001) were consistent with those from the SHRIMP analyses (0.146 ± 0.002). The ID-TIMS results may indicate that discordant parts remained in the analyzed fragments in spite of thermal annealing and chemical etching. The discordance of U–Pb age is not attributable to metamictization because the U contents and 206Pb/238U dates of the SHRIMP spots did not show any particular trend (Figure S1). If the discordance of our ID-TIMS ages reflects the thermal disturbance experienced by LKZ-1, then the concordance of SHRIMP U–Pb dates may have been caused by the selective analysis of inner, unaffected parts of the crystal. The SEM observation was not helpful to identify the discordant parts because the fragments appeared homogeneous in CL and BSE (Figure 1).

Table 4.
U–Pb isotopic data for the LKZ-1 zircon obtained by ID-TIMS.
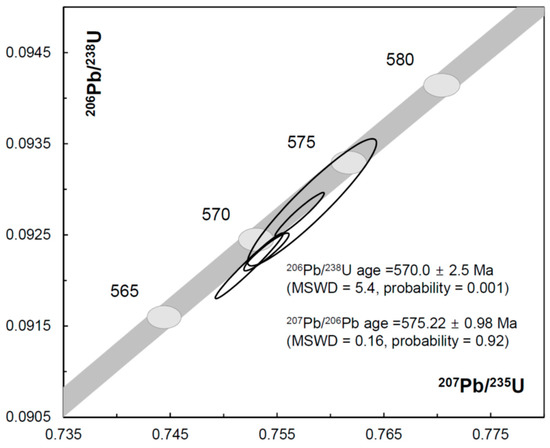
Figure 4.
Concordia diagram showing U–Pb data obtained by ID-TIMS analysis of LKZ-1 zircon with ±2σ error ellipses. The concordia curve is depicted as a band, which incorporates the uncertainty of the U decay constants.
In fact, zircon megacrysts commonly show U–Pb age discordance [6]. The discordancy is typically small. For example, the ID-TIMS results for the 91500 zircon reported by Wiedenbeck et al. (1995) [15] were ~0.3% discordant (207Pb/206Pb age = 1065.4 ± 0.3 Ma, 206Pb/238U age = 1062.4 ± 0.4 Ma). The ID-TIMS results for SL13, listed by Claoué-Long et al. (1995) [43], were also discordant. The weighted mean 207Pb/206Pb and 206Pb/238U ages were 576.3 ± 0.8 Ma and 572.1 ± 0.4 Ma, respectively. The discordance of SL13 data was attributed to the extraneous addition of 231Pa into the crystal or uncertainties of the U decay constants [44,45]. The identical U–Pb ages of the SL13 and LKZ-1 zircons may represent an important geologic event in Sri Lanka.
We conclude that the best 206Pb/238U ratio of LKZ-1 is equivalent to the SHRIMP age (572.6 ± 2.0 Ma), considering that discordant parts in the zircon fragments may have been included in the ID-TIMS analyses. As stated earlier, this age is consistent with the ID-TIMS 206Pb/238U age constrained by concordant results (571.7 ± 1.1 Ma).
3.2. O and Lu–Y–Hf Isotopic Compositions
The O and Lu–Yb–Hf isotopic compositions of LKZ-1 are presented in Table 5 and Table 6, respectively. The δ18OV-SMOW value of LKZ-1 measured by the laser fluorination system was 10.72 ± 0.02 ‰. The oxygen yield of LKZ-1 was 97.8%, meaning that almost all the oxygen was released from the sample. Thus, any kinetic isotopic fractionation effect caused by incomplete fluorination of sample can be ignored.

Table 5.
HR-SIMS O isotopic results for the LKZ-1 zircon.

Table 6.
LA-MC-ICPMS Lu–Yb–Hf results for the LKZ-1 zircon.
