Mineralogy of Zirconium in Iron-Oxides: A Micron- to Nanoscale Study of Hematite Ore from Peculiar Knob, South Australia
Abstract
:1. Introduction
2. Geological Background
3. Methodology
4. Results
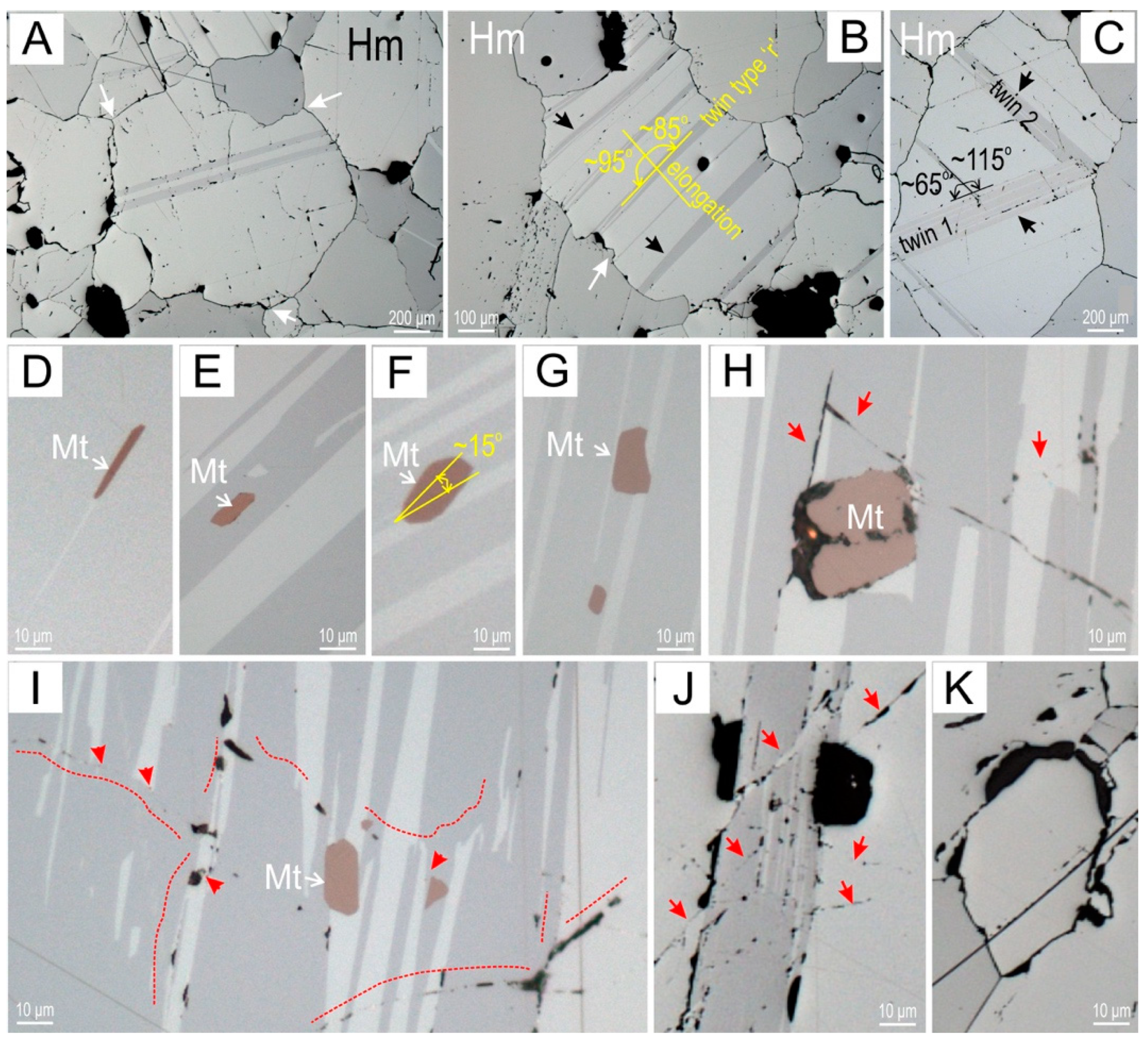
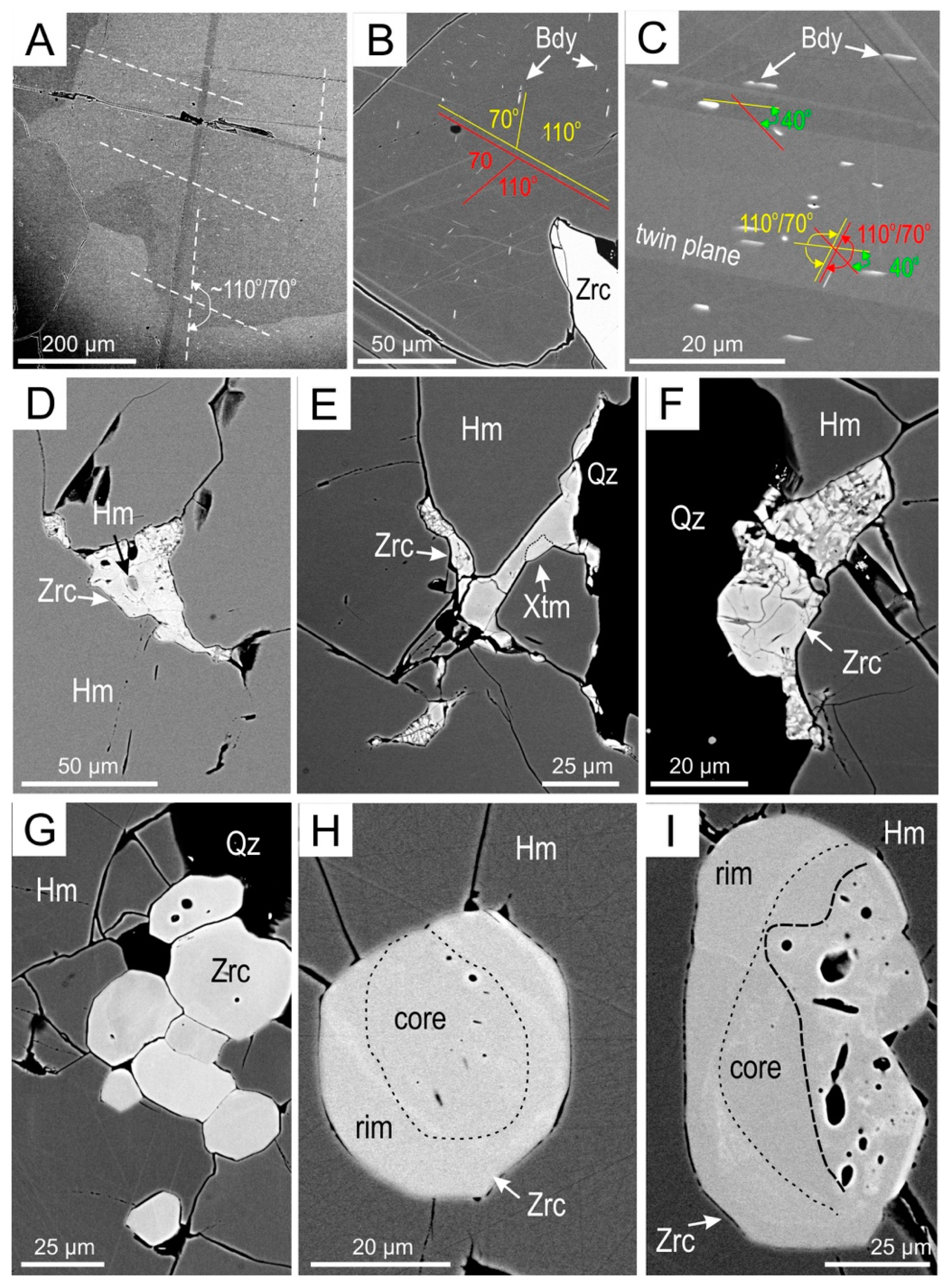
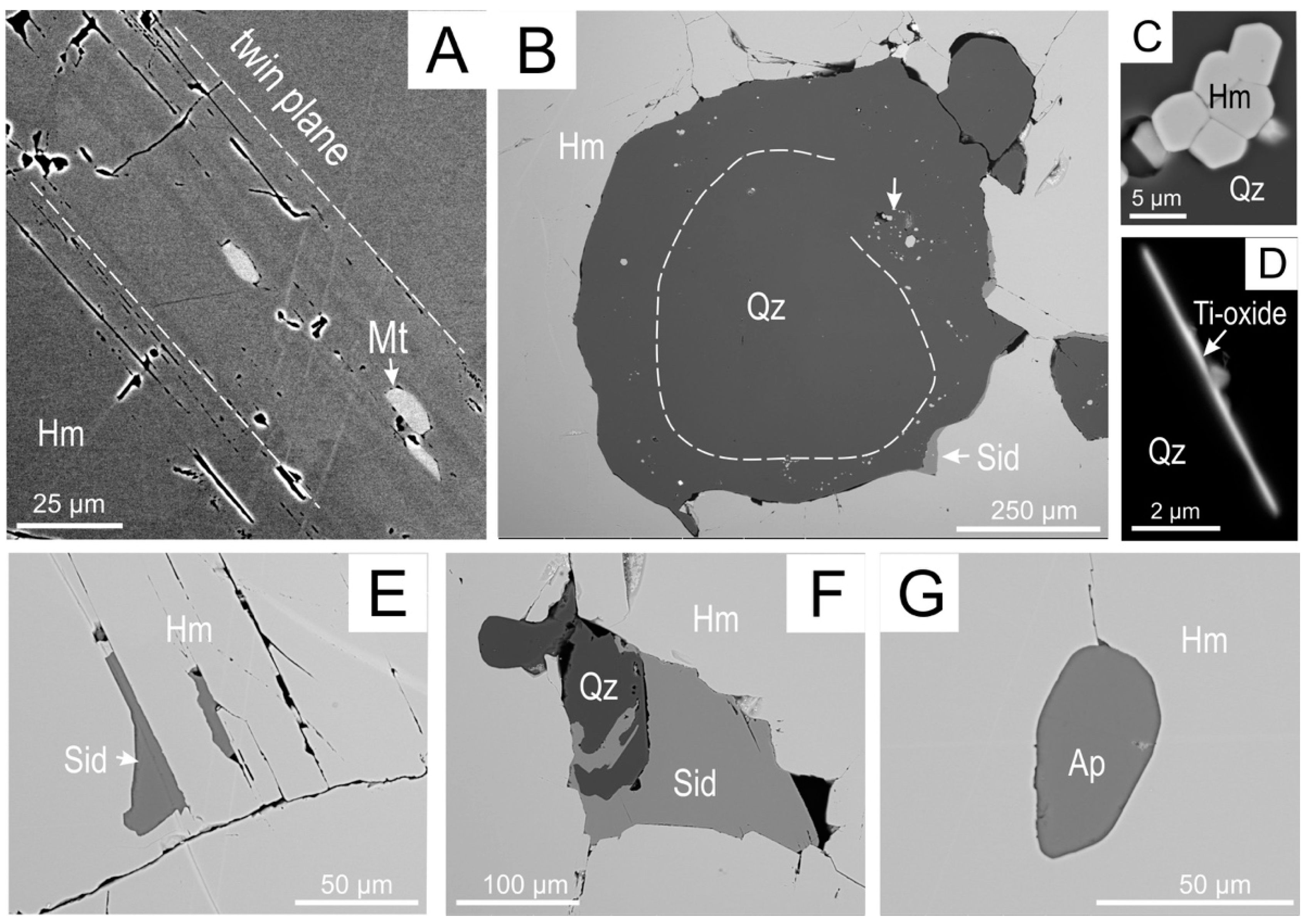
4.1. Petrography
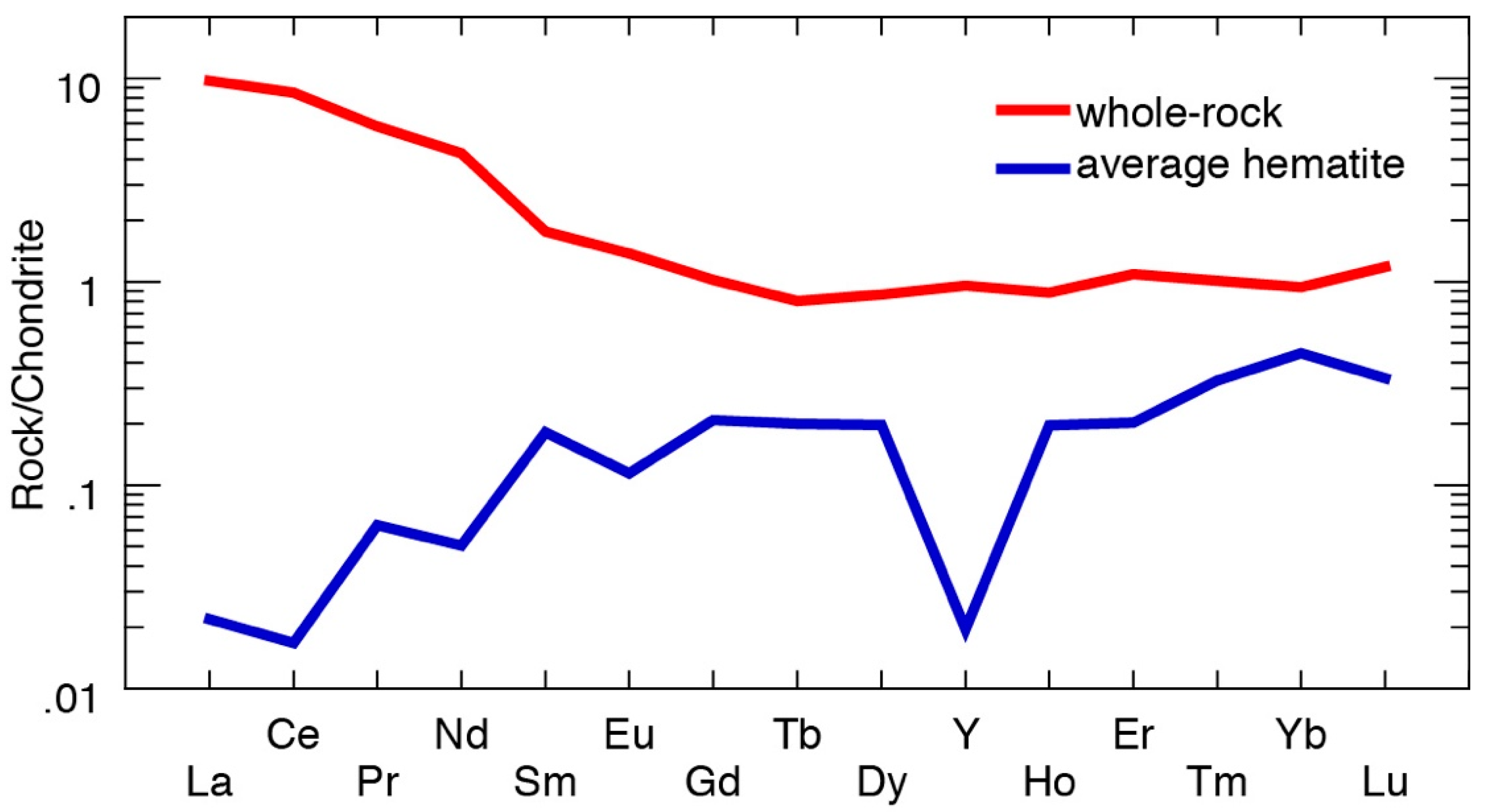
4.2. Geochemistry
4.2.1. Whole-Rock Data
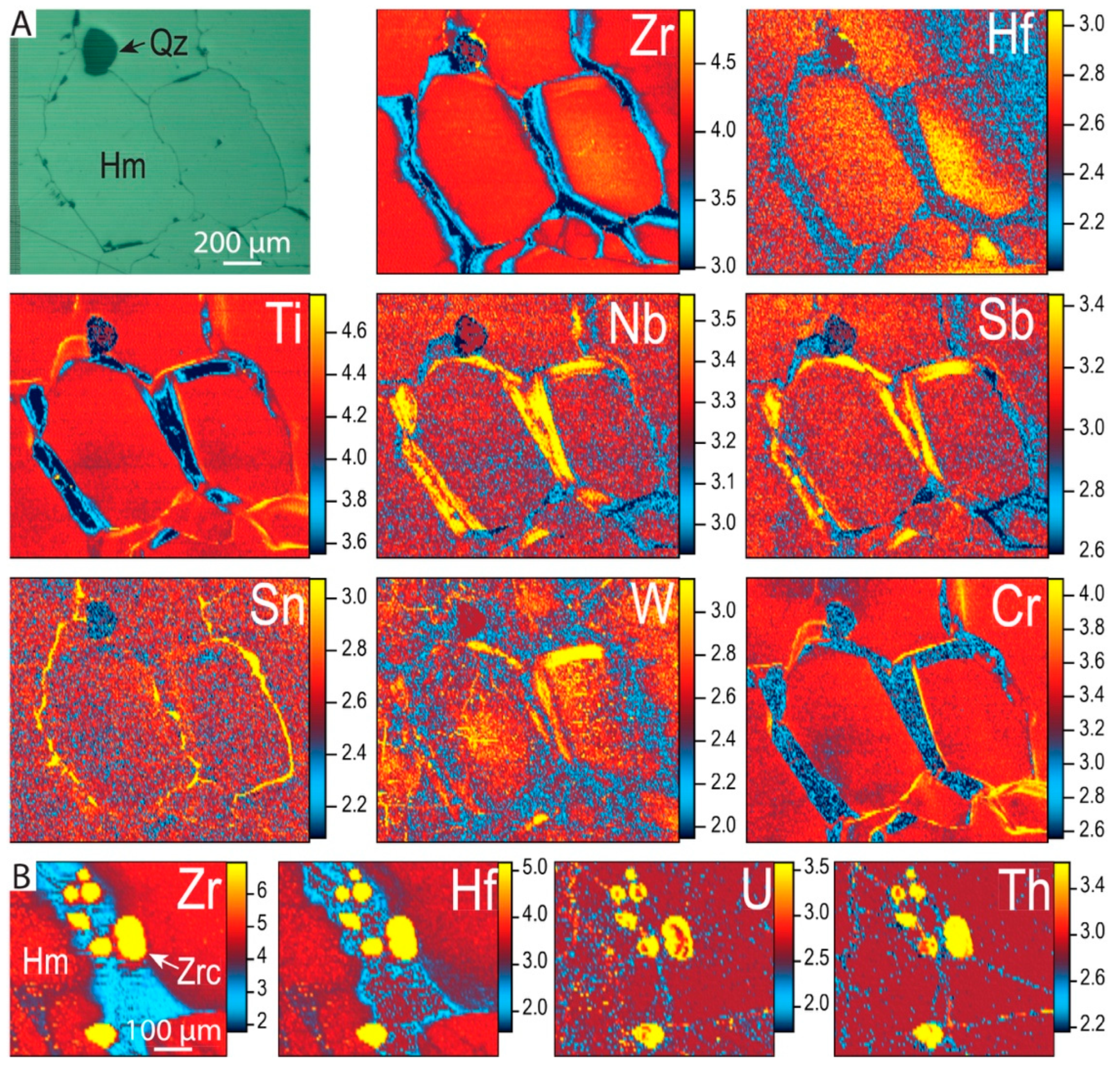
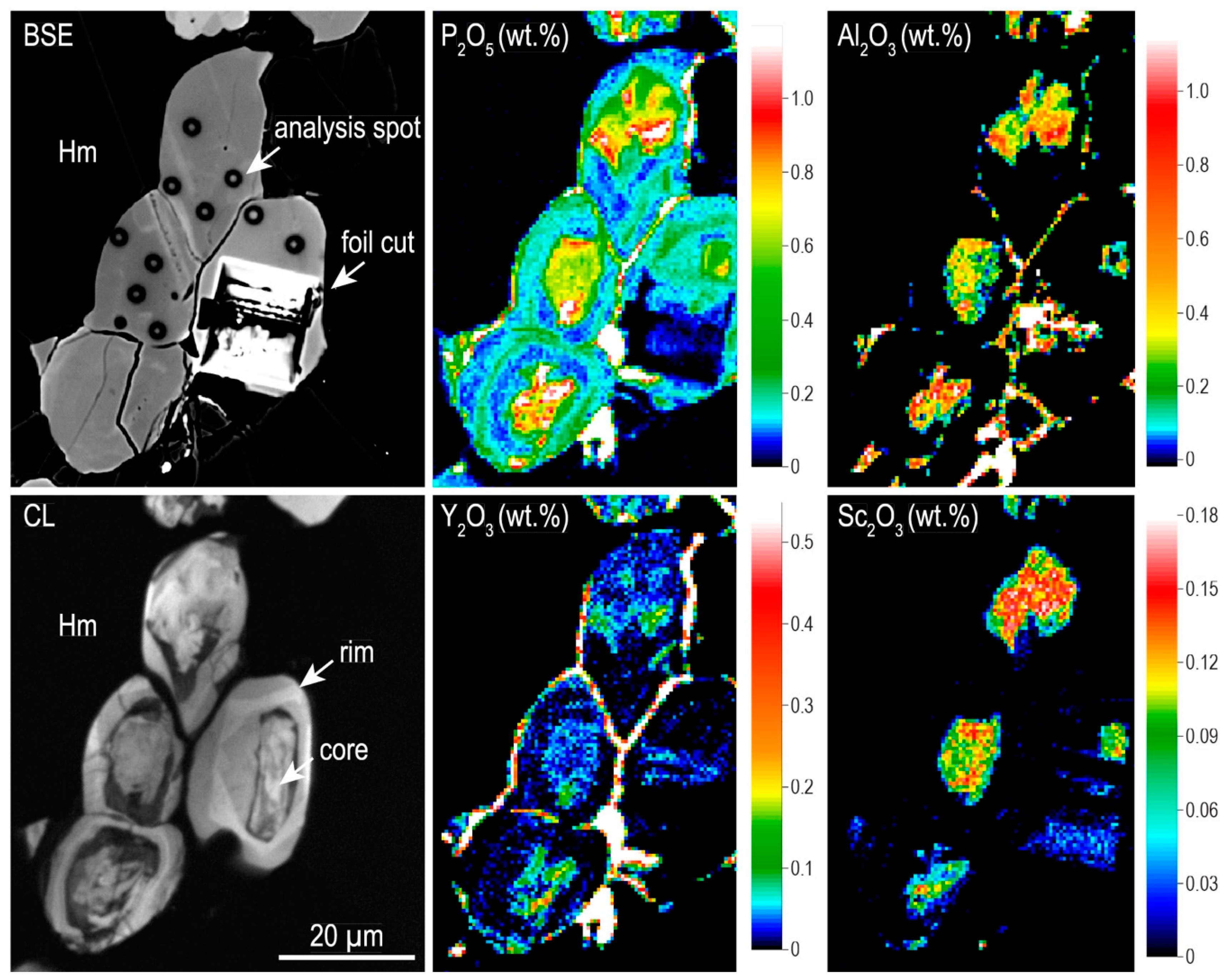
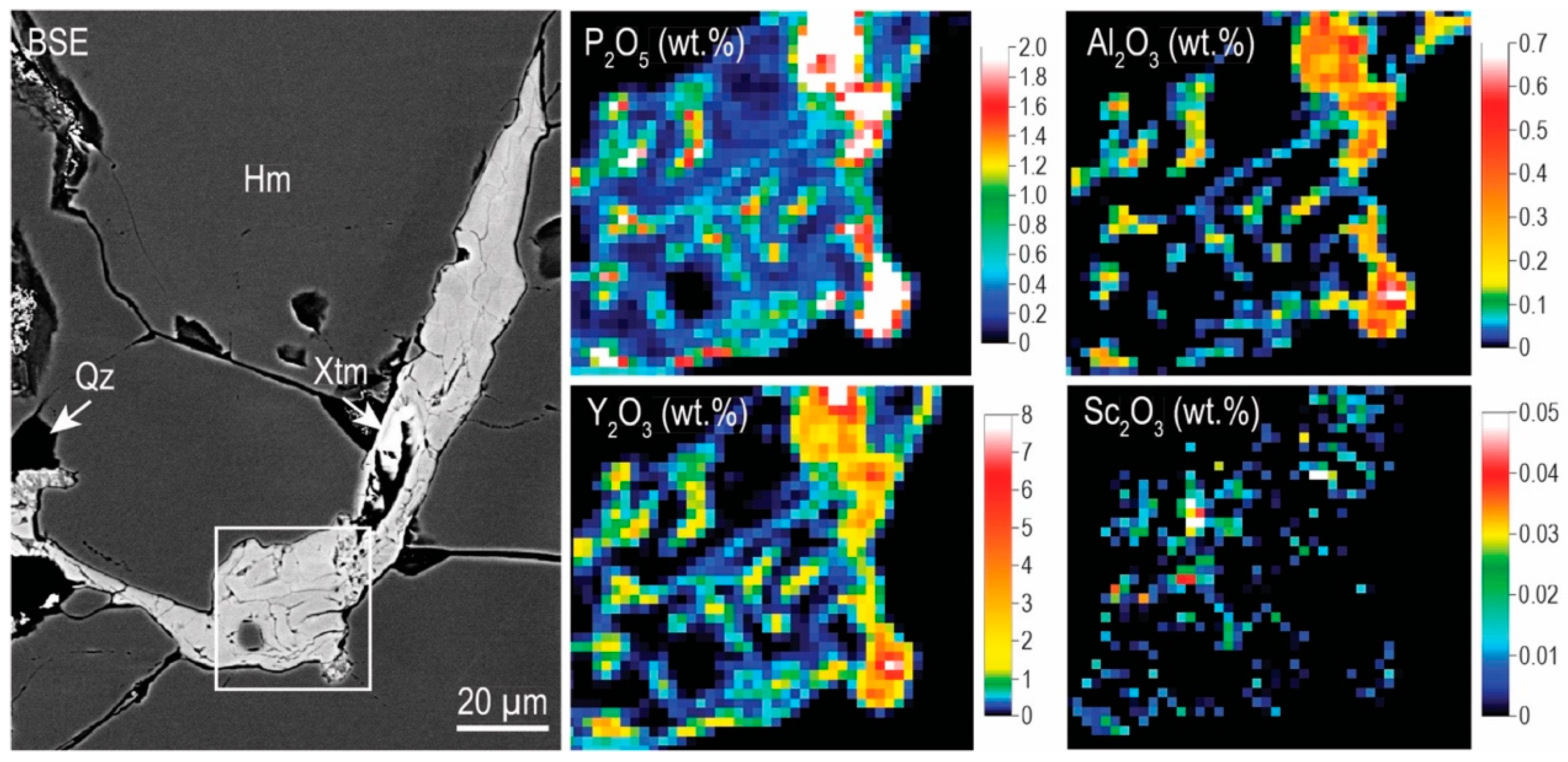
4.2.2. Trace Element Concentrations and Grain-Scale Distribution in Hematite
4.2.3. Zircon Electron Microprobe Data
4.3. Nanoscale Mineral Study
4.3.1. Baddeleyite
4.3.2. Zircon
4.4. U–Pb SHRIMP Zircon Geochronology
5. Discussion
5.1. Recognition of Metamorphic Textures
5.2. Bridging Micron- to Nanoscale Observations in Hematite: Reconstruction of Ore Evolution
5.3. Zirconium Mineralogy: Geological Evolution at Peculiar Knob
6. Conclusions and Implications
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Dawson, J.B.; Steele, I.M.; Smith, J.V.; Rivers, M.L. Minor and trace element chemistry of carbonates, apatites and magnetites in some African carbonatites. Mineral. Mag. 1996, 60, 415–425. [Google Scholar] [CrossRef]
