Impact of Clay Minerals on the Dewatering of Coal Slurry: An Experimental and Molecular-Simulation Study
Abstract
1. Introduction
2. Materials and Methods
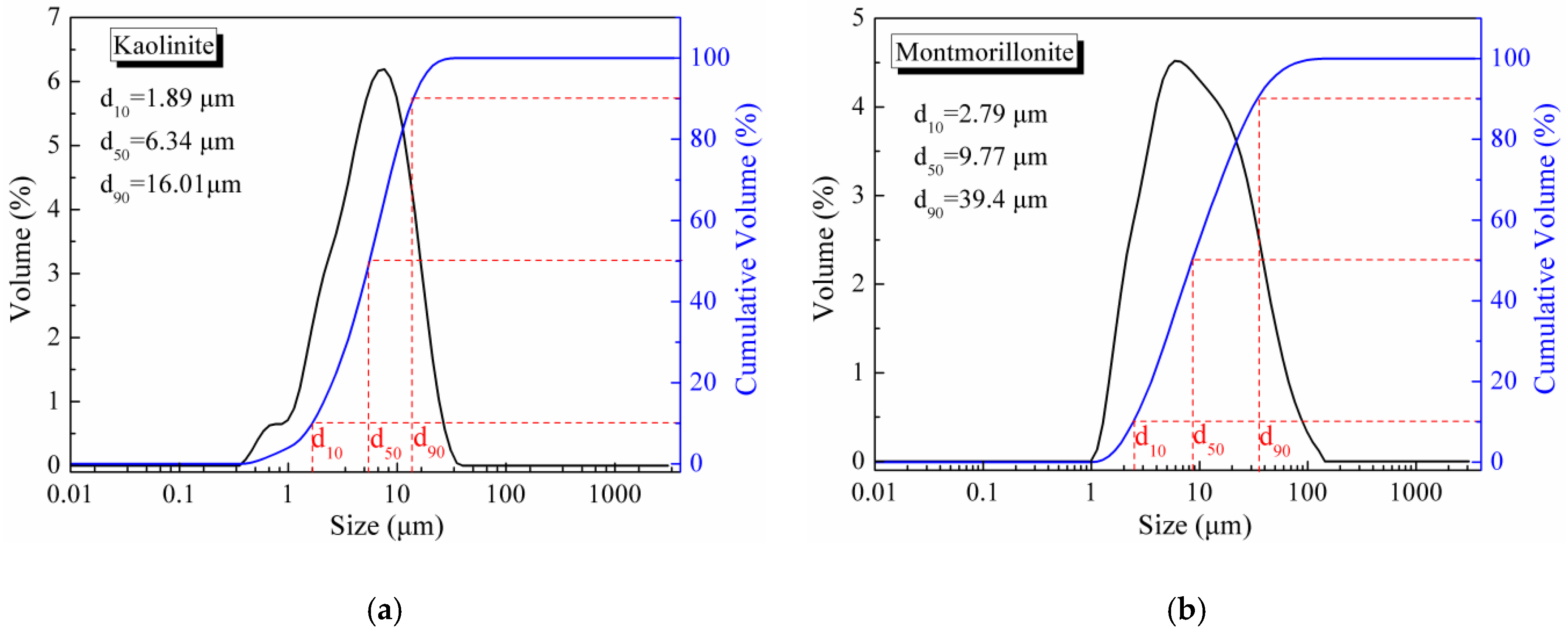
2.1. Materials
2.2. Filtration Experiments
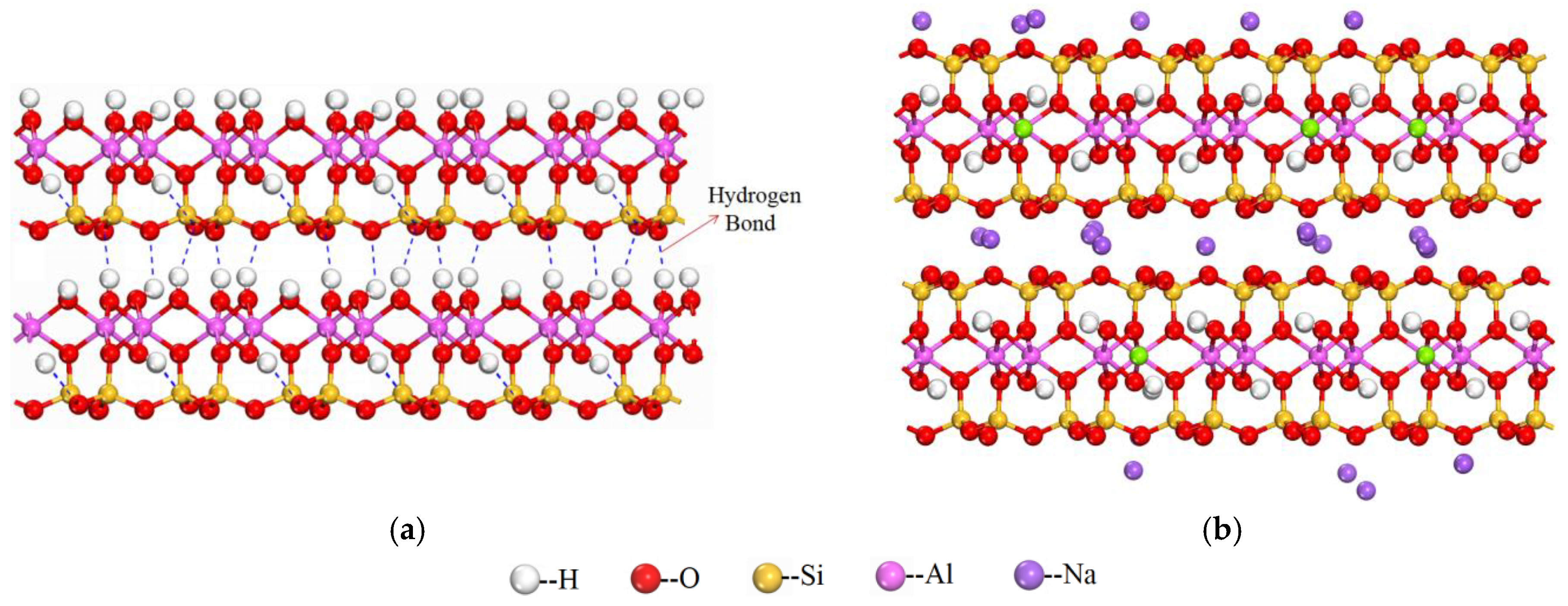
2.3. Molecular Simulation
3. Results and Discussion
3.1. Filtration Experiment Results
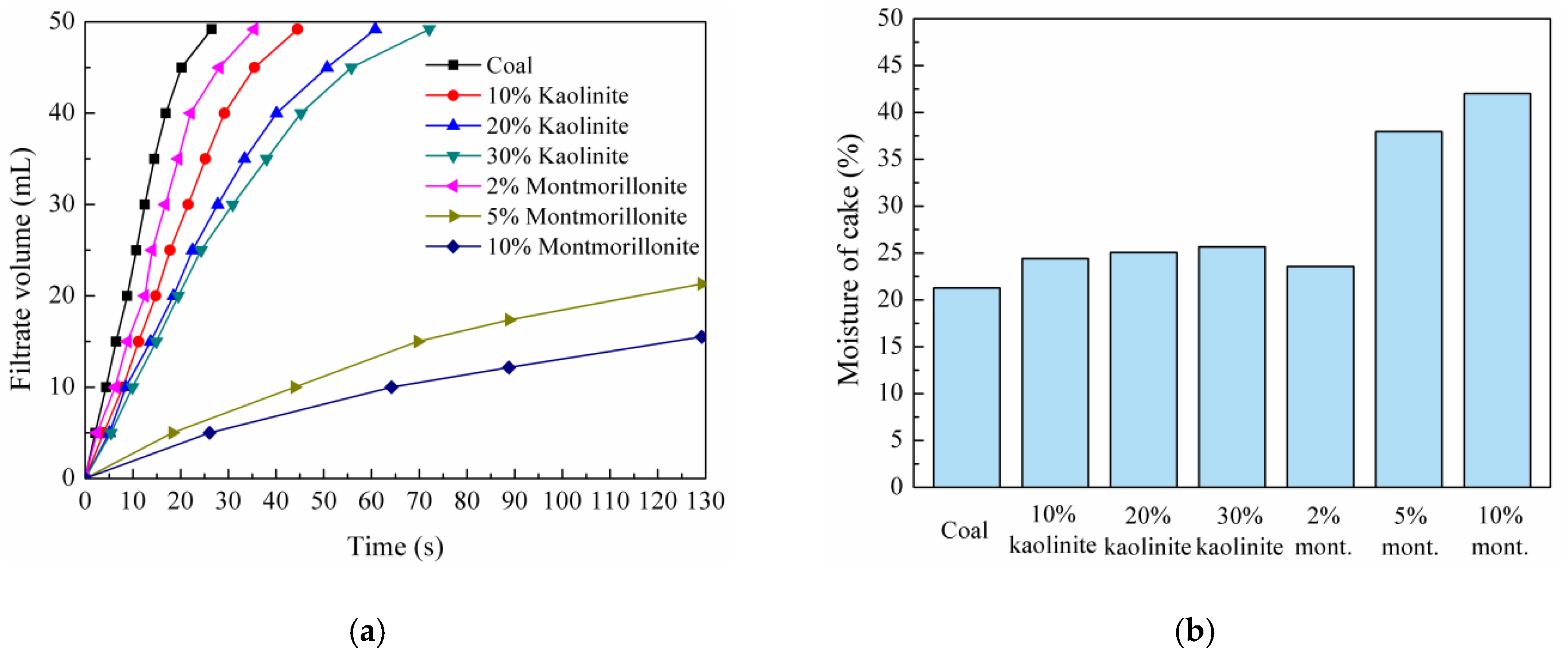
3.1.1. Filtration Velocity
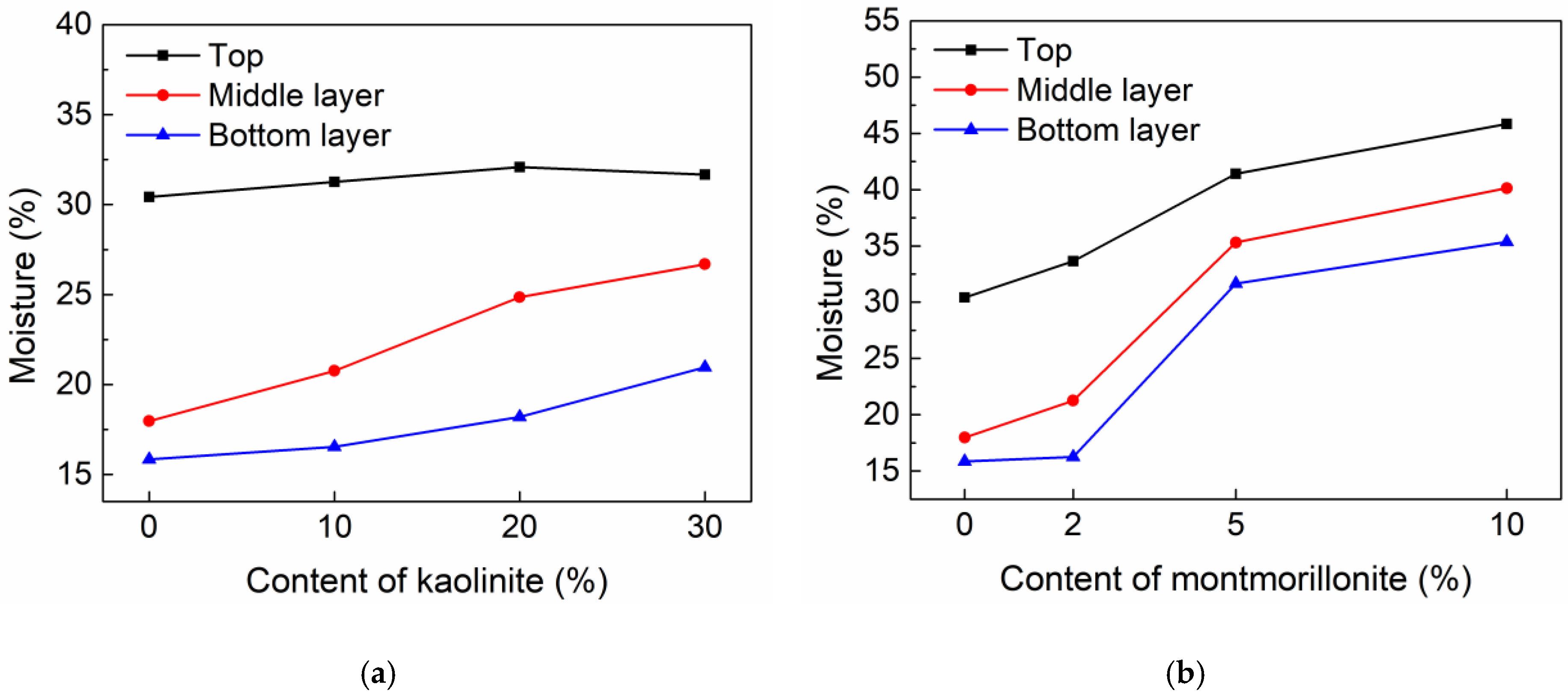
3.1.2. Moisture
3.1.3. Average Specific Resistance
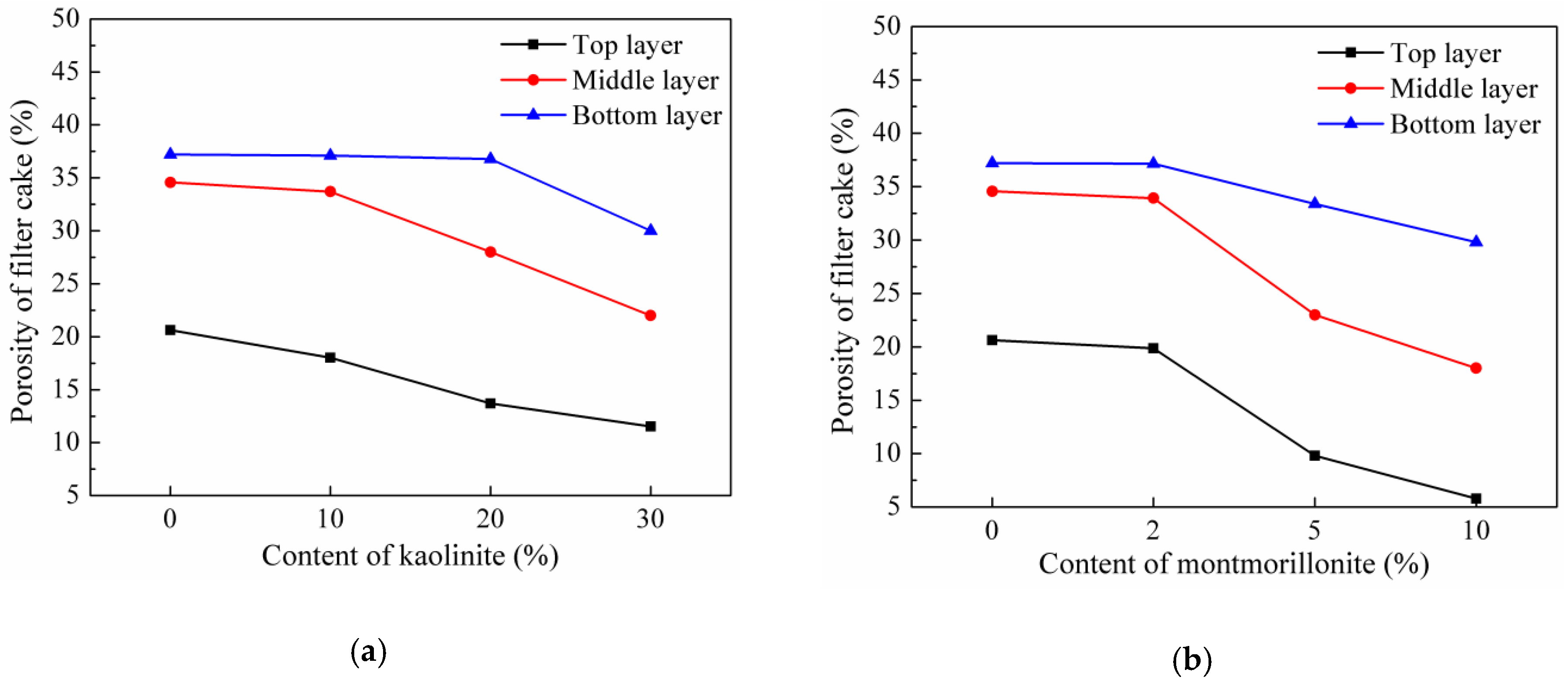
3.1.4. Porosity
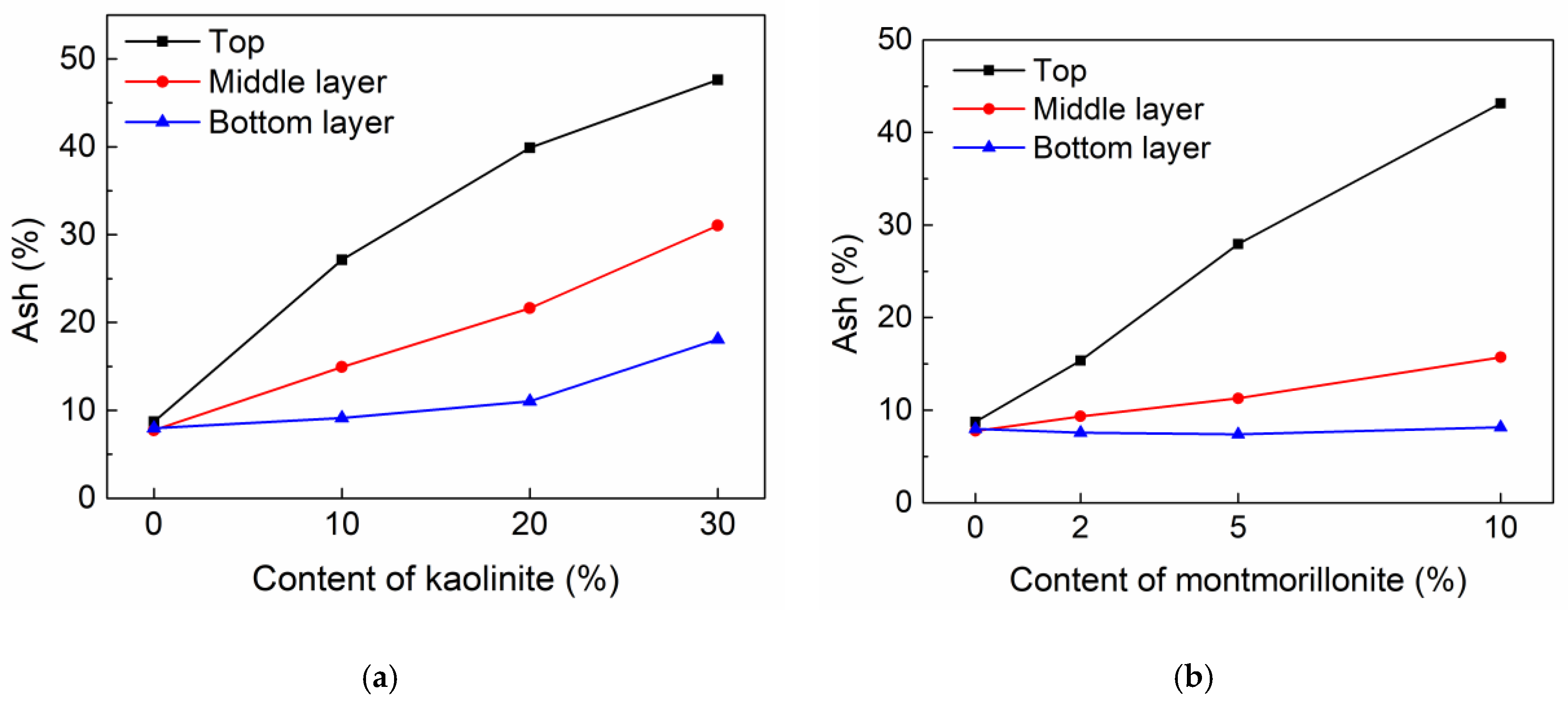
3.1.5. Distribution of Clay Minerals
3.1.6. Distribution of Water
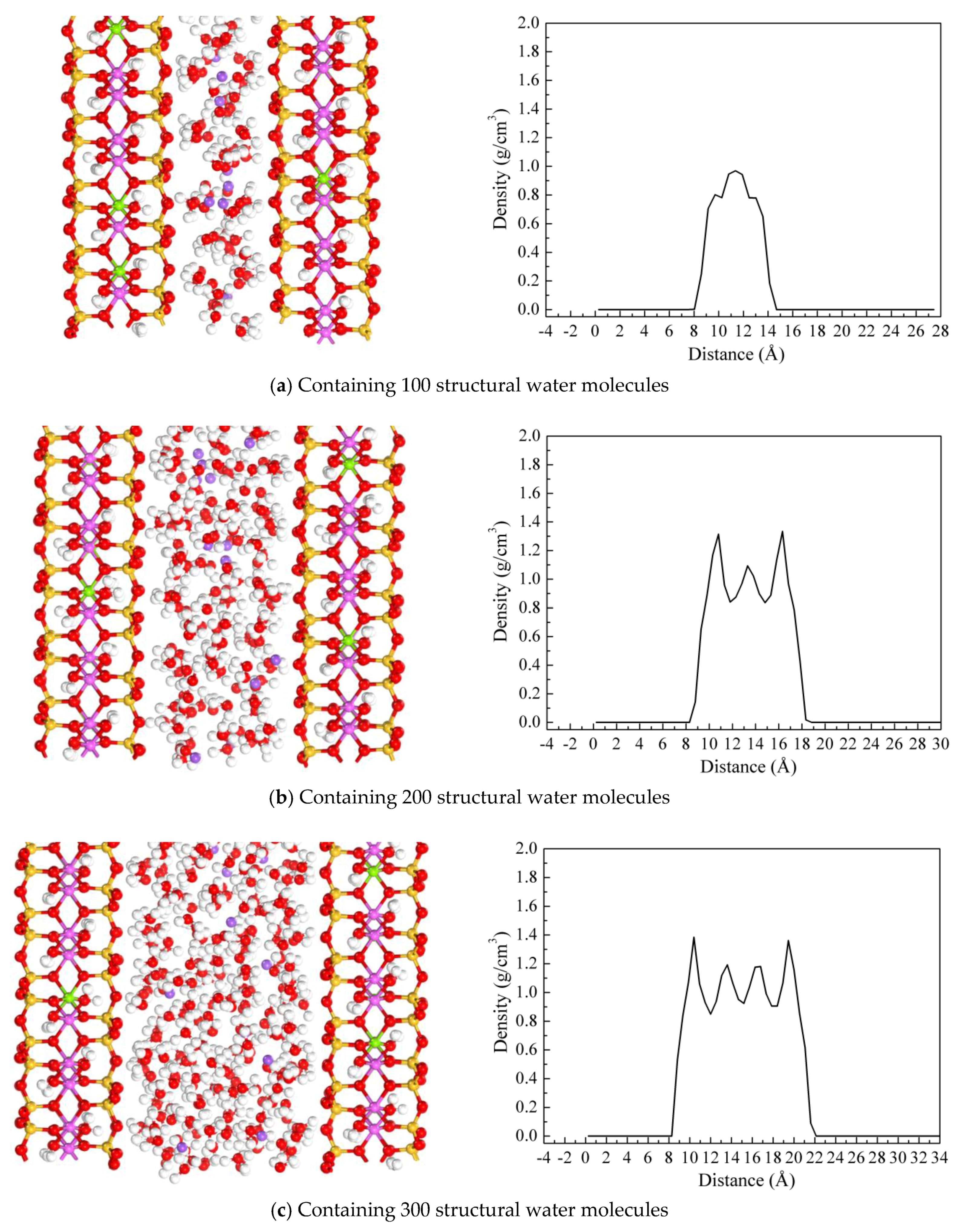
3.2. Molecular Simulations Results
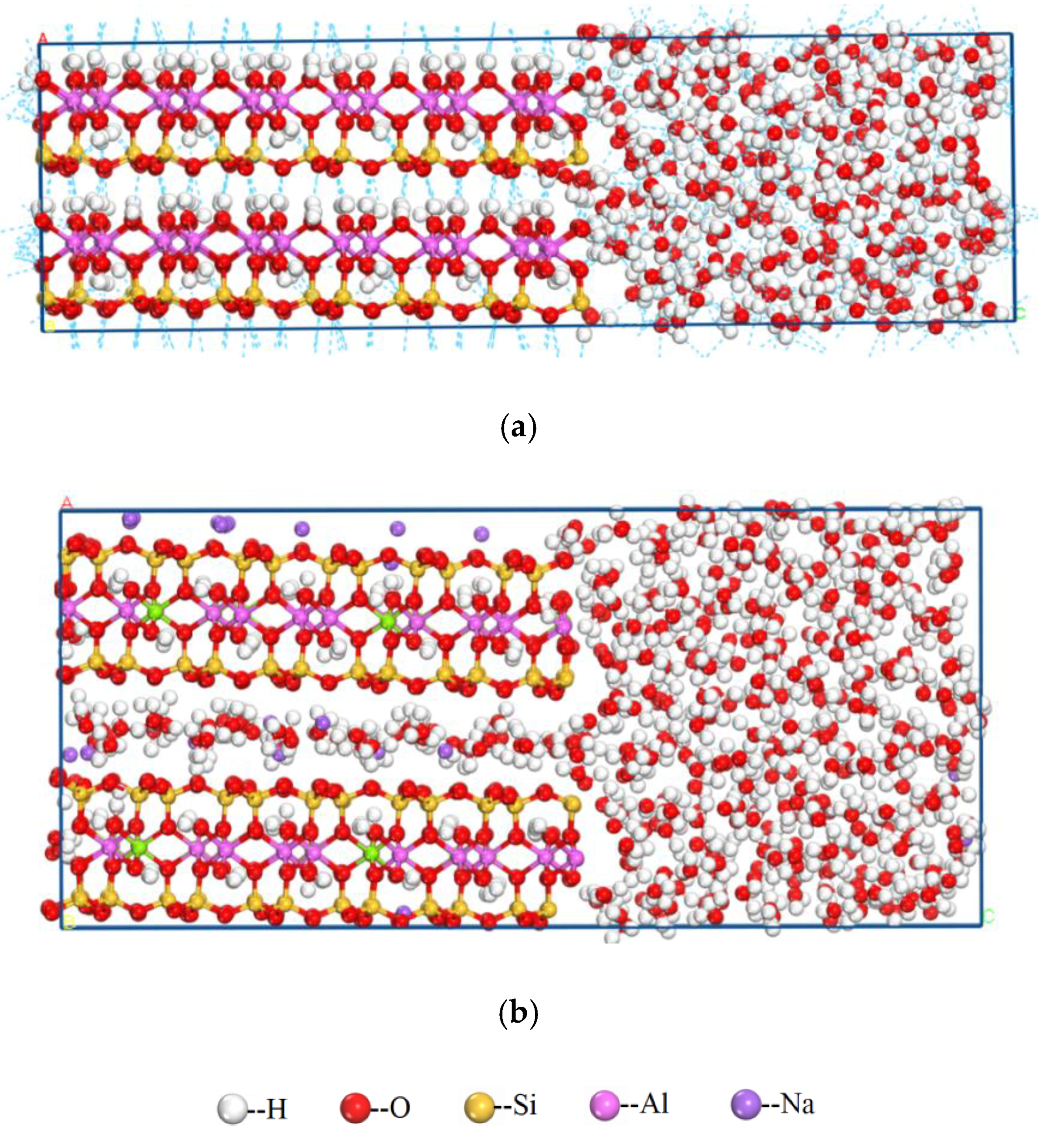
3.2.1. Adsorption of Water on the Side Surface of Kaolinite and Montmorillonite
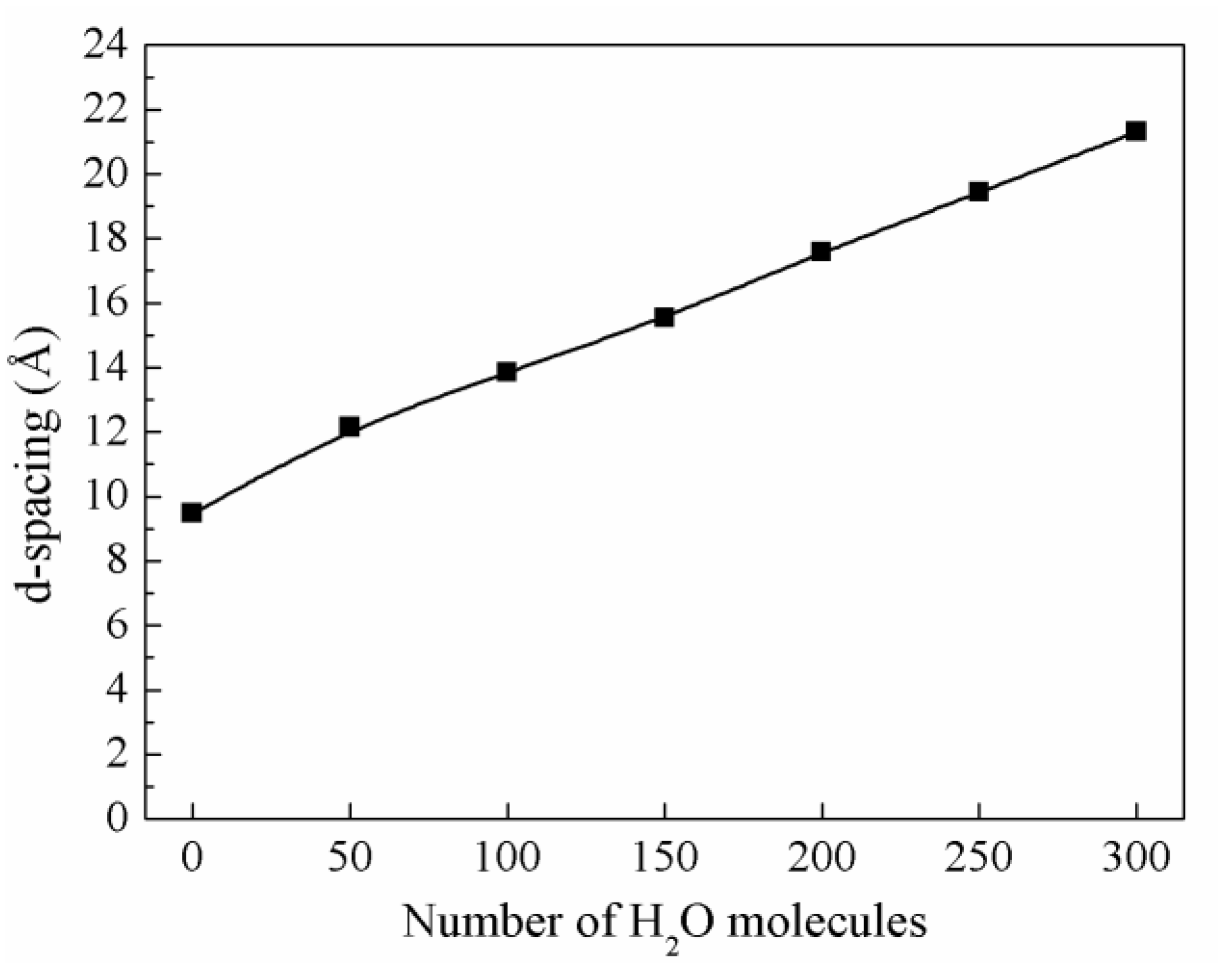
3.2.2. Swelling Curve of Montmorillonite
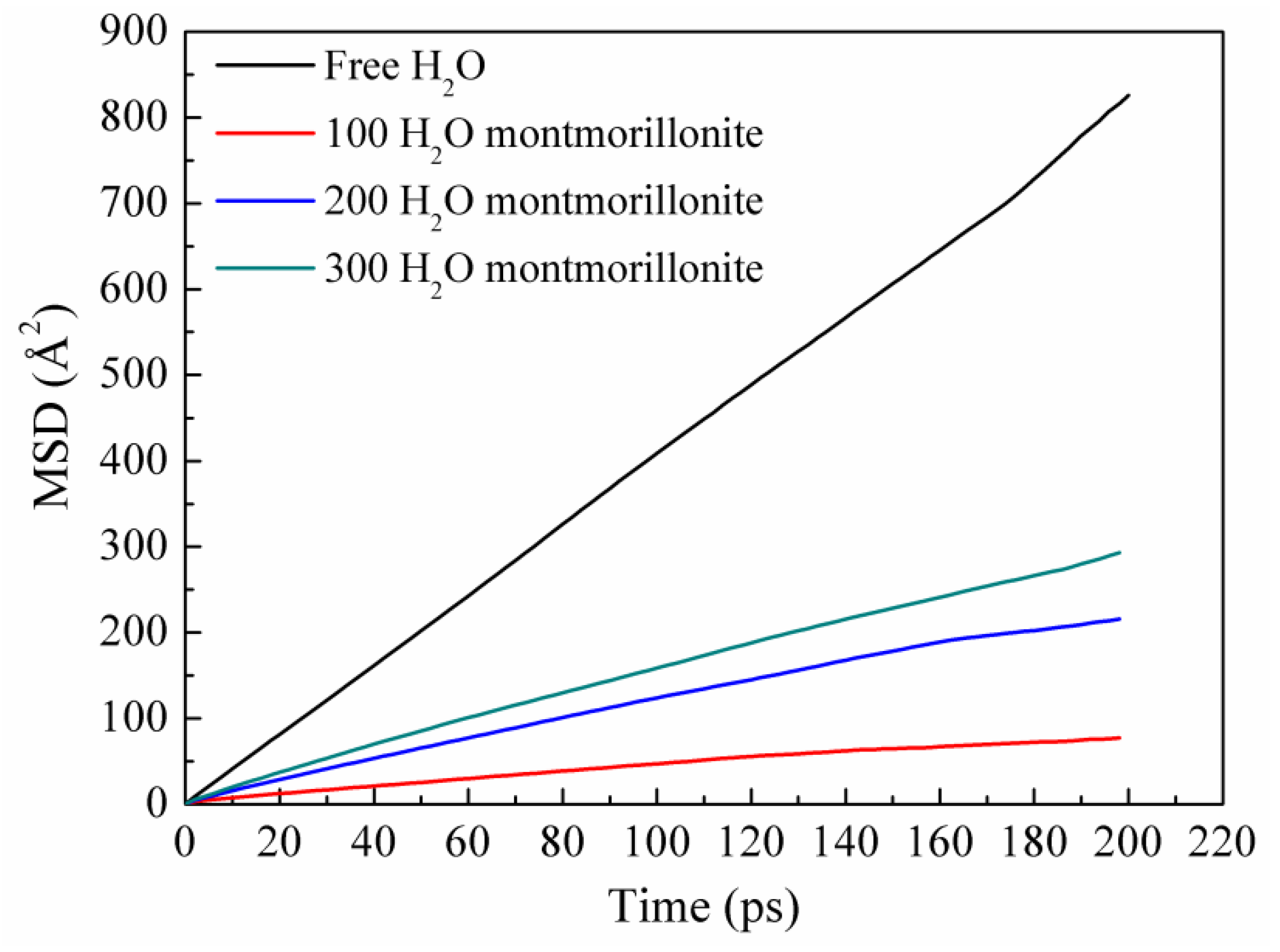
3.2.3. Diffusion Coefficient
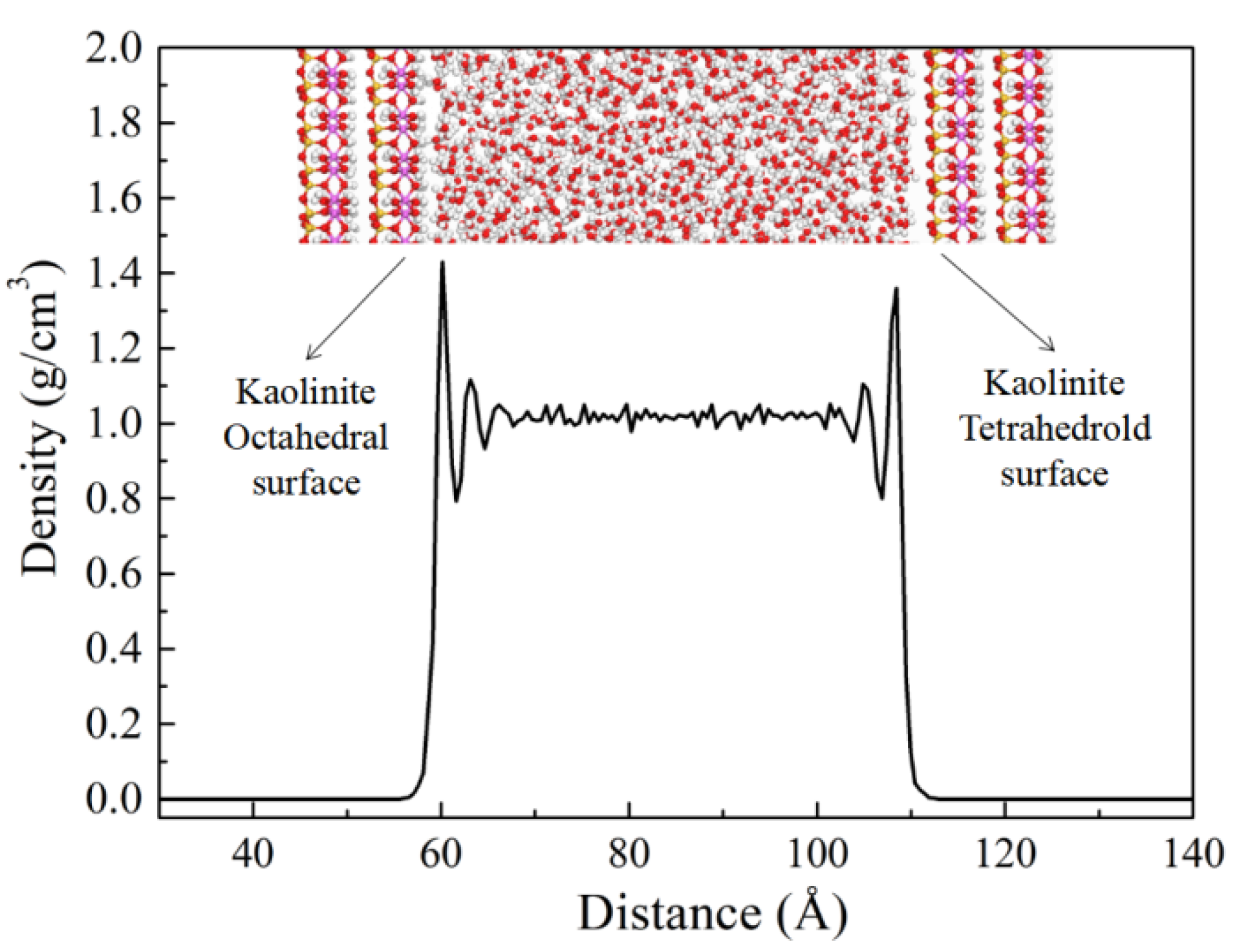
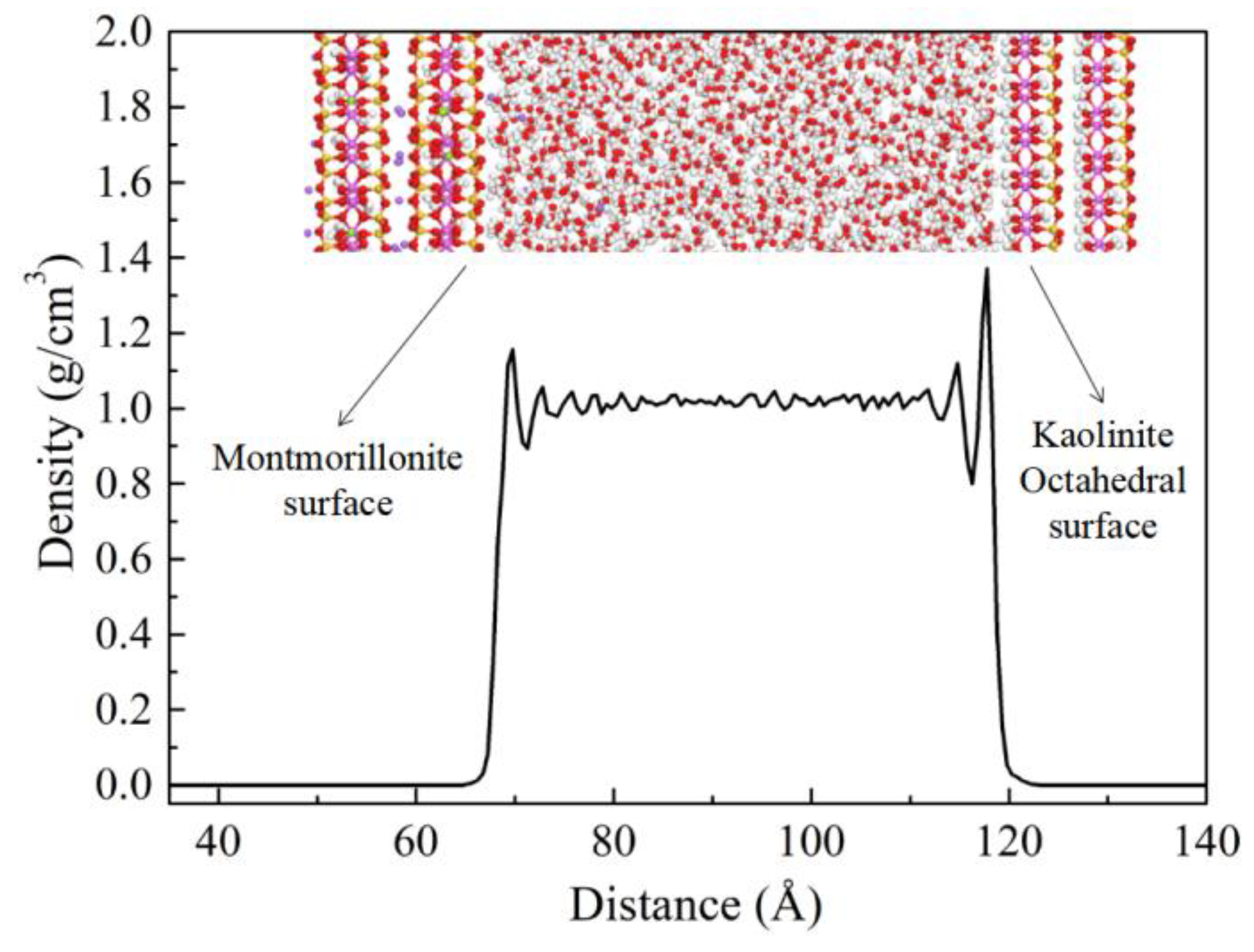
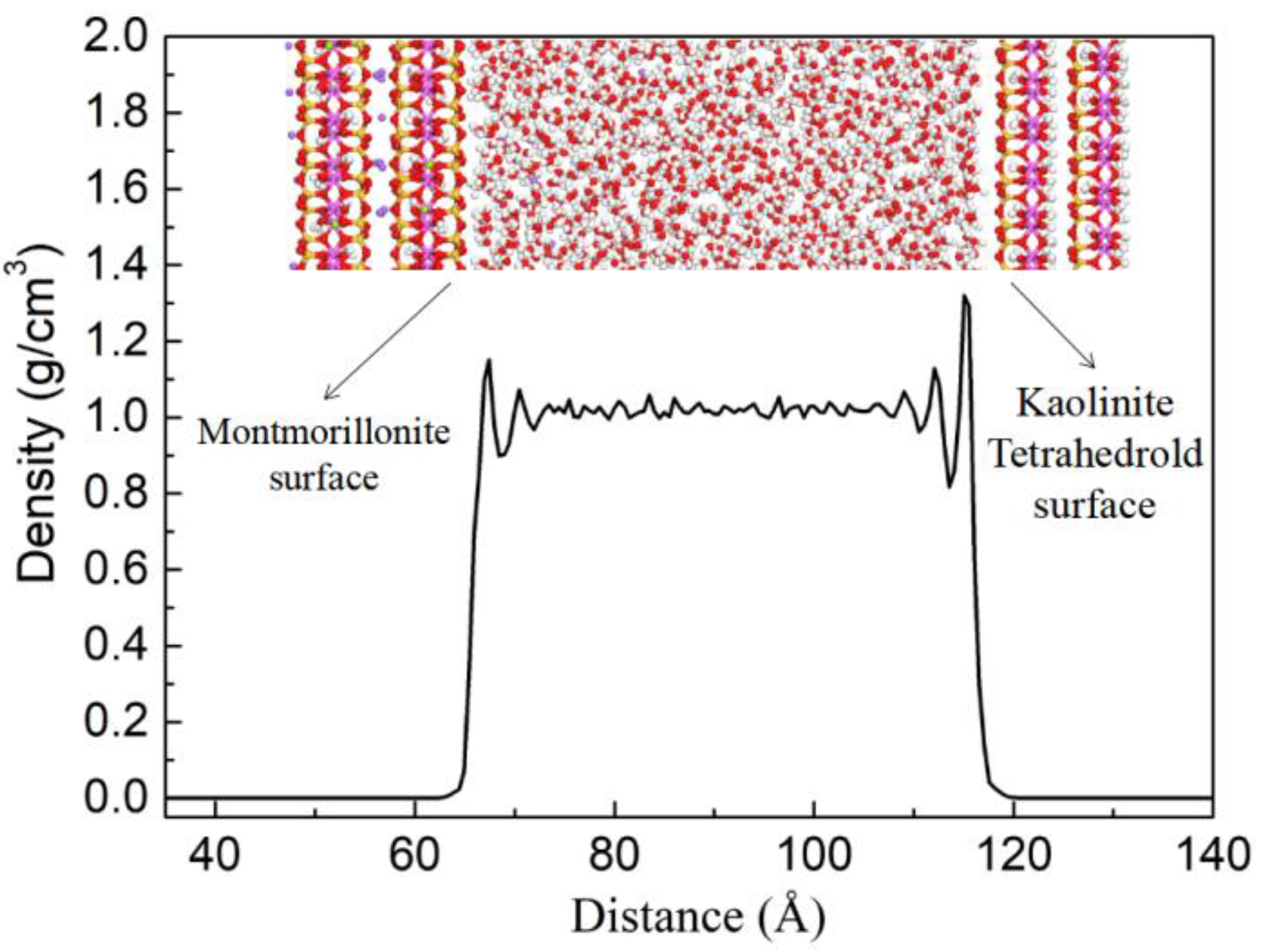
3.2.4. Adsorption of Water on the Surfaces of Kaolinite and Montmorillonite
4. Conclusions
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Devasahayam, S.; Ameen, M.A.; Verheyen, T.V.; Bandyopadhyay, S. Brown coal dewatering using poly (acrylamide-co-potassium acrylic) based super absorbent polymers. Minerals 2015, 5, 623–636. [Google Scholar] [CrossRef]
- Hogg, R. Flocculation and dewatering. Int. J. Miner. Process. 2000, 58, 223–236. [Google Scholar] [CrossRef]
