Fractal Characteristics and Heterogeneity of the Nanopore Structure of Marine Shale in Southern North China
Abstract
:1. Introduction
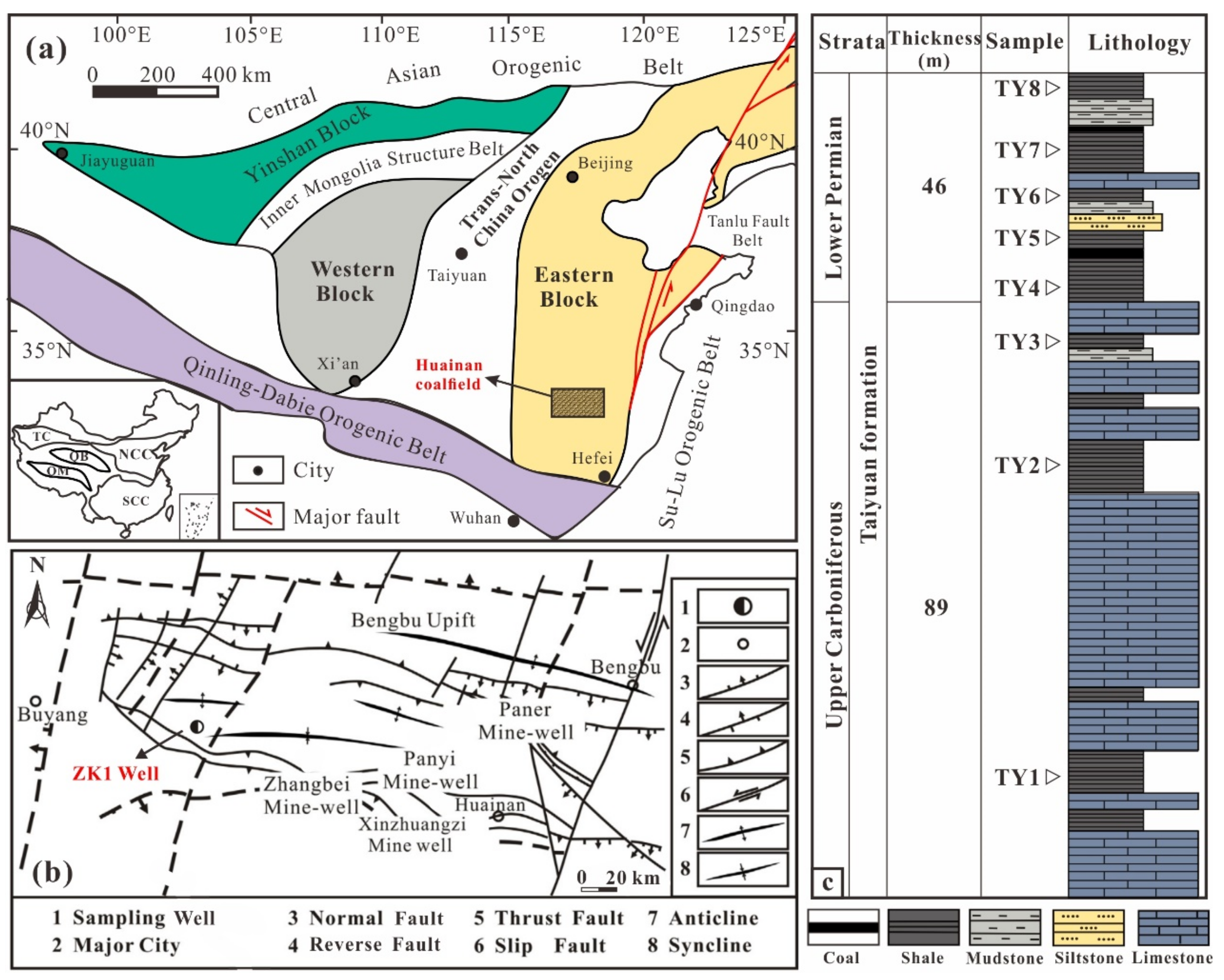
2. Geological Setting
3. Samples and Experiments
3.1. Samples
3.2. Methods
4. Results
4.1. Organic Petrology and Mineralogy
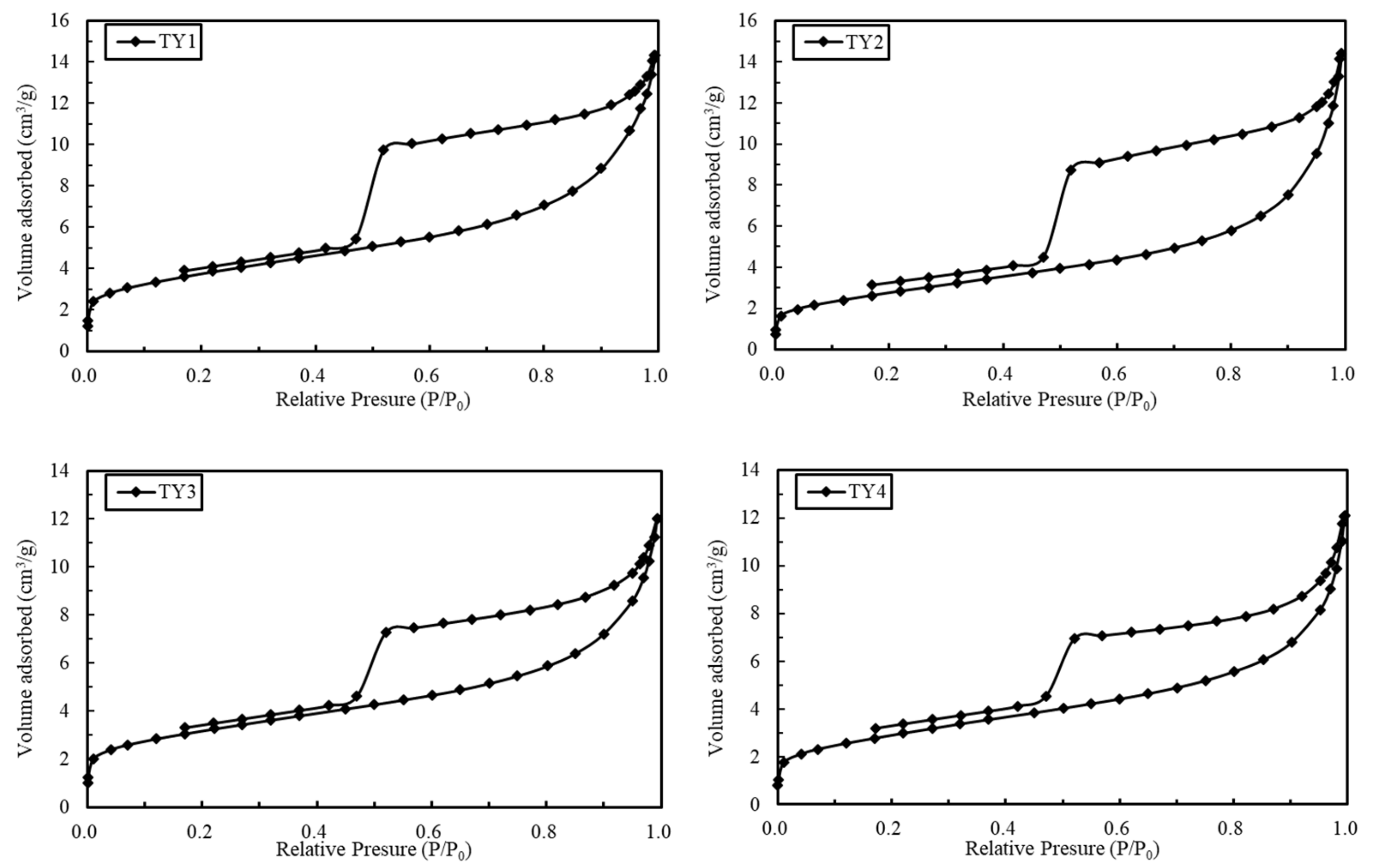
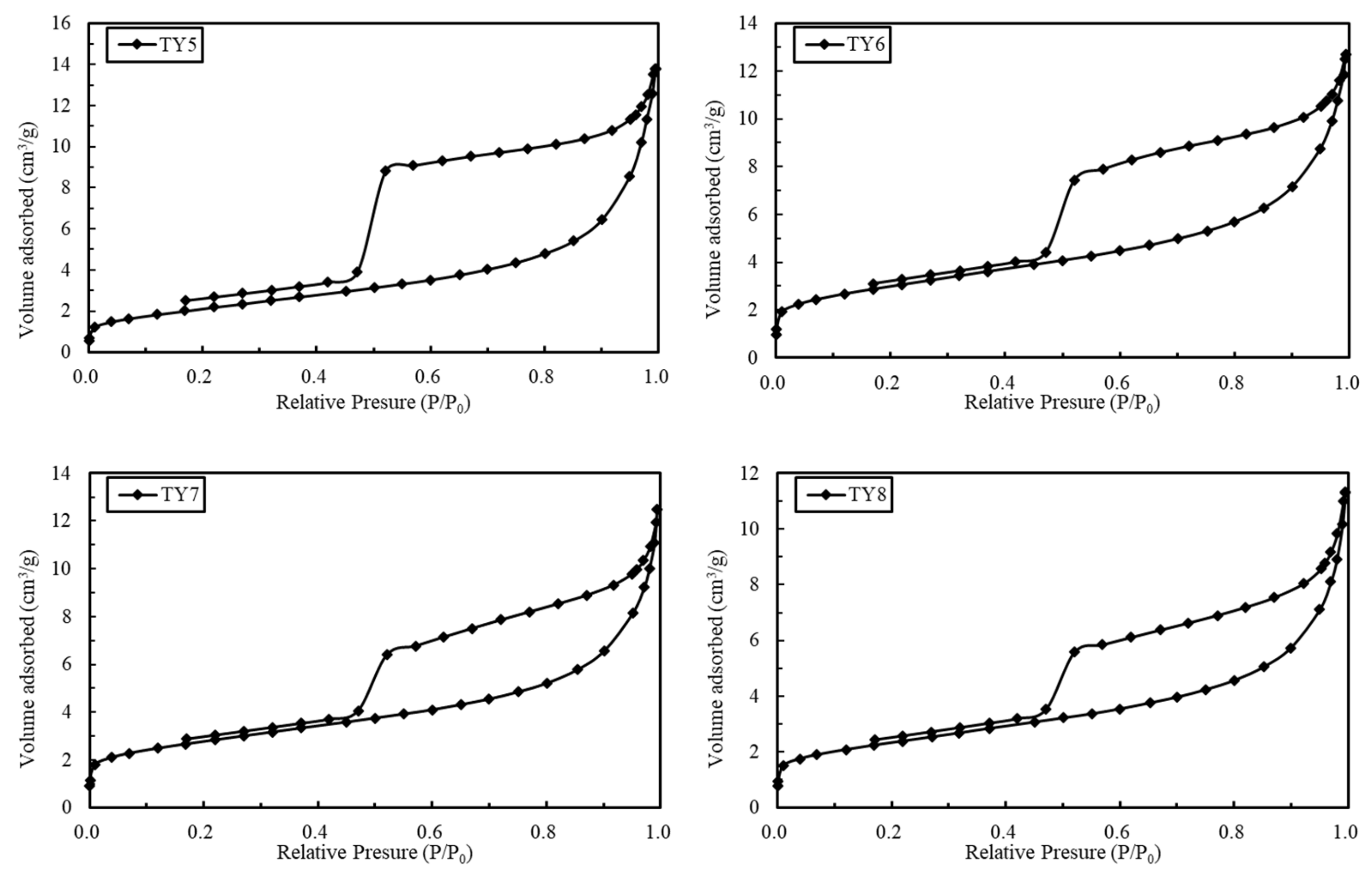
4.2. N2 Adsorption–Desorption Isotherm Characteristics
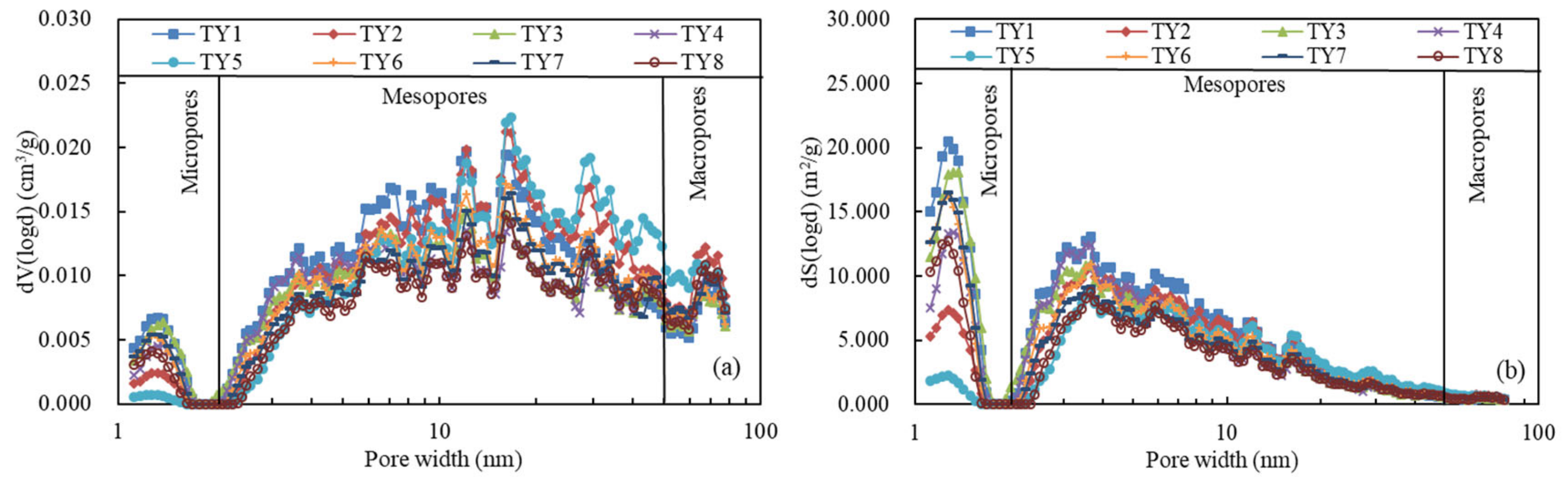
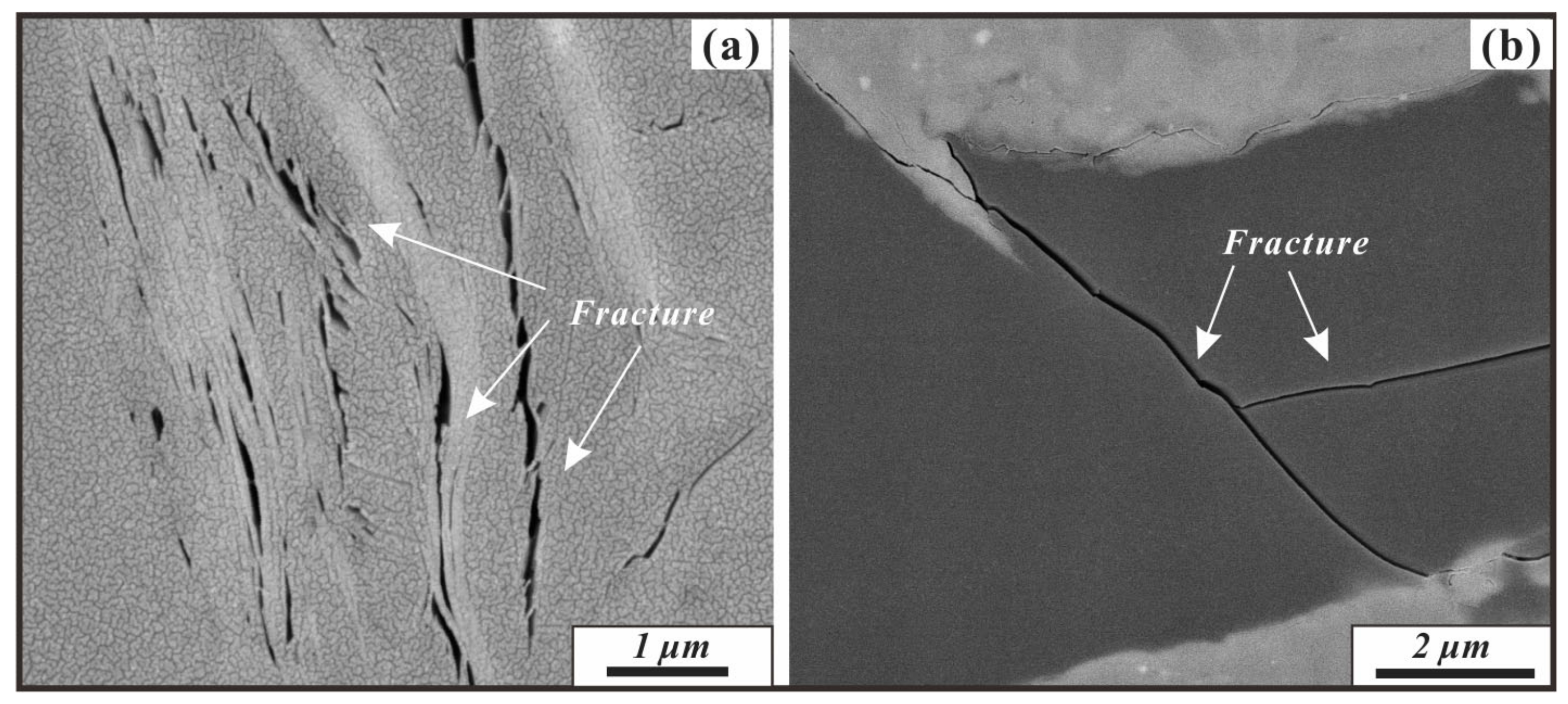
4.3. Pore Structure Parameter Characteristics
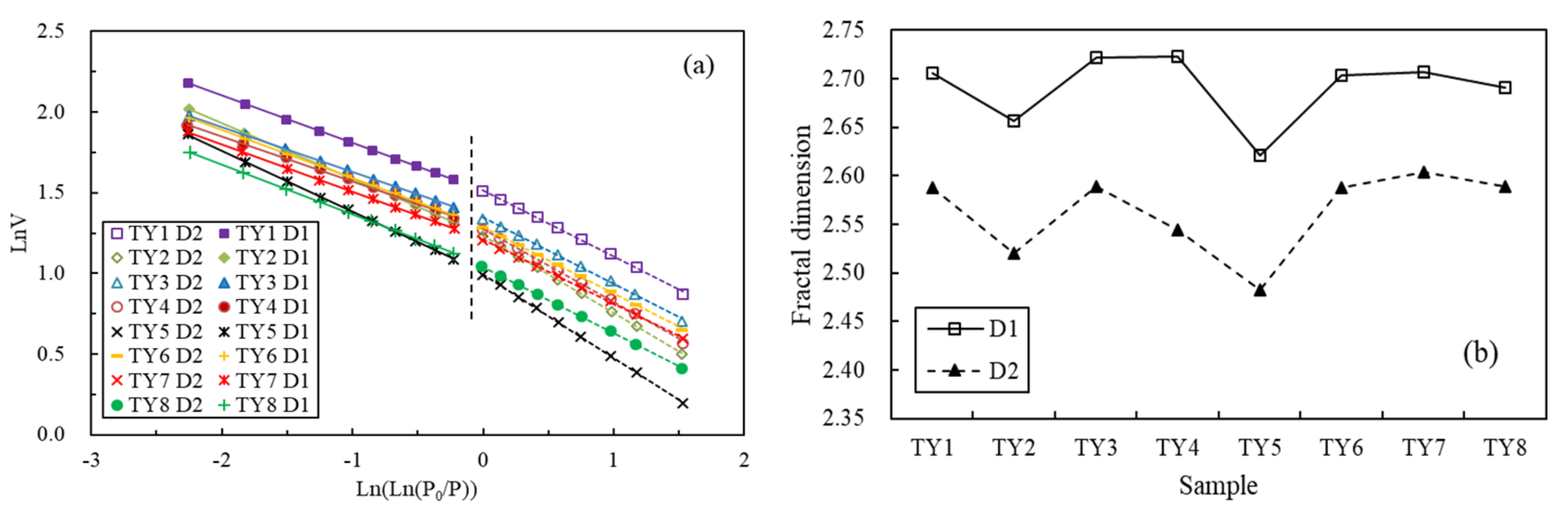
4.4. Fractal Characteristics of the Pore Structure
5. Discussion
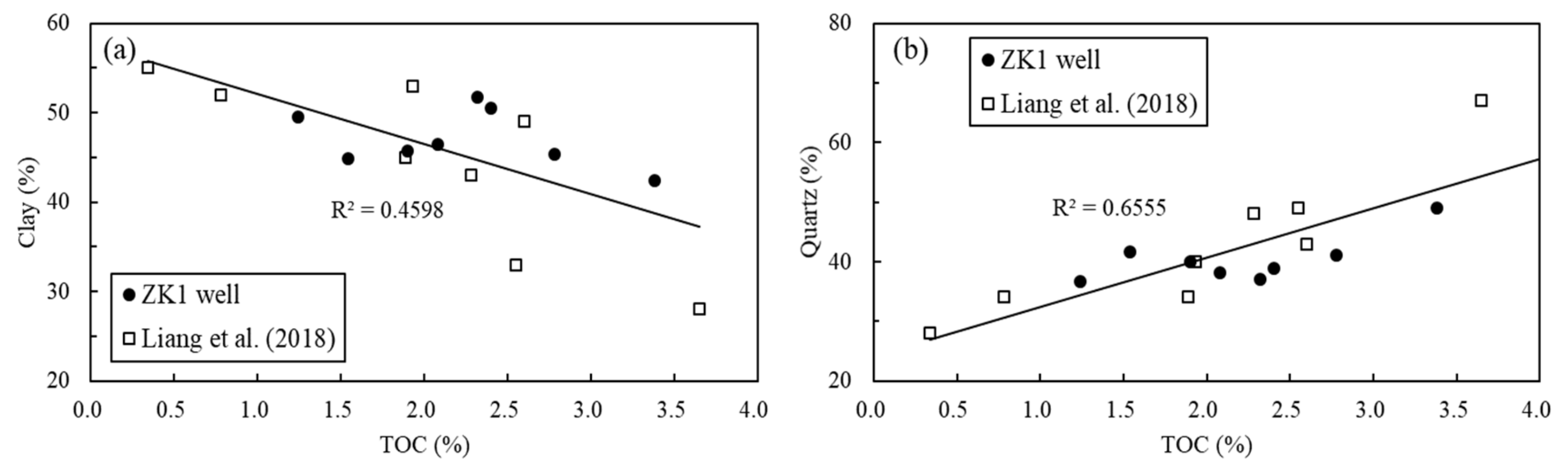
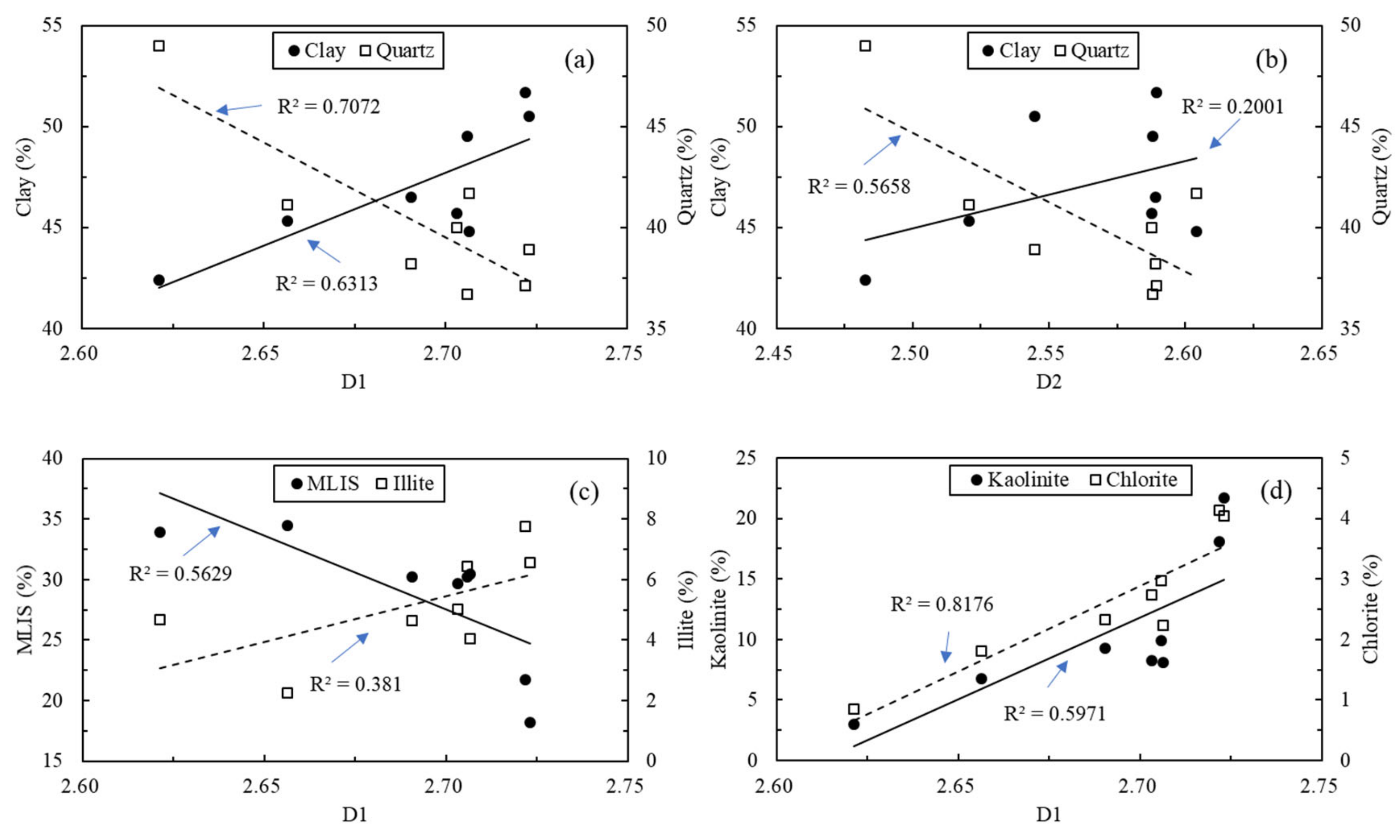
5.1. Relationships between Abundance of Organic Matter and Main Mineral Content
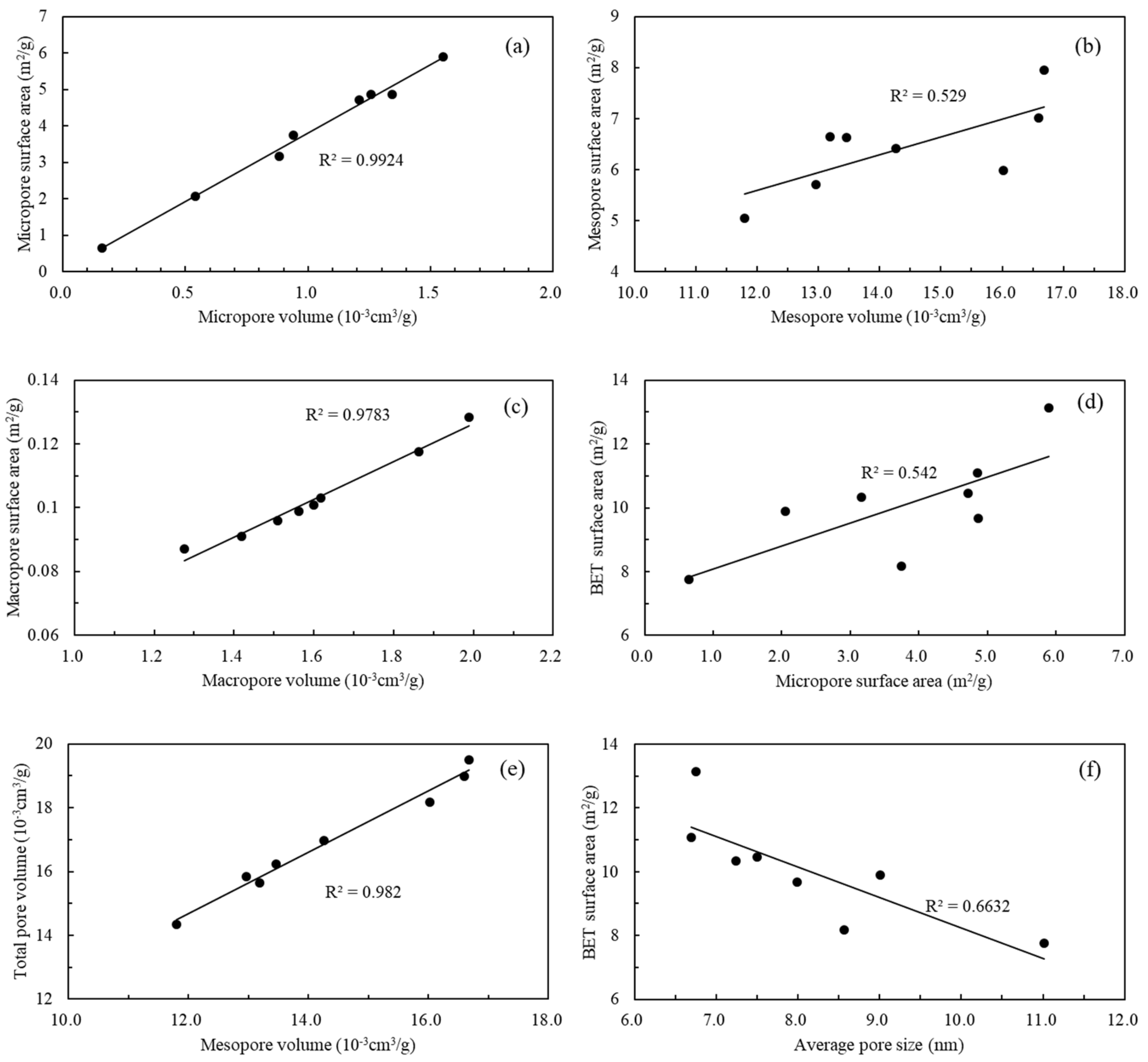
5.2. Relationships among Nanopore Structure Parameters
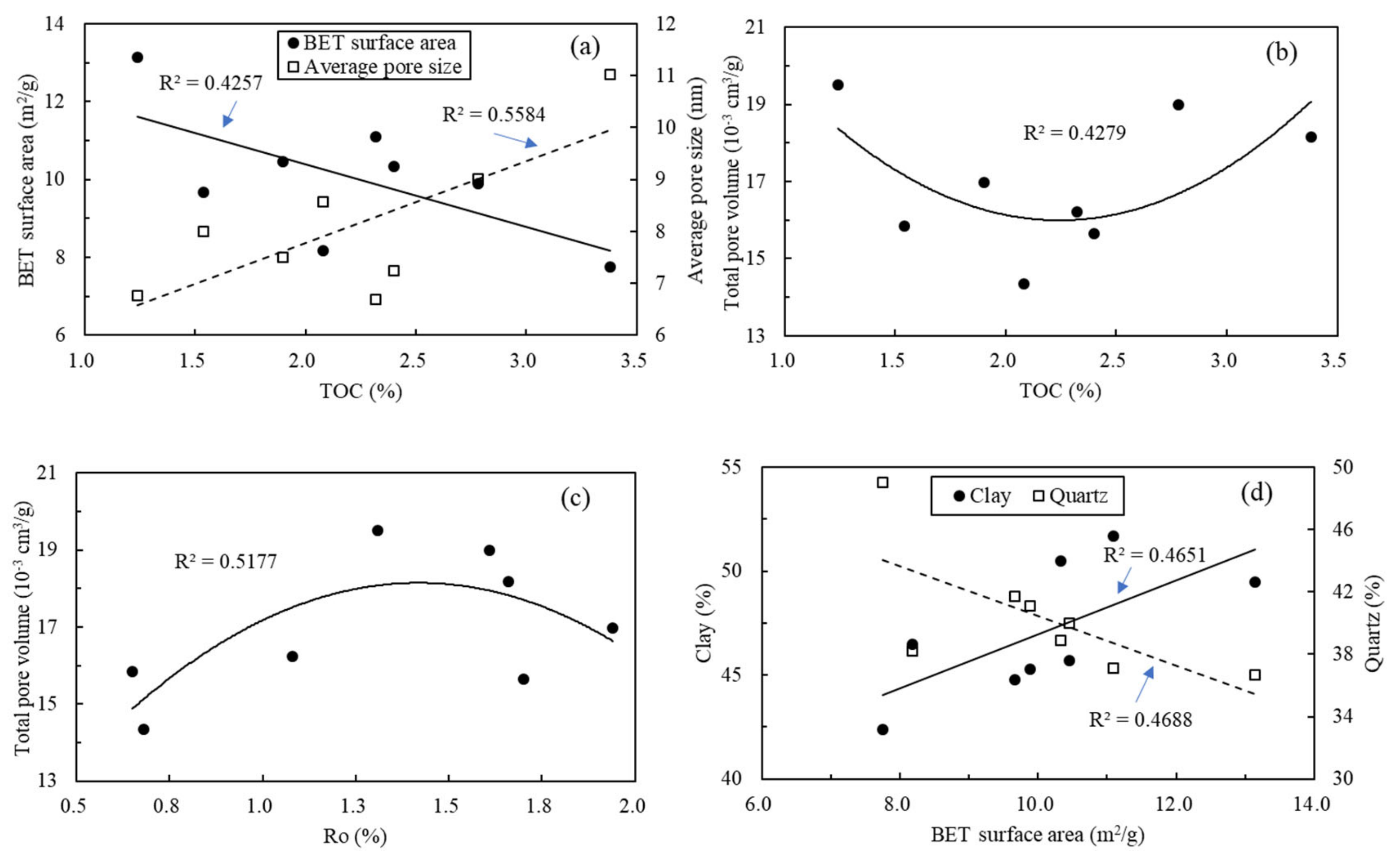
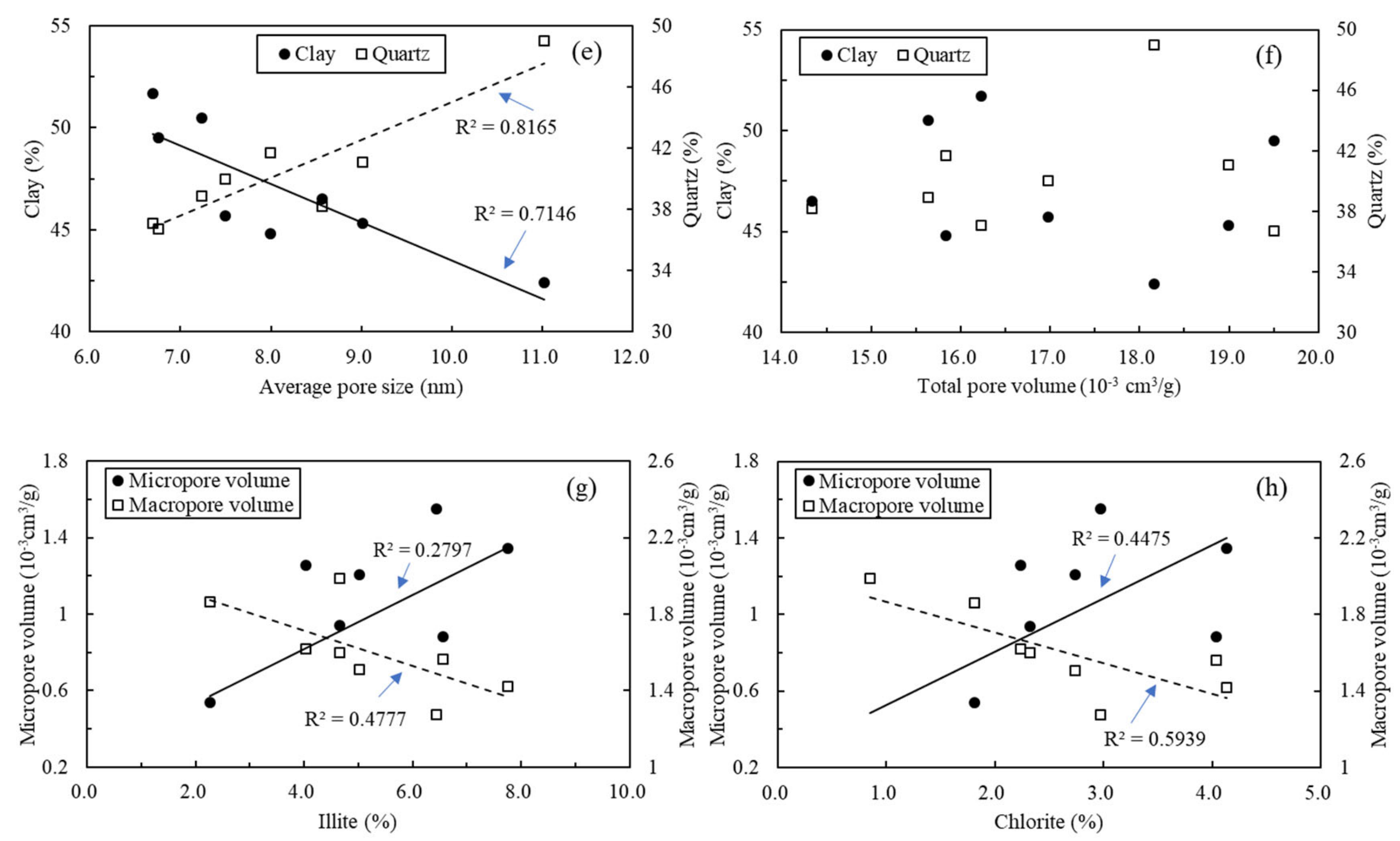
5.3. Relationships between Shale Compositions and Nanopore Structure Parameters
5.4. Relationships between Mineral Composition and Fractal Dimensions
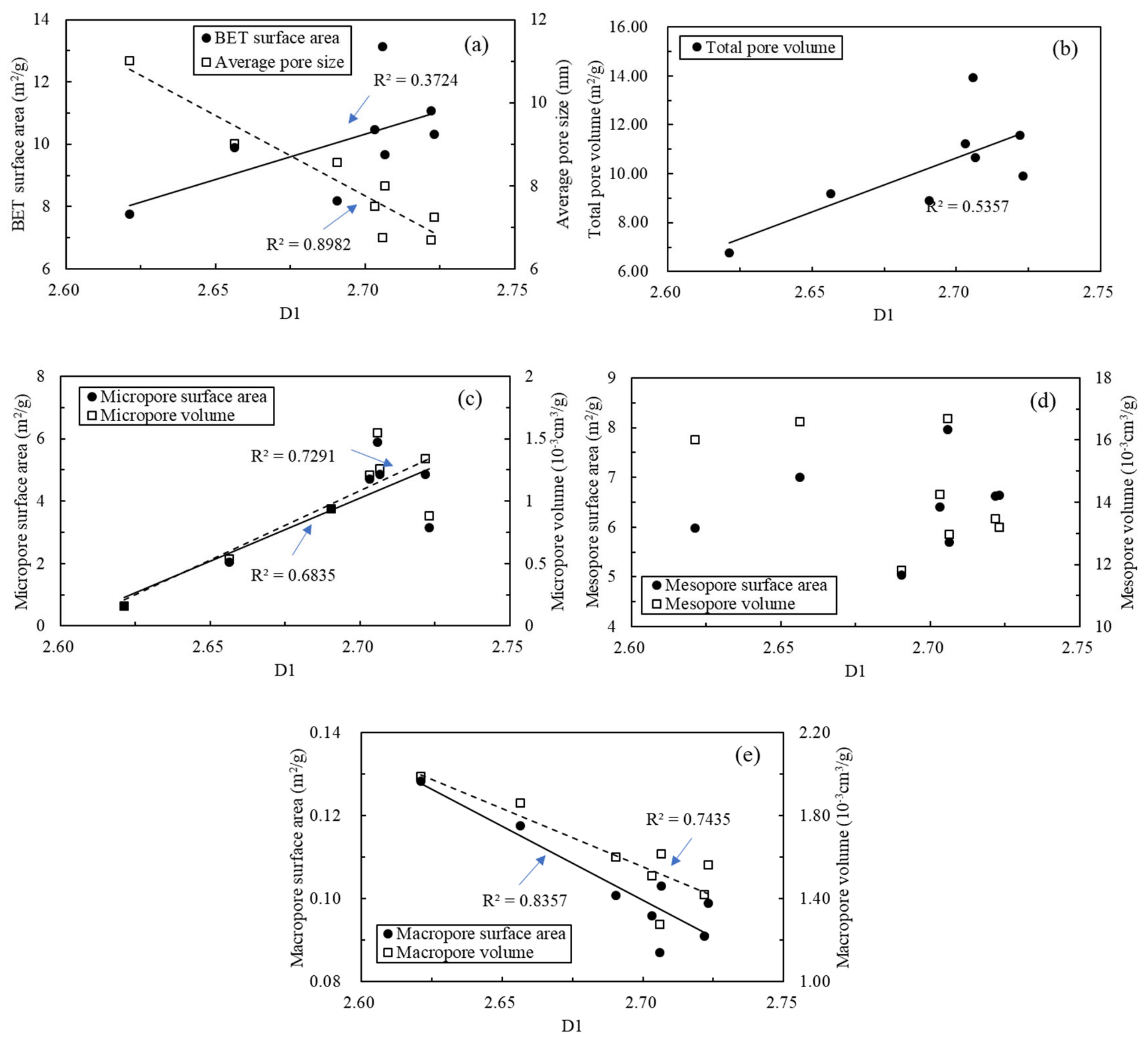
5.5. Relationships between Fractal Dimension and Nanopore Structure Parameters
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Curtis, J.B. Fractured shale-gas systems. AAPG Bull. 2002, 86, 1921–1938. [Google Scholar]
- Chen, S.; Zhu, Y.; Wang, H.; Liu, H.; Wei, W.; Fang, J. Shale gas reservoir characterization: A typical case in the southern Sichuan basin of China. Energy 2011, 36, 6609–6616. [Google Scholar] [CrossRef]
- Gasparik, M.; Bertier, P.; Gensterblum, Y.; Ghanizadeh, A.; Krooss, B.M.; Littke, R. Geological controls on the methane storage capacity in organic-rich shales. Int. J. Coal Geol. 2014, 123, 34–51. [Google Scholar] [CrossRef]
- Clarkson, C.R.; Bustin, R.M. The effect of pore structure and gas pressure upon the transport properties of coal: A laboratory and modeling study. 2. adsorption rate modeling. Fuel 1999, 78, 1345–1362. [Google Scholar] [CrossRef]
- Clarkson, C.R.; Bustin, R.M. Binary gas adsorption/desorption isotherms: Effect of moisture and coal composition upon carbon dioxide selectivity over methane. Int. J. Coal Geol. 2000, 42, 241–271. [Google Scholar] [CrossRef]
