Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings
Abstract
1. Introduction
2. Materials and Methods
2.1. Manganese Nodule Sample
2.2. Tailings
2.3. Reagents Used—Leaching Parameters
2.4. Experimental Design
2.5. Effect of Stirring Speed
2.6. Effect of the MnO2/Fe2O3 Ratio
3. Results and Discussion
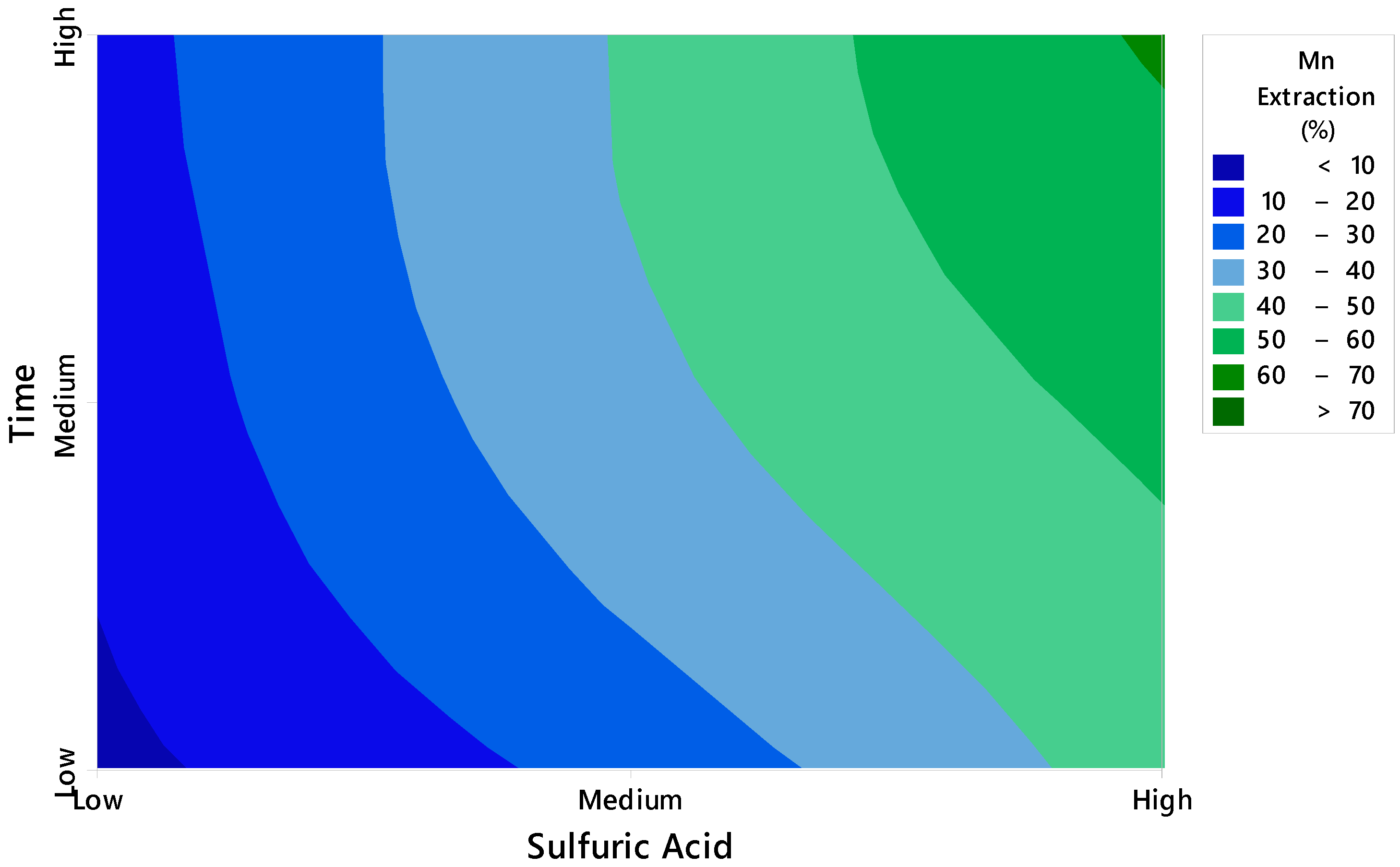
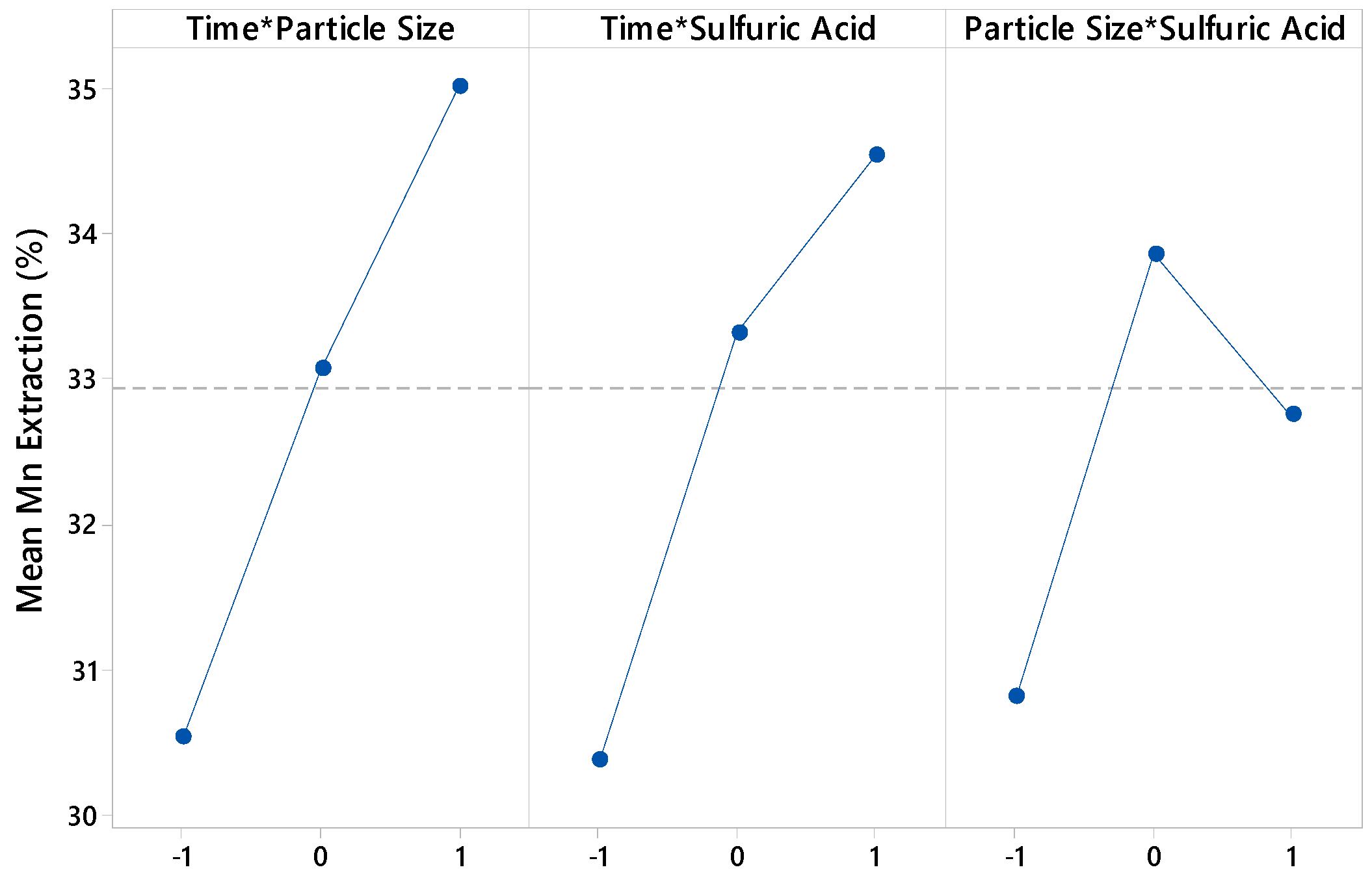
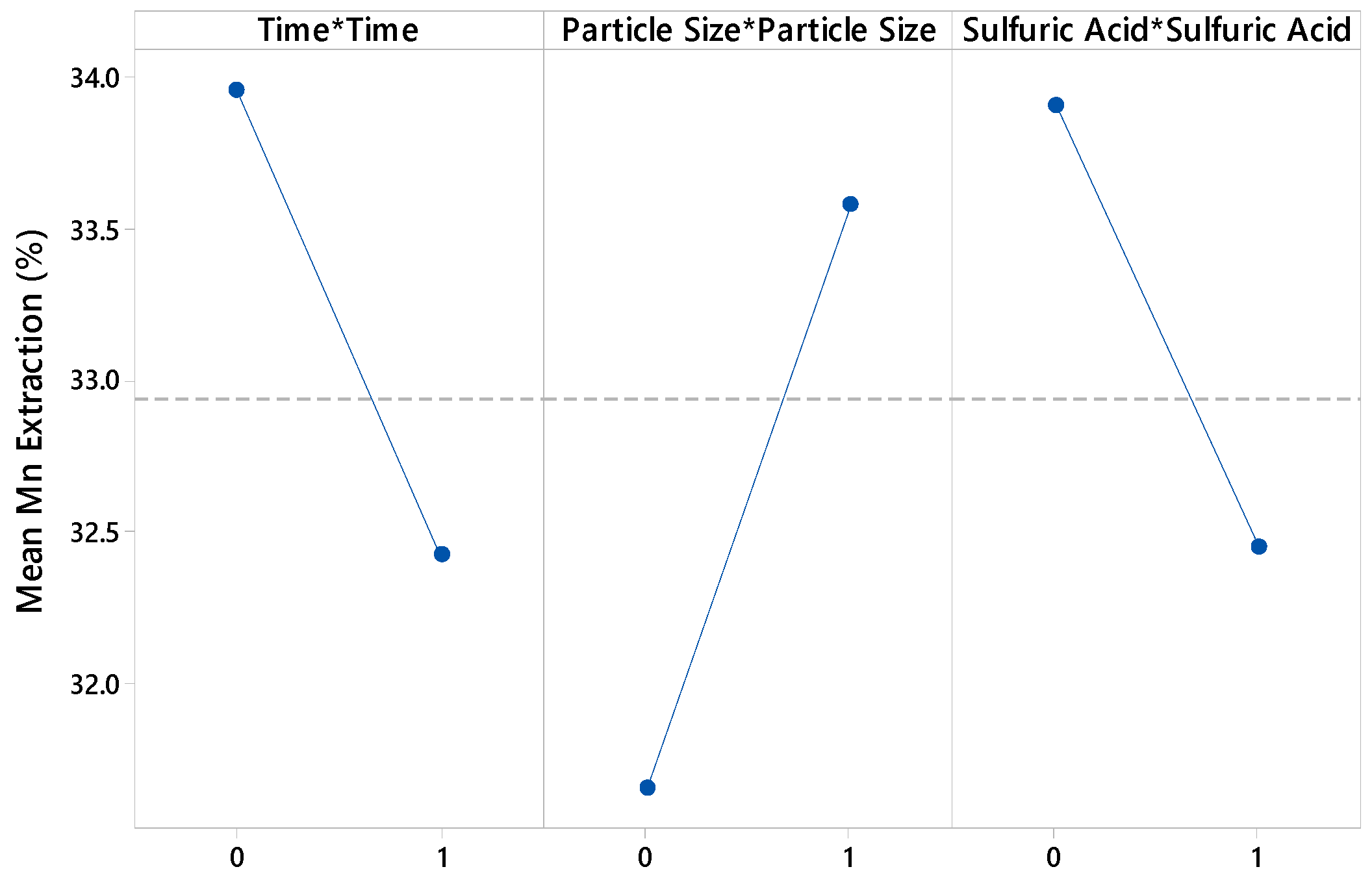