- Kretser, D.R.; Scales, P.J.; Boger, D.V. Improving clay-based tailings disposal: Case study on coal tailings. AIChE J. 1997, 43, 1894–1903. [Google Scholar] [CrossRef]
- Sabah, E.; Yüzer, H.; Celik, M.S. Characterization and dewatering of fine coal tailings by dual-flocculant systems. Int. J. Miner. Process. 2004, 74, 303–315. [Google Scholar] [CrossRef]
- Wang, C.; Harbottle, D.; Liu, Q.X.; Xu, Z.H. Current state of fine mineral tailings treatment: A critical review on theory and practice. Miner. Eng. 2014, 58, 113–131. [Google Scholar] [CrossRef]
- Liu, H.B.; Liu, Z.L. Recycling utilization patterns of coal mining waste in China. Resour. Conserv. Recycl. 2010, 54, 1331–1340. [Google Scholar] [CrossRef]
- Jang, M.; Lee, H.J.; Shim, Y. Rapid removal of fine particles from mine water using sequential processes of coagulation and flocculation. Environ. Technol. 2010, 31, 423–432. [Google Scholar] [CrossRef] [PubMed]
- Salmachi, A.; Karacan, C.Ö. Cross-formational flow of water into coalbed methane reservoirs: Controls on relative permeability curve shape and production profile. Environ. Earth Sci. 2017, 76, 200. [Google Scholar] [CrossRef] [PubMed]
- Nghiem, L.D.; Ren, T.; Aziz, N.; Porter, I.; Regmi, G. Treatment of coal seam gas produced water for beneficial use in Australia: A review of best practices. Desalin. Water Treat. 2011, 32, 316–323. [Google Scholar] [CrossRef]
- Hamawand, I.; Yusaf, T.; Hamawand, S.G. Coal seam gas and associated water: A review paper. Renew. Sustain. Energy Rev. 2013, 22, 550–560. [Google Scholar] [CrossRef]
- Li, S.L.; Liao, Y.F.; Li, G.S.; Li, Z.; Cao, Y.J. Flocculating and dewatering performance of hydrophobic and hydrophilic solids using a thermal-sensitive copolymer. Water Sci. Technol. 2017, 76, 694–704. [Google Scholar] [CrossRef] [PubMed]
