Reflecting on Gold Geomicrobiology Research: Thoughts and Considerations for Future Endeavors
Abstract
1. Introduction
2. Soluble Gold Complexes
3. Gold Biomineralization and Biochemistry
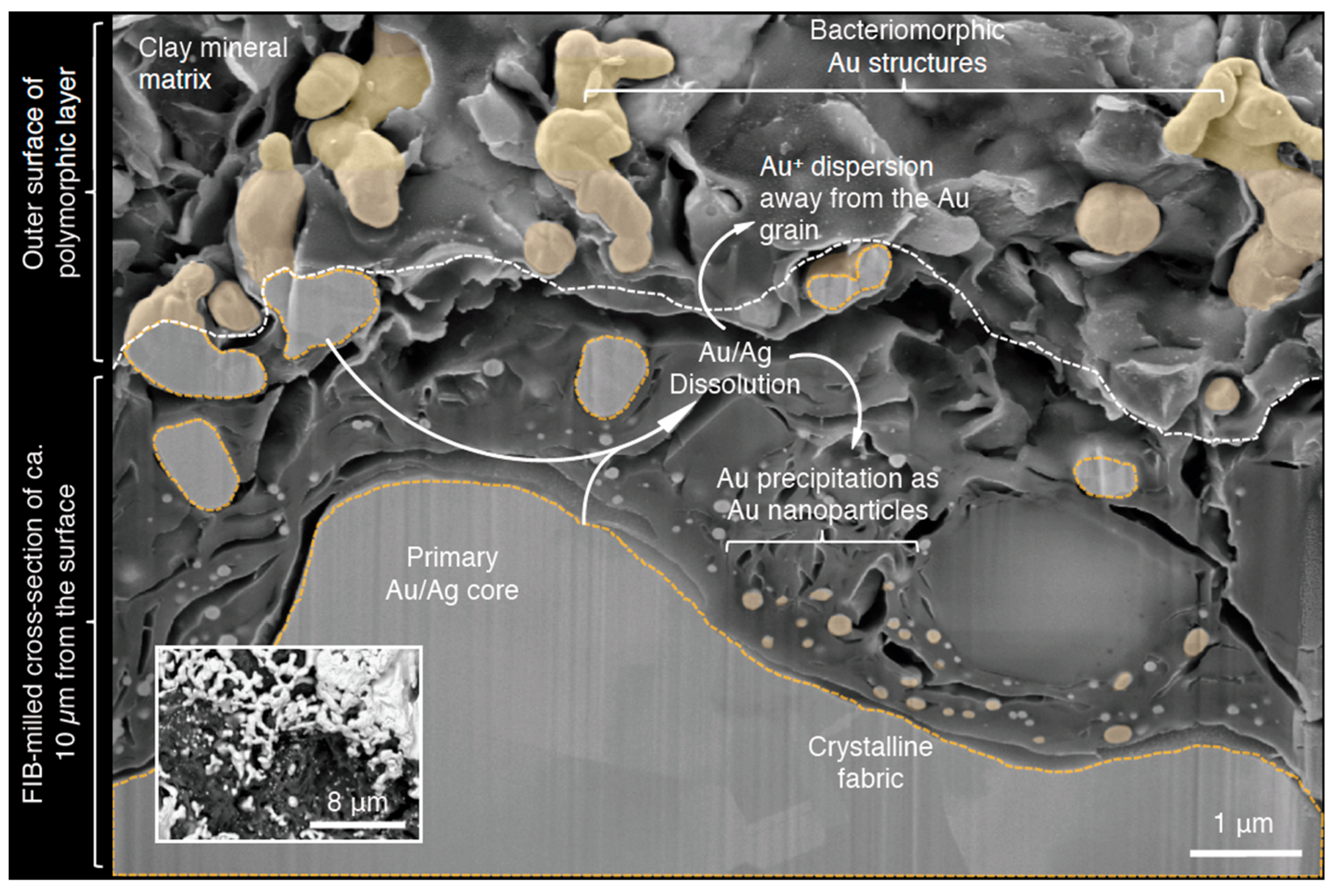
4. Gold Particle Transformation
5. Secondary Gold Transport and Kinetics of Gold Biogeochemical Cycling
6. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Frimmel, H.E. Earth’s continental crustal gold endowment. Earth Planet. Sci. Lett. 2008, 267, 45–55. [Google Scholar] [CrossRef]
- Pitcairn, I.K. Background concentrations of gold in different rock types. Appl. Earth Sci. 2013, 120, 31–38. [Google Scholar] [CrossRef]
- Boyle, R.W. The geochemistry of gold and its deposits. Geol. Surv. Can. Bull. 1979, 280, 1–54. [Google Scholar]
- Mossman, D.J.; Dyer, B.D. The geochemistry of Witwatersrand-type gold deposits and the possible influence of ancient prokaryotic communities on gold dissolution and precipitation. Precambrian Res. 1985, 30, 303–319. [Google Scholar] [CrossRef]
- Sillitoe, R.H. Major gold deposits and belts of the North and South American Cordillera: Distribution, tectonomagmatic settings, and metallogenic considerations. Econ. Geol. 2008, 103, 663–687. [Google Scholar] [CrossRef]
- Sillitoe, R.H.; Lorson, R. Epithermal gold-silver-mercury deposits at Paradise Peak, Nevada: Ore controls, porphyry gold association, detachment faulting and supergene oxidation. Econ. Geol. 1994, 89, 1228–1248. [Google Scholar] [CrossRef]
- Reith, F. Life in the deep subsurface. Geology 2011, 39, 287–288. [Google Scholar] [CrossRef]
- Rothschild, L.J.; Mancinelli, R. Life in extreme environments. Nature 2001, 409, 1092–1101. [Google Scholar] [CrossRef] [PubMed]
- MacLean, L.C.; Tyliszczak, T.; Gilbert, P.U.; Zhou, D.; Pray, T.J.; Onstott, T.C.; Southam, G. A high-resolution chemical and structural study of framboidal pyrite formed within a low-temperature bacterial biofilm. Geobiology 2008, 6, 471–480. [Google Scholar] [CrossRef] [PubMed]
- Wanger, G.; Southam, G.; Onstott, T.C. Structural and Chemical Characterization of a Natural Fracture Surface from 2.8 Kilometers Below Land Surface: Biofilms in the Deep Subsurface. Geomicrobiol. J. 2006, 23, 443–452. [Google Scholar] [CrossRef]
- Southam, G.; Saunders, J.A. The geomicrobiology of ore deposits. Econ. Geol. 2005, 100, 1067–1084. [Google Scholar] [CrossRef]
- Knowles, C.J. Microorganisms and cyanide. Bacteriol. Rev. 1976, 40, 652–680. [Google Scholar] [PubMed]
- Southam, G.; Beveridge, T.J. The in vitro formation of placer gold by bacteria. Geochim. Cosmochim. Acta 1994, 58, 4527–4530. [Google Scholar] [CrossRef]
- Reith, F.; Lengke, M.F.; Falconer, D.; Craw, D.; Southam, G. The geomicrobiology of gold. ISME J. 2007, 1, 567–584. [Google Scholar] [CrossRef] [PubMed]
- Shuster, J.; Bolin, T.; MacLean, L.C.; Southam, G. The effect of iron-oxidising bacteria on the stability of gold (I) thiosulfate complex. Chem. Geol. 2014, 376, 52–60. [Google Scholar] [CrossRef]