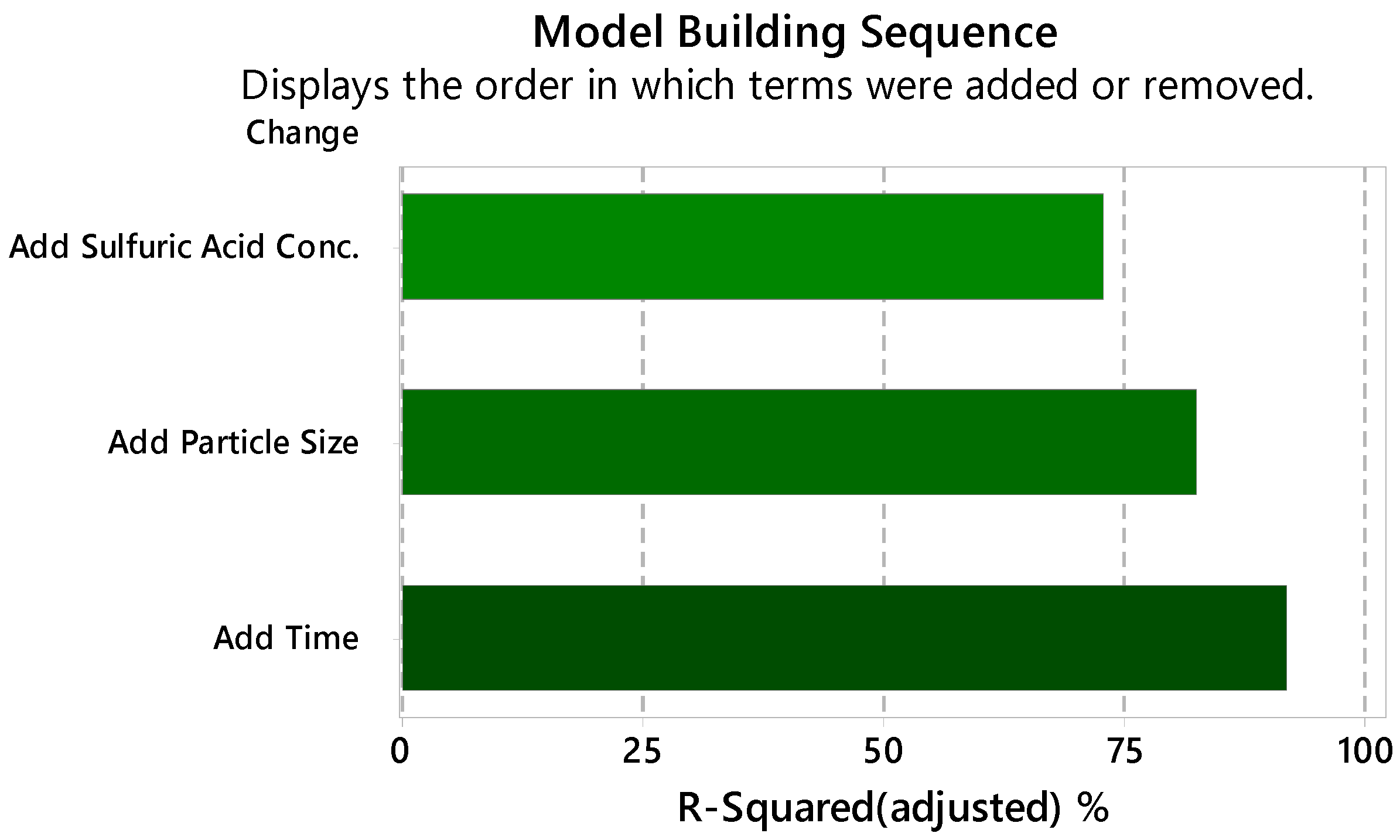
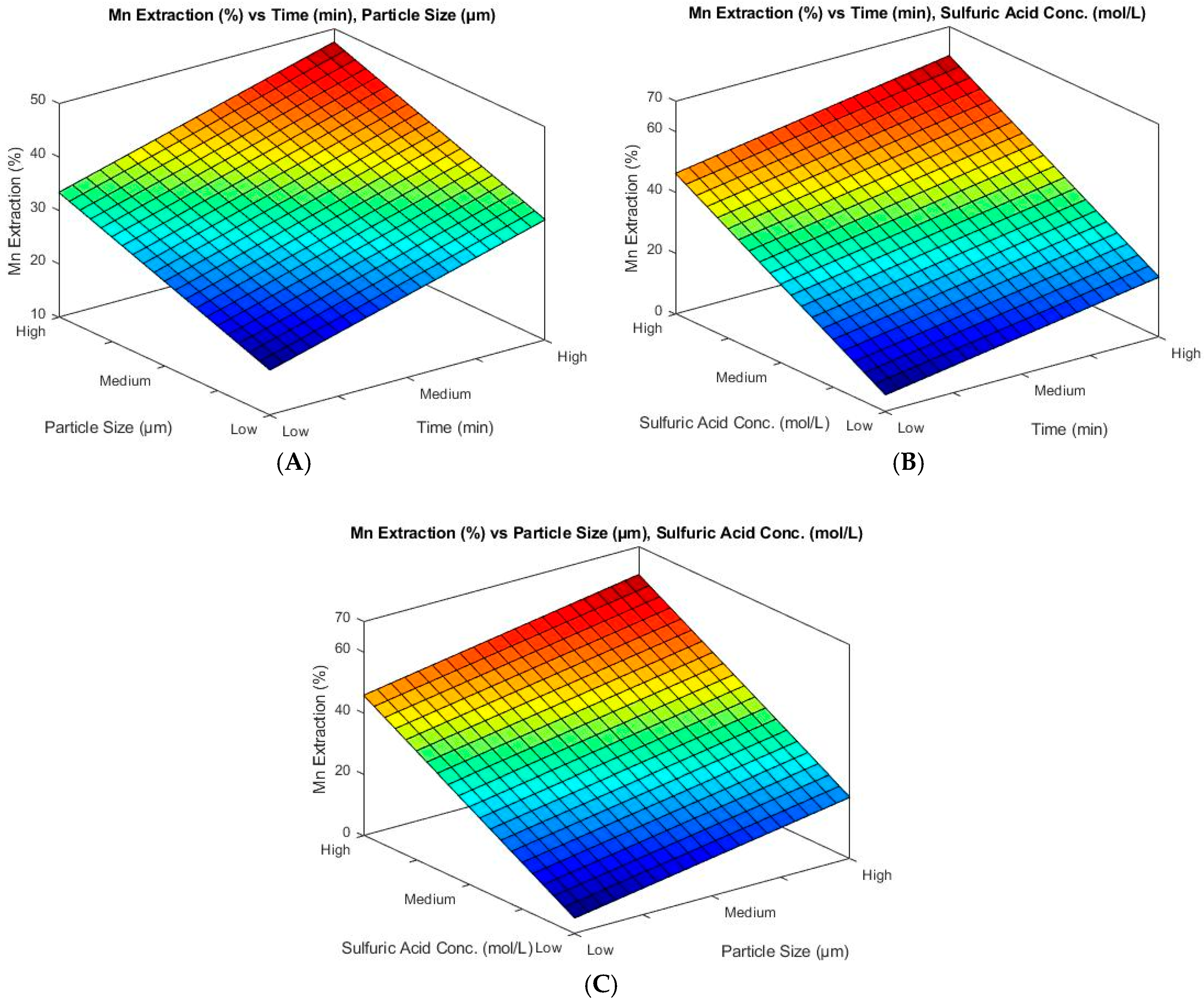
3.1. Effect of Variables
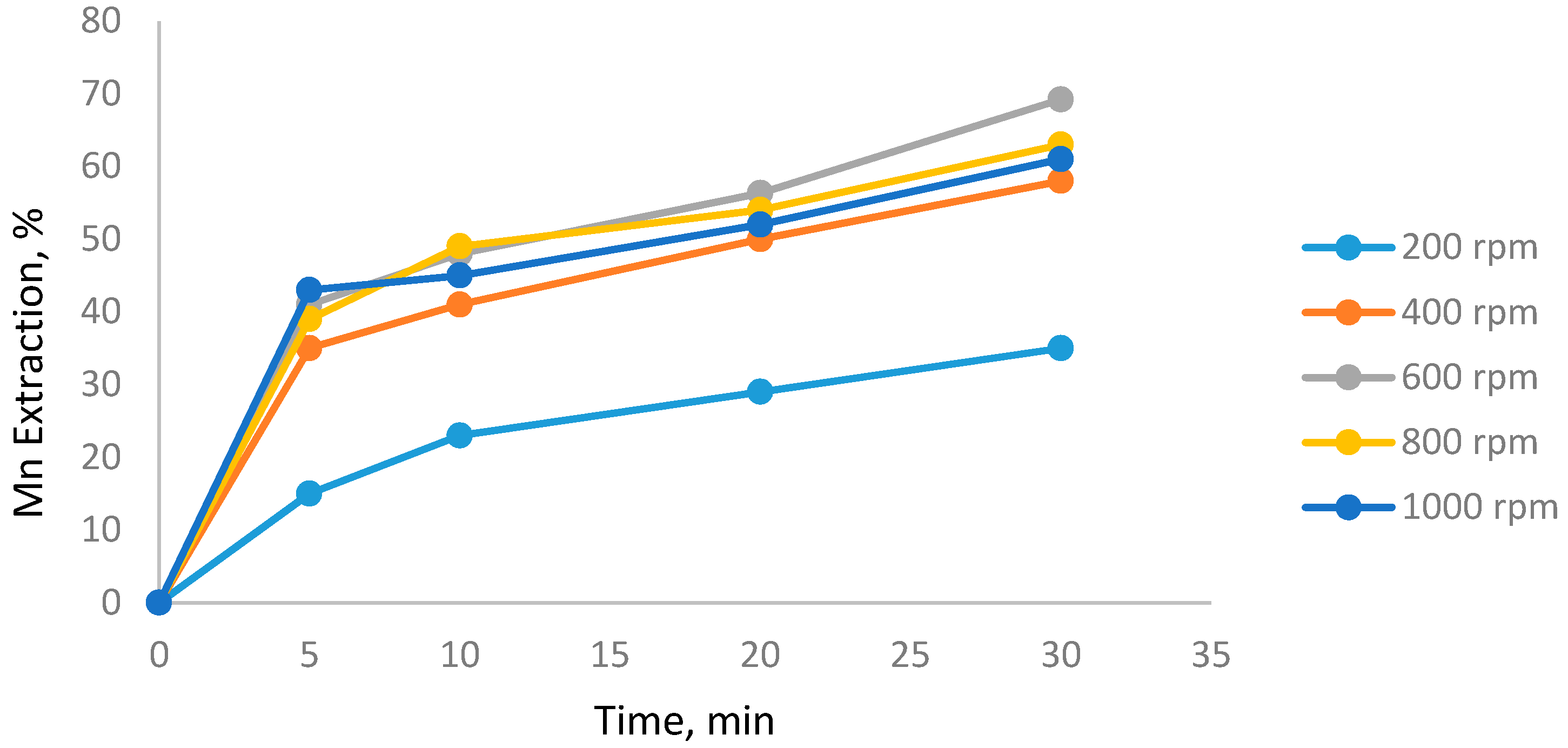
3.2. Effect of Agitation Speed
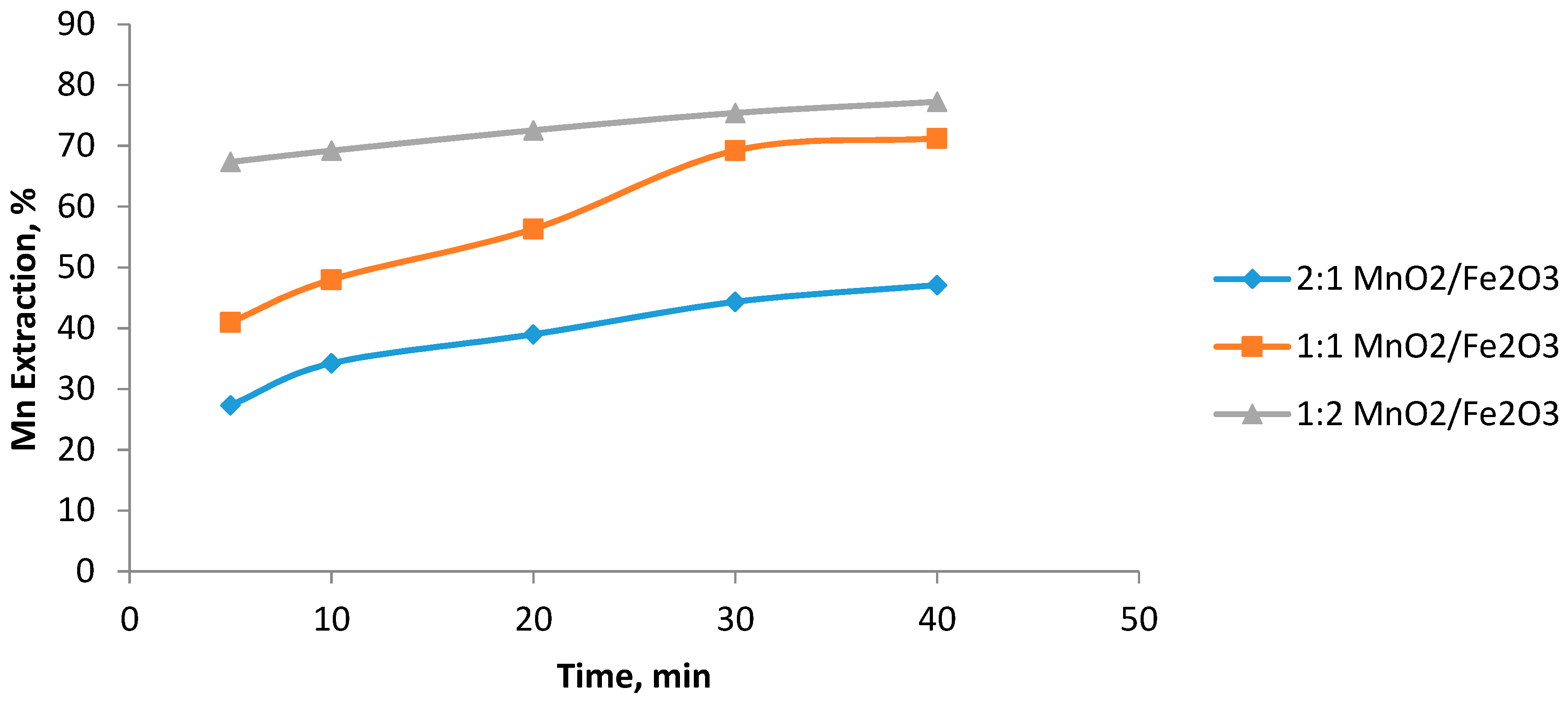
3.3. Effect of the MnO2/Fe2O3 Ratio
4. Conclusions
- (1)
- The ANOVA test indicates that sulfuric acid is the factor that has the greatest impact on manganese extraction under the studied conditions.
- (2)
- The manganese dissolution rate was generally higher when tailings were used instead of slag, possibly because tailings are more reactive to leaching.
- (3)
- Increase of the agitation speed did not significantly increase Mn extraction.
