Abstract
Municipal solid waste incineration bottom ash (MSWI BA) is the main output of the municipal solid waste incineration process, both in mass and volume. It contains some heavy metals that possess market value, but may also limit the utilization of the material. This study illustrates a robust and simple heap leaching method for recovering zinc and copper from MSWI BA. Moreover, the effect of autotrophic and acidophilic bioleaching microorganisms in the system was studied. Leaching yields for zinc and copper varied between 18–53% and 6–44%, respectively. For intensified copper dissolution, aeration and possibly iron oxidizing bacteria caused clear benefits. The MSWI BA was challenging to treat. The main components, iron and aluminum, dissolved easily and unwantedly, decreasing the quality of pregnant leach solution. Moreover, the physical nature and the extreme heterogeneity of the material caused operative requirements for the heap leaching. Nevertheless, with optimized parameters, heap leaching may offer a proper solution for MSWI BA treatment.
1. Introduction
Incineration has become the typical method for treating municipal solid waste (MSW) in European Union, where 68 million tons of MSW was treated with incineration (MSWI) technology in 2016 [1]. During incineration, the organic content of the MSW is converted to thermal energy that can be utilized in generation of heat and power with simultaneous 90 wt % and 75 vol % reduction of initial waste [2]. The main residue of MSWI is bottom ash (BA), representing approximately 80% of solid incineration rejects [3]. The scrap iron and some other metals of MSWI BA can be separated and utilized by the metal refining industry, followed either by reuse of remaining MSWI BA (e.g., as construction materials) or disposal, if the quality does not allow reuse [2,4,5]. Residual heavy/toxic metals can cause challenges in both reuse and disposal of MSWI BA, but also serve as a secondary source for valuable elements [5,6,7].
MSWI BA is an extremely heterogeneous residue stream including glass, synthetic ceramics, natural minerals, unburned organic matter and a variety of metals in different forms [3,8,9,10]. The particle size is heterogeneous varying from a few µm particles to the chunks of several centimeters, effecting also significantly to the elemental composition; in particular, magnetic metals (Fe, contaminated with e.g., Zn and Cu) seem to accumulate to fine fractions, while diamagnetic metals (Al and Cu) are found in all size fractions [3,9]. Moreover, different elements tend to accumulate on different mineralogical fractions. Heavy metals have been reported to concentrate on glass matrix; Cr, Zn and Mn have been incorporated into spinels, while Cu and Pb associated to Fe, Sn and Zn metallic inclusions [9,10]. Despite the incineration process, some metals may be partly in the metallic phase instead of their oxidized form (e.g., copper wires) [3,10]. Also, Cu sulfides may be formed in the process, even though CuO is the dominant form [8].
The above-mentioned characteristics of MSWI BA make the treatment for metal removal/recovery demanding. One proposed solution for the challenge has been hydrometallurgical treatment, however this particular technology has been studied more with other MSWI reject classes (e.g., fly ashes [11,12,13]), despite the fact that a vast majority of Cu and Zn end up in bottom ash [6]. A great majority of current bottom ash research concentrates mainly on more mild leaching conditions (or water leaching/washing), and is linked often more to the metal release during the aging or after landfilling or utilization as material [14,15,16,17]. With MSWI BA, it has been observed that Zn and Cu dissolution start at pH 4–5 and pH 3–4, respectively [14,17,18]. In harsh acidic conditions (acid: 3 M H2SO4, temperature: 80 °C, leaching duration: 2 h, solid/liquid ratio: 20%) and applying reactor apparatus, Mo and V recoveries were high (>80%), but for Ni and Cu low (<40%) [7]. Similar leaching yields for Ni and Cu was observed with HNO3; moreover, Zn leaching yield was found low (<40%) [19]. In addition to low-to-moderate yields of these key metals from MSWI BA, challenges may also be foreseen in high concentrations of iron in the final leachate (treatment costs due to iron removal), as well as in solid-liquid separation due to gel-like formations (filtration costs) [7,19]. Organic acids have also been studied for metals removal from MSWI BA. By using 1 M citric acid and 1% solid/liquid ratio, Cu and Zn leaching yields of >90% have been reached; however, when increasing the solid/liquid ratio to 5%, Cu and Zn leaching yields decreased to <65% [20]. Bioleaching of MSWI BA with iron and sulfur oxidizing microorganisms has been reported to reach high leaching yields for Cu (100%) and Zn (80%) when the system is supplemented with bioleaching microorganisms, elemental sulfur, ferrous iron and 10% (v/v) of MSWI BA [21]. The drawback, compared to chemical leaching tests, was the long duration of leaching.
It has been suggested that acid leaching of MSWI BA may not be reasonable, due to requirement of low pH and low solid/liquid-ratio [5]. In this paper, the approach of acid heap leaching process instead of stirred tank reactor leaching is introduced for treating MSWI BA for improving these factors: in acid heap leaching higher solid/liquid ratio can be applied and residual acid circulated back to the heap decreasing the acid consumption. Additional benefits include the lack of expensive reactor vessels, agitation instruments and motors, as well as tolerance for larger particles, removing the need of comminution. The expected negative impacts of acid heap leaching application were expected to be slow leaching kinetics (i.e., long leaching duration) and blockage/channeling of heap due to release of fine particles. Both these may result in poor leaching yields of target elements. It is noteworthy, that temperature increase in MSWI BA leaching has not dramatically increased leaching yields [7], and therefore, restriction of heap leaching to stay on ambient temperature is expected not to jeopardize the process.