- Bustin, R.M.; Bustin, A.M.M.; Cui, A.; Ross, D.; Pathi, V.M. Impact of shale properties on pore structure and storage characteristics. In Proceedings of the SPE Paper 119892 Society of Petroleum Engineers Shale Gas Production Conference, Fort Worth, TX, USA, 16–18 November 2008. [Google Scholar]
- Ross, D.J.K.; Bustin, R.M. Characterizing the shale gas resource potential of Devonian–Mississippian strata in the Western Canada Sedimentary Basin: Application of an integrated formation evaluation. AAPG Bull. 2008, 92, 87–125. [Google Scholar] [CrossRef]
- Loucks, R.G.; Reed, R.M.; Ruppel, S.C.; Hammes, U. Spectrum of pore types and networks in mudrocks and a descriptive classification for matrixerelatedmudrock pores. AAPG Bull. 2012, 96, 1071–1098. [Google Scholar] [CrossRef]
- Loucks, R.G.; Reed, R.M.; Ruppel, S.C.; Jarvie, D.M. Morphology, genesis, and distribution of nanometerescale pores in siliceous mudstones of the Mississippian Barnett Shale. J. Sediment. Res. 2009, 79, 848–861. [Google Scholar] [CrossRef]
- Lapierre, C.; Leroueil, S.; Locat, J. Mercury intrusion and permeability of louiseville clay. Can. Geotech. J. 1990, 27, 761–773. [Google Scholar] [CrossRef]
- Yao, Y.; Liu, D. Comparison of low-field NMR and mercury intrusion porosimetry in characterizing pore size distributions of coals. Fuel 2012, 95, 152–158. [Google Scholar] [CrossRef]
- Branch, A. Methane and CO2, sorption and desorption measurements on dry argonnepremium coals: Pure components and mixtures. Int. J. Coal Geol. 2003, 55, 205–224. [Google Scholar]
- Chalmers, G.R.; Bustin, R.M.; Power, I.M. Characterization of gas shale pore systems by porosimetry, pycnometry, surface area, and field emission scanning electron microscopy/transmission electron microscopy image analyses: Examples from the Barnett, Woodford, Haynesville, Marcellus, and Doig units. AAPG Bull. 2012, 96, 1099–1119. [Google Scholar]
- Wang, G.; Ju, Y.; Yan, Z.; Li, Q. Pore structure characteristics of coal-bearing shale using fluid invasion methods: A case study in the Huainan-Huaibei coalfield in China. Mar. Pet. Geol. 2015, 62, 1–13. [Google Scholar] [CrossRef]