- (4)
- The highest Mn extraction rate of 77% was obtained at an MnO2/Fe2O3 ratio 0.5, 1 mol/L H2SO4, particle size of −47 + 38 μm, and leaching time of 40 min.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Marino, E.; González, F.J.; Somoza, L.; Lunar, R.; Ortega, L.; Vázquez, J.T.; Reyes, J.; Bellido, E. Strategic and rare elements in Cretaceous-Cenozoic cobalt-rich ferromanganese crusts from seamounts in the Canary Island Seamount Province ( northeastern tropical Atlantic ). Ore Geol. Rev. 2017, 87, 41–61. [Google Scholar] [CrossRef]
- Nishi, K.; Usui, A.; Nakasato, Y.; Yasuda, H. Formation age of the dual structure and environmental change recorded in hydrogenetic ferromanganese crusts from Northwest and Central Paci fi c seamounts. Ore Geol. Rev. 2017, 87, 62–70. [Google Scholar] [CrossRef]
- Konstantinova, N.; Cherkashov, G.; Hein, J.R.; Mirão, J.; Dias, L.; Madureira, P.; Kuznetsov, V.; Maksimov, F. Composition and characteristics of the ferromanganese crusts from the western Arctic Ocean. Ore Geol. Rev. 2017, 87, 88–99. [Google Scholar] [CrossRef]
- Usui, A.; Nishi, K.; Sato, H.; Nakasato, Y.; Thornton, B.; Kashiwabara, T. Continuous growth of hydrogenetic ferromanganese crusts since 17 Myr ago on Takuyo-Daigo Seamount, NW Pacific, at water depths of 800–5500 m. Ore Geol. Rev. 2017, 87, 71–87. [Google Scholar] [CrossRef]
- Josso, P.; Pelleter, E.; Pourret, O.; Fouquet, Y.; Etoubleau, J.; Cheron, S.; Bollinger, C. A new discrimination scheme for oceanic ferromanganese deposits using high fi eld strength and rare earth elements. Ore Geol. Rev. 2017, 87, 3–15. [Google Scholar] [CrossRef]
- Senanayake, G. Acid leaching of metals from deep-sea manganese nodules—A critical review of fundamentals and applications. Miner. Eng. 2011, 24, 1379–1396. [Google Scholar] [CrossRef]
- Randhawa, N.S.; Hait, J.; Jana, R.K. A brief overview on manganese nodules processing signifying the detail in the Indian context highlighting the international scenario. Hydrometallurgy 2016, 165, 166–181. [Google Scholar] [CrossRef]
- Kanungo, S.B. Rate process of the reduction leaching of manganese nodules in dilute HCl in presence of pyrite. Part I. Dissolution behaviour of iron and sulphur species during leaching. Hydrometallurgy 1999, 52, 313–330. [Google Scholar] [CrossRef]
- Kanungo, S.B. Rate process of the reduction leaching of manganese nodules in dilute HCl in presence of pyrite. Part II: leaching behavior of manganese. Hydrometallurgy 1999, 52, 331–347. [Google Scholar] [CrossRef]
- Zakeri, A.; Bafghi, M.S.; Shahriari, S.; Das, S.C.; Sahoo, P.K.; Rao, P.K. Dissolution kinetics of manganese dioxide ore in sulfuric acid in the presence of ferrous ion. Hydrometallurgy 2007, 8, 22–27. [Google Scholar]
- Toro, N.; Herrera, N.; Castillo, J.; Torres, C.; Sepúlveda, R. Initial Investigation into the Leaching of Manganese from Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Foundry Slag—Part I. Minerals 2018, 8, 565. [Google Scholar] [CrossRef]
- Bafghi, M.S.; Zakeri, A.; Ghasemi, Z.; Adeli, M. Reductive dissolution of manganese ore in sulfuric acid in the presence of iron metal. Hydrometallurgy 2008, 90, 207–212. [Google Scholar] [CrossRef]
- Jiang, T.; Yang, Y.; Huang, Z.; Zhang, B.; Qiu, G. Leaching kinetics of pyrolusite from manganese-silver ores in the presence of hydrogen peroxide. Hydrometallurgy 2004, 72, 129–138. [Google Scholar] [CrossRef]
- Su, H.; Liu, H.; Wang, F.; Lü, X.; Wen, Y. Kinetics of reductive leaching of low-grade pyrolusite with molasses alcohol wastewater in H2SO4. Chin. J. Chem. Eng. 2010, 18, 730–735. [Google Scholar] [CrossRef]
- Zhang, Y.; You, Z.; Li, G.; Jiang, T. Manganese extraction by sulfur-based reduction roasting-acid leaching from low-grade manganese oxide ores. Hydrometallurgy 2013, 133, 126–132. [Google Scholar] [CrossRef]
- Velásquez Yévenes, L.; Miki, H.; Nicol, M. The dissolution of chalcopyrite in chloride solutions: Part 2: Effect of various parameters on the rate. Hydrometallurgy 2010, 103, 80–85. [Google Scholar] [CrossRef]
- SERNAGEOMIN. Anuario de la mineria de Chile 2017; SERNAGEOMIN: Santiago, Chile, 2017. [Google Scholar]
- COCHILCO. Sulfuros primarios: desafíos y oportunidades I Comisión Chilena del Cobre; COCHILCO: Santiago, Chile, 2017. [Google Scholar]
- Oyarzun, R.; Oyarzún, J.; Lillo, J.; Maturana, H.; Higueras, P. Mineral deposits and Cu-Zn-As dispersion-contamination in stream sediments from the semiarid Coquimbo Region, Chile. Environ. Geol. 2007, 53, 283–294. [Google Scholar] [CrossRef]
- Baba, A.A.; Ayinla, K.I.; Adekola, F.A.; Ghosh, M.K.; Ayanda, O.S.; Bale, R.B.; Sheik, A.R.; Pradhan, S.R. A Review on Novel Techniques for Chalcopyrite Ore Processing. Int. J. Min. Eng. Miner. Process. 2012, 1, 1–16. [Google Scholar] [CrossRef]
- Centro de Estudios del Cobre y la Minería (CESCO). Available online: http://www.cesco.cl/en/home-en/ (accessed on 11 May 2019).
