Induration Process of MgO Flux Pellet
Abstract
:1. Introduction
2. Materials and Methods
2.1. Raw Materials
2.2. Experimental Methods
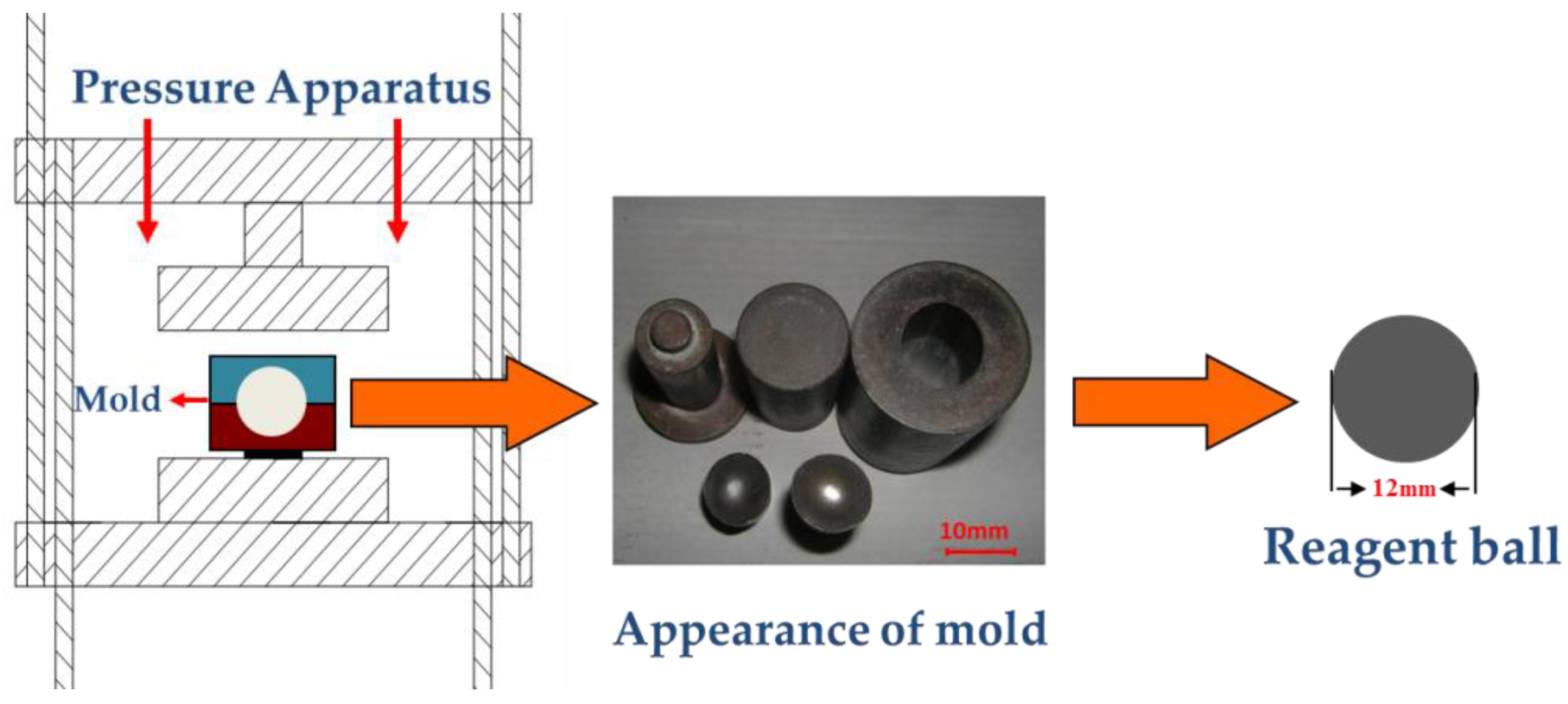
2.2.1. Oxidation Process of Fe3O4
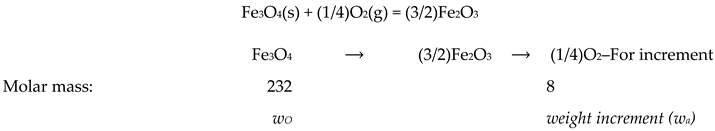
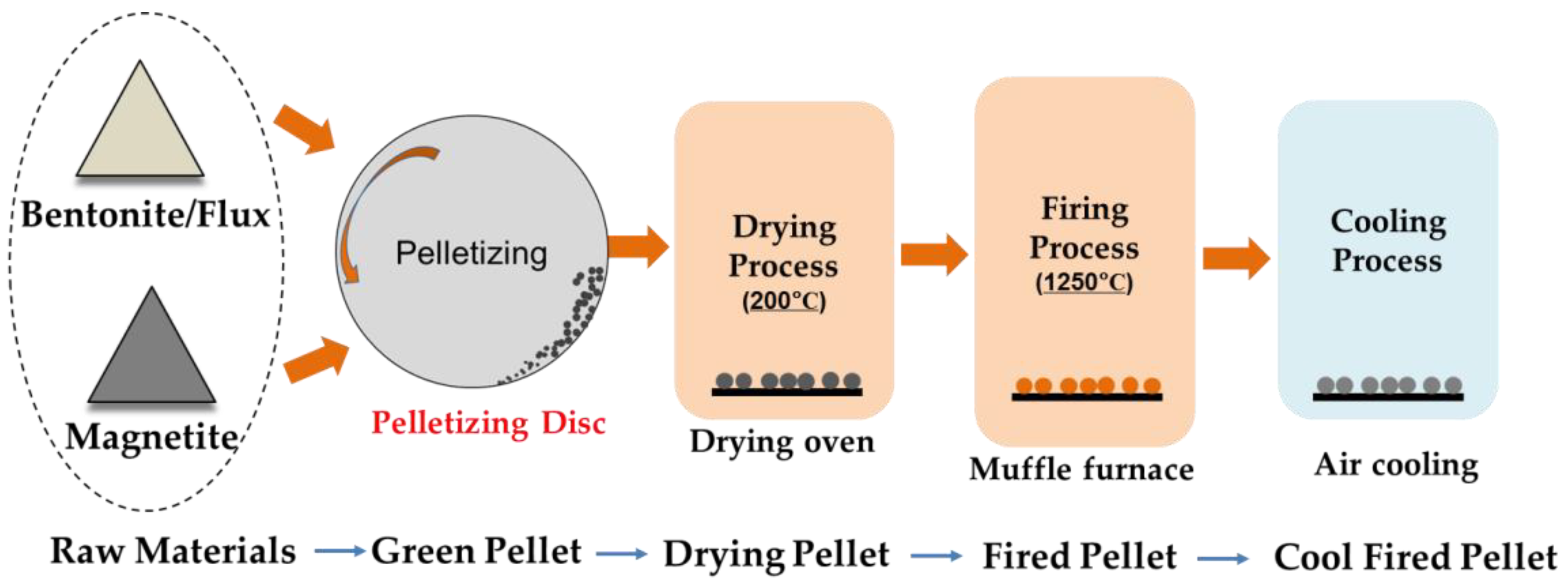
2.2.2. Preparing Pellet with Magnetite Iron Ore
- (a)
- Producing green pellet. The main parameters in this step include, 8.0 ± 0.5% of moisture content in mixed raw materials, 30 min of pelletizing time, and 12.0 mm of size for the green pellet;
- (b)
- Drying of the green pellet. The green pellet is dried at 200 °C for 3 h in a drying oven (H-201, Kexiang Instrument Equipment, Anshan, China);
- (c)
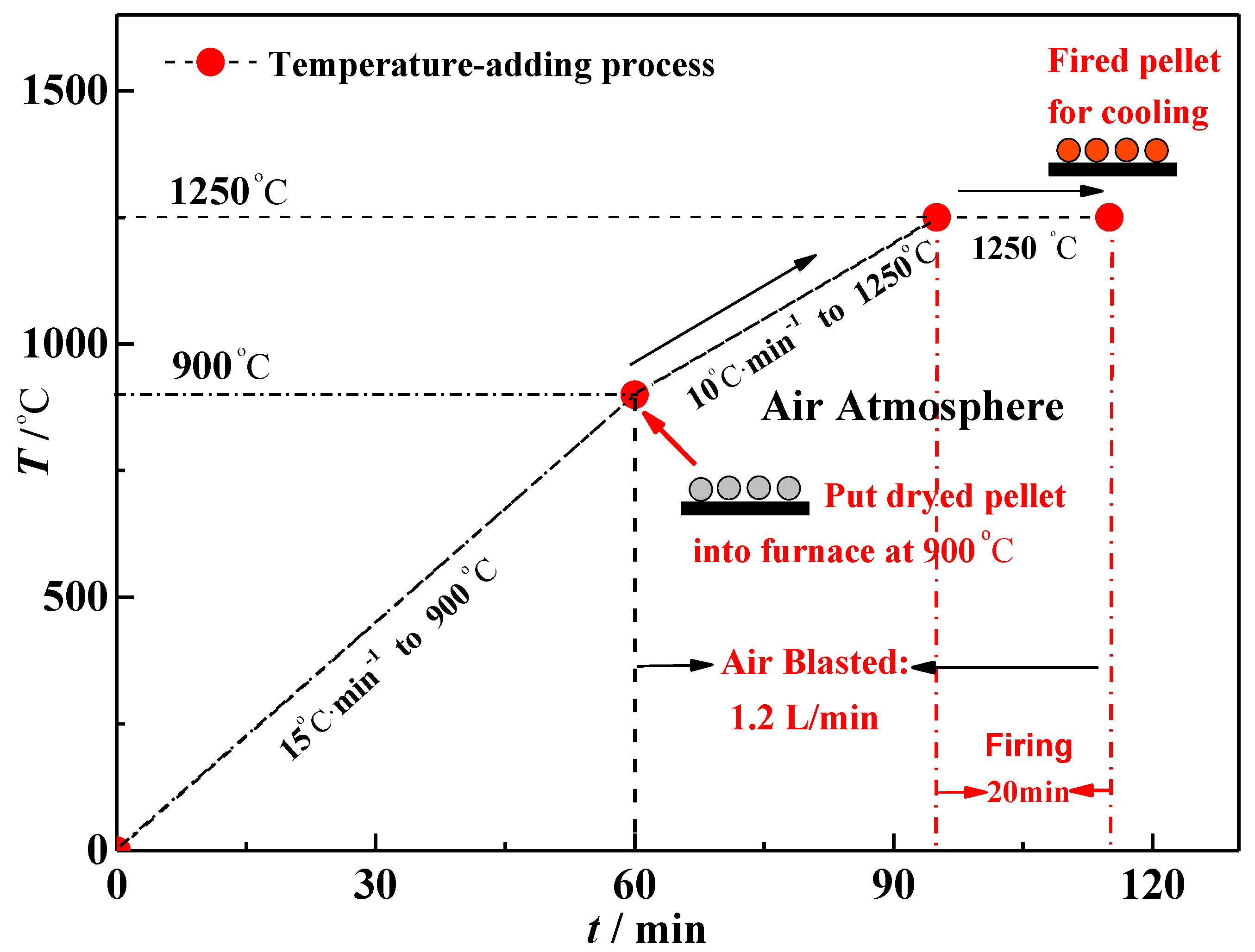
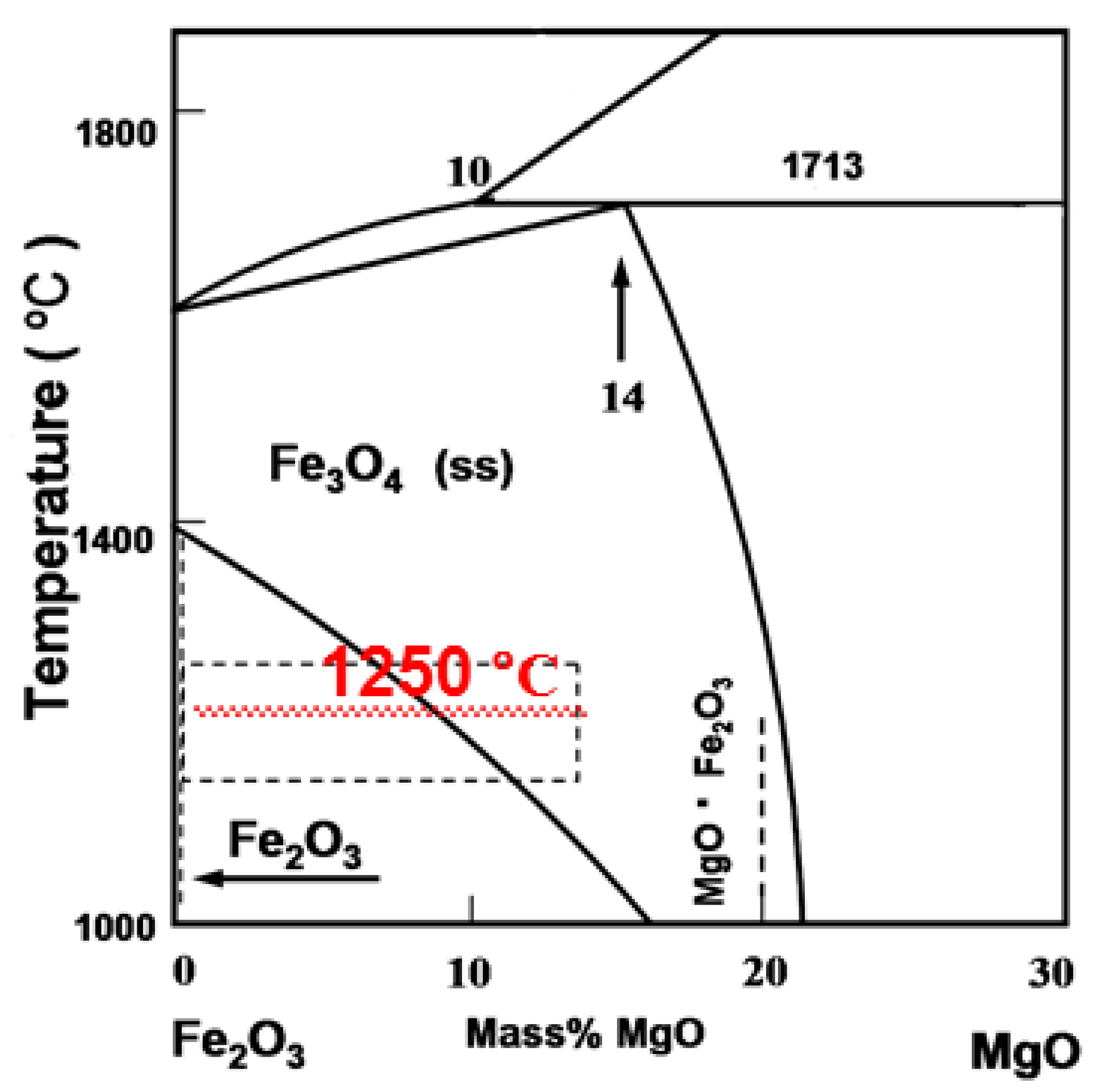
- Firing of the green pellet. In order to physically simulate the firing process of grate-rotary kiln in pellet plants, the dried pellet is put into a preheated muffle furnace (MF400, Great Wall Electronic Furnace, Shenyang, China) at 900 °C, and then the temperature of furnace is increased to 1250 °C and finally maintains at 1250 °C for 20 min. In addition, the air is blasted into the furnace in 1.2 L/min after the dried pellet is put in the furnace. The details of firing process are shown in Figure 5;
- (d)
- Cooling of the fired pellet. Following the process of firing, the pellet is air-cooled to ambient temperature; the cooled pellet is so-called fired pellet;
- (e)
- Compressive strength (CS) testing. The CS is detected according to the ISO4700 standard [19]; the general method is this: 64 selected pellets with diameter of 10–12.5 mm are tested in a compressive tester, the two maximum values and two minimum values are deleted; the average of the remaining 60 values is regarded as the final CS.