For the leaching medium, H2SO4 was selected in this work. This enabled two different leaching strategies: the addition of commercial H2SO4 to the heap or the production of H2SO4 by sulfur oxidizing bacteria from an elemental sulfur source (Equation (1)). The autotrophic sulfur oxidizers, such as Acidithiobacillus thiooxidans, are acidophilic microorganisms, which utilize reduced or elemental sulfur as their energy source by using oxygen as an electron acceptor, and fix carbon from the atmosphere as CO2 (for a review, see [22]).
S0 + 3O2 + 2H2O → 2SO42− + 4H+
Despite the origin of the acid, it will be consumed to overcome the high buffer capacity of MSWI BA by dissolving carbonates, hydroxides, silicates and oxides, but also to dissolve target metals of Zn and Cu. MSWI BA may also contain plenty of metals that have not oxidized in the incineration process, but remain in the metallic form [7]. Especially metallic Cu is resistant to acid attack, but can be efficiently leached with Fe3+ oxidant (Equation (2)), which can be regenerated rapidly by iron oxidizing bacteria (Equation (3)) [23]. Another encountered form of Cu in MWSI BA is Cu2S [8], which can be dissolved by Fe3+ oxidant (Equation (4)), followed by biogenic iron regeneration (Equation (3)) and oxidation of elemental sulfur to sulfuric acid by sulfur oxidizing bacteria (Equation (1)).
Cu + 2Fe3+ → Cu2+ + 2Fe2+
2Fe2+ + 0.5O2 + 2H+ → 2Fe3+ + H2O
Cu2S + 4Fe3+ → 2Cu2+ + 4Fe2+ + S
Typical examples of iron oxidizing bacteria are, e.g., At. ferrooxidans and Leptospirillum ferrooxidans, which are acidophiles and obtain their oxygen and carbon from the atmosphere (for a review, see [22]). The biological oxidation of Fe2+ to Fe3+ can proceed more than million times faster than the abiotic oxidation by oxygen [24]. Therefore, the biogenic iron regeneration process can be an economically attractive method for obtaining the oxidant in certain cases [25].
The objective of this study was to understand how MSWI BA behaves in acid heap leaching process, with the target of dissolving valuable elements of Zn and Cu. In addition to these elements, Fe and Al dissolution was monitored as their dissolution in the process would have negative impacts for the complete process (higher acid consumption, more complex pregnant leach solution). The behavior and effects of bioleaching microorganisms were studied in heap leaching systems to reveal whether they offer advantages for leaching, and preliminary investigate how they could be applied in the heap leaching process.
2. Materials and Methods
2.1. MSWI BA Sample Material
Fortum Environmental Construction Oy provided a 300 kg MSWI BA sample, which was classified to a particle size of 3–40 mm by Geological Survey of Finland (GTK), and then transported to VTT Technical Research Centre of Finland for leaching studies. The selection of wide particle size range was justified by occurrence of target metals (Zn, Cu) in many fractions, and due to their possible adherence to larger particles [3]. Moreover, objective was to study a robust process that does not require pre-treatment, but allows a simple process for the majority of generated MSWI BA. After the transport, the sample was homogenized by mixing and dividing it to four equal shares on a tarpaulin. The material was very brittle, generating small amount of <1 mm powder already in homogenization. The generated small amount of this finer fraction was not removed, but left mixed with the original material for experiments. This decision was justified by maintaining the representativeness of the sample (sieving increased the generation of fine fractions), and possible loss of Cu and Zn. It is noteworthy that fine material in heap leaching may cause disruption in permeability and solution/gas flows. MSWI BA sample was also extremely heterogeneous, as the ash agglomerates occurred in different sizes and shapes, visibly consisting of a variety of materials in different shares. In addition, metallic pieces (e.g., short copper wires, pieces of stainless-steel cutlery) were observed. The heterogeneity caused difficulties in the determination of chemical composition. As reported, the average chemical composition of MSWI BA differs very slightly [3] and therefore, analysis provided by the material owner was used as an average composition (Table 1).

Table 1.
Average composition of municipal solid waste incineration bottom ash (MSWI BA) sample material.
2.2. Adaptation of Microorganisms to the Material
A mixed culture of iron and sulfur oxidizing microorganisms, enriched from acidic mine waters, was used. The mixed culture contained Marinobacter sp., Acidithiobacillus (such as At. ferrooxidans, At. thiooxidans, At. albertensis, At. ferrivorans), Leptospirillum (L. ferrooxidans), Cuniculiplasma, Nitrosotenius and Ferroplasma. The culture was adapted to washed MSWI BA in shaken flasks in incubator-shaker (30 °C and 150 rpm) by gradually increasing the solid/liquid ratio up to 10% (w/v) in 0 K media (composition presented in the Table 2). Solution pH was regularly adjusted to near 2.0 with 95% H2SO4. A separate adaptation with an addition of elemental sulfur was also created (1 g S0/100 mL); here, the bacteria could produce H2SO4 through their own sulfur oxidizing reactions (Equation (1)) and the amount of added H2SO4 was reduced. With elemental sulfur addition, the pH remained lower and the RedOx potential higher than in the adaptations without elemental sulfur, resulting in increased heavy metal accumulation in this adaptation.

Table 2.
The used 0 K medium.