- Salmachi, A.; Talemi, P.; Tooski, Z.Y. Psyllium husk in water-based drilling fluids: An environmentally friendly viscosity and filtration agent. In Proceedings of the Abu Dhabi International Petroleum Exhibition & Conference. Society of Petroleum Engineers, Abu Dhabi, UAE, 7–10 November 2016. [Google Scholar] [CrossRef]
- Gao, Z.Y.; Li, C.W.; Sun, W.; Hu, Y.H. Anisotropic surface properties of calcite: A consideration of surface broken bonds. Colloids Surf. A. 2018, 520, 53–61. [Google Scholar] [CrossRef]
- Murray, H.H. Structure and composition of the clay minerals and their physical and chemical properties. Dev. Clay Sci. 2006, 2, 7–31. [Google Scholar] [CrossRef]
- Azeez, A.A.; Rhee, K.Y.; Park, S.J.; Hui, D. Epoxy clay nanocomposites—Processing, properties and applications: A review. Compos. Part B Eng. 2013, 45, 308–320. [Google Scholar] [CrossRef]
- Zhang, X.; Yi, H.; Zhao, Y.L.; Min, F.F.; Song, S.X. Study on the differences of Na-and Ca-montmorillonites in crystalline swelling regime through molecular dynamics simulation. Adv. Powder Technol. 2016, 27, 779–785. [Google Scholar] [CrossRef]
- Li, H.; Song, S.X.; Dong, X.S.; Min, F.; Zhao, Y.; Peng, C.; Nahmad, Y. Molecular dynamics study of crystalline swelling of montmorillonite as affected by interlayer cation hydration. JOM 2018, 70, 479–484. [Google Scholar] [CrossRef]
- Sun, L.; Hirvi, J.T.; Schatz, T.; Kasa, S.; Pakkanen, T.A. Estimation of montmorillonite swelling pressure: A molecular dynamics approach. J. Phys. Chem. C 2015, 119, 19863–19868. [Google Scholar] [CrossRef]