- Reguir, E.P.; Chakhmouradian, A.R.; Halden, N.M.; Yang, P.; Zaitsev, A.N. Early magmatic and reaction-induced trends in magnetite from the carbonatites of Kerimasi, Tanzania. Can. Mineral. 2008, 46, 879–900. [Google Scholar] [CrossRef]
- Selmi, M.; Lagoeiro, L.E.; Endo, I. Geochemistry of hematite and itabirite, Quadrilátero Ferrífero, Brazil. Revista Escola de Minas 2009, 62, 35–43. [Google Scholar] [CrossRef]
- Cabral, A.R.; Rosière, C.A. The chemical composition of specular hematite from Tilkerode, Marz, Germany: Implications for the genesis of hydrothermal hematite and comparison with the Quadrilátero Ferrífero of Minas Gerais, Brazil. Mineral. Depos. 2013, 48, 907–924. [Google Scholar] [CrossRef]
- Verdugo-Ihl, M.R.; Ciobanu, C.L.; Cook, N.J.; Ehrig, K.; Courtney-Davies, L.; Gilbert, S. Textures and U-W-Sn-Mo signatures in hematite from the Cu-U-Au-Ag orebody at Olympic Dam, South Australia: Defining the archetype for IOCG deposits. Ore Geol. Rev. 2017, 91, 173–195. [Google Scholar] [CrossRef]
