An Experimental Study for the Remediation of Industrial Waste Water Using a Combination of Low Cost Mineral Raw Materials
Abstract
1. Introduction
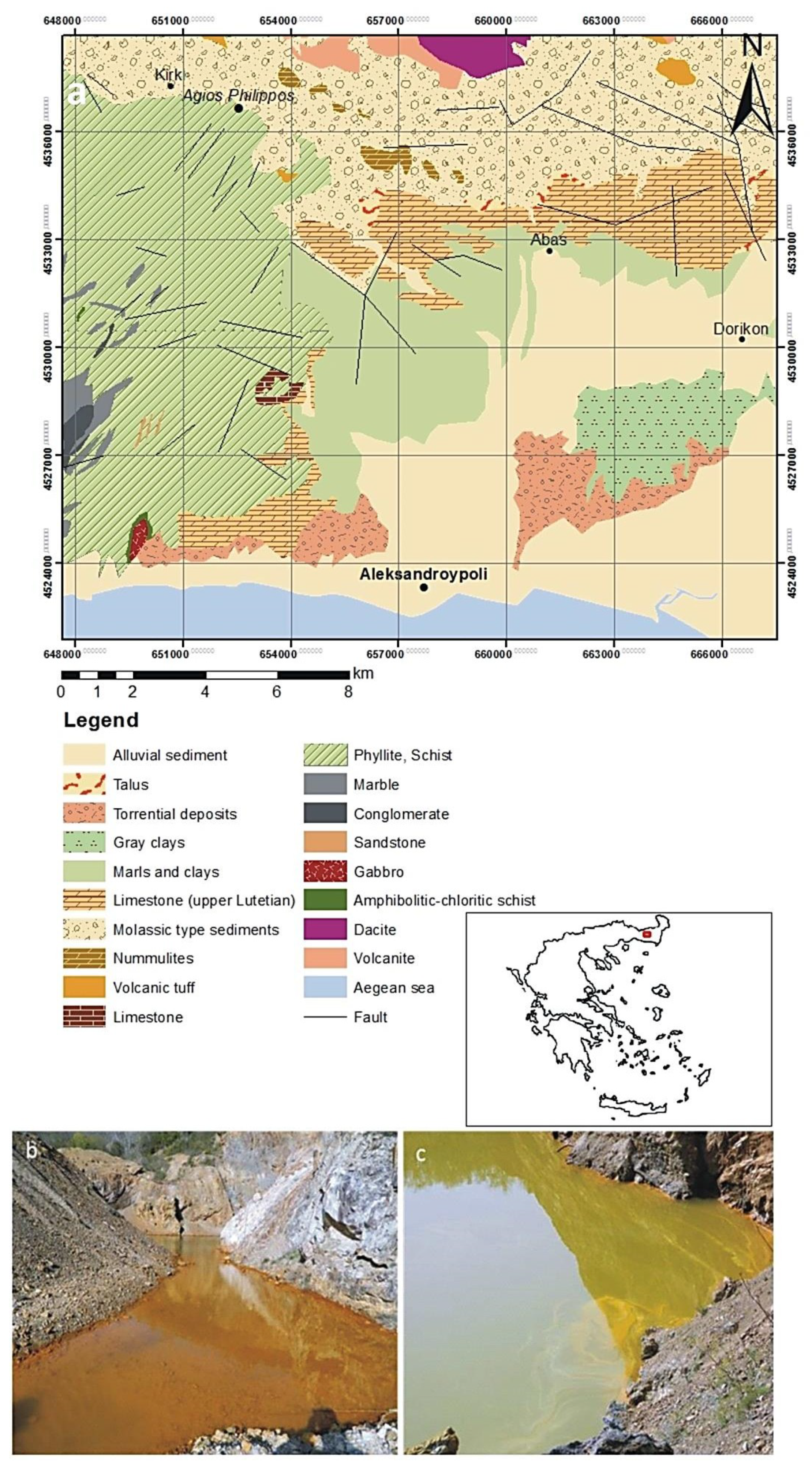
2. Geologic Description
3. Materials and Methods
3.1. Materials
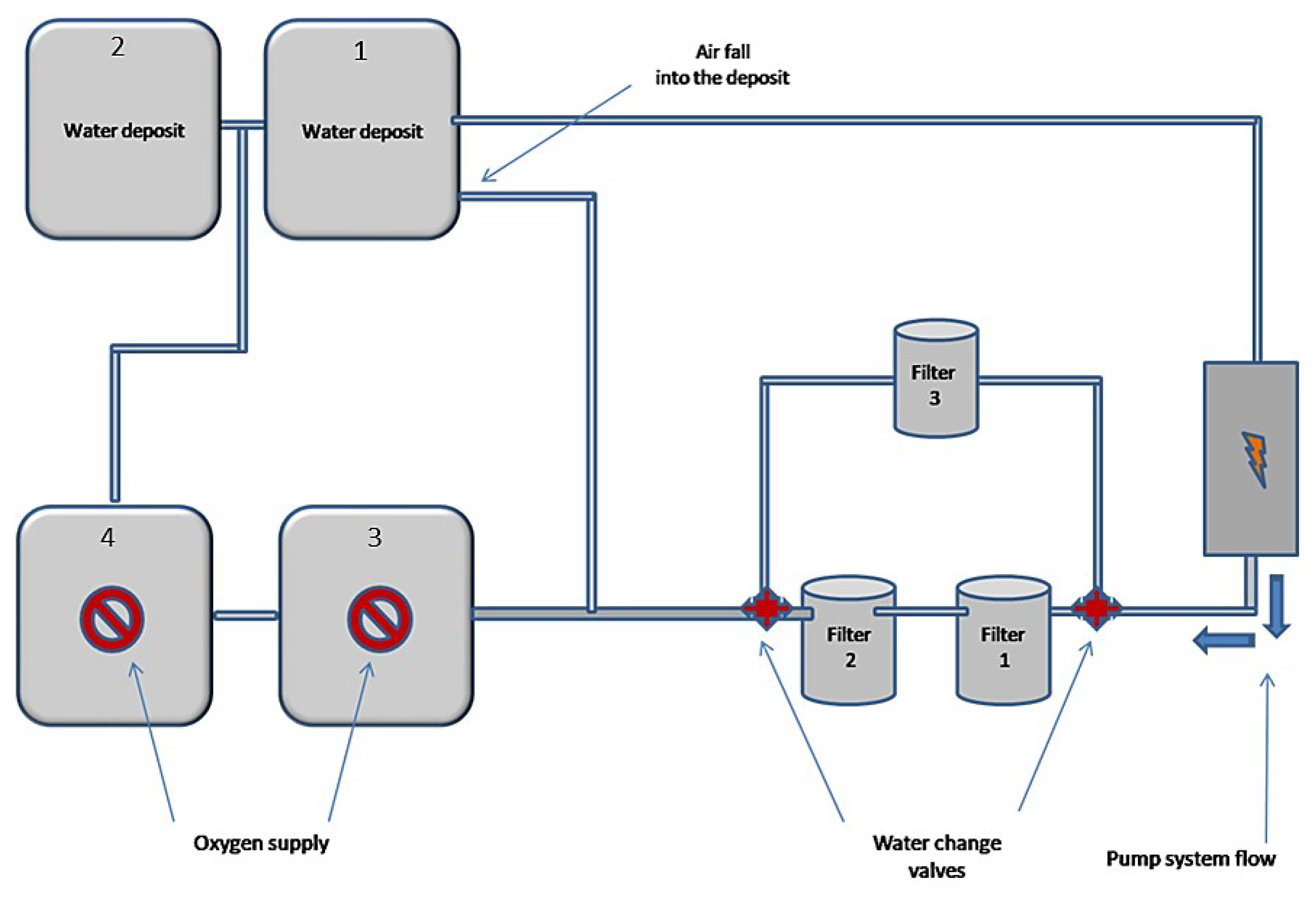
3.2. Methods
4. Results
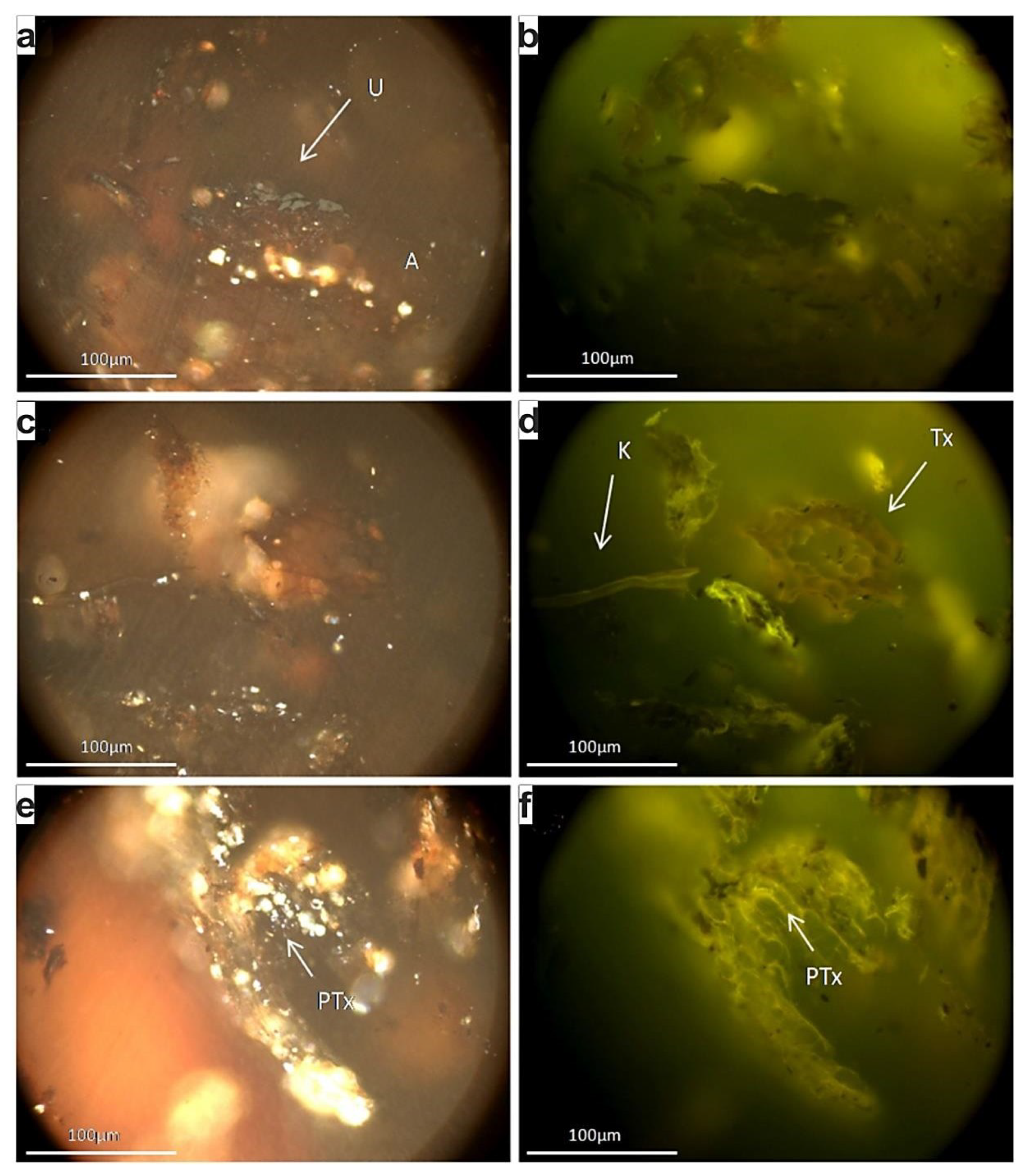
4.1. Petrographic Features of Rock Materials
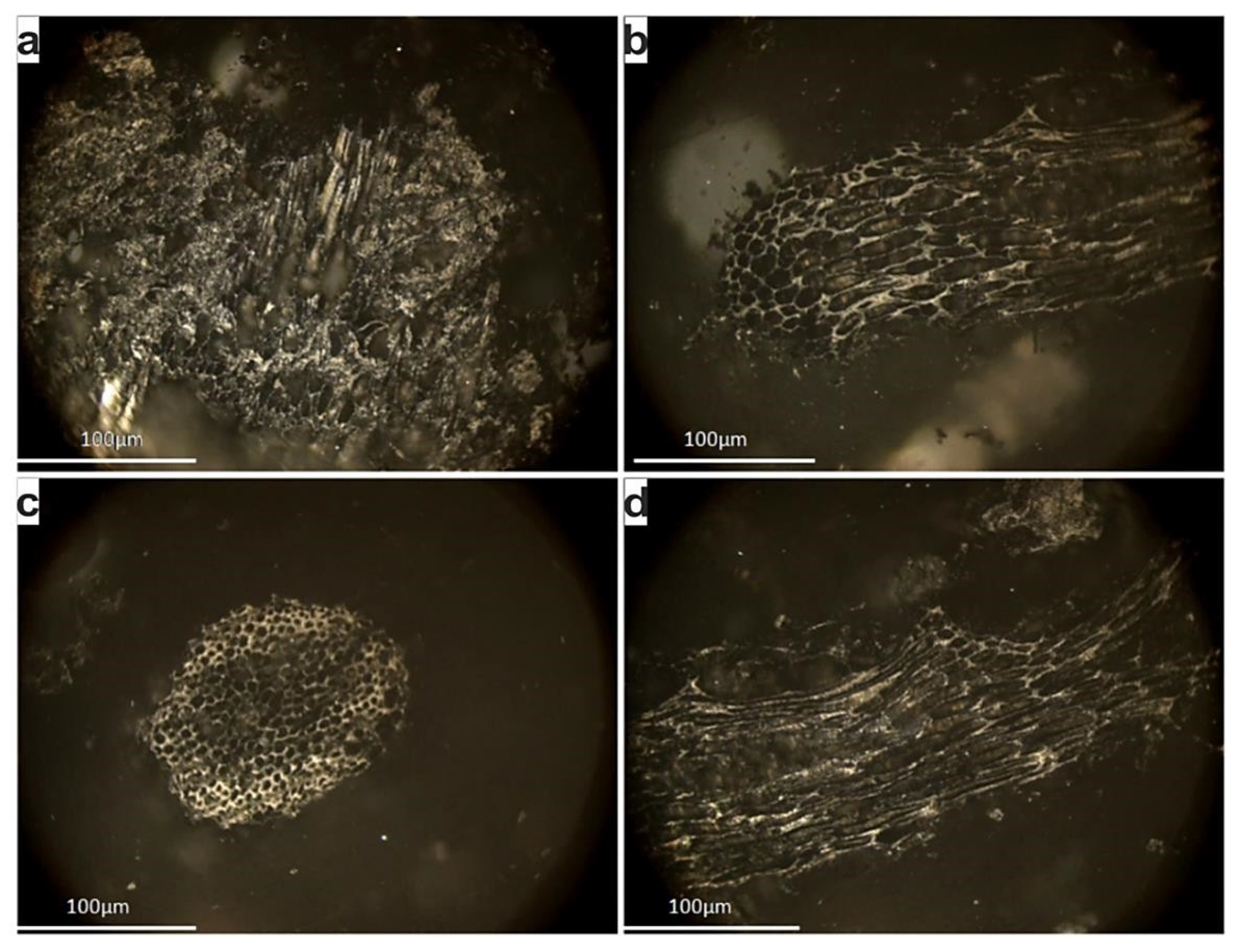
4.2. Petrographic Features of Peat and Biochar
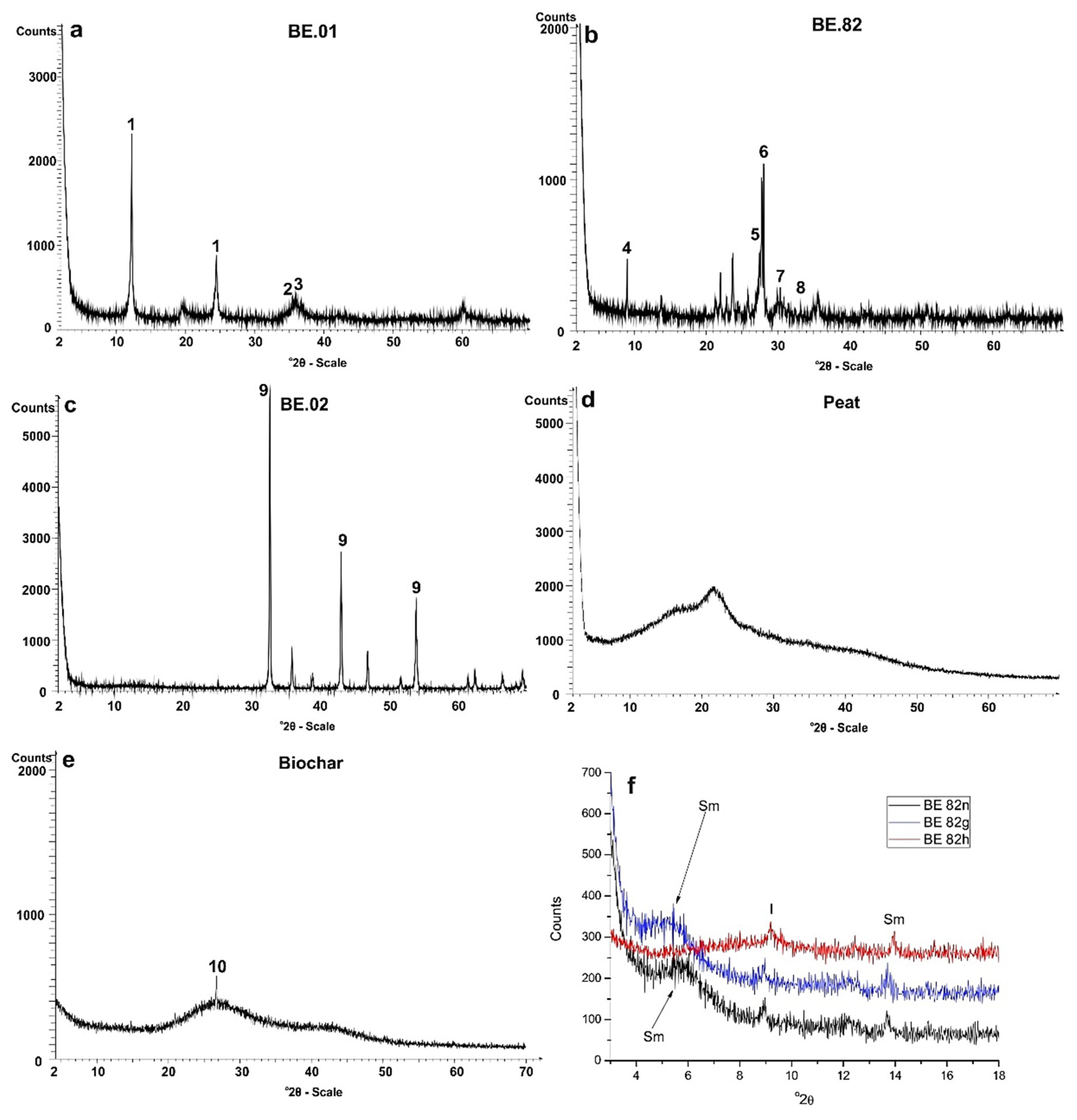
4.3. X-ray Diffractometry of Raw Materials
4.4. Total Porosity of Rock Materials
4.5. Whole-Rock Geochemical Analysis of Rock Materials
4.6. Chemical Composition of the Sludge
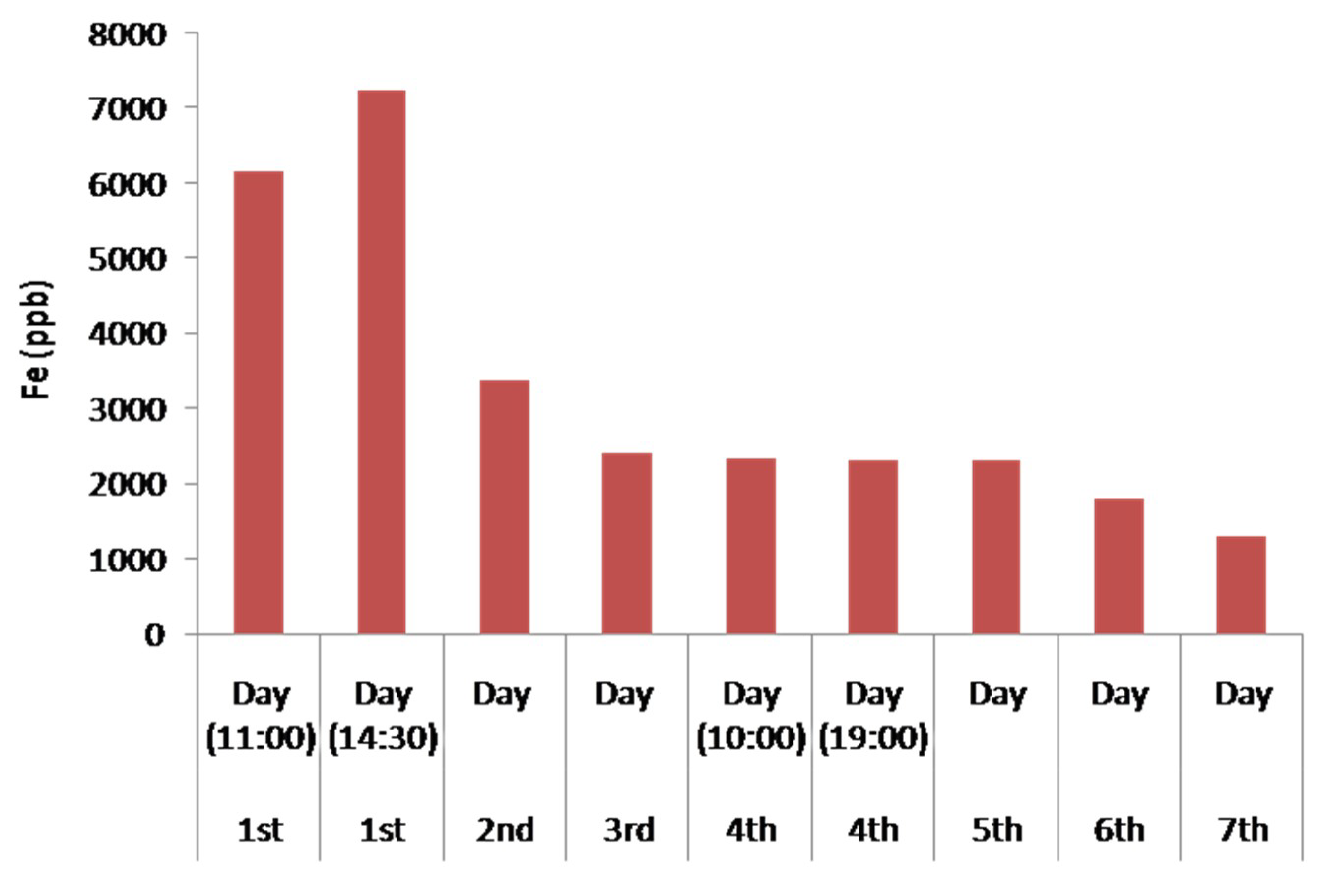
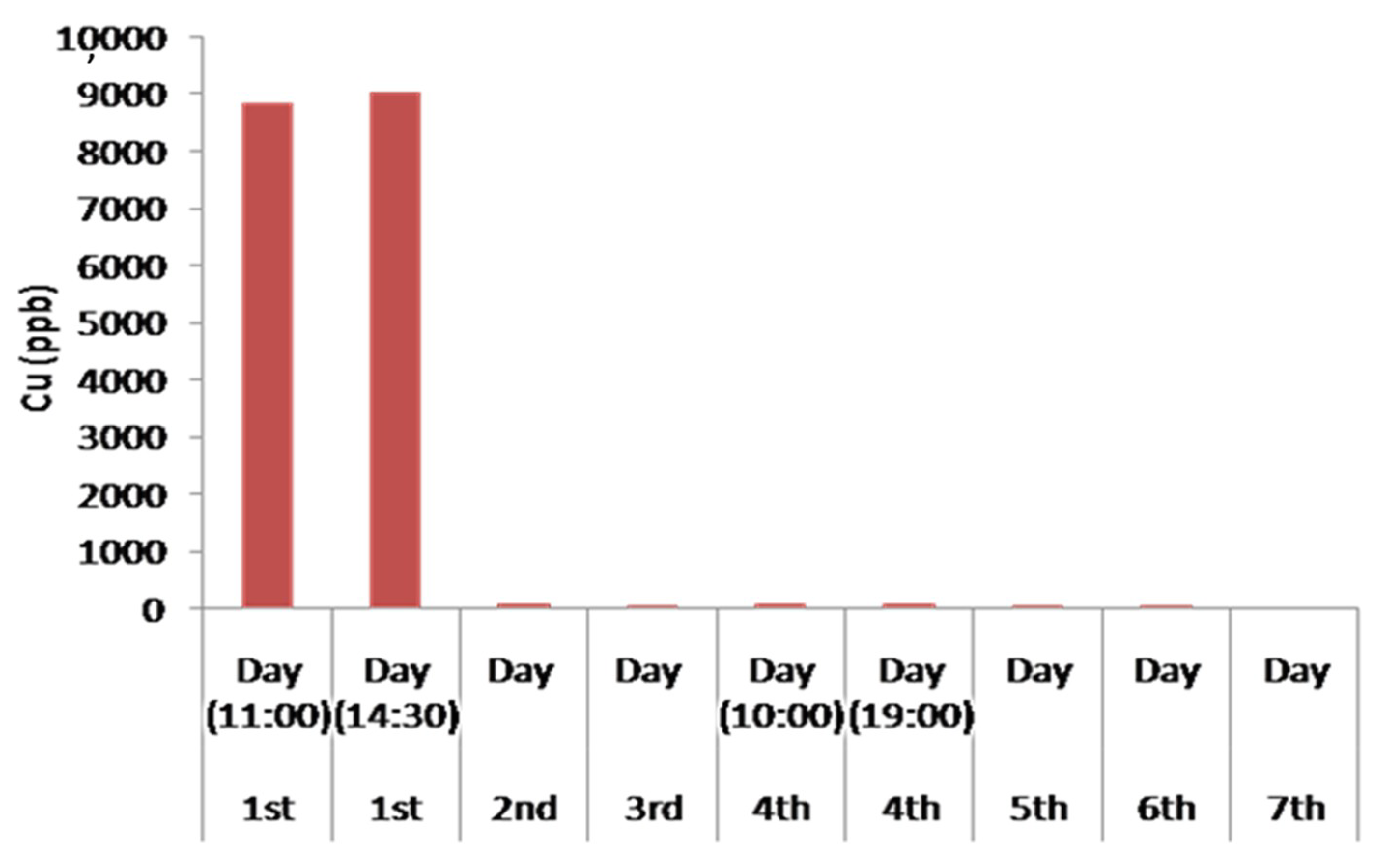
4.7. Results of the Remediation of the Industrial Waste Water during the 7-Day Operation
5. Discussion
5.1. Remediation of pH Values Using the Proposed Experimental Continuous Flow Device
5.2. Removal of Heavy Metals Using the Proposed Experimental Continuous Flow Device
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Weber, W.J.; McGinley, P.M.; Katz, L.E. Sorption phenomena in subsurface systems: Concepts, models and effects on contaminant fate and transport. Water Res. 1991, 25, 499–528. [Google Scholar] [CrossRef]
- Salmon, S.U.; Oldham, C.; Ivey, G.N. Assessing internal and external controls on lake water quality: Limitations on organic carbon-driven alkalinity generation in acidic pit lakes. Water Resour. Res. 2008, 44, W10414. [Google Scholar] [CrossRef]
- Schindler, D.W. The significance of in-lake production of alkalinity. Water Air Soil Pollut. 1986, 30, 931–944. [Google Scholar] [CrossRef]
- Geller, W.; Klapper, H.; Salomons, W. Acidic Mining Lakes; Springer: Berlin/Heidelberg, Germany, 1998. [Google Scholar]
- Geller, W.; Koschorreck, M.; Wendt-Potthoff, K.; Bozau, E.; Herzsprung, P.; Büttner, O.; Schultze, M. A pilot-scale field experiment for the microbial neutralization of a holomictic acidic pit lake. J. Geochem. Explor. 2009, 100, 153–159. [Google Scholar] [CrossRef]
- Plumlee, G.S. The environmental geology of mineral deposits. In The Environmental Geochemistry of Mineral Deposits; Society of Economic Geologists (SEG): Littleton, CO, USA, 1999; pp. 71–116. [Google Scholar]