- Amarasinghe, P.M.; Katti, K.S.; Katti, D.R. Insight into role of clay-fluid molecular interactions on permeability and consolidation behavior of Na-montmorillonite swelling clay. J. Geotech. Geoenviron. Eng. 2011, 138, 138–146. [Google Scholar] [CrossRef]
- Mpofu, P.; Addai-Mensah, J.; Ralston, J. Interfacial chemistry, particle interactions and improved dewatering behaviour of smectite clay dispersions. Int. J. Miner. Process. 2005, 75, 155–171. [Google Scholar] [CrossRef]
- Foster, M.D. The relation between composition and swelling in clays. Clay Clay Min. 1954, 3, 205–220. [Google Scholar] [CrossRef]
- Segad, M.; Jonsson, B.; Åkesson, T.; Cabane, B. Ca/Na montmorillonite: Structure, forces and swelling properties. Langmuir 2010, 26, 5782–5790. [Google Scholar] [CrossRef] [PubMed]
- Segad, M.; Hanski, S.; Olsson, U.; Ruokolainen, J.; Åkesson, T.; Jonsson, B.G. Microstructural and swelling properties of Ca and Na montmorillonite:(in situ) observations with cryo-TEM and SAXS. J. Phys. Chem. C 2012, 116, 7596–7601. [Google Scholar] [CrossRef]
- McFarlane, A.; Bremmell, K.; Addai-Mensah, J. Improved dewatering behavior of clay minerals dispersions via interfacial chemistry and particle interactions optimization. J. Colloid Interface Sci. 2006, 293, 116–127. [Google Scholar] [CrossRef] [PubMed]
- Anderson, R.L.; Ratcliffe, I.; Greenwell, H.C.; Williams, P.A.; Cliffe, S.; Coveney, P.V. Clay swelling—A challenge in the oilfield. Earth-Sci. Rev. 2010, 98, 201–216. [Google Scholar] [CrossRef]
- Zhang, H.Y.; Kuang, Y.L.; Wang, G.H.; Ji, L. Soft sensor model for coal slurry ssh content based on image gray characteristics. Int. J. Coal Prep. Util. 2014, 34, 24–37. [Google Scholar] [CrossRef]