During the analytical session at the CAS, the 91500 and Penglai [46] zircons yielded an average measured δ18OV-SMOW value of 15.16 ± 0.17 ‰ and 10.40 ± 0.21‰, respectively. Detailed data, including 16O intensities, 18O/16O ratios, and spot locations on the mount, are given in Table S2. The interlaboratory calibration work of Wiedenbeck et al. (2004) for the 91500 zircon [47] reported laser fluorination δ18OV-SMOW values between 10.07‰ and 9.74‰. Although the simple mean was 9.86 ± 0.11‰ (n = 13), the high-end value (10.07‰) was preferred in this study to correct the IMF because the simple mean yielded a slightly underestimated δ18OV-SMOW average (5.08‰) for the Penglai zircon (laser fluorination δ18OV-SMOW = 5.31 ± 0.12‰; [46]). An IMF value of –5.09‰, with reference to the 91500 zircon value of 10.07‰, generated an average δ18OV-SMOW value of 10.65 ± 0.14‰ for the LKZ-1 zircon (Figure 5). This value was indistinguishable from that obtained by the laser fluorination method (10.72 ± 0.02‰). The Penglai zircon measured during several analytical sessions yielded an IMF-corrected δ18OV-SMOW of 5.29 ± 0.34 ‰ (n = 23) (Table S2).
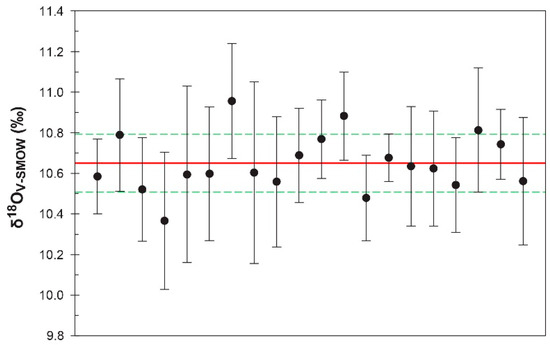
Figure 5.
O isotopic variation in the LKZ-1 zircon based on HR-SIMS analyses. Vertical error bars denote 2 standard errors. Solid and dashed lines represent the average (10.65‰) and standard deviation (±0.14‰), respectively.
The LA-MC-ICPMS analyses produced an average 176Hf/177Hf of 0.282294 ± 0.000021 (n = 41) for the 91500 zircon (Table S3), which was consistent with the MC-ICPMS result obtained by solution chemistry (0.282308 ± 0.000006; [48]). The 176Lu/177Hf results (0.00028 ± 0.00010) for the 91500 zircon (Table S3) were also consistent with the solution chemistry-based data (0.00031 ± 0.00007; [48]). The LKZ-1 zircon showed little intra- and inter-grain variation in 176Hf/177Hf (0.281794 ± 0.000016; Figure 6), 176Lu/177Hf (0.000104 ± 0.000001), and 176Yb/177Hf (0.00358 ± 0.00035) (n = 45). The 178Hf/177Hf ratios were indistinguishable between 91500 (1.467289 ± 0.000059) and LKZ-1 (1.467286 ± 0.000057), with their 176Lu/177Hf and 176Yb/177Hf ratios showing a good positive correlation (R2 = 0.97). The initial εHf values calculated from the 176Hf/177Hf and 176Lu/177Hf ratios and SHRIMP 206Pb/238U age (572.6 Ma) varied narrowly and gave an average value of –22.0 ± 0.6. The 176Hf/177Hf and 176Lu/177Hf ratios did not show a clear trend (Figure S2).
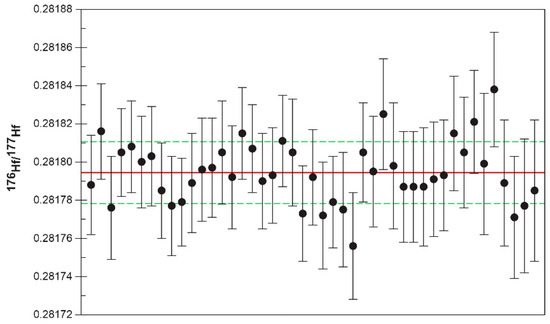
Figure 6.
Hafnium isotopic variation in the LKZ-1 zircon. Vertical error bars denote 2 standard errors. Solid and dashed lines represent the average (0.281794) and standard deviation (±0.000016), respectively.
3.3. Chemical Composition
The LA-quadrupole ICPMS results for the 91500 and LKZ-1 zircons are summarized in Table 7 with literature data [15,47,49,50,51]. The individual spot data analysed in this study are listed in Table S4.