- Keyser, W.M.; Ciobanu, C.L.; Cook, N.J.; Johnson, G.; Feltus, F.; Johnson, S.; Dmitrijeva, M.; Ehrig, K.; Nguyen, P. Petrography and trace element signatures of iron-oxides in deposits from the Middleback Ranges, South Australia: From banded iron formation to ore. Ore Geol. Rev. 2018, 93, 337–360. [Google Scholar] [CrossRef]
- Gao, W.; Ciobanu, C.L.; Cook, N.J.; Huang, F.; Mang, L.; Gao, S. Petrography and trace element signatures in silicates and Fe-Ti-oxides from the Lanjiahuoshan deposit, Panzhihua layered intrusion, Southwest China. Lithos 2017, 294–295, 164–183. [Google Scholar] [CrossRef]
- Jones, T.S.; Kimura, S.I.; Muan, A. Phase relations in the system FeO–Fe2O3–ZrO2–SiO2. J. Am. Ceram. Soc. 1967, 50, 137–142. [Google Scholar] [CrossRef]
- Bechta, S.V.; Krushinov, E.V.; Al’myashev, V.I.; Vitol, S.A.; Mezentseva, L.P.; Petrov, Y.B.; Lopukh, D.B.; Khabenskii, V.B.; Barrachin, M.; Hellmann, S.; et al. Phase relations in the ZrO2–FeO system. Russian J. Inorg. Chem. 2006, 51, 325–331. [Google Scholar]
- Morris, B.J.; Hough, J.K. South Australia Steel and Energy Project, Peculiar Knob Prospect, South Australia; Department of Mines and Energy Resources Report Book 97/9; Department of Primary Industries & Resources: Adelaide, Australia, 1997.