- Ofori, P.; Nguyen, A.V.; Firth, B.; McNally, C.; Ozdemir, O. Shear-induced floc structure changes for enhanced dewatering of coal preparation plant tailings. Chem. Eng. J. 2011, 172, 914–923. [Google Scholar] [CrossRef]
- Mukherjee, S. Environmental impacts of clay-related industries. In The Science of Clays; Springer: Dordrecht, The Netherlands, 2013; pp. 280–295. [Google Scholar]
- Rong, R.X.; Hitchins, J. Preliminary study of correlations between fine coal characteristics and properties and their dewatering behaviour. Miner. Eng. 1995, 8, 293–309. [Google Scholar] [CrossRef]
- Hussain, S.A.; Demirci, Ş.; Özbayoğlu, G. Zeta potential measurements on three clays from Turkey and effects of clays on coal flotation. J. Colloid Interface Sci. 1996, 184, 535–541. [Google Scholar] [CrossRef] [PubMed]
- Alam, N.; Ozdemir, O.; Hampton, M.A.; Nguyen, A.V. Dewatering of coal plant tailings: Flocculation followed by filtration. Fuel 2011, 90, 26–35. [Google Scholar] [CrossRef]
- Dong, X.S.; Hu, X.J.; Yao, S.L.; Peng, W.R.; Wang, Z.Z. Vacuum filter and direct current electro-osmosis dewatering of fine coal slurry. Procedia Earth Planet. Sci. 2009, 1, 685–693. [Google Scholar] [CrossRef]
- Dong, X.S.; Feng, L.H.; Yao, S.L.; Niu, D.F. Study on dewatering of fine coal by combination of electrolysis and filtration. Adv. Mater. Res. 2011, 236, 622–626. [Google Scholar] [CrossRef]
- Min, F.F.; Peng, C.L.; Liu, L.Y. Investigation on hydration layers of fine clay mineral particles in different electrolyte aqueous solutions. Powder Technol. 2015, 283, 368–372. [Google Scholar] [CrossRef]