- Shuster, J.; Marsden, S.; Maclean, L.C.; Ball, J.; Bolin, T.; Southam, G. The immobilization of gold from gold (III) chloride by a halophilic sulfate-reducing bacterial consortium. Geol. Soc. Lond. Spec. Pub. 2013, 393, 249–263. [Google Scholar] [CrossRef]
- Reith, F.; Rogers, S.L.; McPhail, D.C.; Webb, D. Biomineralization of gold: Biofilms on bacterioform gold. Science 2006, 313, 233–236. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, D.E. Heavy metal mining using microbes. Annu. Rev. Microbiol. 2002, 56, 65–91. [Google Scholar] [CrossRef] [PubMed]
- Rawlings, D.E.; Johnson, D.B. The microbiology of biomining: Development and optimization of mineral-oxidizing microbial consortia. Microbiology 2007, 153, 315–324. [Google Scholar] [CrossRef] [PubMed]
- Zammit, C.M.; Cook, N.; Brugger, J.; Ciobanu, C.L.; Reith, F. The future of biotechnology for gold exploration and processing. Miner. Eng 2012, 32, 45–53. [Google Scholar] [CrossRef]
- Reith, F.; Brugger, J.; Zammit, C.; Nies, D.; Southam, G. Geobiological cycling of gold: From fundamental process understanding to exploration solutions. Minerals 2013, 3, 367–394. [Google Scholar] [CrossRef]
- Puddephatt, R.J. The Chemistry of Gold. Topics in Inorganic and General Chemistry: Monograph 16; Elsevier: Amsterdam, The Netherlands, 1978. [Google Scholar]
- Williams-Jones, A.E.; Bowell, R.J.; Migdisov, A.A. Gold in solution. Elements 2009, 5, 281–287. [Google Scholar] [CrossRef]
- Ta, C.; Reith, F.; Brugger, J.; Pring, A.; Lenehan, C.E. Analysis of gold(I/III)-complexes by HPLC-ICP-MS demonstrates gold (III) stability in surface waters. Environ. Sci. Technol. 2014, 48, 5737–5744. [Google Scholar] [CrossRef] [PubMed]
- Ta, C.; Brugger, J.; Pring, A.; Hocking, R.K.; Lenehan, C.E.; Reith, F. Effect of manganese oxide minerals and complexes on gold mobilization and speciation. Chem. Geol. 2015, 407, 10–20. [Google Scholar] [CrossRef]
- Mann, A.W. Mobility of gold and silver in lateritic weathering profiles: Some observations from Western Australia. Econ. Geol. 1984, 79, 38–49. [Google Scholar] [CrossRef]
- Webster, J.G. The solubility of gold and silver in the system Au-Ag-S-O2-H2O at 25 °C and 1 atm. Geochim. Cosmochim. Acta 1986, 50, 1837–1845. [Google Scholar] [CrossRef]
- Benedetti, M.; Boulegue, J. Mechanism of gold transfer and deposition in a supergene environment. Geochim. Cosmochim. Acta 1991, 55, 1539–1547. [Google Scholar] [CrossRef]
- Kerr, G.; Craw, D. Mineralogy and geochemistry of biologically-mediated gold mobilisation and redeposition in a semiarid climate, southern New Zealand. Minerals 2017, 7, 147. [Google Scholar] [CrossRef]
- Schippers, A.; Sand, W. Bacterial leaching of metal sulfides proceeds by two indirect mechanisms via thiosulfate or via polysulfides and sulfur. Appl. Environ. Microbiol. 1999, 65, 319–321. [Google Scholar] [PubMed]