- Nielson, S. Peculiar Knob Resource Model Development; Arrium Mining: Adelaide, Australia, 2014; 55p. [Google Scholar]
- Schmidt, P.W.; McEnroe, S.A.; Clark, D.A.; Robinson, P. Magnetic properties and potential field modeling of the Peculiar Knob metamorphosed iron formation, South Australia: An analog for the source of the intense Martian magnetic anomalies? J. Geophys. Res. 2007, 112, B03102. [Google Scholar] [CrossRef]
- Ciobanu, C.L.; Cook, N.J.; Maunders, C.; Wade, B.P.; Ehrig, K. Focused Ion Beam and Advanced Electron Microscopy for Minerals: Insights and Outlook from Bismuth Sulphosalts. Minerals 2016, 6, 112. [Google Scholar] [CrossRef]
- Flint, R.B.; Benbow, M.C. Geology of the Mount Woods Inlier; Department of Mines, South Australia Report Book 77/134; Department for Energy and Mining: Adelaide, Australia, 1977; 50p.
- Chalmers, N. The Mount Woods Domain: A Geological Review and Discussion on Mineralisation Potential; Department of Primary Industries and Resources Report Book 2007/7; Department of Primary Industries & Resources: Adelaide, Australia, 2007; 62p.
- Parker, A.J.; Lemon, N.M. Reconstruction of the early Proterozoic stratigraphy of the Gawler Craton, South Australia. J. Geol. Soc. Aust. 1982, 29, 221–238. [Google Scholar] [CrossRef]
- Vassallo, J.J.; Wilson, C.J.L. Structural repetition of the Hutchison Group metasediments, Eyre Peninsula, South Australia. Aust. J. Earth Sci. 2001, 48, 331–345. [Google Scholar] [CrossRef]
- Szpunar, M.; Hand, M.; Barovich, K.; Jagodzinski, E.; Belousova, E. Isotopic and geochemical constraints on the Paleoproterozoic Hutchison Group, southern Australia: Implications for Paleoproterozoic continental reconstructions. Precambr. Res. 2011, 187, 99–126. [Google Scholar] [CrossRef]
- Jagodzinski, E.A. Compilation of SHRIMP U-Pb geochronological data, Olympic Domain, Gawler Craton, South Australia, 2001–2003. Geosci. Aust. Rec. 2005, 20, 2005. [Google Scholar]
- Fanning, C.M. Ion-Microprobe U–Pb Zircon Dating of the Mount Woods Inlier, Preliminary Results; Research School of Earth Sciences, Australian National University: Canberra, Australia, 1993; 8p. [Google Scholar]
- Skirrow, R.G.; Bastrakov, E.N.; Barovich, K.; Fraser, G.L.; Creaser, R.A.; Fanning, C.M.; Raymond, O.L.; Davidson, G.J. Timing of iron oxide Cu-Au-(U) hydrothermal activity and Nd isotope constraints on metal sources in the Gawler Craton, South Australia. Econ. Geol. 2007, 102, 1441–1470. [Google Scholar] [CrossRef]
- Forbes, C.J.; Giles, D.; Hand, M.; Betts, P.G.; Suzuki, K.; Chalmers, N.; Dutch, R. Using P-T paths to interpret the tectonothermal setting of prograde metamorphism: An example form the northeastern Gawler Craton, South Australia. Precambr. Res. 2011, 185, 65–85. [Google Scholar] [CrossRef]
- Ambrose, G.J.; Flint, R.B. BILLA KALINA, South Australia. Explanatory Notes 250000 geological series Sheet SH/53-7. Geol. Surv. South Austr. 1981, 1, 36. [Google Scholar]
- Fanning, C.M.; Flint, R.B.; Parker, A.J.; Ludwig, K.R.; Blisset, A.H. Refined Preoterozoic evolution of the Gawler Craton, South Australia, through U–Pb zircon geochronology. Precambr. Res. 1988, 40/41, 363–386. [Google Scholar] [CrossRef]
- Finlay, J. Structural Interpretation of the Mount Woods Inlier. Ph.D. Thesis, Monash University, Melbourne, Australia, 1993. [Google Scholar]
- Holm, O. New geochronology of the Mount Woods Inlier and the central Gawler gold province. In Gawler Craton: State of Play 2004; Report Book; Department of Primary Industries & Resources: Adelaide, Australia, 2004. [Google Scholar]
- Jagodzinski, E.A.; Reid, A.J.; Chalmers, N.; Swain, G.; Frew, R.A.; Foudoulis, C. Compilation of SHRIMP U-Pb Geochronological Data for the Gawler Craton, South Australia; Department of Primary Industries & Resources Report Book 2007/21; Department of Primary Industries & Resources: Adelaide, Australia, 2007.
- Belperio, A.; Flint, R.; Freeman, H. Prominent Hill: A hematite-dominated, iron oxide copper-gold system. Econ. Geol. 2007, 102, 1499–1510. [Google Scholar] [CrossRef]
- Betts, P.G.; Valenta, R.K.; Finlay, J. Evolution of the Mount Woods Inlier, northern Gawler Craton, Southern Australia: An integrated structural and aeromagnetic analysis. Tectonophysics 2003, 366, 83–111. [Google Scholar] [CrossRef]
- Davies, M.B. Iron ore in South Australia; Commodity Review No. 8; Primary Industries and Resources: Adelaide, Australia, 2000; 35p.