- Gui, X.; Liu, J.; Cao, Y.; Miao, Z.; Li, S.; Xing, Y.; Wang, D. Coal preparation technology: Status and development in China. Energy Environ. 2015, 26, 997–1013. [Google Scholar] [CrossRef]
- Heinz, H.; Lin, T.J.; Mishra, R.K.; Emami, F.S. Thermodynamically consistent force fields for the assembly of inorganic, organic, and biological nanostructures: The interface force field. Langmuir 2013, 2, 1754–1765. [Google Scholar] [CrossRef] [PubMed]
- Fan, Y.P.; Dong, X.S.; Li, H. Dewatering effect of fine coal slurry and filter cake structure based on particle characteristics. Vacuum 2015, 114, 54–57. [Google Scholar] [CrossRef]
- Ruth, B.F. Correlating filtration theory with industrial practice. Ind. Eng. Chem. 1946, 38, 564–571. [Google Scholar] [CrossRef]
- Mota, M.; Teixeira, J.A.; Yelshin, A. Influence of cell-shape on the cake resistance in dead-end and cross-flow filtrations. Sep. Purif. Technol. 2002, 27, 137–144. [Google Scholar] [CrossRef]
- Gamba, M.; Kovář, P.; Pospíšil, M.; Sánchez, R.M.T. Insight into thiabendazole interaction with montmorillonite and organically modified montmorillonites. Appl. Clay Sci. 2017, 137, 59–68. [Google Scholar] [CrossRef]
- Bragg, W.H.; Bragg, W.L. The X-Ray spectrometer. Nature 1914, 94, 199–200. [Google Scholar] [CrossRef]
- Schoen, M.; Hoheisel, C. The mutual diffusion coefficient D12 in binary liquid model mixtures. Molecular dynamics calculations based on Lennard-Jones (12-6) potentials. Mol. Phys. 1984, 52, 1029–1042. [Google Scholar] [CrossRef]
- Chang, I.L.; Chu, C.P.; Lee, D.J.; Huang, C.P. Expression dewatering of alum-coagulated clay slurries. Environ. Sci. Technol. 1997, 31. [Google Scholar] [CrossRef]
