Characterization of Microbial Communities Associated with Ceramic Raw Materials as Potential Contributors for the Improvement of Ceramic Rheological Properties
Abstract
1. Introduction
2. Materials and Methods
2.1. Sampling
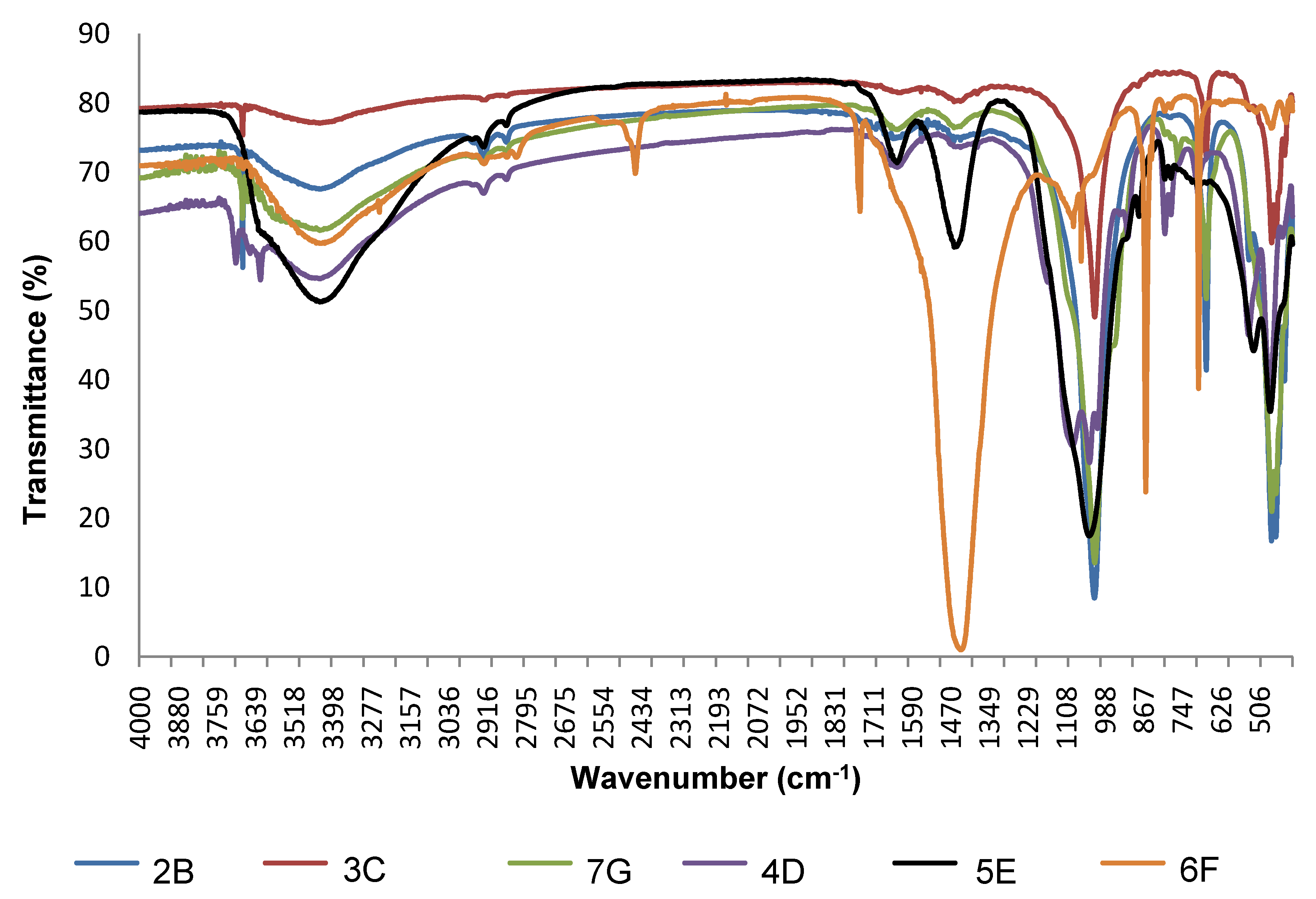
2.2. Mineralogical Characterization by FTIR
2.3. Identification of Microorganisms by Culture Methods
2.4. Identification of Microorganisms by Molecular Biology Techniques
2.4.1. DNA Extraction and PCR Amplification
2.4.2. Clone Library Construction
2.4.3. Sequence Comparisons and Phylogenetic Analysis
3. Results
3.1. Mineralogical Composition of Ceramic Raw Materials
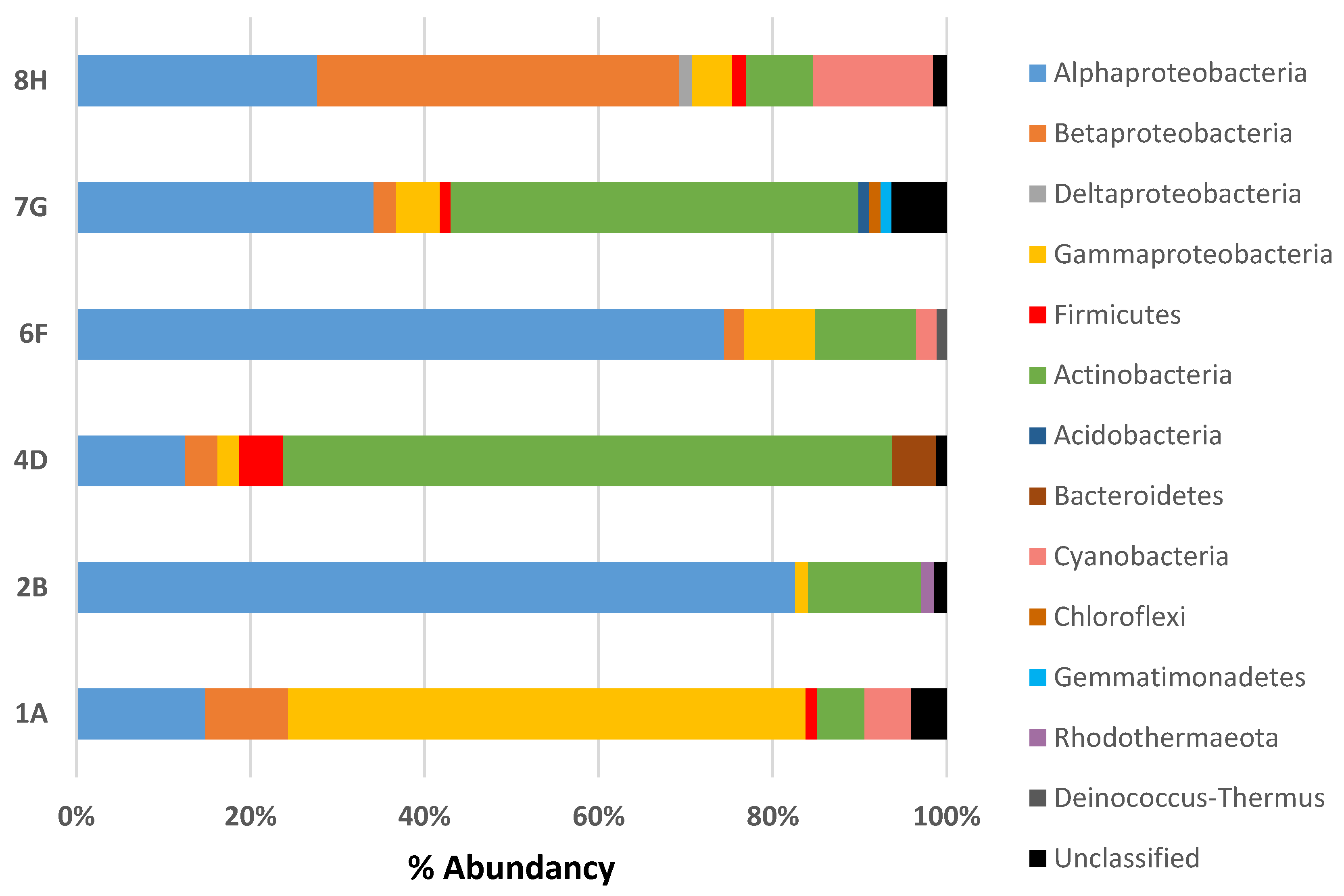
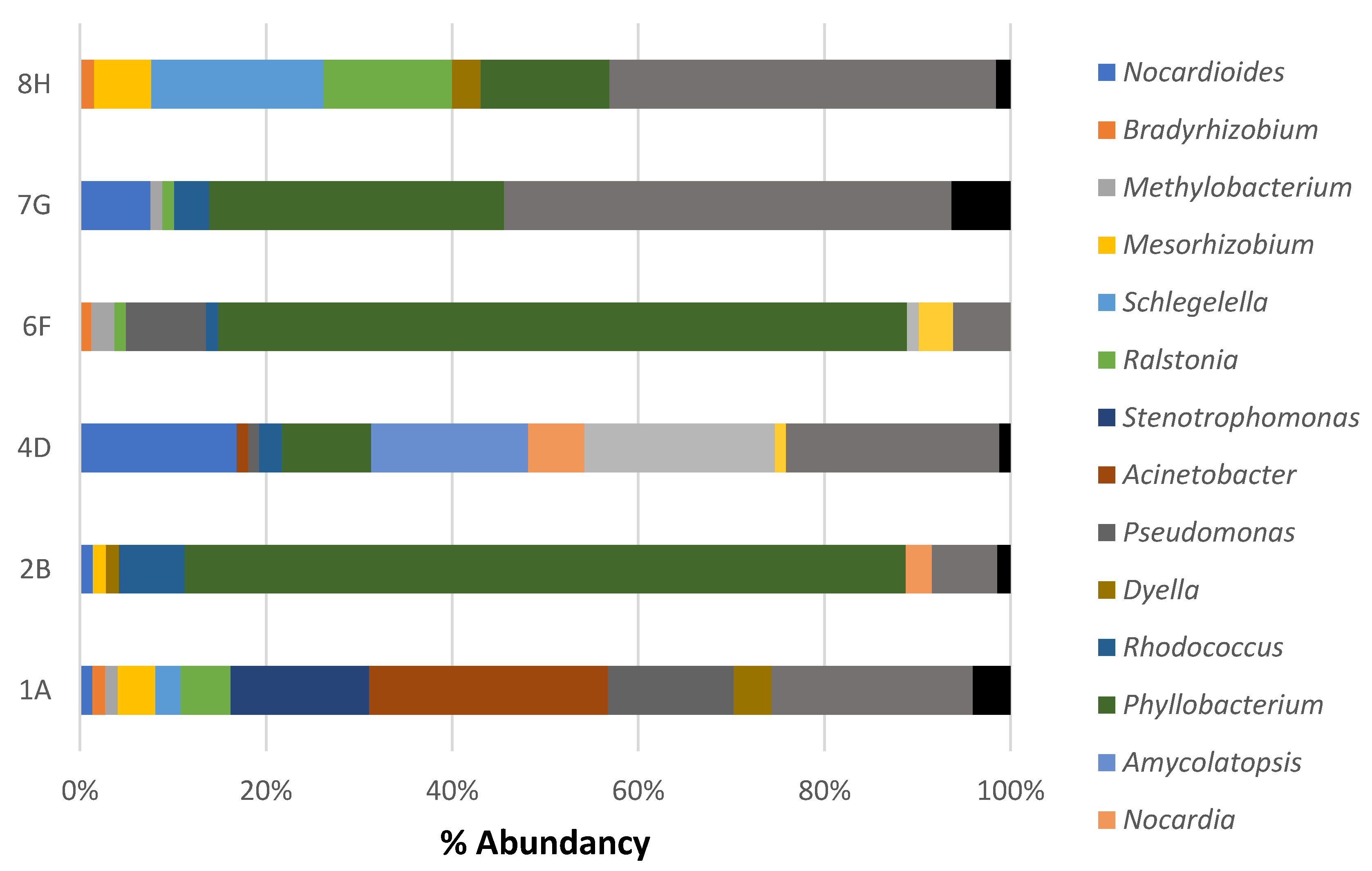
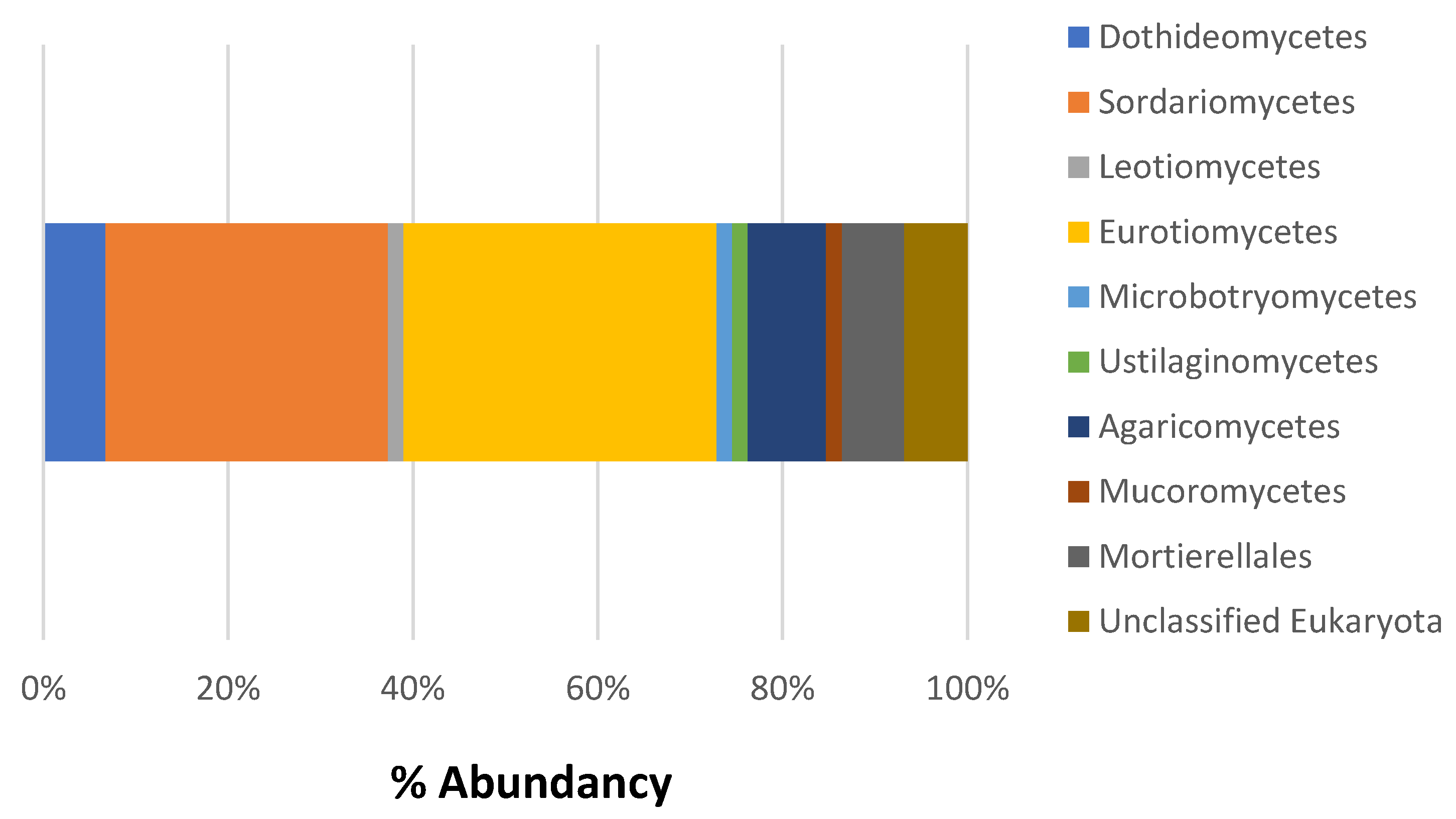
3.2. Composition of Microbial Communities Associated with Ceramic Raw Materials
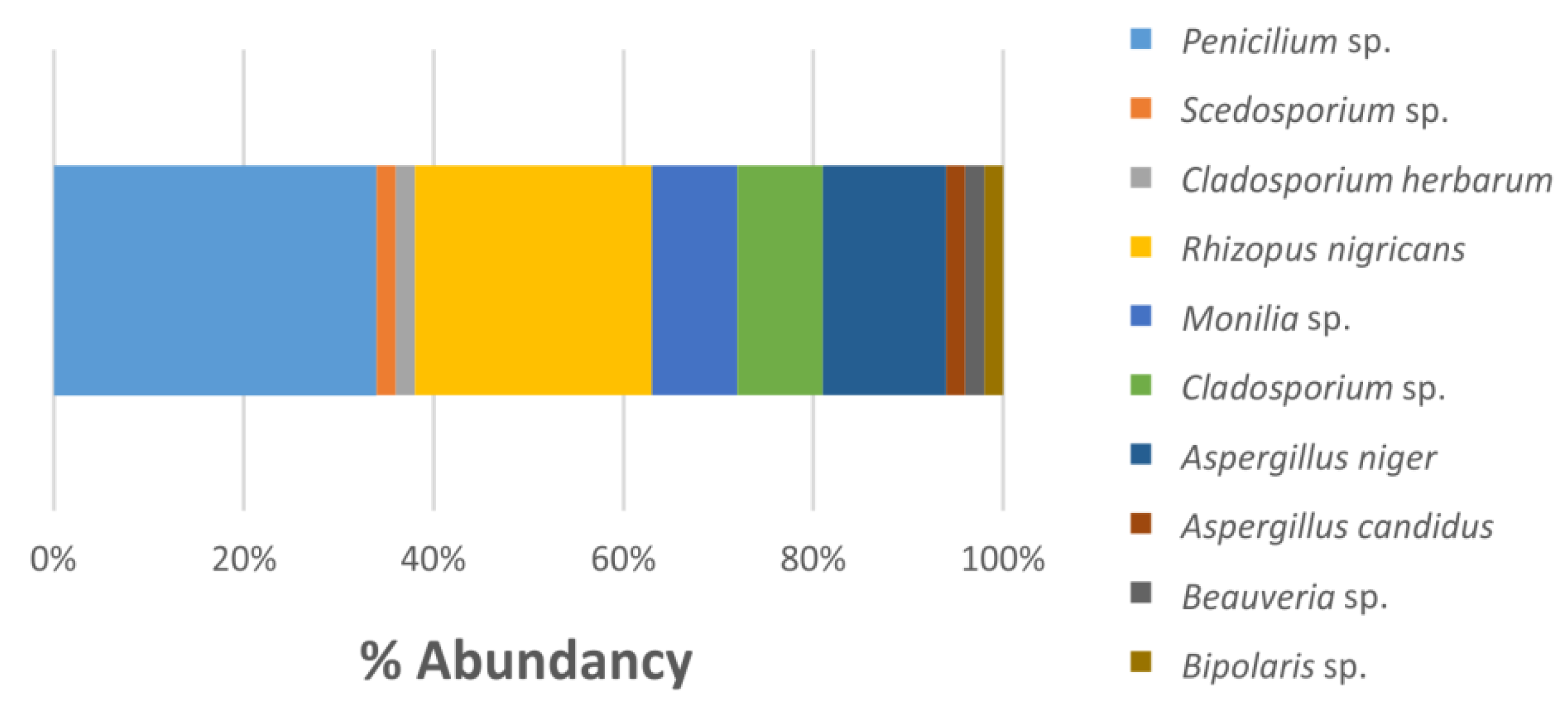
3.3. Isolated Microorganisms
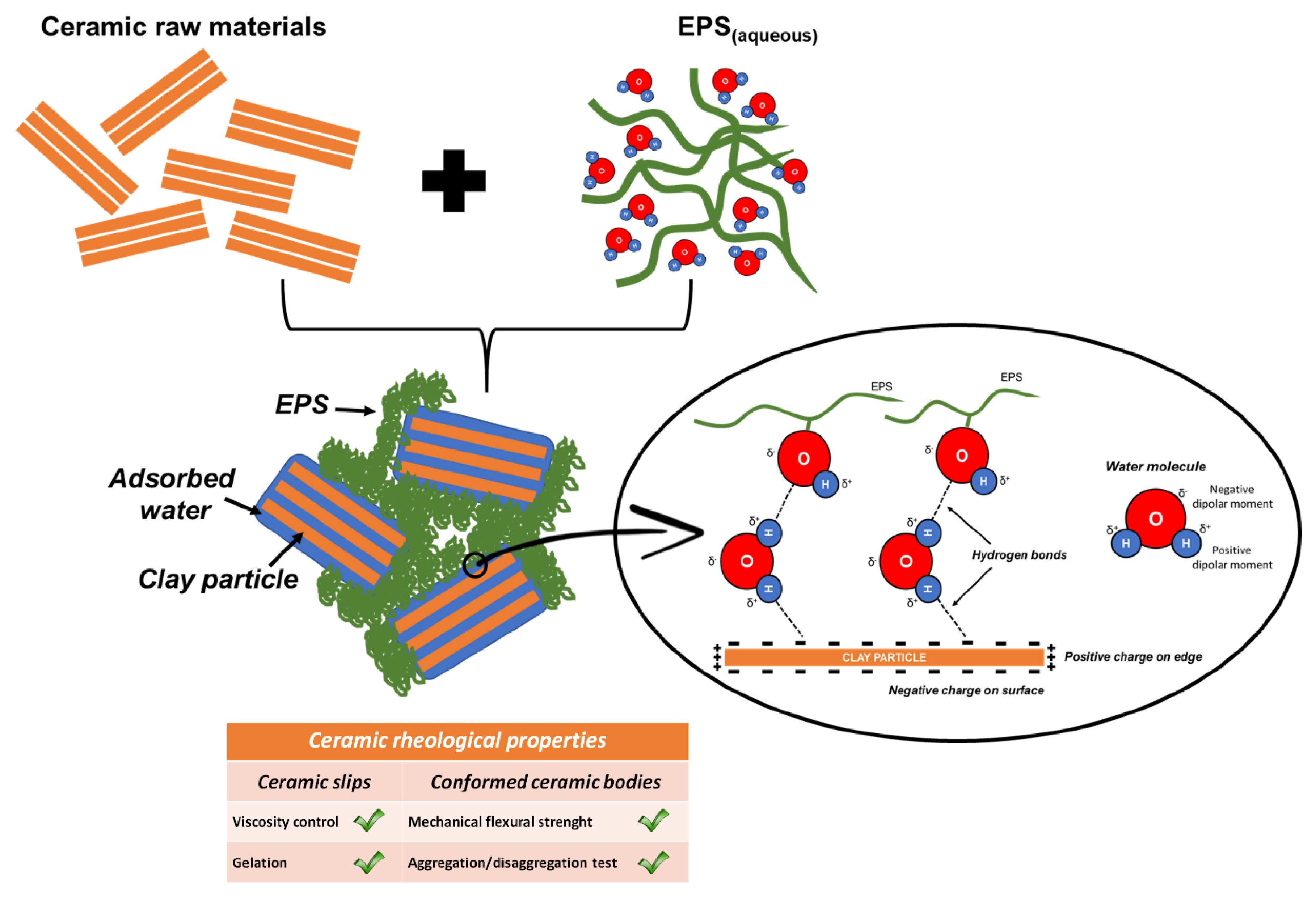
4. Discussion
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Evans, J.R.G. Seventy ways to make ceramics. J. Eur. Ceram. Soc. 2008, 28, 1421–1432. [Google Scholar] [CrossRef]
- Flemming, H.C.; Wingender, J. Relevance of microbial extracellular polymeric substances (EPSs)-Part II: Technical aspects. Water Sci. Technol. 2001, 43, 9–16. [Google Scholar] [CrossRef]
- Ates, O. Systems biology of microbial exopolysaccharides production. Front. Bioeng. Biotechnol. 2015, 3, 200. [Google Scholar] [CrossRef] [PubMed]
- Moscovici, M. Present and future medical applications of microbial exopolysaccharides. Front. Microbiol. 2015, 6, 1012. [Google Scholar] [CrossRef] [PubMed]
- Castillo, N.A.; Valdez, A.L.; Fariña, J.I. Microbial production of scleroglucan and downstream processing. Front. Microbiol. 2015, 15, 1106. [Google Scholar] [CrossRef] [PubMed]
- Russo, P.; de Chiara, M.L.V.; Capozzi, V.; Arena, M.P.; Amodio, M.L.; Rascòn, A.; Dueñas, M.T.; Lòpez, P.; Spano, G. Lactobacillus plantarum strains for multifunctional oat-based foods. Food Sci. Technol. 2016, 68, 288–294. [Google Scholar] [CrossRef]
- Ripari, V. Techno-functional role of exopolysaccharides in cereal-based, yogurt-like beverages. Beverages 2019, 5, 16. [Google Scholar] [CrossRef]
- Frank, J.A.; Reich, C.I.; Sharma, S.; Weisbaum, J.S.; Wilson, B.A.; Olsen, G.J. Critical evaluation of two primers commonly used for amplification of bacterial 16S rRNA genes. Appl. Environ. Microb. 2008, 74, 2461–2470. [Google Scholar] [CrossRef] [PubMed]