2.3. Preliminary Tests
Prior to heap leaching experiments, acid consumption and leaching behavior were studied. First, the sample material was washed with 5-fold weight of distilled water compared to the sample to remove the majority of chloride. The washed sample (20 g) was mixed with water (200 g) in 500 mL Erlenmeyer flask. The flasks were placed to the orbital shaker (150 rpm, 30 ˚C; Stuart SI-500), and pH was maintained at 1.0, 1.5, 2.0, 2.5 or 3.0 for 8 h with 95% H2SO4 using manual titration. In addition to the pH static tests, two experiments with Fe3+ were conducted to clarify the effect of oxidant for leaching. In these experiments, the bioleaching culture was used to oxidize iron by adding inoculum (25 mL), 0K medium (225 mL) and solid FeSO4·7H2O (to reach Fe2+ concentration of 4.5 or 9.0 g/L). The solution was incubated (150 rpm, 30 °C) until the RedOx potential had risen to +650 mV, illustrating biological oxidation of Fe2+ to Fe3+ (Equation (3)). The biologically produced ferric solutions (200 mL, with measured Fe3+ concentrations of 5.3 and 8.4 g/L), were mixed with the sample material (20 g). The working protocol was the same as with the pH-static tests (150 rpm, 30 °C, pH 2.0, 8 h). After the 8-hour leaching tests, all leachates were filtrated (0.45 µm) and analyzed for dissolved metals with ICP-OES (by external accredited laboratory Metropolilab Oy, Helsinki, Finland). The pH and RedOx were measured with a Consort multi-parameter analyzer C3040, with Van London-pHoenix Co. electrodes (Ag/AgCl in 3 M KCl).
2.4. Column Experiments
The experiments were conducted in four columns (height 31 cm, diameter 10 cm) with approximately 2000 grams of MSWI BA sample material. The material was first washed by filling the column containing the material with distilled water for 15 min, then draining the column, and repeating this process for a total of five times. Four different configurations were chosen for the column tests (see Table 3).

Table 3.
Column configurations used in the experiment.
Three columns were operated as bioleaching heaps (COL II, COL III, COL IV) with an addition of 400 mL of adapted inoculum (COL II and COL III adaptation without elemental sulfur, COL IV adaptation with elemental sulfur), while one column (COL I) was kept as a chemical control. In COL IV, 104 grams of elemental sulfur (5% of column material) was agglomerated to the sample material with an addition of 18.73 g of 0.1 M H2SO4 (the same amount of acid was added to the other columns as well). The bottom and the top of the columns contained glass beads (diameter 6 mm) and perforated plates to prevent the washout of the material, as well as to help to distribute the influent evenly to the column. In addition, glass wool was used in the bottom to keep the finer fractions inside the column. The column solutions (0 K media) were circulated through circulation tanks (plastic containers). The solution was not removed from the system, but it was collected and fixed for the pH in the circulation tank, and pumped back to the respective column. Evaporation was compensated by adding distilled water to the circulation tank. The flow rate of solutions was adjusted with peristaltic pumps (Watson Marlow 205S). The pH and RedOx were measured with a Consort multi-parameter analyzer C3040, with Van London-pHoenix Co. electrodes (Ag/AgCl in 3 M KCl) from both column effluents and circulation tanks. Solution samples were taken from circulation tanks, followed by filtration (0.45 µm), and analysis for dissolved metals with ICP-OES (by external accredited laboratories Metropolilab Oy, Helsinki, Finland, and Labtium Oy, Espoo, Finland).
Heap experiments lasted for a total of 139 days with different operational phases. First, the columns were operated as standard irrigation heaps by pumping the solution to the top of the heap (5 L/m2·h, days 0–54). This operation manner and volume flow is typical for acid heap leaching [26]. The pH of the circulation tanks was adjusted daily to approximately 2.0 with 95% H2SO4. After the irrigation trial, the flow direction was reversed on day 54 and the influent was pumped upwards from the bottom, consequently flooding the column (this is a typical operating manner in CEN/TS column tests, often used for MSWI BA research [15]). The solution flow was maintained at 5 L/m2·h. From day 82 onwards, more intense acid addition was deemed necessary to overcome the acid neutralization potential of the material. The circulation tank pH was adjusted daily to 1.3 and the solution flow was increased to 10 L/m2·h. From day 119 onwards, the circulation tanks were aerated by pumping air (1.5 L/min adjusted by a rotameter, Kytola Instruments Oy, Muurame, Finland) through a tube and a plastic nozzle to study the effect of iron oxidizing bacteria. Moreover, the solution flow rate was decreased back to 5 L/m2·h and COL III supplemented with 400 mL of re-inoculation, to secure the presence of active iron oxidizing bacteria.
3. Results
3.1. Preliminary Experiment
Acid consumption varied from 27 to 556 kg 95% H2SO4 per ton of sample material, depending on the selected pH (Figure 1). Acid consumption increased linearly with the decrease of pH from 3.0 to 1.5, but then raised dramatically at pH 1.0. Copper and zinc dissolution increased rather linearly with the pH decrease, while iron dissolution was closer to an exponential rise (Figure 1).
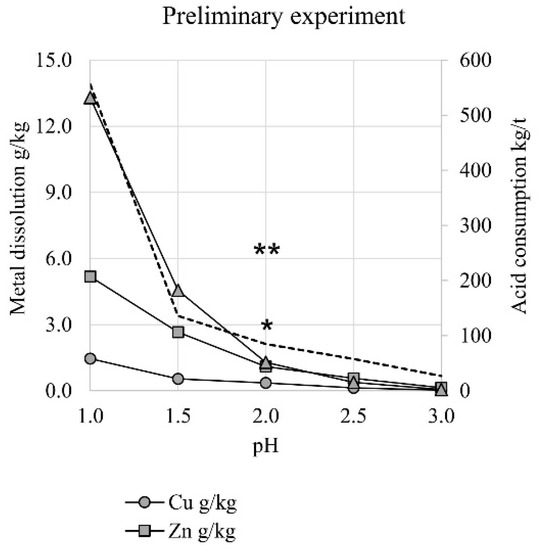
Figure 1.