- Zhu, H.; Ju, Y.; Qi, Y.; Huang, C.; Zhang, L. Impact of tectonism on pore type and pore structure evolution in organic-rich shale: Implications for gas storage and migration pathways in naturally deformed rocks. Fuel 2018, 228, 272–289. [Google Scholar] [CrossRef]
- Lyu, Q.; Long Xi Ranjith, P.G.; Tan, J.; Kang, Y.; Wang, Z. Experimental investigation on the mechanical properties of a low-clay shale with different adsorption times in sub-/super-critical CO2. Energy 2018, 147, 1288–1298. [Google Scholar] [CrossRef]
- Clarkson, C.R.; Freeman, M.; He, L.; Agamalian, M.; Melnichenko, Y.B.; Mastalerz, M.; Bustin, R.M.; Radlinski, A.P.; Blach, T.P. Characterization of tight gas reservoir pore structure using USANS/SANS and gas adsorption analysis. Fuel 2012, 95, 371–385. [Google Scholar] [CrossRef]
- Ross, D.J.K.; Bustin, R.M. The importance of shale composition and pore structure upon gas storage potential of shale gas reservoirs. Mar. Pet. Geol. 2009, 26, 916–927. [Google Scholar] [CrossRef]
- Milliken, K.L.; Rudnicki, M.; Awwiller, D.N.; Zhang, T. Organic matter–hosted pore system, Marcellus formation (Devonian), Pennsylvania. AAPG Bull. 2013, 97, 177–200. [Google Scholar] [CrossRef]
- Ge, X.M.; Fan, Y.R.; Li, J.T.; Zahid, M.A. Pore structure characterization and classification using multifractal theory- an application in tight reservoir of Santanghu basin in Western China. J. Pet. Sci. Eng. 2015, 127, 297–304. [Google Scholar] [CrossRef]
- Pan, J.; Peng, C.; Wan, X.; Zheng, D.; Lv, R.; Wang, K. Pore structure characteristics of coal-bearing organic shale in Yuzhou coalfield, China using low pressure N2 adsorption and FE-SEM methods. J. Pet. Sci. Eng. 2017, 153, 234–243. [Google Scholar] [CrossRef]
- Ju, Y.; Huang, C.; Sun, Y.; Wan, Q.; Lu, X.; Lu, S.; He, H.; Wang, X.; Zou, C.; Wu, J.; et al. Nanogeosciences: Research History, Current Status, and Development Trends. J. Nanosci. Nanotechnol. 2017, 17, 5930–5965. [Google Scholar] [CrossRef]
- Schlueter, E.M.; Zimmerman, R.W.; Witherspoon, P.A.; Cook, N.G.W. The fractal dimension of pores in sedimentary rocks and its influence on permeability. Eng. Geol. 1997, 48, 199–215. [Google Scholar] [CrossRef]