- Daly, S.J.; Fanning, C.M.; Fairclough, M.C. Tectonic evolution and exploration potential of the Gawler Craton South Australia. AGSO J. Aust. Geol. Geophys. 1988, 17, 145–168. [Google Scholar]
- Reid, A.J.; Fabris, A. Influence of preexisting low metamorphic grade sedimentary successions on the distribution of iron oxide copper-gold mineralization in the Olympic Cu-Au Province, Gawler Craton. Econ. Geol. 2015, 110, 2147–2157. [Google Scholar] [CrossRef]
- Guillong, M.; Hametner, K.; Reusser, E.; Wilson, S.A.; Günther, D. Preliminary characteristics of new glass reference materials (GSA-1G, GSC-1G, GSD-1G and GSE-1G) by laser ablation-inductively coupled plasma-mass spectrometry using 193 nm, 213 nm and 266 nm wavelengths. Geostand. Geoanal. Res. 2005, 29, 315–331. [Google Scholar] [CrossRef]
- Jochum, K.P.; Weis, U.; Stoll, B.; Kuzmin, D.; Yang, Q.; Raczek, I.; Jacob, D.E.; Stracke, A.; Gunther, D.; Enzweiler, J. Determination of reference values for NIST SRM 610-617 glasses following ISO guidelines. Geostand. Geoanal. Res. 2011, 35, 397–429. [Google Scholar] [CrossRef]
- Van Achterbergh, E.; Ryan, C.G.; Jackson, S.E.; Griffin, W.L. Data reduction software for LA-ICP-MS. In Laser-ablation-ICPMS in the Earth Sciences: Principles and Applications; Sylvester, P.J., Ed.; Mineralogical Association of Canada: Quebec City, QC, Canada, 2001; pp. 239–243. [Google Scholar]
- Woodhead, J.D.; Hellstrom, J.; Paton, C.; Hergt, J.M.; Greig, A.; Maas, R. A guide to depth profiling and imaging applications of LA-ICM-PS. In Laser Ablation ICP-MS in the Earth Sciences: Current Practices and Outstanding Issues; Mineralogical Association of Canada, Short Course Series 40; Sylvester, P.J., Ed.; Mineralogical Association of Canada: Quebec City, QC, Canada, 2008; pp. 135–145. [Google Scholar]
- Ciobanu, C.L.; Cook, N.J.; Utsunomiya, S.; Pring, A.; Green, L. Focussed ion beam transmission electron microscopy applications in ore mineralogy: Bridging micro and nanoscale observations. Ore Geol. Rev. 2011, 42, 6–31. [Google Scholar] [CrossRef]
- Compston, W.; Williams, I.S.; Meyer, C. U-Pb geochronology of zircon from lunar breccia 73217 using a sensitive high mass-resolution ion microprobe. J. Geophys. Res. 1984, 89, 525–534. [Google Scholar] [CrossRef]
- Williams, I.S. U–Th–Pb geochronology by ion microprobe. In Applications of Microanalytical Techniques to Understanding Mineralizing Processes; McKibben, M.A., Shanks, W.C., Eds.; Society of Economic Geologists: Littleton, CO, USA, 1998; Volume 7, pp. 1–35. [Google Scholar]
- McDonough, W.F.; Sun, S.S. Composition of the Earth. Chem. Geol. 1995, 120, 223–253. [Google Scholar] [CrossRef]
- Swamy, V.; Dubrovinsky, L.S.; Dubrovinskaia, N.A.; Langenhorst, F.; Simionovici, A.S.; Drakopoulos, M.; Dmitriev, V.; Weber, H.-P. Size effects on the structure and phase transition behavior of baddeleyite TiO2. Solid State Commun. 2005, 134, 541–546. [Google Scholar] [CrossRef]
- Craig, J.R.; Vaughan, D.J. Ore Microscopy and Ore Petrography, 2nd ed.; John Wiley & Sons: Hoboken, NJ, USA, 1994. [Google Scholar]
- Löffler, L.; Mader, W. Anisotropic X-ray peak broadening and twin formation in hematite derived from natural and synthetic goethite. J. Eur. Ceram. Soc. 2006, 26, 131–139. [Google Scholar] [CrossRef]
- Avila, C.F.; Lagoeiro, L.; Barbosa, P.F.; Graca, L. EBSD analysis of rhombohedral twinning in hematite crystals of naturally deformed iron formations. J. Appl. Crystallogr. 2015, 48, 212–219. [Google Scholar] [CrossRef]
- Gao, W.; Ciobanu, C.L.; Cook, N.J.; Slattery, A.; Huang, F.; Wang, D. Nanoscale study of lamellar exsolutions in clinopyroxene from olivine gabbro: Recording crystallization sequences in iron-rich layered intrusions. Am. Mineral. 2018, 104, 244–261. [Google Scholar]
- Morisset, C.; Scoates, J.S.; Weis, D.A. Partitioning of trace elements during exsolution in ilmenite-hematite series minerals by LA-ICP-MS. In Proceedings of the AGU Fall Meeting Abstracts, San Francisco, CA, USA, 12–16 December 2010. [Google Scholar]
- Lane, K.; Jagodzinski, E.A.; Dutch, R.; Reid, A.J.; Hand, M. Age constraints on the timing of iron ore mineralisation in the southeastern Gawler Craton. Aust. J. Earth Sci. 2015, 62, 55–75. [Google Scholar] [CrossRef]
- Bingen, B.; Austrheim, H.; Whitehouse, M. Ilmenite as a source for zirconium during high-grade metamorphism? Textural evidence from the Caledonides of western Norway and implications for zircon geochronology. J. Petrol. 2001, 42, 355–375. [Google Scholar] [CrossRef]