- Schabereiter-Gurtner, C.; Lubitz, W.; Rölleke, S. Application of broad-range 16S rRNA PCR amplification and DGGE fingerprinting for detection of tick-infecting bacteria. J. Microbiol. Methods 2003, 52, 251–260. [Google Scholar] [CrossRef]
- White, T.J.; Bruns, T.; Lee, S.; Taylor, J. Amplification and direct sequencing of fungal ribosomal RNA genes for phylogenetics. In PCR Protocols, A Guide to Methods and Applications; Innis, M.A., Gelfand, D.H., Sninsky, J.J., White, T.J., Eds.; Academic Press: New York, NY, USA, 1990; pp. 315–322. [Google Scholar]
- Schloss, P.C.; Westcott, S.L.; Ryabin, T.; Hall, J.R.; Hartmann, M.; Hollister, E.B.; Lesniewski, R.A.; Oakley, B.B.; Parks, D.H.; Robinson, C.J.; et al. Introducing mothur: Open source, platform-independent, community-supported software for describing and comparing microbial communities. Appl. Environ. Microb. 2009, 75, 7537–7541. [Google Scholar] [CrossRef]
- Altschul, S.; Gish, W.; Miller, W.; Myers, E.; Lipman, D. Basic local alignment search tool. J. Mol. Biol. 1990, 215, 403–410. [Google Scholar] [CrossRef]
- Yoon, S.H.; Ha, S.M.; Kwon, S.; Lim, J.; Kim, Y.; Seo, H.; Chun, J. Introducing EzBioCloud: A taxonomically united database of 16S rRNA gene sequences and whole-genome assemblies. Int. J. Syst. Evol. Microbiol. 2017, 67, 1613–1617. [Google Scholar] [PubMed]
- Lagadic, L.; Mitchell, M.K.; Payne, B.D. Highly effective adsorption of heavy metal tons by a thiol-functionalized magnesium phyllosilicate clay. Environ. Sci. Technol. 2001, 35, 84–90. [Google Scholar] [CrossRef]
- Saikia, B.; Parthasarathy, G. Fourier transform infrared spectroscopic characterization of kaolinite from Assam and Meghalaya, Northeastern India. J. Mod. Phys. 2010, 1, 206–210. [Google Scholar] [CrossRef]
- Reddy, T.R.; Kaneko, S.; Endo, T.; Reddy, S.L. Spectroscopic characterization of bentonite. J. Laser Opt. Photonics 2017, 4, 171. [Google Scholar]
- Sabet, M.; Salavati-Niasari, M.; Asgari Fard, Z. Synthesis and characterization of barium carbonate nanostructures via simple hydrothermal method. Synth. React. Inorg. M. 2016, 46, 317–322. [Google Scholar] [CrossRef]
- Janssen, P.H. Identifying the Dominant Soil Bacterial Taxa in Libraries of 16S rRNA and 16S rRNA Genes. Appl. Environ. Microb. 2006, 72, 1719–1728. [Google Scholar] [CrossRef]