Leaching of the MSWI BA sample material in different pH values in shake flasks (pH 1.0–pH 3.0; symbols “*” and “**” illustrate copper dissolution when Fe3+ concentration was 5.3 and 8.4 g/L, respectively). Acid consumption expressed as kg 95% H2SO4 per ton of sample material.
Iron dissolution is an unwanted phenomenon in leaching processes, as it consumes extra acid and decreases the quality of produced leachate. The leaching process was estimated to be the most beneficial at pH 2.0, as it resulted in similar zinc and iron concentrations in the leachate and consumed only 85 kg 95% H2SO4/t of sample material. The introduction of an oxidizing agent (Fe3+) into the leaching media had drastic effects: in the static pH 2.0, the copper concentrations in the solutions rose from 35 mg/L (no added Fe3+) to 300 and 642 mg/L, when the leaching solutions contained 5.3 and 8.4 g/L of Fe3+, respectively (Figure 1).
3.2. Column Experiments
During the material washing, chloride concentration of the washing solution decreased from 680 to 52 mg/L, but only negligible amounts of metals (Cu, Zn, Fe, Al) were detected. After washing, the heap leaching experiments were started by irrigation tests with the target of pH 2.0, according to the preliminary experiments. Despite the identical pH and flow rate of the column influents, differences in effluents were observed. With all inoculated bioleaching columns effluents decreased to the pH 2–4 level. However, this took much longer in the absence of elemental sulfur (COL II) (Figure 2: days 0–54). The pH in the abiotic column (COL I) remained rather stable at pH 7.5–8.5. The acid consumption was clearly lower in the column with supplemented elemental sulfur (COL IV) compared to the other columns (Figure 2: days 0–54); with all columns, acid consumption was remarkably lower than with preliminary experiments. The RedOx-potential rose rapidly to +450–550 mV level in the inoculated columns; some slower increase was also observed in the abiotic column reaching only +300–400 mV level (Figure 3: days 0–54). Despite the rather low pH of the inoculated columns, iron, zinc and copper dissolutions were negligible during the irrigation trial (Figure 3 and Figure 4: days 0–54). It is noteworthy that the increase in copper dissolution during the early days was caused by the inoculum that contained a remarkable copper impurity that was accumulating to the inoculums during the adaptations. The adaptation culture supplemented with elemental sulfur had the highest copper content, which is clearly seen in COL IV (Figure 4).
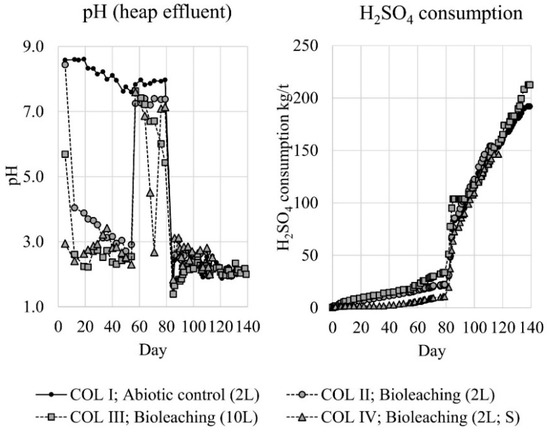
Figure 2.
The pH of the column effluents, and cumulative H2SO4 consumption (kg pure acid per ton of sample material) during the experiments (H2SO4 used to maintain pH of the circulation tank at desired value). Circulation tank volume presented in parentheses; symbol “S” illustrates the column supplemented with elemental sulfur.
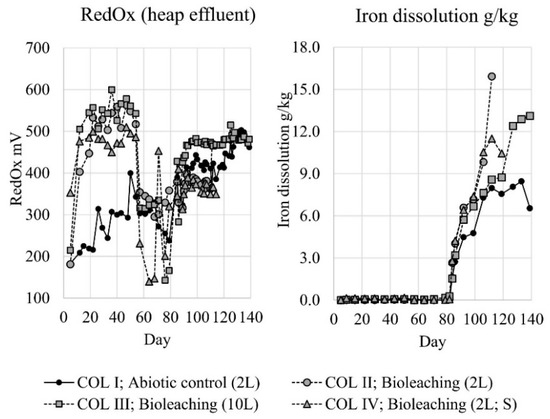
Figure 3.
RedOx-potential in column effluents, and iron dissolution from sample material (grams of dissolved iron per kg of sample material) in column experiments. Circulation tank volume presented in parentheses; symbol “S” illustrates the column supplemented with elemental sulfur.
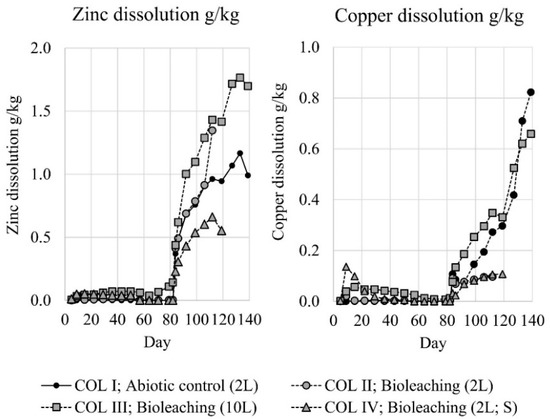
Figure 4.
