Dehydroxylation and Structural Distortion of Kaolinite as a High-Temperature Sorbent in the Furnace
Abstract
1. Introduction
2. Materials and Methods
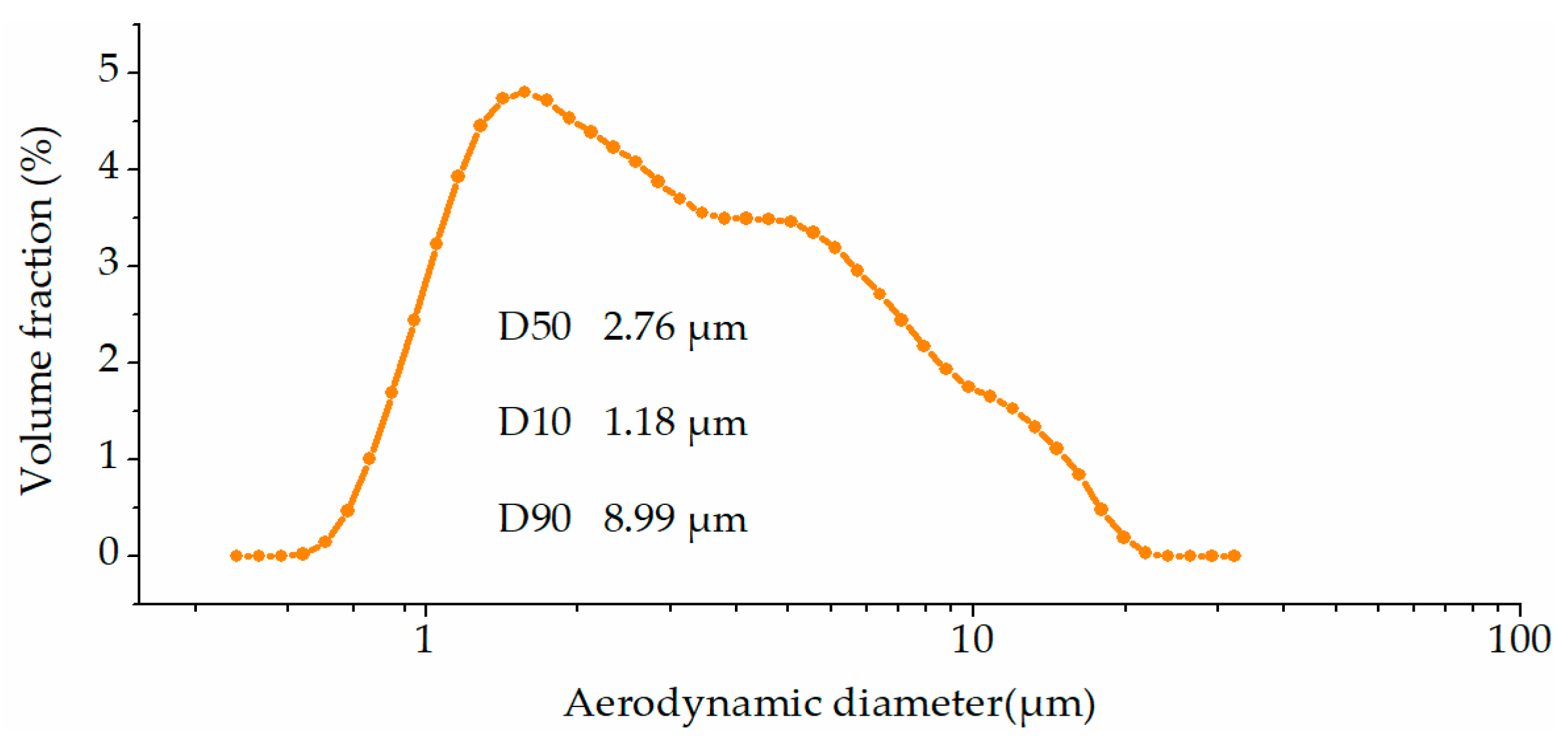
2.1. Experimental Material: Kaolinite
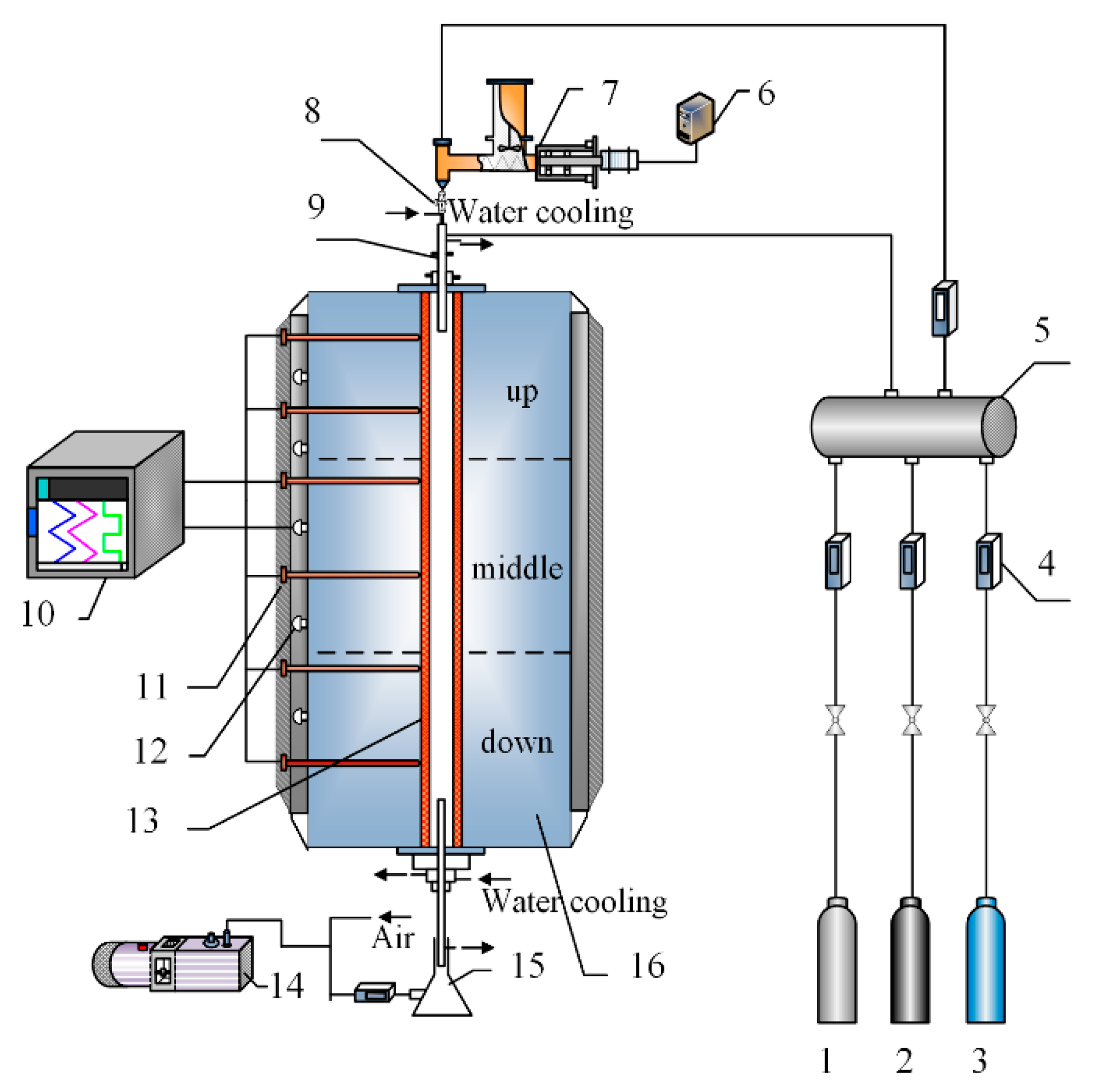
2.2. Exprimental Setup: Drop Tube Furnace
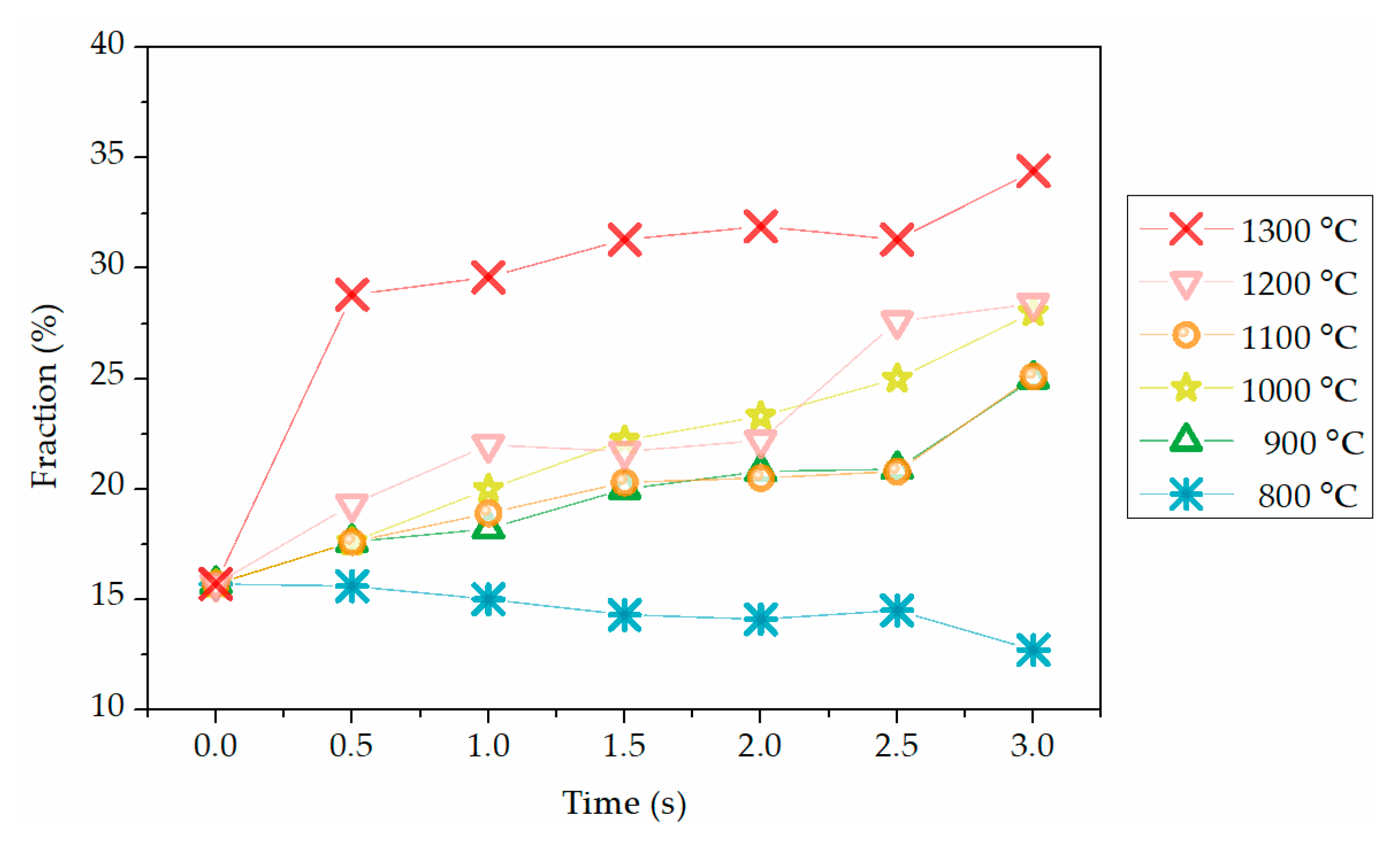
2.3. Dehydroxylation Fraction Determination of Products
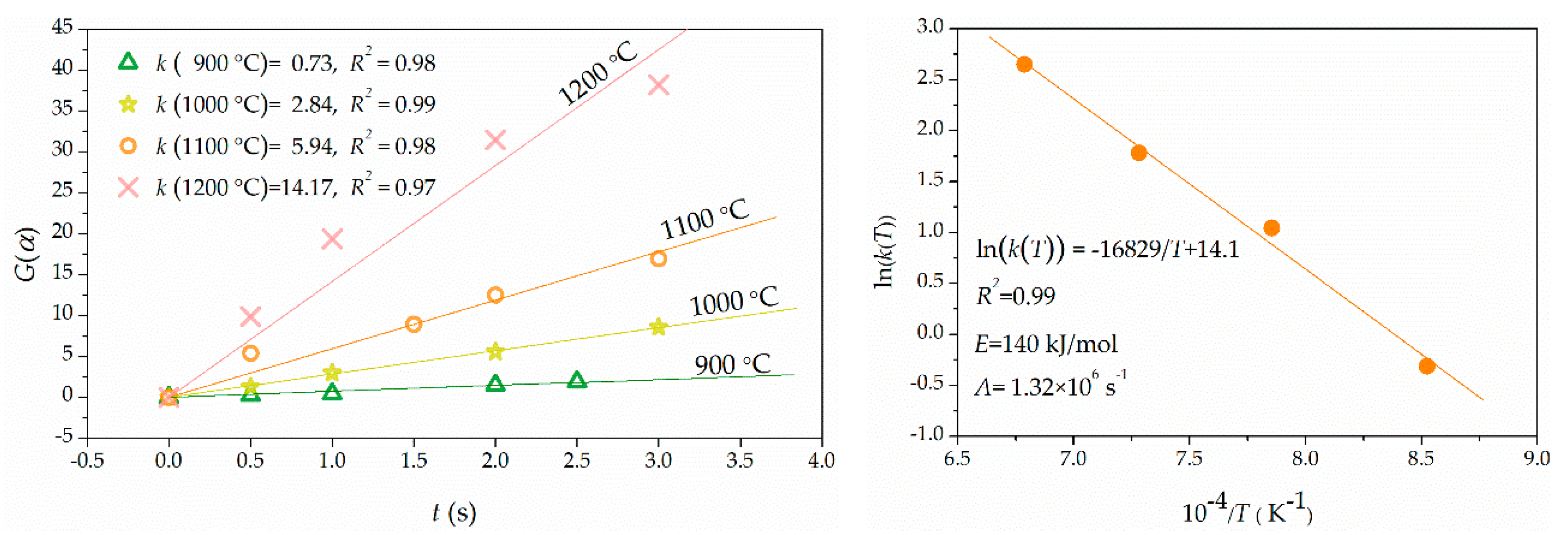
2.4. Kinetics Description of Kaolinte Dehydroxylation
2.5. Structural Characterization Methods
2.6. Choice of Carrier Gas
3. Results
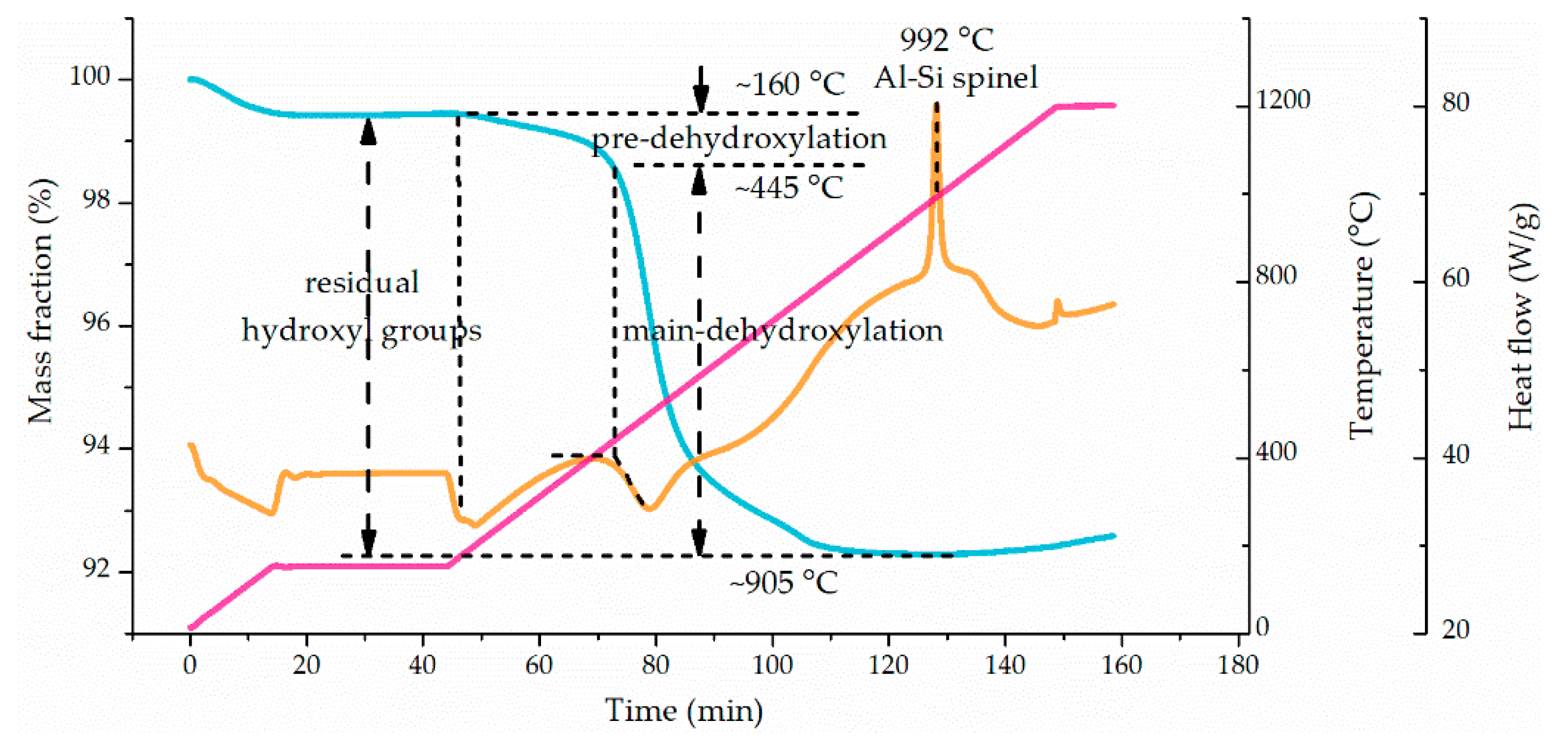
3.1. Dehydroxylation Characteristics of Kaolinite
3.2. Kaolinite Dehydroxylation Kinetics
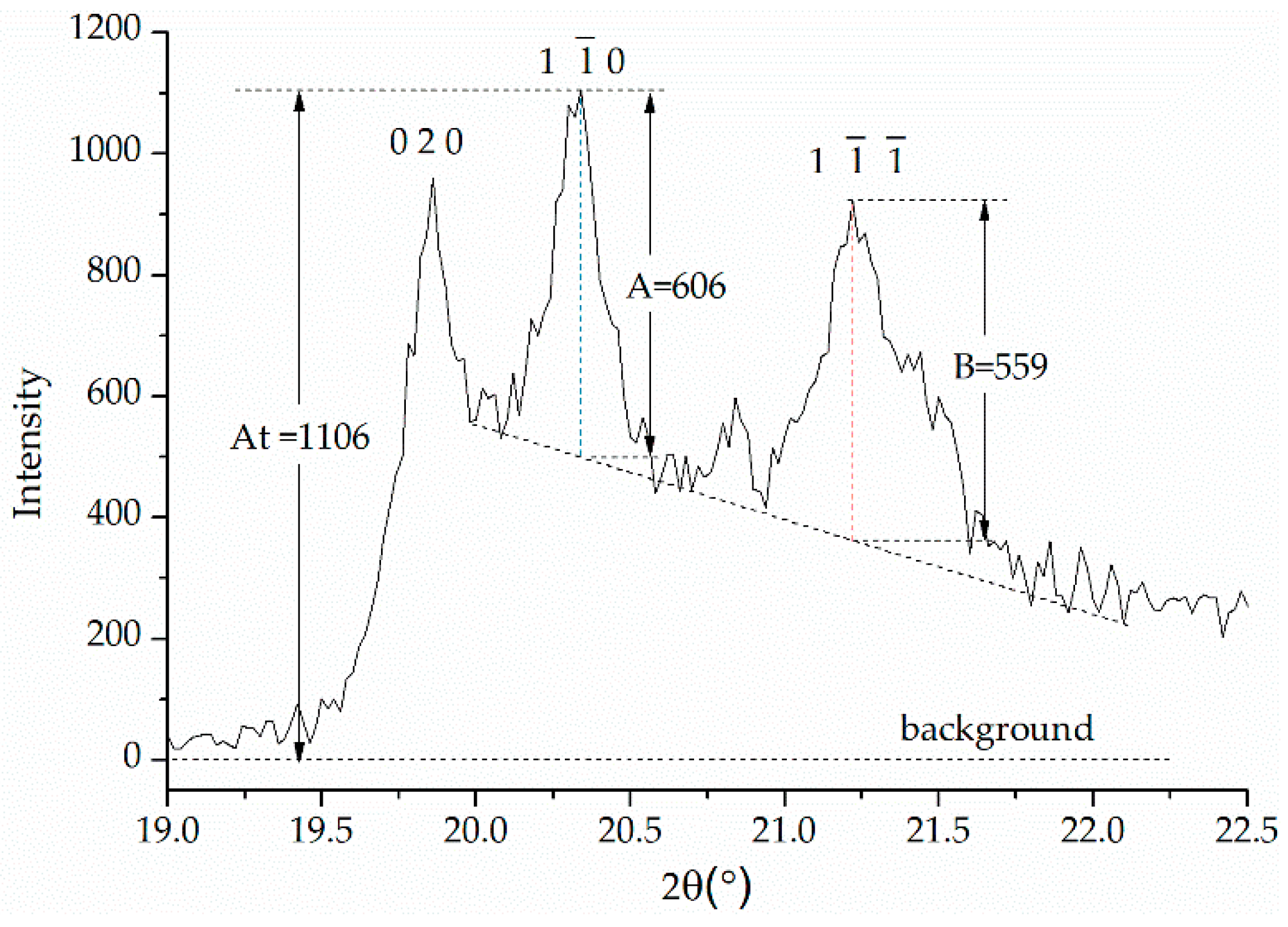
3.3. Kaolinite Crystal Phase Transition
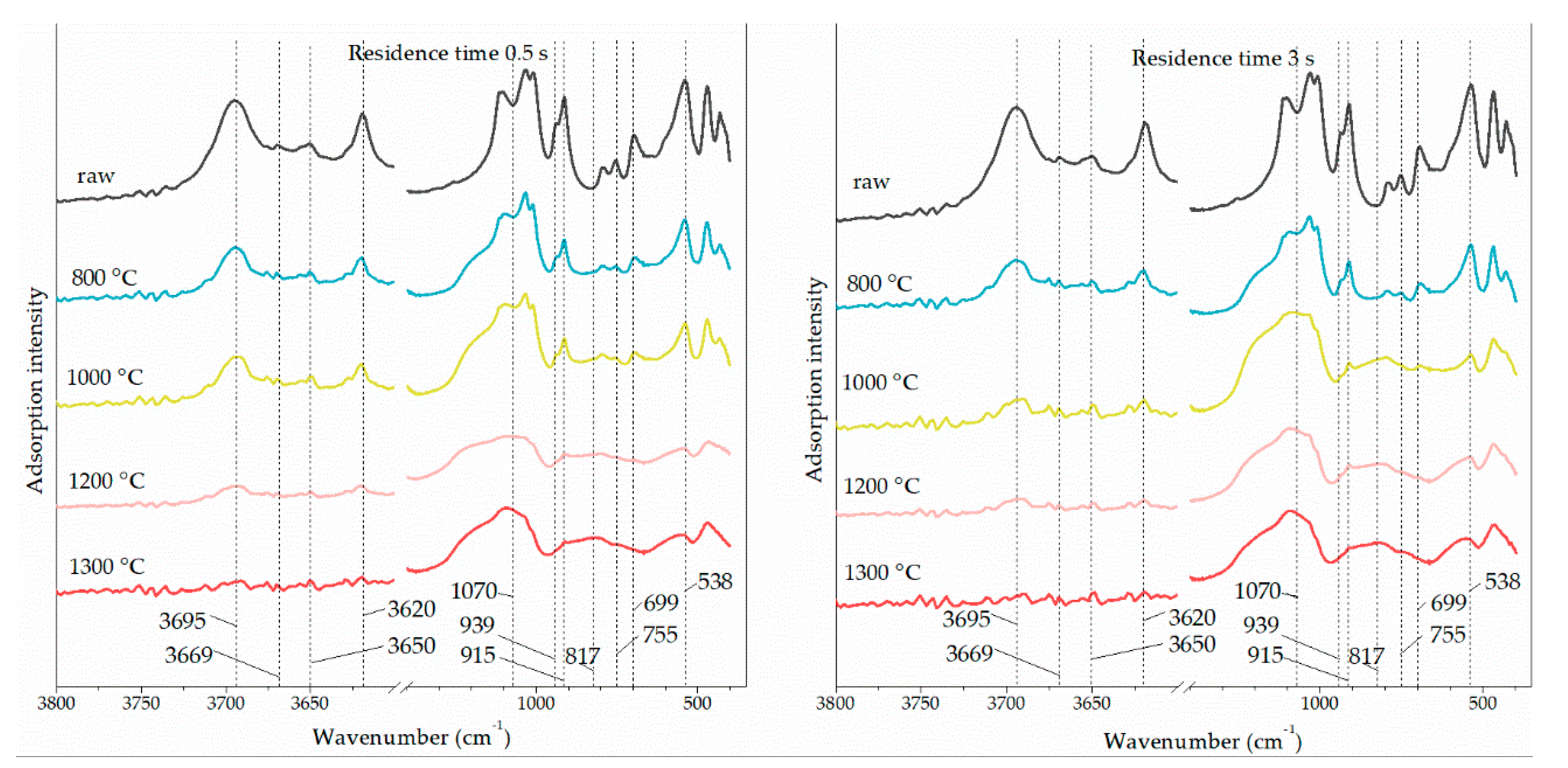
3.4. Kaolinite Surface Functional Groups Change
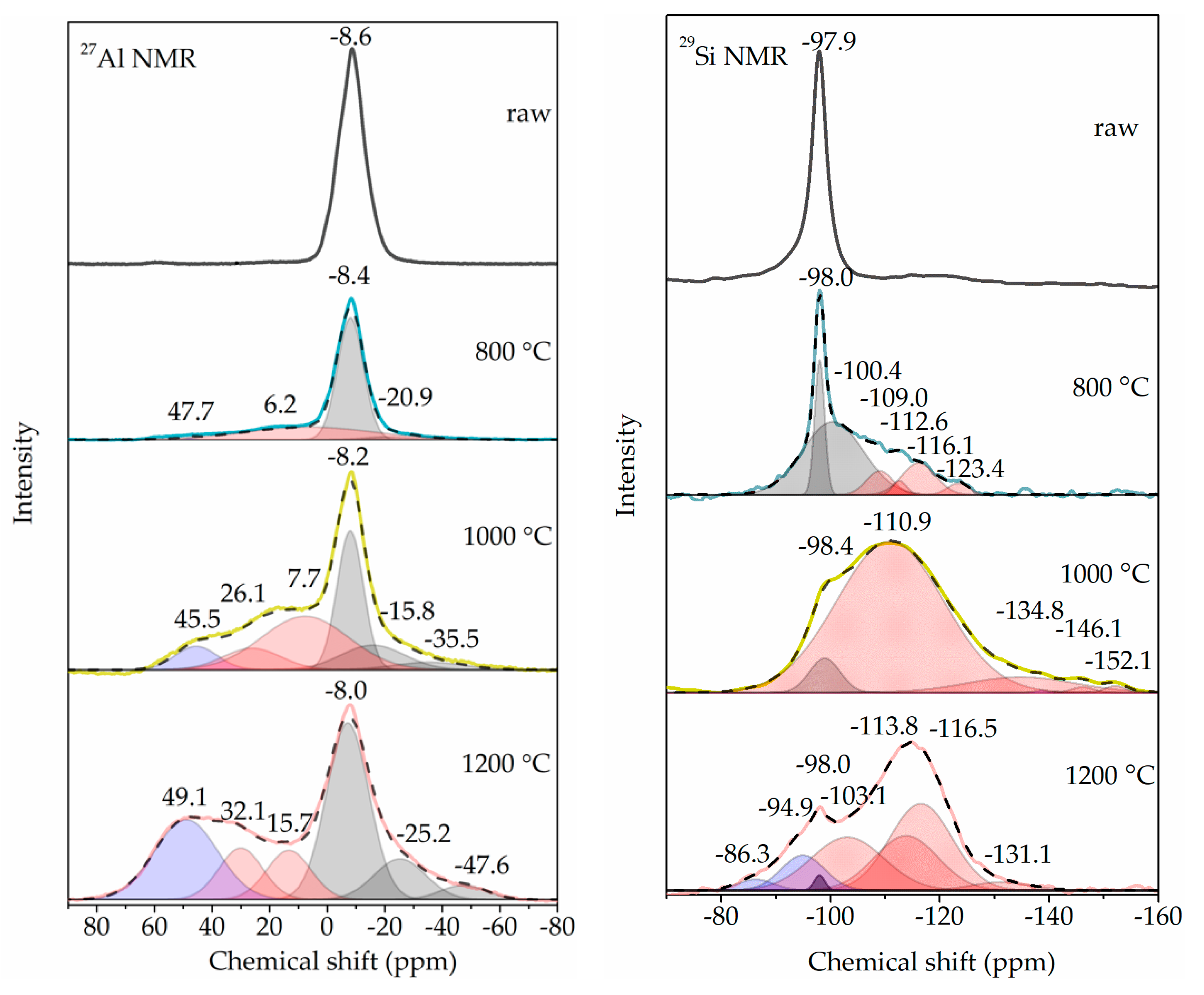
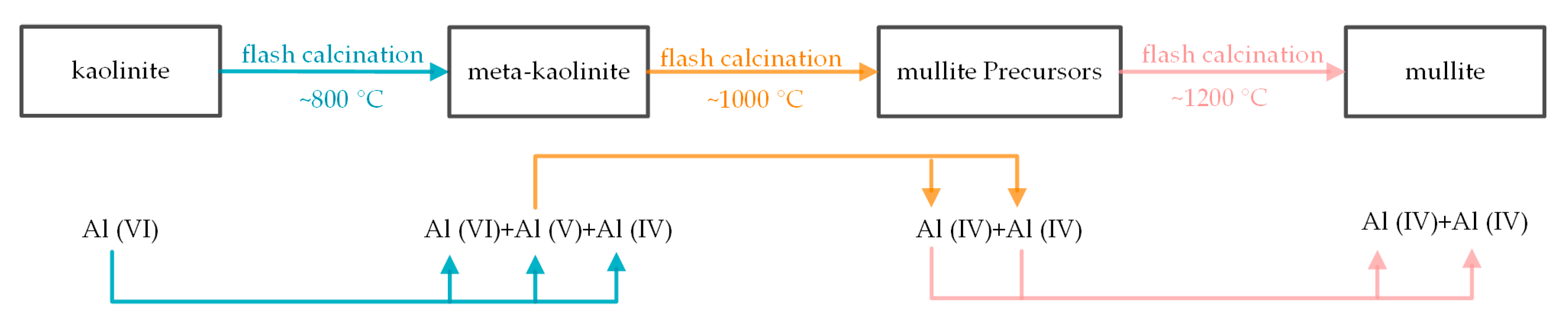
3.5. Coordination Change of Si and Al
4. Discussion
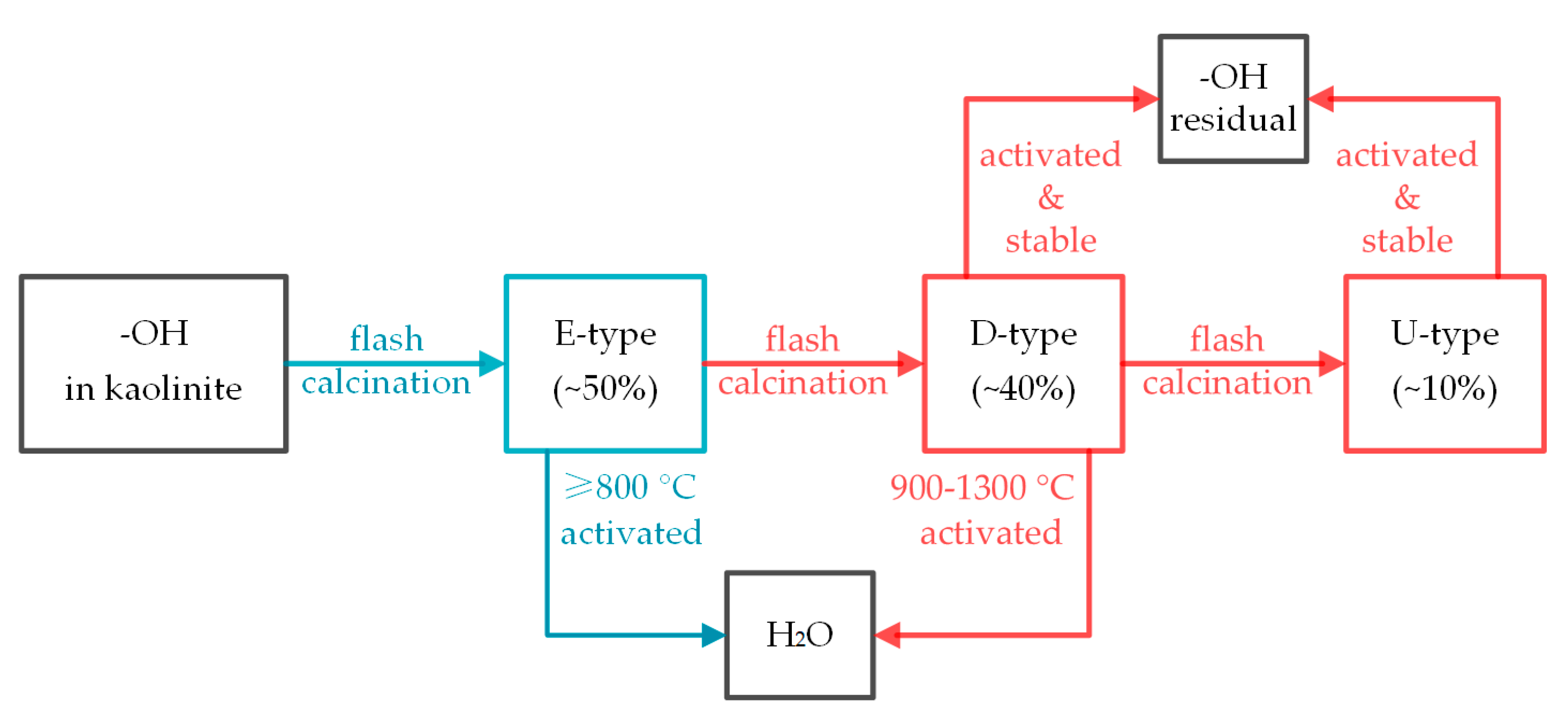
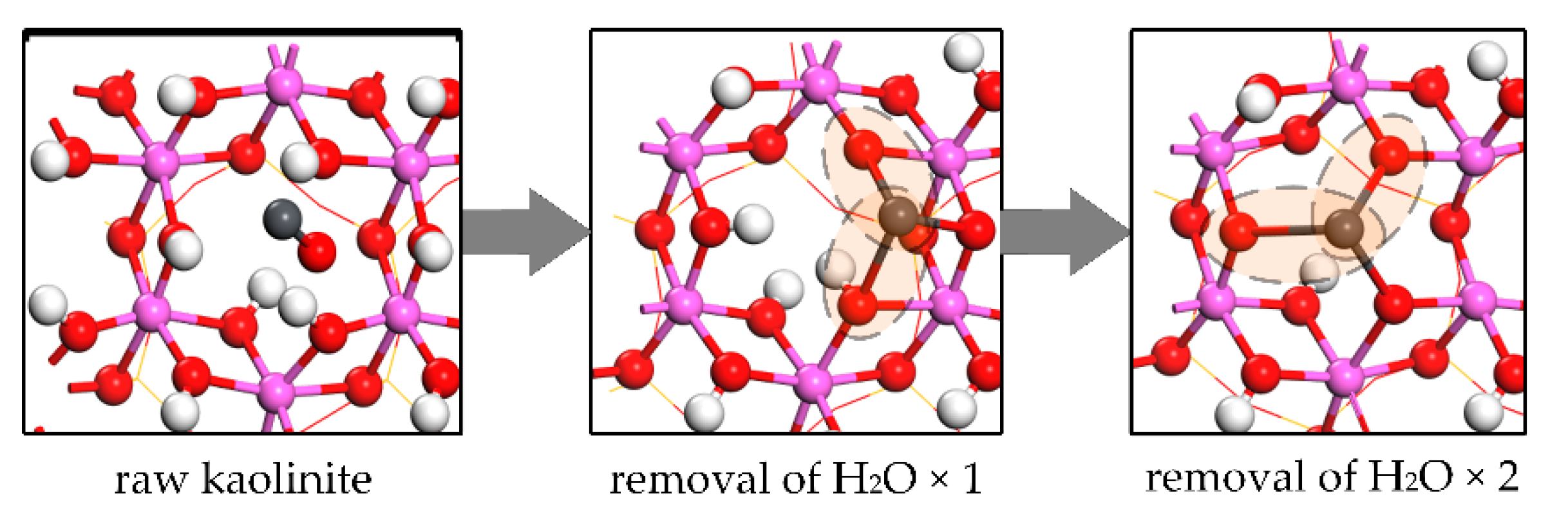
4.1. Process of Kaolinite Dehydroxylation during Flash Calcination
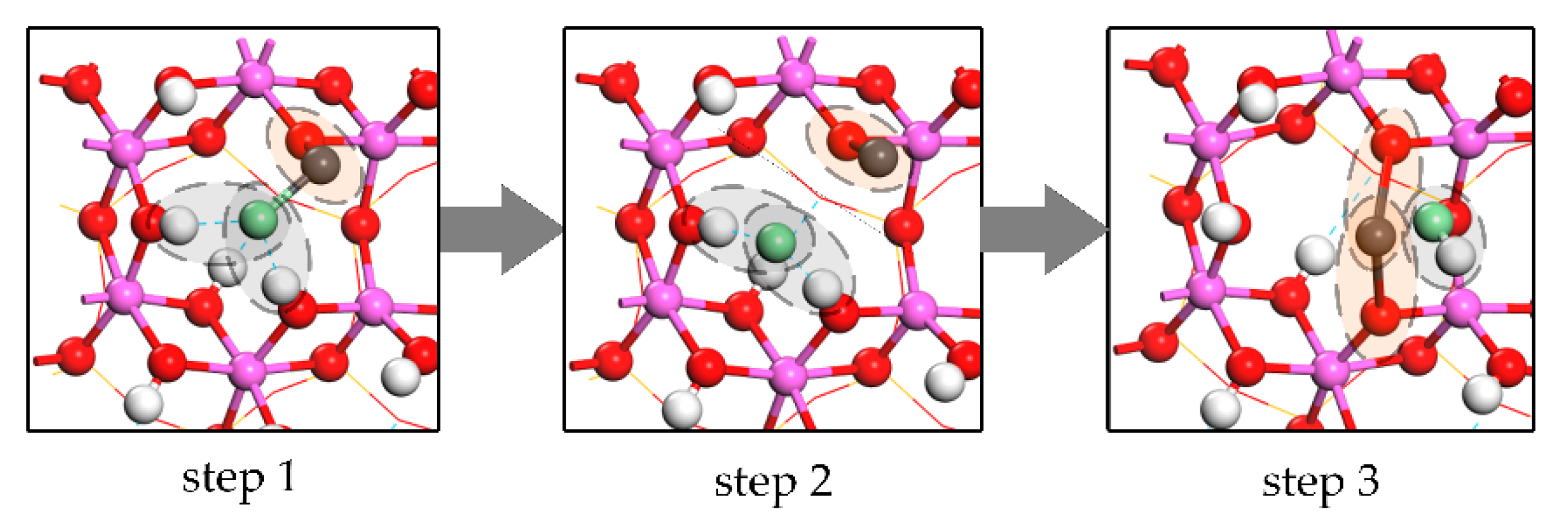
4.2. Process of Kaolinite Structural Distortion during Flash Calcination
4.3. Effect of Dehydroxylation and Structural Distortion on Metal Vapor Adsorption by Kaolinite
5. Conclusions
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Gale, T.K.; Wendt, J. Mechanisms and models describing sodium and lead scavenging by a kaolinite aerosol at high temperatures. Aerosol. Sci. Technol. 2003, 37, 865–876. [Google Scholar] [CrossRef]
