Fiemmeite Cu2(C2O4)(OH)2∙2H2O, a New Mineral from Val di Fiemme, Trentino, Italy
Abstract
1. Introduction
2. Experimental Data
2.1. Mineral Description and Physical Properties
2.2. Chemical Data
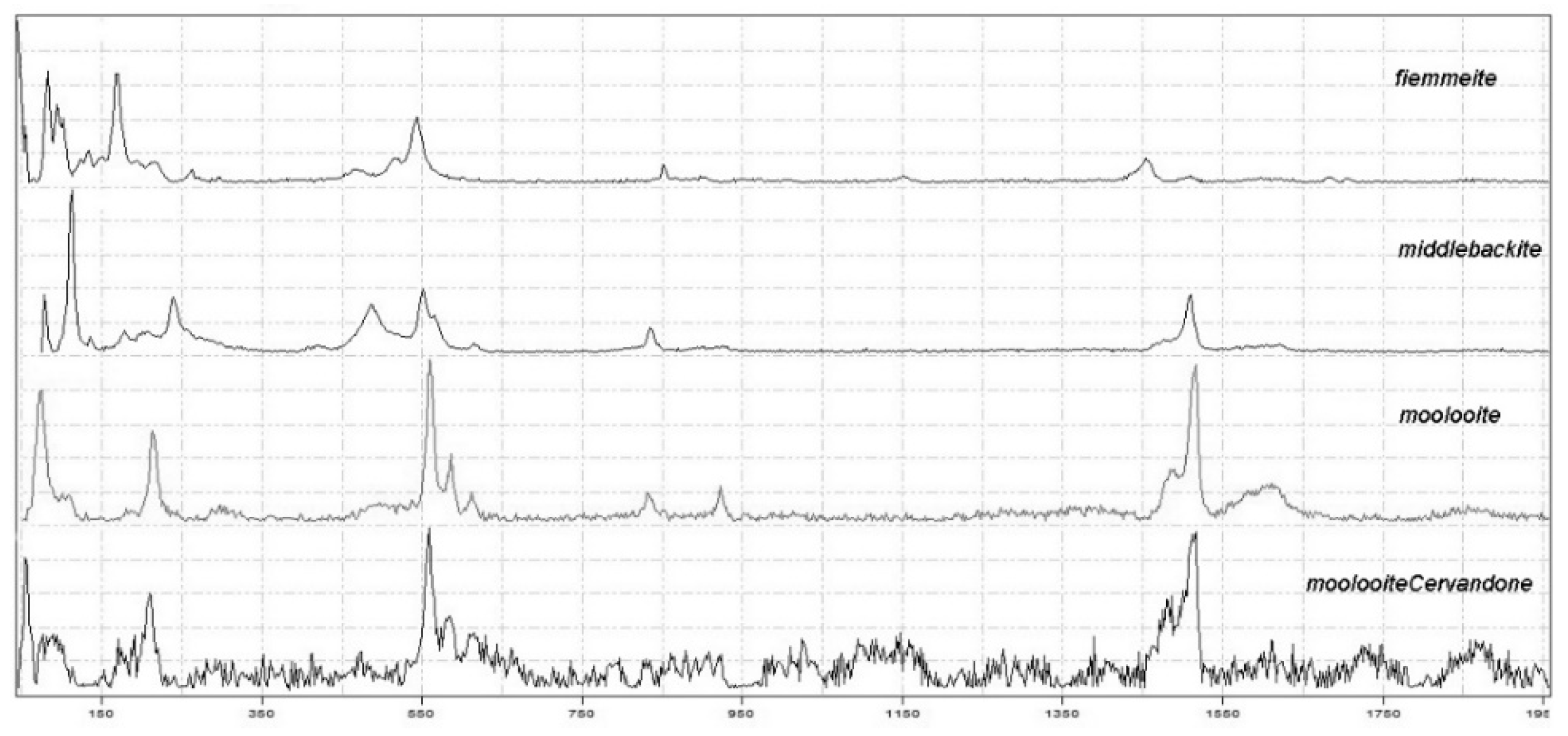
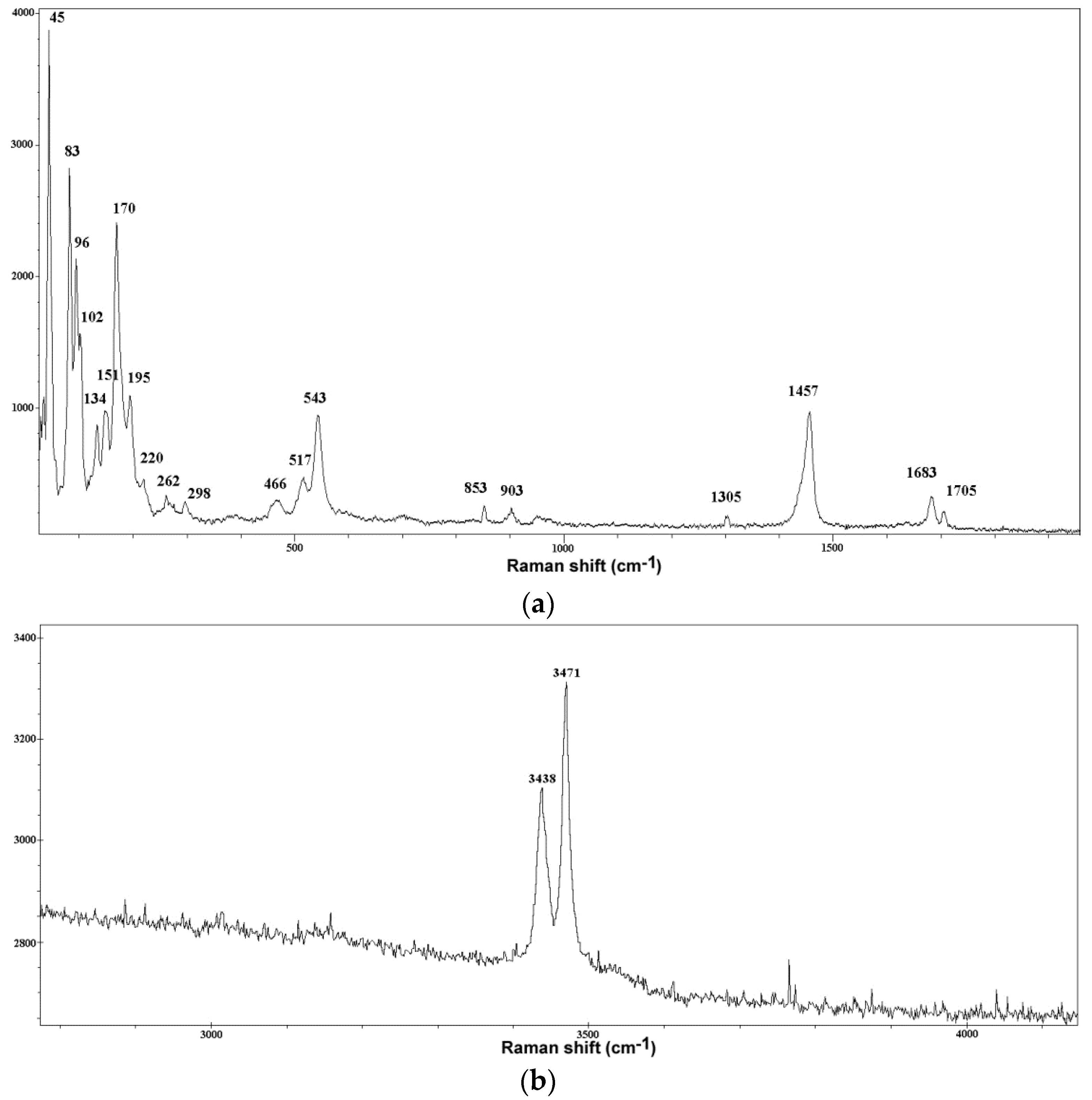
2.3. Micro Raman Spectroscopy
2.4. Crystallography
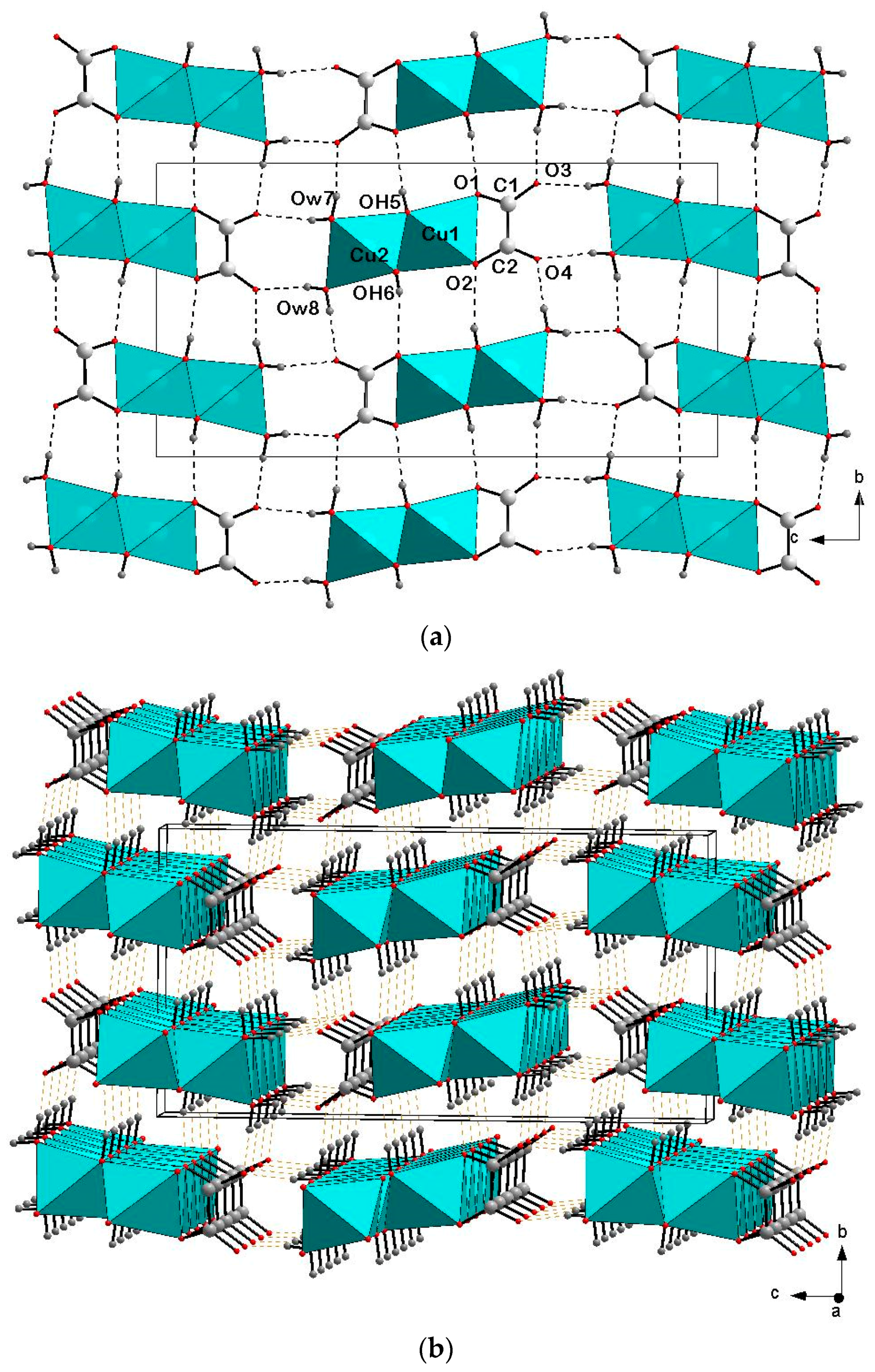
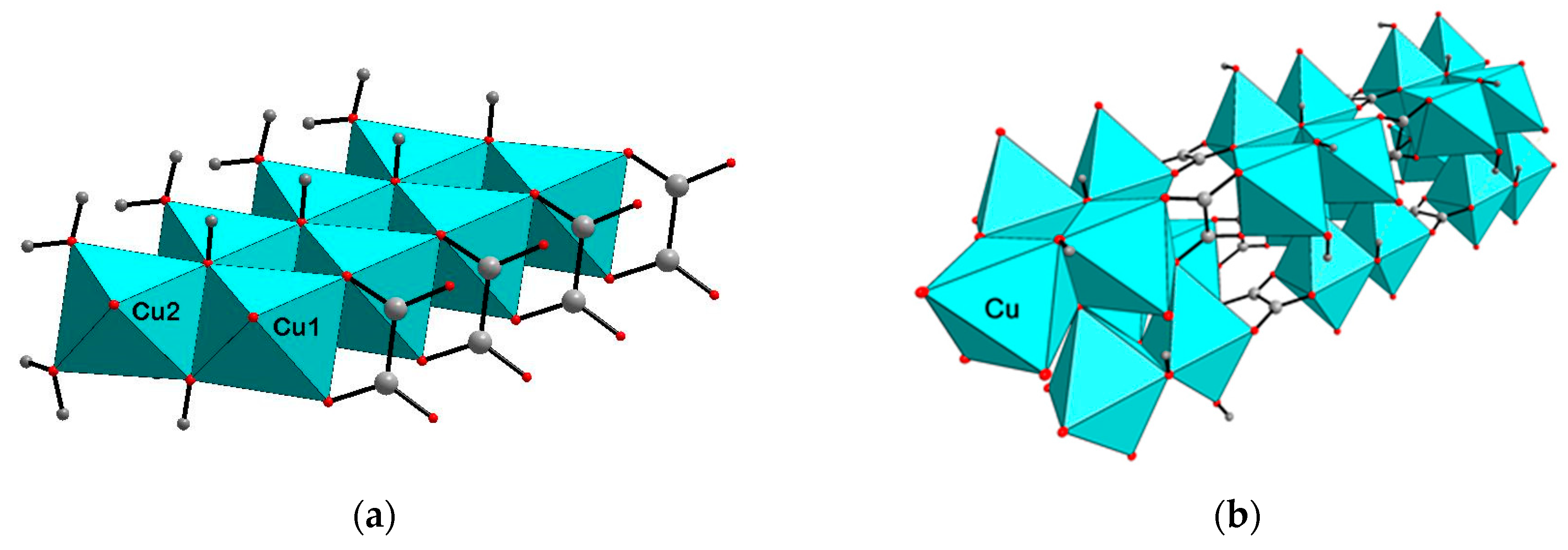
3. Crystal Structure Description and Discussion
Supplementary Materials
Author Contributions
Funding
Acknowledgments
Conflicts of Interest
Appendix A
References
- Massari, F.; Neri, C.; Pittau, P.; Fontana, D.; Stefani, C. Sedimentology, Palynostratigraphy and sequence stratigraphy of a continental to shallow-marine rift-related succession: Upper Permian of the eastern Southern Alps (Italy). Mem. Sci. Geol. Padova 1994, 46, 119–243. [Google Scholar]
- Wopfner, H.; Drake-Brockman, J. Base metal and uranium mineralization in the Groeden Sandstone of South Tyrol. Geo. Alp 2017, 14, 11–23. [Google Scholar]
- Demartin, F.; Campostrini, I.; Ferretti, P.; Rocchetti, I. Second global occurrence of middlebackite near the Passo di San Lugano (Carano, Trento, Italy). Geo. Alp 2017, 14, 35–38. [Google Scholar]
- Elliott, P. Middlebackite, IMA 2015-115. CNMNC Newsletter No. 30, April 2016, page 411. Mineral. Mag. 2016, 80, 407–413. [Google Scholar]
- Mandarino, J.A. The Gladstone-Dale relationship I. Derivation of new constants. Can. Mineral. 1976, 14, 498–502. [Google Scholar]
