1. Introduction
Since 1960, the number of described uranyl mineral species has been multiplied by three, and to date, more than 250 mineral species containing uranium as a main structural element are reported in nature. This tendency is still going on and many novel uranyl minerals showing novel chemical compositions and structural arrangements are still reported each year. The most remarkable and recent discovery is certainly ewingite, Mg
8Ca
8(UO
2)
24(CO
3)
30O
4(OH)
12(H
2O)
138, the most structurally complex mineral [
1]. Based on unexampled nanoscale uranyl carbonate cages, the crystal structure of ewingite emphasizes how uranyl carbonate minerals are important in the control of the mobility of uranium in groundwater under neutral and alkaline conditions. In addition, this discovery highlights that the study of the alteration processes of uraninite, as well as the intermediate and final products of these processes, are crucial for the understanding of the evolution of used nuclear fuel in the case of long-term disposal in geological repositories.
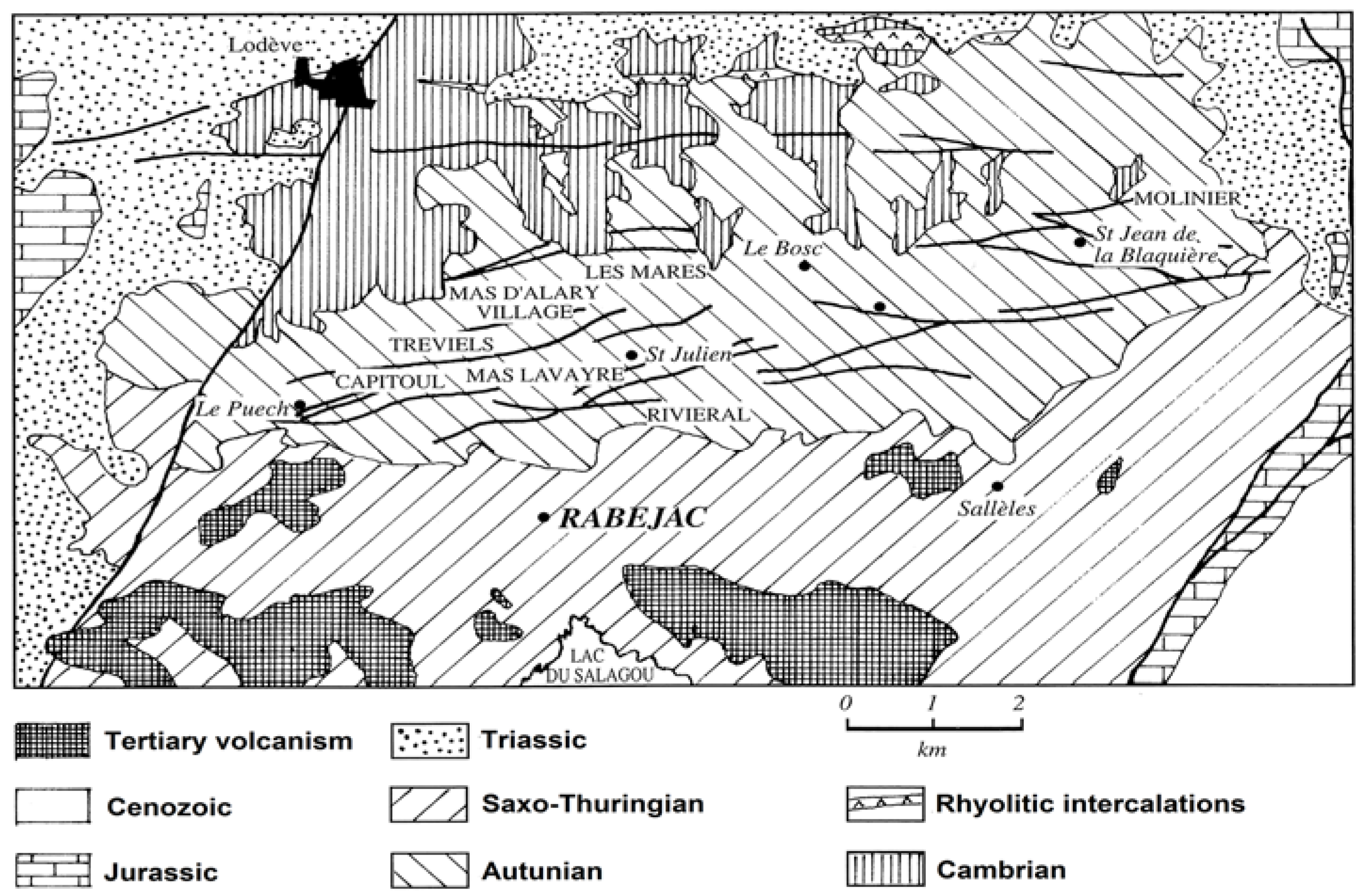
The Rabejac deposit has provided very interesting samples to study the weathering of primary uraninite and the formation of secondary uranyl minerals. The mineralogical associations that were observed in Rabejac are common with those observed in the other uranium mines of the Lodève basin. However, the Rabejac mine contains in addition rare species, such as uranyl arsenates and uranyl molybdates. The Rabejac mine is also the type locality for three uranyl mineral species: fontanite Ca[(UO
2)
3O
2(CO
3)
2](H
2O)
6 [
2,
3], seelite Mg[(UO
2)
2(As
3+O
3)
2](H
2O)
7 [
4,
5], and rabejacite Ca
2[(UO
2)
4O
4(SO
4)
2](H
2O)
8 [
6,
7].
The mining history of the Lodève uranium deposits started in 1957 with the discovery of a radiometric anomaly in the locality of Rabejac. However, the first mining investigations in the area, one shaft of 27 m and 156 m of galleries gave a disappointing estimation of the volume of uranium ore [