- Zhang, Y.; Gong, G.Q.; Wu, G.G.; Wang, Y.L. Physical properties and filter cake structure of fine clean coal from flotation. Int. J. Min. Sci. Technol. 2014, 24, 281–284. [Google Scholar] [CrossRef]
- Mpofu, P.; Addai-Mensah, J.; Ralston, J. Flocculation and dewatering behaviour of smectite dispersions: Effect of polymer structure type. Miner. Eng. 2004, 17, 411–423. [Google Scholar] [CrossRef]
- Teich-McGoldrick, S.L.; Greathouse, J.A.; Jove-Colon, C.F.; Cygan, R.T. Swelling properties of montmorillonite and beidellite clay minerals from molecular simulation: Comparison of temperature, interlayer cation, and charge location effects. J. Phys. Chem. C 2015, 119, 20880–20891. [Google Scholar] [CrossRef]
- Vao-soongnern, V.; Pipatpanukul, C.; Horpibulsuk, S. A combined X-ray absorption spectroscopy and molecular dynamic simulation to study the local structure potassium ion in hydrated montmorillonite. J. Mater. Sci. 2015, 50, 7126–7136. [Google Scholar] [CrossRef]
- Croteau, T.; Bertram, A.K.; Patey, G.N. Simulation of water adsorption on kaolinite under atmospheric conditions. J. Phys. Chem. A 2009, 113, 7826–7833. [Google Scholar] [CrossRef] [PubMed]




| Sample | Moisture (%) | Ash (%) | Volatile Matter (%) | Fixed Carbon (%) | Density (g/cm3) |
|---|---|---|---|---|---|
| Coal | 1.21 | 14.59 | 12.22 | 71.98 | 1.42 |
| Size Classes (mm) | Mass Fraction (%) | Ash Content (%) | Accumulation | |
|---|---|---|---|---|
| Mass Distribution (%) | Ash Content (%) | |||
| 0.25–0.5 | 6.56 | 12.64 | 6.45 | 12.64 |
| 0.125–0.25 | 15.76 | 11.34 | 22.32 | 11.72 |
| 0.075–0.125 | 22.68 | 13.53 | 45.01 | 12.63 |
| 0.045–0.075 | 13.32 | 13.79 | 58.32 | 12.90 |
| −0.045 | 41.68 | 17.02 | 100.00 | 14.62 |
| Total | 100.00 | 14.62 | ||