- Qi, Y.; Ju, Y.; Huang, C.; Zhu, H.; Bao, Y.; Wu, J.; Meng, S.; Chen, W. Influences of organic matter and kaolinite on pore structures of transitional organic-rich mudstone with an emphasis on S2 controlling specific surface area. Fuel 2019, 237, 860–873. [Google Scholar] [CrossRef]
- Pérez Bernal, J.L.; Bello, M.A. Fractal geometry and mercury porosimetry: Comparison and application of proposed models on building stones. Appl. Surf. Sci. 2001, 185, 99–107. [Google Scholar] [CrossRef]
- Watt-Smith, M.J.; Edler, K.J.; Rigby, S.P. An experimental study of gas adsorption on fractal surfaces. Langmuir 2005, 21, 2281–2292. [Google Scholar] [CrossRef]
- Ahmad, A.L.; Idrus, N.F.; AbdShukor, S.R. Surface fractal dimension of Perovskite-doped alumina membrane: Influence of calcining temperature. J. Am. Ceram. Soc. 2006, 89, 1694–1698. [Google Scholar] [CrossRef]
- Khalili, N.R.; Pan, M.; Sandí, G. Determination of fractal dimensions of solid carbons from gas and liquid phase adsorption isotherms. Carbon 2000, 38, 573–588. [Google Scholar] [CrossRef]
- Fu, X.; Qin, Y.; Zhang, W.; Wei, Z.; Zhou, R. Pore fractal classification and natural classification based on the coal bed methane migration. Chin. Sci. Bull. 2005, 50, 51–55. [Google Scholar] [CrossRef]
- Zhang, S.; Tang, S.; Tang, D.; Huang, W.; Pan, Z. Determining fractal dimensions of coal pores by FHH model: Problems and effects. J. Nat. Gas Sci. Eng. 2014, 21, 929–939. [Google Scholar] [CrossRef]
- Bu, H.; Ju, Y.; Tan, J.; Wang, G.; Li, X. Fractal characteristics of pores in non-marine shales from the Huainan coalfield, eastern China. J. Nat. Gas Sci. Eng. 2015, 24, 166–177. [Google Scholar] [CrossRef]
- Yang, R.; He, S.; Yi, J.; Hu, Q. Nano-scale pore structure and fractal dimension of organic-rich Wufeng-Longmaxi shale from Jiaoshiba area, Sichuan Basin: Investigations using FE-SEM, gas adsorption and helium pycnometry. Mar. Pet. Geol. 2016, 70, 27–45. [Google Scholar] [CrossRef]
- Liu, J.; Yao, Y.; Liu, D.; Cai, Y.; Cai, J. Comparison of pore fractal characteristics between marine and continental shales. Fractals 2018, 26, 1840016. [Google Scholar] [CrossRef]