- Labbé, D.; Margesin, R.; Schinner, F.; Whyte, L.G.; Greer, C.W. Comparative phylogenetic analysis of microbial communities in pristine and hydrocarbon-contaminated Alpine soils. FEMS Microbiol. Ecol. 2007, 59, 466–475. [Google Scholar] [CrossRef] [PubMed]
- Niepceron, M.; Martin-Laurent, F.; Crampon, M.; Portet-Koltalo, F.; Akpa-Vinceslas, M.; Legras, M.; Bru, D.; Bureau, F.; Bodilis, J. GammaProteobacteria as a potential bioindicator of a multiple contamination by polycyclic aromatic hydrocarbons (PAHs) in agricultural soils. Environ. Pollut. 2013, 180, 199–205. [Google Scholar] [CrossRef]
- Das, N.; Tripathi, N.; Basu, S.; Bose, C.; Maitra, S.; Khurana, S. Progress in the development of gelling agents for improved culturability of microorganisms. Front. Microbiol. 2015, 6, 698. [Google Scholar] [CrossRef]
- Osmałek, T.; Froelich, A.; Tasarek, S. Application of gellan gum in pharmacy and medicine. Int. J. Pharm. 2014, 466, 328–340. [Google Scholar] [CrossRef]
- Miller, A.Z.; Hernández-Mariné, M.; Jurado, V.; Dionísio, A.; Barquinha, P.; Fortunato, E.; Afonso, M.J.; Chaminé, H.I.; Saiz-Jimenez, C. Enigmatic reticulated filaments in subsurface granite. Env. Microbiol. Rep. 2012, 4, 596–603. [Google Scholar] [CrossRef] [PubMed]
- Miller, A.Z.; Garcia-Sanchez, A.M.; Martin-Sanchez, P.M.; Pereira, M.F.C.; Spangenberg, J.E.; Jurado, V.; Dionisio, A.; Afonso, M.J.; Chaminé, H.I.; Hermosin, B.; et al. Origin of abundant moonmilk deposits in a subsurface granitic environment. Sedimentology 2018, 65, 1482–1503. [Google Scholar] [CrossRef]
- Djuric, A.; Gojgic-Cvijovic, G.; Jakovljevic, D.; Kekez, B.; Kojic, J.S.; Mattinen, M.L.; Harju, I.E.; Vrvic, M.M.; Beskoski, V.P. Brachybacterium sp. CH-KOV3 isolated from an oil-polluted environment—A new producer of levan. Int. J. Biol. Macromol. 2017, 104, 311–321. [Google Scholar] [CrossRef]
- Broszat, M.; Nacke, H.; Blasi, R.; Siebe, C.; Huebner, J.; Daniel, R.; Grohmann, E. Wastewater irrigation increases the abundance of potentially harmful Gammaproteobacteria in soils in Mezquital Valley, Mexico. Appl. Environ. Microb. 2014, 80, 5282–5291. [Google Scholar] [CrossRef] [PubMed]
- Paul, S.; Dukare, A.S.; Bandeppa; Manjunatha, B.S.; Annapurna, K. Plant growth-promoting rhizobacteria for abiotic stress alleviation in crops. In Advances in Soil Microbiology: Recent Trends and Future Prospects. Microorganisms for Sustainability; Adhya, T., Mishra, B., Annapurna, K., Verma, D., Kumar, U., Eds.; Springer: Singapore, 2017; Volume 4. [Google Scholar]