- Vlassopoulos, D.; Wood, S.A. Gold speciation in natural waters: I. Solubility and hydrolysis reactions of gold in aqueous solution. Geochim. Cosmochim. Acta 1990, 54, 3–12. [Google Scholar] [CrossRef]
- Leybourne, M.I.; Goodfellow, W.D.; Boyle, D.R.; Hall, G.E.M. Form and distribution of gold mobilized into surface waters and sediments from a gossan tailings pile, Murray Brook massive sulphide desposit, New Brunswick Canada. Appl. Geochem. 2000, 15, 629–646. [Google Scholar] [CrossRef]
- Campell, S.C.; Olson, G.J.; Clark, T.R.; McFetters, G. Biogenic production of cyanide and its application to gold recovery. J. Ind. Microbiol. Biotechnol. 2001, 26, 134–139. [Google Scholar] [CrossRef]
- Fairbrother, L.; Shapter, J.; Brugger, J.; Southam, G.; Pring, A.; Reith, F. Effect of the cyanide-producing bacterium Chromobacterium violaceum on ultraflat Au surfaces. Chem. Geol. 2009, 265, 313–320. [Google Scholar] [CrossRef]
- Baker, W.E. The rold of humic acid in the transport of gold. Geochim. Cosmochim. Acta 1978, 42, 645–649. [Google Scholar] [CrossRef]
- Bowell, R.J. Supergene gold mineralogy at Ashanti, Ghana: Implications for the supergene behaviour of gold. Mineral. Mag. 1992, 56, 545–560. [Google Scholar] [CrossRef]
- Bowell, R.J.; Gize, A.P.; Foster, R.P. The role of fulic acid in the supergene migration of gold in tropical rain forest soils. Geochim. Cosmochim. Acta 1993, 57, 4179–4190. [Google Scholar] [CrossRef]
- Butt, C.R.M. Supergene gold deposits. AGSO J. Aust. Geol. Geophys. 1998, 14, 89–96. [Google Scholar]
- Fetzer, W.G. Humic acids and true organic acids as solvents of minerals. Econ. Geol. 1946, 41, 47–56. [Google Scholar] [CrossRef]
- Freise, F.W. The transportation of gold by organic underground solutions. Econ. Geol. 1931, 26, 421–431. [Google Scholar] [CrossRef]
- Carey, M.L.; McPhail, D.C.; Taufen, P.M. Groundwater flow in playa lake environments-Impact on gold and pathfinder element distributions in groundwaters surrounding mesothermal gold deposits, St. Ives area, Eastern Goldfields, Western Australia. Geochem. Explor. Environ. Anal. 2003, 3, 57–71. [Google Scholar] [CrossRef]
- Goldschmidt, V.M. The principles of distribution of chemical elements in minerals and rocks. J. Chem. Soc. 1937, 655–673. [Google Scholar] [CrossRef]
- Lowenstam, H.A. Minerals formed by organisms. Science 1981, 211, 1126–1131. [Google Scholar] [CrossRef] [PubMed]
- Beveridge, T.J.; Fyfe, W.S. Metal fixation by bacterial cell walls. Can. J. Earth Sci. 1985, 22, 1893–1898. [Google Scholar] [CrossRef]
- Daughney, C.J.; Fein, J.B.; Yee, N. A comparison of the thermodynamics of metal adsorption onto two common bacteria. Chem. Geol. 1998, 144, 161–176. [Google Scholar] [CrossRef]
- Southam, G.; Beveridge, T.J. The occurrence of sulfur and phosphorus within bacteriall derived crystalline and pseudocrastalline octahedral gold formed in vitro. Geochim. Cosmochim. Acta 1996, 60, 4369–4376. [Google Scholar] [CrossRef]