- Yu, K.; Shao, C.; Ju, Y.; Qu, Z. The genesis and controlling factors of micropore volume in transitional coal-bearing shale reservoirs under different sedimentary environments. Mar. Pet. Geol. 2019, 102, 426–438. [Google Scholar] [CrossRef]
- Yu, K.; Ju, Y.; Qian, J.; Qu, Z.; Shao, C.; Yu, K.; Shi, Y. Burial and thermal evolution of coal-bearing strata and its mechanisms in the southern North China Basin since the late Paleozoic. Int. J. Coal Geol. 20198, 198, 110–115. [Google Scholar] [CrossRef]
- Zaslow, J.; Orloff, T. Zircon U–Pb age and Lu–Hf isotope constraints on Precambrian evolution of continental crust in the Songshan area, the south-central North China Craton. Precambrian Res. 2013, 226, 1–20. [Google Scholar]
- Ju, Y.; Sun, Y.; Tan, J.; Bu, H.; Han, K.; Li, X. The composition, pore structure characterization and deformation mechanism of coal-bearing shales from tectonically altered coalfields in eastern China. Fuel 2018, 234, 626–642. [Google Scholar] [CrossRef]
- Determination of Carbon and Hydrogen in Coal; General Administration of Quality Supervision; Inspection and Quarantine of China: Beijing, China, 2008; No. GB/T4762008.
- Method of Determining Microscopically the Reflectance of Vitrinite in Sedimentary; National Energy Administration: Beijing, China, 1995; No. SY/T5124-1995.
- Analysis Method for Clay Minerals and Ordinary Ono-Clay Minerals in Sedimentary Rocks by the X-ray Diffraction; National Energy Administration: Beijing, China, 2010; No. SY/T5163-2010.
- Gregg, S.J.; Sing, K.S.W. Adsorption, Surface Area and Porosity, 2nd ed.; Academic Press: London, UK, 1982. [Google Scholar]
- Lastoskie, C.; Gubbins, K.E.; Quirke, N. Pore size distribution analysis of microporous carbons: A density functional theory approach. J. Phys. Chem. 1993, 97, 1012–1016. [Google Scholar] [CrossRef]
- Seaton, N.A.; Walton JP, R.B.; Quirke, N. A new analysis method for the determination of the pore size distribution of porous carbons from nitrogen adsorption measurements. Carbon 1989, 27, 853–861. [Google Scholar] [CrossRef]
- Rouquerol, J.; Avnir, D.; Fairbridge, C.W.; Everett, D.H.; Haynes, J.M.; Pernicone, N.; Ramsay, J.D.F.; Sing, K.S.W.; Unger, K.K. Recommendations for the characterization of porous solids. Int. Union Pure Appl. Chem. 1994, 66, 1739–1758. [Google Scholar] [CrossRef]