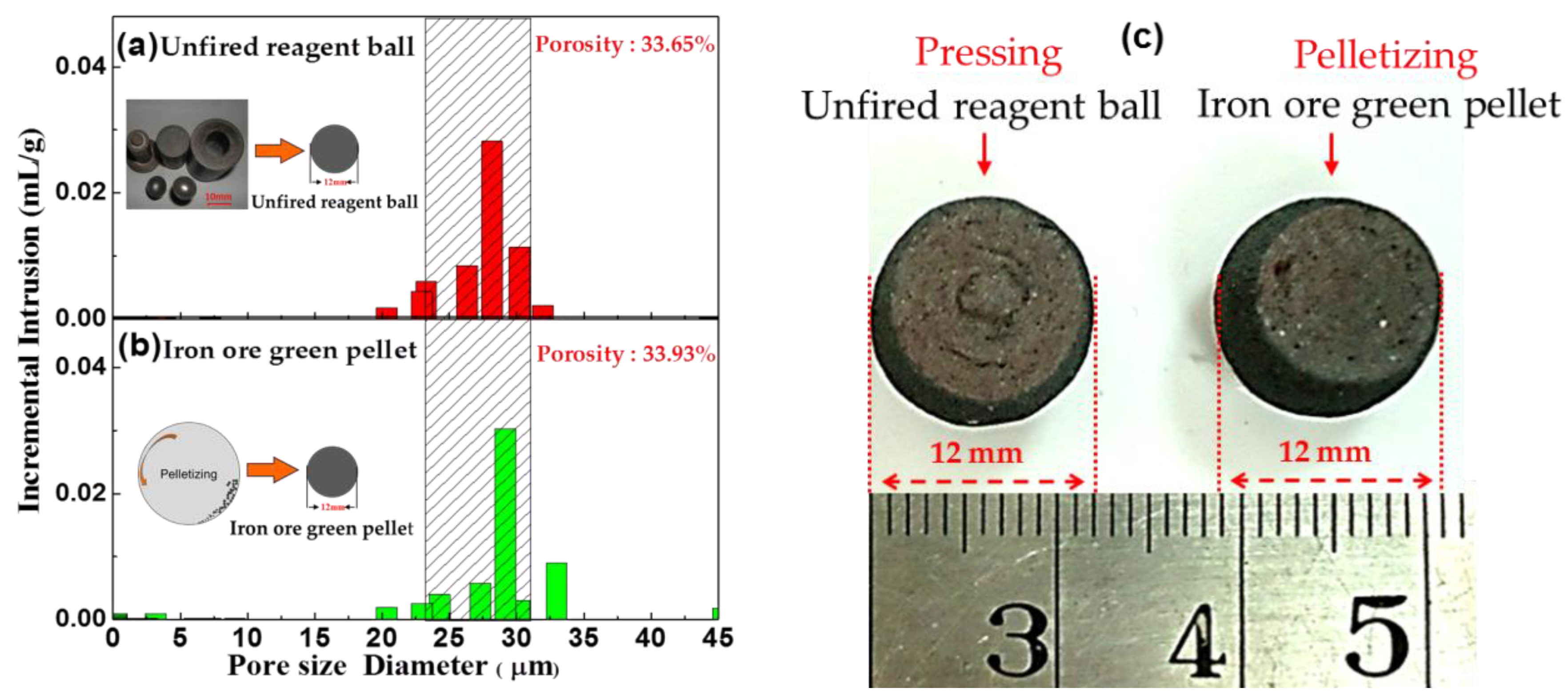
2.2.3. Densification Process of Pellet
2.2.4. Reproducible Experiment
3. Results
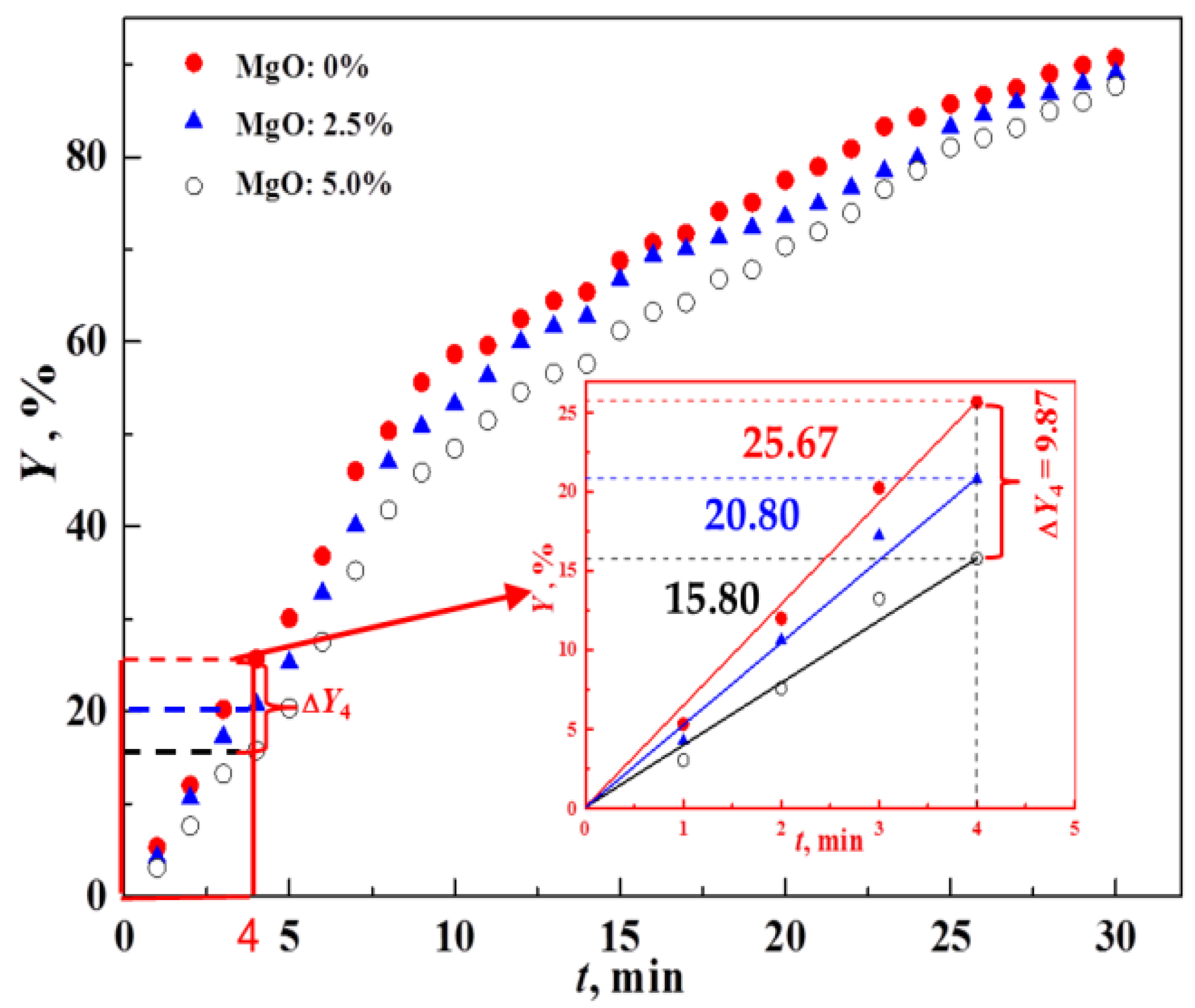
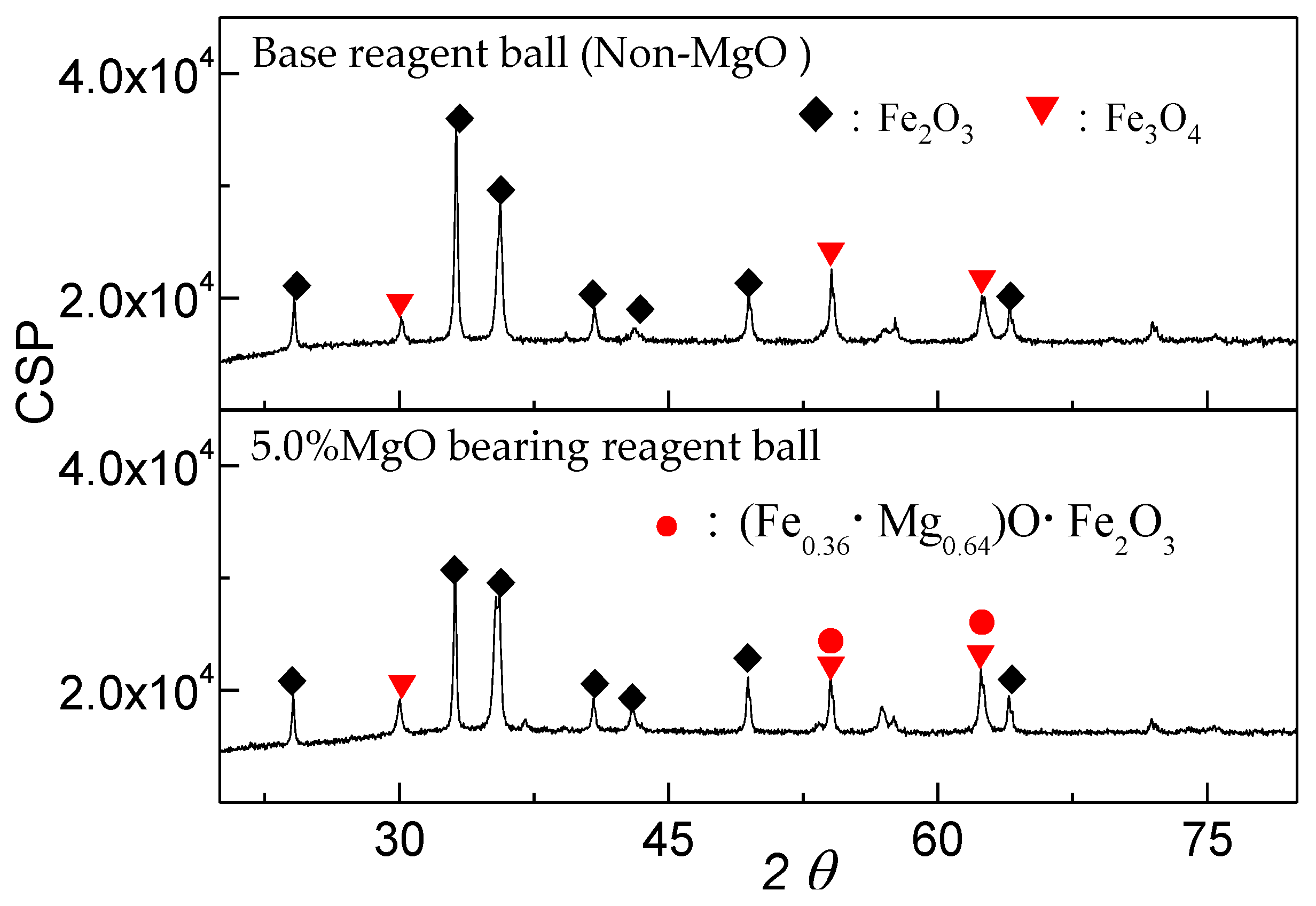
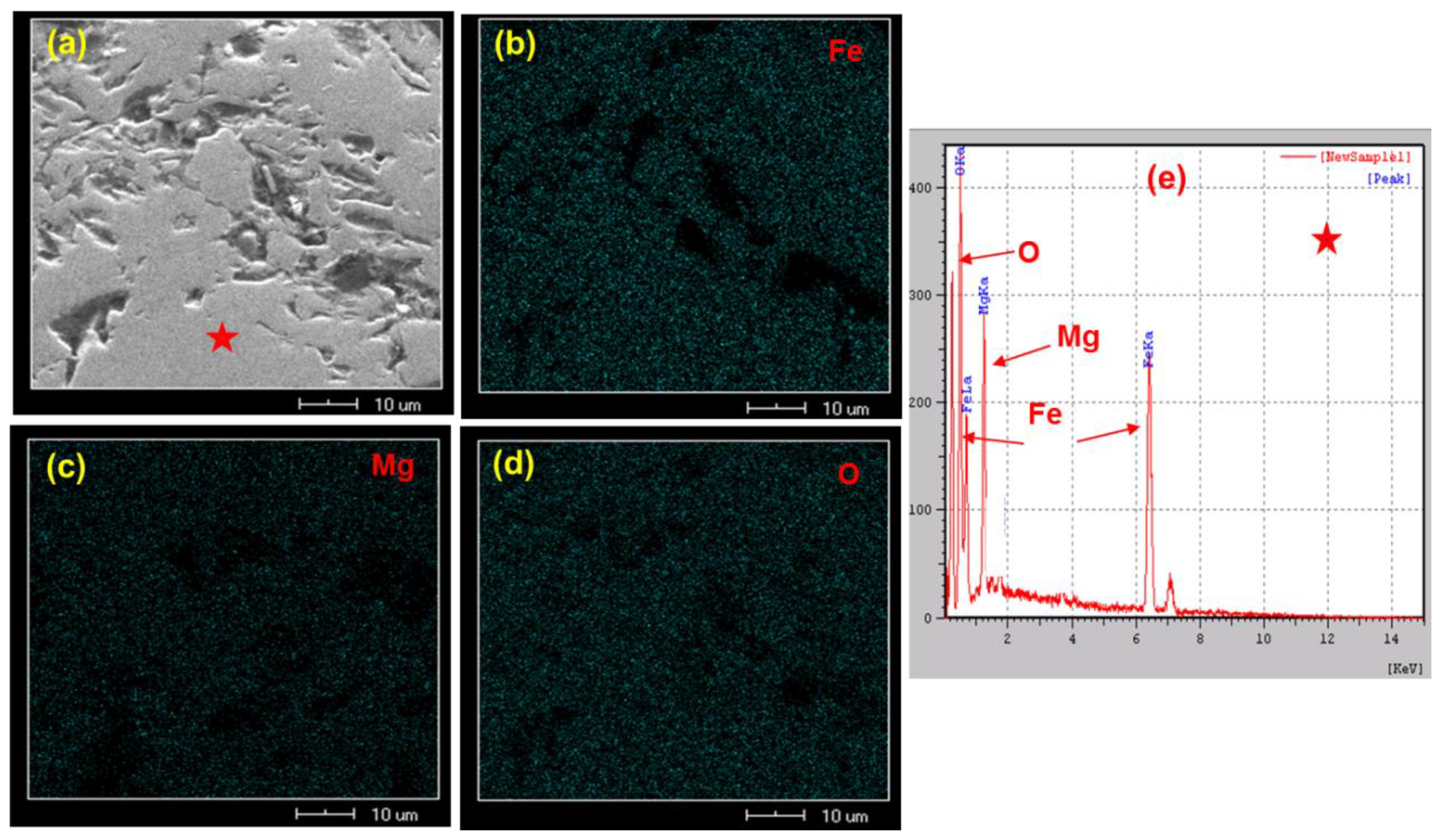
3.1. Effect of MgO on the Oxidation Process of Fe3O4
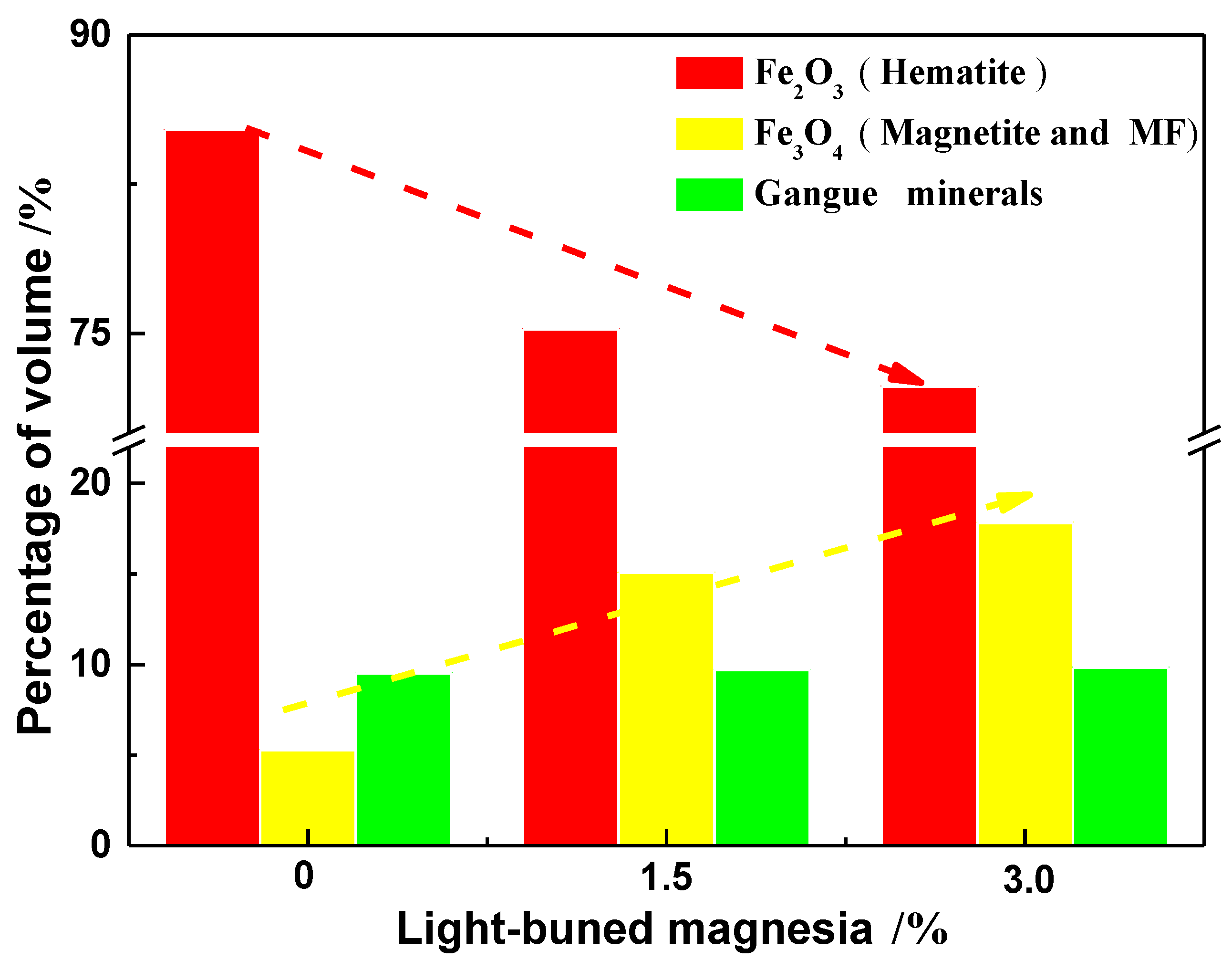
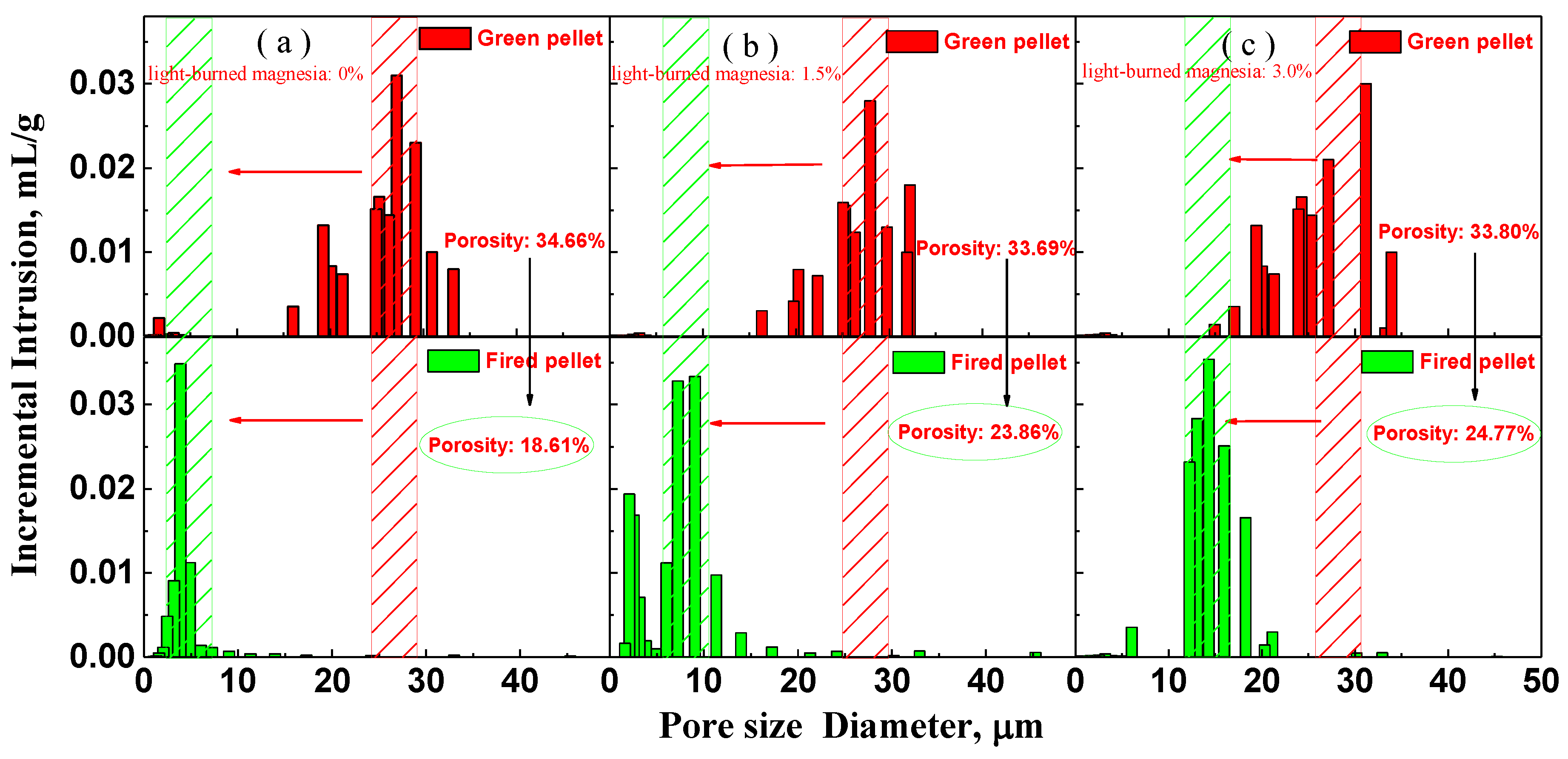
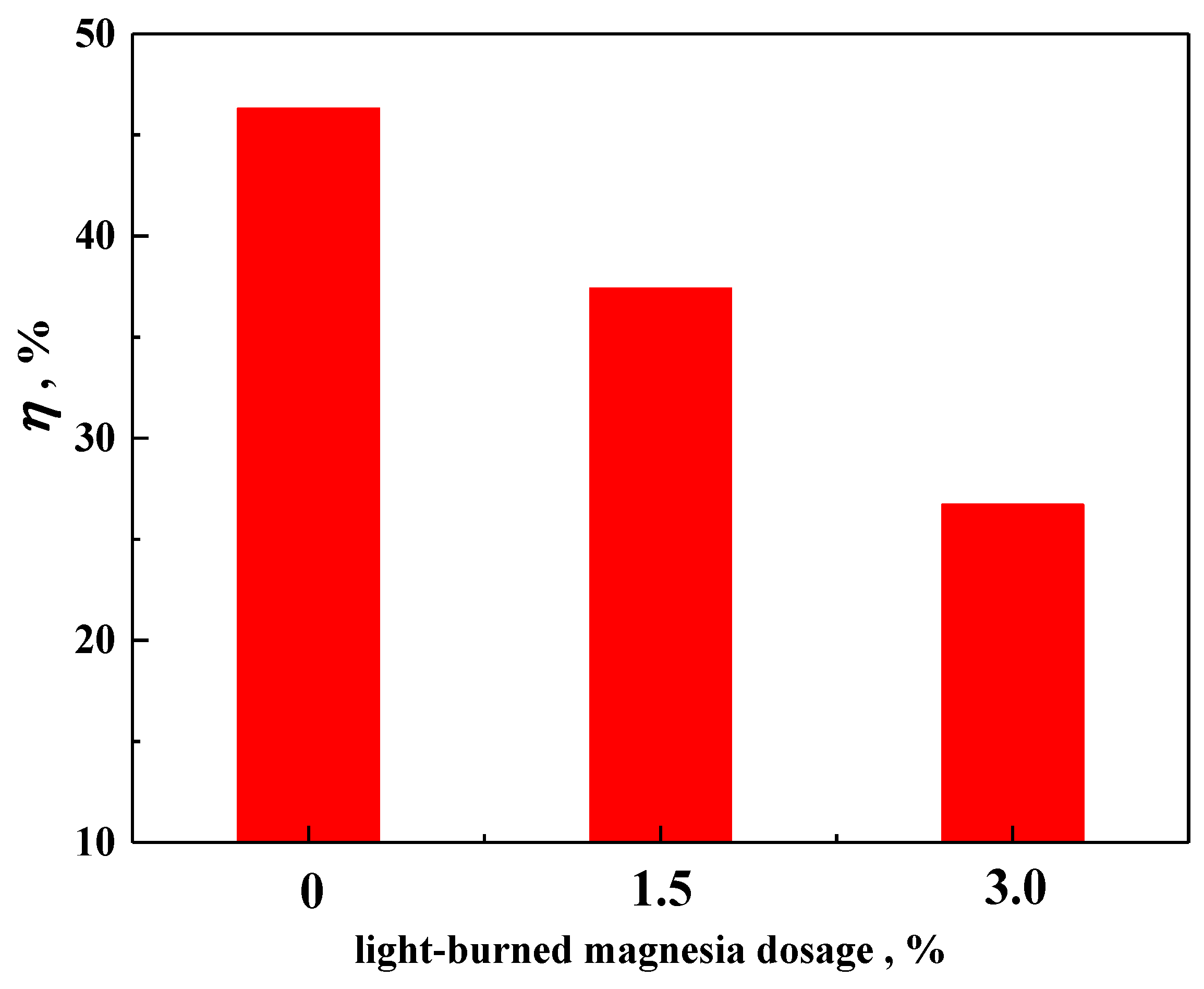