- Mandarino, J.A. The Gladstone-Dale relationship IV. The compatibility concept and its applications. Can. Mineral. 1981, 19, 441–450. [Google Scholar]
- Frost, R.L. Raman spectroscopy of natural oxalates. Anal. Chim. Acta 2004, 517, 207–214. [Google Scholar] [CrossRef]
- Libowitzky, E. Correlation of O-H stretching frequencies and O-H…O hydrogen bond lengths in minerals. Monatsh. Chem. 1999, 130, 1047–1059. [Google Scholar]
- Holland, T.J.B.; Redfern, S.A.T. Unit cell refinement from powder diffraction data: The use of regression diagnostics. Mineral. Mag. 1997, 61, 65–77. [Google Scholar] [CrossRef]
- Sheldrick, G.M. SADABS Area-Detector Absorption Correction Program; Bruker AXS Inc.: Madison, WI, USA, 2000. [Google Scholar]
- Altomare, A.; Burla, M.C.; Camalli, M.; Cascarano, G.L.; Giacovazzo, C.; Gagliardi, A.; Moliterni, A.G.; Polidori, G.; Spagna, R. SIR97, a new tool for crystal structure determination and refinement. J. Appl. Crystallogr. 1999, 32, 115–119. [Google Scholar] [CrossRef]
- Sheldrick, G.M. Crystal structure refinement with SHELXL. Acta Crystallogr. C Struct. Chem. 2015, 71, 3–8. [Google Scholar] [CrossRef] [PubMed]
- Fischer, R.X.; Tillmanns, E. The equivalent isotropic displacement factor. Acta Crystallogr. Sect. C Cryst. Struct. Commun. 1988, 44, 775–776. [Google Scholar] [CrossRef]
- Brown, I.D. Recent developments in the methods and applications of the Bond Valence Model. Chem. Rev. 2009, 109, 6858–6919. [Google Scholar] [CrossRef] [PubMed]