- Li, G.; Li, S.; Huang, Q.; Yao, Q. Fine particulate formation and ash deposition during pulverized coal combustion of high-sodium lignite in a down-fired furnace. Fuel 2015, 143, 430–437. [Google Scholar] [CrossRef]
- Wang, G.; Jensen, P.A.; Wu, H.; Frandsen, F.J.; Sander, B.; Glarborg, P. Potassium capture by kaolin, part 1: KOH. Energy Fuels 2018, 32, 1851–1862. [Google Scholar] [CrossRef]
- Xu, Y.; Liu, X.; Wang, H.; Zeng, X.; Zhang, Y.; Han, J.K.; Xu, M.; Pan, S. Influences of in-furnace kaolin addition on the formation and emission characteristics of PM2.5 in a 1000 MW coal-fired power station. Environ. Sci. Technol. 2018, 52, 8718–8724. [Google Scholar] [CrossRef] [PubMed]
- Song, M.; Wei, Y.; Cai, S.; Yu, L.; Zhong, Z.; Jin, B. Study on adsorption properties and mechanism of Pb2+ with different carbon based adsorbents. Sci. Total Environ. 2018, 618, 1416–1422. [Google Scholar] [CrossRef] [PubMed]
- Wang, X.; Huang, Y.; Zhong, Z.; Yan, Y.; Niu, M.; Wang, Y. Control of inhalable particulate lead emission from incinerator using kaolin in two addition modes. Fuel Process. Technol. 2014, 119, 228–235. [Google Scholar] [CrossRef]
- Sun, W.; Liu, X.; Xu, Y.; Zhang, Y.; Chen, D.; Chen, Z.; Xu, M. Effects of the modified kaolin sorbents on the reduction of ultrafine particulate matter (PM0.2) emissions during pulverized coal combustion. Fuel 2018, 215, 153–160. [Google Scholar] [CrossRef]
- Wang, G.; Jensen, P.A.; Wu, H.; Frandsen, F.J.; Sander, B.; Glarborg, P. Potassium capture by kaolin, part 2: K2CO3, KCl, and K2SO4. Energy Fuels 2018, 32, 3566–3578. [Google Scholar] [CrossRef]