Table 7.
Summary of chemical data for the 91500 and LKZ-1 zircons (unit: ppm).
For the 91500 zircon, the internal normalization by 91Zr generated concentration results close to the literature data, except for U and Th. We employed 29Si-normalization for the calculation of U and Th concentrations because the 91Zr-normalization for these elements yielded results consistently ~15% lower than the recommended values. The 29Si-normalized results for U (83 ± 14 ppm) and Th (30 ± 6 ppm), and the 91Zr-normalized result for Hf (5780 ± 340 ppm) agreed well with the ID-TIMS data ([U] = 81.2 ppm, [Th] = 28.61 ppm, [Hf] = 5895 ppm; [15]). The 91Zr-normalized results for P (14 ± 7 ppm) and Y (148 ± 36 ppm) were marginally or closely similar to the literature data [[P] = 35 ± 17 ppm, [Y] = 153 ± 14 ppm; [47]). As shown in Figure 7a, the chondrite-normalized pattern of our 91Zr-normalized rare earth element (REE) data generally matched the recommended values. The slightly negative Eu anomalies in our data (Eu/Eu* = 0.72 ± 0.17) are consistent with the data previously obtained by inter-laboratory SIMS analyses [47].
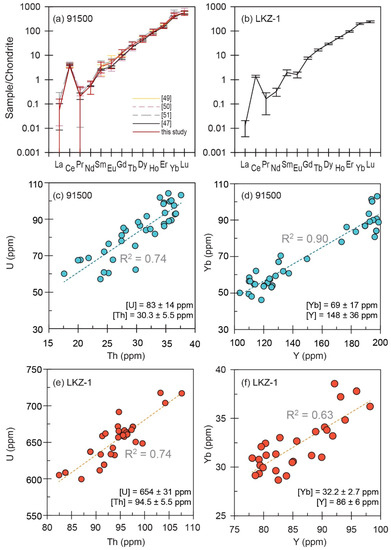
Figure 7.
Chondrite-normalized REE patterns of 91500 (a) and LKZ-1 (b) zircons. Chondrite values were taken from McDonough and Sun (1995) [52]. Vertical error bars denote 1 standard deviation of our data, or uncertainties reported in previous works. (c–f) U vs. Th and Yb vs. Y variations for 91500 and LKZ-1 zircons.
For most elements >1 ppm, the relative standard deviation values for the concentration data for the LKZ-1 zircon were <10%. The chondrite-normalized REE pattern of LKZ-1 (Figure 7b) was highly consistent, with prominently positive Ce (Ce/Ce* = 44 ± 32) and negative Eu (Eu/Eu* = 0.43 ± 0.17) anomalies. The ratios between the heavy and middle REEs were also consistent ((Lu/Gd)N = 31 ± 3). The U (654 ± 31 ppm) and Th (94 ± 6 ppm) concentrations, and their ratios (Th/U = 0.144 ± 0.004), were comparable to the SHRIMP results. The positive correlations observed between U and Th concentrations and Yb and Y concentrations (Figure 7c–f) indicate that our data reflect inter- and intra-grain chemical variation, rather than random analytical errors.
4. Conclusions
The results of our ID-TIMS, HR-SIMS, and LA-MC-ICPMS analyses demonstrate that the LKZ-1 zircon is nearly concordant in U–Pb age, with negligible common Pb content and a consistent Th/U ratio. LKZ-1 is fairly homogeneous in O–Hf isotopic and chemical composition. These consistencies suggest that LKZ-1 can be used as a suitable working standard for geochronological and isotopic/geochemical analyses. Fragments of the LKZ-1 zircon are available upon request from the lead author of this paper.
Supplementary Materials
The following are available online at https://www.mdpi.com/2075-163X/9/5/325/s1, Figure S1: Plot of 206Pb/238U dates and U concentrations for the SHRIMP spots of the LKZ-1 zircon. Figure S2: 176Hf/177Hf vs. 176Lu/177Hf plot for the LKZ-1 zircon. Table S1: SHRIMP U–Th–Pb results for the Plešovice zircon. Table S2: HR-SIMS (Cameca 1280) oxygen isotope data for the analyzed zircons. Table S3: LA-MC-ICPMS Lu–Yb–Hf isotopic results for the 91500 zircon. Table S4: LA-ICPMS results for the 91500 and LKZ-1 zircons.