Zinc and copper dissolution from sample material (grams of dissolved iron per kg of sample material) in column experiments. Circulation tank volume presented in parentheses; symbol “S” illustrates the column supplemented with elemental sulfur.
The low metals dissolution was most likely caused by uneven distribution and channeling of leaching solution inside the columns. Therefore, the test protocol was changed on day 54 to flooding in order to maximize the contact between solution and sample material. In flooding, the pH rose rapidly in all columns to pH >7 (Figure 2: days 54–79); as the influent parameters were maintained at pH 2.0 and 5 L/m2·h, all acid was consumed completely inside the column. The RedOx-potential decreased in all columns to +150–350 mV (Figure 3: days 54–79), referring possibly to acid and/or oxygen depletion in columns. Virtually no metals (Cu, Zn, Fe, Al) were leached during this flooding period (Figure 3 and Figure 4: days 54–79). However, an anomaly was observed in pH and RedOx-potential between days 68–71 in the column supplemented with elemental sulfur (COL IV). The pH decreased to pH <3, and the RedOx-potential peaked to +450 mV. The reason for the anomaly was not found, but an explanation could be an oxygen leak in the junction of the column top and effluent tubing. This would cause the oxidation of elemental (or intermediate) sulfur found from the tubings.
With the original circulation tank pH and flow rate back to the columns there was not enough acid to perform the targeted leaching of metals. Therefore, the flooding trial protocol was changed on day 79, by decreasing the pH in circulation tanks to pH 1.3 and increasing the influent flow rate to 10 L/m2·h. This caused a drastic drop in the pH with all columns to the level of 1–3 and later stabilizing to pH 2.0–2.5 (Figure 2: days 79–119) with subsequent increase in the acid consumption. The rise of the RedOx-potential was clearly lower with inoculated COL II and COL IV. The highest RedOx-potential was reached with the inoculated column with 10L circulation solution volume (COL III), followed by the abiotic column (COL I) (Figure 3: days 79–119). Iron and zinc dissolution started rapidly when introducing more acid to the columns (Figure 3 and Figure 4: days 79–119). The highest iron dissolution seemed to occur in the inoculated columns COL II and COL IV. However, the iron dissolution in the column supplemented with elemental sulfur (COL IV) collapsed later on day 112, but even intensified without elemental sulfur (COL II). For zinc dissolution, the inoculation of microorganisms had a positive effect, but supplemented elemental sulfur decreased the dissolution. For copper, the dissolution was generally slower and tended to level, especially with the columns that had a lower RedOx-potential (COL II and COL IV). The dissolution of copper increased linearly only with the COL III. Unfortunately, two columns were lost during the intensified flooding trial: the COL IV on day 114 and the COL II on day 119. Column tubings broke during the night, and all column and circulation tank solutions were lost.
After the loss of two inoculated columns, a final flooding test was done for the remaining columns (COL I and COL III) by adding aeration to the circulation tanks on day 119. Simultaneously, the influent flow rate was decreased back to 5 L/m2·h, and the inoculated column COL III was re-inoculated to secure the presence of active iron oxidizing bacteria. These changes did not have remarkable effects on the pH or acid consumption (Figure 2: days 119–139). The RedOx-potential remained stable with the inoculated column COL III, but increased with the abiotic column COL I, so that both operative columns reached +500 mV level (Figure 3: days 119–139). During the aeration, iron and zinc dissolution tended to level off. However, copper dissolution intensified clearly in both columns (Figure 3 and Figure 4: days 119–139).
In this study, aluminum dissolution was not monitored as often as iron. Nevertheless, with several measurements it was seen that aluminum required stronger acid addition (that occurred from day 79 onwards) for rapid leaching, reaching the final 6–12 g/kg leaching yield level. Therefore, aluminum and iron behaved similarly in terms of leaching. The final leaching yields for zinc, copper, iron and aluminum are presented in Table 4.

Table 4.
The final leaching yields in column experiments.
4. Discussion
When approaching the challenge of removing heavy metals from MSWI BA, several factors must be stressed. Iron and aluminum were the main metallic components of the sample material, while targeted zinc and copper occurred in lower concentrations. Therefore, certain leaching selectivity must be obtained to avoid extra use of leaching chemicals, but also to produce a solution that can be economically purified to metal products and inert residues. It was seen that all heavy metals required rather strong acid addition and leaching was not possible before the effluent reached pH < 3. This was slightly controversial to some earlier leaching studies [14,17,18]. However, it is noteworthy that refereed studies were done in agitated bottom ash slurry, where leaching may be different, as compared to heap leaching; moreover, these experiments were done in batch-wise while this study examined continuous heap leaching. However, the difference does not possibly link only to the actual dissolution reaction inside the column, but to later hydroxide precipitation of heavy metals when the pH rises, if strongly neutralizing sectors still occur in the heap. For example, the precipitation of Fe(OH)3, Al(OH)3, Cu(OH)2, Zn(OH)2 and Fe(OH)2 occur at pH > 3.5, pH > 4.0, pH > 6.5, pH > 8.0 and pH > 8.5, respectively (the exact pH depends greatly of residual concentration of a studied metal) [27,28]. Therefore, the whole heap must reach and maintain acidic conditions to enable the liberation of heavy metals. In this study, the actual leaching was shown to require a much lower pH than required by the formation of hydroxides (preliminary experiment; Figure 1), indicating that not much metal hydroxides are present in MSWI BA, which has also been shown earlier [8,9,10]. However, the preliminary experiment also showed that too high an acid addition caused drastic liberation of iron, and that the heap should not be operated below pH 2.0. Later in the column experiments, it was seen that when pH 2.0 was reached in the column effluents, iron and aluminum dissolutions were already tenfold, as compared to zinc and copper. On day 112 (the last recorded acid consumption before two columns were lost), acid consumption in the columns had reached 142–154 kg 95% H2SO4/t, compared to 85 kg 95% H2SO4/t of preliminary experiment at pH 2.0. Therefore, it can be estimated that the acid addition was too strong and too rapid in columns. When the first parts of the columns reached pH < 2 and started to liberate iron and aluminum, the final parts of columns still possessed some acid neutralization potential and the effluent remained at pH >2. This led to prolonged acid addition and uncontrolled dissolution of unwanted elements. However, we assume that this can be neglected by increasing the influent flow rate but maintaining the influent pH at 2.0–3.0. This secures that enough acid is supplemented to the heap, but the pH gradient in the heap is not too severe. It was also observed that MSWI BA may require a more rapid influent flow rate than typical applications with ores, due to the material characteristics and the risk of channeling effect.