- Lattanzi, P.; Da Pelo, S.; Musu, E.; Atzei, D.; Elsener, B.; Fantauzzi, M.; Rossi, A. Enargite oxidation: A review. Earth Sci. Rev. 2008, 86, 62–88. [Google Scholar] [CrossRef]
- Sperling, E.; Grandschamp, C.A.P. Possible water uses in mining lakes: Case study of Agua Claras, Brazil. In Access to Sanitation and Safe Water-Global Partnerships and Local Actions: Proceedings of the 33rd WEDC International Conference, Accra, Ghana, 7–11 April 2008; WEDC, Loughborough University: Loughborough, UK, 2008; pp. 375–380. [Google Scholar]
- Shevenell, L.; Connors, K.A.; Henry, C.D. Controls on pit lake water quality at sixteen open-pit mines in Nevada. Appl. Geochem. 1999, 14, 669–687. [Google Scholar] [CrossRef]
- Argun, M.E.; Dursun, S. A new approach to modification of natural adsorbent for heavy metal adsorption. Bioresour. Technol. 2008, 99, 2516–2527. [Google Scholar] [CrossRef]
- Meena, A.K.; Kadirvelu, K.; Mishra, G.K.; Rajagopal, C.; Nagar, P.N. Adsorptive removal of heavy metals from aqueous solution by treated sawdust (Acacia arabica). J. Hazard. Mater. 2008, 150, 604–611. [Google Scholar] [CrossRef] [PubMed]
- Panayotova, M.; Velikov, B. Influence of zeolite transformation in a homoionic form on the removal of some heavy metal ions from wastewater. J. Environ. Sci. Health. 2003, 38, 545–554. [Google Scholar] [CrossRef]
- Amarasinghe, B.M.W.P.K.; Williams, R.A. Tea waste as a low cost adsorbent for the removal of Cu and Pb from wastewater. Chem. Eng. J. 2007, 132, 299–309. [Google Scholar] [CrossRef]
- Barkat, M.A. New trends in removing heavy metals from industrial wastewater. Arab. J. Chem. 2011, 4, 361–377. [Google Scholar] [CrossRef]
- Bai, Y.; Bartkiewicz, B. Removal of cadmium from wastewater using ion exchange resin Amberjet 1200H columns. Pol. J. Environ. Stud. 2009, 18, 1191–1195. [Google Scholar]
- Hegazi, H.A. Removal of heavy metals from wastewater using agricultural and industrial wastes as adsorbents. Hbrc J. 2013, 9, 276–282. [Google Scholar] [CrossRef]
- Lakherwal, D. Adsorption of heavy metals, a review. Int. J. Environ. Res. Dev. 2014, 4, 41–48. [Google Scholar]