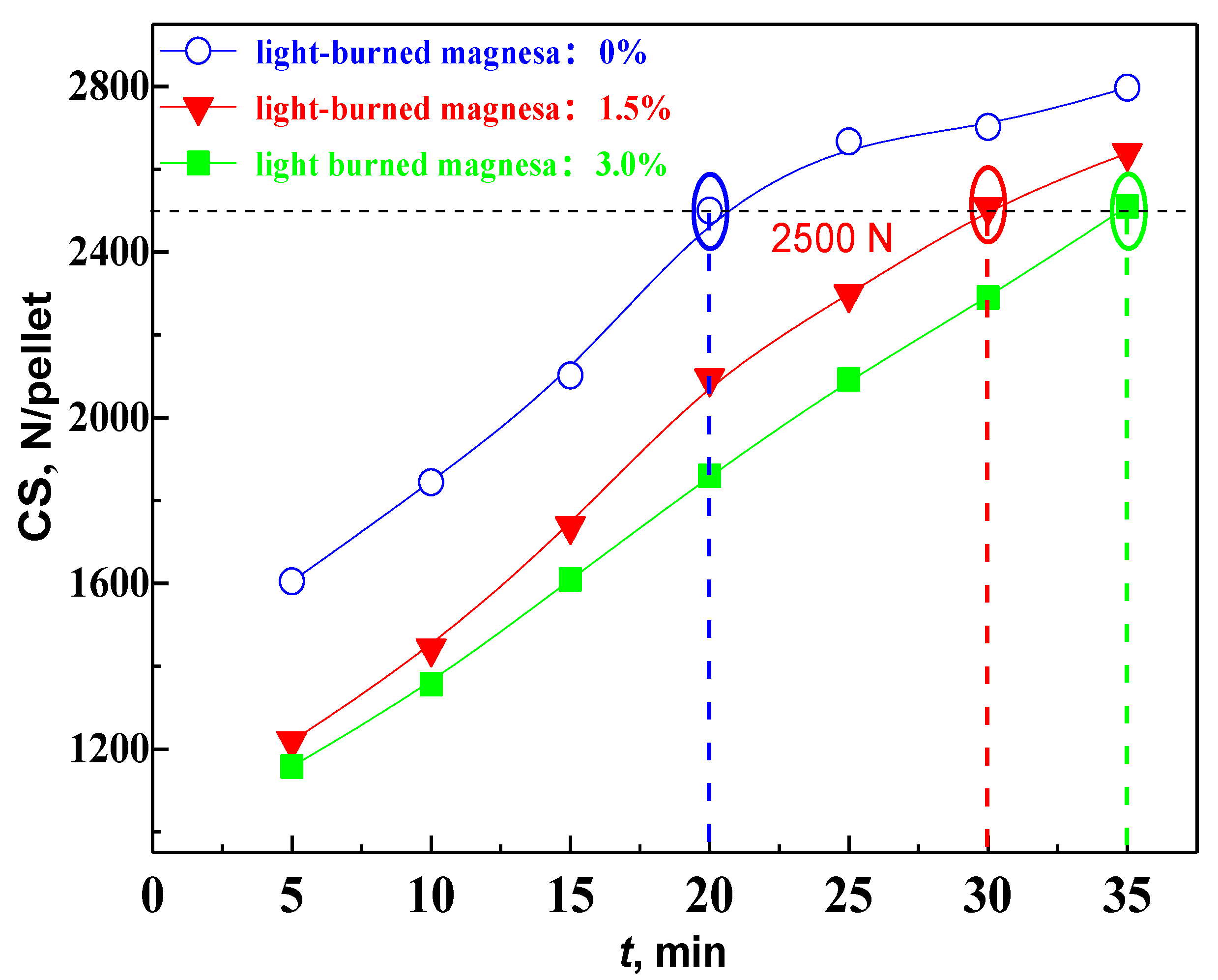
3.2. Effect of MgO on the Densification Process of Pellet
4. Analysis and Discussion
5. Conclusions
- (1)
- MgO is found to adversely affect the oxidation process of Fe3O4. The oxidation rate of Fe3O4 for the MgO bearing reagent ball (w(Fe3O4)/w(MgO) = 95.0%/5.0%) is slower than that for the base reagent ball (w(Fe3O4) = 100%).
- (2)
- Based on the unreacted core model, the relationship between the oxidation ratio of Fe3O4(Y) and time (t) was proposed. The calculated results coincide well with the experimental data according to the verification experiments. The unreacted core model is therefore suitable to discuss the oxidation of Fe3O4 in pellet induration process.
- (3)
- The densification of the pellet is limited after the addition of MgO. The oxide densification index (ODI) of MgO flux pellet is lower compared with that of Non-MgO flux pellet, which makes CS of the MgO flux pellet maintain at a lower level.
- (4)
- MgO negatively affects pellet induration and then restrains the pellet strength. However, prolonging the fired time and adjusting the MgO dosage are available to overcome these drawbacks. Therefore, the MgO flux pellet will be a prospective candidate for ironmaking units if an appropriate balance can be made between the merits and drawbacks of MgO flux pellet.