8]. During the following year, more surface radiometric anomalies were detected in the areas of Mas d’Alary, Riviéral, Les Mares, Usclas, and Le Puech. In 1959, new underground-prospecting works in the Rabejac and Mas d’Alary deposits reveal a potential of 1300 and 2000 tons of uranium, respectively. In 1962, 800 m of galleries and one shaft of 27 m are already dug in Mas d’Alary in order to start the mining exploitation, as well as the construction of one refinement factory in Forez. In the seventies, due to the first oil crisis and the huge increase of the oil price, the French government decided to build new nuclear stations. In order to supply these new reactors with nuclear fuel the prospections in the Lodève basin were heightened and the global potential of the deposits was estimated to be 20,000 tons of uranium. At the beginning of 1976, the COGEMA (Compagnie Générale des Matières Nucléaires) company is created to control the mining operations, and the production peak is reached in 1983 with more than 1032 tons of uranium refined. From 1988 the exchange rates of uranium slumped and the production rate of the Lodève mines was decreased. In 1993, the COGEMA group reduced the workforce by half, and the production rate was decreased from 950 tons to 500 tons of uranium per year. The mining activities were definitively shut down in 1997, the refinement factories were dismantled, and the mining sites were restored. In total, the Lodève mines have produced more than 14,000 tons of uranium [
9]. The Rabejac deposit was mined between 1989 and 1992, and it provided around 40 tons of uranium.
In the present study we report a mineralogical description as well as new chemical and crystallographic data for the following mineral species: (meta)zeunerite, (meta)nováčekite, (meta)uranospinite, heinrichite, nováčekite-I, arsenuranospathite, umohoite, calcurmolite, becquerelite, billietite, and liebigite. In addition, a description of the alteration of the primary uraninite and of the formation of secondary uranyl minerals is proposed in agreement with the observed mineral assemblages.