- Teir, S.; Eloneva, S.; Fogelholm, C.; Zevenhoven, R. Stability of calcium carbonate and magnesium carbonate in rainwater and nitric acid solutions. Energy Convers. Manag. 2006, 47, 3059–3068. [Google Scholar] [CrossRef]
- Smith, M. Panasqueira the tungsten giant at 100+. Oper. Focus Int. Min. 2006, 33, 10–14. [Google Scholar]
- Petrounias, P.; Giannakopoulou, P.P.; Rogkala, A.; Stamatis, P.M.; Tsikouras, B.; Papoulis, D.; Lampropoulou, P.; Hatzipanagiotou, K. The influence of alteration of aggregates on the quality of the concrete: A case study from serpentinites and andesites from central macedonia (North Greece). Geosciences 2018, 8, 115. [Google Scholar] [CrossRef]
- Giannakopoulou, P.P.; Petrounias, P.; Rogkala, A.; Tsikouras, B.; Stamatis, P.M.; Pomonis, P.; Hatzipanagiotou, K. The influence of the mineralogical composition of ultramafic rocks on their engineering performance: A case study from the veria-naousa and gerania ophiolite complexes (Greece). Geosciences 2018, 8, 251. [Google Scholar] [CrossRef]
- Petrounias, P.; Giannakopoulou, P.P.; Rogkala, A.; Lampropoulou, P.; Koutsopoulou, E.; Papoulis, D.; Tsikouras, B.; Hatzipanagiotou, K. The Impact of Secondary Phyllosilicate Minerals on the Engineering Properties of Various Igneous Aggregates from Greece. Minerals 2018, 8, 329. [Google Scholar] [CrossRef]
- Petrounias, P.; Giannakopoulou, P.P.; Rogkala, A.; Stamatis, P.M.; Lampropoulou, P.; Tsikouras, B.; Hatzipanagiotou, K. The Effect of petrographic characteristics and physico-mechanical properties of aggregates on the quality of concrete. Minerals 2018, 8, 577. [Google Scholar] [CrossRef]
- Giannakopoulou, P.P.; Petrounias, P.; Tsikouras, B.; Kalaitzidis, S.; Rogkala, A.; Hatzipanagiotou, K.; Tombros, S. Using factor analysis to determine the interrelationships between the engineering properties of aggregates from igneous rocks in Greece. Minerals 2018, 8, 580. [Google Scholar] [CrossRef]
- Coupal, B.; Lalancette, J.M. The treatment of waste waters with peat moss. War. Res. 1976, 10, 1071–1076. [Google Scholar] [CrossRef]
- McLellan, J.K.; Rock, C.A. Pretreating landfill leachate with peat to remove metals. Water Air Soil Pollut. 1988, 37, 203–215. [Google Scholar] [CrossRef]
- Chaney, R.L.; Hundemann, P.T. Use of peat moss columns to remove cadmium from wastewaters. J. Water Pollut. Control Fed. 1979, 51, 17–21. [Google Scholar]