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
References
- Nabeel, M.; Karasev, A.; Jönsson, P.G. Evaluation of dust generation during mechanical wear of iron ore pellets. ISIJ Int. 2016, 56, 960–966. [Google Scholar] [CrossRef]
- Fu, J.; Zhu, D. Basic Principles, Techniques and Equipment of the Iron Ore Oxidized Pellets; Central South University Press: Changsha, China, 2005; pp. 323–336. ISBN 9787811050516. [Google Scholar]
- Nishimura, T.; Higuchi, K.; Naito, M.; Kunitomo, K. Evaluation of softening, shrinking and melting reduction behavior of raw materials for blast furnace. ISIJ Int. 2011, 51, 1316–1321. [Google Scholar] [CrossRef]
- Iljana, M.; Kemppainen, A.; Paananen, T.; Mattila, O.; Heikkinen, E.P.; Fabritius, T. Evaluating the reduction-softening behaviour of blast furnace burden with an advanced Test. ISIJ Int. 2016, 56, 1705–1714. [Google Scholar] [CrossRef]
- Jiang, X.; Li, X.; Shen, F. Study on improving the softening-melting properties of MgO bearing iron ores. J. Northeast. Univ. Nat. Sci. 2007, 28, 365–369. [Google Scholar]
- Gao, Q.; Shen, F.; Wei, G.; Jiang, X. Gas-solid reduction kinetic model of MgO-fluxed pellets. Int. J. Miner. Metall. Mater. 2014, 21, 12–17. [Google Scholar] [CrossRef]
- Kemppainen, A.; Mattila, O.; Heikkinen, E.; Paananen, T.; Fabritius, T. Effect of H2-H2O on the reduction of olivine pellets in CO-CO2 Gas. ISIJ Int. 2012, 52, 1973–1978. [Google Scholar] [CrossRef]
- Matsumura, M.; Hoshi, M.; Kawaguchi, T. Improve of sinter softening property and reducibility by controlling chemical compositions. ISIJ Int. 2005, 45, 598–607. [Google Scholar] [CrossRef]
- Shen, F.; Gao, Q.; Wei, G.; Shen, Y. Densification process of MgO bearing pellets. Steel Res. Int. 2015, 86, 644–650. [Google Scholar] [CrossRef]
- Ahmed, H.; Semberg, P.; Andersson, C.; Björkman, B. Effect of added olivine on iron ore agglomerate during induration. ISIJ Int. 2018, 58, 446–452. [Google Scholar] [CrossRef]
- Li, S.; Chen, T.; Zhang, Y.; Zhao, J. Experimental study on application of Mg-bearing additives in iron ore pellet. Sinter. Pelletizing 2011, 36, 33–37. [Google Scholar]
- Semberg, P.; Andersson, C.; Björkman, B. Interaction between iron oxides and olivine in magnetite pellets during reduction to wustite at temperatures of 1000–1300 °C. ISIJ Int. 2013, 53, 391–398. [Google Scholar] [CrossRef]
- Singh, M.; Björkman, B. Effect of reduction conditions on the swelling behaviour of cement-bonded briquettes. ISIJ Int. 2004, 44, 294–299. [Google Scholar] [CrossRef]
- Gao, Q.; Wei, G.; Jiang, X.; Zheng, H.; Shen, F. Characteristics of light-burned magnesia and its application on oxidized pellet production. J. Iron Steel Res. Int. 2014, 21, 408–412. [Google Scholar] [CrossRef]
- Chun, T.; Zhu, D.; Pan, J. Influence of sulfur content in raw materials on oxidized pellets. J. Cent. South Univ. Technol. 2011, 18, 1924–1929. [Google Scholar] [CrossRef]
- Gao, Q.; Shen, F.; Wei, G.; Jiang, X.; Zheng, H. Effects of MgO containing additive on low-temperature metallurgical properties of oxidized pellet. J. Iron Steel Res. Int. 2013, 20, 25–30. [Google Scholar] [CrossRef]
- Sandeep Kumar, T.; Viswanathan, N.; Ahmed, H.P.; Andersson, C.; Björkman, B. Estimation of sintering kinetics of oxidized magnetite pellet using optical dilatometer. Metall. Mater. Trans. B 2015, 46, 635–643. [Google Scholar] [CrossRef]