- Montgomery, D.C. Design and Analysis of Experiments; Wiley: Hoboken, NJ, USA, 2012. [Google Scholar]
- Bezerra, M.A.; Santelli, R.E.; Oliveira, E.P.; Villar, L.S.; Escaleira, L.A. Response surface methodology (RSM) as a tool for optimization in analytical chemistry. Talanta 2008, 76, 965–977. [Google Scholar] [CrossRef] [PubMed]
- Komnitsas, K.; Zaharaki, D.; Perdikatsis, V. Effect of synthesis parameters on the compressive strength of low-calcium ferronickel slag inorganic polymers. J. Hazard. Mater. 2009, 161, 760–768. [Google Scholar] [CrossRef] [PubMed]
- Komnitsas, K.; Manousaki, K.; Zaharaki, D. Assessment of reactivity of sulphidic tailings and river sludges. Geochemistry Explor. Environ. Anal. 2009, 9, 313–318. [Google Scholar] [CrossRef]
- Bartzas, G.; Komnitsas, K.; Paspaliaris, I. Laboratory evaluation of Fe0 barriers to treat acidic leachates. Miner. Eng. 2006, 19, 505–514. [Google Scholar] [CrossRef]




| Component | MgO | Al2O3 | SiO2 | P2O5 | SO3 | K2O | CaO | TiO2 | MnO2 | Fe2O3 |
|---|---|---|---|---|---|---|---|---|---|---|
| Weight (%) | 3.54 | 3.69 | 2.97 | 7.20 | 1.17 | 0.33 | 22.48 | 1.07 | 29.85 | 26.02 |
| Mineral | Amount % (w/w) |
|---|---|
| Chalcopyrite/Bornite | 0.47 |
| Tennantite/Tetrahedrite | 0.03 |
| Other Cu Minerals | 0.63 |
| Cu–Fe Hydroxides | 0.94 |
| Pyrite | 0.12 |
| Magnetite | 58.52 |
| Specular Hematite | 0.89 |
| Hematite | 4.47 |
| Ilmenite/Titanite/Rutile | 0.04 |
| Siderite | 0.22 |
| Chlorite/Biotite | 3.13 |
| Other Phyllosilicates | 11.61 |
| Fayalite | 4.59 |
| Dicalcium Silicate (Si) | 8.30 |
| Kirschsteinite (CaFeSi ) | 3.40 |
| Forsterita (Si) | 2.30 |
| Baritine (BaS) | 0.08 |
| Zinc Oxide (ZnO) | 0.02 |
| Lead Oxide (PbO) | 0.01 |
| Sulfate (S) | 0.20 |
| Others | 0.03 |
| Total | 100.00 |
| Exp. No. | Time (min) | Sieve Fraction (Tyler Mesh) | Particle Size (µm) | Sulfuric Acid Conc. (mol/L) | Mn Extraction (%) |
|---|---|---|---|---|---|
| 1 | 10 | −320 + 400 | −47 + 38 | 0.1 | 8.12 |
| 2 | 20 | −100 + 140 | −150 + 106 | 0.5 | 29.10 |
| 3 | 20 | −320 + 400 | −47 + 38 | 1 | 55.51 |
| 4 | 30 | −320 + 400 | −47 + 38 | 1 | 71.00 |
| 5 | 10 | −200 + 270 | −75 + 53 | 0.5 | 19.12 |
| 6 | 20 | −100 + 140 | −150 + 106 | 0.1 | 7.63 |
| 7 | 30 | −100 + 140 | −150 + 106 | 1 | 49.8 |
| 8 | 30 | −200 + 270 | −75 + 53 | 0.1 | 17.79 |
| 9 | 10 | −100 + 140 | −150 + 106 | 0.5 | 13.98 |
| 10 | 10 | −100 + 140 | −150 + 106 | 1 | 41.22 |
| 11 | 20 | −320 + 400 | −47 + 38 | 0.5 | 52.51 |
| 12 | 30 | −100 + 140 | −150 + 106 | 0.1 | 10.89 |
| 13 | 20 | −320 + 400 | −47 + 38 | 0.1 | 19.12 |
| 14 | 10 | −100 + 140 | −150 + 106 | 0.1 | 5.24 |
| 15 | 10 | −320 + 400 | −47 + 38 | 1 | 46.23 |