- Clarke, R.M.; Williams, I.R. Moolooite, a naturally occurring hydrated copper oxalate from western Australia. Mineral. Mag. 1986, 50, 295–298. [Google Scholar] [CrossRef]
- Rouse, R.C.; Peacor, D.R.; Dunn, P.J.; Simmons, W.B.; Newbury, D. Wheatleyite, Na2Cu(C2O4)2∙2H2O, a natural sodium copper salt of oxalic acid. Am. Mineral. 1986, 71, 1240–1242. [Google Scholar]
- Chukanov, N.V.; Aksenov, S.M.; Rastsvetaeva, R.K.; Lyssenko, K.A.; Belakovskiy, D.I.; Färber, G.; Möhn, G.; Van, K.V. Antipinite, KNa3Cu2(C2O4)4, a new mineral species from guano deposits at Pabellón de Pica, Chile. Mineral. Mag. 2014, 78, 797–804. [Google Scholar] [CrossRef]
- Smith, D.G.W.; Nickel, E.H. A system for codification for unnamed minerals: Report of the Subcommittee for Unnamed Minerals of the IMA Commission on New Minerals, Nomenclature and Classification. Can. Mineral. 2007, 45, 983–1055. [Google Scholar] [CrossRef]

| Constituent | wt % | Range | Stand. Dev. | Probe Standard | wt % ** |
|---|---|---|---|---|---|
| Cu | 44.00 | 43.79–44.24 | 0.19 | Synth. CuO | 44.57 |
| Zn | 0.09 | 0.06–0.12 | 0.02 | Synth. ZnO | 0 |
| O | 44.40 * | 44.89 | |||
| C | 8.34 * | 8.42 | |||
| H | 2.10 * | 2.12 | |||
| Total | 98.93 | 100.00 |
| dobs (Å) | Iobs | dcalc (Å) $ | Icalc $ | h, k, l | dobs (Å) | Iobs | dcalc (Å) $ | Icalc $ | h, k, l |
|---|---|---|---|---|---|---|---|---|---|
| 9.71 | 55 | 9.698 | 68 | 0 0 2 | 2.251 | 5 | 2.247 | 3 | 0 4 4 |