- Mandelbrot, B.B. The Fractal Geometry of Nature; Freeman: New York, NY, USA, 1983. [Google Scholar]
- Mahamud, M.M.; Novo, M.F. The use of fractal analysis in the textural characterization of coals. Fuel 2008, 87, 222–231. [Google Scholar] [CrossRef]
- Jaroniec, M. Evaluation of the fractal dimension from a single adsorption isotherm. Langmuir 1995, 11, 2316–2317. [Google Scholar] [CrossRef]
- Dong, T.; Harris, N.B.; Ayranci, K.; Twemlow, C.E.; Nassichuk, B.R. Porosity characteristics of the Devonian Horn River shale, Canada: Insights from lithofacies classification and shale composition. Int. J. Coal Geol. 2015, 141, 74–90. [Google Scholar] [CrossRef]
- Tian, H.; Pan, L.; Xiao, X.; Wilkins RW, T.; Meng, Z.; Huang, B. A preliminary study on the pore characterization of lower Silurian black shales in the Chuandong thrust fold belt, southwestern China using low pressure N2 adsorption and FE-SEM methods. Mar. Pet. Geol. 2013, 48, 8–19. [Google Scholar] [CrossRef]
- Liang, C.; Jiang, Z.; Zhang, C.; Guo, L.; Yang, Y.; Li, J. The shale characteristics and shale gas exploration prospects of the lower Silurian Longmaxi shale, Sichuan basin, south China. J. Nat. Gas Sci. Eng. 2014, 21, 636–648. [Google Scholar] [CrossRef]
- Xi, Z.; Tang, S.; Wang, J.; Yi, J.; Guo, Y.; Wang, K. Pore structure and fractal characteristics of Niutitang shale from China. Minerals 2018, 8, 163. [Google Scholar] [CrossRef]
- Yang, F.; Ning, Z.; Liu, H. Fractal characteristics of shales from a shale gas reservoir in the Sichuan basin, China. Fuel 2014, 115, 378–384. [Google Scholar] [CrossRef]
- Liang, Q.; Zhang, X.; Tian, J.; Sun, X.; Chang, H. Geological and geochemical characteristics of marine-continental transitional shale from the Lower Permian Taiyuan Formation, Taikang Uplift, southern North China Basin. Mar. Pet. Geol. 2018, 98, 229–242. [Google Scholar] [CrossRef]
- Wang, P.; Jiang, Z.; Ji, W.; Zhang, C.; Yuan, Y.; Chen, L. Heterogeneity of intergranular, intraparticle and organic pores in Longmaxi shale in Sichuan basin, south China: Evidence from SEM digital images and fractal and multifractal geometries. Mar. Pet. Geol. 2016, 72, 122–138. [Google Scholar] [CrossRef]