- Wang, X.; Huang, Y.; Pan, Z.; Wang, Y.; Liu, C. Theoretical investigation of lead vapor adsorption on kaolinite surfaces with DFT calculations. J. Hazard. Mater. 2015, 295, 43–54. [Google Scholar] [CrossRef]
- Wang, X.; Huang, Y.; Zhong, Z.; Pan, Z.; Liu, C. Theoretical investigation of cadmium vapor adsorption on kaolinite surfaces with DFT calculations. Fuel 2016, 166, 333–339. [Google Scholar] [CrossRef]
- Gasparini, E.; Tarantino, S.C.; Ghigna, P.L.; Riccardi, M.P.; Cedillo-González, E.I.; Siligardi, C.; Zema, M. Thermal dehydroxylation of kaolinite under isothermal conditions. Appl. Clay Sci. 2013, 80, 417–425. [Google Scholar] [CrossRef]
- Sperinck, S.; Raiteri, P.; Marks, N.; Wright, K. Dehydroxylation of kaolinite to metakaolin—A molecular dynamics study. J. Mater. Chem. 2011, 21, 2118–2125. [Google Scholar] [CrossRef]
- Xing, H.; Liu, H.; Zhang, X.; Deng, H.; Hu, H.; Yao, H. Enhanced sodium adsorption capacity of kaolinite using a combined method of thermal pre-activation and intercalation-exfoliation: Alleviating the problems of slagging and fouling during the combustion of Zhundong coal. Fuel 2019, 239, 312–319. [Google Scholar] [CrossRef]
- Favergeon, L.; Morandini, J.; Pijolat, M.; Soustelle, M. A general approach for kinetic modeling of solid-gas reactions at reactor scale: Application to kaolinite dehydroxylation. Oil Gas Sci. Technol. 2013, 68, 1039–1048. [Google Scholar] [CrossRef]
- Slade, R.; Davies, T.; Atakül, H.; Hooper, R.M.; Jones, D.J. Flash calcines of kaolinite: Effect of process variables on physical characteristics. J. Mater. Sci. 1992, 27, 2490–2500. [Google Scholar] [CrossRef]
- Wendt, J.; Lee, S.J. High-temperature sorbents for Hg, Cd, Pb, and other trace metals: mechanisms and applications. Fuel 2010, 89, 894–903. [Google Scholar] [CrossRef]