- Sandeep Kumar, T.; Viswanathan, N.; Ahmed, H.P.; Andersson, C.; Björkman, B. Estimation of sintering kinetics of oxidized magnetite pellet using optical dilatometer. Metall. Mater. Trans. B 2016, 47, 309–319. [Google Scholar] [CrossRef]
- China Metallurgical Construction Association. Code for Design of Iron Pelletizing Engineering; China Planning Press: Beijing, China, 2009; pp. 20–25. ISBN 1580177244. [Google Scholar]
- Biswas, A.K. Principles of Blast Furnace Ironmaking; Cootha Publishing House: Brisbane, Australia, 1981; pp. 33–39. ISBN 0949917001. [Google Scholar]
- Szekely, J.; Evans, J.; Sohn, H. Gas-Solid Reductions; Academic Press: New York, NY, USA, 1976; pp. 97–102. ISBN 0126808503. [Google Scholar]
- Sohn, H.; Wadsworth, M. Rate Processes of Extractive Metallurgy; Plenum Press: New York, NY, USA, 1979; pp. 115–117. ISBN 030631102X. [Google Scholar]
- Eisenhüttenleute, V.D.; Allibert, M. Verlag Stahleisen. In Slag Atlas, 1st ed.; Verlag Stahleisen GmbH: Düsseldorf, Germany, 1995; p. 128. ISBN 3514004579. [Google Scholar]
- Bao, Y.; Jin, Z. Size effects and a mean-strength criterion for ceramic. Fatingue Fract. Eng. Mater. Struct. 1998, 8, 55–61. [Google Scholar] [CrossRef]
- Gibson, L.; Ashby, M. Cellular Solids: Structure and Propertie; Cambridge University Press: Cambridge, UK, 1997; pp. 150–158. ISBN 9780521499118. [Google Scholar]
- Suresh, S. Fatigue of Materials; Cambridge University Press: Cambridge, UK, 1998; pp. 70–75. ISBN 9780521578479. [Google Scholar]
- Iljana, M.; Mattila, O.; Alatarvas, T.; Kurikkala, J.; Paananen, T.; Fabritius, T. Effect of circulating elements on the dynamic reduction swelling behaviour of olivine and acid iron ore pellets under simulated blast furnace shaft conditions. ISIJ Int. 2013, 53, 419–426. [Google Scholar] [CrossRef]
- Shen, F.; Gao, Q.; Jiang, X.; Wei, G.; Zheng, H. Effect of magnesia on the compressive strength of pellets. Int. J. Miner. Metall. Mater. 2014, 21, 431–437. [Google Scholar] [CrossRef]
- Gao, Q.; Shen, Y.; Wei, G.; Jiang, X.; Shen, F. Diffusion behavior and distribution regulation of MgO in MgO-bearing pellets. Int. J. Miner. Metall. Mater. 2016, 23, 1011–1018. [Google Scholar] [CrossRef]
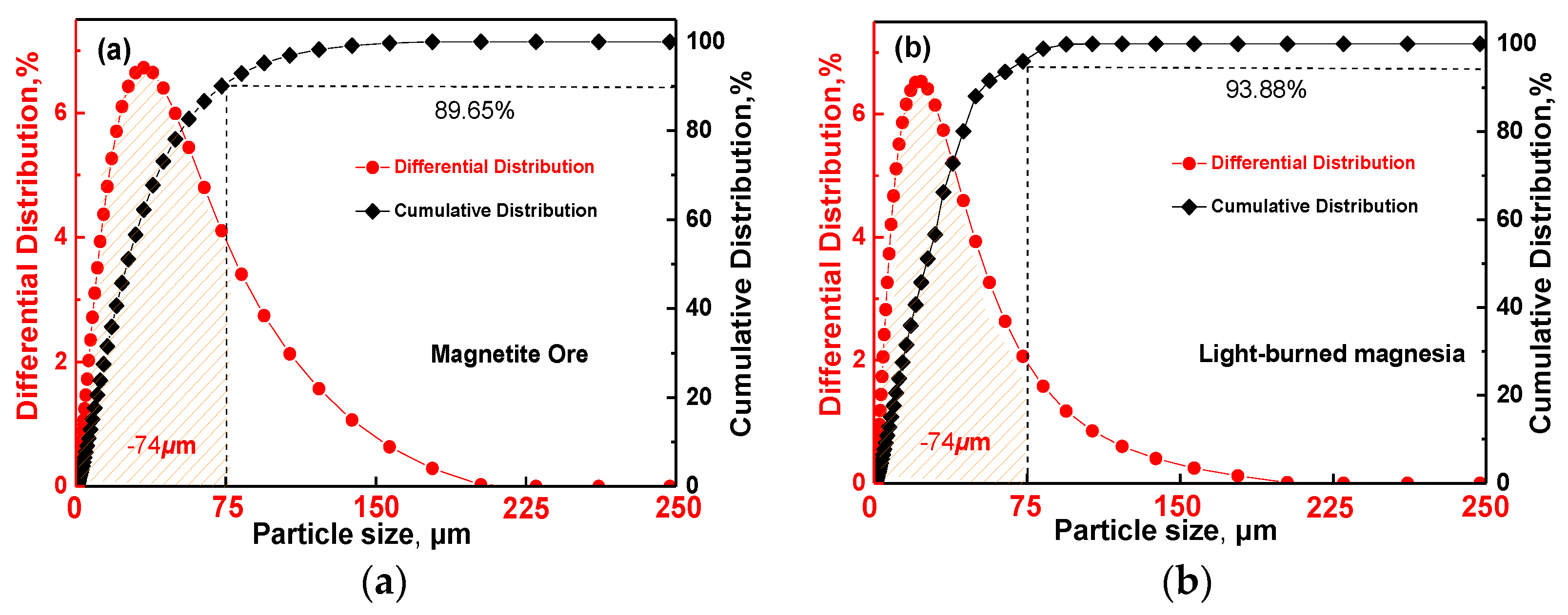
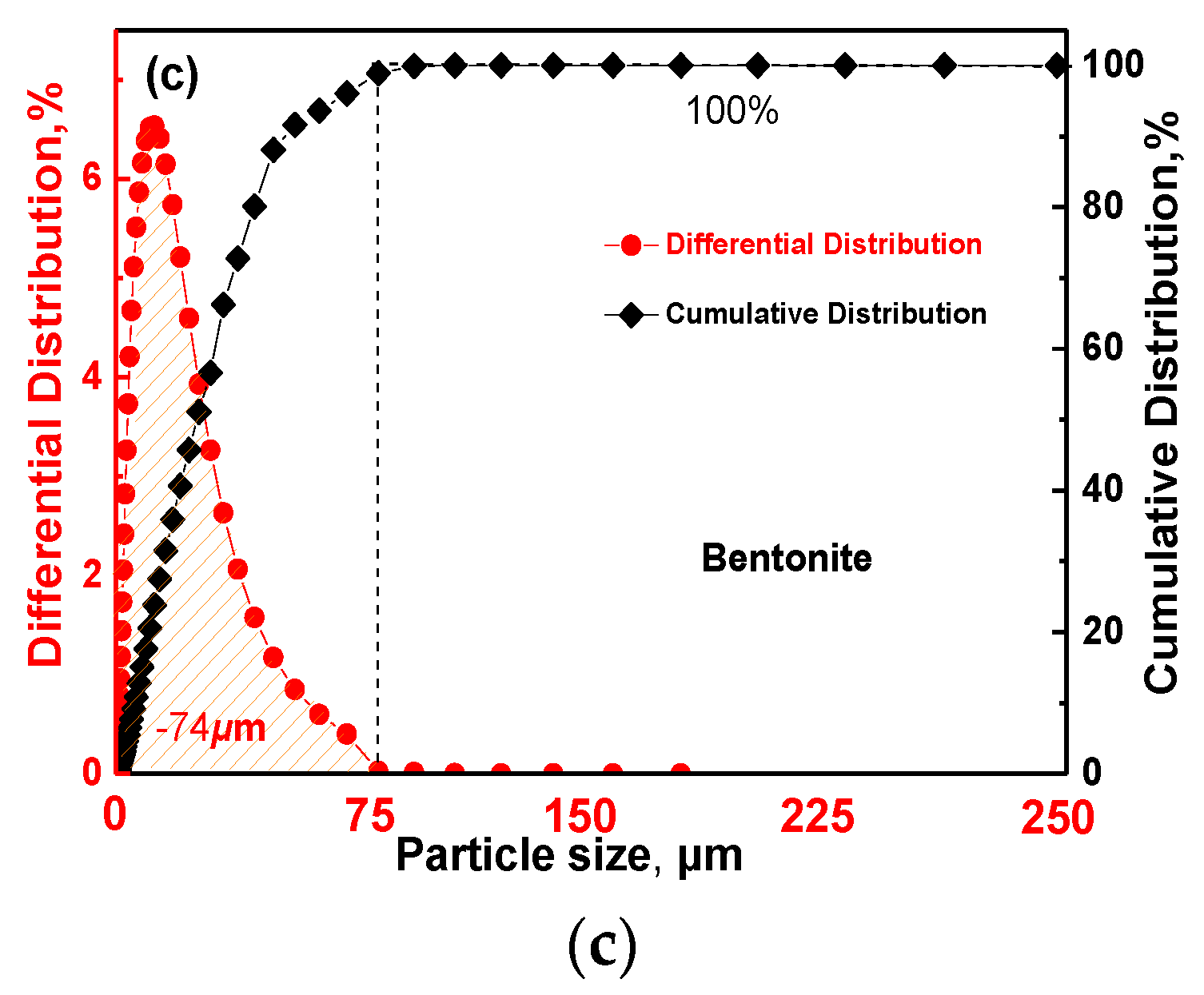
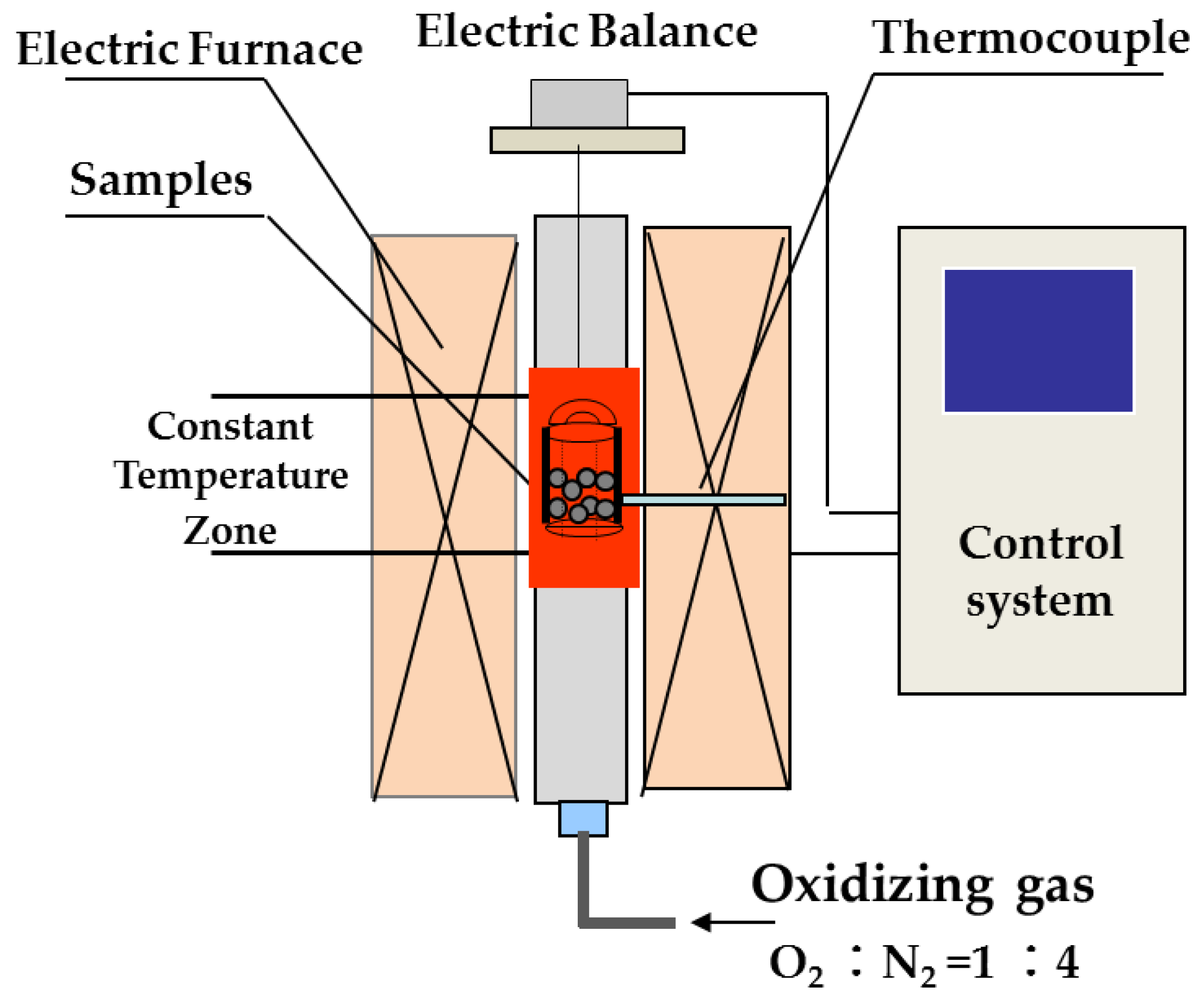



| Pelletizing Materials | TFe | MgO | CaO | SiO2 | Al2O3 | K2O + Na2O | LOI |
|---|---|---|---|---|---|---|---|
| Magnetite iron ore | 65.5 | 0.14 | 0.56 | 5.07 | 0.12 | - | - |
| Light-burned magnesia | - | 84.50 | 1.2 | 3.11 | 0.72 | - | 9.32 |
| Bentonite | 1.93 | 0.16 | 0.55 | 70.89 | 19.65 | 4.45 | - |
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Gao, Q.; Jiang, X.; Zheng, H.; Shen, F. Induration Process of MgO Flux Pellet. Minerals 2018, 8, 389. https://doi.org/10.3390/min8090389
Gao Q, Jiang X, Zheng H, Shen F. Induration Process of MgO Flux Pellet. Minerals. 2018; 8(9):389. https://doi.org/10.3390/min8090389
Chicago/Turabian StyleGao, Qiangjian, Xin Jiang, Haiyan Zheng, and Fengman Shen. 2018. "Induration Process of MgO Flux Pellet" Minerals 8, no. 9: 389. https://doi.org/10.3390/min8090389
APA StyleGao, Q., Jiang, X., Zheng, H., & Shen, F. (2018). Induration Process of MgO Flux Pellet. Minerals, 8(9), 389. https://doi.org/10.3390/min8090389