| 16 | 10 | −200 + 270 | −75 + 53 | 0.1 | 9.54 |
| 17 | 20 | −200 + 270 | −75 + 53 | 0.1 | 11.11 |
| 18 | 20 | −200 + 270 | −75 + 53 | 0.5 | 29.41 |
| 19 | 30 | −320 + 400 | −47 + 38 | 0.1 | 19.43 |
| 20 | 30 | −320 + 400 | −47 + 38 | 0.5 | 59.16 |
| 21 | 10 | −200 + 270 | −75 + 53 | 1 | 46.77 |
| 22 | 20 | −200 + 270 | −75 + 53 | 1 | 54.00 |
| 23 | 20 | −100 + 140 | −150 + 106 | 1 | 47.24 |
| 24 | 30 | −200 + 270 | −75 + 53 | 0.5 | 33.67 |
| 25 | 10 | −320 + 400 | −47 + 38 | 0.5 | 38.23 |
| 26 | 30 | −200 + 270 | −75 + 53 | 1 | 63.50 |
| 27 | 30 | −100 + 140 | −150 + 106 | 0.5 | 30.00 |
| Parameters/Values | Low | Medium | High |
|---|---|---|---|
| Sieve fraction (Tyler mesh) | −100 + 140 | −200 + 270 | −320 + 400 |
| Particle size (µm) | −150 + 106 | −75 + 53 | −47 + 38 |
| Time (in min) | 10 | 20 | 30 |
| H2SO4 (mol/L) | 0.1 | 0.5 | 1 |
| Source | F-Value | p-Value |
|---|---|---|
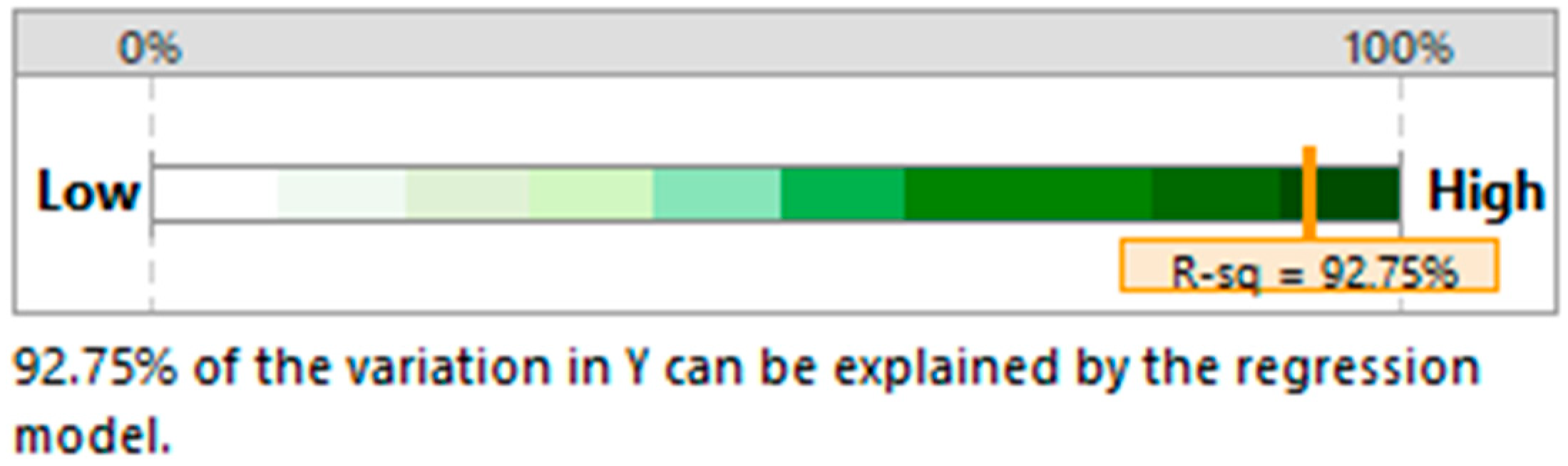
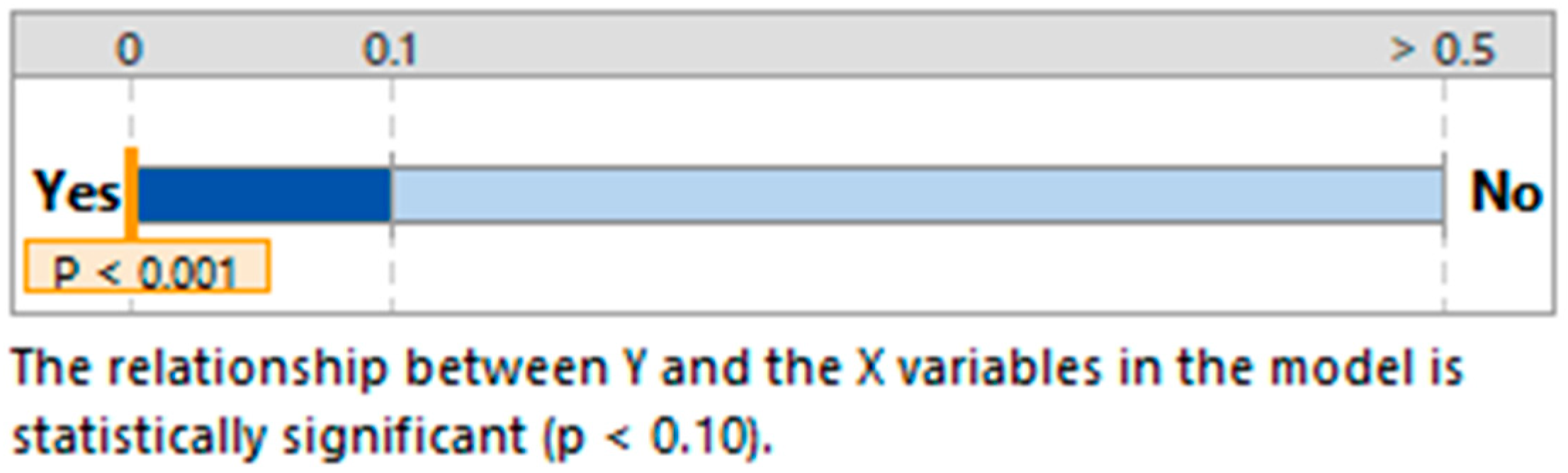
| Regression | 32.13 | 0.000 |
| Time | 27.12 | 0.000 |
| Particle size | 30.39 | 0.000 |
| Sulfuric acid | 226.50 | 0.000 |
| Time × Time | 0.43 | 0.522 |
| Particle size × Particle size | 0.67 | 0.423 |
| Sulfuric acid × Sulfuric acid | 0.39 | 0.542 |
| Time × Particle size | 1.81 | 0.196 |
| Time × Sulfuric acid | 1.57 | 0.228 |
| Particle size × Sulfuric acid | 0.34 | 0.568 |
| Experimental Conditions | Toro et al. [11] (2018) | Present Investigation |
|---|---|---|
| Temperature (°C) | 25 | 25 |
| Particle size of Mn nodules and slag/tailings (μm) | −75 + 53 | −75 + 53 |
| H2SO4 concentration (mol/L) | 1 | 1 |
| MnO2/Fe2O3 ratio | 1/2 | 1/2 |
| Mn dissolution rate at 5 min (%) | 68 | 67 |
| Mn dissolution rate at 40 min (%) | 70 | 77 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Toro, N.; Saldaña, M.; Castillo, J.; Higuera, F.; Acosta, R. Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings. Minerals 2019, 9, 289. https://doi.org/10.3390/min9050289
Toro N, Saldaña M, Castillo J, Higuera F, Acosta R. Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings. Minerals. 2019; 9(5):289. https://doi.org/10.3390/min9050289
Chicago/Turabian StyleToro, Norman, Manuel Saldaña, Jonathan Castillo, Freddy Higuera, and Roxana Acosta. 2019. "Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings" Minerals 9, no. 5: 289. https://doi.org/10.3390/min9050289
APA StyleToro, N., Saldaña, M., Castillo, J., Higuera, F., & Acosta, R. (2019). Leaching of Manganese from Marine Nodules at Room Temperature with the Use of Sulfuric Acid and the Addition of Tailings. Minerals, 9(5), 289. https://doi.org/10.3390/min9050289