- Wang, G.; Jensen, P.A.; Wu, H.; Frandsen, F.J.; Bøjer, M.; Glarborg, P. Entrained flow reactor study of k-capture by solid additives. In Proceedings of the 24th European Biomass Conference and Exhibition, Amsterdam, The Netherlands, 6–9 June 2016; pp. 762–766. [Google Scholar]
- San, R.; Cyr, M.; Escadeillas, G. Characteristics and applications of flash metakaolins. Appl. Clay Sci. 2013, 83, 253–262. [Google Scholar]
- Sarı, M.; Kalpaklı, Y.; Pişkin, S. Thermal behavior and dehydroxylation kinetics of naturally occurring sepiolite and bentonite. J. Therm. Anal. Calorim. 2013, 114, 1191–1199. [Google Scholar] [CrossRef]
- Yao, H.; Mkilaha, I.S.N.; Naruse, I. Screening of sorbents and capture of lead and cadmium compounds during sewage sludge combustion. Fuel 2004, 83, 1001–1007. [Google Scholar] [CrossRef]
- Ilić, B.R.; Mitrović, A.A.; Miličić, L.R. Thermal treatment of kaolin clay to obtain metakaolin. Hem. Ind. 2010, 64, 351–356. [Google Scholar]
- Hinckley, D.N. Variability in “crystallinity” Values among the kaolin deposits of the coastal plain of georgia and south carolina. Clay Clay Miner. 1962, 11, 229–235. [Google Scholar] [CrossRef]
- Ptček, P.; Opravil, T.; Soukal, F.; Wasserbauer, J.; Másilko, J.; Baráček, J. The influence of structure order on the kinetics of dehydroxylation of kaolinite. J. Eur. Ceram. Soc. 2013, 33, 2793–2799. [Google Scholar] [CrossRef]
- Ebrahimi-Kahrizsangi, R.; Abbasi, M.H. Evaluation of reliability of Coats-Redfern method for kinetic analysis of non-isothermal TGA. Trans. Nonferrous Met. Soc. China 2008, 18, 217–221. [Google Scholar] [CrossRef]