- Kashefi, K.; Tor, J.M.; Nevin, K.P.; Lovley, D.R. Reductive precipitation of gold by dissimilatory Fe(III)-reducing bacteria and archaea. Appl. Environ. Microbiol. 2001, 67, 3275–3279. [Google Scholar] [CrossRef] [PubMed]
- Lengke, M.F.; Fleet, M.E.; Southam, G. Bioaccumulation of gold by filamentous cyanobacteria between 25 and 200 °C. Geomicrobiol. J. 2006, 23, 591–597. [Google Scholar] [CrossRef]
- Lengke, M.F.; Southam, G. Bioaccumulation of gold by sulfate-reducing bacteria cultured in the presence of gold(I)-thiosulfate complex. Geochim. Cosmochim. Acta 2006, 70, 3646–3661. [Google Scholar] [CrossRef]
- Lengke, M.F.; Southam, G. The deposition of elemental gold from gold (I) thiosulfate complexes mediated by sulfate-reducing bacterial conditions. Econ. Geol. 2007, 102, 109–126. [Google Scholar] [CrossRef]
- Kenney, J.P.L.; Song, Z.; Bunker, B.A.; Fein, J.B. An experimental study of Au removal from solution by non-metabolizing bacterial cells and their exudates. Geochim. Cosmochim. Acta 2012, 87, 51–60. [Google Scholar] [CrossRef]
- Song, Z.; Kenney, J.P.L.; Fein, J.B.; Bunker, B.A. An X-ray Absorption Fine Structure study of Au adsorbed onto the non-metabolizing cells of two soil bacterial species. Geochim. Cosmochim. Acta 2012, 86, 103–117. [Google Scholar] [CrossRef]
- Fairbrother, L. Cupriavidus Metallidurans and the Biomineralization of Gold: The Role of Bacteria in the Formation of Secondary Gold on Grains in the Australian Regolith. Ph.D. Thesis, Flinders University, Adelaide, Australia, 2013. [Google Scholar]
- Fairbrother, L.; Etschmann, B.; Brugger, J.; Shapter, J.; Southam, G.; Reith, F. Biomineralization of gold in biofilms of Cupriavidus metallidurans. Environ. Sci. Technol. 2013, 47, 2628–2635. [Google Scholar] [CrossRef] [PubMed]
- Karthikeyan, S.; Beveridge, T. Pseudomonas aeruginosa biofilms react with and precipitate toxic soluble gold. Environ. Microbiol. 2002, 4, 667–675. [Google Scholar] [CrossRef] [PubMed]
- Johnston, C.W.; Wyatt, M.A.; Li, X.; Ibrahim, A.; Shuster, J.; Southam, G.; Magarvey, N.A. Gold biomineralization by a metallophore from a gold-associated microbe. Nat. Chem. Biol. 2013, 9, 241–243. [Google Scholar] [CrossRef] [PubMed]
- Butof, L.; Wiesemann, N.; Herzberg, M.; Altzschner, M.; Holleitner, A.; Reith, F.; Nies, D.H. Synergistic gold-copper detoxification at the core of gold biomineralisation in cupriavidus metallidurans. Metallomics 2018, 10, 278–286. [Google Scholar] [CrossRef] [PubMed]
- Wiesemann, N.; Butof, L.; Herzberg, M.; Hause, G.; Berthold, L.; Etschmann, B.; Brugger, J.; Martinez-Criado, G.; Dobritzsch, D.; Baginsky, S.; et al. Synergistic toxicity of copper and gold compounds in Cupriavidus metallidurans. Appl. Environ. Microbiol. 2017, 83, 1–17. [Google Scholar] [CrossRef] [PubMed]