- Roberson, E.B.; Firestone, M.K. Relationship between desiccation and exopolysaccharide production in a soil Pseudomonas spp. Appl. Environ. Microb. 1992, 58, 1284–1291. [Google Scholar]
- Kaushal, M.; Wani, S.P. Plant-growth-promoting rhizobacteria: Drought stress alleviators to ameliorate crop production in drylands. Ann. Microbiol. 2016, 66, 35–42. [Google Scholar] [CrossRef]
- Chun, J.; Oren, A.; Ventosa, A.; Christensen, H.; Arahal, D.; da Costa, M.; Rooney, A.; Yi, H.; Xu, X.; De Meyer, S.; et al. Proposed minimal standards for the use of genome data for the taxonomy of prokaryotes. Int. J. Syst. Evol. Microbiol. 2018, 68, 461–466. [Google Scholar] [CrossRef]
- Fox, G.E.; Wisotzkey, J.D.; Jurtshuk, P., Jr. How close is close: 16s rRNA sequence identity may not be sufficient to guarantee species identity. Int. J. Syst. Bacteriol. 1992, 42, 166–170. [Google Scholar] [CrossRef] [PubMed]
- Bent, S.J.; Forney, L.J. The tragedy of the uncommon: Understanding limitations in the analysis of microbial diversity. ISME J. 2008, 2, 689–695. [Google Scholar] [CrossRef] [PubMed]
- Xie, F.H.; Ma, S.; Quan, D.; Liu, G.; Chao, C.Y.; Qian, S. Pseudomonas kunmingensis sp. nov., an exopolysaccharide-producing bacterium isolated from a phosphate mine. Int. J. Syst. Evol. Microbiol. 2014, 64, 559–564. [Google Scholar] [CrossRef] [PubMed]
- Maalej, H.; Boisset, C.; Hmidet, N.; Buon, L.; Heyraud, A.; Nasri, M. Purification and structural data of a highly substituted exopolysaccharide from Pseudomonas stutzeri AS22. Carbohydr. Polym. 2014, 112, 404–411. [Google Scholar] [CrossRef] [PubMed]
- Wu, S.; Zheng, R.; Sha, Z.; Sun, C. Genome Sequence of Pseudomonas stutzeri 273 and Identification of the Exopolysaccharide EPS273 Biosynthesis Locus. Mar. Drugs 2017, 15, 218. [Google Scholar] [CrossRef] [PubMed]
- Hultberg, M.; Bengtsson, T.; Liljeroth, E. Late blight on potato is suppressed by the biosurfactant-producing strain Pseudomonas koreensis 2.74 and its biosurfactant. Biocontrol 2010, 55, 543–550. [Google Scholar] [CrossRef]
- Hultberg, M.; Alsberg, T.; Khalil, S.; Alsanius, B. Suppression of disease in tomato infected by Pythium ultimum with a biosurfactant produced by Pseudomonas koreensis. Biocontrol 2010, 55, 435–444. [Google Scholar] [CrossRef]
- Toribio, J.; Escalante, A.E.; Caballero-Mellado, J.; González-González, A.; Zavala, S.; Souza, V.; Soberón-Chávez, G. Characterization of a novel biosurfactant producing Pseudomonas koreensis lineage that is endemic to Cuatro Ciénegas Basin. Syst. Appl. Microbiol. 2011, 34, 531–535. [Google Scholar] [CrossRef] [PubMed]