- Zhang, J.; Wang, R.; Zhai, X.; Zhao, J.; Yang, H.; Mo, L. Determination of mechanism function and kinetic Parameters of thermal decomposition of the 2,2’-Dipyridine tris (p-methoxybenzoate) Europium(III) with non-isothermal TG and DTG curves. Chin. J. Inorg. Chem. 2000, 16, 103–110. [Google Scholar]
- Zemenová, P.; Kloužková, A.; Kohoutková, M.; Král, R.M. Investigation of the first and second dehydroxylation of kaolinite. J. Therm. Anal. Calorim. 2014, 116, 633–639. [Google Scholar] [CrossRef]
- Meinhold, R.H.; Slade, R.C.T.; Davies, T.W. High-field 27Al mas NMR studies of the formation of metakaolinite by flash calcination of kaolinite. Appl. Magn. Reson. 1993, 4, 141–155. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, H.; Xing, H.; Wang, G.; Deng, H.; Hu, H.; Li, X.; Yao, H. Correlations between the sodium adsorption capacity and the thermal behavior of modified kaolinite during the combustion of Zhundong coal. Fuel 2019, 237, 170–177. [Google Scholar] [CrossRef]
- Ortega, A.; Mac, A.M.; Gotor, F.J. The multistep nature of the kaolinite dehydroxylation: Kinetics and mechanism. J. Am. Ceram. Soc. 2010, 93, 197–203. [Google Scholar] [CrossRef]
- Stoch, L. Significance of structural factors in dehydroxylation of kaolinite polytypes. J. Therm. Anal. 1984, 29, 919–931. [Google Scholar] [CrossRef]
- Meinhold, R.H.; Atakul, H.; Daviesb, T.W.; Robert, C.T. Flash calcines of kaolinite: Kinetics of isothermal dehydroxyla.tion of partially dehydroxylated flash calcines and of flash calcination itself. J. Mater. Chem. 1992, 2, 913–921. [Google Scholar] [CrossRef]
- Redfern, S. The kinetics of dehydroxylation of kaolinite. Clay Miner. 1987, 22, 447–456. [Google Scholar] [CrossRef]