- Reith, F.; Etschmann, B.; Grosse, C.; Moors, H.; Benotmane, M.A.; Monsieurs, P.; Grass, G.; Doonan, C.; Vogt, S.; Lai, B.; et al. Mechanisms of gold biomineralization in the bacterium Cupriavidus metallidurans. Proc. Natl. Acad. Sci. USA 2009, 106, 17757–17762. [Google Scholar] [CrossRef] [PubMed]
- Etschmann, B.; Brugger, J.; Fairbrother, L.; Grosse, C.; Nies, D.H.; Martinez-Criado, G.; Reith, F. Applying the Midas touch: Differing toxicity of mobile gold and platinum complexes drives biomineralization in the bacterium Cupriavidus metallidurans. Chem. Geol. 2016, 438, 103–111. [Google Scholar] [CrossRef]
- Rea, M.A.; Zammit, C.; Reith, F. Bacterial biofilms on gold grains—Implications for geomicrobial transformations of gold. FEMS Microbiol. Ecol. 2016, 92, 1–12. [Google Scholar] [CrossRef] [PubMed]
- Reith, F.; Fairbrother, L.; Nolze, G.; Wilhelmi, O.; Clode, P.L.; Gregg, A.; Parsons, J.E.; Wakelin, S.A.; Pring, A.; Hough, R.; et al. Nanoparticle factories: Biofilms hold the key to gold dispersion and nugget formation. Geology 2010, 38, 843–846. [Google Scholar] [CrossRef]
- Reith, F.; McPhail, D.C. Effect of resident microbiota on the solubilization of gold in soil from the Tomakin Park Gold Mine, New South Wales, Australia. Geochim. Cosmochim. Acta 2006, 70, 1421–1438. [Google Scholar] [CrossRef]
- Reith, F.; Stewart, L.; Wakelin, S.A. Supergene gold transformation: Secondary and nano-particulate gold from southern New Zealand. Chem. Geol. 2012, 320, 32–45. [Google Scholar] [CrossRef]
- Reith, F.; Wakelin, S.A.; Gregg, A.L.; Mumm, A.S. A microbial pathway for the formation of gold-anomalous calcrete. Chem. Geol. 2009, 258, 315–326. [Google Scholar] [CrossRef]
- Shuster, J.; Reith, F.; Cornelis, G.; Parsons, J.E.; Parsons, J.M.; Southam, G. Secondary gold structures: Relics of past biogeochemical transformations and implications for colloidal gold dispersion in subtropical environments. Chem. Geol. 2017, 450, 154–164. [Google Scholar] [CrossRef]
- Rea, M.A.; Standish, C.D.; Shuster, J.; Bissett, A.; Reith, F. Progressive biogeochemical transformation of placer gold particles drives compositional changes in associated biofilm communities. FEMS Microbiol. Ecol. 2018, 94, fiy080. [Google Scholar] [CrossRef] [PubMed]
- Townley, B.K.; Herail, G.; Maksaev, V.; Palacios, C.; de Parseval, P.; Sepulveda, F.; Orellana, R.; Rivas, P.; Ulloa, C. Gold grain morphology and composition as an exploration tool-application to gold exploration in covered areas. Geochem. Explor. Environ. Anal. 2003, 3, 29–38. [Google Scholar] [CrossRef]
- Kesler, S.E.; Chryssoulis, S.L.; Simon, G. Gold in poryphyry copper deposits: Its abundance and fate. Ore Geol. Rev. 2002, 21, 103–124. [Google Scholar] [CrossRef]
- Bonev, I.K.; Kerestedjian, T.; Atanassova, R.; Andrew, C.J. Morphogenesis and composition of native gold in the Chelopech volcanic-hosted Au-Cu epithermal deposit, Srednogorie zone, Bulgaria. Miner. Depos. 2002, 37, 614–629. [Google Scholar] [CrossRef]