Author Contributions
Conceptualization, A.C.-s.C.; methodology, Y.-J.J., S.L., K.Y., H.J.J., H.-S.L., C.P., N.K.K., X.-H.L. and S.L.K.; compiling and writing of the manuscript, A.C.-s.C.
Funding
This research was supported by a Korea Basic Science Institute grant (C39709) and a National Research Foundation of Korea grant funded by the government of Korea’s Ministry of Science and ICT (2016R1A2B4007283), awarded to A.C.C.
Acknowledgments
We are grateful for the assistance of Sook Ju Kim, Seon Gyu Kim, and Guo-Qiang Tang in the laboratory work. Insightful reviews by the editor and three anonymous journal reviewers improved the manuscript substantially.
Conflicts of Interest
The funders had no role in the design of the study; in the collection, analyses, or interpretation of data; in the writing of the manuscript, or in the decision to publish the results.
References
- Jeffery, P.G.; Hutchison, D. Chemical Methods of Rock Analysis, 3rd ed.; Pergamon Press: Oxford, UK, 1981; p. 379. [Google Scholar]
- Potts, P.J. Handbook of Rock Analysis; Viridian Publishing: Surrey, UK, 2003; p. 622. [Google Scholar]
- Bardo, J.; Ryerson, F.J.; Weber, P.K.; Ricolleau, A.; Fallon, S.J.; Hutcheon, I.D. Chemical imaging with NanoSIMS: A window into deep-Earth geochemistry. Earth Planet. Sci. Lett. 2007, 262, 543–551. [Google Scholar]
- Valley, J.W.; Cavosie, A.J.; Ushikubo, T.; Reinhard, D.A.; Lawrence, D.F.; Larson, D.J.; Clifton, P.H.; Kelly, T.F.; Wilde, S.A.; Moser, D.E.; et al. Hadean age for a post-magma-ocean zircon confirmed by atom-probe tomography. Nat. Geosci. 2014, 7, 219–223. [Google Scholar] [CrossRef]
- Davidson, J.P.; Morgan, D.J.; Charlier, B.L.A.; Harlou, R.; Hora, J.M. Microsampling and isotopic analysis of igneous rocks: Implications for the study of magmatic systems. Annu. Rev. Earth Planet. Sci. 2007, 35, 273–311. [Google Scholar] [CrossRef]
- Ireland, T.R.; Williams, I.S. Considerations in zircon geochronology by SIMS. Rev. Mineral. Geochem. 2003, 53, 215–241. [Google Scholar] [CrossRef]
- Valley, J.W. Oxygen isotopes in zircon. Rev. Mineral. Geochem. 2003, 53, 343–385. [Google Scholar] [CrossRef]
- Košler, J.; Sylvester, P.J. Present trends and the future of zircon in geochronology: Laser ablation ICP-MS. Rev. Mineral. Geochem. 2003, 53, 243–275. [Google Scholar] [CrossRef]
- Yokoyama, T.D.; Suzuki, T.; Kon, Y.; Hirata, T. Determinations of rare earth element abundance and U-Pb age of zircons using multisport laser ablation-inductively coupled plasma mass spectrometry. Anal. Chem. 2011, 83, 8892–8899. [Google Scholar] [CrossRef] [PubMed]
- Cherniak, D.J.; Watson, B. Diffusion in zircon. Rev. Mineral. Geochem. 2003, 53, 113–143. [Google Scholar] [CrossRef]
- Hawkesworth, C.J.; Kemp, A.I.S. Using hafnium and oxygen isotopes in zircons to unravel the record of crustal evolution. Chem. Geol. 2006, 226, 144–162. [Google Scholar] [CrossRef]
- Kemp, A.I.S.; Hawkesworth, C.J.; Paterson, B.A.; Kinny, P.D. Episodic growth of the Gondwana supercontinent from hafnium and oxygen isotopes in zircon. Nature 2006, 439, 580–583. [Google Scholar] [CrossRef]