- Sharma, D.C.; Forster, C.F. Removal of hexavalent chromium using sphagnum moss peat. Water Res. 1993, 27, 1201–1208. [Google Scholar] [CrossRef]
- Gardea-Torresdey, J.L.; Tang, L.; Salvador, J.M. Copper adsorption by esterified and unesterified fractions of sphagnum peat moss and its different humic substances. J. Hazard. Mater. 1996, 48, 191–206. [Google Scholar] [CrossRef]
- Zhipei, Z.; Junlu, Y.; Zenghui, W.; Piya, C. A preliminary study of the removal of Pb(2+), Cd(2+), Zn(2+), Ni(2+), and Cr(2+) from wastewaters with several Chinese peats. In Proceedings of the Seventh International Peat Congress, Dublin, Ireland, 18–23 June 1984; pp. 147–152. [Google Scholar]
- Dennehy, C.; Lawlor, P.G.; Jiang, Y.; Gardiner, G.E.; Xie, S.; Nghiem, L.D.; Zhan, X. Green-house gas emissions from different pig manure management techniques: A critical analysis. Front. Environ. Sci. Eng. 2017, 11, 11. [Google Scholar] [CrossRef]
- Feng, Z.; Zhu, L. Sorption of phenanthrene to biochar modified by base. Front. Environ. Sci. Eng. 2017, 12, 1. [Google Scholar] [CrossRef]
- Li, Z.; Wang, F.; Bai, T.; Tao, J.; Guo, J.; Yang, M.; Wang, S.; Hu, S. Lead immobilization by geological fluorapatite and fungus Aspergillus niger. J. Hazard. Mater. 2016, 320, 386–392. [Google Scholar] [CrossRef]
- Li, J.-S.; Beiyuan, J.; Tsang, D.C.; Wang, L.; Poon, C.S.; Li, X.-D.; Fendorf, S. Arsenic-containing soil from geogenic source in Hong Kong: Leaching characteristics and stabilization/solidification. Chemosphere 2017, 182, 31–39. [Google Scholar] [CrossRef]
- Shen, Z.; Zhang, Y.; Jin, F.; McMillan, O.; Al-Tabbaa, A. Qualitative and quantitative characterisation of adsorption mechanisms of lead on four biochars. Sci. Total Environ. 2017, 609, 1401–1410. [Google Scholar] [CrossRef]
- Lee, S.J.; Park, J.H.; Ahn, Y.T.; Chung, J.W. Comparison of heavy metal adsorption by peat moss and peat moss-derived biochar produced under different carbonization conditions. Water Soil Air Pollut. 2015, 226, 9. [Google Scholar] [CrossRef]
- Skarpelis, N. The Agios Philippos ore deposit, Kirki (Western Thrace). A base metal part of a high sulfidation epithermal system. Bull. Geol. Soc. Gr. 1999, 33, 51–60. [Google Scholar]