- Hough, R.M.; Butt, C.R.M.; Buhner, J.F. The crystallography, metallography and composition of gold. Elements 2009, 5, 297–302. [Google Scholar] [CrossRef]
- Zavalía, F.M.; Craig, J.R.; Solberg, T.N. Duranusite, product of realgar alteration, Mina Capillitas, Argentina. Can. Mineral. 1999, 37, 1255–1259. [Google Scholar]
- Zavalía, F.M.; Craig, J.R. Tellurium and precious-metal ore minerals at Mina Capillitas, Northwestern Argentina. Neues Jahrbuch für Mineralogie Monatshefte 2004, 4, 176–192. [Google Scholar] [CrossRef]
- Sampson, M.I.; Phillips, C.V.; Ball, A.S. Investigation of the attachment of Thiobacillus ferrooxidans to mineral sulfides using scanning electron microscopy analysis. Miner. Eng. 2000, 13, 643–656. [Google Scholar] [CrossRef]
- Jones, R.A.; Koval, S.F.; Nesbitt, H.W. Surface alteration of arsenopyrite (FeAsS) by Thiobacillus ferrooxidans. Geochim. Cosmochim. Acta 2003, 67, 955–965. [Google Scholar] [CrossRef]
- Thurston, R.S.; Mandernack, K.W.; Shanks, W.C. Laboratory chalcopyrite oxidation by Acidithiobacillus ferrooxidans: Oxygen and sulfur isotope fractionation. Chem. Geol. 2010, 269, 252–261. [Google Scholar] [CrossRef]
- Shuster, J.; Lengke, M.F.; Zavalía, F.M.; Southam, G. Floating gold grains and nanophase particles produced from the biogeochemical weathering of a gold-bearing ore. Econ. Geol. 2016, 111, 1485–1494. [Google Scholar] [CrossRef]
- Reith, F.; Rea, M.A.D.; Sawley, P.; Zammit, C.M.; Nolze, G.; Reith, T.; Rantanen, K.; Bissett, A. Biogeochemical cycling of gold: Transforming gold particles from arctic Finland. Chem. Geol. 2018, 483, 511–529. [Google Scholar] [CrossRef]
- Reith, F.; Etschmann, B.; Dart, R.C.; Brewe, D.L.; Vogt, S.; Mumm, A.S.; Brugger, J. Distribution and speciation of gold in biogenic and abiogenic calcium carbonates—Implications for the formation of gold anomalous calcrete. Geochim. Cosmochim. Acta 2011, 75, 1942–1956. [Google Scholar] [CrossRef]
- Craw, D.; Lilly, K. Gold nugget morphology and geochemical environments of nugget formation, southern New Zealand. Ore Geol. Rev. 2016, 79, 301–315. [Google Scholar] [CrossRef]
- Shuster, J.; Johnston, C.W.; Magarvey, N.A.; Gordon, R.A.; Barron, K.; Banerjee, N.R.; Southam, G. Structural and chemical characterization of placer gold grains: Implications for bacterial contributions to grain formation. Geomicrobiol. J. 2015, 32, 158–169. [Google Scholar] [CrossRef]
- Cohen, D.R.; Waite, T.D. Interaction of aqueous Au species with goethite, smectite and kaolinite. Geochem. Explor. Environ. Anal. 2004, 4, 279–287. [Google Scholar] [CrossRef]
- Hanlie, H.; Liyun, T. Deposition of gold on kaolinite surfaces from AuCl4- solution. Geochem. Int. 2006, 44, 1246–1249. [Google Scholar]
- Hanlie, H.; Zhengyi, F.; Xinmin, M. The adsorption of [Au(HS)2]− on kaolinite surfaces: Quantum chemistry calculations. Can. Mineral. 2001, 39, 1591–1596. [Google Scholar] [CrossRef]