- Jo, H.J.; Cheong, A.C.S.; Ryu, J.S.; Kim, N.; Yi, K.; Jung, H.; Li, X.H. In situ oxygen isotope records of crustal self-cannibalization selectively captured by zircon crystals from high-δ26Mg granitoids. Geology 2016, 44, 339–342. [Google Scholar] [CrossRef]
- Grimes, C.B.; John, B.E.; Kelemen, P.B.; Mazdab, F.K.; Wooden, J.L.; Cheadle, M.J.; Hanghøj, K.; Schwartz, J.J. Trace element chemistry of zircons from oceanic crust: A method for distinguishing detrital zircon provenance. Geology 2007, 35, 643–646. [Google Scholar] [CrossRef]
- Wiedenbeck, M.; Allé, P.; Corfu, F.; Griffin, W.L.; Meier, M.; Oberli, F.; von Quadt, A.; Roddick, J.C.; Speigel, W. Three natural zircon standards for U-Th-Pb, Lu-Hf, trace element and REE analyses. Geostand. Newslett. 1995, 19, 1–23. [Google Scholar] [CrossRef]
- Paces, J.B.; Miller, J.D., Jr. Precise U–Pb ages of Duluth Complex and related mafic intrusions, Northeastern Minnesota: geochronological insights to physical, petrogenic, paleomagnetic, and tectonomagmatic processes associated with the 1.1 Ga midcontinent rift system. J. Geophys. Res. 1993, 98, 13997–14013. [Google Scholar] [CrossRef]
- Black, L.P.; Kamo, S.L.; Allen, C.M.; Aleinikoff, J.N.; Davis, D.W.; Korsch, R.J.; Foudoulis, C. TEMORA 1: A new zircon standard for Phanerozoic U–Pb geochronology. Chem. Geol. 2003, 200, 155–170. [Google Scholar] [CrossRef]
- Black, L.P.; Kamo, S.L.; Allen, C.M.; Davis, D.W.; Aleinikoff, J.N.; Valley, J.W.; Mundil, R.; Campbell, I.H.; Korsch, R.J.; Williams, I.S.; et al. Improved 206Pb/238U microprobe geochronology by the monitoring of a trace-element-related matrix effect; SHRIMP, ID-TIMS, ELA-ICP-MS and oxygen isotope documentation for a series of zircon standards. Chem. Geol. 2004, 205, 115–140. [Google Scholar] [CrossRef]
- Sláma, J.; Košler, J.; Condon, D.J.; Crowley, J.L.; Gerdes, A.; Hanchar, J.M.; Horstwood, M.S.A.; Morris, G.A.; Nasdala, L.; Norberg, N.; et al. Plešovice zircon—A new natural reference material for U-Pb and Hf isotopic microanalysis. Chem. Geol. 2008, 249, 1–35. [Google Scholar] [CrossRef]
- Takehara, M.; Horie, K.; Hokada, T.; Kiyokawa, S. New insight into disturbance of U-Pb and trace-element systems in hydrothermally altered zircon via SHRIMP analyses of zircon from the Duluth Gabbro. Chem. Geol. 2018, 484, 168–178. [Google Scholar] [CrossRef]
- Jeong, Y.J.; Lee, S.; Kim, S.J.; Jo, H.J.; Yi, K.; Cheong, A.C.S. U–Th isotopic microanalysis of zircon reference materials and KBSI working standards. J. Anal. Sci. Technol. 2018, 9, 16. [Google Scholar] [CrossRef]
- Kim, S.J.; Lee, T.H.; Yi, K.; Jeong, Y.J.; Cheong, C.S. Characterization of new zircon and monazite working standards LKZ-1, BRZ-1, COM-1 and BRM-1. In Proceedings of the 1st Japan-Korea SHRIMP meeting, Higashi-Hiroshima, Japan, 14–16 September 2015. [Google Scholar]
- Williams, I.S. U-Th-Pb geochronology by ion microprobe. In Applications of Microanalytical Techniques to Understanding Mineralizing Processes; McKibben, M.A., Shanks, W.C., III, Rindley, W.I., Eds.; Society of Economic Geologists: Littleton, CO, USA, Reviews in Economic Geology; 1998; Volume 7, pp. 1–35. [Google Scholar]
- Ludwig, K.R. User’s manual for Squid 2.50; Berkeley Geochronology Center: Berkeley, CA, USA, Special Publication No. 5; 2009; 110p. [Google Scholar]