- Babu, A.G.; Shea, P.J.; SudhakaraIk-BooJung, D.; Oh, B.T. Potential use of Pseudomonas koreensis AGB-1 in association with Miscanthus sinensis to remediate heavy metal(loid)-contaminated mining site soil. J. Environ. Manag. 2015, 151, 160–166. [Google Scholar] [CrossRef]
- Anbu, P. Characterization of an extracellular lipase by Pseudomonas koreensis BK-L07 isolated from soil. Prep. Biochem. Biotech. 2014, 44, 266–280. [Google Scholar] [CrossRef]
- Mujumdar, S.; Joshi, P.; Karve, N. Production, characterization, and applications of bioemulsifiers (BE) and biosurfactants (BS) produced by Acinetobacter spp.: A review. J. Basic Microb. 2019, 59, 277–287. [Google Scholar] [CrossRef]
- Jagtap, S.; Yavankar, S.; Pardesi, K.; Chopade, B. Production of bioemulsifier by Acinetobacter species isolated from healthy human skin. Indian J. Exp. Biol. 2010, 48, 70–76. [Google Scholar]
- Mohamed, S.S.; Amer, S.K.; Selim, M.S.; Rifaat, H.M. Characterization and applications of exopolysaccharide produced by marine Bacillus altitudinis MSH2014 from Ras Mohamed, Sinai, Egypt. Egypt. J. Basic Appl. Sci. 2018, 5, 204–209. [Google Scholar] [CrossRef]
- Kiliç, N.K.; Dönmez, G. Environmental conditions affecting exopolysaccharide production by Pseudomonas aeruginosa, Micrococcus sp., and Ochrobactrum sp. J. Hazard Mater. 2008, 154, 1019–1024. [Google Scholar] [CrossRef] [PubMed]
- Boretska, M.; Bellenberg, S.; Moshynets, O.; Pokholenko, I.; Sand, W. Change of extracellular polymeric substances composition of Thiobacillus thioparus in presence of sulfur and steel. J. Microb. Biochem. Technol. 2013, 5, 68–73. [Google Scholar] [CrossRef]
- Ojeda, J.J.; Romero-González, M.E.; Banwart, S.A. Analysis of bacteria on steel surfaces using reflectance Micro-Fourier Transform Infrared Spectroscopy. Anal. Chem. 2009, 81, 6467–6473. [Google Scholar] [CrossRef]
- Ge, X.; Kitten, T.; Chen, Z.; Lee, S.P.; Munro, C.L.; Xu, P. Identification of Streptococcus sanguinis genes required for biofilm formation and examination of their role in endocarditis virulence. Infect. Immun. 2008, 76, 2551–2559. [Google Scholar] [CrossRef] [PubMed]
- Belgini, D.R.B.; Dias, R.S.; Siqueira, V.M.; Valadares, L.A.B.; Albanese, J.M.; Souza, R.S.; Torres, A.P.R.; Sousa, M.P.; Silva, C.C.; De Paula, S.O.; et al. Culturable bacterial diversity from a feed water of a reverse osmosis system, evaluation of biofilm formation and biocontrol using phages. World J. Microb. Biotechnol. 2014, 30, 2689–2700. [Google Scholar] [CrossRef]