- Bridson, D.; Davies, T.; Harrison, D. Properties of flash-calcined kaolinite. Clay Clay Miner. 1985, 33, 258–260. [Google Scholar] [CrossRef]
- Zhang, X.; Liu, H.; Xing, H.; Li, H.; Hu, H.; Li, A.; Yao, H. Improved sodium adsorption by modified kaolinite at high temperature using intercalation-exfoliation method. Fuel 2017, 191, 198–203. [Google Scholar] [CrossRef]
- Mackenzie, K.; Brown, I.; Meinhold, R.H.; Bowden, M. Outstanding problems in the kaolinite-mullite reaction sequence investigated by 29Si and 27Al Solid-state nuclear magnetic resonance: I, metakaolinite. J. Am. Ceram. Soc. 1985, 68, 293–297. [Google Scholar] [CrossRef]
- White, C.E.; Provis, J.L.; Proffen, T.; Riley, D.P.; van Deventer, J.S.J. Density functional modeling of the local structure of kaolinite subjected to thermal dehydroxylation. J. Phys. Chem. A 2010, 114, 4988–4996. [Google Scholar] [CrossRef] [PubMed]
- Badogiannis, E.; Kakali, G.; Tsivilis, S. Metakaolin as supplementary cementitious material: Optimization of kaolin to metakaolin conversion. J. Therm. Anal. Calorim. 2005, 81, 457–462. [Google Scholar] [CrossRef]
- Fabbri, B.; Gualtieri, S.; Leonardi, C. Modifications induced by the thermal treatment of kaolin and determination of reactivity of metakaolin. Appl. Clay Sci. 2013, 73, 2–10. [Google Scholar] [CrossRef]
- Rocha, J.; Klinowski, J. 29Si and 27Al magic-angle-spinning NMR studies of the thermal transformation of kaolinite. Phys. Chem. Miner. 1990, 17, 179–186. [Google Scholar] [CrossRef]
- Wang, X.; Chen, M.; Liu, C.; Bu, C.; Zhang, J.; Zhao, C.; Huang, Y. Typical gaseous semi-volatile metals adsorption by meta-kaolinite: A DFT study. Int. J. Environ. Res. Public Health 2018, 15, 2154. [Google Scholar] [CrossRef] [PubMed]