| 7.02 | 28 | 7.009 | 34 | 0 1 2 | 2.190 | 12 | 2.187 | 16 | 0 2 8 |
| 5.452 | 3 | 0 1 3 | 2.162 | 6 | −1 −3 4 | ||||
| 5.079 | 100 | 5.071 | 100 | 0 2 0 | 2.151 | 13 | 2.147 | 17 | 1 3 4 |
| 4.914 | 12 | 4.906 | 10 | 0 2 1 | 2.122 | 3 | 1 2 6 | ||
| 4.855 | 2 | 4.849 | 5 | 0 0 4 | 2.052 | 1 | −1 −3 5 | ||
| 4.501 | 50 | 4.493 | 40 | 0 2 2 | 2.036 | 2 | 1 3 5 | ||
| 3.996 | 4 | 3.990 | 6 | 0 2 3 | 2.028 | 6 | −1 −4 1 | ||
| 3.330 | 2 | 0 3 1 | 2.017 | 2 | 0 5 1 | ||||
| 3.241 | 4 | −1 0 2 | 1.997 | 1 | −1 −4 2 | ||||
| 3.237 | 5 | 3.233 | 7 | 0 0 6 | 1.998 | 3 | 1.995 | 7 | 0 4 6 |
| 3.193 | 1 | −1 −1 1 | 1.972 | 1 | 1 2 7 | ||||
| 3.198 | 5 | 3.192 | 7 | 0 3 2 | 1.953 | 1 | −1 1 8 | ||
| 3.072 | 58 | 3.087 | 65 | 1 1 2 | 1.943 | 8 | 1.938 | 13 | −1 3 6 |
| 3.001 | 2 | 2.996 | 6 | 0 3 3 | 1.935 | 4 | 0 5 3 | ||
| 2.891 | 20 | 2.886 | 18 | 1 1 3 | 1.905 | 3 | 0 1 10 | ||
| 2.813 | 2 | −1 0 4 | 1.883 | 2 | −1 −4 4 | ||||
| 2.811 | 5 | −1 −2 1 | 1.873 | 2 | 1 4 4 | ||||
| 2.731 | 2 | −1 2 2 | 1.873 | 5 | 1.870 | 6 | 0 4 7 | ||
| 2.730 | 15 | 2.726 | 9 | 0 2 6 | 1.824 | 6 | −1 3 7 | ||
| 2.711 | 13 | −1 −1 4 | 1.809 | 2 | −1 −4 5 | ||||
| 2.686 | 25 | 2.682 | 32 | 1 1 4 | 1.797 | 4 | 0 5 5 | ||
| 2.608 | 2 | −1 −2 3 | 1.755 | 7 | 1.752 | 10 | 0 4 8 | ||
| 2.589 | 1 | 1 2 3 | 1.745 | 1 | 1 5 0 | ||||
| 2.552 | 3 | 2.549 | 6 | 0 3 5 | 1.719 | 1 | −1 −5 2 | ||
| 2.514 | 1 | 0 4 1 | 1.716 | 3 | 1 5 2 | ||||