- Kumar, C.G.; Sujitha, P. Kocuran, an exopolysaccharide isolated from Kocuria rosea strain BS-1 and evaluation of its in vitro immunosuppression activities. Enzyme Microb. Technol. 2014, 55, 113–120. [Google Scholar] [CrossRef] [PubMed]
- Gu, D.; Jiao, Y.; Wu, J.; Liu, Z.; Chen, Q. Optimization of EPS production and characterization by a halophilic bacterium, Kocuria rosea ZJUQH from Chaka Salt Lake with response surface methodology. Molecules 2017, 22, 814. [Google Scholar] [CrossRef] [PubMed]
- Öner, E.T.; Hernández, L.; Combie, J. Review of Levan polysaccharide: From a century of past experiences to future prospects. Biotechnol. Adv. 2016, 34, 827–844. [Google Scholar] [CrossRef]
- Costa, R.R.; NetoI, A.I.; Calgeris, I.; Correia, C.R.; Pinho, A.C.M.; Fonseca, J.; Toksoy Öner, E.; Mano, J.F. Adhesive nanostructured multilayer films using a bacterial exopolysaccharide for biomedical applications. J. Mater. Chem. 2013, 1, 2367–2374. [Google Scholar] [CrossRef]
- Chen, X.; Gao, H.; Ploehn, H.J. Montmorillonite-levan nanocomposites with improved thermal and mechanical properties. Carbohydr. Polym. 2014, 101, 565–573. [Google Scholar] [CrossRef]
- Hong, B.H.; Joe, M.M.; Selvakumar, G.; Kim, K.Y.; Choi, J.H.; Sa, T.M. Influence of salinity variations on exocellular polysaccharide production, biofilm formation and flocculation in halotolerant bacteria. J. Environ. Biol. 2017, 38, 657–664. [Google Scholar] [CrossRef]
- Rusznyák, A.; Akob, D.M.; Nietzsche, S.; Eusterhues, K.; Totsche, K.U.; Neu, T.R.; Frosch, T.; Popp, J.; Keiner, R.; Geletneky, J.; et al. Calcite biomineralization by bacterial isolates from the recently discovered pristine karstic herrenberg cave. Appl. Environ. Microb. 2012, 78, 1157–1167. [Google Scholar] [CrossRef]
- Badireddy, A.R.; Chellam, S.; Yanina, S.; Gassman, P.; Rosso, K.M. Bismuth dimercaptopropanol (BisBAL) inhibits the expression of extracellular polysaccharides and proteins by Brevundimonas diminuta: Implications for membrane microfiltration. Biotechnol. Bioeng. 2008, 99, 634–643. [Google Scholar] [CrossRef]
- Zhou, F.; Wu, Z.; Jin Han, C.C.; Ai, L.; Guo, B. Exopolysaccharides produced by Rhizobium radiobacter S10 in whey and their rheological properties. Food Hydrocoloids 2014, 36, 362–368. [Google Scholar] [CrossRef]