- Ludwig, K.R. User’s manual for Isoplot 3.6: A geochronological toolkit for Microsoft Excel; Berkeley Geochronology Center: Berkeley, CA, USA, Special Publication No. 4; 2008; 77p. [Google Scholar]
- Mattinson, J.M. Zircon U-Pb chemical abrasion (“CA-TIMS”) method: Combined annealing and multi-step partial dissolution analysis for improved precision and accuracy of zircon ages. Chem. Geol. 2005, 220, 47–66. [Google Scholar] [CrossRef]
- Krogh, T.E. A low contamination method for hydrothermal decomposition of zircon and extraction of U and Pb for isotopic age determinations. Geochim. Cosmochim. Acta 1973, 37, 485–494. [Google Scholar] [CrossRef]
- Gerstenberger, H.; Haase, G. A highly effective emitter substance for mass spectrometric Pb isotope ratio determinations. Chem. Geol. 1997, 136, 309–312. [Google Scholar] [CrossRef]
- Jaffey, A.H.; Flynn, K.F.; Glendenin, L.E.; Bentley, W.C.; Essling, A.M. Precision measurement of half-lives and specific activities of 235U and 238U. Phys. Rev. 1971, 4, 1889–1906. [Google Scholar]
- Ludwig, K.R. User’s manual for Isoplot 3.00: A geochronological toolkit for Microsoft Excel; Berkeley Geochronology Center: Berkeley, CA, USA, Special Publication No. 4; 2003; p. 71. [Google Scholar]
- Kim, N.K.; Kusakabe, M.; Park, C.; Lee, J.I.; Nagao, K.; Enokido, Y.; Yamashita, S.; Park, S.Y. An automated laser fluorination technique for high-precision analysis of three oxygen isotopes in silicates. Rapid Commun. Mass Sp. 2019, 33, 641–649. [Google Scholar] [CrossRef] [PubMed]
- Baertschi, P. Absolute 18O content of Standard Mean Ocean Water. Earth Planet. Sci. Lett. 1976, 31, 341–344. [Google Scholar] [CrossRef]
- Paton, C.; Hellstrom, J.; Paul, B.; Woodhead, J.; Hergt, J. Iolite: freeware for the visualisation and processing of mass spectrometric data. J. Anal. At. Spectrom. 2011, 26, 2508–2518. [Google Scholar] [CrossRef]
- Chu, N.C.; Taylor, R.N.; Chavagnac, V.; Nesbitt, R.W.; Boella, M.; Milton, J.A. Hf isotope ratio analysis using multi-collector inductively coupled plasma mass spectrometry: An evaluation of isobaric interference corrections. J. Anal. At. Spectrom. 2002, 17, 1567–1574. [Google Scholar] [CrossRef]
- Vervoort, J.D.; Patchett, P.J.; Söderlund, U.; Baker, M. Isotopic composition of Yb and the determination of Lu concentrations and Lu/Hf ratios by isotope dilution using MC-ICPMS. Geochem. Geophys. Geosyst. 2004, 5, Q11002. [Google Scholar] [CrossRef]
- Choi, M.S.; Cheong, C.S.; Kim, J.; Shin, H.S. Hafnium isotope analysis of mixed standard solutions by multi-collector inductively coupled plasma mass spectrometry: an evaluation of isobaric interference corrections. J. Anal. Sci. Technol. 2013, 4, 1. [Google Scholar] [CrossRef][Green Version]
- Iizuka, T.; Hirata, T. Improvements of precision and accuracy in in-situ Hf isotope microanalysis of zircon using the laser ablation-MC-ICPMS technique. Chem. Geol. 2005, 220, 121–137. [Google Scholar] [CrossRef]
- Scherer, E.; Münker, C.; Mezger, K. Calibration of the lutetium-hafnium clock. Science 2001, 293, 683–687. [Google Scholar] [CrossRef] [PubMed]