- Triantafyllidis, S. Environmental risk assessment of mining and processing activities and rehabilitation proposals in evros and rhodope prefectures (Thrace, NE Greece). Ph.D. Thesis, Faculty of Geology & Geoenvironment, University of Athens, Athens, Greece, 2006; 361p. (In Greek with English abstract). [Google Scholar]
- Triantafyllidis, S.; Skarpelis, N. Mineral formation in an acid pit lake from a high-sulfidation ore deposit: Kirki, NE Greece. J. Geoch. Explor. 2006, 88, 68–71. [Google Scholar] [CrossRef]
- Maratos, G.; Andronopoulos, V.; Koukouzas, K. Geological Map of Greece. Alexandroupolis Sheet. 1:50.000; IGME (Institute of Geology and Mineral Exploration): Athens, Greece, 1965. [Google Scholar]
- Petrounias, P.; Rogkala, A.; Kalpogiannaki, M.; Tsikouras, B.; Hatzipanagiotou, K. Comparative study of physico-mechanical properties of ultrabasic rocks (Veria-Naousa ophiolite) and andesites from central Macedonia (Greece). Bull. Geol. Soc. Gr. 2016, 50, 1989–1998. [Google Scholar]
- Rogkala, A.; Petrounias, P.; Tsikouras, B.; Hatzipanagiotou, K. Petrogenetic significance of spinel from serpentinised peridotites from Veria-Naousa ophiolite. Bull. Geol. Soc. Gr. 2016, 50, 1999–2008. [Google Scholar]
- Rogkala, A.; Petrounias, P.; Tsikouras, B.; Hatzipanagiotou, K. New occurrence of pyroxenites in the Veria-Naousa ophiolite (north Greece): Implications on their origin and petrogenetic evolution. Geosciences 2017, 7, 92. [Google Scholar] [CrossRef]
- Rogkala, A.; Petrounias, P.; Tsikouras, B.; Giannakopoulou, P.P.; Hatzipanagiotou, K. Mineralogical Evidence for Partial Melting and Melt-Rock Interaction Processes in the Mantle Peridotites of Edessa Ophiolite (North Greece). Minerals 2019, 9, 120. [Google Scholar] [CrossRef]
- Lester, E.; Alvarez, D.; Borrego, A.G.; Valentim, B.; Flores, D.; Clift, D.A.; Rosenberg, P.; Kwiecińska, B.; Barranco, R.; Petersen, H.I.; et al. The procedure used to develop a coal char classification—Commission III combustion working group of the international committee for coal and organic petrology. Int. J. Coal Geol. 2010, 81, 333–342. [Google Scholar] [CrossRef]
- Sýkorová, I.; Pickel, W.; Christanis, K.; Wolf, M.; Taylor, G.H.; Flores, D. Classification of huminite—ICCP System 1994. Int. J. Coal Geol. 2005, 62, 85–106. [Google Scholar] [CrossRef]