- Kohama, T.; Fujimoto, M.; Kuninaka, A.; Yoshino, H. Structure of malonogalactan, an acidic polysaccharide of Penicillium citrinum. Agric. Biol. Chem. 1974, 38, 127–134. [Google Scholar]
- Guo, S.; Mao, W.; Li, Y.; Tian, J.; Xu, J. Structural elucidation of the exopolysaccharide produced by fungus Fusarium oxysporum Y24-2. Carbohydr. Res. 2013, 365, 9–13. [Google Scholar] [CrossRef]
- Seviour, R.J.; Stasinopoulos, S.J.; Auer, D.P.F.; Gibbs, P.A. Production of pullulan and other exopolysaccharides by filamentous fungi. Crit. Rev. Biotechnol. 1992, 12, 279–298. [Google Scholar] [CrossRef]
- Pitson, S.M.; Seviour, R.J.; McDougall, B.M. Effect of carbon source on extracellular (1 → 3)- and (1 → 6)-β-glucanase production by Acremonium persicinum. Can. J. Microbiol. 1997, 43, 432–439. [Google Scholar] [CrossRef]
- Takita, J.; Katohda, S.; Sugiyama, H. Structural determination of an exocellular mannan from Rhodotorula mucilaginosa YR-2 using ab initio assignment of proton and carbon NMR spectra. Carbohydr. Res. 2001, 335, 133–139. [Google Scholar] [CrossRef]
- Vazquez-Rodriguez, A.; Vasto-Anzaldo, X.G.; Barboza Perez, D.; Vázquez-Garza, E.; Chapoy-Villanueva, H.; García-Rivas, G.; Garza-Cervantes, J.A.; Gómez-Lugo, J.J.; Gomez-Loredo, A.E.; Garza Gonzalez, M.T.; et al. Microbial competition of Rhodotorula mucilaginosa UANL-001L and E. coli increase biosynthesis of non-toxic exopolysaccharide with applications as a wide-spectrum antimicrobial. Sci. Rep. 2018, 8, 798. [Google Scholar] [CrossRef] [PubMed]
- Yeh, C.W.; Zang, C.Z.; Lin, C.C.; Kan, S.C.; Chang, W.F.; Shieh, C.J.; Liu, Y.C. Quantitative and morphologic analysis on exopolysaccharide and biomass production from a truffle endophytic fungus Hypocreales sp. NCHU01. J. Taiwan Inst. Chem. Eng. 2014, 45, 108–114. [Google Scholar] [CrossRef]
- Orlandelli, R.C.; Vasconcelos, A.F.D.; Azevedo, J.L.; Corradi da Silva, M.L.; Pamphile, J.A. Screening of endophytic sources of exopolysaccharides: Preliminary characterization of crude exopolysaccharide produced by submerged culture of Diaporthe sp. JF766998 under different cultivation time. Biochim. Open 2016, 2, 33–40. [Google Scholar] [CrossRef] [PubMed]
- Leathers, T.D. Biotechnological production and applications of pullulan. Appl. Microbiol. Biotechnol. 2003, 62, 468–473. [Google Scholar] [CrossRef] [PubMed]
- Singh, R.S.; Sainia, G.K.; Kennedy, J.F. Pullulan: Microbial sources, production and applications. Carbohydr. Polym. 2008, 4, 515–531. [Google Scholar] [CrossRef] [PubMed]