2. Geological Setting
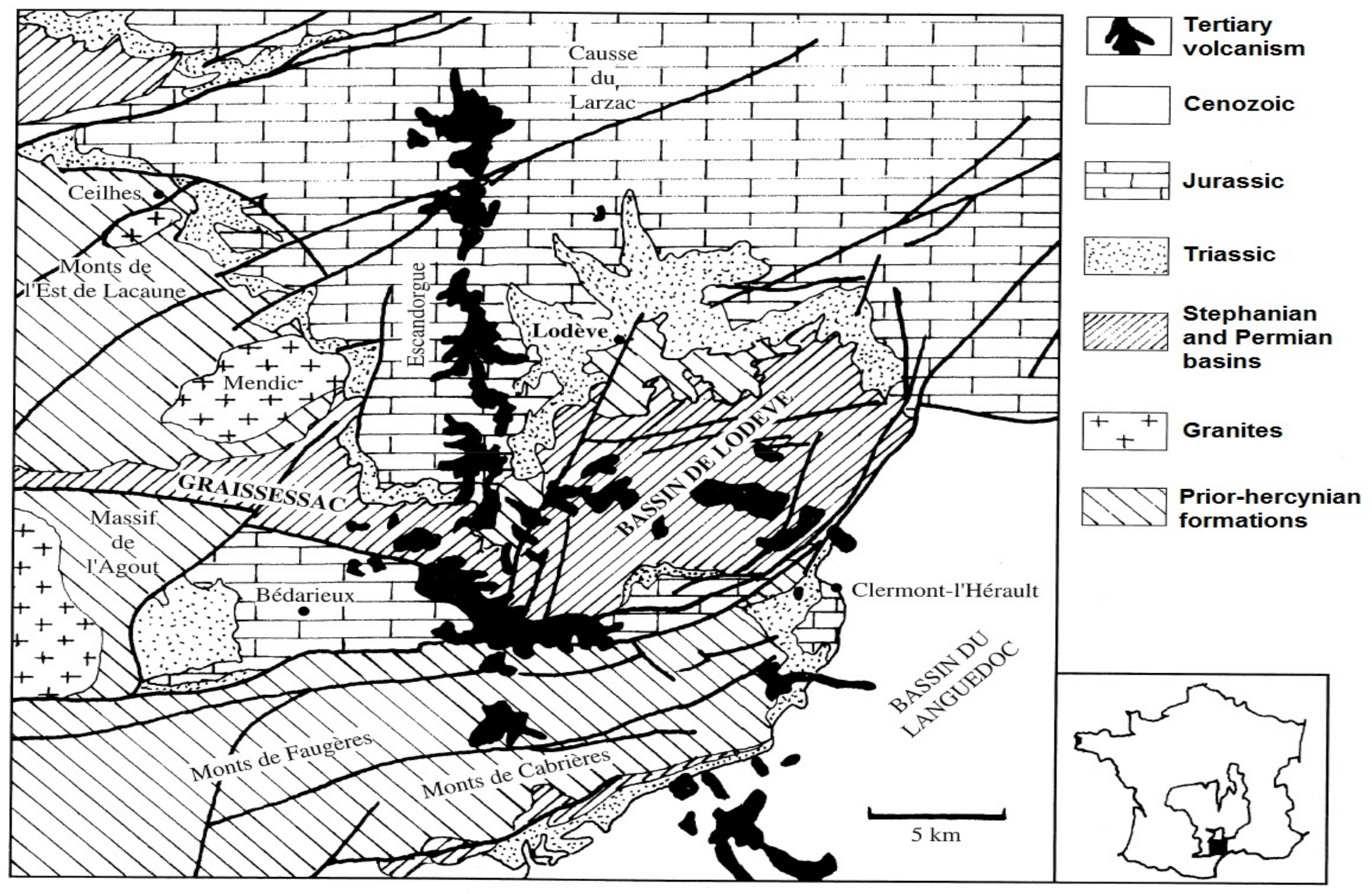
The Rabejac deposit is located in the Permian Lodève basin that is limited by the locality of Lodève, Saint-Privas, Clermont-l’Hérault, La Tour-sur-Orb, and Lumas. The Lodève basin is a geomorphological unit that is located in the south of the Central Massif, France (
Figure 1). This basin has a surface area of 25 × 10 km
2 and is surrounded by the Black Mountain in the north, the Causses of Larzac in the north and north-east, and the plains of Languedoc in the south-east.