- Moëlo, Y.; Lulzac, Y.; Rouer, O.; Palvadeau, P.; Gloaguen, É.; Léone, P. Scandium mineralogy: Pretulite with scandian zircon and xenotime-(Y) within an apatite-rich oolitic ironstone from Saint-Aubin-des-Châteaux, Armorican Massif, France. Can. Mineral. 2002, 40, 1657–1673. [Google Scholar] [CrossRef]
- Cook, N.J.; Ciobanu, C.L.; O’Reilly, D.; Wilson, R.; Das, K.; Wade, B. Mineral chemistry of rare earth element (REE) mineralization, Browns Range, Western Australia. Lithos 2013, 172, 192–213. [Google Scholar] [CrossRef]
- Harley, S.L.; Kelly, N.M.; Möller, A. Zircon behavior and the thermal histories of mountain chains. Elements 2007, 3, 25–30. [Google Scholar] [CrossRef]
- Kohn, M.J.; Corrie, S.L.; Markley, C. The fall and rise of metamorphic zircon. Am. Mineral. 2015, 100, 897–908. [Google Scholar] [CrossRef]
- Keyser, W.M.; Ciobanu, C.L.; Cook, N.J.; Courtney-Davies, L.; Ehrig, K.; Gilbert, S.; McPhie, J. Links between sedimentary protoliths and IOCG Links between sedimentary protoliths and IOCG-skarn skarn alteration, Island Dam, South Australia. In Proceedings of the 14th SGA Biennial Meeting, Quebec City, QC, Canada, 20–23 August 2017; pp. 935–938. [Google Scholar]
- Cook, N.J.; Ciobanu, C.L.; George, L.; Zhu, Z.-Y.; Wade, B.; Ehrig, K. Trace Element Analysis of Minerals in Magmatic-Hydrothermal Ores by Laser Ablation Inductively-Coupled Plasma Mass Spectrometry: Approaches and Opportunities. Minerals 2016, 6, 111. [Google Scholar] [CrossRef]
- Ludwig, K.R.A. SQUID, version 2.50; Special Publication No. 5; A User’s Manual; Berkeley Geochronology Center: Berkeley, CA, USA, 2009; p. 110. [Google Scholar]
- Steiger, R.H.; Jäger, E. Subcommission of geochronology: Convention on the use of decay constants in geo- and cosmochronology. Earth Plan. Sci. Lett. 1977, 36, 359–362. [Google Scholar] [CrossRef]
- Armstrong, J.T. Quantitative analysis of silicate and oxide minerals: Comparison of Monte Carlo, ZAF, and φ(ρz) procedures. In Microbeam Analysis; Newbury, D.E., Ed.; San Francisco Press: San Francisco, CA, USA, 1988; pp. 239–246. [Google Scholar]
- Claoué-Long, J.C.; Compston, W.; Roberts, J.; Fanning, C.M. Two carboniferous ages: A comparison of SHRIMP zircon dating with conventional zircon ages and 40Ar/39Ar analysis. In Berggren, Geochronology Time Scales and Global Stratigraphic Correlation; Berggren, W.A., Kent, D.V., Aubrey, M.P., Hardenbol, J., Eds.; Society of Sedimentary Geology Special Publication: Tulsa, OK, USA, 1995; pp. 3–21. [Google Scholar]
- Goemann, K.; Donovan, J.J. Electron probe microanalysis of complex natural sulphides using shared background measurements. In Proceedings of the 14th Biennial Australian Microbeam Analysis Symposium (AMAS 2017), Brisbane, Australia, 6–10 February 2017; pp. 61–62, ISBN 978-0-9580408-6-0. [Google Scholar]
- Goemann, K.; Donovan, J.J.; Feig, S.T.; Thompson, J. “Sharing” Background Measurements in Wavelength Dispersive Electron Probe Microanalysis. In Proceedings of the Micronalysis Society—Topical Conference on Electron-Probe Microanalysis, Program Guide with Abstracts, Madison, WI, USA, 16–19 May 2016; pp. 28–29, ISBN 978-1-5323-0217-6. [Google Scholar]
- Goemann, K.; Donovan, J.J.; Feig, S.T.; Thompson, J. Shared backgrounds in wavelength-dispersive electron probe microanalysis. In Proceedings of the 15th European Workshop on Modern Developments and Applications in Microbeam Analysis, 7th Meeting of the International Union of Microbeam Analysis Societies, Konstanz, Germany, 7–11 May 2017; pp. 378–379, ISBN 978-90-8827-693-0. [Google Scholar]
- Donovan, J.J.; Rowe, M. Techniques for Improving Quantitative Analysis of Mineral Glasses. Geochim. Cosmochim. Acta 2005. Goldschmidt abstracts. [Google Scholar]
- Donovan, J.J.; Tingle, T.N. An Improved Mean Atomic Number Background Correction for Quantitative Microanalysis. Microsc. Microanal. 1996, 1, 1–7. [Google Scholar] [CrossRef]
- Donovan, J.J.; Singer, J.W.; Armstrong, J.T. A new EPMA method for fast trace element analysis in simple matrices. Am. Mineral. 2016, 101, 1839–1853. [Google Scholar] [CrossRef]


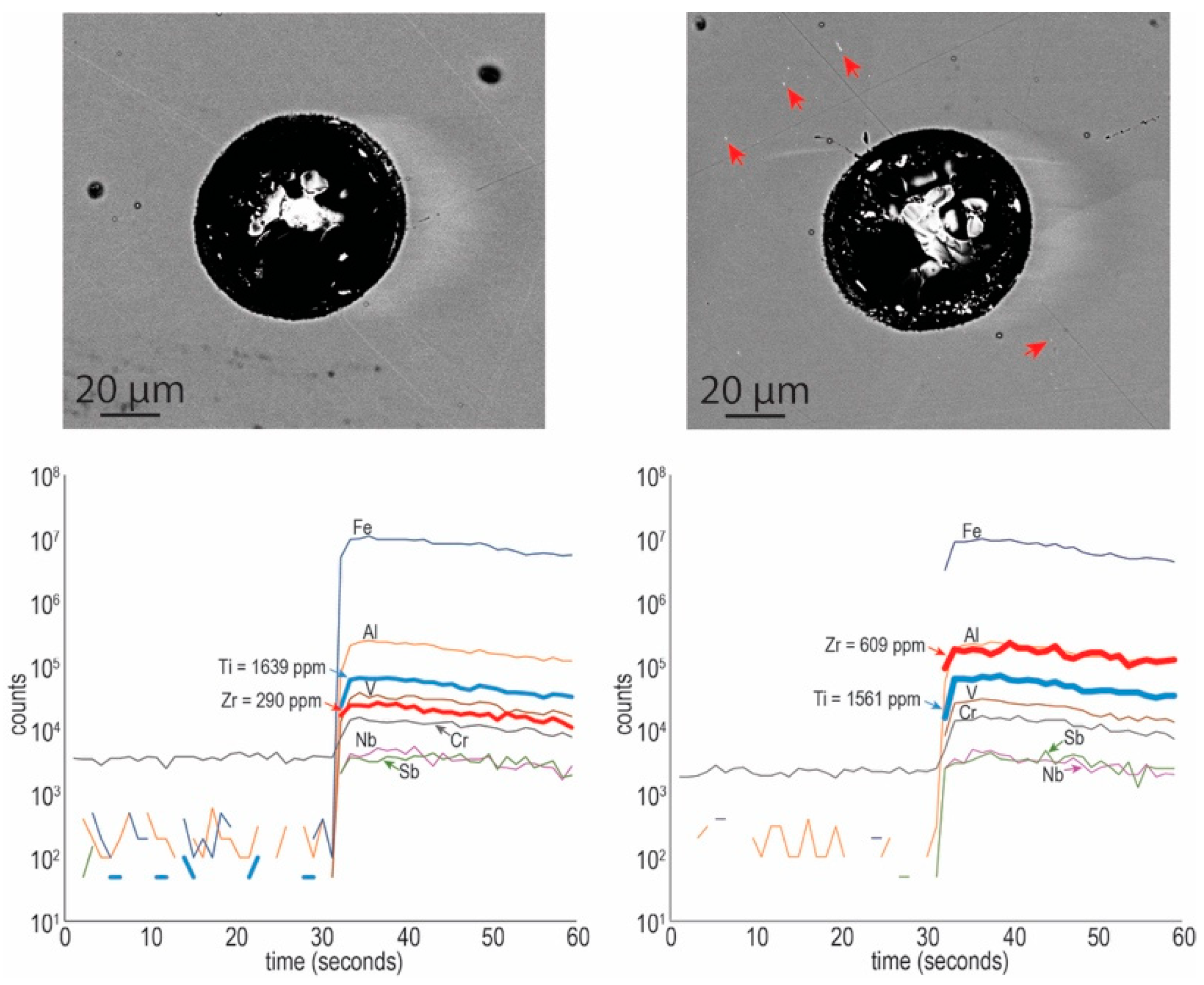
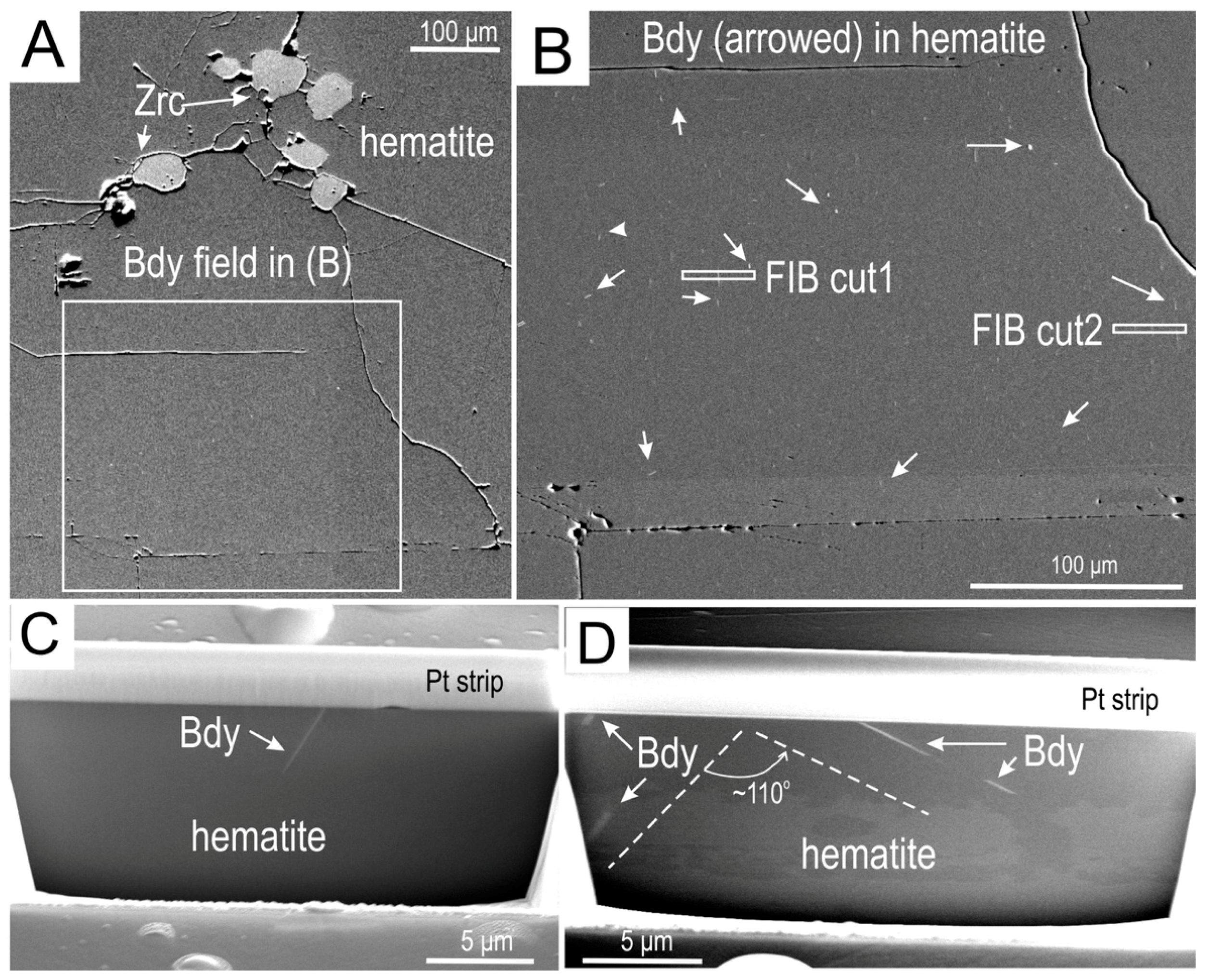

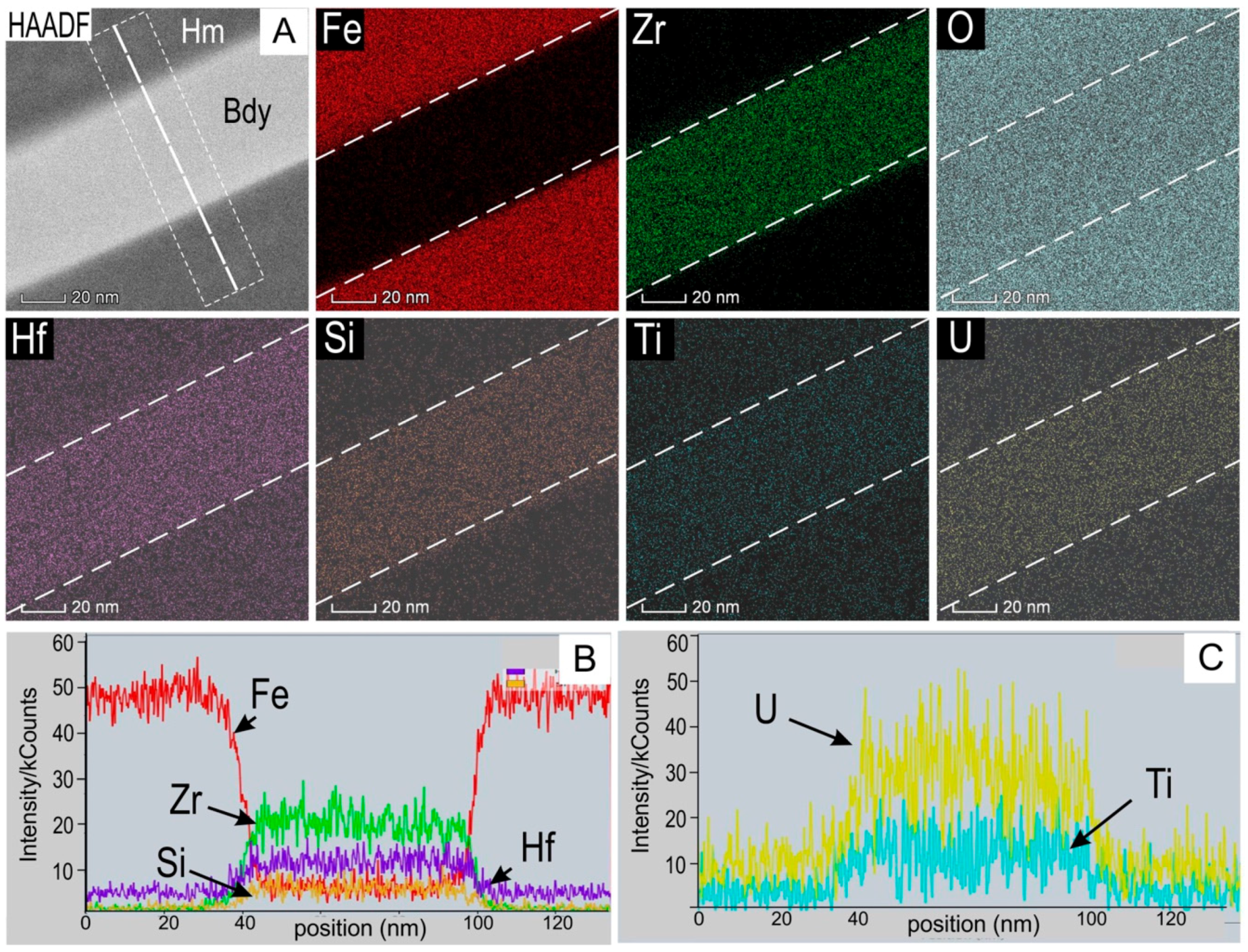
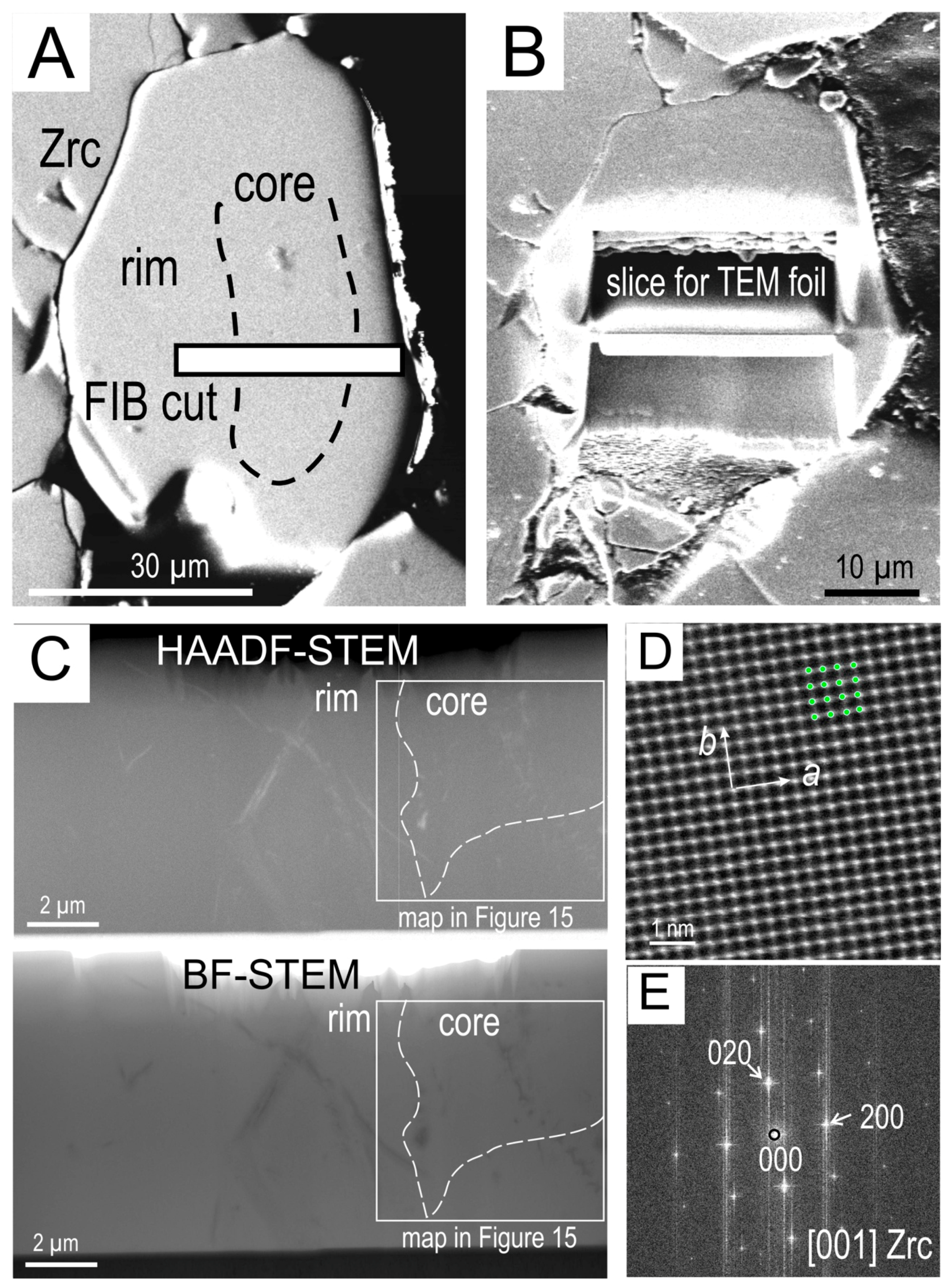

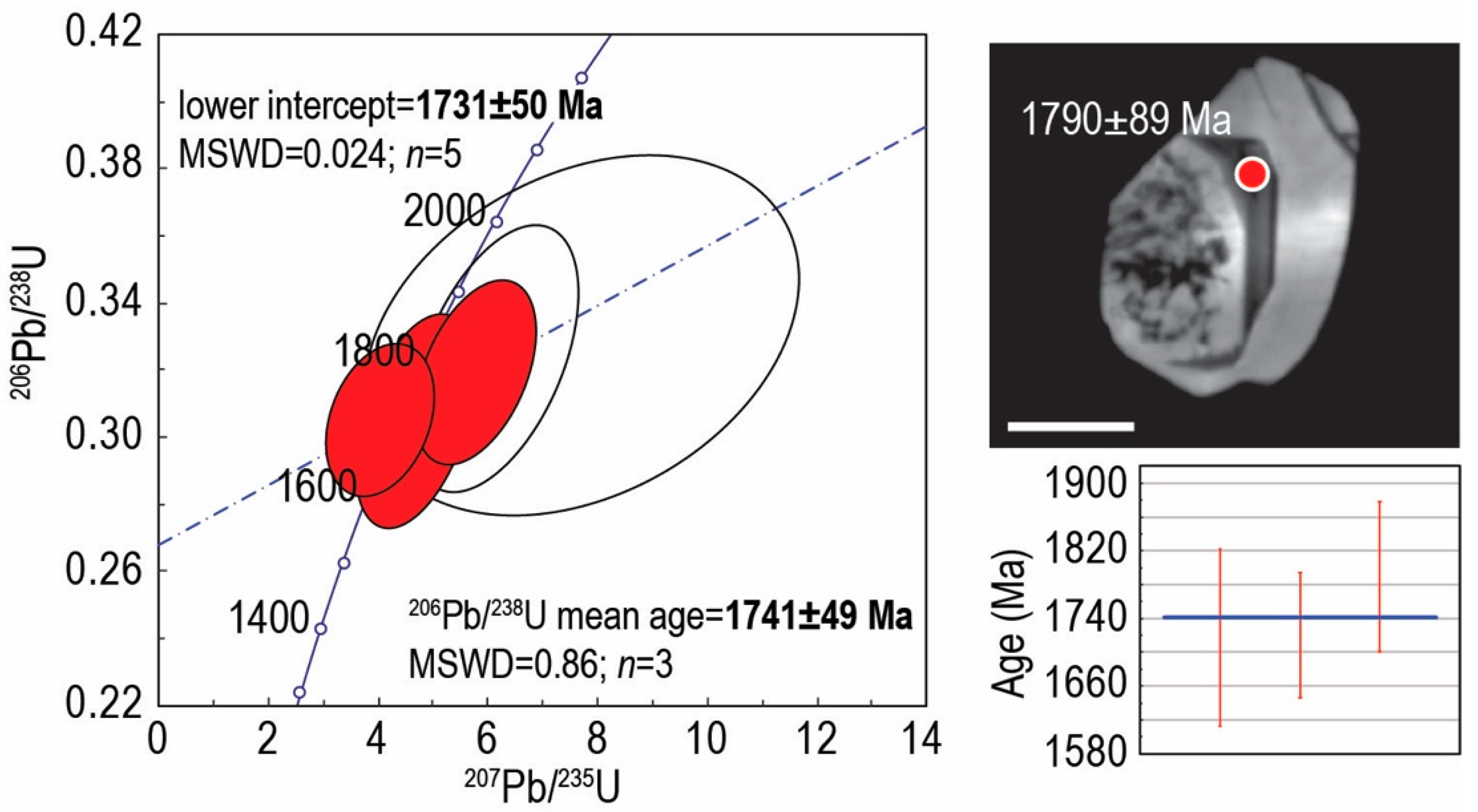
| Element (wt. %) | Element (ppm) | ||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|
| SiO2 | 0.96 | Li | 1.4 | V | 25 | Sb | 7.5 | La | 2.3 | Tm | <0.05 |
| Al2O3 | 0.06 | Ba | 1 | Nb | 5.8 | Bi | <0.05 | Ce | 5.2 | Yb | 0.16 |
| MgO | 0.02 | Sr | 1.2 | Ta | 0.3 | Pb | <5 | Pr | 0.55 | Lu | 0.03 |