| 2.511 | 2 | 2.503 | 6 | −1 −1 5 | 1.697 | 2 | −1 0 10 | ||
| 2.468 | 2 | 2.474 | 7 | −1 −2 4 | 1.695 | 9 | 1.689 | 7 | −2 0 2 |
| 2.453 | 2 | 0 4 2 | 1.688 | 6 | 2 1 0 | ||||
| 2.442 | 4 | 2.438 | 5 | 1 2 4 | 1.687 | 3 | −1 −5 3 | ||
| 2.431 | 3 | 0 2 7 | 1.666 | 2 | −2−1 2 | ||||
| 2.424 | 1 | 0 0 8 | 1.657 | 3 | 1.656 | 6 | 1 1 10 | ||
| 2.390 | 1 | −1 −3 1 | 1.639 | 5 | −1 −5 4 | ||||
| 2.360 | 2 | 0 4 3 | 1.642 | 4 | 0 4 9 | ||||
| 2.340 | 2 | −1 −3 2 | 1.636 | 3 | 1 4 7 | ||||
| 2.336 | 3 | 0 3 6 | 1.609 | 3 | −1 2 10 | ||||
| 2.336 | 2 | 1 0 6 | 1.603 | 4 | −2 −2 2 | ||||
| 2.330 | 3 | 1 3 2 | 1.598 | 4 | 1 3 9 | ||||
| 2.310 | 6 | 2.303 | 9 | −1 1 6 | 1.596 | 2 | 0 6 4 | ||
| 2.261 | 2 | −1 −3 3 | 1.578 | 2 | −2 −2 3 | ||||
| 2.248 | 4 | 1 3 3 | 1.544 | 3 | −2 −2 4 |
| Crystal System | Monoclinic |
|---|---|
| Space Group | P21/c (No. 14) |
| a (Å) | 3.4245(6) |
| b (Å) | 10.141(2) |
| c (Å) | 19.397(3) |
| β (°) | 90.71(1) |
| V (Å3) | 673.6(2) |
| Z | 4 |
| Radiation | MoKα (λ = 0.71073 Å) |
| μ (mm−1) | 6.322 |
| Dcalc (g·cm−3) | 2.802 |
| Measured reflections | 7512 |
| Rint | 0.0294 |
| Independent reflections | 2118 |
| Range of h, k, l | −5 ≤ h ≤ 4, −14 ≤ k ≤ 14, −28 ≤ l ≤ 28 |
| Observed reflections [I > 2σ(I)] | 1942 |
| Parameters refined | 133 |
| Final R1 [I > 2σ(I)] and wR2 (all data) | 0.0386, 0.0905 |
| GooF | 1.176 |
| Max/min residuals (e/Å3) | 1.37/−0.73 |
| Atom | x/a | y/b | z/c | Ueq | ||
| Cu1 | 0.19372(13) | 0.24133(4) | 0.50020(2) | 0.0126(1) | ||