The main geological feature of the Lodève basin is described as a monoclinal structure tilted 15° to the south. The base of the basin is constituted by Cambrian dolomite, which is covered by Stephanian and Permian rocks [
11]. The Permian record is about 2500 m and is divided into three series: the Autunian (160–240 m) and Saxonian (200 m) series constituted by a succession of grey sandstones, siltstones, arenites, and organic matter-bearing carbonate shales; and, the Thuringian series (2000 m) characterized by sandstones and pyrite-bearing schists [
8]. The Thuringian typically contains several white ash layers (5 to 50 cm). The Permian deposits are highly faulted by several orogenic phases, and the faults that were formed by these tectonic events have contributed to the remobilization of the uranium during the Lias, Aptian-Albian, Oligocene, and Miocene periods [
8,
12].
The Rabejac deposit is the only one located in the Thuringian series, while all the other uranium mines are located in the Autunian and Saxonian series (
Figure 2). The uranium mineralization generally occurs as fracture fillings and around the fault zones crossing the Thuringian series. The uranium ore also occurs as metrical stratiform lens and plurimetrical massive uraninite (variety pitchblende) near the surface, giving the Rabejac deposit a very high uranium concentration [
6,
8,
10].
5. Evolution of the Supergene Mineralization in the Rabejac Deposit
The origin of the uranium in the Lodève basin is still debatable and several hypotheses are discussed in the literature. The first hypothesis is a syngenetic process that is related to the erosion of the uranium-bearing granitic plutons of the Central Massif [
60,
61]. The second hypothesis is an origin from deep layers that have been affected by hydrothermal activities. The hydrothermal alteration has occurred through the major faults of the basement and before the beginning of the Permian sedimentation (~299 Ma) [
62]. The third hypothesis is based on acid volcanism (ignimbrite), which is often linked with a uranium supply. This hypothesis is corroborated by the presence of volcanic ash layers and the abundance of molybdenum and zirconium in the Thuringian series (~270 Ma) [
12,
63].
Despite the fact that the origin of the uranium is not clear, the emplacement of the uranium deposits is correlated with the migration of the bitumen coming from the maturation of the organic matter trapped by the Permian sediments [
12]. Uranium was concentrated during the Autunian-Saxonian sedimentation (~299 Ma), and the diagenesis occurring during the Thuringian (more than 2000 m of sediments) leads to the migration of bitumen and uranium along the faults network oriented east-west. U–Pb geochronological studies indicated two phases of uranium remobilization afterwards. The first uranium migration has occurred around 173 Ma (Lias) and the second around 110 Ma (Aptian-Albian boundary) [
64].
The mineral associations that were observed in the Rabejac mine are typical of the supergene alteration zone of the uranium deposits. These secondary uranyl minerals show a wide diversity, especially in the fault zones where the oxido-reduction processes have occurred deeply, and therefore, all the succession in the alteration products of the uraninite is observed [
8]. Firstly, in close association with uraninite (variety pitchblende), schoepite, vandendriesscheite, fourmarierite, masuyite, becquerelite and billietite are observed. These uranyl-oxide-hydroxy-hydrate minerals are very typical products of initial in-situ alteration of uraninite [
65,
66,
67]. The presence of Pb-uranyl-oxide minerals (vandendriesscheite, fourmarierite, and masuyite) indicates that the uraninite is an
old uraninite, as the majority of Pb is radiogenic, coming from the decay of uranium [
66,
68]. The small red plates of masuyite are often associated with uranyl-silicate sklodowskite [
8]. The occurrence of the rare billietite is an evidence that the Ba activity was greater than the Ca activity during some period of the weathering.