- Pickel, W.; Kus, J.; Flores, D.; Kalaitzidis, S.; Christanis, K.; Cardott, B.J.; Misz-Kennan, M.; Rodrigues, S.; Hentschel, A.; Hamor-Vido, M.; et al. Classification of liptinite—ICCP System 1994. Int. J. Coal Geol. 2017, 169, 40–61. [Google Scholar] [CrossRef]
- Bish, D.L.; Post, J.E. Quantitative mineralogical analysis using the Rietveld full pattern fitting method. Am. Mineral. 1993, 78, 932–940. [Google Scholar]
- Brown, E. ISRM Suggested Methods. Rock Characterization Testing and Monitoring; London Royal School of Mines: London, UK, 1981. [Google Scholar]
- Maschio, G.; Koufopanos, C.; Lucchesi, A. Pyrolysis, a Promising Route for Biomass Utilization. Biores. Technol. 1992, 42, 219–231. [Google Scholar] [CrossRef]
- González, J.F.; Román, S.; Encinar, J.M.; Martínez, G. Pyrolysis of various biomass residues and char utilization for the production of activated carbons. J. Anal. Appl. Pyrolysis 2009, 85, 134–141. [Google Scholar] [CrossRef]
- Hawke, M.I.; Martini, I.P.; Stasiuk, L.D. A comparison of temperate and boreal peats from Ontario, Canada: Possible modern analogues for Permian coals. Int. J. Coal Geol. 1999, 41, 213–238. [Google Scholar] [CrossRef]
- Pengwu, H.; Zhao, L.; Min, C.; Huimin, H.; Zhiwu, L.; Qiwu, Z.; Wenyi, Y. Mechanochemical activation of serpentine for recovering CU (II) from waste water. Appl. Clay Sci. 2017, 149, 1–7. [Google Scholar]
- Kleiv, R.A.; Thornhill, M. Adsortive retention of copper from acidic mine water at the disused sulphide mine at Løkken, central Norway-initial experiments using olivine. Miner. Eng. 2004, 17, 195–203. [Google Scholar] [CrossRef]
- Handols, T. Det nya Sattet att Hejda Forsurningen; 1983. [Google Scholar]
- Crundwell, F.K. The mechanism of dissolution of minerals in acidic and alkaline solutions: Part I—A new theory of non-oxidation dissolution. Hydrometallurgy 2014, 149, 252–264. [Google Scholar] [CrossRef]
- Ohta, K.; Ahsan, S.; Kaneco, S.; Suzuki, T.; Mizuno, T.; Kani, K. Treatment of waste water with rocks (andesite, granite, marble), refuse concrete and refuse cement. In Proceedings of the Fourth Asian Symposium on Academic Activity for Waste Management and Resources; pp. 162–168.