- Chen, S.; Han, Y.; Fu, C.; Zhang, H.; Zhu, Y.; Zuo, Z. Micro and nano-size pores of clay minerals in shale reservoirs: Implication for the accumulation of shale gas. Sediment. Geol. 2016, 342, 180–190. [Google Scholar] [CrossRef]
- Malekani, K. Comparison of techniques for determining the fractal dimensions of clay minerals. Clays Clay Miner. 1996, 44, 677–685. [Google Scholar] [CrossRef]
- Yao, Y.; Liu, D.; Tang, D.; Tang, S.; Huang, W. Fractal characterization of adsorption-pores of coals from North China: An investigation on CH4 adsorption capacity of coals. Int. J. Coal Geol. 2008, 73, 27–42. [Google Scholar] [CrossRef]


| Sample | Depth (m) | TOC (%) | Ro (%) | Kerogen Type |
|---|---|---|---|---|
| TY8 | 1013.1 | 2.18 | 0.71 | III |
| TY7 | 1023.3 | 1.56 | 0.65 | III |
| TY6 | 1034.5 | 1.91 | 1.94 | III |
| TY5 | 1045.7 | 3.38 | 1.66 | III |
| TY4 | 1076.5 | 2.40 | 1.7 | III |
| TY3 | 1083.2 | 2.32 | 1.08 | III |
| TY2 | 1107.3 | 2.78 | 1.61 | III |
| TY1 | 1113.7 | 1.24 | 1.31 | III |
| Sample | Quartz | K-Feldspar | Plagioclase | Calcite | Dolomite | Siderite | Pyrite | Clay | MLIS | Illite | Kaolinite | Chlorite |
|---|---|---|---|---|---|---|---|---|---|---|---|---|
| TY8 | 39.3 | 1.1 | 3.5 | 0 | 0 | 8.2 | 1.5 | 45.5 | 31.7 | 4.4 | 8.9 | 2.5 |
| TY7 | 41.7 | 4.4 | 3.8 | 0 | 0 | 3.3 | 2.1 | 44.7 | 32.5 | 3.0 | 9.1 | 2.4 |
| TY6 | 40.6 | 1.6 | 3.2 | 0 | 0 | 7.5 | 1.5 | 45.6 | 28.5 | 3.8 | 8.6 | 2.3 |
| TY5 | 49 | 0 | 0 | 0 | 0 | 0 | 8.6 | 42.4 | 33.9 | 4.7 | 3.0 | 0.8 |
| TY4 | 31.9 | 0.9 | 1.1 | 0 | 0 | 4.5 | 4.1 | 57.5 | 20.7 | 7.5 | 24.7 | 4.6 |
| TY2 | 37.1 | 1.6 | 0 | 0 | 0 | 8.6 | 1 | 51.7 | 21.7 | 7.8 | 18.1 | 4.1 |
| TY2 | 41.1 | 1.2 | 3.7 | 0 | 1.3 | 0 | 7.4 | 45.3 | 34.4 | 2.3 | 6.8 | 1.8 |
| TY1 | 36.7 | 0 | 0 | 3.7 | 2.9 | 0 | 7.2 | 49.5 | 30.2 | 6.4 | 9.9 | 3.0 |
| Sample | SA (m2/g) | APS (nm) | PV (10−3 cm3/g) | V1 (10−3 cm3/g) | V2 (10−3 cm3/g) | V3 (10−3 cm3/g) |
|---|---|---|---|---|---|---|
| TY1 | 13.138 | 6.754 | 19.508 | 1.551 | 16.681 | 1.276 |
| TY2 | 9.891 | 9.012 | 18.994 | 0.539 | 16.592 | 1.863 |
| TY3 | 11.086 | 6.696 | 16.225 | 1.344 | 13.461 | 1.420 |
| TY4 | 10.331 | 7.239 | 15.638 | 0.883 | 13.193 | 1.562 |
| TY5 | 7.752 | 11.014 | 18.166 | 0.161 | 16.017 | 1.988 |