Following the crystallization of uranyl-oxide-hydroxy-hydrate minerals, the formation of uranyl silicate phases is a common step as the Si
4+ activity is increasing due to the alteration of the silicate minerals from the surrounding rocks [
69]. In the Rabejac deposits, the uranyl silicates are characterized only by minerals of the uranophane group: uranophane-α, uranophane-β, kasolite, sklodowskite, and cuprosklodowskite. Some samples show an association between uranophane-α, rutherfordine, and zeunerite. Soddyite is not reported in the Rabejac deposit, and was not observed in the samples that are investigated in the present study.
The weathering processes affecting the uraninite have also lead to the formation of several uranyl carbonates. The most common phase is the metal-free carbonate rutherfordine, often found associated with uranophane-α. Crystallization of rutherfordine indicates an increase of the
pCO
2 values and a near neutral to acid pH [
66,
70]. The increase of the pH (>8) leads to the formation of uranyl carbonates with UO
2:CO
3 ratio of 1:3, and especially of liebigite. The other observed uranyl carbonates are fontanite, which was discovered for the first time in the Rabejac deposit [
2], and the very rare urancalcarite [
71]. The UO
2:CO
3 ratio of these two species is uncommon, 3:2 and 3:1 for fontanite and urancalcarite, respectively. The conditions occurring at the moment of crystallization of these phases are not clear; however, the crystallization of fontanite and urancalcarite can be considered as an indication of the increase of Ca activity, a pH below 6, and a slight decrease of the