- Acheampong, M.A.; Meulepas, R.J.W.; Lens, P.N.L. Removal of heavy metals and cyanide from gold mine wastewater. J. Chem. Technol. Biotechnol. 2010, 85, 590–613. [Google Scholar] [CrossRef]
- Farooq, U.; Kozinski, J.A.; Khan, M.A.; Athar, M. Biosorption of heavy metal ions using wheat based biosorbents—A review of the recent literature. Bioresour. Technol. 2010, 101, 5043–5053. [Google Scholar] [CrossRef] [PubMed]
- Vijayaraghavan, K.; Yun, Y.-S. Bacterial biosorbents and biosorption. Biotechnol. Adv. 2008, 26, 266–291. [Google Scholar] [CrossRef]
- Sen, T.K.; Gomez, D. Adsorption of zinc (Zn2+) from aqueous solution on natural bentonite. Desalination 2011, 267, 286–294. [Google Scholar] [CrossRef]
- Thornbern, M.R. Supergene alteration of sulphides. VII. Distribution of elements during the gossan-forming process. Chem. Geol. 1985, 53, 279–301. [Google Scholar]
- Nolan, A.L.; McLaughlin, M.J.; Mason, S.D. Chemical speciation of Zn, Cd, Cu and Pb in pore waters of agricultural and contaminated soils using Donnan analysis. Environ. Sci. Technol. 2003, 37, 90–98. [Google Scholar] [CrossRef]
- Chen, M.; Li, Z.; Li, X.W.; Qu, J.; Zhang, Q.W. Mechanochemically extracting tungsten through caustic processing of scheelite by controlling calcium dissolution. Int. J. Refract. Met. Hard. Mater. 2016, 58, 211–215. [Google Scholar] [CrossRef]
- James, S.L.; Adams, C.J.; Bolm, C.; Braga, D.; Collier, P.; Friscic, T.; Grepioni, F.; Harris, K.D.; Hyett, G.; Jones, W.; et al. Mechanochemistry: Opportunities for new and cleaner synthesis. Chem. Soc. Rev. 2012, 41, 413–447. [Google Scholar] [CrossRef]
- Li, J.; Hitch, M. Mechanical activation of ultramafic mine waste rock in dry condition for enhanced mineral carbonation. Miner. Eng. 2016, 95, 1–4. [Google Scholar] [CrossRef]
- Li, Z.; Chen, M.; Zhang, Q.W.; Liu, X.Z.; Saito, F. Mechanochemical processing of molybdenum and vanadium sulfides for metal recovery from spent catalysts wastes. Waste Manag. 2016, 60, 734–738. [Google Scholar] [CrossRef]
- Vdović, N.; Jurina, I.; Škapin, S.D.; Sondi, I. The surface properties of clay minerals modified by intensive dry milling—Revisited. Appl. Clay Sci. 2010, 48, 575–580. [Google Scholar] [CrossRef]
- Gossett, T.; Trancart, J.-L.; Thevenot, D.R. Batch metal removal by peat kinetics and thermodynamics. Water Res. 1986, 20, 21–26. [Google Scholar] [CrossRef]
- Ho, Y.S.; Wase, D.A.J.; Forster, C.F. Batch nickel removal from aqueous solution by Sphagnum moss peat. Water Res. 1995, 29, 1327–1332. [Google Scholar] [CrossRef]
- Chen, X.; Chen, G.; Chen, L.; Chen, Y.; Lehmann, J.; McBride, M.B.; Hay, A.G. Adsorption of copper and zinc by biochars produced from pyrolysis of hardwood and corn straw in aqueous solution. Bioresour. Technol. 2011, 102, 8877–8884. [Google Scholar] [CrossRef] [PubMed]
- Xu, X.; Cao, X.; Zhao, L.; Wang, H. Removal of Cu, Zn, and Cd from aqueous solutions by the dairy manure-derived biochar. Environ. Sci. Pollut. Res. 2013, 20, 358–368. [Google Scholar] [CrossRef] [PubMed]
- Park, J.H.; Wang, J.J.; Kim, S.H.; Cho, J.S.; Kang, S.W.; Delaune, R.D.; Han, K.J.; Seo, D.C. Recycling of rice straw through pyrolysis and its adsorption behaviors for Cu and Zn ions in aqueous solution. Colloids Surf. 2017, 533, 330–337. [Google Scholar] [CrossRef]




| Filter | Beginning-Pause of the Operation | Sample Number | Participation Ratio in Filter | Lithotype |
|---|---|---|---|---|
| Filter 1 | 1st day–4th day | BE.01 | 30% | serpentinite |
| BE.02 | 70% | magnesite | ||
| Filter 2 | 1st day–4th day | BE.82 | 100% | andesite |
| Filter 3 | 4th day–7th day | P.1 | 50% | peat |
| B.1 | 50% | biochar |
| Lithotype | Total Porosity ((nt) %) |
|---|---|
| Serpentinite (BE.01) | 6.49 |
| Andesite (BE.82) | 10.76 |
| Magnesite (ED.18) | 1.32 |
| Sample | BE.01 | BE.82 | BE.02 |
|---|---|---|---|
| Rock-Type | Serpentinite | Andesite | Magnesite |
| Major Elements (wt %) | |||
| SiO2 | 39.82 | 56.39 | 0.3 |
| TiO2 | - | 0.63 | - |
| Al2O3 | 1.01 | 17.84 | - |
| Fe2O3 t | 8.86 | 5.91 | - |
| MnO | 0.11 | 0.09 | |
| MgO | 34.17 | 2.25 | 47.06 |
| CaO | 0.10 | 5.28 | 0.42 |
| Na2O | - | 3.78 | - |
| K2O | - | 4.80 | - |
| P2O5 | - | 0.39 | - |
| LOI | 14.6 | 2.0 | 51.4 |
| Total | 98.67 | 99.36 | 99.22 |
| Trace Elements (ppm) | |||
| Cr | 2963 | - | - |
| Co | 91.1 | 17.3 | 0.6 |
| Ni | 2655.8 | 11.6 | 409.8 |
| Cu | 12.9 | 29.5 | 0.6 |
| Zn | 8 | 26 | 2 |
| Rb | 0.4 | 203.5 | 0.3 |
| Sr | 2.0 | 1896.2 | 5.2 |
| Y | 0.9 | 27.0 | - |