| Cu2 | 0.61198(13) | 0.29545(4) | 0.37303(2) | 0.0131(1) | ||
| C1 | 0.0211(11) | 0.1394(3) | 0.6268(2) | 0.0130(6) | ||
| C2 | −0.1380(10) | 0.2827(4) | 0.6259(2) | 0.0132(6) | ||
| O1 | 0.2069(8) | 0.1067(3) | 0.5731(1) | 0.0160(5) | ||
| O2 | −0.1081(8) | 0.3439(3) | 0.5690(1) | 0.0169(5) | ||
| O3 | −0.0369(9) | 0.0695(3) | 0.6773(1) | 0.0228(6) | ||
| O4 | −0.2941(9) | 0.3269(3) | 0.6784(1) | 0.0221(6) | ||
| OH5 | 0.6196(7) | 0.1665(2) | 0.4475(1) | 0.0130(5) | ||
| OH6 | 0.1952(8) | 0.3715(3) | 0.4273(1) | 0.0139(5) | ||
| Ow7 | 0.9519(9) | 0.1899(3) | 0.3121(1) | 0.0193(5) | ||
| Ow8 | 0.5910(10) | 0.4305(3) | 0.3023(1) | 0.0263(7) | ||
| H5 | 0.666(16) | 0.0765(16) | 0.441(3) | 0.032(15) | ||
| H6 | 0.182(14) | 0.4626(13) | 0.434(3) | 0.026(14) | ||
| H71 | 0.861(16) | 0.193(6) | 0.2664(11) | 0.037(16) | ||
| H72 | 0.932(19) | 0.0986(14) | 0.318(3) | 0.046(18) | ||
| H81 | 0.706(17) | 0.423(7) | 0.2589(15) | 0.047(19) | ||
| H82 | 0.504(15) | 0.517(2) | 0.309(3) | 0.028(14) | ||
| Atom | U11 | U22 | U33 | U23 | U13 | U12 |
| Cu1 | 0.0151(2) | 0.0118(2) | 0.01106(19) | 0.00217(14) | 0.00295(14) | 0.00301(15) |
| Cu2 | 0.0156(2) | 0.0120(2) | 0.01179(19) | 0.00236(14) | 0.00336(14) | 0.00312(15) |
| C1 | 0.0148(15) | 0.0107(14) | 0.0137(14) | −0.0003(11) | 0.0008(11) | 0.0013(12) |
| C2 | 0.0096(14) | 0.0151(15) | 0.0149(15) | −0.0014(12) | −0.0002(11) | 0.0009(12) |
| O1 | 0.0211(13) | 0.0124(12) | 0.0147(11) | 0.0011(9) | 0.0044(9) | 0.0056(10) |