| Fe2O3 | 97.81 | Be | <0.5 | Zr | 94 | Te | 0.1 | Nd | 2 | ΣREE | 12.8 |
| CaO | 0.01 | Rb | <0.1 | Hf | 2.2 | Se | <1 | Sm | 0.27 | ||
| K2O | 0.01 | Cs | <0.05 | Sc | <2 | Tl | <0.02 | Eu | 0.08 | ||
| Na2O | 0.01 | Cr | 27 | Ga | 1.7 | Mo | <1 | Gd | 0.21 | ||
| P2O5 | 0.16 | Ni | <1 | Ge | 0.1 | Re | <0.05 | Tb | 0.03 | ||
| TiO2 | 0.26 | Co | 0.2 | In | <0.05 | Sn | 4 | Dy | 0.22 | ||
| MnO | 0.01 | Cu | <1 | Au | <0.01 | W | 4 | Y | 1.5 | ||
| LOI | 0.03 | Zn | <1 | Ag | <0.1 | Th | 0.14 | Ho | 0.05 | ||
| Total + LOI | 99.47 | Cd | <0.05 | As | 1 | U3O8 | 0.16 | Er | 0.18 | ||
| Element | Geomean | Max. | Min. | s.t.d. | Element | Geomean | Max. | Min. | s.t.d. |
|---|---|---|---|---|---|---|---|---|---|
| Mg | 6.3 | 48 | 1.3 | 11 | Hf | 6.7 | 24 | 1.3 | 6.6 |
| Al | 421 | 444 | 382 | 14 | Ta | 0.50 | 0.59 | 0.42 | 0.04 |
| Si | 206 | 325 | 174 | 32 | W | 5.0 | 9.4 | 1.1 | 2.4 |
| P | 14 | 22 | 9.6 | 3.1 | 206Pb | 0.24 | 1.3 | 0.05 | 0.30 |
| Ca | 63 | 99 | 40 | 30 | 208Pb | 0.17 | 0.66 | 0.04 | 0.16 |
| Sc | 1.1 | 1.5 | 1.0 | 0.15 | Th | 0.03 | 1.2 | 0.002 | 0.48 |
| Ti | 1492 | 1881 | 1218 | 124 | U | 0.06 | 1.0 | 0.002 | 0.29 |
| V | 36 | 39 | 34 | 1.2 | La | 0.005 | 0.03 | 0.002 | 0.007 |
| Cr | 22 | 40 | 16 | 4.7 | Ce | 0.01 | 0.13 | 0.002 | 0.04 |
| Mn | 3.1 | 3.9 | 2.4 | 0.41 | Pr | 0.006 | 0.02 | 0.001 | 0.007 |
| Co | 0.69 | 1.1 | 0.31 | 0.25 | Nd | 0.02 | 0.09 | 0.008 | 0.03 |
| Ni | 0.72 | 1.2 | 0.32 | 0.25 | Sm | 0.03 | 0.10 | 0.01 | 0.03 |
| Cu | 0.55 | 0.68 | 0.41 | 0.11 | Eu | 0.007 | 0.02 | 0.002 | 0.008 |
| Zn | 0.28 | 0.55 | 0.16 | 0.12 | Gd | 0.04 | 0.34 | 0.01 | 0.11 |
| Ga | 1.8 | 2.1 | 1.5 | 0.16 | Tb | 0.007 | 0.08 | 0.002 | 0.03 |
| As | 1.3 | 3.4 | 0.44 | 0.78 | Dy | 0.05 | 0.58 | 0.006 | 0.20 |
| Sr | 0.03 | 0.55 | 0.01 | 0.16 | Y | 0.03 | 5.6 | 0.003 | 1.5 |
| Zr | 260 | 609 | 68 | 145 | Ho | 0.01 | 0.09 | 0.002 | 0.03 |
| Nb | 5.3 | 5.9 | 4.2 | 0.40 | Er | 0.03 | 0.56 | 0.005 | 0.19 |
| Mo | 0.76 | 1.0 | 0.59 | 0.09 | Tm | 0.008 | 0.09 | 0.002 | 0.03 |
| Sn | 6.4 | 11 | 4.8 | 1.6 | Yb | 0.08 | 0.77 | 0.01 | 0.25 |
| Sb | 20 | 25 | 12 | 3.2 | Lu | 0.009 | 0.14 | 0.002 | 0.04 |
| Ba | 0.16 | 0.53 | 0.08 | 0.16 | |||||
| (n = 20 hematite analyses) | |||||||||
| Texture | Granular | Skeletal | |||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Core | Rim | ||||||||||||
| # | 1 | 2 | 3 | 4 | 5 ** | 1 | 2 | 3 | 4 | 5 | 1 | 2 | 3 |
| SiO2 | 32.91 | 33.76 | 33.16 | 33.15 | 29.47 | 33.64 | 33.18 | 33.32 | 33.08 | 33.73 | 33.52 | 33.09 | 33.37 |
| Al2O3 | 0.25 | 0.27 | 0.28 | 0.26 | 0.09 | 0.19 | 0.19 | 0.18 | 0.24 | 0.21 | <mdl | <mdl | 0.19 |
| Fe2O3 * | 1.33 | 1.25 | 0.81 | 0.88 | 2.18 | 0.67 | 1.96 | 1.26 | 1.11 | 1.06 | 1.55 | 1.03 | 0.80 |
| MgO | 0.05 | 0.03 | 0.05 | 0.05 | 0.02 | 0.02 | 0.02 | 0.01 | 0.02 | 0.03 | 0.01 | 0.01 | 0.02 |
| CaO | <mdl | <mdl | 0.01 | <mdl | 0.09 | 0.02 | <mdl | <mdl | 0.01 | <mdl | <mdl | 0.03 | <mdl |
| Na2O | <mdl | 0.03 | <mdl | 0.02 | 0.05 | <mdl | <mdl | <mdl | 0.02 | <mdl | 0.04 | 0.05 | <mdl |
| P2O5 | 0.72 | 0.51 | 0.69 | 0.70 | 0.23 | 0.08 | 0.18 | 0.10 | 0.18 | 0.13 | 0.17 | 0.09 | 0.10 |
| Sc2O3 | 0.18 | 0.13 | 0.11 | 0.13 | 0.03 | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl |
| ZrO2 | 65.85 | 65.35 | 65.92 | 65.86 | 65.73 | 65.94 | 65.67 | 66.42 | 65.35 | 66.53 | 67.35 | 67.03 | 66.39 |
| HfO2 | 1.36 | 1.40 | 1.46 | 1.55 | 0.98 | 1.39 | 1.64 | 1.65 | 1.73 | 1.77 | 0.99 | 1.03 | 1.55 |
| PbO | <mdl | 0.02 | <mdl | 0.03 | <mdl | 0.02 | 0.02 | <mdl | 0.02 | <mdl | <mdl | <mdl | <mdl |
| Y2O3 | 0.04 | 0.03 | 0.05 | 0.06 | 0.10 | 0.02 | 0.02 | 0.01 | 0.16 | 0.01 | 0.06 | 0.03 | 0.02 |
| Eu2O3 | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | <mdl | 0.04 | <mdl |
| Gd2O3 | <mdl | <mdl | <mdl | <mdl | 0.05 | 0.05 | <mdl | <mdl | <mdl | <mdl | 0.07 | <mdl | <mdl |
| Tb2O3 | 0.03 | <mdl | <mdl | <mdl | 0.07 | <mdl | 0.04 | <mdl | <mdl | <mdl | <mdl | <mdl | 0.03 |
| Dy2O3 | 0.05 | <mdl | <mdl | <mdl | <mdl | 0.03 | 0.04 | <mdl | <mdl | 0.03 | <mdl | <mdl | <mdl |
| Er2O3 | 0.05 | <mdl | 0.03 | 0.03 | <mdl | <mdl | 0.05 | <mdl | 0.05 | 0.03 | 0.03 | <mdl | <mdl |
| Yb2O3 | 0.09 | 0.09 | 0.09 | 0.11 | <mdl | 0.06 | 0.06 | <mdl | 0.09 | 0.04 | <mdl | <mdl | 0.05 |
| Cl | <mdl | <mdl | <mdl | <mdl | <mdl | 0.01 | <mdl | <mdl | <mdl | <mdl | <mdl | 0.01 | <mdl |
| Total | 101.58 | 101.62 | 101.84 | 101.96 | 96.91 | 101.47 | 101.11 | 101.69 | 100.95 | 102.51 | 102.23 | 101.42 | 101.73 |
| Zircon formula: (ABZ4); calculated based on 4 oxygen per formula unit | |||||||||||||