- Huang, Y.; Wang, X.; Liu, C.; Wang, Y.; Dong, L. Kaolinite induced control of particulate lead and cadmium emissions during fluidized bed waste incineration. Asia-Pac. J. Chem. Eng. 2017, 12, 321–331. [Google Scholar] [CrossRef]
- Zhang, Y.; Liu, X.; Xu, Y.; Sun, W.; Xu, M. Investigation of reducing ultrafine particulate matter formation by adding modified montmorillonite during coal combustion. Fuel Process. Technol. 2017, 158, 264–271. [Google Scholar] [CrossRef]
- Wang, X.; Huang, Y.; Niu, M.; Wang, Y.; Liu, C. Effect of multi-factors interaction on trace lead equilibrium during municipal solid waste incineration. J. Mater. Cycles Waste 2016, 18, 287–295. [Google Scholar] [CrossRef]
- Zhang, Z.; Liu, J.; Yang, Y.; Shen, F.; Zhang, Z. Theoretical investigation of sodium capture mechanism on kaolinite surfaces. Fuel 2018, 234, 318–325. [Google Scholar] [CrossRef]



| Element | SiO2 | Al2O3 | K2O | SO3 | TiO2 | Fe2O3 | MgO | P2O5 | Others |
|---|---|---|---|---|---|---|---|---|---|
| Content (%) | 53.00 | 44.17 | 0.66 | 0.66 | 0.48 | 0.45 | 0.22 | 0.17 | 0.19 |
| Band (cm−1) | Functional Group | Band (cm−1) | Functional Group | Band (cm−1) | Functional Group |
|---|---|---|---|---|---|
| 538 | octahedral O-Al-OH | 817 | tetrahedral Al-O | 3620 | inner –OH |
| 699 | octahedral O-Al-OH | 915 | inner –OH | 3650 | inner-surface –OH |
| 755 | octahedral O-Al-OH | 939 | inner-surface –OH | 3669 | inner-surface –OH |
| 1070 | meta-kaolinite | 3695 | inner-surface –OH |
| Form of Al Atom | Fraction | |||
|---|---|---|---|---|
| Raw | 800 °C | 1000 °C | 1200 °C | |
| VI-coordinated Al | 100% | 67% | 45% | 52% |