- Mohammadnejad, S.; Provis, J.L.; van Deventer, J.S. Gold sorption by silicates in acidic and alkaline chloride media. Int. J. Mineral. Process. 2011, 100, 149–156. [Google Scholar] [CrossRef]
- Mohammadnejad, S.; Provis, J.L.; van Deventer, J.S. Reduction of gold(III) chloride to gold(0) on silicate surfaces. J. Colloid Interface Sci. 2013, 389, 252–259. [Google Scholar] [CrossRef] [PubMed]
- Reith, F.; Cornelis, G. Effect of soil properties on gold- and platinum nanoparticle mobility. Chem. Geol. 2017, 466, 446–453. [Google Scholar] [CrossRef]
- Hanlie, H.; Liyun, T.; Qiujuan, B.; Yong, Z. Interface characteristics between colloidal gold and kaolinite surface by XPS. J. Wuhan Univ. Technol. Mater. Sci. Ed. 2006, 21, 90–93. [Google Scholar] [CrossRef]
- Zhu, L.; Letaief, S.; Liu, Y.; Gervais, F.; Detellier, C. Clay mineral-supported gold nanoparticles. Appl. Clay Sci. 2009, 43, 439–446. [Google Scholar] [CrossRef]
- Shuster, J.; Southam, G. The in-vitro “growth” of gold grains. Geology 2014, 43, 79–82. [Google Scholar] [CrossRef]
- Turkevich, J.; Stevenson, P.C.; Hillier, J. A study of the nucleation and growth processes in the synthesis of colloidal gold. Discuss. Faraday Soc. 1951, 11, 55–75. [Google Scholar] [CrossRef]
- Fairbrother, L.; Brugger, J.; Shapter, J.; Laird, J.S.; Southam, G.; Reith, F. Supergene gold transformation: Biogenic secondary and nano-particulate gold from arid Australia. Chem. Geol. 2012, 320, 17–31. [Google Scholar] [CrossRef]
- Campbell, G.; MacLean, L.; Reith, F.; Brewe, D.; Gordon, R.; Southam, G. Immobilisation of platinum by cupriavidus metallidurans. Minerals 2018, 8, 10. [Google Scholar] [CrossRef]
- Shuster, J.; Reith, F.; Izawa, M.; Flemming, R.; Banerjee, N.; Southam, G. Biogeochemical cycling of silver in acidic, weathering environments. Minerals 2017, 7, 218. [Google Scholar] [CrossRef]
- Reith, F.; Zammit, C.M.; Shar, S.S.; Etschmann, B.; Bottrill, R.; Southam, G.; Ta, C.; Kilburn, M.; Oberthür, T.; Ball, A.S.; et al. Biological role in the transformation of platinum-group mineral grains. Nat. Geosci. 2016, 9, 294. [Google Scholar] [CrossRef]


© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Shuster, J.; Reith, F. Reflecting on Gold Geomicrobiology Research: Thoughts and Considerations for Future Endeavors. Minerals 2018, 8, 401. https://doi.org/10.3390/min8090401
Shuster J, Reith F. Reflecting on Gold Geomicrobiology Research: Thoughts and Considerations for Future Endeavors. Minerals. 2018; 8(9):401. https://doi.org/10.3390/min8090401
Chicago/Turabian StyleShuster, Jeremiah, and Frank Reith. 2018. "Reflecting on Gold Geomicrobiology Research: Thoughts and Considerations for Future Endeavors" Minerals 8, no. 9: 401. https://doi.org/10.3390/min8090401
APA StyleShuster, J., & Reith, F. (2018). Reflecting on Gold Geomicrobiology Research: Thoughts and Considerations for Future Endeavors. Minerals, 8(9), 401. https://doi.org/10.3390/min8090401