pCO
2 value, in comparison to the conditions leading to the formation of rutherfordine [
66,
70].
Despite the fact that uranyl phosphates constitute the most diverse group of uranyl minerals in the world, they are relatively scarce in the Rabejac deposit. Only autunite, saléeite, torbernite, and uranocircite-I are observed, as well as their dehydrated equivalent. Minerals of the phosphuranylite group, which are indicators of alkaline and neutral condition (pH > 7), are not reported [
72]. The scarcity of phosphates of the autunite-group minerals is explained by the low P activity in the ground water due to the low amount of primary phosphate-bearing minerals (e.g., apatite) in the host rocks.
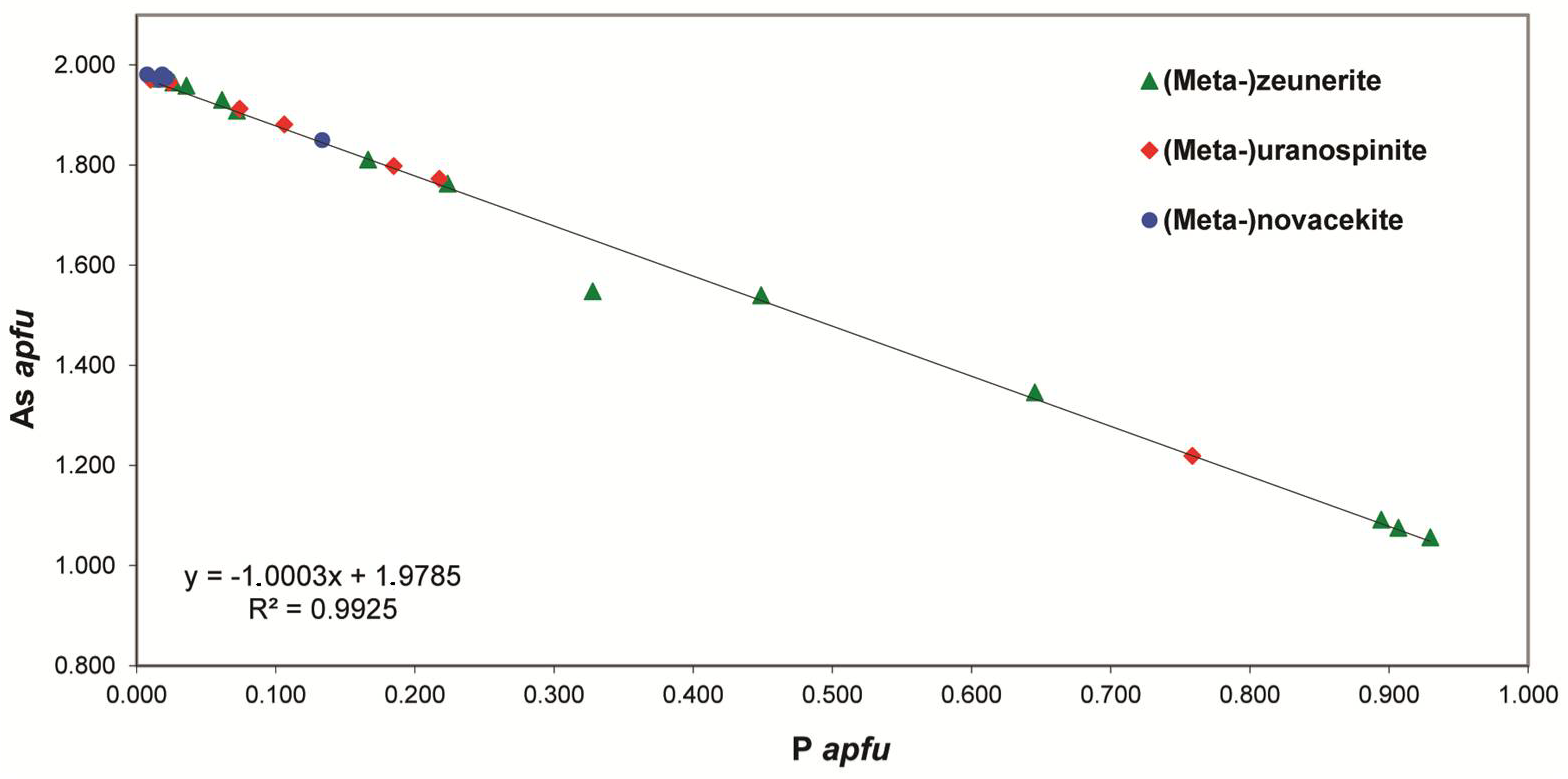
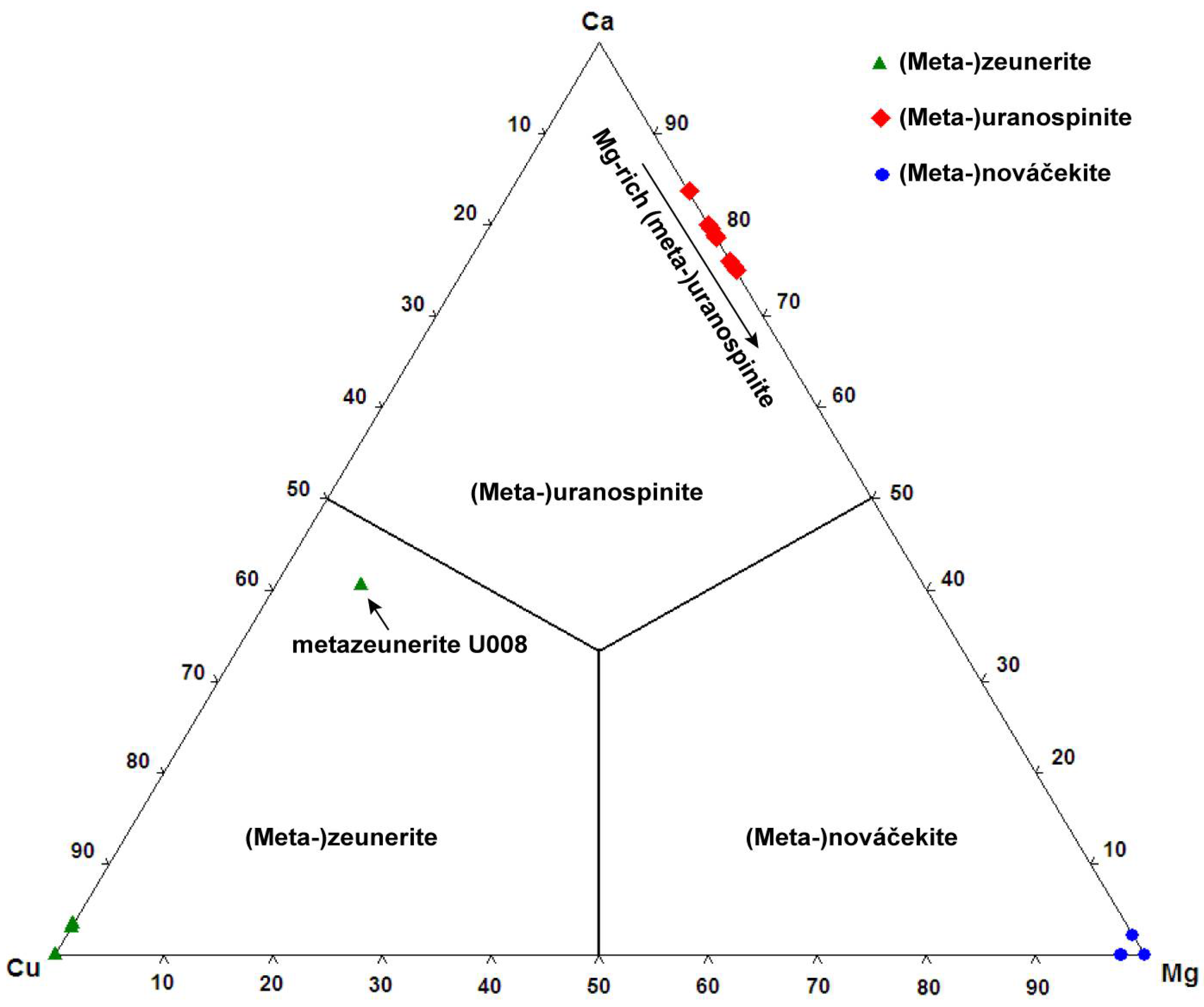
As shown in the mineralogical description part, the uranyl arsenates are very abundant in the Rabejac deposit and nearly all the uranyl arsenates of the autunite group can be observed. The origin of As is the sulphides and arsenides (such as chalcocite, bornite, domeykite, and koutekite) concentrated in the schists of the Thuringian series [
11]. The high arsenic activity has also lead to the formation of secondary arsenolite in close association with uranyl arsenates, such as nováčekite and heinrichite. Uranyl arsenates are often found in association with uranyl phosphates, uranyl silicates, and uranyl sulphates, and