| (from original peak) | 0 | (64%) | (29%) | (38%) |
| (from new peaks) | 0 | (3%) | (16%) | (14%) |
| V-coordinated Al | 0 | 33% | 47% | 22% |
| IV-coordinated Al | 0 | 0 | 8% | 26% |
| Form of Si Atom | ||||
| Kaolinite-characterized Si | 100% | 73% | 6% | 1% |
| meta-kaolinite-characterized Si + SiO2-characterized Si | 0 | 27% | 94% | 86% |
| Mullite-characterized Si | 0 | 0 | 0 | 13% |
| HI of Raw Kaolinite | Residence Time (s) | HI of Product Calcined at | ||
|---|---|---|---|---|
| 800 °C | 900 °C | 1000 °C | ||
| 1.05 | 0.5 | 0.46 | 0.37 | 0.24 |
| 3 | 0.22 | 0.20 | — | |
© 2019 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Cheng, Y.; Xing, J.; Bu, C.; Zhang, J.; Piao, G.; Huang, Y.; Xie, H.; Wang, X. Dehydroxylation and Structural Distortion of Kaolinite as a High-Temperature Sorbent in the Furnace. Minerals 2019, 9, 587. https://doi.org/10.3390/min9100587
Cheng Y, Xing J, Bu C, Zhang J, Piao G, Huang Y, Xie H, Wang X. Dehydroxylation and Structural Distortion of Kaolinite as a High-Temperature Sorbent in the Furnace. Minerals. 2019; 9(10):587. https://doi.org/10.3390/min9100587
Chicago/Turabian StyleCheng, Yun, Jingru Xing, Changsheng Bu, Jubing Zhang, Guilin Piao, Yaji Huang, Hao Xie, and Xinye Wang. 2019. "Dehydroxylation and Structural Distortion of Kaolinite as a High-Temperature Sorbent in the Furnace" Minerals 9, no. 10: 587. https://doi.org/10.3390/min9100587
APA StyleCheng, Y., Xing, J., Bu, C., Zhang, J., Piao, G., Huang, Y., Xie, H., & Wang, X. (2019). Dehydroxylation and Structural Distortion of Kaolinite as a High-Temperature Sorbent in the Furnace. Minerals, 9(10), 587. https://doi.org/10.3390/min9100587