For copper, it was shown in the preliminary and column experiments that dissolution was intensified rapidly by raising RedOx-potential, by aeration and/or iron oxidizing bacteria. This is most likely linked to the presence of metallic copper in the MSWI BA, which requires oxidant, e.g., Fe3+, to dissolve. The role of iron oxidizing bacteria would be intensified Fe2+ to Fe3+ regeneration for improved copper leaching (the iron regeneration loop presented in Equations (2) and (3)). In addition, iron oxidation may benefit heap leaching by precipitating excess iron as jarosite, schwertmannite or goethite in sulfate rich solutions [29]; these reactions also generate acid that could be reused in heap leaching. For the above-mentioned reasons, it may be justified to add aeration to the heap leaching process of MSWI BA. However, the role of iron oxidizing bacteria is more complex. In the column experiments presented here, no clear benefit was seen between the abiotic aerated (COL I) and the inoculated aerated column (COL III) in copper leaching; both reached high RedOx-potential. However, it is very possible that the abiotic column and/or circulation tank was taken over by microorganisms during the aerated period, as the system was not completely sealed, and the material was not sterilized. Despite the fact that no microorganisms were found in a microscopic examination of the circulation tank solution, the presence of bacteria could not be ruled out. Nevertheless, an industrial heap leaching process cannot be operated in a sterile mode, and most likely, the circulation pond would offer suitable conditions for iron oxidizing bacteria. Another studied biochemical possibility, the oxidation of supplemented elemental sulfur and biogenic sulfuric acid production by sulfur oxidizing bacteria, was also not completely understood in this experiment. During the early days of operation (days 0–54), the inoculated column with supplemented elemental sulfur (COL IV) consumed only 25–35% acid compared to the other columns, which proved the original theory of using elemental sulfur in biological treatment of MSWI BA to decrease the acid consumption. However, as the irrigation approach resulted in slow leaching, the operative mode was changed to flooding to speed up the leaching (day 54 onwards), which caused the dramatic pH increase to the unsuitable levels for, e.g., At. thiooxidans [22]. Therefore, it is justified to assume that the sulfur oxidizing bacteria perished, or at least were strongly inhibited by the high pH. Later, the acid consumption of the column with supplemented elemental sulfur was similar to the other columns, showing that the bacteria were no longer active enough, and the true biological acid generation potential remained unclear. However, it was seen that elemental sulfur disrupted at least zinc and copper dissolution, perhaps due to the decreased RedOx-potential.
A simple examination of chemical costs (i.e., H2SO4) versus the market value of liberated metals (Cu and Zn) was done (Table 5) to check if the process had any economic viability. The data of COL I was used for these studies (end of experiment on day 139) as it seemed most successful in terms of economic aspects (high Cu and Zn dissolution, lowest Fe dissolution). With the sulfuric acid price of 200 $/t the chemical costs were app. 4.5 times higher than value of liberated metals. As many other costs would be generated by the complete process and also metals would be recovered from the leachate, it is obvious that (1) great improvements must be obtained in the process in terms of leaching yields and acid consumption, (2) an extremely cheap sulfuric acid source must be utilized and (3) added value must be generated for treated MSWI BA.

Table 5.
Comparison of sulfuric acid costs to the value of liberated valuable metals (per ton of treated MSWI BA) [30,31].
After the heap leaching, valuable metals must be recovered from the pregnant leach solution. This can be done by conventional technologies, such as chemical precipitation, solvent-extraction or ion-exchange [27,28,32]. Moreover, other elements and compounds must be removed from the leachate, to allow water discharge back to environment or circulation back to process. The iron and aluminum removal can be conducted by chemical precipitation with lime, which also results in precipitation of calcium sulfate from the solution [32,33]. Residual elements can also be removed before discharge/recycling by chemical precipitation with lime, if pH is raised to basic area [27,33].
Finally, it is noteworthy that sulfuric acid leaching system has its limitations as some elements are not mobile with this chemistry (e.g., lead forms insoluble PbSO4 in H2SO4 leaching) [34]. One possibility is to utilize organic acids that are known to dissolve variety of metals, including Pb [18,20,34]. Organic acids can also be produced by heterotrophic bioleaching method, studied with MSWI fly ashes [35,36]. Currently, it cannot be stated which leaching chemistry is superior for the MSWI BA treatment, and more research is needed regarding acid consumption (and consequent acid cost), leaching duration and leaching yields, obtained leachate composition and the effect of these factors on the down-stream processing possibilities with water circulation. Moreover, sequential leaching, as well as additive chemicals (e.g., pH buffers) may be considered to utilize the strengths of different leaching chemistries and avoid the limitations.