| O2 | 0.0236(14) | 0.0124(12) | 0.0147(11) | 0.0019(9) | 0.0056(9) | 0.0053(10) |
| O3 | 0.0360(17) | 0.0152(13) | 0.0175(13) | 0.0055(10) | 0.0091(11) | 0.0069(12) |
| O4 | 0.0328(16) | 0.0182(13) | 0.0155(12) | −0.0012(10) | 0.0075(11) | 0.0090(12) |
| OH5 | 0.0149(12) | 0.0097(11) | 0.0145(11) | 0.0019(9) | 0.0029(9) | 0.0042(9) |
| OH6 | 0.0159(12) | 0.0116(11) | 0.0144(11) | 0.0026(9) | 0.0034(9) | 0.0033(9) |
| Ow7 | 0.0301(15) | 0.0139(12) | 0.0141(12) | −0.0007(9) | 0.0045(10) | 0.0041(11) |
| Ow8 | 0.0406(18) | 0.0192(14) | 0.0194(13) | 0.0079(11) | 0.0132(12) | 0.0133(13) |
| Atom1-Atom2 | Distance | vu | Atom1-Atom2 | Distance | vu |
| Cu1-OH6 | 1.935(3) | 0.50 | Cu2-Ow8 | 1.939(3) | 0.46 |
| Cu1-OH5 | 1.946(2) | 0.46 | Cu2-OH5 | 1.948(3) | 0.45 |
| Cu1-O1 | 1.966(3) | 0.43 | Cu2-OH6 | 1.943(3) | 0.46 |
| Cu1-O2 | 1.992(3) | 0.40 | Cu2-Ow7 | 1.983(3) | 0.41 |
| Cu1-OH5#1 | 2.332(3) | 0.16 | Cu2-OH6#2 | 2.375(3) | 0.14 |
| Cu1-O2#2 | 2.916(3) | 0.03 | Cu2-Ow7#1 | 2.754(3) | 0.05 |
| Total | 1.98 | Total | 1.97 | ||
| Cu1-Cu2 | 2.9198(7) | C1-C2 | 1.552(5) | ||
| C1-O3 | 1.228(4) | C2-O4 | 1.240(4) | ||
| C1-O1 | 1.271(4) | C2-O2 | 1.270(4) | ||
| Atom1-Atom2-Atom3 | angle | Atom1-Atom2-Atom3 | angle | ||
| O1-C1-O3 | 126.2(3) | O1-C1-C2 | 114.5(3) | ||
| C1-C2-O2 | 115.8(3) | O2-C2-O4 | 125.2(3) | ||
| C1-C2-O4 | 118.9(3) | C2-C1-O3 | 119.3(3) | ||
| Hydrogen bond interactions | |||||