| Slurry Sample | Components of Solid in Slurry (Mass Fraction) | Average Specific Resistance (m/kg) |
|---|---|---|
| 1 | 100% coal | 1.98 × 107 |
| 2 | 90% coal + 10% kaolinite | 2.35 × 108 |
| 3 | 80% coal + 20% kaolinite | 3.68 × 108 |
| 4 | 70% coal + 30% kaolinite | 4.51 × 108 |
| 5 | 98% coal + 2% montmorillonite | 9.80 × 107 |
| 6 | 95% coal + 5% montmorillonite | 4.28 × 109 |
| 7 | 90% coal + 10% montmorillonite | 8.05 × 109 |
| Conditions | Diffusion Coefficient of Water (10−5 cm2/s) |
|---|---|
| Free water molecules | 6.77 |
| 300 structural water molecules | 2.40 |
| 200 structural water molecules | 1.83 |
| 100 structural water molecules | 0.63 |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Ma, X.; Fan, Y.; Dong, X.; Chen, R.; Li, H.; Sun, D.; Yao, S. Impact of Clay Minerals on the Dewatering of Coal Slurry: An Experimental and Molecular-Simulation Study. Minerals 2018, 8, 400. https://doi.org/10.3390/min8090400
Ma X, Fan Y, Dong X, Chen R, Li H, Sun D, Yao S. Impact of Clay Minerals on the Dewatering of Coal Slurry: An Experimental and Molecular-Simulation Study. Minerals. 2018; 8(9):400. https://doi.org/10.3390/min8090400
Chicago/Turabian StyleMa, Xiaomin, Yuping Fan, Xianshu Dong, Ruxia Chen, Hongliang Li, Dong Sun, and Suling Yao. 2018. "Impact of Clay Minerals on the Dewatering of Coal Slurry: An Experimental and Molecular-Simulation Study" Minerals 8, no. 9: 400. https://doi.org/10.3390/min8090400
APA StyleMa, X., Fan, Y., Dong, X., Chen, R., Li, H., Sun, D., & Yao, S. (2018). Impact of Clay Minerals on the Dewatering of Coal Slurry: An Experimental and Molecular-Simulation Study. Minerals, 8(9), 400. https://doi.org/10.3390/min8090400