| Zr | 0.1 | 290.5 | - |
| Nb | 0.3 | 20.6 | - |
| Pb | 21.7 | 50.0 | 4.2 |
| Ba | 1 | 2020 | 3 |
| V | 60 | 115 | - |
| Sc | 11 | 12 | - |
| Ga | 3.1 | 20.0 | - |
| Hf | - | 7.4 | - |
| As | 7.2 | 8.9 | - |
| Hg | - | - | - |
| Ta | - | 1.2 | - |
| Th | - | 62.7 | - |
| U | 0.2 | 17.8 | - |
| Be | - | 4 | - |
| Au (ppb) | 3.4 | 1.0 | 0.9 |
| S (ppm) | Fe (ppm) | Pd (ppm) | Zn (ppm) | Cu (ppm) | Cd (ppm) | Mn (ppm) | Ni (ppm) | Ag (ppm) | Cr (ppm) | Co (ppm) | Sb (ppm) |
|---|---|---|---|---|---|---|---|---|---|---|---|
| 32,485 | 31,200 | 4801 | 3720 | 1382 | 27 | 1053 | 25 | 10 | 4 | 2.19 | 26.87 |
| Day | 1st | 1st | 1st | 2nd | 3rd | 4th | 4th | 5th | 6th | 7th |
|---|---|---|---|---|---|---|---|---|---|---|
| Day (11:00) | Day (14:30) | Day (15:00) | Day | Day | Day (10:00) | Day (19:00) | Day | Day | Day | |
| pH | 2.99 | 3.79 | 4.70 | 6.04 | 6.45 | 7.13 | 6.65 | 6.57 | 6.54 | 6.82 |
| Τwater (°C) | 25.2 | 26.0 | 29.0 | 28.0 | 29.0 | 37.6 | 37.8 | 39.8 | 38.0 | 37.6 |
| Days of Sampling→ Elements (ppb)↓ | 1st Water Analysis at the Beginning (Time: 11:00) | 1st (Time: 15:00) | 2nd (Time: 11:00) | 2nd (Time: 15:00) | 3rd | 4th | 5th | 6th | 7th |
|---|---|---|---|---|---|---|---|---|---|
| Ag | 0.18 | 0.19 | 0.12 | 0.75 | 0.15 | 0.66 | 0.09 | 0.08 | 0.14 |
| As | - | - | 1.49 | 1.37 | 0.81 | - | 1.21 | 1.25 | - |
| Ba | 20.54 | 26.31 | 120.65 | 78.56 | 86.09 | 88.12 | 86.62 | 90.88 | 71.86 |
| Be | 24.11 | 18.46 | 0.65 | 1.31 | 1.27 | 1.17 | 1.39 | 1.38 | 1.30 |
| Cd | 1717.73 | 1806.63 | 1943.44 | 1827.29 | 1840.01 | 1779.79 | 1802.34 | 1887.21 | 1995.75 |
| Co | 213.11 | 234.75 | 209.71 | 160.83 | 166.05 | 158.24 | 175.00 | 186.58 | 198.51 |
| Cr | - | - | 17.39 | 18.51 | - | - | - | - | - |
| Cs | 13.70 | 11.41 | 24.48 | 22.35 | 23.71 | 23.19 | 25.62 | 27.23 | 29.97 |
| Cu | 8847.21 | 9038.43 | 74.62 | 67.88 | 88.58 | 79.72 | 65.12 | 45.73 | 35.10 |
| Ga | 3.90 | 4.38 | 4.71 | 3.74 | 3.85 | 3.90 | 3.68 | 3.89 | 3.19 |
| Li | 25.96 | 26.82 | 48.17 | 28.01 | 29.96 | 28.75 | 38.75 | 40.62 | 40.43 |
| Mn | 70,982.00 | 75,014.04 | 79,519.93 | 62,665.14 | 65,927.83 | 64,350.15 | 73,595.50 | 78,936.65 | 84,328.29 |
| Pb | 812.77 | 329.79 | 88.58 | 37.01 | 43.93 | 30.71 | 23.27 | 20.46 | 19.94 |
| Rb | 60.42 | 47.28 | 44.67 | 43.71 | 46.72 | 45.55 | 32.50 | 25.28 | 11.11 |
| Sr | 520.57 | 460.55 | 957.43 | 972.86 | 1025.03 | 1001.78 | 1169.42 | 1261.13 | 1330.17 |
| V | - | - | - | 13.17 | 4.60 | - | - | - | - |
| U | 111.96 | 113.76 | 0.07 | 0.17 | 0.49 | 0.00 | 0.04 | 0.00 | 0.00 |
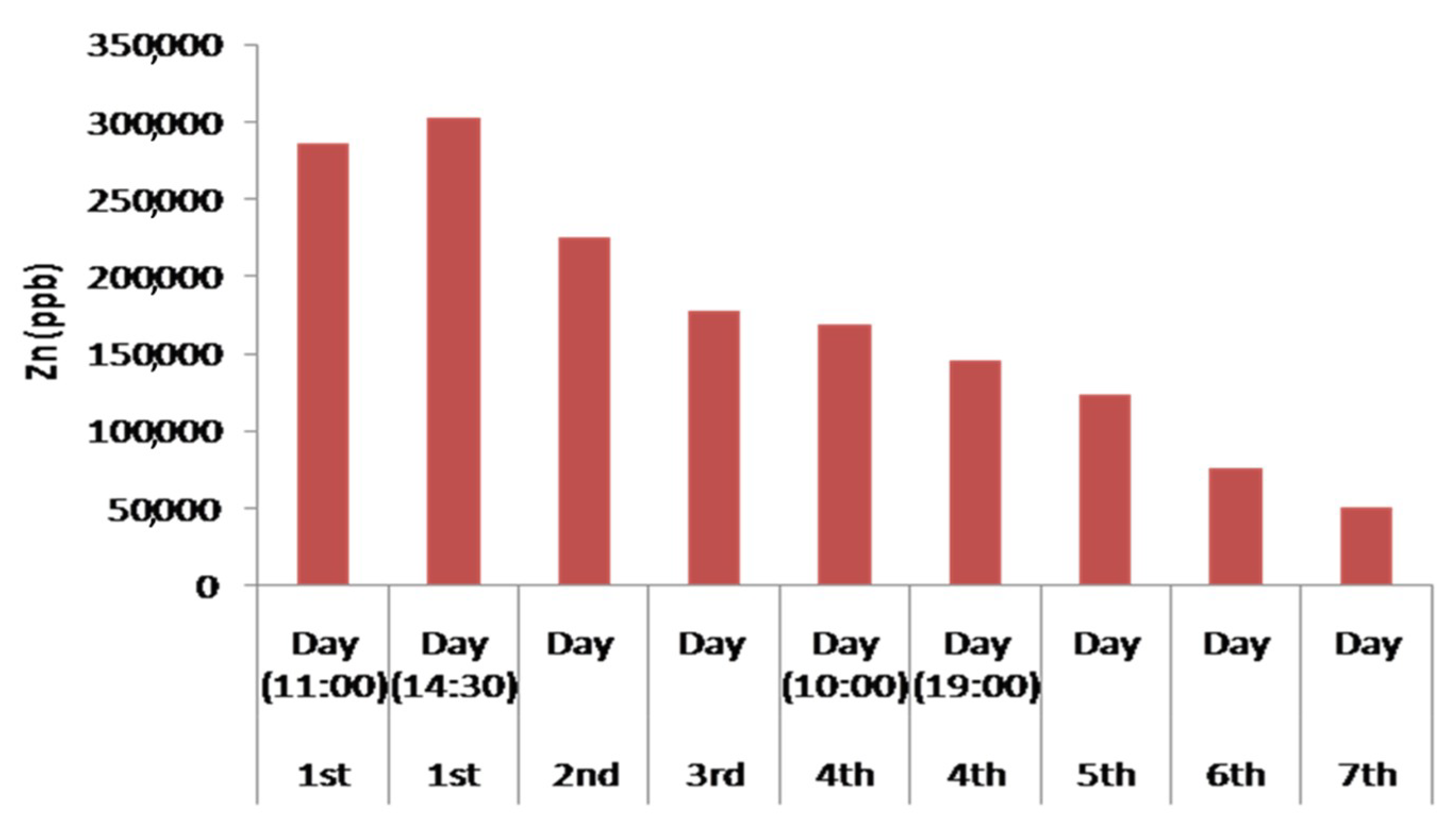
| Zn | 285,458.55 | 302,248.01 | 224,694.59 | 177,796.45 | 168,876.79 | 145,508.28 | 123,173.02 | 75,591.12 | 50,157.35 |
| Se | 24.91 | 21.10 | 18.89 | 16.77 | 13.94 | 13.36 | 16.43 | 15.74 | 20.69 |
| Ni | 1149.40 | 1227.28 | 1715.19 | 2492.34 | 2776.00 | 2959.62 | 3446.81 | 3651.29 | 3627.29 |
| Fe | 6149.02 | 7230.94 | 3368.72 | 2414.98 | 2340.99 | 2300.44 | 2298.15 | 1800.93 | 1300.57 |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Petrounias, P.; Rogkala, A.; Giannakopoulou, P.P.; Tsikouras, B.; Lampropoulou, P.; Kalaitzidis, S.; Hatzipanagiotou, K.; Lambrakis, N.; Christopoulou, M.A. An Experimental Study for the Remediation of Industrial Waste Water Using a Combination of Low Cost Mineral Raw Materials. Minerals 2019, 9, 207. https://doi.org/10.3390/min9040207
Petrounias P, Rogkala A, Giannakopoulou PP, Tsikouras B, Lampropoulou P, Kalaitzidis S, Hatzipanagiotou K, Lambrakis N, Christopoulou MA. An Experimental Study for the Remediation of Industrial Waste Water Using a Combination of Low Cost Mineral Raw Materials. Minerals. 2019; 9(4):207. https://doi.org/10.3390/min9040207
Chicago/Turabian StylePetrounias, Petros, Aikaterini Rogkala, Panagiota P. Giannakopoulou, Basilios Tsikouras, Paraskevi Lampropoulou, Stavros Kalaitzidis, Konstantin Hatzipanagiotou, Nicolaos Lambrakis, and Marina A. Christopoulou. 2019. "An Experimental Study for the Remediation of Industrial Waste Water Using a Combination of Low Cost Mineral Raw Materials" Minerals 9, no. 4: 207. https://doi.org/10.3390/min9040207
APA StylePetrounias, P., Rogkala, A., Giannakopoulou, P. P., Tsikouras, B., Lampropoulou, P., Kalaitzidis, S., Hatzipanagiotou, K., Lambrakis, N., & Christopoulou, M. A. (2019). An Experimental Study for the Remediation of Industrial Waste Water Using a Combination of Low Cost Mineral Raw Materials. Minerals, 9(4), 207. https://doi.org/10.3390/min9040207