5. Conclusions
MSWI BA is the main output of municipal solid waste incineration process, both in mass and volume. It contains some heavy metals that possess market value, but may also limit the reuse. In this study, we illustrated a robust and simple heap leaching method for recovering zinc and copper from the MSWI BA. Leaching yields for zinc and copper varied between 18–53% and 6–44%, respectively. For intensified copper dissolution, aeration was needed. The main contaminants, iron and aluminum, were easily liberated from the material by sulfuric acid, setting limitations for the industrial utilization of the process. Moreover, the extreme heterogeneity (elemental composition, mineralogy, size, dimension, porosity and wearing fragility) of the material is challenging and seems to cause very different physical heap behavior and requirements compared to the heap leaching of, e.g., ores. With the current results, economics are very challenging for the acid heap leaching process, requiring improvements on leaching efficiency, introducing a cheaper sulfuric acid source, and clear value increase for treated MSWI BA. Despite the challenges, we stress that the heap leaching of MSWI BA should be further studied, with the two main aims of optimizing the leaching chemistry and duration, and physical questions of irrigation.
Author Contributions
Conceptualization, J.M., M.S., J.S. and P.K.; methodology, J.M., M.S. and P.K.; validation, formal analysis and investigation, J.M. and M.S.; resources and data curation M.S. and J.M.; writing—original draft preparation, review and editing, J.M., M.S., J.S. and P.K. visualization, J.M.
Funding
The authors greatly acknowledge the Business Finland Circular Metal Ecosystem (CMEco) project (8161/31/2016) for funding the research.
Conflicts of Interest
The authors declare no conflict of interest.
References
- Eurostat, Eurostat Database, Municipal Waste. 2018. Available online: https://ec.europa.eu/eurostat/statistics-explained/index.php/Municipal_waste_statistics (accessed on 5 November 2018).
- Sabbas, T.; Polettini, A.; Pomi, R.; Astrup, T.; Hjelmar, O.; Mostbauer, P.; Cappai, G.; Magel, G.; Salhofer, S.; Speiser, C.; et al. Management of municipal solid waste incineration residues. Waste Manag. 2003, 23, 61–88. [Google Scholar] [CrossRef]
- Chimenos, J.M.; Segarra, M.; Fernández, M.A.; Espiell, F. Characterization of the bottom ash in municipal solid waste incinerator. J. Hazard. Mater. 1999, 64, 211–222. [Google Scholar] [CrossRef]
- Müller, U.; Rübner, K. The microstructure of concrete made with municipal waste incinerato bottom ash as an aggregate component. Cem. Concr. Res. 2006, 36, 1434–1443. [Google Scholar] [CrossRef]
- Todorovic, J.; Ecke, H. Treatment of MSWI Residues for Utilization as Secondary Construction Minerals: A Review of Methods. Miner. Energy 2006, 3, 45–59. [Google Scholar] [CrossRef]
- Kuo, N.-W.; Ma, H.-W.; Yang, Y.-M.; Hsiao, T.-Y.; Huang, C.-M. An investigation on the potential of metal recovery from the municipal waste incinerator in Taiwan. Waste Manag. 2007, 27, 1673–1679. [Google Scholar] [CrossRef]
- Agcasulu, I.; Akcil, A. Metal Recovery from Bottom Ash of An Incineration Plant: Laboratory Reactor Tests. Miner. Process. Extr. Metall. Rev. 2017, 38, 199–206. [Google Scholar] [CrossRef]
- Bayuseno, A.P.; Schmahl, W.W. Understanding the chemical and mineralogical properties of the inorganic portion of MSWI bottom ash. Waste Manag. 2010, 30, 1509–1520. [Google Scholar] [CrossRef] [PubMed]
- Kowalski, P.R.; Kasina, M.; Michalik, M. Metallic Elements Fractionation in Municipal Solid Waste Incineration Residues. Energy Procedia 2016, 97, 31–36. [Google Scholar] [CrossRef]
- Wei, Y.; Shimaoka, T.; Saffarzadeh, A.; Takahashi, F. Mineralogical characterization of municipal waste incineration bottom ash with an emphasis on heavy metal-bearing phases. J. Hazard. Mater. 2011, 187, 534–543. [Google Scholar] [CrossRef] [PubMed]
- Fedje, K.K.; Ekberg, C.; Skarnemark, G.; Steenari, B.-M. Removal of hazardous metals from MSW fly ash—An evaluation of ash leaching methods. J. Hazard. Mater. 2010, 173, 310–317. [Google Scholar] [CrossRef]
- Weibel, G.; Eggenberger, U.; Kulik, D.A.; Hummel, W.; Schlumberger, S.; Klink, W.; Fisch, M.; Mäder, U.K. Extraction of heavy metals from MSWI fly ash using hydrochloric acid and sodium chloride solution. Waste Manag. 2018, 76, 457–471. [Google Scholar] [CrossRef] [PubMed]