| Atom1…Atom2 | distance | Atom1-Atom2…Atom3 | angle | ||
| OH5…O1#3 | 2.862(4) | OH5-H5…O1#3 | 177(5) | ||
| OH6…O2#4 | 2.903(4) | OH6-H6…O2#4 | 169(5) | ||
| Ow7…O3#3 | 2.655(4) | Ow7-H72…O3#3 | 163(6) | ||
| Ow7…O4#5 | 2.722(4) | Ow7-H71…O4#5 | 171(6) | ||
| Ow8…O4#4 | 2.691(4) | Ow8-H82…O4#4 | 175(5) | ||
| Ow8…O3#5 | 2.753(4) | Ow8-H81…O3#5 | 172(6) | ||
© 2018 by the authors. Licensee MDPI, Basel, Switzerland. This article is an open access article distributed under the terms and conditions of the Creative Commons Attribution (CC BY) license (http://creativecommons.org/licenses/by/4.0/).
Share and Cite
Demartin, F.; Campostrini, I.; Ferretti, P.; Rocchetti, I. Fiemmeite Cu2(C2O4)(OH)2∙2H2O, a New Mineral from Val di Fiemme, Trentino, Italy. Minerals 2018, 8, 248. https://doi.org/10.3390/min8060248
Demartin F, Campostrini I, Ferretti P, Rocchetti I. Fiemmeite Cu2(C2O4)(OH)2∙2H2O, a New Mineral from Val di Fiemme, Trentino, Italy. Minerals. 2018; 8(6):248. https://doi.org/10.3390/min8060248
Chicago/Turabian StyleDemartin, Francesco, Italo Campostrini, Paolo Ferretti, and Ivano Rocchetti. 2018. "Fiemmeite Cu2(C2O4)(OH)2∙2H2O, a New Mineral from Val di Fiemme, Trentino, Italy" Minerals 8, no. 6: 248. https://doi.org/10.3390/min8060248
APA StyleDemartin, F., Campostrini, I., Ferretti, P., & Rocchetti, I. (2018). Fiemmeite Cu2(C2O4)(OH)2∙2H2O, a New Mineral from Val di Fiemme, Trentino, Italy. Minerals, 8(6), 248. https://doi.org/10.3390/min8060248