| TY6 | 10.390 | 7.372 | 16.723 | 1.212 | 14.271 | 1.512 |
| TY7 | 9.728 | 7.883 | 15.901 | 1.266 | 12.972 | 1.609 |
| TY8 | 8.263 | 8.497 | 14.298 | 0.952 | 11.806 | 1.603 |
| Sample | Equation (1) | R2 | K1 | D1 | Equation (2) | R2 | K2 | D2 |
|---|---|---|---|---|---|---|---|---|
| TY1 | y = −0.2941x + 1.5134 | 0.9999 | −0.2941 | 2.7059 | y = −0.4119x + 1.5149 | 0.9987 | −0.4119 | 2.5881 |
| TY2 | y = −0.3436x + 1.2421 | 0.9999 | −0.3436 | 2.6564 | y = −0.4793x + 1.2333 | 0.9996 | −0.4793 | 2.5207 |
| TY3 | y = −0.2781x + 1.3496 | 0.9999 | −0.2781 | 2.7219 | y = −0.4107x + 1.3456 | 0.9981 | −0.4107 | 2.5893 |
| TY4 | y = −0.2769x + 1.2954 | 0.9987 | −0.2769 | 2.7231 | y = −0.4552x + 1.2775 | 0.9988 | −0.4552 | 2.5448 |
| TY5 | y = −0.3788x + 1.0028 | 0.9998 | −0.3788 | 2.6212 | y = −0.5175x + 0.9936 | 0.9998 | −0.5175 | 2.4825 |
| TY6 | y = −0.2968x + 1.2963 | 0.9999 | −0.2968 | 2.7032 | y = −0.4124x + 1.2883 | 0.9995 | −0.4124 | 2.5876 |
| TY7 | y = −0.2935x + 1.2129 | 0.9998 | −0.2935 | 2.7065 | y = −0.3958x + 1.2069 | 0.9995 | −0.3958 | 2.6042 |
| TY8 | y = −0.3095x + 1.0558 | 0.9998 | −0.3095 | 2.6905 | y = −0.4111x + 1.0416 | 0.9999 | −0.4111 | 2.5889 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Yu, K.; Ju, Y.; Qi, Y.; Qiao, P.; Huang, C.; Zhu, H.; Feng, H. Fractal Characteristics and Heterogeneity of the Nanopore Structure of Marine Shale in Southern North China. Minerals 2019, 9, 242. https://doi.org/10.3390/min9040242
Yu K, Ju Y, Qi Y, Qiao P, Huang C, Zhu H, Feng H. Fractal Characteristics and Heterogeneity of the Nanopore Structure of Marine Shale in Southern North China. Minerals. 2019; 9(4):242. https://doi.org/10.3390/min9040242
Chicago/Turabian StyleYu, Kun, Yiwen Ju, Yu Qi, Peng Qiao, Cheng Huang, Hongjian Zhu, and Hongye Feng. 2019. "Fractal Characteristics and Heterogeneity of the Nanopore Structure of Marine Shale in Southern North China" Minerals 9, no. 4: 242. https://doi.org/10.3390/min9040242
APA StyleYu, K., Ju, Y., Qi, Y., Qiao, P., Huang, C., Zhu, H., & Feng, H. (2019). Fractal Characteristics and Heterogeneity of the Nanopore Structure of Marine Shale in Southern North China. Minerals, 9(4), 242. https://doi.org/10.3390/min9040242