- Herck, P.V.; Bruggen, V.; Vogels, G.; Vandecasteele, C. Application of computer modelling to predict the leaching behaviour of heavy metals from MSWI fly ash and comparison with a sequential extraction method. Waste Manag. 2000, 20, 203–210. [Google Scholar] [CrossRef]
- Meima, J.A.; Comans, R.N. The leaching of trace elements from municipal solid waste incinerator bottom ash at different stages of weathering. Appl. Geochem. 1999, 14, 159–171. [Google Scholar] [CrossRef]
- Hyks, J.; Astrup, T.; Christensen, T.H. Leaching from MSWI bottom ash: Evaluation of non-equilibrium in column percolation experiments. Waste Manag. 2009, 29, 522–529. [Google Scholar] [CrossRef]
- Sivula, L.; Sormunen, K.; Rintala, J. Leachate formation and characteristics from gasification and grate incineration bottom ash under landfill conditions. Waste Manag. 2012, 32, 780–788. [Google Scholar] [CrossRef]
- Yao, J.; Li, W.; Xia, F.; Wang, J.; Fang, C.; Shen, D. Investigation of Cu leaching from municipal solid waste incinerator bottom ash with a comprehensive approach. Front. Energy 2011, 5, 340–348. [Google Scholar] [CrossRef]
- Zhang, H.; He, P.-J.; Shao, L.-M.; Li, X.-J. Leaching behavior of heavy metals from municipal solid waste incineration bottom ash and its geochemical modeling. J. Mater. Cycles. Waste Manag. 2008, 10, 7–13. [Google Scholar] [CrossRef]
- Tang, J.; Steenari, B.-M. Leaching optimization of municipal solid waste incineration ash for resource recovery: A case study of Cu, Zn, Pb and Cd. Waste Manag. 2016, 48, 315–322. [Google Scholar] [CrossRef]
- Jadhav, U.U.; Biswal, B.K.; Chen, Z.; Yang, E.-H.; Hocheng, H. Leaching of Metals from Incineration Bottom Ash Using Organic Acids. J. Sustain. Metall. 2018, 4, 115–125. [Google Scholar] [CrossRef]
- Funari, V.; Gomes, H.; Cappelletti, M.; Dinnelli, E.; Fedi, S.; Rogerson, M.; Mayes, W. Bioleaching of fly ash and bottom ash from Municipal Solid Waste Incineration for metal recovery. In Proceedings of the NAXOS 2018 6th International Conference on Sustainable Solid Waste Management, Naxos Island, Greece, 13–16 June 2018. [Google Scholar]
- Rawlings, D.E. Heavy Metal Mining Using Microbes. Annu. Rev. Microbiol. 2002, 56, 65–91. [Google Scholar] [CrossRef] [PubMed]
- Mäkinen, J.; Bachér, J.; Kaartinen, T.; Wahlström, M.; Salminen, J. The effect of flotation and parameters for bioleaching of printed circuit boards. Miner. Eng. 2015, 75, 26–31. [Google Scholar] [CrossRef]
- Brierley, C. Microbial mining. Sci. Am. 1982, 247, 42–50. [Google Scholar] [CrossRef]
- Ballor, N.R.; Nesbitt, C.C.; Lueking, D.R. Recovery of scrap iron metal value using biogenerated ferric iron. Biotechnol. Bioeng. 2006, 93, 1089–1094. [Google Scholar] [CrossRef] [PubMed]
- Halinen, A.-K. Heap Bioleaching of Low-Grade Multimetal Sulphidic Ore in Boreal Conditions. Ph.D. Thesis, Tampere University of Technology, Tampere, Finland, 2015. [Google Scholar]
- Monhemius, J. Precipitation diagrams for metal hydroxides, sulfides, arsenates and phosphates. Tran. Inst. Min. Metall. Sect. C 1977, 86, 202–206. [Google Scholar]
- Lewis, A.E. Review of metal sulphide precipitation. Hydrometallurgy 2010, 104, 222–234. [Google Scholar] [CrossRef]
- Bigham, J.M.; Schwertmann, U.; Pfab, G. Influence of pH on mineral speciation in a bioreactor simulating acid mine drainage. Appl. Geochem. 1996, 11, 845–849. [Google Scholar] [CrossRef]
- London Metal Exchange. Available online: https://www.lme.com/ (accessed on 19 March 2019).
- Alibaba. Available online: https://www.alibaba.com/showroom/sulfuric-acid.html (accessed on 19 March 2019).
- Free, M.L. Hydrometallurgy: Fundamentals and Applications, 1st ed.; John Wiley & Sons, Inc.: Hoboken, NJ, USA, 2013; 444p. [Google Scholar]
- Kinnunen, P.; Kyllönen, H.; Kaartinen, T.; Mäkinen, J.; Heikkinen, J.; Miettinen, V. Sulphate removal from mine water with chemical, biological and membrane technologies. Water Sci. Technol. 2017, 1, 194–205. [Google Scholar] [CrossRef]
- Huang, K.; Inoue, K.; Harada, H.; Kawakita, H.; Ohto, K. Leaching of heavy metals by citric acid from fly ash generated in municipal waste incineration plants. J. Mater. Cycles Waste Manag. 2011, 13, 118–126. [Google Scholar] [CrossRef]
- Xu, T.-J.; Ting, Y.-P. Fungal bioleaching of incinerator fly ash: Metal extraction and modeling growth kinetics. Enzyme Microb. Technol. 2009, 44, 323–328. [Google Scholar] [CrossRef]
- Wang, Q.; Yang, J.; Wang, Q.; Wu, T. Effects of water-washing pretreatment on bioleaching of heavy metals from municipal solid waste incineration fly ash. J. Hazard. Mater. 2009, 162, 812–818. [Google Scholar] [CrossRef]
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).