| Al | 0.009 | 0.010 | 0.010 | 0.009 | 0.003 | 0.007 | 0.007 | 0.006 | 0.008 | 0.007 | - | - | 0.007 |
| Mg | 0.002 | 0.001 | 0.002 | 0.002 | 0.001 | 0.001 | 0.001 | - | 0.001 | 0.001 | 0.001 | - | 0.001 |
| Ca | - | - | - | - | 0.003 | 0.001 | - | - | - | - | - | 0.001 | - |
| Na | - | 0.001 | - | 0.001 | 0.003 | - | - | - | 0.001 | - | 0.002 | 0.003 | - |
| Sc | 0.005 | 0.003 | 0.003 | 0.003 | 0.001 | - | - | - | - | - | - | - | - |
| Zr | 0.964 | 0.952 | 0.962 | 0.960 | 1.029 | 0.967 | 0.969 | 0.975 | 0.966 | 0.968 | 0.982 | 0.987 | 0.974 |
| Hf | 0.012 | 0.012 | 0.012 | 0.013 | 0.009 | 0.012 | 0.014 | 0.014 | 0.015 | 0.015 | 0.008 | 0.009 | 0.013 |
| Pb | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Y | 0.001 | 0.001 | 0.001 | 0.001 | 0.002 | - | - | - | 0.003 | - | 0.001 | 0.001 | - |
| Eu | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Gd | - | - | - | - | 0.001 | - | - | - | - | - | 0.001 | - | - |
| Tb | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Dy | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Er | 0.001 | - | - | - | - | - | 0.001 | - | - | - | - | - | - |
| Yb | 0.001 | 0.001 | 0.001 | 0.001 | - | 0.001 | 0.001 | - | 0.001 | - | - | - | - |
| Tot. (A site) | 0.994 | 0.981 | 0.991 | 0.992 | 1.052 | 0.989 | 0.993 | 0.996 | 0.996 | 0.993 | 0.995 | 1.001 | 0.996 |
| ΣREY | 0.003 | 0.001 | 0.002 | 0.002 | 0.002 | 0.002 | 0.002 | 0.000 | 0.004 | 0.001 | 0.002 | 0.001 | 0.001 |
| Si | 0.988 | 1.008 | 0.992 | 0.991 | 0.946 | 1.011 | 1.004 | 1.003 | 1.003 | 1.006 | 1.002 | 0.999 | 1.004 |
| P | 0.018 | 0.013 | 0.017 | 0.018 | 0.006 | 0.002 | 0.005 | 0.003 | 0.005 | 0.003 | 0.004 | 0.002 | 0.003 |
| Tot. (B site) | 1.006 | 1.021 | 1.009 | 1.009 | 0.953 | 1.013 | 1.009 | 1.005 | 1.007 | 1.009 | 1.006 | 1.001 | 1.006 |
| O | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 |
| Cl | - | - | - | - | - | - | - | - | - | - | - | - | - |
| Tot. (Z site) | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 | 4.000 |
| Age | Isotopic Ratio | |||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| % | U | Th | 206Pb * | 232Th | 206Pb | 207Pb | 208Pb | % | 207Pb * | 207Pb * | 206Pb * | Err | ||||||||
| # | 206Pbc | ppm | ppm | ppm | /238U | ±% | /238U | ±1σ | /206Pb | ±1σ | /232Th | ±1σ | Disc. | /206Pb * | ±% | /235U | ±% | /238U | ±% | Corr |
| 1 | −1.54 | 3.2 | 7 | 0.84 | 2.3 | 1.99 | 1717 | 105 | 1805 | 253 | 1883 | 175 | 6 | 0.110 | 14 | 4.64 | 16 | 0.305 | 6.9 | 0.4 |
| 2 | 1.92 | 6.8 | 18 | 1.8 | 2.7 | 2.92 | 1720 | 74 | 1538 | 292 | 1512 | 109 | −13 | 0.096 | 16 | 4.03 | 16 | 0.306 | 4.9 | 0.3 |
| 3 | −2.03 | 2.4 | 11 | 0.68 | 4.6 | 2.25 | 1809 | 128 | 2186 | 250 | 1850 | 233 | 20 | 0.137 | 14 | 6.11 | 17 | 0.324 | 8.1 | 0.5 |
| 4 | 2.37 | 7.3 | 23 | 1.6 | 3.2 | 1.30 | 1449 | 63 | 1183 | 335 | 1376 | 90 | −25 | 0.079 | 17 | 2.76 | 18 | 0.252 | 4.9 | 0.3 |
| 5 | −2.58 | 12 | 34 | 2.7 | 3.0 | 2.62 | 1537 | 87 | 1843 | 265 | 1624 | 140 | 19 | 0.113 | 15 | 4.18 | 16 | 0.269 | 6.3 | 0.4 |
| 6 | −2.23 | 7.5 | 21 | 2.1 | 3.0 | 1.63 | 1790 | 89 | 2103 | 201 | 1938 | 173 | 17 | 0.130 | 11 | 5.75 | 13 | 0.320 | 5.7 | 0.4 |
| 7 | −3.44 | 1.8 | 27 | 0.52 | 15 | 5.43 | 1842 | 171 | 2545 | 543 | 1514 | 186 | 32 | 0.169 | 32 | 7.69 | 34 | 0.331 | 11 | 0.3 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Keyser, W.; Ciobanu, C.L.; Cook, N.J.; Feltus, H.; Johnson, G.; Slattery, A.; Wade, B.P.; Ehrig, K. Mineralogy of Zirconium in Iron-Oxides: A Micron- to Nanoscale Study of Hematite Ore from Peculiar Knob, South Australia. Minerals 2019, 9, 244. https://doi.org/10.3390/min9040244
Keyser W, Ciobanu CL, Cook NJ, Feltus H, Johnson G, Slattery A, Wade BP, Ehrig K. Mineralogy of Zirconium in Iron-Oxides: A Micron- to Nanoscale Study of Hematite Ore from Peculiar Knob, South Australia. Minerals. 2019; 9(4):244. https://doi.org/10.3390/min9040244
Chicago/Turabian StyleKeyser, William, Cristiana L. Ciobanu, Nigel J. Cook, Holly Feltus, Geoff Johnson, Ashley Slattery, Benjamin P. Wade, and Kathy Ehrig. 2019. "Mineralogy of Zirconium in Iron-Oxides: A Micron- to Nanoscale Study of Hematite Ore from Peculiar Knob, South Australia" Minerals 9, no. 4: 244. https://doi.org/10.3390/min9040244
APA StyleKeyser, W., Ciobanu, C. L., Cook, N. J., Feltus, H., Johnson, G., Slattery, A., Wade, B. P., & Ehrig, K. (2019). Mineralogy of Zirconium in Iron-Oxides: A Micron- to Nanoscale Study of Hematite Ore from Peculiar Knob, South Australia. Minerals, 9(4), 244. https://doi.org/10.3390/min9040244