- Blichert-Toft, J.; Albarède, F. The Lu-Hf isotope geochemistry of chondrites and the evolution of the mantle-crust system. Earth Planet. Sci. Lett. 1997, 148, 243–258. [Google Scholar] [CrossRef]
- Guillong, M.; Günther, D. Effect of particle size distribution on ICP-induced elemental fractionation in laser ablation-inductively coupled plasma-mass spectrometry. J. Anal. At. Spectrom. 2002, 17, 831–837. [Google Scholar] [CrossRef]
- Jochum, K.P.; Weis, U.; Stoll, B.; Kuzmin, D.; Yang, Q.; Raczek, I.; Jacob, D.E.; Stracke, A.; Birbaum, K.; Frick, D.A.; et al. Determination of reference values for NIST SRM 610-617 glasses following ISO guidelines. Goestan. Geoanal. Res. 2011, 35, 397–429. [Google Scholar] [CrossRef]
- Stacey, J.S.; Kramers, J.D. Approximation of terrestrial lead isotope evolution by a two-stage model. Earth Planet. Sci. Lett. 1975, 26, 207–221. [Google Scholar] [CrossRef]
- Claoué-Long, J.C.; Compston, W.; Roberts, J.; Fanning, C.M. Two Carboniferous ages: A comparison of SHRIMP zircon dating with conventional zircon ages and 40Ar/39Ar analysis. In Geochronology Time Scales and Global Stratigraphic Correlation; Berggren, W.A., Kent, D.V., Aubrey, M.-P., Hardenbol, J., Eds.; Society for Sedimentary Geology Special Publication: Tulsa, OK, USA, 1995; Volume 54, pp. 3–21. [Google Scholar]
- Mattinson, J.M. U–Pb ages of zircons: A basic examination of error propagation. Chem. Geol. 1987, 66, 151–162. [Google Scholar] [CrossRef]
- Black, L.P.; Kamo, S.L.; Williams, I.S.; Mundil, R.; Davis, D.W.; Korsch, R.J.; Foudoulis, C. The application of SHRIMP to Phanerozoic geochronology; a critical appraisal of four zircon standards. Chem. Geol. 2003, 200, 171–188. [Google Scholar] [CrossRef]
- Li, X.H.; Long, W.G.; Li, Q.L.; Liu, Y.; Zheng, Y.F.; Yang, Y.H.; Chamberlain, K.R.; Wan, D.F.; Guo, C.H.; Wang, X.C.; et al. Penglai zircon megacrysts: A potential new working reference microbeam determination of Hf–O isotopes and U–Pb age. Geostand. Geoanal. Res. 2010, 34, 117–134. [Google Scholar] [CrossRef]
- Wiedenbeck, M.; Hanchar, J.M.; Peck, W.H.; Sylvester, P.; Valley, J.; Whitehouse, M.; Kronz, A.; Morishita, Y.; Nasdala, L.; Fiebig, J.; et al. Further characterisation of the 91500 zircon crystal. Geostand. Geoanal. Res. 2004, 28, 9–39. [Google Scholar] [CrossRef]
- Blichert-Toft, J. The Hf isotopic composition of zircon reference material 91500. Chem. Geol. 2008, 253, 252–257. [Google Scholar] [CrossRef]
- Belousova, E.A.; Griffin, W.L.; O’Reilly, S.Y.; Fisher, N.I. Igneous zircon: Trace element composition as an indicator of source rock type. Contrib. Mineral. Petrol. 2002, 143, 602–622. [Google Scholar] [CrossRef]
- Sano, Y.; Terada, K.; Fukuoka, T. High mass resolution ion microprobe analysis of rare earth elements in silicate glass, apatite and zircon: lack of matrix dependency. Chem. Geol. 2002, 184, 217–230. [Google Scholar] [CrossRef]
- Iizuka, T.; Hirata, T. Simultaneous determinations of U–Pb age and REE abundances for zircons using ArF excimer laser ablation-ICPMS. Geochem. J. 2004, 38, 229–241. [Google Scholar] [CrossRef]
- McDonough, W.F.; Sun, S.S. The composition of the Earth. Chem. Geol. 1995, 120, 223–253. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).