some remarkable samples show close association between three different uranyl arsenates (e.g., zeunerite (Cu), uranospinite (Ca) and nováčekite (Mg)). The Rabejac deposit is also the type locality, as well as the Talmessi mine (Iran), of the mixed-valence uranyl arsenite-arsenate mineral seelite. This mineral does not belong to the autunite nor to the phosphuranylite group, as its structure is based upon sheets showing the uranophane-anion topology. The formation conditions of this mineral are unclear; in the Talmessi mine seelite occurs in association with arsenide (nickeline, NiAs) but also with arsenates (e.g., annabergite, Ni
2(AsO
4)
2(H
2O)
8) [
4]. It can be assumed that local reducing conditions have allowed for the crystallization of seelite. Moreover, the uranophane-type sheets allow to stabilize the (As
3+O
3)
3− anionic group, and to prevent its oxidation into the (As
5+O
4)
3− anionic group, even under the oxidizing conditions of the surface.
Finally, the Rabejac deposit is characterized by the presence of uranyl vanadates (sengierite and tyuyamunite) and rare uranyl molybdates minerals (calcurmolite, iriginite, and umohoite). In the present study, tyuyamunite was observed in association with mottramite, PbCu(VO
4)(OH). V is certainly coming from the alteration of the organic-rich shales located in the Autunian and Saxonian series [
68], while Mo is likely coming from the alteration of molydenite (MoS
2) and Mo-rich sulphides concentrated in the schists of the Thuringian series.