| Sample ID | Sample Description | Source |
|---|---|---|
| 1A | Ceramic suspension collected from the mill used to mix and mill all the raw materials with water | Rauschert Portuguesa SA. |
| 2B | Talc 1 collected from individual packing bag * | Rauschert Portuguesa SA. |
| 3C | Talc 2 collected from individual packing bag * | Rauschert Portuguesa SA. |
| 4D | Clay collected from individual packing bag * | Rauschert Portuguesa SA. |
| 5E | Bentonite collected from individual packing bag * | Rauschert Portuguesa SA. |
| 6F | Barium carbonate collected from individual packing bag * | Rauschert Portuguesa SA. |
| 7G | Talc 3 collected from individual packing bag * | Rauschert Portuguesa SA. |
| 8H | Ceramic suspension collected from a mixing tank used to prepare the ceramic slip for further spray-drying | Rauschert Portuguesa SA. |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Garcia-Sanchez, A.M.; Machado-Moreira, B.; Freire, M.; Santos, R.; Monteiro, S.; Dias, D.; Neves, O.; Dionísio, A.; Miller, A.Z. Characterization of Microbial Communities Associated with Ceramic Raw Materials as Potential Contributors for the Improvement of Ceramic Rheological Properties. Minerals 2019, 9, 316. https://doi.org/10.3390/min9050316
Garcia-Sanchez AM, Machado-Moreira B, Freire M, Santos R, Monteiro S, Dias D, Neves O, Dionísio A, Miller AZ. Characterization of Microbial Communities Associated with Ceramic Raw Materials as Potential Contributors for the Improvement of Ceramic Rheological Properties. Minerals. 2019; 9(5):316. https://doi.org/10.3390/min9050316
Chicago/Turabian StyleGarcia-Sanchez, Angela M., Bernardino Machado-Moreira, Mário Freire, Ricardo Santos, Sílvia Monteiro, Diamantino Dias, Orquídia Neves, Amélia Dionísio, and Ana Z. Miller. 2019. "Characterization of Microbial Communities Associated with Ceramic Raw Materials as Potential Contributors for the Improvement of Ceramic Rheological Properties" Minerals 9, no. 5: 316. https://doi.org/10.3390/min9050316
APA StyleGarcia-Sanchez, A. M., Machado-Moreira, B., Freire, M., Santos, R., Monteiro, S., Dias, D., Neves, O., Dionísio, A., & Miller, A. Z. (2019). Characterization of Microbial Communities Associated with Ceramic Raw Materials as Potential Contributors for the Improvement of Ceramic Rheological Properties. Minerals, 9(5), 316. https://doi.org/10.3390/min9050316









